Coffee Decreases the Risk of Endometrial Cancer: A Dose–Response Meta-Analysis of Prospective Cohort Studies
Abstract
1. Introduction
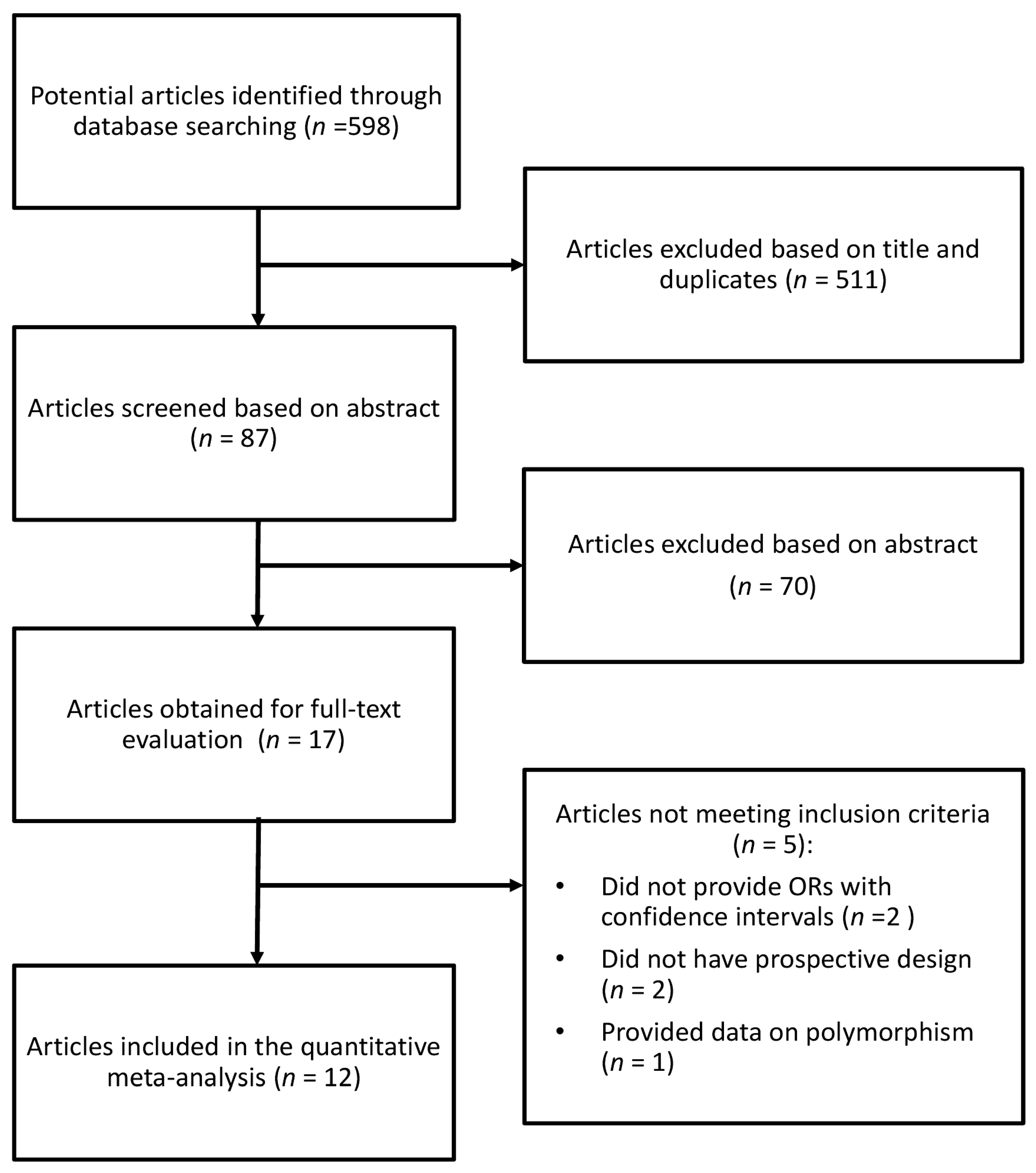
2. Methods
2.1. Search Strategy
2.2. Data Extraction
2.3. Statistical Analysis
3. Results
3.1. Study Characteristics
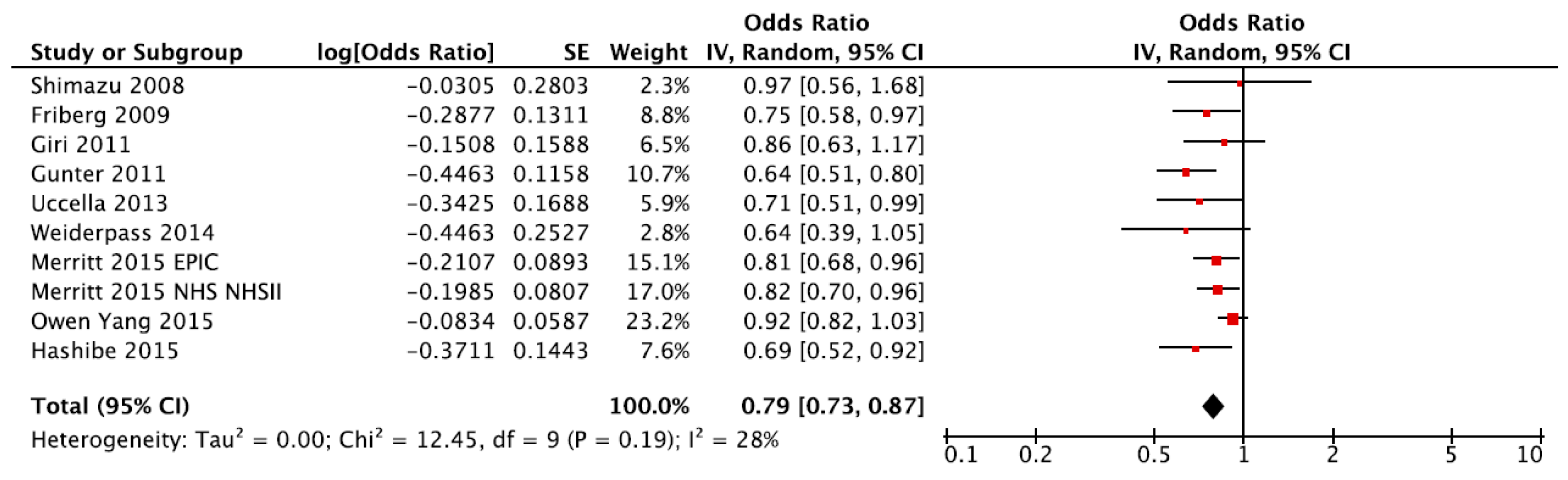
