Vitamin D Every Day to Keep the Infection Away?
Abstract
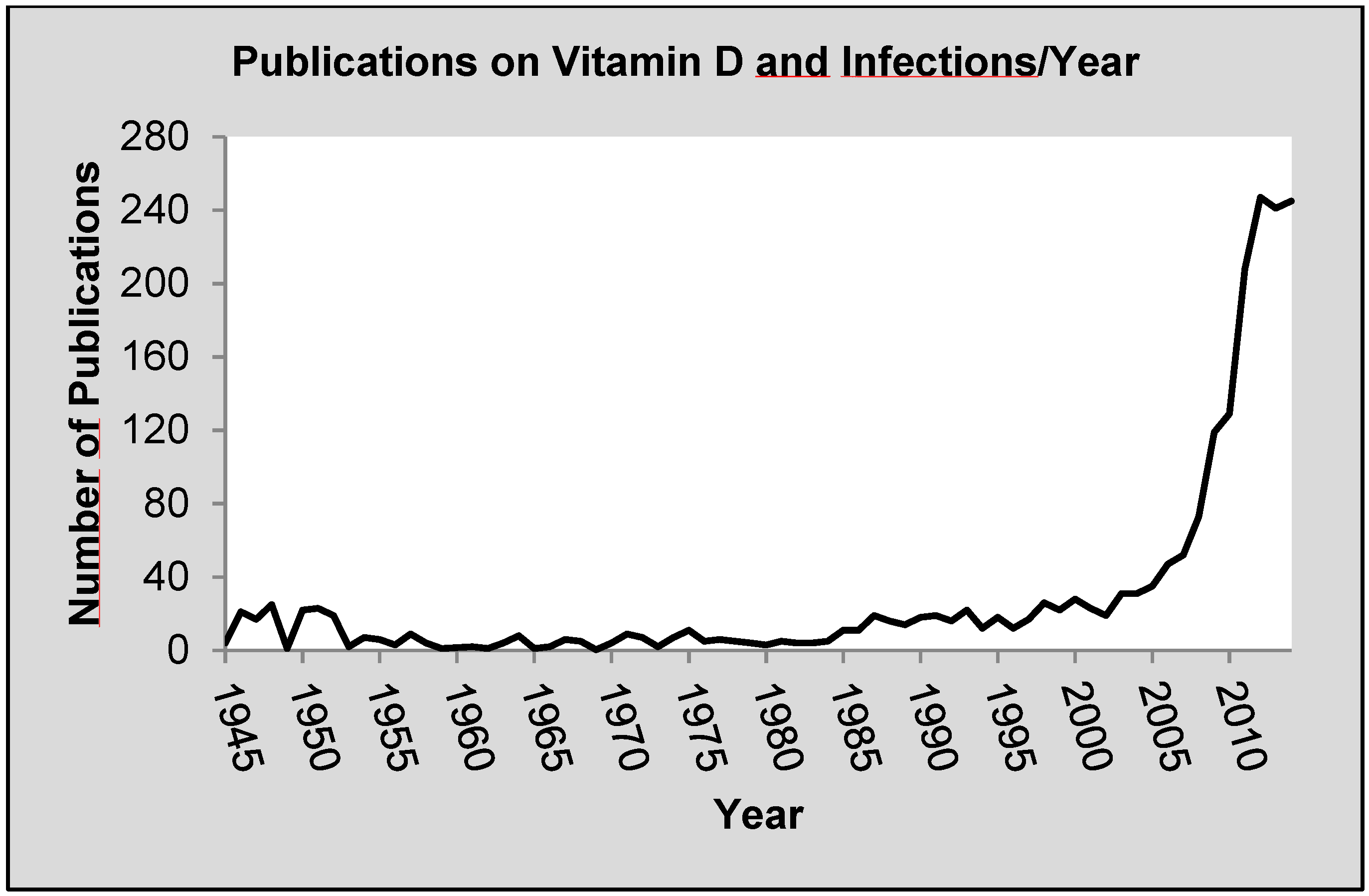
:1. Introduction
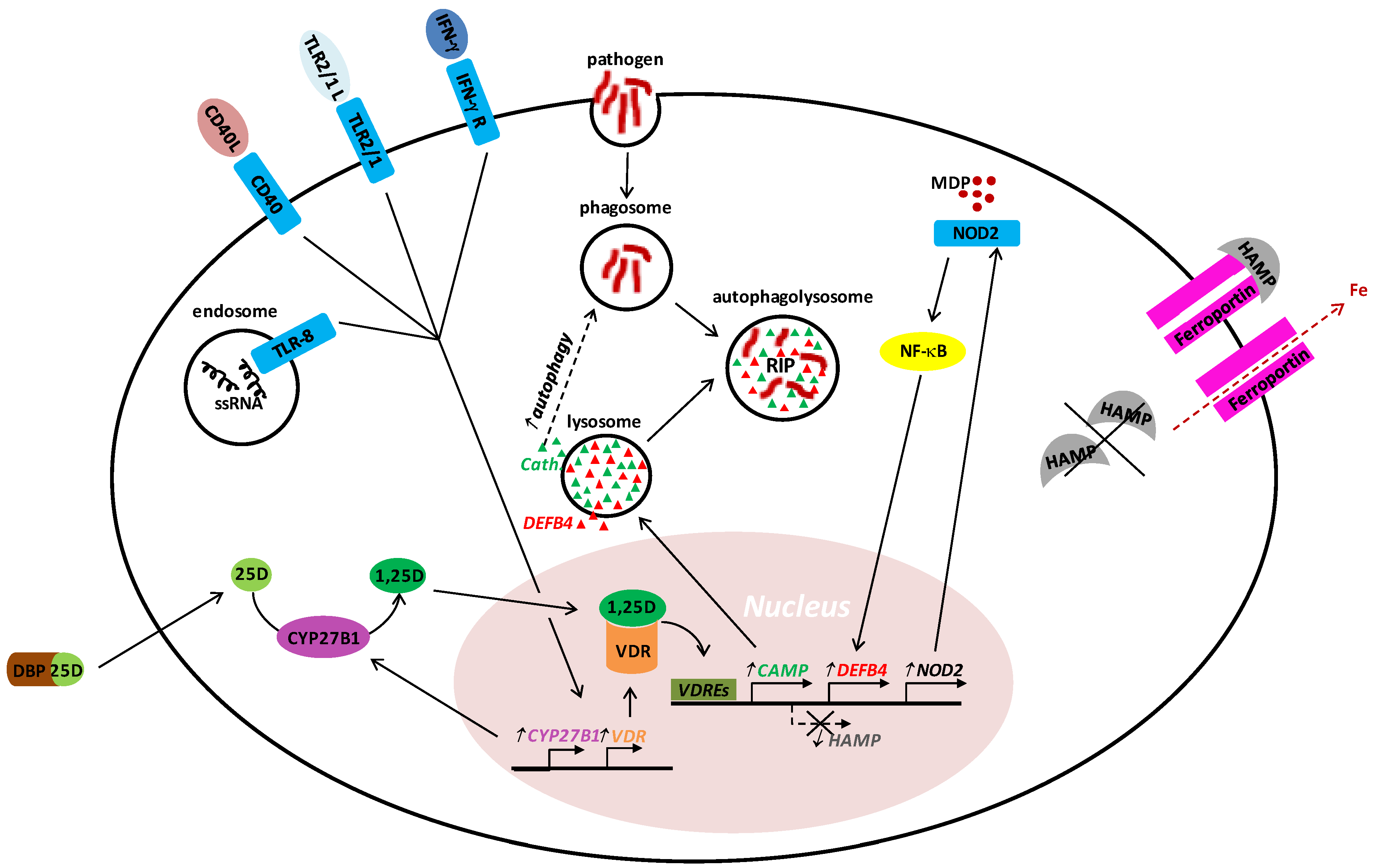
2. Vitamin D in Innate Host Defense
3. Vitamin D in Acquired Host Defense
4. Mouse Models of Infection
5. Comments on Therapeutic Intervention
Acknowledgements
Author Contributions
Conflict of Interests
References
- Norman, A.W. From vitamin D to hormone D: Fundamentals of the vitamin D endocrine system essential for good health. Am. J. Clin. Nutr. 2008, 88, 491S–499S. [Google Scholar] [PubMed]
- Hossein-nezhad, A.; Holick, M.F. Vitamin D for health: A global perspective. Mayo Clin. Proc. 2013, 88, 720–755. [Google Scholar] [CrossRef] [PubMed]
- Hossein-nezhad, A.; Spira, A.; Holick, M.F. Influence of vitamin D status and vitamin D3 supplementation on genome wide expression of white blood cells: A randomized double-blind clinical trial. PLoS ONE 2013, 8, e58725. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Naughton, D.P. Vitamin D in health and disease: Current perspectives. Nutr. J. 2010, 9, 65. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D.D. Vitamin D: Newly discovered actions require reconsideration of physiologic requirements. Trends Endocrinol. Metab. 2010, 21, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Tsoukas, C.D.; Provvedini, D.M.; Manolagas, S.C. 1,25-dihydroxyvitamin d 3: A novel immunoregulatory hormone. Science 1984, 224, 1438–1440. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M. Vitamin D and the intracrinology of innate immunity. Mol. Cell Endocrinol. 2010, 321, 103–111. [Google Scholar] [CrossRef]
