Impact of Food Components on in vitro Calcitonin Gene-Related Peptide Secretion—A Potential Mechanism for Dietary Influence on Migraine
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Extractions
2.3. Cell Culture Maintenance
2.4. Cytotoxicity Assay
2.5. CGRP Assay
2.6. Calcium Influx
2.7. Statistical Analysis
3. Results
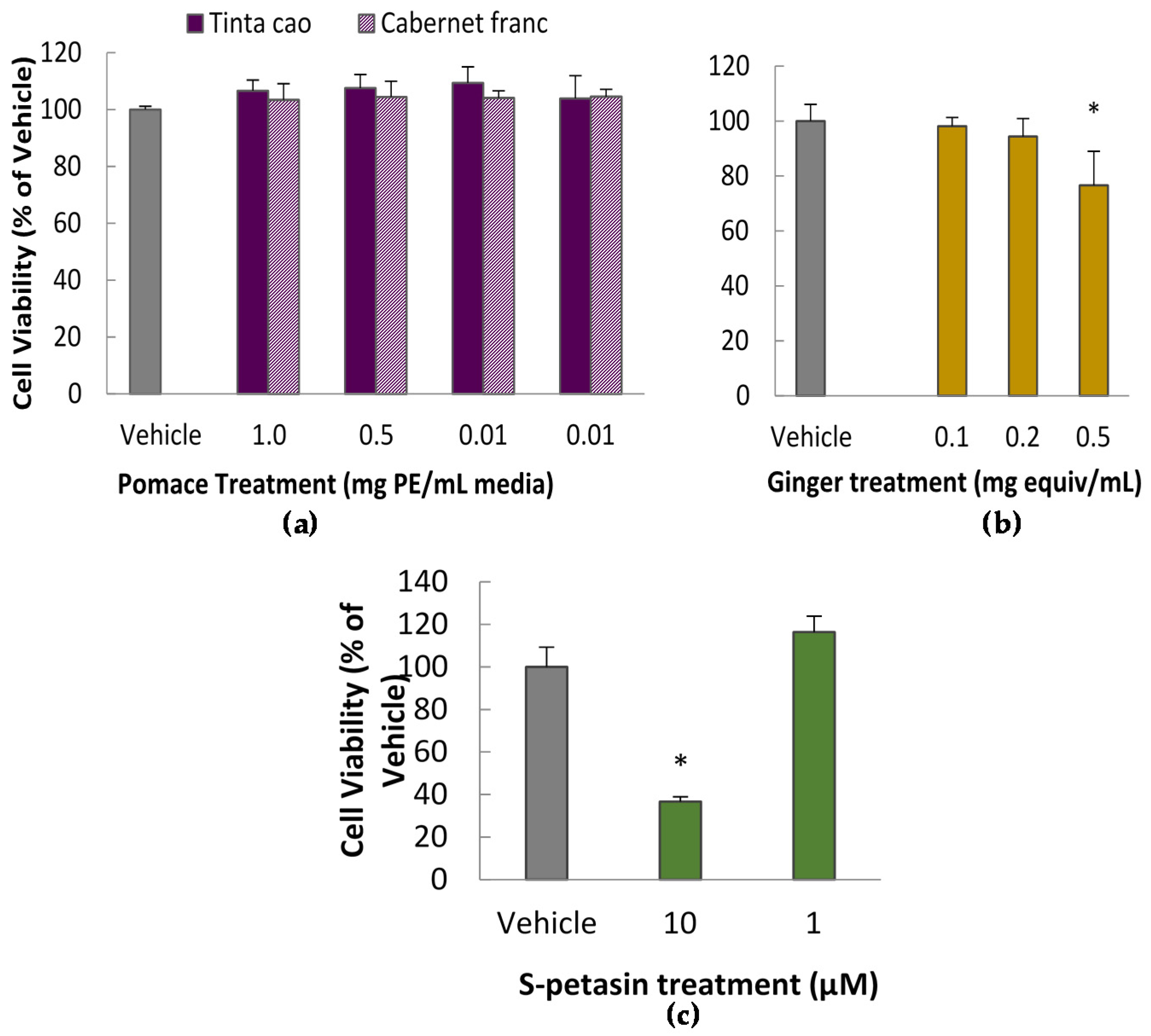
3.1. Cytotoxicity Assay
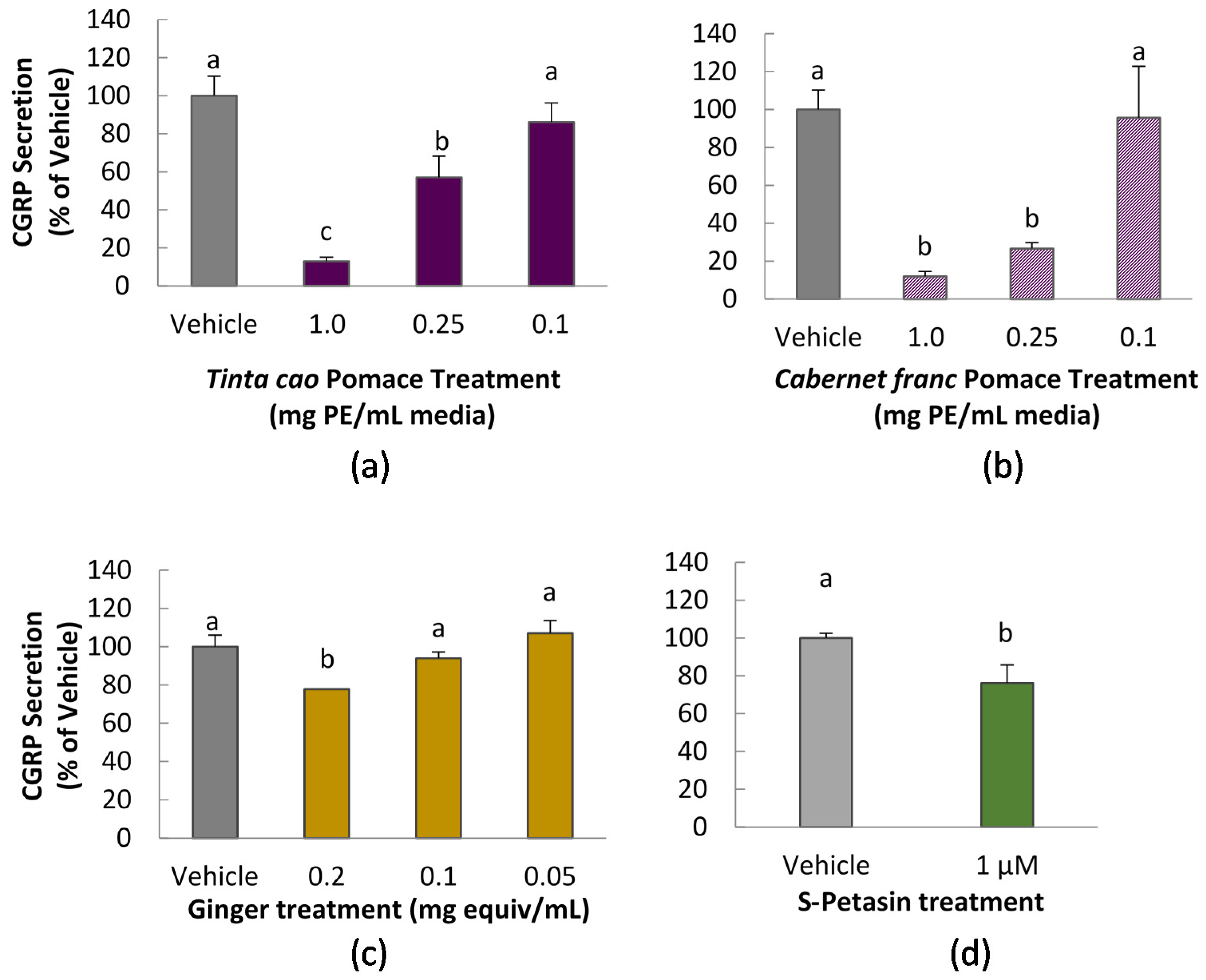
3.2. CGRP Secretion
3.3. Effect on Calcium Influx
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Stewart, W.; Wood, C.; Reed, M.; Roy, J.; Lipton, R. Cumulative lifetime migraine incidence in women and men. Cephalalgia 2008, 28, 1170–1178. [Google Scholar] [CrossRef] [PubMed]
- Vos, T.; Flaxman, A.D.; Naghavi, M.; Lozano, R.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S.; Aboyans, V. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2013, 380, 2163–2196. [Google Scholar] [CrossRef]
- Baillie, L.D.; Ahn, A.H.; Mulligan, S.J. Sumatriptan inhibition of N-type calcium channel mediated signaling in dural CGRP terminal fibres. Neuropharmacology 2012, 63, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Recober, A.; Russo, A.F. Calcitonin gene-related peptide: An update on the biology. Curr. Opin. Neurol. 2009, 22, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Nahas, S.J.; Peterlin, B.L. Chemical mediators of migraine: Preclinical and clinical observations. Headache J. Head Face Pain 2011, 51, 1029–1045. [Google Scholar] [CrossRef] [PubMed]
- Silberstein, S.D.; Edvinsson, L. Is CGRP a marker for chronic migraine? Neurology 2013, 81, 1184–1185. [Google Scholar] [CrossRef] [PubMed]
- Lassen, L.H.; Haderslev, P.A.; Jacobsen, V.B.; Iversen, H.K.; Sperling, B.; Olesen, J. CGRP may play a causative role in migraine. Cephalalgia 2002, 22, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Goadsby, P.J.; Edvinsson, L. The trigeminovascular system and migraine: Studies characterizing cerebrovascular and neuropeptide changes seen in humans and cats. Ann. Neurol. 1993, 33, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Zelissen, P.M.; Koppeschaar, H.P.; Lips, C.J.; Hackeng, W.H. Calcitonin gene-related peptide in human obesity. Peptides 1991, 12, 861–863. [Google Scholar] [CrossRef]