3.2. Summary Relative Risk for Highest vs. Lowest Category of Coffee Consumption
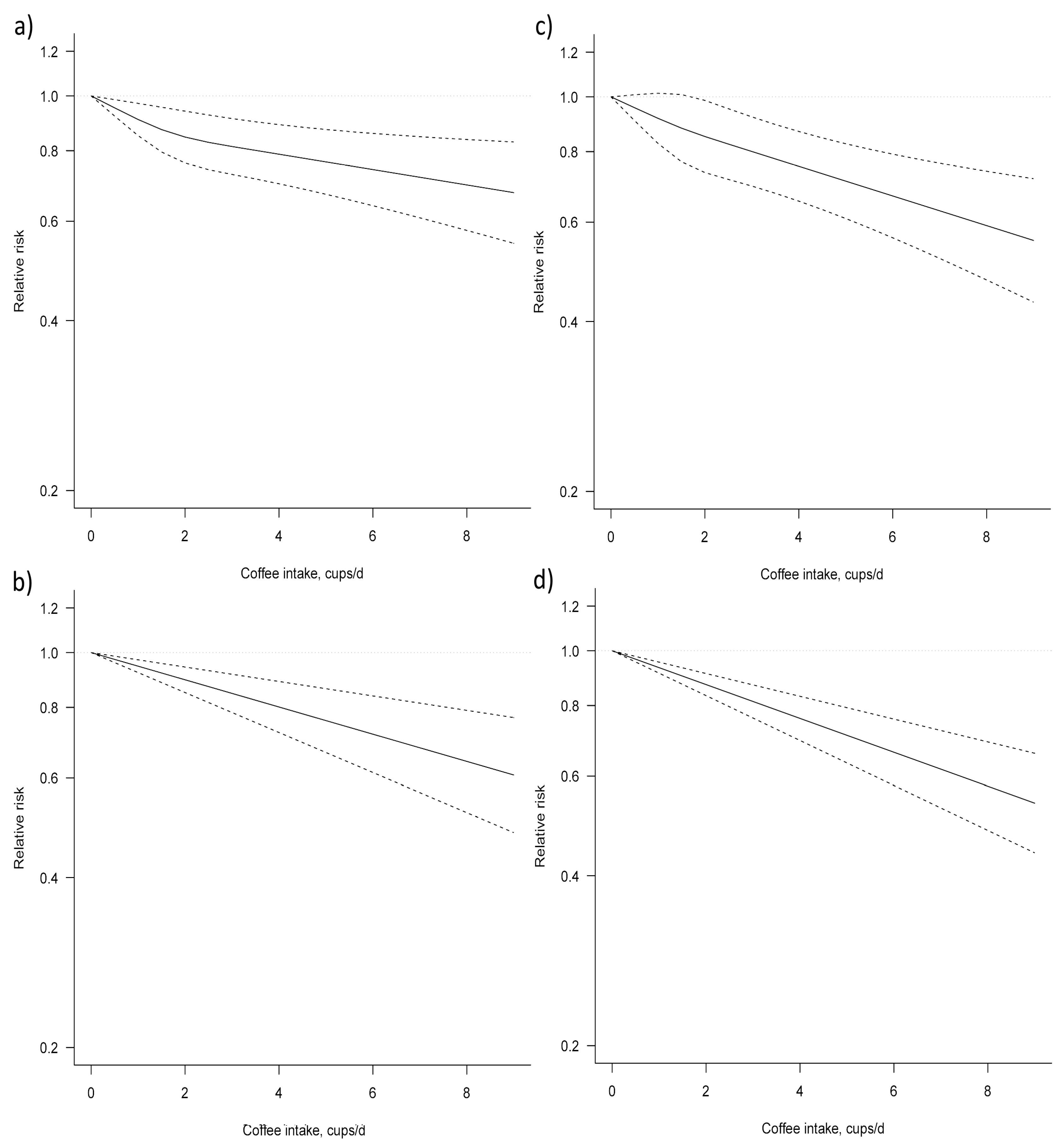
3.3. Dose–Response Meta-Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Word Health Oranganization. Available online: http://globocan.iarc.fr/Default.aspx (accessed on 4 November 2017).
