In Pursuit of Vitamin D in Plants
Abstract
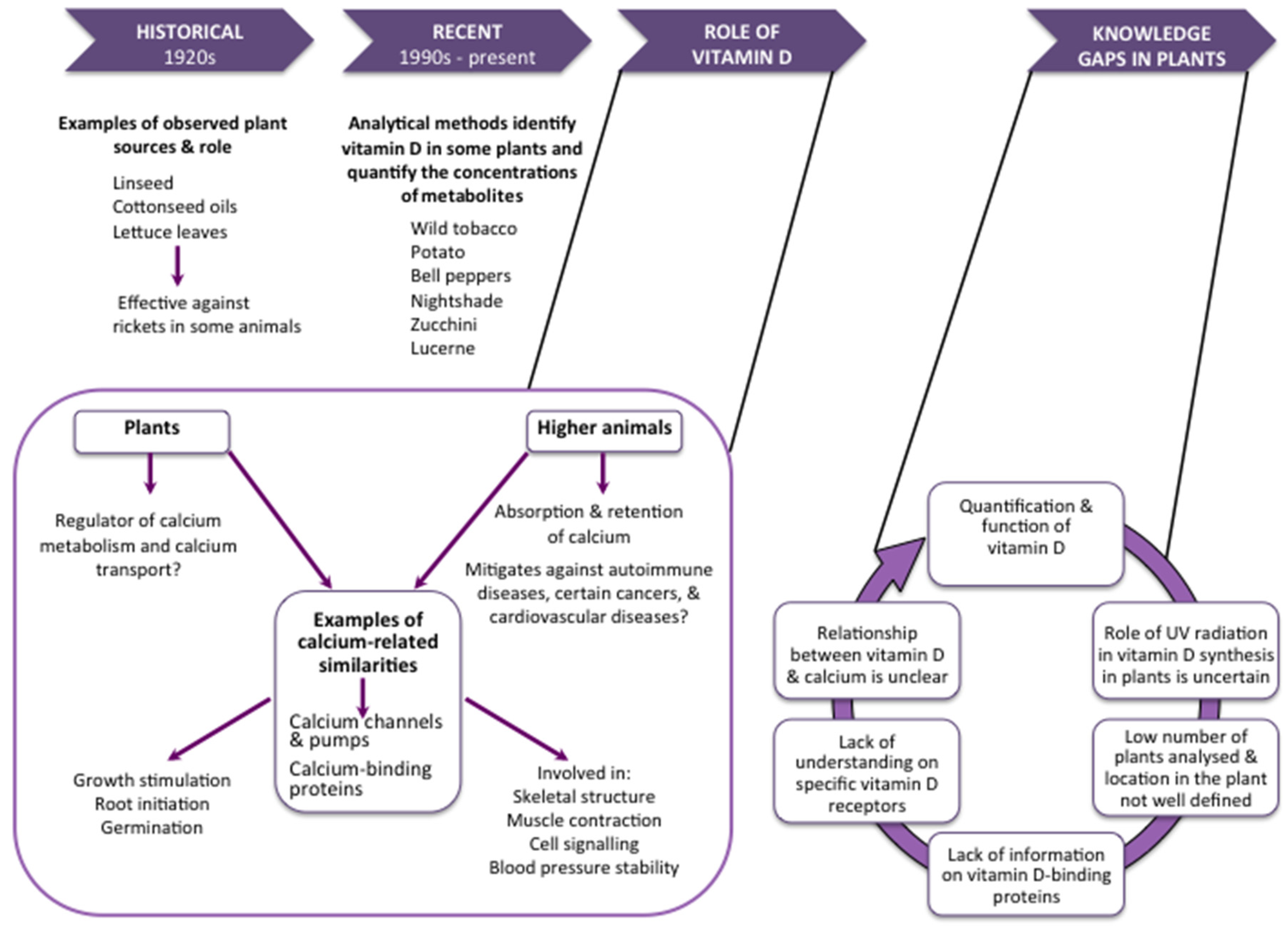
:1. Introduction
2. Metabolism of Vitamin D in Plants
3. The Function of Vitamin D in Plants
4. Vitamin D2 Content of Plants
5. Vitamin D3 Content of Plants
6. Research Gaps
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| UV-B | Ultraviolet-B radiation |
| 25(OH)D | 25-hydroxyvitamin D |
| 1,25(OH)2D | 1,25-dihydroxyvitamin D |
| VDR | Vitamin D receptor |
| LC-MS/MS | Liquid-chromatography tandem mass spectrometry |
References
- Basu, S.; Gupta, R.; Mitra, M.; Ghosh, A. Prevalence of vitamin D deficiency in a pediatric hospital of Eastern India. Indian J. Clin. Biochem. 2015, 30, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Strobel, N.; Buddhadasa, S.; Adorno, P.; Stockham, K.; Greenfield, H. Vitamin D and 25-hydroxyvitamin D determination in meats by LC-IT-MS. Food Chem. 2013, 138, 1042–1047. [Google Scholar] [CrossRef] [PubMed]
- Jäpelt, R.B.; Didion, T.; Smedsgaard, J.; Jakobsen, J. Seasonal variation of provitamin D2 and vitamin D2 in perennial ryegrass (Lolium perenne L.). J. Agric. Food Chem. 2011, 59, 10907–10912. [Google Scholar] [CrossRef] [PubMed]
- Jäpelt, R.B.; Silvestro, D.; Smedsgaard, J.; Jensen, P.E.; Jakobsen, J. LC-MS/MS with atmospheric pressure chemical ionization to study the effect of UV treatment on the formation of viatmin D3 and sterols in plants. Food Chem. 2011, 129, 217–225. [Google Scholar] [CrossRef]
- Jäpelt, R.B.; Jakobsen, J. Vitamin D in plants: A review of occurrence, analysis, and biosynthesis. Front. Plant Sci. 2013, 4, 136. [Google Scholar] [CrossRef] [PubMed]
- Darby, H.H.; Clarke, H.T. The plant origin of a vitamin D. Science 1937, 318, 318–319. [Google Scholar] [CrossRef] [PubMed]
- Sunita Rao, D.; Raghuramulu, N. Food chain as origin of vitamin D in fish. Comp. Biochem. Physiol. 1996, 114A, 15–19. [Google Scholar] [CrossRef]
- Boland, R.L. Plants as a source of vitamin D3 metabolites. Nutr. Rev. 1986, 44, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Phillips, K.M.; Horst, R.L.; Koszewski, N.J.; Simon, R.R. Vitamin D4 in mushrooms. PLoS ONE 2012, 7, e40702. [Google Scholar] [CrossRef] [PubMed]
- Krings, U.; Berger, R.G. Dynamics of sterols and fatty acids during UV-B treatment of oyster mushroom. Food Chem. 2014, 149, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Björn, L.; Widell, S.; Wang, T. Evolution of UV-B regulation and protection in plants. Adv. Space Res. 2002, 30, 1557–1562. [Google Scholar] [CrossRef]
- Frega, N.; Bocci, F.; Conte, L.S.; Testa, F. Chemical composition of tobacco seeds (Nicotiana tabacum L.). J. Am. Oil Chem. Soc. 1991, 68, 29–33. [Google Scholar] [CrossRef]