- Abbey, M.J.; Patil, V.V.; Vause, C.V.; Durham, P.L. Repression of calcitonin gene-related peptide expression in trigeminal neurons by a Theobroma cacao extract. J. Ethnopharmacol. 2008, 115, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Cady, R.J.; Durham, P.L. Cocoa-enriched diets enhance expression of phosphatases and decrease expression of inflammatory molecules in trigeminal ganglion neurons. Brain Res. 2010, 1323, 18–32. [Google Scholar] [CrossRef] [PubMed]
- Cady, R.J.; Hirst, J.J.; Durham, P.L. Dietary grape seed polyphenols repress neuron and glia activation in trigeminal ganglion and trigeminal nucleus caudalis. Mol. Pain 2010, 6, 91. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.-Y.; Jing, M.-Y.; Wang, J.-F.; Weng, X.-Y. The approach to the mechanism of calcitonin gene-related peptide-inducing inhibition of food intake: Calcitonin gene-related peptide and food intake. J. Anim. Physiol. Anim. Nutr. 2010, 94, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Holland, S.; Silberstein, S.D.; Freitag, F.; Dodick, D.W.; Argoff, C.; Ashman, E. Evidence-based guideline update: NSAIDS and other complementary treatments for episodic migraine prevention in adults report of the quality standards subcommittee of the American Academy of Neurology and the American Headache Society. Neurology 2012, 78, 1346–1353. [Google Scholar] [CrossRef] [PubMed]
- Mahady, G.B.; Pendland, S.L.; Yun, G.S.; Lu, Z.-Z.; Stoia, A. Ginger (Zingiber officianale roscoe) and the Gingerols inhibit the growth of Cag A+ Strains of Helicobacter pylori. Anticancer Res. 2003, 23, 3699–3702. [Google Scholar] [PubMed]