- Forouzanfar, M.H.; Alexander, L.; Anderson, H.R.; Bachman, V.F.; Biryukov, S.; Brauer, M.; Burneet, R.; Casey, D.; Coates, M.M.; Cohen, A.; et al. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2013: A systematic analysis for the global burden of disease study 2013. Lancet 2016, 388, 2287–2323. [Google Scholar] [CrossRef]
- Grosso, G.; Bella, F.; Godos, J.; Sciacca, S.; Del Rio, D.; Ray, S.; Galvano, F.; Giovannucci, E. Possible role of diet in cancer: Systematic review and multiple meta-analyses of dietary patterns, lifestyle factors, and cancer risk. Nut. Rev. 2017, 75, 405–419. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; AlMazroa, M.A.; Amann, M.; Anderson, H.R.; Andrews, K.G.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the global burden of disease study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef]
- Grosso, G.; Godos, J.; Galvano, F.; Giovannucci, E.L. Coffee, caffeine, and health outcomes: An umbrella review. Annu. Rev. Nutr. 2017, 37, 131–156. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Micek, A.; Godos, J.; Sciacca, S.; Pajak, A.; Martinez-Gonzalez, M.A.; Giovannucci, E.L.; Galvano, F. Coffee consumption and risk of all-cause, cardiovascular, and cancer mortality in smokers and non-smokers: A dose-response meta-analysis. Eur. J. Epidemiol. 2016, 31, 1191–1205. [Google Scholar] [CrossRef] [PubMed]
- Marventano, S.; Salomone, F.; Godos, J.; Pluchinotta, F.; Del Rio, D.; Mistretta, A.; Grosso, G. Coffee and tea consumption in relation with non-alcoholic fatty liver and metabolic syndrome: A systematic review and meta-analysis of observational studies. Clin. Nutr. 2016, 35, 1269–1281. [Google Scholar] [CrossRef] [PubMed]
- Godos, J.; Micek, A.; Marranzano, M.; Salomone, F.; Rio, D.D.; Ray, S. Coffee consumption and risk of biliary tract cancers and liver cancer: A dose-response meta-analysis of prospective cohort studies. Nutrition 2017, 9, 950. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Hu, G.H.; Wang, X.C.; Huang, T.B.; Xu, L.; Lai, P.; Guo, Z.F.; Xu, Y.F. Coffee consumption and prostate cancer risk: A meta-analysis of cohort studies. Nutr. Cancer 2015, 67, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Ran, H.Q.; Wang, J.Z.; Sun, C.Q. Coffee consumption and pancreatic cancer risk: An update meta-analysis of cohort studies. Pak. J. Med. Sci. 2016, 32, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Gan, Y.; Wu, J.; Zhang, S.; Li, L.; Cao, S.; Mkandawire, N.; Ji, K.; Herath, C.; Gao, C.; Xu, H.; et al. Association of coffee consumption with risk of colorectal cancer: A meta-analysis of prospective cohort studies. Oncotarget 2016, 8, 18699. [Google Scholar] [CrossRef] [PubMed]
- Caprioli, G.; Cortese, M.; Sagratini, G.; Vittori, S. The influence of different types of preparation (espresso and brew) on coffee aroma and main bioactive constituents. Int. J. Food Sci. Nutr. 2015, 66, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Godos, J.; Pluchinotta, F.R.; Marventano, S.; Buscemi, S.; Li Volti, G.; Galvano, F.; Grosso, G. Coffee components and cardiovascular risk: Beneficial and detrimental effects. Int. J. Food Sci. Nutr. 2014, 65, 925–936. [Google Scholar] [CrossRef] [PubMed]
- Je, Y.; Giovannucci, E. Coffee consumption and risk of endometrial cancer: Findings from a large up-to-date meta-analysis. Int. J. Cancer 2012, 131, 1700–1710. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Luo, M.L.; Li, H.; Li, M.; Zhou, J.G. Coffee consumption and risk of endometrial cancer: A dose-response meta-analysis of prospective cohort studies. Sci. Rep. 2015, 5, 13410. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.O.; Crowe, F.; Cairns, B.J.; Reeves, G.K.; Beral, V. Tea and coffee and risk of endometrial cancer: Cohort study and meta-analysis. Am. J. Clin. Nutr. 2015, 101, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Federation, E.C. Coffee Consumption in Europe. Available online: https://www.ecf-coffee.org/index.php (accessed on 4 November 2017).
