Reduced Anxiety in Forensic Inpatients after a Long-Term Intervention with Atlantic Salmon
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Sample
2.2. Measures
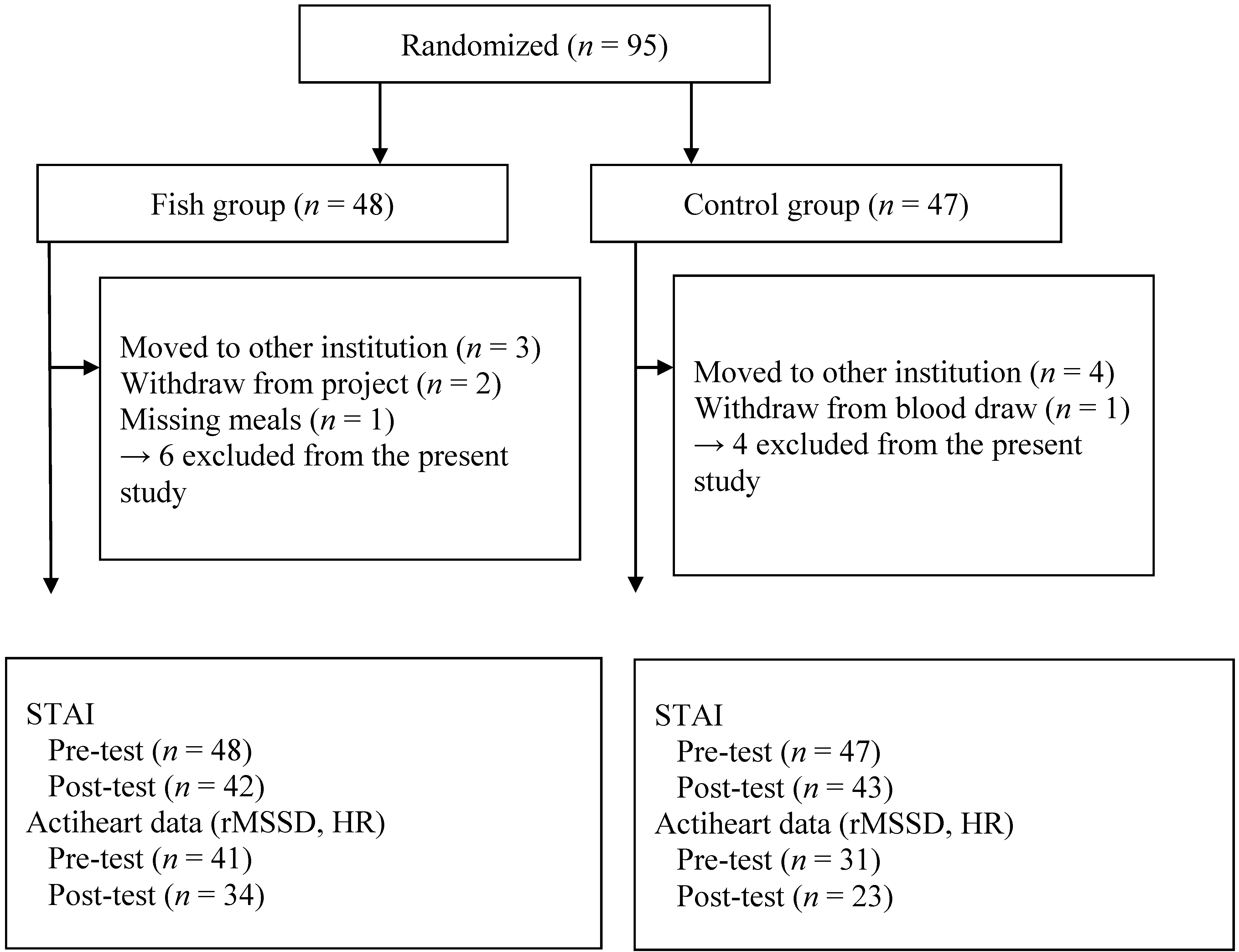
2.3. Ethics and Procedure
2.4. Statistical Analysis
3. Results
3.1. Descriptive Statistics
| Clinical Characteristics | Control Group (n = 43) | Fish Group (n = 42) | ||||
|---|---|---|---|---|---|---|
| Pre-test | Post-test | p | Pre-test | Post-test | p | |
| PD | 39% (n = 33) | 38% (n = 32) | ||||
| AD | 14% (n = 12) | 16% (n = 14) | ||||
| DD | 11% (n = 9) | 7% (n = 6) | ||||
| PD+AD/DD | 19% (n = 9) | 12% (n = 10) | ||||
| Age | 40.84 ± 9.14 | 42.76 ± 9.72 | 0.35 | |||
| IQ | 95.84 ± 13.02 | 95.79 ± 11.10 | 0.98 | |||
| PCL-R | 25.64 ± 5.58 | 25.30 ± 7.03 | 0.80 | |||