- Parry, J.; Su, L.; Moore, J.; Cheng, Z.; Luther, M.; Rao, J.N.; Wang, J.-Y.; Yu, L.L. Chemical compositions, antioxidant capacities, and antiproliferative activities of selected fruit seed flours. J. Agric. Food Chem. 2006, 54, 3773–3778. [Google Scholar] [CrossRef] [PubMed]
- Parry, J.W.; Li, H.; Liu, J.-R.; Zhou, K.; Zhang, L.; Ren, S. Antioxidant activity, antiproliferation of colon cancer cells, and chemical composition of grape pomace. Food Nutr. Sci. 2011, 2, 530–540. [Google Scholar] [CrossRef]
- Yoon, H.; Liu, R.H. Effect of selected phytochemicals and apple extracts on NF-κB activation in human breast cancer MCF-7 cells. J. Agric. Food Chem. 2007, 55, 3167–3173. [Google Scholar] [CrossRef] [PubMed]
- Durham, P.L.; Masterson, C.G. Two mechanisms involved in trigeminal CGRP release: Implications for migraine treatment. Headache J. Head Face Pain 2013, 53, 67–80. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Ahmedna, M. Functional components of grape pomace: Their composition, biological properties and potential applications. Int. J. Food Sci. Technol. 2013, 48, 221–237. [Google Scholar] [CrossRef]
- Ali, B.H.; Blunden, G.; Tanira, M.O.; Nemmar, A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): A review of recent research. Food Chem. Toxicol. 2008, 46, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.-X.; Tang, X.-D.; Wang, F.-Y.; Duan, Z.-J.; Li, Y.-C.; Qiu, J.-J.; Guo, H.-S. Effect of gingerol on colonic motility via inhibition of calcium channel currents in rats. World J. Gastroenterol. 2015, 21, 13466–13472. [Google Scholar] [CrossRef] [PubMed]
- Cady, R.K.; Goldstein, J.; Nett, R.; Mitchell, R.; Beach, M.E.; Browning, R. A double-blind placebo-controlled pilot study of sublingual feverfew and ginger (LipiGesicTMM) in the treatment of migraine: July/August 2011. Headache J. Head Face Pain 2011, 51, 1078–1086. [Google Scholar] [CrossRef] [PubMed]