- Grigg, D. The worlds of tea and coffee: Patterns of consumption. GeoJournal 2002, 57, 283–294. [Google Scholar] [CrossRef]
- Wang, A.; Wang, S.; Zhu, C.; Huang, H.; Wu, L.; Wan, X.; Yang, X.; Zhang, H.; Miao, R.; He, L.; et al. Coffee and cancer risk: A meta-analysis of prospective observational studies. Sci. Rep. 2016, 6, 33711. [Google Scholar] [CrossRef] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis of observational studies in epidemiology (moose) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (nos) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses; Ottawa Health Research Institute: Ottawa, ON, Canada, 1999. [Google Scholar]
- Greenland, S. Quantitative methods in the review of epidemiologic literature. Epidemiol. Rev. 1987, 9, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Greenland, S.; Longnecker, M.P. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am. J. Epidemiol. 1992, 135, 1301–1309. [Google Scholar] [CrossRef] [PubMed]
- Orsini, N.B.R.; Greenland, S. Generalized least squares for trend estimation of summarized dose-response data. Stata J. 2006, 6, 40–57. [Google Scholar]
- Orsini, N.; Li, R.; Wolk, A.; Khudyakov, P.; Spiegelman, D. Meta-analysis for linear and nonlinear dose-response relations: Examples, an evaluation of approximations, and software. Am. J. Epidemiol. 2012, 175, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Friberg, E.; Orsini, N.; Mantzoros, C.S.; Wolk, A. Coffee drinking and risk of endometrial cancer—A population-based cohort study. Int. J. Cancer 2009, 125, 2413–2417. [Google Scholar] [CrossRef] [PubMed]
- Gavrilyuk, O.; Braaten, T.; Skeie, G.; Weiderpass, E.; Dumeaux, V.; Lund, E. High coffee consumption and different brewing methods in relation to postmenopausal endometrial cancer risk in the Norwegian women and cancer study: A population-based prospective study. BMC Women's Health 2014, 14, 48. [Google Scholar] [CrossRef] [PubMed]
- Giri, A.; Sturgeon, S.R.; Luisi, N.; Bertone-Johnson, E.; Balasubramanian, R.; Reeves, K.W. Caffeinated coffee, decaffeinated coffee and endometrial cancer risk: A prospective cohort study among us postmenopausal women. Nutrition 2011, 3, 937–950. [Google Scholar] [CrossRef] [PubMed]
- Gunter, M.J.; Schaub, J.A.; Xue, X.; Freedman, N.D.; Gaudet, M.M.; Rohan, T.E.; Hollenbeck, A.R.; Sinha, R. A prospective investigation of coffee drinking and endometrial cancer incidence. Int. J. Cancer 2012, 131, E530–E536. [Google Scholar] [CrossRef] [PubMed]
- Hashibe, M.; Galeone, C.; Buys, S.S.; Gren, L.; Boffetta, P.; Zhang, Z.F.; La Vecchia, C. Coffee, tea, caffeine intake, and the risk of cancer in the plco cohort. Br. J. Cancer 2015, 113, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Je, Y.; Hankinson, S.E.; Tworoger, S.S.; De Vivo, I.; Giovannucci, E. A prospective cohort study of coffee consumption and risk of endometrial cancer over a 26-year follow-up. Cancer Epidemiol. Biomark. Prev. 2011, 20, 2487–2495. [Google Scholar] [CrossRef] [PubMed]
- Merritt, M.A.; Tzoulaki, I.; Tworoger, S.S.; De Vivo, I.; Hankinson, S.E.; Fernandes, J.; Tsilidis, K.K.; Weiderpass, E.; Tjonneland, A.; Petersen, K.E.; et al. Investigation of dietary factors and endometrial cancer risk using a nutrient-wide association study approach in the epic and nurses’ health study (NHS) and NHSII. Cancer Epidemiol. Biomark. Prev. 2015, 24, 466–471. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, L.M.; Johansson, I.; Lenner, P.; Lindahl, B.; Van Guelpen, B. Consumption of filtered and boiled coffee and the risk of incident cancer: A prospective cohort study. Cancer Causes Control 2010, 21, 1533–1544. [Google Scholar] [CrossRef] [PubMed]
- Shimazu, T.; Inoue, M.; Sasazuki, S.; Iwasaki, M.; Kurahashi, N.; Yamaji, T.; Tsugane, S.; JPHC Study Group Members of the Japan Public Health Center-based Prospective Study. Coffee consumption and risk of endometrial cancer: A prospective study in Japan. Int. J. Cancer 2008, 123, 2406–2410. [Google Scholar] [CrossRef] [PubMed]