- Lang, P.O.; Samaras, N.; Samaras, D.; Aspinall, R. How important is vitamin D in preventing infections? Osteoporos. Int. 2013, 24, 1537–1553. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, L.E.; Wood, A.M.; Qureshi, O.S.; Hou, T.Z.; Gardner, D.; Briggs, Z.; Kaur, S.; Raza, K.; Sansom, D.M. Availability of 25-hydroxyvitamin D3 to apcs controls the balance between regulatory and inflammatory t cell responses. J. Immunol. 2012, 189, 5155–5164. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, L.E.; Burke, F.; Mura, M.; Zheng, Y.; Qureshi, O.S.; Hewison, M.; Walker, L.S.; Lammas, D.A.; Raza, K.; Sansom, D.M. 1,25-dihydroxyvitamin D3 and il-2 combine to inhibit T cell production of inflammatory cytokines and promote development of regulatory T cells expressing ctla-4 and foxp3. J. Immunol. 2009, 183, 5458–5467. [Google Scholar] [CrossRef] [PubMed]
- Chun, R.F.; Lauridsen, A.L.; Suon, L.; Zella, L.A.; Pike, J.W.; Modlin, R.L.; Martineau, A.R.; Wilkinson, R.J.; Adams, J.; Hewison, M. Vitamin D-binding protein directs monocyte responses to 25-hydroxy- and 1,25-dihydroxyvitamin D. J. Clin. Endocrinol. Metab. 2010, 95, 3368–3376. [Google Scholar] [CrossRef] [PubMed]
- Grzybowski, A.; Pietrzak, K. Tadeusz reichstein (1897‒1996): A cofounder of modern steroid treatment in dermatology. Clin. Dermatol. 2012, 30, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Kearns, M.D.; Alvarez, J.A.; Seidel, N.; Tangpricha, V. Impact of vitamin D on infectious disease. Am. J. Med. Sci. 2015, 349, 245–262. [Google Scholar] [CrossRef] [PubMed]
- PubMed. Available online: http://www.ncbi.nlm.nih.gov/pubmed/ (accessed on 28 January 2015).
