Anti-Stress, Behavioural and Magnetoencephalography Effects of an l-Theanine-Based Nutrient Drink: A Randomised, Double-Blind, Placebo-Controlled, Crossover Trial
Abstract
:1. Introduction
2. Methods
2.1. Participants
| Variable | Full Sample (n = 34) | Subset MEG Participants (n = 17) | |
|---|---|---|---|
| Gender | Male | 15 | 8 |
| Female | 19 | 9 | |
| Age (years) | Mean | 26.53 | 27.03 |
| SD | (5.04) | (5.61) | |
| BMI (kg/m2) | Mean | 22.66 | 22.99 |
| SD | (2.86) | (2.86) | |
| Trait Anxiety | Mean | 37.56 | 37.76 |
| (STAI-T) | SD | (7.44) | (6.95) |
| Years of Education | Mean SD | 17.25 (2.85) | 17.74 (3.10) |
2.2. Procedure
2.3. Treatments
Analysis of Theanine Content
2.4. Cognitive Stressor: Multi-Tasking Framework
2.5. Assessment of Stress, Mood and Fatigue
2.5.1. Bond–Lader Visual Analogue Mood Scales
2.5.2. Stress and Fatigue Visual Analogue Mood Scales
2.5.3. State-Trait Anxiety Inventory: State Subscale
2.6. Salivary Cortisol
2.7. MEG Subset
2.8. Data Analysis: Data Screening and MTF Stressor Responders
2.9. Data Analysis: Mood, Cognitive Performance and Cortisol
2.10. Data Analysis: MEG Data
3. Results
3.1. Primary Outcome: Subjective Stress Response to MTF
| 1 h | 3 h | ||||
|---|---|---|---|---|---|
| N | M | (SD) | M | (SD) | |
| Placebo | 26 | 1.58 | (21.54) | −6.08 | (21.70) |
| Active | 26 | −13.73 | (18.49) | −6.38 | (23.48) |
| ** | N.S. | ||||
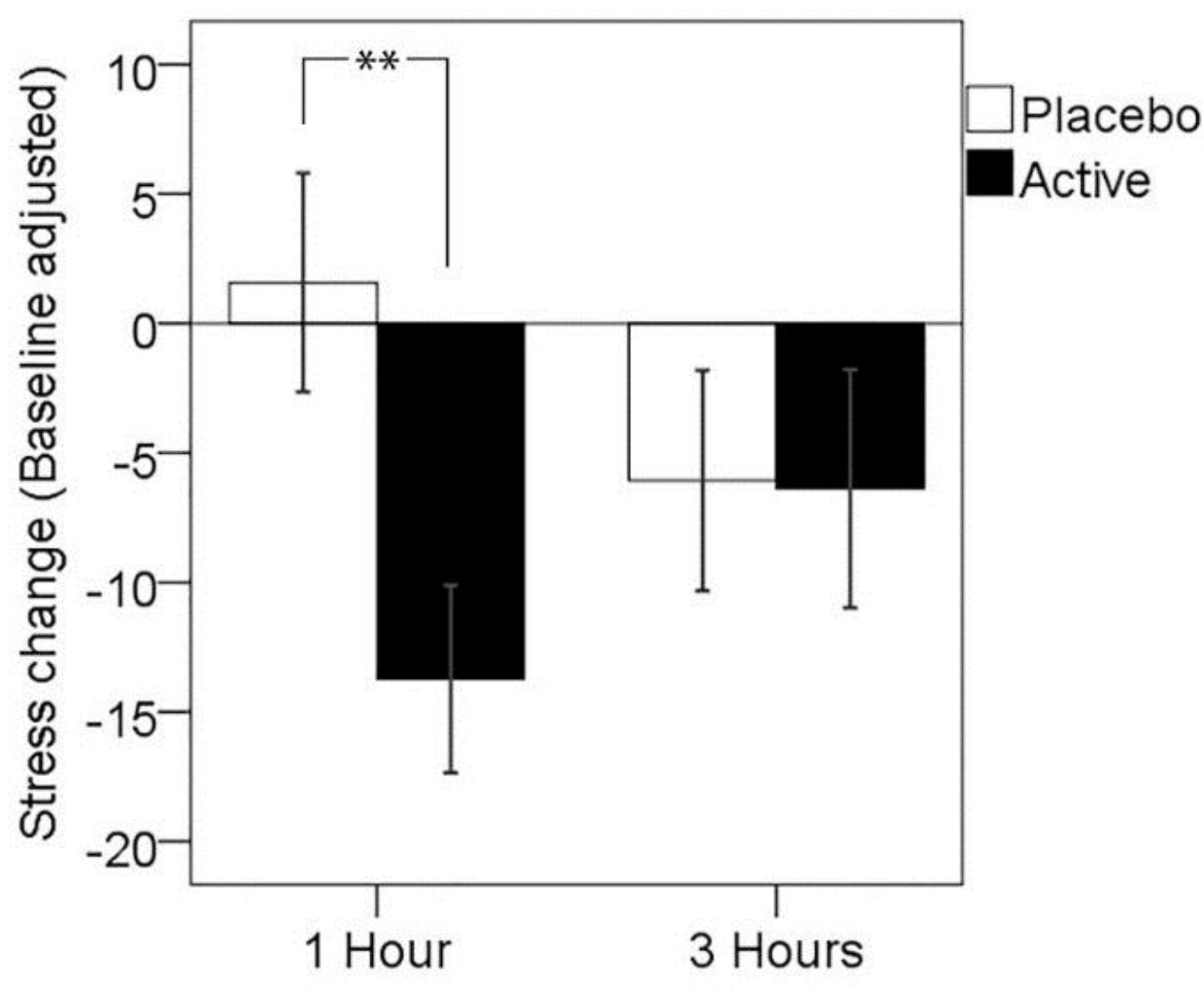
3.2. Mood and Fatigue Response to MTF
| 1 h | 3 h | Treatment × Assessment Time p-Values | |||||
|---|---|---|---|---|---|---|---|
| Placebo Active | Placebo Active | ||||||
| n | M | M | M | M | Treatment | Interaction | |
| (SD) | (SD) | (SD) | (SD) | ||||
| Fatigue | 25 | −3.32 | −5.96 | −4.80 | −7.44 | 0.663 | 0.999 |
| (23.63) | (22.48) | (23.99) | (23.62) | ||||
| Alertness | 26 | −0.72 | 3.86 | 5.28 | 5.69 | 0.375 | 0.267 |
| (14.19) | (15.44) | (11.77) | (11.30) | ||||
| Contentedness | 26 | −0.56 | 1.41 | 1.99 | 0.62 | 0.885 | 0.152 |
| (7.90) | (7.81) | (9.29) | (7.89) | ||||
| Calmness | 26 | −0.31 | 3.87 | 0.25 | 0.04 | 0.624 | 0.418 |
| (18.39) | (21.36) | (13.61) | (14.64) | ||||
| Anxiety (STAI-S) | 26 | 1.50 | −1.81 | 0.12 | −1.00 | 0.201 | 0.251 |
| (7.12) | (6.20) | (5.88) | (6.36) | ||||
“Tranquil-Troubled” Rating at Rest
| 1 h | 3 h | ||||
|---|---|---|---|---|---|
| n | M | (SD) | M | (SD) | |
| Placebo | 26 | 4.23 | (13.61) | 2.54 | (11.83) |
| Active | 26 | 0.69 | (9.69) | 1.50 | (15.19) |
3.3. Cortisol Response to MTF
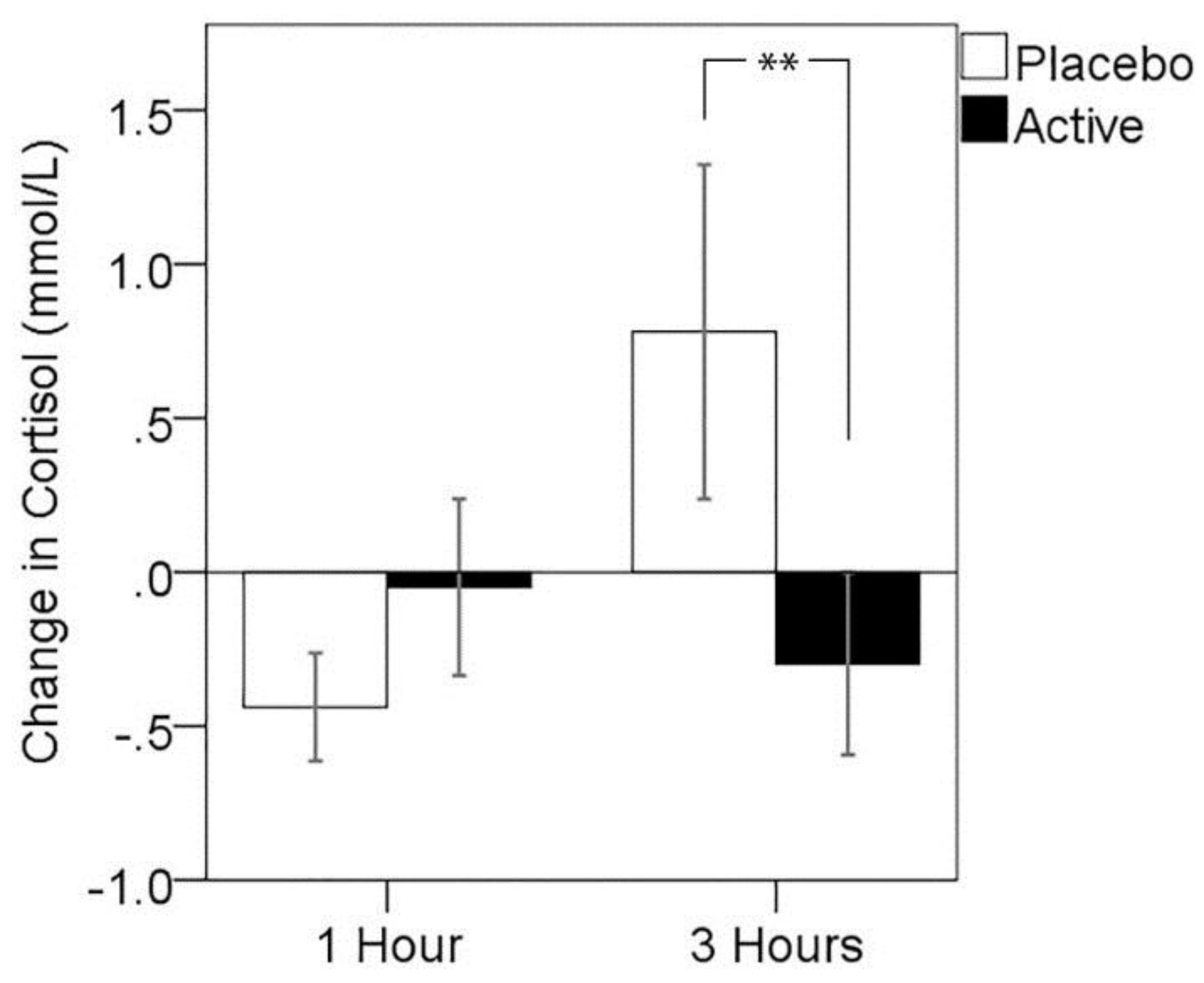