- Ko, W.-C.; Lei, C.-B.; Lin, Y.-L.; Chen, C.-F. Mechanisms of relaxant action of S-Petasin and S-Isopetasin sesquiterpenes of Petasites formosanus, in isolated guinea pig trachea. Planta Med. 2001, 67, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.-N.; Chen, H.; Lin, Y.-L. The mechanism of inhibitory actions of S-petasin, a sesquiterpene of Petasites formosanus, on l-type calcium current in NG108-15 neuronal cells. Planta Med. 2003, 69, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.-J.; Shum, A.Y.-C.; Lin, Y.-L.; Wu, X.-C.; Ren, J.; Chen, C.-F. Calcium channel blockade in vascular smooth muscle cells- major hypotensive mechansim of S-petasin, a hypotensive sesquiterpene from Petasites formosanus. J. Pharmacol. Exp. Ther. 2001, 297, 240–247. [Google Scholar] [PubMed]
- Sheykhzade, M.; Smajilovic, S.; Issa, A.; Haunso, S.; Christensen, S.B.; Tfelt-Hansen, J. S-petasin and butterbur lactones dilate vessels through blockage of voltage gated calcium channels and block DNA synthesis. Eur. J. Pharmacol. 2008, 593, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Fiebich, B.L.; Grozdeva, M.; Hess, S.; Hull, M.; Danesch, U.; Bodensieck, A.; Bauer, R. Petasites hybridus extracts in vitro inhibit COX-2 and PGE2 release by direct interaction with the enzyme and by preventing p42/44 MAP kinase activation in rat primary microglial cells. Planta Med. 2005, 71, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Thomet, O.A.R.; Wiesmann, U.N.; Schapowal, A.; Bizer, C.; Simon, H.-U. Thomet 2001—Role of petasin in the potential anti-inflmmatory activity of a plant extract of petasites hybridus. Biochem. Pharmacol. 2001, 61, 1041–1047. [Google Scholar] [CrossRef]
- Aydın, A.A.; Zerbes, V.; Parlar, H.; Letzel, T. The medical plant butterbur (Petasites): Analytical and physiological (re)view. J. Pharm. Biomed. Anal. 2013, 75, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Pedersen-Bjergaard, U.; Host, U.; Kelbaek, H.; Schifter, S.; Rehfeld, J.F.; Faber, J.; Christensen, N.J. Influence of meal composition on postprandial peripheral plasma concentrations of vasoactive peptides in man. Scand. J. Clin. Lab. Investig. 1996, 56, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Borkum, J.M. Migraine triggers and oxidative stress: A narrative review and synthesis: Headache. Headache J. Head Face Pain 2016, 56, 12–35. [Google Scholar] [CrossRef] [PubMed]