- Liu, P.T.; Stenger, S.; Li, H.; Wenzel, L.; Tan, B.H.; Krutzik, S.R.; Ochoa, M.T.; Schauber, J.; Wu, K.; Meinken, C.; et al. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science 2006, 311, 1770–1773. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.T.; Schenk, M.; Walker, V.P.; Dempsey, P.W.; Kanchanapoomi, M.; Wheelwright, M.; Vazirnia, A.; Zhang, X.; Steinmeyer, A.; Zugel, U.; et al. Convergence of il-1beta and vdr activation pathways in human tlr2/1-induced antimicrobial responses. PLoS ONE 2009, 4, e5810. [Google Scholar] [CrossRef] [PubMed]
- Fabri, M.; Stenger, S.; Shin, D.M.; Yuk, J.M.; Liu, P.T.; Realegeno, S.; Lee, H.M.; Krutzik, S.R.; Schenk, M.; Sieling, P.A.; et al. Vitamin D is required for ifn-gamma-mediated antimicrobial activity of human macrophages. Sci. Transl. Med. 2011, 3, 104ra102. [Google Scholar] [CrossRef] [PubMed]
- Klug-Micu, G.M.; Stenger, S.; Sommer, A.; Liu, P.T.; Krutzik, S.R.; Modlin, R.L.; Fabri, M. Cd40l and ifn-gamma induce an antimicrobial response against M. Tuberculosis in human monocytes. Immunology 2013, 139, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Yuk, J.M.; Shin, D.M.; Lee, H.M.; Yang, C.S.; Jin, H.S.; Kim, K.K.; Lee, Z.W.; Lee, S.H.; Kim, J.M.; Jo, E.K. Vitamin D3 induces autophagy in human monocytes/macrophages via cathelicidin. Cell Host. Microbe 2009, 6, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Sato, E.; Imafuku, S.; Ishii, K.; Itoh, R.; Chou, B.; Soejima, T.; Nakayama, J.; Hiromatsu, K. Vitamin D-dependent cathelicidin inhibits mycobacterium marinum infection in human monocytic cells. J. Dermatol. Sci. 2013, 70, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.T.; Wheelwright, M.; Teles, R.; Komisopoulou, E.; Edfeldt, K.; Ferguson, B.; Mehta, M.D.; Vazirnia, A.; Rea, T.H.; Sarno, E.N.; et al. Microrna-21 targets the vitamin D-dependent antimicrobial pathway in leprosy. Nat. Med. 2012, 18, 267–273. [Google Scholar] [PubMed]
- Edfeldt, K.; Liu, P.T.; Chun, R.; Fabri, M.; Schenk, M.; Wheelwright, M.; Keegan, C.; Krutzik, S.R.; Adams, J.S.; Hewison, M.; et al. T-cell cytokines differentially control human monocyte antimicrobial responses by regulating vitamin D metabolism. Proc. Natl. Acad. Sci. USA 2010, 107, 22593–22598. [Google Scholar] [PubMed]
- Reich, K.M.; Fedorak, R.N.; Madsen, K.; Kroeker, K.I. Vitamin D improves inflammatory bowel disease outcomes: Basic science and clinical review. World J. Gastroenterol. 2014, 20, 4934–4947. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.T.; Dabbas, B.; Laperriere, D.; Bitton, A.J.; Soualhine, H.; Tavera-Mendoza, L.E.; Dionne, S.; Servant, M.J.; Bitton, A.; Seidman, E.G.; et al. Direct and indirect induction by 1,25-dihydroxyvitamin D3 of the nod2/card15-defensin beta2 innate immune pathway defective in crohn disease. J. Biol. Chem. 2010, 285, 2227–2231. [Google Scholar] [CrossRef] [PubMed]
- Gombart, A.F.; Borregaard, N.; Koeffler, H.P. Human cathelicidin antimicrobial peptide (camp) gene is a direct target of the vitamin D receptor and is strongly up-regulated in myeloid cells by 1,25-dihydroxyvitamin d3. FASEB J. 2005, 19, 1067–1077. [Google Scholar] [CrossRef] [PubMed]
- White, J.H. Vitamin D metabolism and signaling in the immune system. Rev. Endocr. Metab. Disord. 2012, 13, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Yim, S.; Dhawan, P.; Ragunath, C.; Christakos, S.; Diamond, G. Induction of cathelicidin in normal and cf bronchial epithelial cells by 1,25-dihydroxyvitamin D(3). J. Cyst. Fibros. 2007, 6, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Peric, M.; Koglin, S.; Kim, S.M.; Morizane, S.; Besch, R.; Prinz, J.C.; Ruzicka, T.; Gallo, R.L.; Schauber, J. Il-17a enhances vitamin D3-induced expression of cathelicidin antimicrobial peptide in human keratinocytes. J. Immunol. 2008, 181, 8504–8512. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.Y.; Chen, Y.; Thai, P.; Wachi, S.; Huang, F.; Kim, C.; Harper, R.W.; Wu, R. Il-17 markedly up-regulates beta-defensin-2 expression in human airway epithelium via jak and nf-kappab signaling pathways. J. Immunol. 2004, 173, 3482–3491. [Google Scholar] [CrossRef] [PubMed]
- Schauber, J.; Dorschner, R.A.; Coda, A.B.; Buchau, A.S.; Liu, P.T.; Kiken, D.; Helfrich, Y.R.; Kang, S.; Elalieh, H.Z.; Steinmeyer, A.; et al. Injury enhances tlr2 function and antimicrobial peptide expression through a vitamin D-dependent mechanism. J. Clin. Invest. 2007, 117, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Campbell, G.R.; Spector, S.A. Vitamin D inhibits human immunodeficiency virus type 1 and mycobacterium tuberculosis infection in macrophages through the induction of autophagy. PLoS Pathog. 2012, 8, e1002689. [Google Scholar] [CrossRef] [PubMed]