3.4. Cognitive Performance on MTF
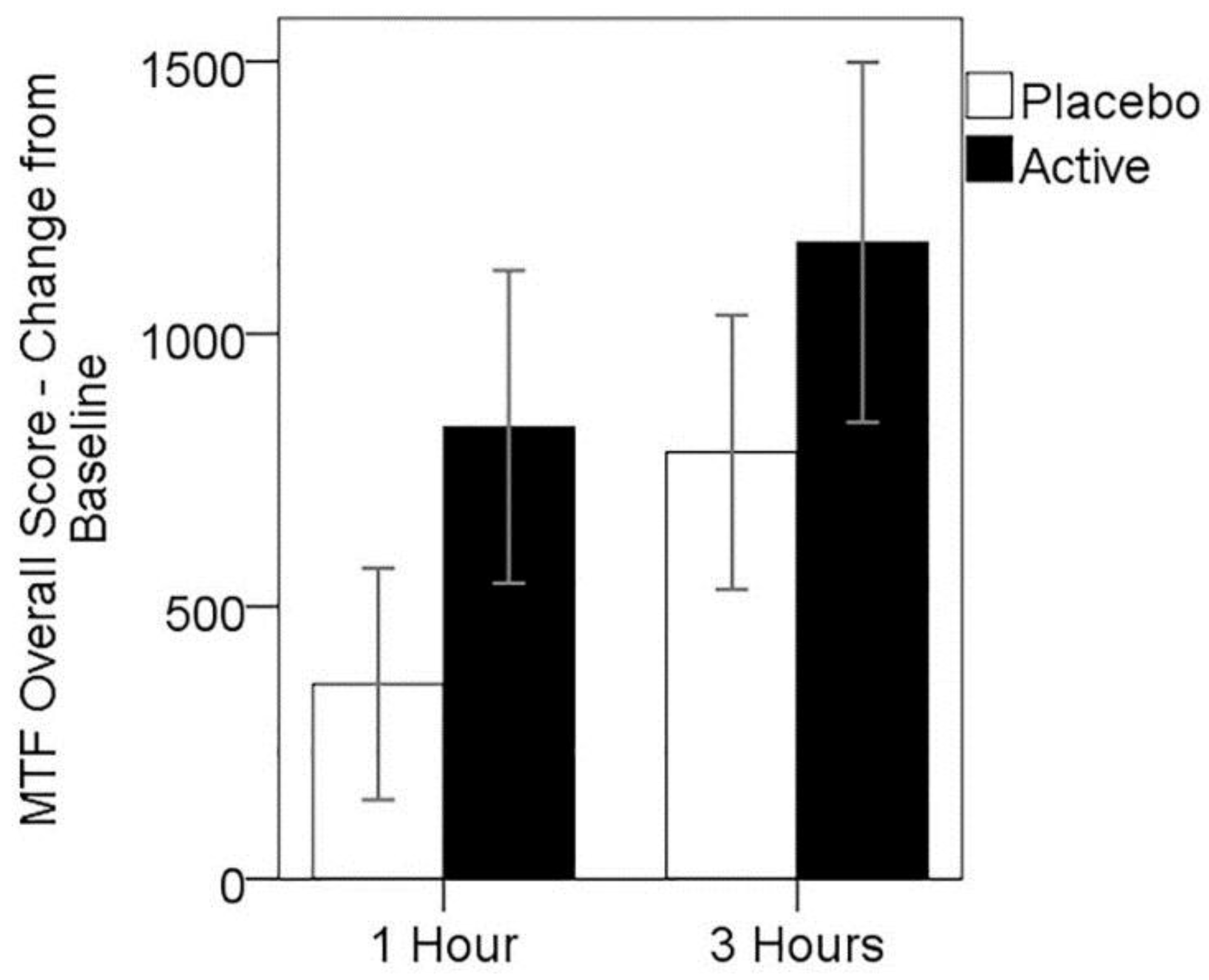
3.5. MEG Resting State Alpha Activity
3.5.1. Resting State Alpha Oscillatory Activity Changes and Trait Anxiety
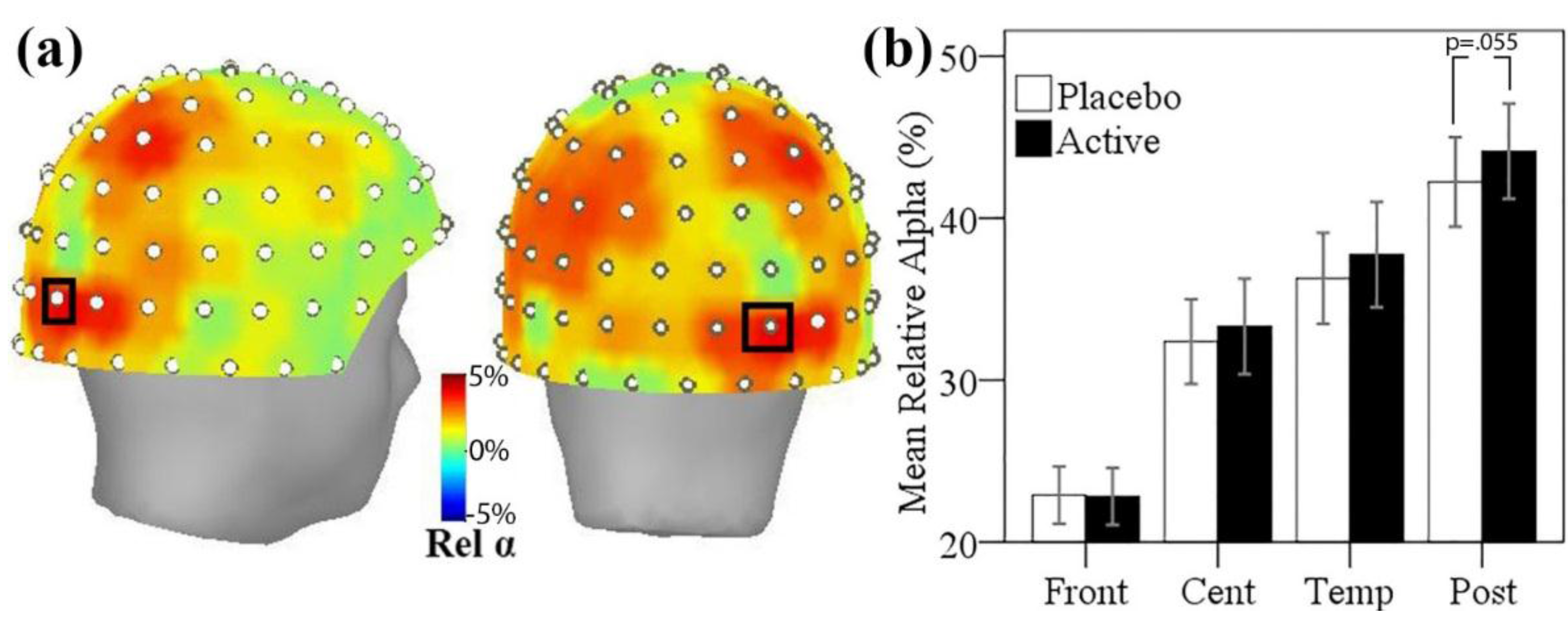
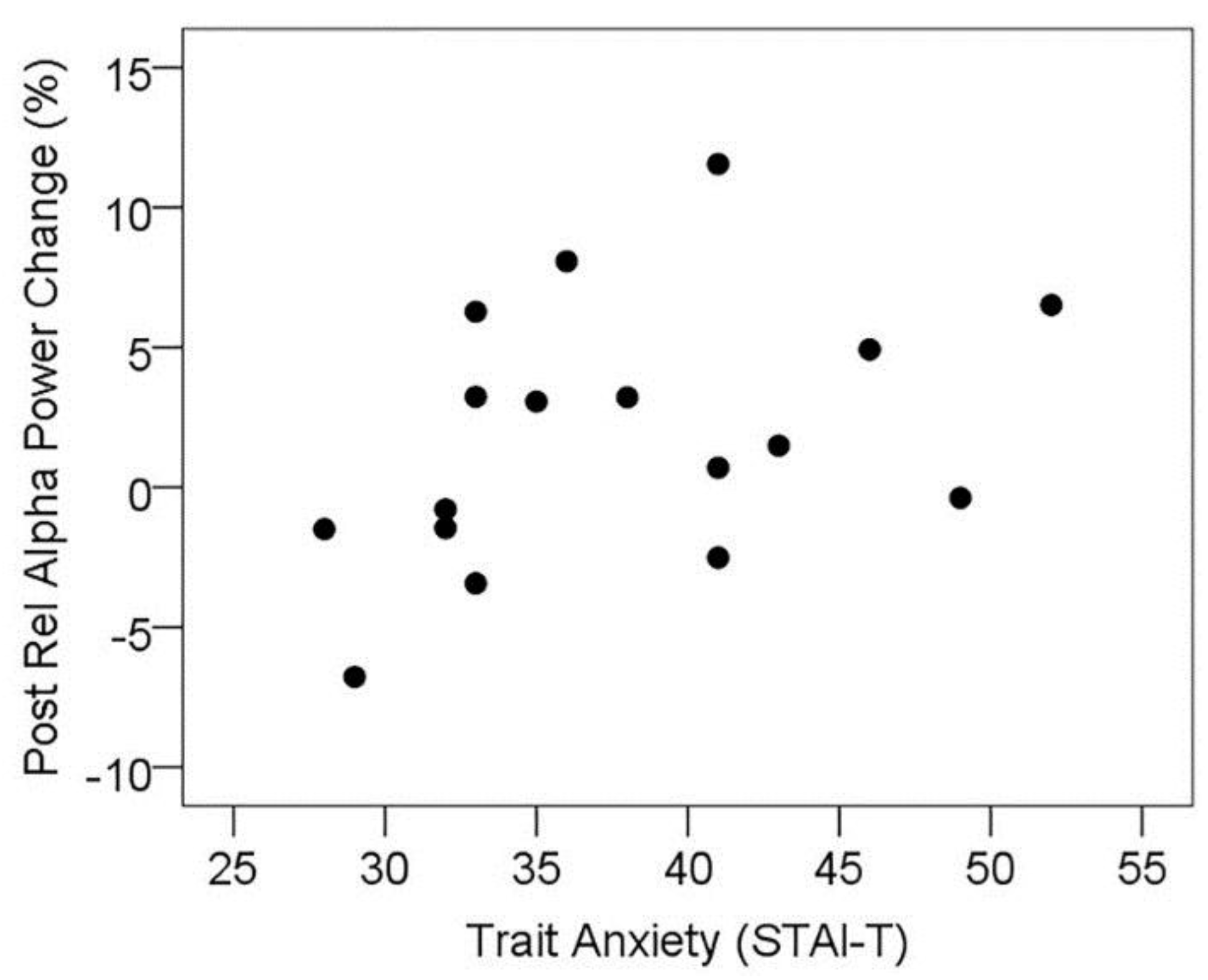
3.5.2. Resting State Alpha Oscillatory Activity Changes and Other Treatment Effects
| Low Anxiety (n = 8) | High Anxiety (n = 9) | |||
|---|---|---|---|---|
| M | (SD) | M | (SD) | |
| Placebo | 43.12 | (11.94) | 41.46 | (11.56) |
| Active | 42.95 | (14.28) | 45.19 | (10.63) |
| N.S. | * | |||
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Feldheim, W.; Yongvanit, P.; Cummings, P.H. Investigation of the presence and significance of theanine in the tea plant. J. Sci. Food Agric. 1986, 37, 527–534. [Google Scholar] [CrossRef]
- Finger, A.; Kuhr, S.; Engelhardt, U.H. Chromatography of tea constituents. J. Chromatogr. 1992, 624, 293–315. [Google Scholar] [CrossRef]
- Keenan, E.K.; Finnie, M.D.A.; Jones, P.S.; Rogers, P.J.; Priestley, C.M. How much theanine in a cup of tea? Effects of tea type and method of preparation. Food Chem. 2011, 125, 588–594. [Google Scholar] [CrossRef]
- Yokogoshi, H.; Kobayashi, M.; Mochizuki, M.; Terashima, T. Effect of theanine, r-glutamylethylamide, on brain monoamines and striatal dopamine release in conscious rats. Neurochem. Res. 1998, 23, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Unno, T.; Suzuki, Y.; Kakuda, T.; Hayakawa, T.; Tsuge, H. Metabolism of theanine, γ-glutamylethylamide, in rats. J. Agric. Food Chem. 1999, 47, 1593–1596. [Google Scholar] [CrossRef] [PubMed]
- Van der Pijla, P.; Chenb, L.; Muldera, T. Human disposition of l-theanine in tea or aqueous solution. J. Funct. Foods 2010, 2, 239–244. [Google Scholar] [CrossRef]
- Kakuda, T.; Nozawa, A.; Sugimoto, A.; Niino, H. Inhibition by theanine of binding of [3H] AMPA, [3H] kainate, and [3H] MDL 105,519 to glutamate receptors. Biosci. Biotechnol. Biochem. 2002, 66, 2683–2686. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T.; Sadzuka, Y.; Tanaka, K.; Sonobe, T. Inhibition of glutamate transporter by theanine enhances the therapeutic efficacy of doxorubicin. Toxicol. Lett. 2001, 121, 89–96. [Google Scholar] [CrossRef]