- Uccella, S.; Mariani, A.; Wang, A.H.; Vierkant, R.A.; Cliby, W.A.; Robien, K.; Anderson, K.E.; Cerhan, J.R. Intake of coffee, caffeine and other methylxanthines and risk of Type I vs. Type II endometrial cancer. Br. J. Cancer 2013, 109, 1908–1913. [Google Scholar] [CrossRef] [PubMed]
- Weiderpass, E.; Sandin, S.; Lof, M.; Oh, J.K.; Inoue, M.; Shimazu, T.; Tsugane, S.; Adami, H.O. Endometrial cancer in relation to coffee, tea, and caffeine consumption: A prospective cohort study among middle-aged women in Sweden. Nutr. Cancer 2014, 66, 1132–1143. [Google Scholar] [CrossRef] [PubMed]
- Bohn, S.K.; Blomhoff, R.; Paur, I. Coffee and cancer risk, epidemiological evidence, and molecular mechanisms. Mol. Nutr. Res. 2014, 58, 915. [Google Scholar] [CrossRef] [PubMed]
- Boettler, U.; Sommerfeld, K.; Volz, N.; Pahlke, G.; Teller, N.; Somoza, V.; Lang, R.; Hofmann, T.; Marko, D. Coffee constituents as modulators of Nrf2 nuclear translocation and ARE (EpRE)-dependent gene expression. J. Nutr. Biochem. 2011, 22, 426–440. [Google Scholar] [CrossRef] [PubMed]
- Gaascht, F.; Dicato, M.; Diederich, M. Coffee provides a natural multitarget pharmacopeia against the hallmarks of cancer. Genes Nutr. 2015, 10, 51. [Google Scholar] [CrossRef] [PubMed]
- Niseteo, T.; Komes, D.; Belščak-Cvitanović, A.; Horžić, D.; Budeč, M. Bioactive composition and antioxidant potential of different commonly consumed coffee brews affected by their preparation technique and milk addition. Food Chem. 2012, 134, 1870–1877. [Google Scholar] [CrossRef] [PubMed]
- Liang, N.; Kitts, D.D. Antioxidant property of coffee components: Assessment of methods that define mechanisms of action. Molecules 2014, 19, 19180. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Godos, J.; Lamuela-Raventos, R.; Ray, S.; Micek, A.; Pajak, A.; Sciacca, S.; D’Orazio, N.; Del Rio, D.; Galvano, F. A comprehensive meta-analysis on dietary flavonoid and lignan intake and cancer risk: Level of evidence and limitations. Mol. Nutr. Food Res. 2017, 61, 1600930. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Micek, A.; Godos, J.; Pajak, A.; Sciacca, S.; Galvano, F.; Giovannucci, E.L. Dietary flavonoid and lignan intake and mortality in prospective cohort studies: Systematic review and dose-response meta-analysis. Am. J. Epidemiol. 2017, 185, 1304–1316. [Google Scholar] [CrossRef] [PubMed]
- Kotsopoulos, J.; Eliassen, A.H.; Missmer, S.A.; Hankinson, S.E.; Tworoger, S.S. Relationship between caffeine intake and plasma sex hormone concentrations in premenopausal and postmenopausal women. Cancer 2009, 115, 2765–2774. [Google Scholar] [CrossRef] [PubMed]
- Ferrini, R.L.; Barrett-Connor, E. Caffeine intake and endogenous sex steroid levels in postmenopausal women. The rancho bernardo study. Am. J. Epidemiol. 1996, 144, 642–644. [Google Scholar] [CrossRef] [PubMed]
- Nagata, C.; Kabuto, M.; Shimizu, H. Association of coffee, green tea, and caffeine intakes with serum concentrations of estradiol and sex hormone-binding globulin in premenopausal Japanese women. Nutr. Cancer 1998, 30, 21. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; Schulze, M.B.; Hu, F.B.; Hankinson, S.E.; Holmes, M.D. A dietary pattern derived to correlate with estrogens and risk of postmenopausal breast cancer. Breast Cancer Res. Treat. 2012, 132, 1157. [Google Scholar] [CrossRef] [PubMed]
- Sisti, J.S.; Hankinson, S.E.; Caporaso, N.E.; Gu, F.; Tamimi, R.M.; Rosner, B.; Xu, X.; Ziegler, R.; Eliassen, A.H. Caffeine, coffee, and tea intake and urinary estrogens and estrogen metabolites in premenopausal women. Cancer Epidemiol. Biomark. Prev. 2015, 24, 1174. [Google Scholar] [CrossRef] [PubMed]
- Akash, M.S.; Rehman, K.; Chen, S. Effects of coffee on type 2 diabetes mellitus. Nutrition 2014, 30, 755. [Google Scholar] [CrossRef] [PubMed]