- Campbell, G.R.; Spector, S.A. Toll-like receptor 8 ligands activate a vitamin D mediated autophagic response that inhibits human immunodeficiency virus type 1. PLoS Pathog. 2012, 8, e1003017. [Google Scholar] [CrossRef] [PubMed]
- Schauber, J.; Oda, Y.; Buchau, A.S.; Yun, Q.C.; Steinmeyer, A.; Zugel, U.; Bikle, D.D.; Gallo, R.L. Histone acetylation in keratinocytes enables control of the expression of cathelicidin and cd14 by 1,25-dihydroxyvitamin d3. J. Invest. Dermatol. 2008, 128, 816–824. [Google Scholar] [CrossRef] [PubMed]
- Muehleisen, B.; Bikle, D.D.; Aguilera, C.; Burton, D.W.; Sen, G.L.; Deftos, L.J.; Gallo, R.L. Pth/pthrp and vitamin D control antimicrobial peptide expression and susceptibility to bacterial skin infection. Sci. Transl. Med. 2012, 4, 135ra166. [Google Scholar] [CrossRef] [PubMed]
- Love, J.F.; Tran-Winkler, H.J.; Wessels, M.R. Vitamin D and the human antimicrobial peptide ll-37 enhance group a streptococcus resistance to killing by human cells. MBio 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Bacchetta, J.; Zaritsky, J.J.; Sea, J.L.; Chun, R.F.; Lisse, T.S.; Zavala, K.; Nayak, A.; Wesseling-Perry, K.; Westerman, M.; Hollis, B.W.; et al. Suppression of iron-regulatory hepcidin by vitamin D. J. Am. Soc. Nephrol. 2014, 25, 564–572. [Google Scholar] [CrossRef] [PubMed]
- Cherayil, B.J. The role of iron in the immune response to bacterial infection. Immunol. Res. 2011, 50, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Chun, R.F.; Liu, P.T.; Modlin, R.L.; Adams, J.S.; Hewison, M. Impact of vitamin D on immune function: Lessons learned from genome-wide analysis. Front. Physiol. 2014, 5, 151. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Leung, D.Y.; Richers, B.N.; Liu, Y.; Remigio, L.K.; Riches, D.W.; Goleva, E. Vitamin D inhibits monocyte/macrophage proinflammatory cytokine production by targeting mapk phosphatase-1. J. Immunol. 2012, 188, 2127–2135. [Google Scholar] [CrossRef]
- Khoo, A.L.; Chai, L.Y.; Koenen, H.J.; Oosting, M.; Steinmeyer, A.; Zuegel, U.; Joosten, I.; Netea, M.G.; van der Ven, A.J. Vitamin D(3) down-regulates proinflammatory cytokine response to mycobacterium tuberculosis through pattern recognition receptors while inducing protective cathelicidin production. Cytokine 2011, 55, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Khoo, A.L.; Chai, L.Y.; Koenen, H.J.; Kullberg, B.J.; Joosten, I.; van der Ven, A.J.; Netea, M.G. 1,25-dihydroxyvitamin D3 modulates cytokine production induced by candida albicans: Impact of seasonal variation of immune responses. J. Infect. Dis. 2011, 203, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M. Vitamin D and the immune system: New perspectives on an old theme. Endocrinol. Metab. Clin. North Am. 2010, 39, 365–379. [Google Scholar] [CrossRef] [PubMed]
- Avila, E.; Diaz, L.; Halhali, A.; Larrea, F. Regulation of 25-hydroxyvitamin D3 1alpha-hydroxylase, 1,25-dihydroxyvitamin D3 24-hydroxylase and vitamin d receptor gene expression by 8-bromo cyclic amp in cultured human syncytiotrophoblast cells. J. Steroid. Biochem. Mol. Biol. 2004, 89, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Verway, M.; Bouttier, M.; Wang, T.T.; Carrier, M.; Calderon, M.; An, B.S.; Devemy, E.; McIntosh, F.; Divangahi, M.; Behr, M.A.; et al. Vitamin D induces interleukin-1beta expression: Paracrine macrophage epithelial signaling controls m. Tuberculosis infection. PLoS Pathog. 2013, 9, e1003407. [Google Scholar]
- Larcombe, L.; Orr, P.; Turner-Brannen, E.; Slivinski, C.R.; Nickerson, P.W.; Mookherjee, N. Effect of vitamin D supplementation on mycobacterium tuberculosis-induced innate immune responses in a canadian dene first nations cohort. PLoS ONE 2012, 7, e40692. [Google Scholar] [CrossRef] [PubMed]
- Kleijwegt, F.S.; Laban, S.; Duinkerken, G.; Joosten, A.M.; Zaldumbide, A.; Nikolic, T.; Roep, B.O. Critical role for tnf in the induction of human antigen-specific regulatory T cells by tolerogenic dendritic cells. J. Immunol. 2010, 185, 1412–1418. [Google Scholar] [CrossRef] [PubMed]
- Unger, W.W.; Laban, S.; Kleijwegt, F.S.; van der Slik, A.R.; Roep, B.O. Induction of treg by monocyte-derived dc modulated by vitamin D3 or dexamethasone: Differential role for pd-l1. Eur. J. Immunol. 2009, 39, 3147–3159. [Google Scholar] [PubMed]
- Chamorro, S.; Garcia-Vallejo, J.J.; Unger, W.W.; Fernandes, R.J.; Bruijns, S.C.; Laban, S.; Roep, B.O.; t Hart, B.A.; van Kooyk, Y. Tlr triggering on tolerogenic dendritic cells results in tlr2 up-regulation and a reduced proinflammatory immune program. J. Immunol. 2009, 183, 2984–2994. [Google Scholar] [CrossRef] [PubMed]