- Kimura, R.; Murata, T. Influence of alkylamides of glutamic acid and related compounds on the central nervous I. central depressant effect of theanine. Chem. Pharm. Bull. 1971, 19, 1257–1261. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Terashima, T.; Kawano, S.; Furuno, R.; Okubo, T.; Juneja, L.; Yokogoshi, H. Theanine, γ-glutamylethylamide, a unique amino acid in tea leaves, modulates neurotransmitter concentrations in the brain striatum interstitium in conscious rats. Amino Acids 2009, 36, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Yokogoshi, H.; Kato, Y.; Sagesaka, Y.M.; Takihara-Matsuura, T.; Kakuda, T.; Takeuchi, N. Reduction effect of theanine on blood pressure and brain 5-hydroxyindoles in spontaneously hypertensive rats. Biosci. Biotechnol. Biochem. 1995, 59, 615–618. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, C.; Numakawa, T.; Ninomiya, M.; Chiba, S.; Kunugi, H. Behavioral and molecular evidence for psychotropic effects in l-theanine. Psychopharmacology 2012, 219, 1099–1109. [Google Scholar] [CrossRef] [PubMed]
- Tamano, H.; Fukura, K.; Suzuki, M.; Sakamoto, K.; Yokogoshi, H.; Takeda, A. Advantageous effect of theanine intake on cognition. Nutr. Neurosci. 2014, 17, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Kakuda, T. Neuroprotective effects of theanine and its preventive effects on cognitive dysfunction. Pharmacol. Res. 2011, 64, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Di, X.; Yan, J.; Zhao, Y.; Zhang, J.; Shi, Z.; Chang, Y.; Zhao, B. l-theanine protects the APP (Swedish mutation) transgenic SH-SY5Y cell against glutamate-induced excitotoxicity via inhibition of the NMDA receptor pathway. Neuroscience 2010, 168, 778–786. [Google Scholar] [CrossRef] [PubMed]
- Einother, S.J.; Martens, V.E. Acute effects of tea consumption on attention and mood. Am. J. Clin. Nutr. 2013, 98, 1700S–1708S. [Google Scholar] [CrossRef] [PubMed]
- Camfield, D.A.; Stough, C.; Farrimond, J.; Scholey, A.B. Acute effects of tea constituents l-theanine, caffeine, and epigallocatechin gallate on cognitive function and mood: A systematic review and meta-analysis. Nutr. Rev. 2014, 72, 507–522. [Google Scholar] [CrossRef] [PubMed]
- Haskell, C.F.; Dodd, F.L.; Wightman, E.L.; Kennedy, D.O. Behavioural effects of compounds co-consumed in dietary forms of caffeinated plants. Nutr. Res. Rev. 2013, 26, 49–70. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.; Gray, M.A.; Oliver, C.; Liley, D.T.; Harrison, B.J.; Bartholomeusz, C.F.; Phan, K.L.; Nathan, P.J. The acute effects of l-theanine in comparison with alprazolam on anticipatory anxiety in humans. Hum. Psychopharmacol. 2004, 19, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Bond, A.; Lader, M. The use of analogue scales in rating subjective feelings. Br. J. Med. Psychol. 1974, 47, 211–218. [Google Scholar] [CrossRef]