- Ortega, Á.; Berná, G.; Rojas, A.; Martín, F.; Soria, B. Gene-diet interactions in type 2 diabetes: The chicken and egg debate. Int. J. Mol. Sci. 2017, 18, 1188. [Google Scholar] [CrossRef] [PubMed]
- Lees, B.; Leath, C.A. The impact of diabetes on gynecologic cancer: Current status and future directions. Curr. Obstet. Gynecol. Rep. 2015, 4, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Alicandro, G.; Tavani, A.; La Vecchia, C. Coffee and cancer risk: A summary overview. Eur. J. Cancer Prev. 2017, 26, 424–432. [Google Scholar] [CrossRef] [PubMed]
- Stocks, T.; Bjorge, T.; Ulmer, H.; Manjer, J.; Haggstrom, C.; Nagel, G.; Engeland, A.; Johansen, D.; Hallmans, G.; Selmer, R.; et al. Metabolic risk score and cancer risk: Pooled analysis of seven cohorts. Int. J. Epidemiol. 2015, 44, 1353–1363. [Google Scholar] [CrossRef] [PubMed]
- Renehan, A.G.; Tyson, M.; Egger, M.; Heller, R.F.; Zwahlen, M. Body-mass index and incidence of cancer: A systematic review and meta-analysis of prospective observational studies. Lancet 2008, 371, 569–578. [Google Scholar] [CrossRef]
- De Pergola, G.; Silvestris, F. Obesity as a major risk factor for cancer. J. Obes. 2013, 2013, 291546. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, P.R.; Ferrari, G.S.; Ferrari, C.K. Diet, sleep and metabolic syndrome among a legal amazon population, Brazil. Clin. Nutr. Res. 2015, 4, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Driessen, M.T.; Koppes, L.L.; Veldhuis, L.; Samoocha, D.; Twisk, J.W. Coffee consumption is not related to the metabolic syndrome at the age of 36 years: The amsterdam growth and health longitudinal study. Eur. J. Clin. Nutr. 2009, 63, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.S.; Chang, Y.F.; Liu, P.Y.; Chen, C.Y.; Tsai, Y.S.; Wu, C.H. Smoking, habitual tea drinking and metabolic syndrome in elderly men living in rural community: The tianliao old people (TOP) study 02. PLoS ONE 2012, 7, e38874. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Marventano, S.; Galvano, F.; Pajak, A.; Mistretta, A. Factors associated with metabolic syndrome in a mediterranean population: Role of caffeinated beverages. J. Epidemiol. 2014, 24, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Stepaniak, U.; Micek, A.; Topor-Madry, R.; Pikhart, H.; Szafraniec, K.; Pajak, A. Association of daily coffee and tea consumption and metabolic syndrome: Results from the polish arm of the hapiee study. Eur. J. Nutr. 2015, 54, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Lutsey, P.L.; Steffen, L.M.; Stevens, J. Dietary intake and the development of the metabolic syndrome: The atherosclerosis risk in communities studies. Circulation 2008, 117, 754–761. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, H.; Mure, K.; Nishio, N.; Kitano, N.; Nagai, N.; Takeshita, T. Relationship between coffee consumption and prevalence of metabolic syndrome among Japanese civil servants. J. Epidemiol. 2012, 22, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Nordestgaard, A.T.; Thomsen, M.; Nordestgaard, B.G. Coffee intake and risk of obesity, metabolic syndrome and type 2 diabetes: A Mendelian randomization study. Int. J. Epidemiol. 2015, 44, 551–565. [Google Scholar] [CrossRef] [PubMed]