- Srikiatkhachorn, A.; Grand, S.M.; Supornsilpchai, W.; Storer, R.J. Pathophysiology of medication overuse headache—An update. Headache J. Head Face Pain 2014, 54, 204–210. [Google Scholar] [CrossRef] [PubMed]
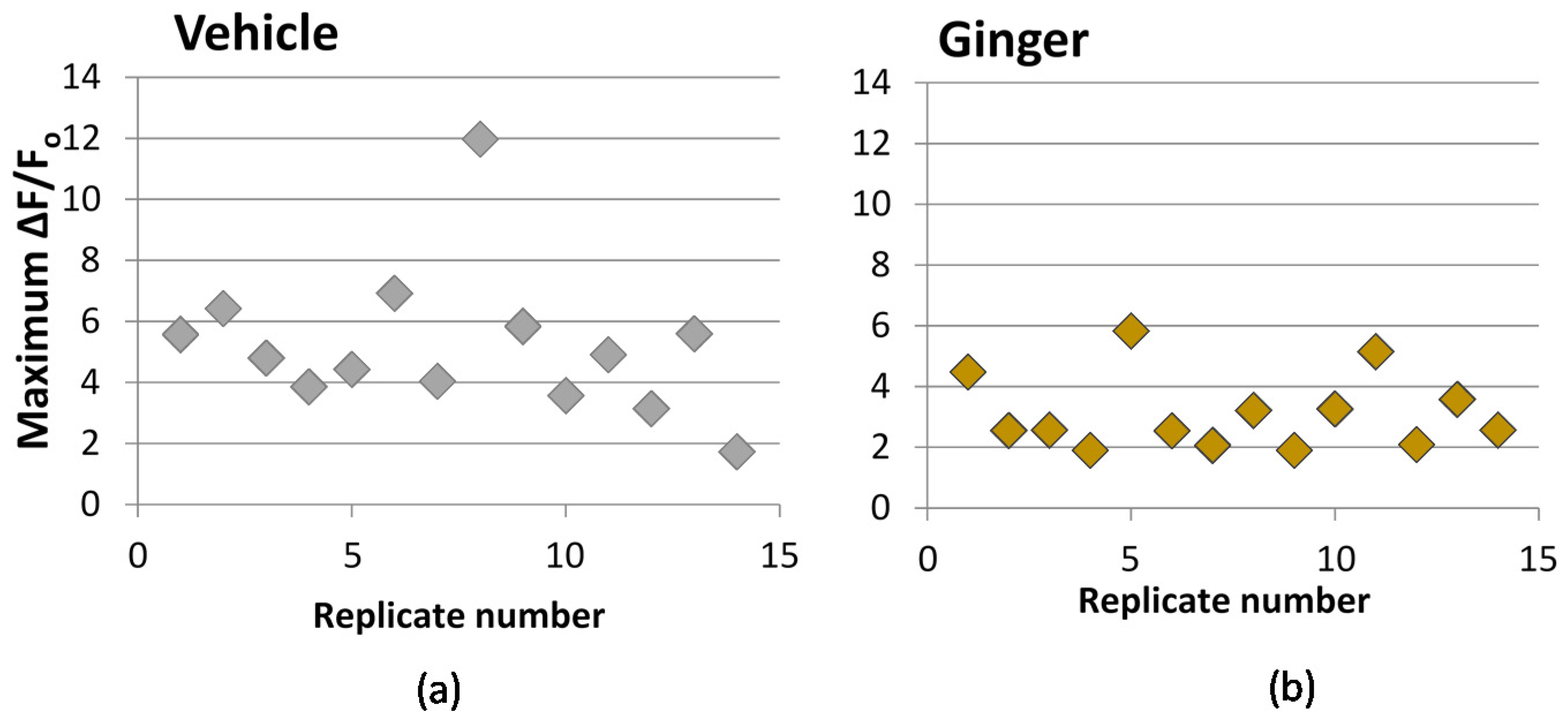
| Sample | Treatment Concentration | ΔF/Fo (% Vehicle) 2 | n | P |
|---|---|---|---|---|
| Tinta cao extract | 1.0 mg PE/mL 3 | 66.2 (55.7) | 13 | 0.146 |
| Cabernet franc extract | 1.0 mg PE/mL 3 | 76.8 (43.2) | 14 | 0.365 |
| Ginger extract | 0.2 mg equivalent/mL | 60.1 (23.9) | 14 | 0.007 |
| S-Petasin | 10 μM | 22.3 (8.4) | 6 | <0.001 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Slavin, M.; Bourguignon, J.; Jackson, K.; Orciga, M.-A. Impact of Food Components on in vitro Calcitonin Gene-Related Peptide Secretion—A Potential Mechanism for Dietary Influence on Migraine. Nutrients 2016, 8, 406. https://doi.org/10.3390/nu8070406
Slavin M, Bourguignon J, Jackson K, Orciga M-A. Impact of Food Components on in vitro Calcitonin Gene-Related Peptide Secretion—A Potential Mechanism for Dietary Influence on Migraine. Nutrients. 2016; 8(7):406. https://doi.org/10.3390/nu8070406
Chicago/Turabian StyleSlavin, Margaret, Julia Bourguignon, Kyle Jackson, and Michael-Angelo Orciga. 2016. "Impact of Food Components on in vitro Calcitonin Gene-Related Peptide Secretion—A Potential Mechanism for Dietary Influence on Migraine" Nutrients 8, no. 7: 406. https://doi.org/10.3390/nu8070406
APA StyleSlavin, M., Bourguignon, J., Jackson, K., & Orciga, M.-A. (2016). Impact of Food Components on in vitro Calcitonin Gene-Related Peptide Secretion—A Potential Mechanism for Dietary Influence on Migraine. Nutrients, 8(7), 406. https://doi.org/10.3390/nu8070406