- Kimura, K.; Ozeki, M.; Juneja, L.R.; Ohira, H. l-theanine reduces psychological and physiological stress responses. Biol. Psychol. 2007, 74, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Yoto, A.; Motoki, M.; Murao, S.; Yokogoshi, H. Effects of l-theanine or caffeine intake on changes in blood pressure under physical and psychological stresses. J. Physiol. Anthropol. 2012, 31, 28. [Google Scholar] [CrossRef] [PubMed]
- Shacham, S. A shortened version of the Profile of Mood States. J. Pers. Assess. 1983, 47, 305–306. [Google Scholar] [CrossRef] [PubMed]
- Higashiyama, A.; Htay, H.H.; Ozeki, M.; Juneja, L.R.; Kapoor, M.P. Effects of l-theanine on attention and reaction time response. J. Funct. Foods 2011, 3, 171–181. [Google Scholar] [CrossRef]
- Rogers, P.J.; Smith, J.E.; Heatherley, S.V.; Pleydell-Pearce, C. Time for tea: Mood, blood pressure and cognitive performance effects of caffeine and theanine administered alone and together. Psychopharmacology 2008, 195, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Haskell, C.F.; Kennedy, D.O.; Milne, A.L.; Wesnes, K.A.; Scholey, A.B. The effects of l-theanine, caffeine and their combination on cognition and mood. Biol. Psychol. 2008, 77, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Juneja, L.R.; Chu, D.C.; Okubo, T.; Nagato, Y.; Yokogoshi, H. l-theanine—A unique amino acid of green tea and its relaxation effect in humans. Trends Food Sci. Technol. 1999, 10, 199–204. [Google Scholar] [CrossRef]
- Ito, K.; Nagato, Y.; Aoi, N.; Juneja, L.R.; Kim, M.; Yamamoto, H.; Sugimoto, S. Effects of l-theanine on the release of alpha-brain waves in human volunteers. Nippon Nōgei Kagakukaishi 1998, 72, 153–157. [Google Scholar]
- Nobre, A.C.; Rao, A.; Owen, G.N. l-theanine, a natural constituent in tea, and its effect on mental state. Asia Pac. J. Clin. Nutr. 2008, 17, 167–168. [Google Scholar] [PubMed]
- Kim, K.S.; Song, C.H.; Oh, H.J. Effects of theanine on the release of brain alpha wave in adult males. FASEB J. 2004, 18, A541–A542. [Google Scholar]
- Barry, R.J.; Clarke, A.R.; Johnstone, S.J. Caffeine and opening the eyes have additive effects on resting arousal measures. Clin. Neurophysiol. 2011, 122, 2010–2015. [Google Scholar] [CrossRef] [PubMed]
- Barry, R.J.; Rushby, J.A.; Wallace, M.J.; Clarke, A.R.; Johnstone, S.J.; Zlojutro, I. Caffeine effects on resting-state arousal. Clin. Neurophysiol. 2005, 116, 2693–2700. [Google Scholar] [CrossRef] [PubMed]
- Freyer, F.; Becker, R.; Dinse, H.R.; Ritter, P. State-dependent perceptual learning. J. Neurosci. 2013, 33, 2900–2907. [Google Scholar] [CrossRef] [PubMed]