- Haussler, M.R.; Haussler, C.A.; Bartik, L.; Whitfield, G.K.; Hsieh, J.C.; Slater, S.; Jurutka, P.W. Vitamin D receptor: Molecular signaling and actions of nutritional ligands in disease prevention. Nutr. Rev. 2008, 66, S98–S112. [Google Scholar] [CrossRef] [PubMed]
- Boyan, B.D.; Chen, J.; Schwartz, Z. Mechanism of pdia3-dependent 1 α,25-dihydroxyvitamin D3 signaling in musculoskeletal cells. Steroids 2012, 77, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Doroudi, M.; Plaisance, M.C.; Boyan, B.D.; Schwartz, Z. Membrane actions of 1α,25(OH)2D3 are mediated by Ca2+/calmodulin-dependent protein kinase II in bone and cartilage cells. J. Steroid Biochem. Mol. Biol. 2015, 145, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D. Vitamin D: An ancient hormone. Exp. Dermatol. 2010, 20, 7–13. [Google Scholar] [CrossRef]
- Milanesi, L.; Boland, R. Presence of vitamin D3 receptor (VDR)-like proteins in Solanum glaucophyllum. Physiol. Plant 2006, 128, 341–350. [Google Scholar] [CrossRef]
- Esparza, M.S.; Vega, M.; Boland, R.L. Synthesis and composition of vitamin D3 metabolites in Solanum malacoxylon. Biochim. Biophys. Acta 1982, 719, 633–640. [Google Scholar] [CrossRef]
- Hughes, M.R.; McCain, T.A.; Chang, S.Y.; Haussler, M.R.; Villareale, M.; Wasserman, R.H. Presence of 1,25 dihydroxyvitamin D3-glycoside in the calcinogenic plant Cestrum diurnum. Nature 1977, 268, 347–349. [Google Scholar] [CrossRef] [PubMed]
- Rambeck, W.A.; Weiser, H.; Zucker, H. Biological activity of glycosides of vitamin D3 and 1 alpha-hydroxyvitamin D3. Int. J. Vitam Nutr. Res. 1984, 54, 25–34. [Google Scholar] [PubMed]
- Cranney, A.; Horsley, T.; O’Donnell, S.; Weiler, H.A.; Puil, L.; Ooi, D.S.; Atkinson, S.A.; Ward, L.M.; Moher, D.; Hanley, D.A.; et al. Effectiveness and Safety of Vitamin D in Relation to Bone Health; Evidence Report/Technology Assessment No. 158 (Prepared by the University of Ottawa Evidence-Based Practice Center (uo-epc) under Contract No. 290-02-0021. Ahrq publication No. 07-e013; University of Ottawa Evidence-Based Practice Center: Rockville, MD, USA, 2007. [Google Scholar]
- Dodd, A.N.; Kudla, J.; Sanders, D. The language of calcium signaling. Annu. Rev. Plant Biol. 2010, 61, 593–620. [Google Scholar] [CrossRef] [PubMed]
- De Souza Leite, T.; Cnossen-Fassoni, A.; Pereira, O.L.; Mizubuti, E.S.; de Araujo, E.F.; de Queiroz, M.V. Novel and highly diverse fungal endophytes in soybean revealed by the consortium of two different techniques. J. Microbiol. 2013, 51, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Gonzaga, L.L.; Costa, L.E.; Santos, T.T.; Araujo, E.F.; Queiroz, M.V. Endophytic fungi from the genus Colletotrichum are abundant in the Phaseolus vulgaris and have high genetic diversity. J. Appl. Microbiol. 2015, 118, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Schulz, B.; Boyle, C. The endophytic continuum. Mycol. Res. 2005, 109, 661–686. [Google Scholar] [CrossRef] [PubMed]