- Wobke, T.K.; Sorg, B.L.; Steinhilber, D. Vitamin D in inflammatory diseases. Front. Physiol. 2014, 5, 244. [Google Scholar] [PubMed]
- Mahon, B.D.; Gordon, S.A.; Cruz, J.; Cosman, F.; Cantorna, M.T. Cytokine profile in patients with multiple sclerosis following vitamin D supplementation. J. Neuroimmunol. 2003, 134, 128–132. [Google Scholar] [CrossRef]
- Sigmundsdottir, H.; Pan, J.; Debes, G.F.; Alt, C.; Habtezion, A.; Soler, D.; Butcher, E.C. Dcs metabolize sunlight-induced vitamin D3 to “program” T cell attraction to the epidermal chemokine ccl27. Nat. Immunol. 2007, 8, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Penna, G.; Amuchastegui, S.; Giarratana, N.; Daniel, K.C.; Vulcano, M.; Sozzani, S.; Adorini, L. 1,25-dihydroxyvitamin D3 selectively modulates tolerogenic properties in myeloid but not plasmacytoid dendritic cells. J. Immunol. 2007, 178, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Brennan, A.; Katz, D.R.; Nunn, J.D.; Barker, S.; Hewison, M.; Fraher, L.J.; O'Riordan, J.L. Dendritic cells from human tissues express receptors for the immunoregulatory vitamin D3 metabolite, dihydroxycholecalciferol. Immunology 1987, 61, 457–461. [Google Scholar] [PubMed]
- Hewison, M.; Freeman, L.; Hughes, S.V.; Evans, K.N.; Bland, R.; Eliopoulos, A.G.; Kilby, M.D.; Moss, P.A.; Chakraverty, R. Differential regulation of vitamin D receptor and its ligand in human monocyte-derived dendritic cells. J. Immunol. 2003, 170, 5382–5390. [Google Scholar] [CrossRef] [PubMed]
- Fritsche, J.; Mondal, K.; Ehrnsperger, A.; Andreesen, R.; Kreutz, M. Regulation of 25-hydroxyvitamin D3-1 alpha-hydroxylase and production of 1 alpha,25-dihydroxyvitamin D3 by human dendritic cells. Blood 2003, 102, 3314–3316. [Google Scholar] [CrossRef] [PubMed]
- Kundu, R.; Chain, B.M.; Coussens, A.K.; Khoo, B.; Noursadeghi, M. Regulation of cyp27b1 and cyp24a1 hydroxylases limits cell-autonomous activation of vitamin D in dendritic cells. Eur. J. Immunol. 2014, 44, 1781–1790. [Google Scholar] [CrossRef] [PubMed]
- Penna, G.; Adorini, L. 1 alpha,25-dihydroxyvitamin D3 inhibits differentiation, maturation, activation, and survival of dendritic cells leading to impaired alloreactive t cell activation. J. Immunol. 2000, 164, 2405–2411. [Google Scholar] [CrossRef] [PubMed]
- Piemonti, L.; Monti, P.; Sironi, M.; Fraticelli, P.; Leone, B.E.; Dal, C.E.; Allavena, P.; Di, C.V. Vitamin D3 affects differentiation, maturation, and function of human monocyte-derived dendritic cells. J. Immunol. 2000, 164, 4443–4451. [Google Scholar] [CrossRef] [PubMed]
- Griffin, M.D.; Lutz, W.; Phan, V.A.; Bachman, L.A.; McKean, D.J.; Kumar, R. Dendritic cell modulation by 1alpha,25 dihydroxyvitamin D3 and its analogs: A vitamin d receptor-dependent pathway that promotes a persistent state of immaturity in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2001, 98, 6800–6805. [Google Scholar] [CrossRef] [PubMed]
- van Halteren, A.G.; van Etten, E.; de Jong, E.C.; Bouillon, R.; Roep, B.O.; Mathieu, C. Redirection of human autoreactive T-cells upon interaction with dendritic cells modulated by tx527, an analog of 1,25 dihydroxyvitamin D(3). Diabetes 2002, 51, 2119–2125. [Google Scholar]
- Berer, A.; Stockl, J.; Majdic, O.; Wagner, T.; Kollars, M.; Lechner, K.; Geissler, K.; Oehler, L. 1,25-dihydroxyvitamin D(3) inhibits dendritic cell differentiation and maturation in vitro. Exp. Hematol. 2000, 28, 575–583. [Google Scholar] [CrossRef]
- Pedersen, A.W.; Holmstrom, K.; Jensen, S.S.; Fuchs, D.; Rasmussen, S.; Kvistborg, P.; Claesson, M.H.; Zocca, M.B. Phenotypic and functional markers for 1alpha,25-dihydroxyvitamin D(3)-modified regulatory dendritic cells. Clin. Exp. Immunol. 2009, 157, 48–59. [Google Scholar] [PubMed]
- Veldman, C.M.; Cantorna, M.T.; DeLuca, H.F. Expression of 1,25-dihydroxyvitamin D(3) receptor in the immune system. Arch. Biochem. Biophys. 2000, 374, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Provvedini, D.M.; Tsoukas, C.D.; Deftos, L.J.; Manolagas, S.C. 1,25 dihydroxyvitamin D3 receptors in human leukocytes. Science 1983, 221, 1181–1183. [Google Scholar] [CrossRef] [PubMed]
- von Essen, M.R.; Kongsbak, M.; Schjerling, P.; Olgaard, K.; Odum, N.; Geisler, C. Vitamin D controls t cell antigen receptor signaling and activation of human t cells. Nat. Immunol. 2010, 11, 344–349. [Google Scholar]
- Kongsbak, M.; von Essen, M.R.; Levring, T.B.; Schjerling, P.; Woetmann, A.; Odum, N.; Bonefeld, C.M.; Geisler, C. Vitamin D-binding protein controls t cell responses to vitamin D. BMC Immunol. 2014, 15, 35. [Google Scholar] [CrossRef] [PubMed]