- Foxe, J.J.; Morie, K.P.; Laud, P.J.; Rowson, M.J.; de Bruin, E.A.; Kelly, S.P. Assessing the effects of caffeine and theanine on the maintenance of vigilance during a sustained attention task. Neuropharmacology 2012, 62, 2320–2327. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Ramirez, M.; Higgins, B.A.; Rycroft, J.A.; Owen, G.N.; Mahoney, J.; Shpaner, M.; Foxe, J.J. The deployment of intersensory selective attention: A high-density electrical mapping study of the effects of theanine. Clin. Neuropharmacol. 2007, 30, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Ramirez, M.; Kelly, S.P.; Montesi, J.L.; Foxe, J.J. The effects of l-theanine on alpha-band oscillatory brain activity during a visuo-spatial attention task. Brain Topogr. 2009, 22, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Mu, W.; Zhang, T.; Jiang, B. An overview of biological production of l-theanine. Biotechnol. Adv. 2015, 33, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Kullenberg, D.; Taylor, L.A.; Schneider, M.; Massing, U. Health effects of dietary phospholipids. Lipids Health Dis. 2012, 11, 3. [Google Scholar] [CrossRef] [PubMed]
- Benton, D.; Donohoe, R.T.; Sillance, B.; Nabb, S. The influence of phosphatidylserine supplementation on mood and heart rate when faced with an acute stressor. Nutr. Neurosci. 2001, 4, 169–178. [Google Scholar] [PubMed]
- Hellhammer, J.; Fries, E.; Buss, C.; Engert, V.; Tuch, A.; Rutenberg, D.; Hellhammer, D. Effects of soy lecithin phosphatidic acid and phosphatidylserine complex (PAS) on the endocrine and psychological responses to mental stress. Stress 2004, 7, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Amsterdam, J.D.; Li, Y.; Soeller, I.; Rockwell, K.; Mao, J.J.; Shults, J. A Randomized, Double-Blind, Placebo-Controlled Trial of Oral Matricaria recutita (Chamomile) Extract Therapy for Generalized Anxiety Disorder. J. Clin. Psychopharmacol. 2009, 29, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Terashima, T.; Takido, J.; Yokogoshi, H. Time-dependent changes of amino acids in the serum, liver, brain and urine of rats administered with theanine. Biosci. Biotechnol. Biochem. 1999, 63, 615–618. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.J.; Armstrong, D.W. Analysis of derivatized and underivatized theanine enantiomers by high-performance liquid chromatography/atmospheric pressure ionization-mass spectrometry. Rapid Commun. Mass Spectrom. 