- Takami, H.; Nakamoto, M.; Uemura, H.; Katsuura, S.; Yamaguchi, M.; Hiyoshi, M.; Sawachika, F.; Juta, T.; Arisawa, K. Inverse correlation between coffee consumption and prevalence of metabolic syndrome: Baseline survey of the japan multi-institutional collaborative cohort (J-micc) study in Tokushima, Japan. J. Epidemiol. 2013, 23, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Suliga, E.; Koziel, D.; Ciesla, E.; Rebak, D.; Gluszek, S. Coffee consumption and the occurrence and intensity of metabolic syndrome: A cross-sectional study. Int. J. Sci. Nutr. 2016, 68, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Micek, A.; Grosso, G.; Polak, M.; Kozakiewicz, K.; Tykarski, A.; Puch Walczak, A.; Drygas, W.; Kwasniewska, M.; Pajak, A. Association between tea and coffee consumption and prevalence of metabolic syndrome in Poland—Results from the WOBASZ II study (2013–2014). Int. J. Food Sci. Nutr. 2017, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Aoyagi, R.; Funakoshi-Tago, M.; Fujiwara, Y.; Tamura, H. Coffee inhibits adipocyte differentiation via inactivation of pparγ. Biol. Pharm. Bull. 2014, 37, 1820–1825. [Google Scholar] [CrossRef] [PubMed]
- Kearney, J.M.; Kearney, M.J.; McElhone, S.; Gibney, M.J. Methods used to conduct the pan-European Union survey on consumer attitudes to physical activity, body weight and health. Public Health Nutr. 1999, 2, 79–86. [Google Scholar] [CrossRef] [PubMed]
| Author, Year | Cohort Name, Country | Years of Study, Follow-Up | Cases; Total Population | Age Range | Adjustments |
|---|---|---|---|---|---|
| Shimazu, 2008 | JPHC, Japan | 1990–2005, 15 years (maximum) | 117; 53,724 | 40–59 years | Age, study area, BMI (body mass index), menopausal status, age at menopause for postmenopausal women, parity, use of exogenous female hormones, smoking status, green vegetable consumption, beef consumption, pork consumption, and green tea consumption. |
| Friberg, 2009 | SMC, Sweden | 1992–2007, 17.6 years (mean) | 677; 60,634 | 40–76 years | Age, BMI, smoking. |
| Nilsson, 2010 | VIP, Sweden | 1985–2007, 6 years (median) | 108; 32,178 | 30–60 years | Age, sex, BMI, smoking, education, recreational physical activity. |
| Giri, 2011 | WHI, USA | 1993–2005, 7.5 years (average) | 427; 45,696 | 50–79 years | Age, ethnicity, unopposed estrogen use, progestin + estrogen use, smoking, BMI. |
| Je, 2011 | NHS, USA | 1980–2006, 26 years (maximum) | 672; 67,470 | 34–59 years | Age, BMI, age at menopause, age at menarche, parity and age at last birth, duration of oral contraceptive use, postmenopausal hormone use, pack-years of smoking, alcohol intake, and total energy intake, tea analysis. |
| Gunter, 2011 | NIH-AARP, USA | 1995–2006, 9.3 years (mean) | 1486; 111,429 | 50–71 years | Age, smoking, BMI, age at menarche, age at first child’s birth, parity, age at menopause, HT (hormonal therapy) use, oral contraceptive use, diabetes and physical activity. |
| Ucella, 2013 | IWHS, USA | 1986–2005, 20 years (maximum) | 542; 23,356 | 55–69 years | Age, diabetes, duration of HT use, hypertension, age at menarche, age at menopause, BMI, waist-to-hip ratio, smoking status, pack years of smoking, total energy and alcohol use. |
| Gavrilyuk, 2011 | NOWAC, Norway | 1991–2007, 10.9 (average) | 462; 97,926 | 30–70 years | Parity, smoking status, BMI, duration of OC (oral contracception) and HRT use. |
| Weiderpass, 2014 | WLH, Sweden | 1991–2009, 18 years (maximum) | 144; 42,270 | 30-49 years | Age, education, duration of hormonal contraceptive use, parity, duration of breastfeeding, smoking status and number of cigarettes/day, menopausal status, BMI, and diabetes mellitus. |
| Merritt, 2015 | EPIC, Multicentre; NHS/NHSII USA | EPIC 1992-NA, 11 years (mean); NHS 1976–2010, 25 years (mean); NHSII 1989–2011, 25 years (mean) | EPIC 1303; 301,107; NHS/NHSII 1531; 155,406 | EPIC 25–70 years; NHS 30–55 years; NHSII 25–42 years | BMI, total energy intake, smoking status, age at menarche, oral contraceptive use, a combined variable for menopausal status and postmenopausal hormone (PMH) use, parity, and was stratified by the age of recruitment, and the study centre. |
| Owen Yang, 2015 | MWS, UK | 1996–2001, 9.3 years (average) | 4067; 560,356 | ~60 years (mean) | Age, region, socioeconomic status, height, age at menarche, parity, duration of oral contraceptive use, age and status of menopause at study baseline, duration of hormone therapy for menopause, BMI, smoking, alcohol consumption, strenuous exercise, tea consumption, and other nonalcohol fluid intake. |