- Baeke, F.; Korf, H.; Overbergh, L.; van Etten, E.; Verstuyf, A.; Gysemans, C.; Mathieu, C. Human t lymphocytes are direct targets of 1,25-dihydroxyvitamin D3 in the immune system. J. Steroid Biochem. Mol. Biol. 2010, 121, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Correale, J.; Ysrraelit, M.C.; Gaitan, M.I. Immunomodulatory effects of vitamin d in multiple sclerosis. Brain 2009, 132, 1146–1160. [Google Scholar] [CrossRef] [PubMed]
- Bhalla, A.K.; Amento, E.P.; Serog, B.; Glimcher, L.H. 1,25-dihydroxyvitamin D 3 inhibits antigen-induced t cell activation. J. Immunol. 1984, 133, 1748–1754. [Google Scholar] [PubMed]
- Lemire, J.M.; Adams, J.S.; Kermani-Arab, V.; Bakke, A.C.; Sakai, R.; Jordan, S.C. 1,25-dihydroxyvitamin D 3 suppresses human t helper/inducer lymphocyte activity in vitro. J. Immunol. 1985, 134, 3032–3035. [Google Scholar] [PubMed]
- Kongsbak, M.; von Essen, M.R.; Boding, L.; Levring, T.B.; Schjerling, P.; Lauritsen, J.P.; Woetmann, A.; Odum, N.; Bonefeld, C.M.; Geisler, C. Vitamin d up-regulates the vitamin D receptor by protecting it from proteasomal degradation in human cd4+ t cells. PLoS ONE 2014, 9, e96695. [Google Scholar] [CrossRef] [PubMed]
- Rigby, W.F.C.; Stacy, T.; Fanger, M.W. Inhibition of t lymphocyte mitogenesis by 1,25-dihydroxyvitamin D 3 (calcitriol). J. Clin. Invest. 1984, 74, 1451–1455. [Google Scholar] [PubMed]
- Rigby, W.F.; Denome, S.; Fanger, M.W. Regulation of lymphokine production and human t lymphocyte activation by 1,25-dihydroxyvitamin D3. Specific inhibition at the level of messenger rna. J. Clin. Invest. 1987, 79, 1659–1664. [Google Scholar] [PubMed]
- Alroy, I.; Towers, T.L.; Freedman, L.P. Transcriptional repression of the interleukin-2 gene by vitamin d3: Direct inhibition of nfatp/ap-1 complex formation by a nuclear hormone receptor. Mol. Cell. Biol. 1995, 15, 5789–5799. [Google Scholar] [PubMed]
- Cippitelli, M.; Santoni, A. Vitamin D3: A transcriptional modulator of the interferon-gamma gene. Eur. J. Immunol. 1998, 28, 3017–3030. [Google Scholar] [PubMed]
- Rigby, W.F.; Waugh, M.; Graziano, R.F. Regulation of human monocyte HLA-DR and CD4 antigen expression, and antigen presentation by 1,25-dihydroxyvitamin D3. Blood 1990, 76, 189–197. [Google Scholar] [PubMed]
- Ikeda, U.; Wakita, D.; Ohkuri, T.; Chamoto, K.; Kitamura, H.; Iwakura, Y.; Nishimura, T. 1alpha,25-dihydroxyvitamin D3 and all-trans retinoic acid synergistically inhibit the differentiation and expansion of th17 cells. Immunol. Lett. 2010, 134, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Pantalena, L.C.; Liu, X.K.; Gaffen, S.L.; Liu, H.; Rohowsky-Kochan, C.; Ichiyama, K.; Yoshimura, A.; Steinman, L.; Christakos, S.; et al. 1,25-dihydroxyvitamin D(3) ameliorates th17 autoimmunity via transcriptional modulation of interleukin-17a. Mol. Cell Biol. 2011, 31, 3653–3669. [Google Scholar] [PubMed]
- Tang, J.; Zhou, R.; Luger, D.; Zhu, W.; Silver, P.B.; Grajewski, R.S.; Su, S.B.; Chan, C.C.; Adorini, L.; Caspi, R.R. Calcitriol suppresses antiretinal autoimmunity through inhibitory effects on the th17 effector response. J. Immunol. 2009, 182, 4624–4632. [Google Scholar] [CrossRef] [PubMed]
- Palmer, M.T.; Lee, Y.K.; Maynard, C.L.; Oliver, J.R.; Bikle, D.D.; Jetten, A.M.; Weaver, C.T. Lineage-specific effects of 1,25-dihydroxyvitamin D(3) on the development of effector cd4 T cells. J. Biol. Chem. 2011, 286, 997–1004. [Google Scholar] [PubMed]
- Boonstra, A.; Barrat, F.J.; Crain, C.; Heath, V.L.; Savelkoul, H.F.; O’Garra, A. 1alpha,25-dihydroxyvitamin D3 has a direct effect on naive cd4(+) T cells to enhance the development of th2 cells. J. Immunol. 2001, 167, 4974–4980. [Google Scholar] [PubMed]
- Barrat, F.J.; Cua, D.J.; Boonstra, A.; Richards, D.F.; Crain, C.; Savelkoul, H.F.; de Waal-Malefyt, R.; Coffman, R.L.; Hawrylowicz, C.M.; O’Garra, A. In vitro generation of interleukin 10-producing regulatory cd4(+) t cells is induced by immunosuppressive drugs and inhibited by T helper type 1 (th1)- and th2-inducing cytokines. J. Exp. Med. 2002, 195, 603–616. [Google Scholar] [CrossRef] [PubMed]
- Pludowski, P.; Holick, M.F.; Pilz, S.; Wagner, C.L.; Hollis, B.W.; Grant, W.B.; Shoenfeld, Y.; Lerchbaum, E.; Llewellyn, D.J.; Kienreich, K.; et al. Vitamin D effects on musculoskeletal health, immunity, autoimmunity, cardiovascular disease, cancer, fertility, pregnancy, dementia and mortality-a review of recent evidence. Autoimmun. Rev. 2013, 12, 976–989. [Google Scholar] [CrossRef] [PubMed]
- Baeke, F.; Korf, H.; Overbergh, L.; Verstuyf, A.; Thorrez, L.; Van Lommel, L.; Waer, M.; Schuit, F.; Gysemans, C.; Mathieu, C. The vitamin D analog, tx527, promotes a human cd4+cd25highcd127low regulatory t cell profile and induces a migratory signature specific for homing to sites of inflammation. J. Immunol. 2011, 186, 132–142. [Google Scholar] [CrossRef] [PubMed]