2004, 18, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Wetherell, M.A.; Carter, K. The multitasking framework: The effects of increasing workload on acute psychobiological stress reactivity. Stress Health 2014, 30, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Scholey, A.; Savage, K.; O’Neill, B.V.; Owen, L.; Stough, C.; Priestley, C.; Wetherell, M. Effects of two doses of glucose and a caffeine-glucose combination on cognitive performance and mood during multi-tasking. Hum. Psychopharmacol. 2014, 29, 434–445. [Google Scholar] [CrossRef] [PubMed]
- Scholey, A.; Haskell, C.; Robertson, B.; Kennedy, D.; Milne, A.; Wetherell, M. Chewing gum alleviates negative mood and reduces cortisol during acute laboratory psychological stress. Physiol. Behav. 2009, 97, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Benson, S.; Downey, L.A.; Stough, C.; Wetherell, M.; Zangara, A.; Scholey, A. An acute, double-blind, placebo-controlled cross-over study of 320 mg and 640 mg doses of Bacopa monnieri (CDRI 08) on multitasking stress reactivity and mood. Phytother. Res. 2014, 28, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Pipingas, A.; Camfield, D.A.; Stough, C.; Cox, K.H.; Fogg, E.; Tiplady, B.; Sarris, J.; White, D.J.; Sali, A.; Wetherell, M.A.; et al. The effects of multivitamin supplementation on mood and general well-being in healthy young adults. A laboratory and at-home mobile phone assessment. Appetite 2013, 69, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Spielberger, C.D.; Gorsuch, R.L.; Lushene, R.E.; Vagg, P.R.; Jacobs, G.A. Manual for the State-Trait Anxiety Inventory; Consulting Psychologists Press: Palo Alto, CA, USA, 1983. [Google Scholar]
- Ahearn, E.P. The use of visual analog scales in mood disorders: A critical review. J. Psychiatr. Res. 1997, 31, 569–579. [Google Scholar] [CrossRef]
- Rossi, V.; Pourtois, G. Transient state-dependent fluctuations in anxiety measured using STAI, POMS, PANAS or VAS: A comparative review. Anxiety Stress Coping 2011, 25, 603–645. [Google Scholar] [CrossRef] [PubMed]
- Pruessner, J.C.; Wolf, O.T.; Hellhammer, D.H.; Buske-Kirschbaum, A.; von Auer, K.; Jobst, S.; Kaspers, F.; Kirschbaum, C. Free Cortisol Levels after Awakening: A Reliable Biological Marker for the Assessment of Adrenocortical Activity. Life Sci. 1997, 61, 2539–2549. [Google Scholar] [CrossRef]