- Hess, A.F.; Weinstock, M. Antirachitic properties imparted to inert fluids and to green vegetables by ultra-violet irradiation. J. Biol. Chem. 1924, 62, 301–313. [Google Scholar]
- Bechtel, H.E.; Huffman, C.F.; Ducan, C.W.; Hoppert, C.A. Vitamin D studies in cattle. J. Dairy Sci. 1936, 19, 359–372. [Google Scholar] [CrossRef]
- Wasserman, R.H.; Corradino, R.A.; Krook, L.; Hughes, M.R.; Haussler, M.R. Studies on the 1α, 25-dihydroxycholecalciferol-like activity in a calcinogenic plant, Cestrum diurnum, in the chick. J. Nutr. 1976, 106, 457–465. [Google Scholar] [PubMed]
- Jakobsen, J.; Saxholt, E. Vitamin D metabolites in bovine milk and butter. J. Food Comp. Anal. 2009, 22, 472–478. [Google Scholar] [CrossRef]
- Rodriguez, R.; Redman, R. More than 400 million years of evolution and some plants still can’t make it on their own: Plant stress tolerance via fungal symbiosis. J. Exp. Biol. 2008, 59, 1109–1114. [Google Scholar] [CrossRef] [PubMed]
- Scudamore, K.A.; Livesey, C.T. Occurrence and significance of mycotoxins in forage crops and silage: A review. J. Sci. Food Agric. 1998, 77, 1–17. [Google Scholar] [CrossRef]
- Thacher, T.D.; Fischer, P.R.; Obadofin, M.O.; Levine, M.A.; Singh, R.J.; Pettifor, J.M. Comparison of metabolism of vitamins D2 and D3 in children with nutritional rickets. J. Bone Miner. Res. 2010, 25, 1988–1995. [Google Scholar] [CrossRef] [PubMed]
- Fisk, C.M.; Theobald, H.E.; Sanders, T.A. Fortified malted milk drinks containing low-dose ergocalciferol and cholecalciferol do not differ in their capacity to raise serum 25-hydroxyvitamin D concentrations in healthy men and women not exposed to UV-B. J. Nutr. 2012, 142, 1286–1290. [Google Scholar] [CrossRef] [PubMed]
- Stephensen, C.B.; Zerofsky, M.; Burnett, D.J.; Lin, Y.P.; Hammock, B.D.; Hall, L.M.; McHugh, T. Ergocalciferol from mushrooms or supplements consumed with a standard meal increases 25-hydroxyergocalciferol but decreases 25-hydroxycholecalciferol in the serum of healthy adults. J. Nutr. 2012, 142, 1246–1252. [Google Scholar] [CrossRef] [PubMed]
- Armas, L.A.; Hollis, B.W.; Heaney, R.P. Vitamin D2 is much less effective than vitamin D3 in humans. J. Clin. Endocrinol. Metab. 2004, 89, 5387–5391. [Google Scholar] [CrossRef] [PubMed]
- Prema, T.P.; Raghuramulu, N. Vitamin D3 and its metabolites in the tomato plant. Phytochemistry 1996, 42, 617–620. [Google Scholar] [CrossRef]
- Curino, A.; Skliar, M.; Boland, R. Identification of 7-dehydrocholesterol, vitamin D3, 25(OH)-vitamin D3 and 1,25(OH)2-vitamin D3 in Solanum glaucophyllum cultures grown in absence of light. Biochim. Biophys. Acta 1998, 1425, 485–492. [Google Scholar] [CrossRef]