| Hashibe, 2015 | PLCO, USA | 1992–2011, 13 years (maximum) | 254; 50,563 | 55–74 years | Age, sex, race, education, smoking status, smoking frequency, smoking duration, time since stopping smoking for past smokers, and drinking frequency. |
| Subgroup/Additional Analysis | No. of Datasets | RR (95% CI) | I2 | Pheterogeneity |
|---|---|---|---|---|
| Total | 10 | 0.79 (0.73, 0.87) | 28% | 0.19 |
| Geographical area | ||||
| North America | 5 | 0.75 (0.67, 0.84) | 6% | 0.37 |
| Europe | 4 | 0.84 (0.74, 0.94) | 29% | 0.24 |
| Asia | 1 | 0.97 (0.56, 1.68) | NA | NA |
| Menopausal status | ||||
| Postmenopausal | 7 | 0.70 (0.63, 0.78) | 0% | 0.60 |
| Premenopausal | 2 | 0.76 (0.49, 1.19) | 16% | 0.27 |
| Coffee type | ||||
| Caffeinated | 4 | 0.65 (0.50, 0.85) | 64% | 0.04 |
| Decaffeinated | 4 | 0.76 (0.62, 0.93) | 0% | 0.72 |
| BMI | ||||
| <25 kg/m2 | 7 | 0.99 (0.86, 1.14) | 0% | 0.58 |
| >25 kg/m2 | 7 | 0.79 (0.61, 1.01) | 66% | 0.004 |
| >30 kg/m2 | 5 | 0.75 (0.63, 0.88) | 22% | 0.27 |
| Smoking status | ||||
| Never smoker | 8 | 0.78 (0.68, 0.88) | 7% | 0.38 |
| Ever smoker (former/current) | 8 | 0.74 (0.57, 0.98) | 68% | 0.003 |
| Adjusted for smoking | ||||
| No | 0 | NA | NA | NA |
| Yes | 10 | 0.79 (0.73, 0.87) | 28% | 0.19 |
| Adjusted for BMI | ||||
| No | 1 | 0.69 (0.52, 0.91) | NA | NA |
| Yes | 9 | 0.80 (0.74, 0.88) | 27% | 0.20 |
| Adjusted for education | ||||
| No | 8 | 0.81 (0.74, 0.89) | 30% | 0.19 |
| Yes | 2 | 0.68 (0.53, 0.87) | 0% | 0.80 |
| Adjusted for alcohol intake | ||||
| No | 8 | 0.77 (0.71, 0.84) | 0% | 0.56 |
| Yes | 2 | 0.85 (0.67, 1.07) | 52% | 0.15 |
| No. of Datasets (No. of Studies) | Coffee Intake (Cups/Day) | I2(%) | Pheterogeneity | Pnon-linearity | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | |||||
| Total analysis | ||||||||||||
| Non-linear | 11 (9) | Ref. | 0.91 (0.85, 0.97) | 0.85 (0.76, 0.94) | 0.81 (0.73, 0.91) | 0.79 (0.70, 0.89) | 0.76 (0.67, 0.87) | 0.74 (0.64, 0.86) | 0.72 (0.61, 0.85) | 30.98 | 0.09 | 0.09 |
| Linear | 11 (9) | Ref. | 0.95 (0.92, 0.97) | 0.90 (0.85, 0.94) | 0.85 (0.78, 0.92) | 0.80 (0.72, 0.89) | 0.76 (0.67, 0.86) | 0.72 (0.61, 0.84) | 0.68 (0.57, 0.81) | 59.21 | 0.01 | NA |
| Postmenopausal | ||||||||||||
| Non-linear | 7 (6) | Ref. | 0.92 (0.83, 1.01) | 0.85 (0.73, 0.99) | 0.80 (0.69, 0.92) | 0.75 (0.65, 0.87) | 0.71 (0.61, 0.83) | 0.67 (0.56, 0.79) | 0.63 (0.52, 0.76) | 0 | 0.64 | 0.67 |
| Linear | 7 (6) | Ref. | 0.93 (0.91, 0.95) | 0.87 (0.83, 0.91) | 0.81 (0.76, 0.87) | 0.76 (0.69, 0.83) | 0.71 (0.63, 0.79) | 0.66 (0.58, 0.76) | 0.62 (0.53, 0.72) | 0 | 0.46 | NA |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lafranconi, A.; Micek, A.; Galvano, F.; Rossetti, S.; Del Pup, L.; Berretta, M.; Facchini, G. Coffee Decreases the Risk of Endometrial Cancer: A Dose–Response Meta-Analysis of Prospective Cohort Studies. Nutrients 2017, 9, 1223. https://doi.org/10.3390/nu9111223
Lafranconi A, Micek A, Galvano F, Rossetti S, Del Pup L, Berretta M, Facchini G. Coffee Decreases the Risk of Endometrial Cancer: A Dose–Response Meta-Analysis of Prospective Cohort Studies. Nutrients. 2017; 9(11):1223. https://doi.org/10.3390/nu9111223
Chicago/Turabian StyleLafranconi, Alessandra, Agnieszka Micek, Fabio Galvano, Sabrina Rossetti, Lino Del Pup, Massimiliano Berretta, and Gaetano Facchini. 2017. "Coffee Decreases the Risk of Endometrial Cancer: A Dose–Response Meta-Analysis of Prospective Cohort Studies" Nutrients 9, no. 11: 1223. https://doi.org/10.3390/nu9111223
APA StyleLafranconi, A., Micek, A., Galvano, F., Rossetti, S., Del Pup, L., Berretta, M., & Facchini, G. (2017). Coffee Decreases the Risk of Endometrial Cancer: A Dose–Response Meta-Analysis of Prospective Cohort Studies. Nutrients, 9(11), 1223. https://doi.org/10.3390/nu9111223