- Debono, M.; Ghobadi, C.; Rostami-Hodjegan, A.; Huatan, H.; Campbell, M.J.; Newell-Price, J.; Darzy, K.; Merke, D.P.; Arlt, W.; Ross, R.J. Modified-release hydrocortisone to provide circadian cortisol profiles. J. Clin. Endocrinol. Metab. 2009, 94, 1548–1554. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O.; Little, W.; Scholey, A.B. Attenuation of Laboratory-Induced Stress in Humans After Acute Administration of Melissa officinalis (Lemon Balm). Psychosom. Med. 2004, 66, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O.; Little, W.; Haskell, C.F.; Scholey, A.B. Anxiolytic effects of a combination of Melissa officinalis and Valeriana officinalis during laboratory induced stress. Phytother. Res. 2006, 20, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O.; Pace, S.; Haskell, C.; Okello, E.J.; Milne, A.; Scholey, A.B. Effects of cholinesterase inhibiting sage (Salvia officinalis) on mood, anxiety and performance on a psychological stressor battery. Neuropsychopharmacolog 2006, 31, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Taulu, S.; Hari, R. Removal of magnetoencephalographic artifacts with temporal signal-space separation: Demonstration with single-trial auditory-evoked responses. Hum. Brain Mapp. 2009, 30, 1524–1534. [Google Scholar] [CrossRef] [PubMed]
- Tadel, F.; Baillet, S.; Mosher, J.C.; Pantazis, D.; Leahy, R.M. Brainstorm: A user-friendly application for MEG/EEG analysis. Comput. Intell. Neurosci. 2011, 2011, 879716. [Google Scholar] [CrossRef] [PubMed]
- Uusitalo, M.A.; Ilmoniemi, R.J. Signal-space projection method for separating MEG or EEG into components. Med. Biol. Eng. Comput. 1997, 35, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Gross, J.; Baillet, S.; Barnes, G.R.; Henson, R.N.; Hillebrand, A.; Jensen, O.; Jerbi, K.; Litvak, V.; Maess, B.; Oostenveld, R.; et al. Good practice for conducting and reporting MEG research. NeuroImage 2013, 65, 349–363. [Google Scholar] [CrossRef] [PubMed]
- Blair, R.C.; Karniski, W. An alternative method for significance testing of waveform difference potentials. Psychophysiology 1993, 30, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Groppe, D.M.; Urbach, T.P.; Kutas, M. Mass univariate analysis of event-related brain potentials/fields I: A critical tutorial review. Psychophysiology 2011, 48, 1711–1725. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Tanida, N.; Ishii, N.; Yamamoto, H.; Iguchi, K.; Hoshino, M.; Takeda, A.; Ozawa, H.; Ohkubo, T.; Juneja, L.R.; et al. Anti-stress effect of theanine on students during pharmacy practice: Positive correlation among salivary alpha-amylase activity, trait anxiety and subjective stress. Pharmacol. Biochem. Behav. 