- Weissenberg, M.; Levy, A.; Wasserman, R.H. Distribution of calcitriol activity in Solanum glaucophyllum plants and cell cultures. Phytochemistry 1989, 28, 795–798. [Google Scholar] [CrossRef]
- Curino, A.; Milanesi, L.; Benassati, S.; Skliar, M.; Boland, R. Effect of culture conditions on the synthesis of vitamin D3 metabolites in Solanum glaucophyllum grown in vitro. Phytochemistry 2001, 58, 81–89. [Google Scholar] [CrossRef]
- Simon, R.R.; Phillips, K.M.; Horst, R.L.; Munro, I.C. Vitamin D mushrooms: Comparison of the composition of button mushrooms (Agaricus bisporus) treated postharvest with UVB light or sunlight. J. Agric. Food Chem. 2011, 59, 8724–8732. [Google Scholar] [CrossRef] [PubMed]
- Jäpelt, R.B.; Silvestro, D.; Smedsgaard, J.; Jensen, P.E.; Jakobsen, J. Quantification of vitamin D3 and its hydroxylated metabolites in waxy leaf nightshade (Solanum glaucophyllum Desf.), tomato (Solanum lycopersicum L.) and bell pepper (Capsicum annuum L.). Food Chem. 2013, 138, 1206–1211. [Google Scholar] [CrossRef] [PubMed]

| Species | 7-dehydrocholesterol (μg/g) | Vitamin D3 (μg/g) | 25(OH)D3 (μg/g) | 1,25(OH)2D3 (μg/g) |
|---|---|---|---|---|
| Non-irradiated | ||||
| Lycopersicon esculentum (tomato plant) | 0.47 [4] 1 | Not identified [4] 1 0.0017 [41] 2 | <0.00002 [41] 2 | <0.0001 [41] 2 |
| Solanum glaucophyllum (waxy leaf nightshade) | 0.67 [4] 1 | Not identified [4] 1 0.0032 [41] 2 | 0.0008 [41] 2 | <0.0001 [41] 2 |
| Capsicum annuum (bell pepper) | 0.03 [4] 1 | Not identified [4] 1 <0.00002 [41] 2 | <0.00002 [41] 2 | <0.0001 [41] 2 |
| UV-irradiated | ||||
| Lycopersicon esculentum (tomato plant) | 0.23 [4] 1 | 0.09 [4] 1 0.1 [41] 2 | 0.0043 [41] 2 | <0.0001 [41] 2 |
| Solanum glaucophyllum (waxy leaf nightshade) | 1.26 [4] 1 | 0.21 [4] 1 0.2 [41] 2 | 0.031 [41] 2 | 0.032 [41] 2 |
| Capsicum annuum (bell pepper) | 0.03 [4] 1 | Not identified [4] 1 0.0029 [41] 2 | 0.0005 [41] 2 | <0.0001 [41] 2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Black, L.J.; Lucas, R.M.; Sherriff, J.L.; Björn, L.O.; Bornman, J.F. In Pursuit of Vitamin D in Plants. Nutrients 2017, 9, 136. https://doi.org/10.3390/nu9020136
Black LJ, Lucas RM, Sherriff JL, Björn LO, Bornman JF. In Pursuit of Vitamin D in Plants. Nutrients. 2017; 9(2):136. https://doi.org/10.3390/nu9020136
Chicago/Turabian StyleBlack, Lucinda J., Robyn M. Lucas, Jill L. Sherriff, Lars Olof Björn, and Janet F. Bornman. 2017. "In Pursuit of Vitamin D in Plants" Nutrients 9, no. 2: 136. https://doi.org/10.3390/nu9020136
APA StyleBlack, L. J., Lucas, R. M., Sherriff, J. L., Björn, L. O., & Bornman, J. F. (2017). In Pursuit of Vitamin D in Plants. Nutrients, 9(2), 136. https://doi.org/10.3390/nu9020136