- Vyas, J.M.; Van der Veen, A.G.; Ploegh, H.L. The known unknowns of antigen processing and presentation. Nat. Rev. Immunol. 2008, 8, 607–618. [Google Scholar] [CrossRef] [PubMed]
- Fabri, M.; Realegeno, S.E.; Jo, E.K.; Modlin, R.L. Role of autophagy in the host response to microbial infection and potential for therapy. Curr. Opin. Immunol. 2011, 23, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Blanchet, F.P.; Piguet, V. Immunoamphisomes in dendritic cells amplify tlr signaling and enhance exogenous antigen presentation on mhc-ii. Autophagy 2010, 6, 816–818. [Google Scholar] [CrossRef] [PubMed]
- Jagannath, C.; Lindsey, D.R.; Dhandayuthapani, S.; Xu, Y.; Hunter, R.L., Jr.; Eissa, N.T. Autophagy enhances the efficacy of bcg vaccine by increasing peptide presentation in mouse dendritic cells. Nat. Med. 2009, 15, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Deretic, V.; Saitoh, T.; Akira, S. Autophagy in infection, inflammation and immunity. Nat. Rev. Immunol. 2013, 13, 722–737. [Google Scholar] [CrossRef] [PubMed]
- Cantorna, M.T.; Hullett, D.A.; Redaelli, C.; Brandt, C.R.; Humpal-Winter, J.; Sollinger, H.W.; Deluca, H.F. 1,25-dihydroxyvitamin D3 prolongs graft survival without compromising host resistance to infection or bone mineral density. Transplantation 1998, 66, 828–831. [Google Scholar] [PubMed]
- Hullett, D.A.; Cantorna, M.T.; Redaelli, C.; Humpal-Winter, J.; Hayes, C.E.; Sollinger, H.W.; Deluca, H.F. Prolongation of allograft survival by 1,25-dihydroxyvitamin D3. Transplantation 1998, 66, 824–828. [Google Scholar] [CrossRef] [PubMed]
- Cantorna, M.T. Mechanisms underlying the effect of vitamin D on the immune system. Proc. Nutr. Soc. 2010, 69, 286–289. [Google Scholar] [CrossRef] [PubMed]
- Cantorna, M.T.; Mahon, B.D. Mounting evidence for vitamin D as an environmental factor affecting autoimmune disease prevalence. Exp. Biol. Med. (Maywood.) 2004, 229, 1136–1142. [Google Scholar] [PubMed]
- Whitcomb, J.P.; Deagostino, M.; Ballentine, M.; Fu, J.; Tenniswood, M.; Welsh, J.; Cantorna, M.; McDowell, M.A. The role of vitamin D and vitamin D receptor in immunity to leishmania major infection. J. Parasitol. Res. 2012, 2012, 134645. [Google Scholar] [PubMed]
- Ehrchen, J.; Helming, L.; Varga, G.; Pasche, B.; Loser, K.; Gunzer, M.; Sunderkotter, C.; Sorg, C.; Roth, J.; Lengeling, A. Vitamin d receptor signaling contributes to susceptibility to infection with leishmania major. FASEB J. 2007, 21, 3208–3218. [Google Scholar] [CrossRef] [PubMed]
- Bruce, D.; Whitcomb, J.P.; August, A.; McDowell, M.A.; Cantorna, M.T. Elevated non-specific immunity and normal listeria clearance in young and old vitamin D receptor knockout mice. Int. Immunol. 2009, 21, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.F.; Zhang, Z.H.; Chang, Z.Q.; Tang, K.L.; Lin, D.Z.; Xu, J.Z. Vitamin D deficiency affects the immunity against mycobacterium tuberculosis infection in mice. Clin. Exp. Med. 2013, 13, 265–270. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Ananaba, G.A.; Patrickson, J.; Pitts, S.; Yi, Y.; Yan, F.; Eko, F.O.; Lyn, D.; Black, C.M.; Igietseme, J.U.; et al. Chlamydial infection in vitamin D receptor knockout mice is more intense and prolonged than in wild-type mice. J. Steroid. Biochem. Mol. Biol. 2013, 135, 7–14. [Google Scholar] [PubMed]
- Chen, J.; Waddell, A.; Lin, Y.D.; Cantorna, M.T. Dysbiosis caused by vitamin D receptor deficiency confers colonization resistance to citrobacter rodentium through modulation of innate lymphoid cells. Mucosal. Immunol. 2014, 8, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Yuzefpolskiy, Y.; Baumann, F.M.; Penny, L.A.; Studzinski, G.P.; Kalia, V.; Sarkar, S. Vitamin D receptor signals regulate effector and memory cd8 T cell responses to infections in mice. J. Nutr. 2014, 144, 2073–2082. [Google Scholar] [CrossRef] [PubMed]
- Bruce, D.; Ooi, J.H.; Yu, S.; Cantorna, M.T. Vitamin D and host resistance to infection? Putting the cart in front of the horse. Exp. Biol. Med. (Maywood) 2010, 235, 921–927. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.L.; Chen, K.; McAleer, J.; Kolls, J.K. Vitamin D regulation of ox40 ligand in immune responses to aspergillus fumigatus. Infect. Immun. 2013, 81, 1510–1519. [Google Scholar] [CrossRef] [PubMed]
- Enioutina, E.Y.; Bareyan, D.; Daynes, R.A. Tlr-induced local metabolism of vitamin D3 plays an important role in the diversification of adaptive immune responses. J. Immunol. 2009, 182, 4296–4305. [Google Scholar] [CrossRef] [PubMed]