2013, 111, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Steptoe, A.; Gibson, E.L.; Vuononvirta, R.; Williams, E.D.; Hamer, M.; Rycroft, J.A.; Erusalimsky, J.D.; Wardle, J. The effects of tea on psychophysiological stress responsivity and post-stress recovery: A randomised double-blind trial. Psychopharmacolog 2007, 190, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Granger, D.A.; Kivlighan, K.T.; el-Sheikh, M.; Gordis, E.B.; Stroud, L.R. Salivary alpha-amylase in biobehavioral research: Recent developments and applications. Ann. N. Y. Acad. Sci. 2007, 1098, 122–144. [Google Scholar] [CrossRef] [PubMed]
- Foley, P.; Kirschbaum, C. Human hypothalamus–pituitary–adrenal axis responses to acute psychosocial stress in laboratory settings. Neurosci. Biobehav. Rev. 2010, 35, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Crawford, J.; Cayley, C.; Lovibond, P.F.; Wilson, P.H.; Hartley, C. Percentile Norms and Accompanying Interval Estimates from an Australian General Adult Population Sample for Self-Report Mood Scales (BAI, BDI, CRSD, CES-D, DASS, DASS-21, STAI-X, STAI-Y, SRDS, and SRAS). Aust. Psychol. 2011, 46, 3–14. [Google Scholar] [CrossRef]
- Hughes, J.R.; John, E.R. Conventional and Quantitative Electroencephalography in Psychiatry. J. Neuropsychiatry Clin. Neurosci. 1999, 11, 190–208. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
White, D.J.; De Klerk, S.; Woods, W.; Gondalia, S.; Noonan, C.; Scholey, A.B. Anti-Stress, Behavioural and Magnetoencephalography Effects of an l-Theanine-Based Nutrient Drink: A Randomised, Double-Blind, Placebo-Controlled, Crossover Trial. Nutrients 2016, 8, 53. https://doi.org/10.3390/nu8010053
White DJ, De Klerk S, Woods W, Gondalia S, Noonan C, Scholey AB. Anti-Stress, Behavioural and Magnetoencephalography Effects of an l-Theanine-Based Nutrient Drink: A Randomised, Double-Blind, Placebo-Controlled, Crossover Trial. Nutrients. 2016; 8(1):53. https://doi.org/10.3390/nu8010053
Chicago/Turabian StyleWhite, David J., Suzanne De Klerk, William Woods, Shakuntla Gondalia, Chris Noonan, and Andrew B. Scholey. 2016. "Anti-Stress, Behavioural and Magnetoencephalography Effects of an l-Theanine-Based Nutrient Drink: A Randomised, Double-Blind, Placebo-Controlled, Crossover Trial" Nutrients 8, no. 1: 53. https://doi.org/10.3390/nu8010053
APA StyleWhite, D. J., De Klerk, S., Woods, W., Gondalia, S., Noonan, C., & Scholey, A. B. (2016). Anti-Stress, Behavioural and Magnetoencephalography Effects of an l-Theanine-Based Nutrient Drink: A Randomised, Double-Blind, Placebo-Controlled, Crossover Trial. Nutrients, 8(1), 53. https://doi.org/10.3390/nu8010053