- Enioutina, E.Y.; Bareyan, D.; Daynes, R.A. Vitamin D3-mediated alterations to myeloid dendritic cell trafficking in vivo expand the scope of their antigen presenting properties. Vaccine 2007, 25, 1236–1249. [Google Scholar] [CrossRef] [PubMed]
- Kaposzta, R.; Tree, P.; Marodi, L.; Gordon, S. Characteristics of invasive candidiasis in gamma interferon- and interleukin-4-deficient mice: Role of macrophages in host defense against candida albicans. Infect. Immun. 1998, 66, 1708–1717. [Google Scholar] [PubMed]
- Lucas, R.M.; Gorman, S.; Geldenhuys, S.; Hart, P.H. Vitamin d and immunity. F1000Prime Rep. 2014, 6, 118. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.J.; Zhou, Y.; Bu, F.; Travers-Gustafson, D.; Ye, A.; Xu, X.; Hamm, L.; Gorsage, D.M.; Fang, X.; Deng, H.W.; et al. Factors predicting vitamin D response variation in non-hispanic white postmenopausal women. J. Clin. Endocrinol. Metab. 2012, 97, 2699–2705. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.; Yu, K.; Stolzenberg-Solomon, R.; Simon, K.C.; McCullough, M.L.; Gallicchio, L.; Jacobs, E.J.; Ascherio, A.; Helzlsouer, K.; Jacobs, K.B.; et al. Genome-wide association study of circulating vitamin D levels. Hum. Mol. Genet. 2010, 19, 2739–2745. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.J.; Zhang, F.; Richards, J.B.; Kestenbaum, B.; van Meurs, J.B.; Berry, D.; Kiel, D.P.; Streeten, E.A.; Ohlsson, C.; Koller, D.L.; et al. Common genetic determinants of vitamin D insufficiency: A genome-wide association study. Lancet 2010, 376, 180–188. [Google Scholar] [CrossRef]
- Houghton, L.A.; Vieth, R. The case against ergocalciferol (vitamin D2) as a vitamin supplement. Am. J. Clin. Nutr. 2006, 84, 694–697. [Google Scholar] [PubMed]
- Tai, K.; Need, A.G.; Horowitz, M.; Chapman, I.M. Vitamin D, glucose, insulin, and insulin sensitivity. Nutrition 2008, 24, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Bendik, I.; Friedel, A.; Roos, F.F.; Weber, P.; Eggersdorfer, M. Vitamin D: A critical and essential micronutrient for human health. Front. Physiol. 2014, 5, 248. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D: Importance in the prevention of cancers, type 1 diabetes, heart disease, and osteoporosis. Am. J. Clin. Nutr. 2004, 79, 362–371. [Google Scholar] [PubMed]
- Clemens, T.L.; Adams, J.S.; Henderson, S.L.; Holick, M.F. Increased skin pigment reduces the capacity of skin to synthesise vitamin D3. Lancet 1982, 1, 74–76. [Google Scholar] [CrossRef]
- MacLaughlin, J.; Holick, M.F. Aging decreases the capacity of human skin to produce vitamin D3. J. Clin. Invest. 1985, 76, 1536–1538. [Google Scholar] [CrossRef] [PubMed]
- Vimaleswaran, K.S.; Cavadino, A.; Berry, D.J.; Whittaker, J.C.; Power, C.; Jarvelin, M.R.; Hypponen, E. Genetic association analysis of vitamin D pathway with obesity traits. Int. J. Obes. (Lond.) 2013, 37, 1399–1406. [Google Scholar] [CrossRef] [PubMed]
- Koutkia, P.; Lu, Z.; Chen, T.C.; Holick, M.F. Treatment of vitamin D deficiency due to crohn's disease with tanning bed ultraviolet B radiation. Gastroenterology 2001, 121, 1485–1488. [Google Scholar] [CrossRef] [PubMed]
- Yamshchikov, A.V.; Desai, N.S.; Blumberg, H.M.; Ziegler, T.R.; Tangpricha, V. Vitamin D for treatment and prevention of infectious diseases: A systematic review of randomized controlled trials. Endocr. Pract. 2009, 15, 438–449. [Google Scholar] [CrossRef] [PubMed]
- Fetahu, I.S.; Hobaus, J.; Kallay, E. Vitamin D and the epigenome. Front. Physiol. 2014, 5, 164. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, C. Genome-wide (over)view on the actions of vitamin D. Front. Physiol. 2014, 5, 167. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, C.; Seuter, S.; de Mello, V.D.; Schwab, U.; Voutilainen, S.; Pulkki, K.; Nurmi, T.; Virtanen, J.; Tuomainen, T.P.; Uusitupa, M. Primary vitamin D target genes allow a categorization of possible benefits of vitamin D(3) supplementation. PLoS ONE 2013, 8, e71042. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kroner, J.D.C.; Sommer, A.; Fabri, M. Vitamin D Every Day to Keep the Infection Away? Nutrients 2015, 7, 4170-4188. https://doi.org/10.3390/nu7064170
Kroner JDC, Sommer A, Fabri M. Vitamin D Every Day to Keep the Infection Away? Nutrients. 2015; 7(6):4170-4188. https://doi.org/10.3390/nu7064170
Chicago/Turabian StyleKroner, Juliana De Castro, Andrea Sommer, and Mario Fabri. 2015. "Vitamin D Every Day to Keep the Infection Away?" Nutrients 7, no. 6: 4170-4188. https://doi.org/10.3390/nu7064170
APA StyleKroner, J. D. C., Sommer, A., & Fabri, M. (2015). Vitamin D Every Day to Keep the Infection Away? Nutrients, 7(6), 4170-4188. https://doi.org/10.3390/nu7064170