| BMI | 32 ± 13 1 | 29 ± 7 2 | 30 ± 5 1 | 31 ± 5 2 | 1 0.56 2 0.11 | |
| EPA | 11 ± 8 | 17 ± 16 2 | 0.02 | 10 ± 6 | 31 ± 12 2 | 0.001 2 0.001 |
| DHA | 79 ± 34 | 88 ± 47 2 | 1 | 73 ± 25 | 158 ± 50 2 | 0.001, 2 0.001 |
| Anxiety | ||||||
| State-Anxiety | 32.37 ± 10.24 | 32.42 ± 9.98 | 0.97 | 33.71 ± 9.70 | 29.79 ± 8.42 | 0.006 |
| Trait-Anxiety | 38.14 ± 10.10 | 36.63 ± 8.58 | 0.16 | 37.60 ± 8.23 | 36.93 ± 8.44 | 0.54 |
| Psychology | (n = 23) | (n = 34) | ||||
| rMSSD | 1.48 ± 0.31 | 1.39 ± 0.33 | 0.18 | 1.38 ± 0.26 | 1.57 ± 0.29 | 0.001 |
| HR | 73 ± 12 | 78 ± 11 | 0.03 | 76 ± 12 | 72 ± 12 | 0.01 |
3.2. Fatty Fish Consumption and Psychophysiology
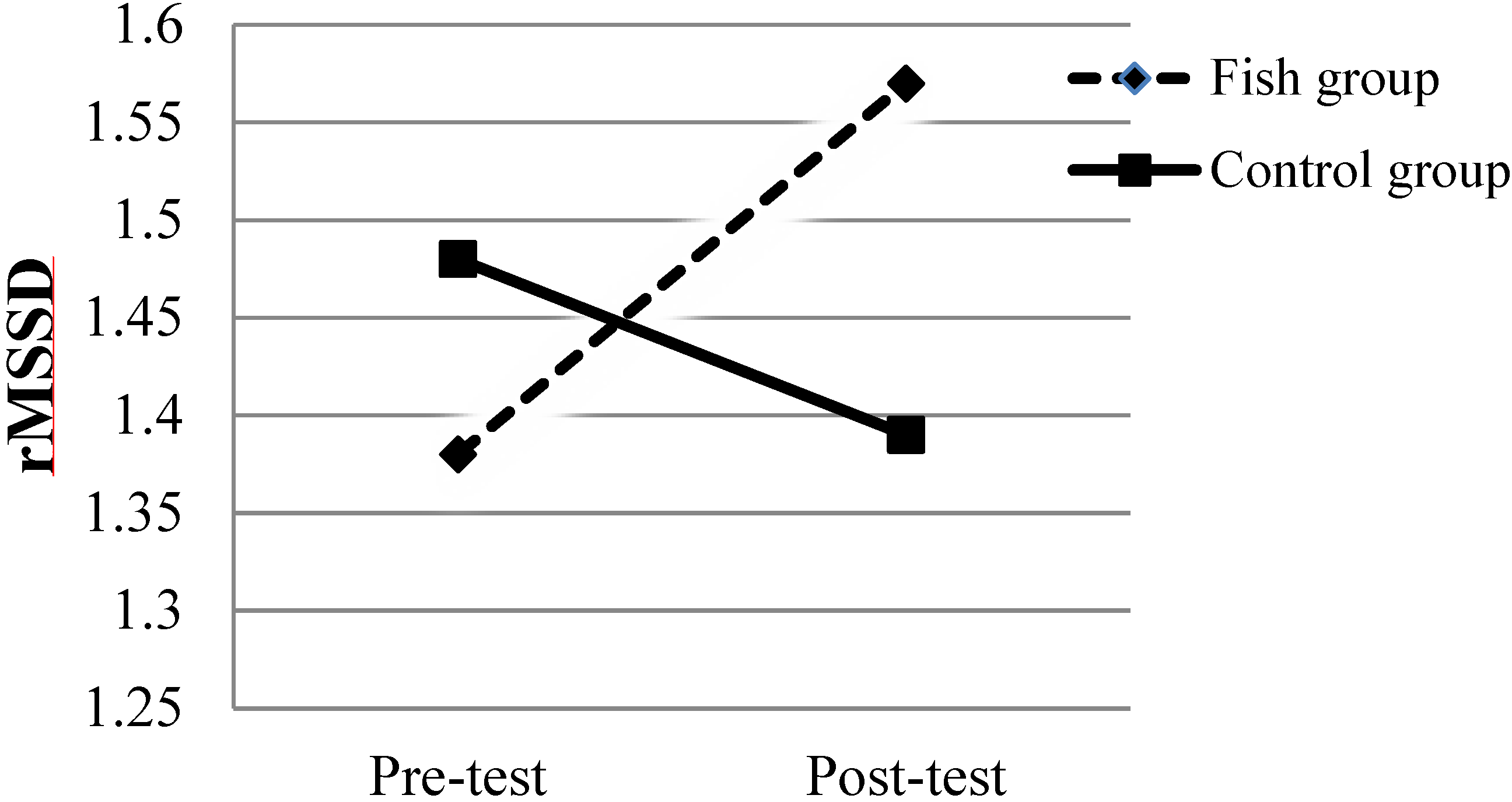
3.3. Fatty Fish Consumption and Anxiety

3.4. Correlations between HRV Parameters and Specific Nutrients
3.5. Correlations between Anxiety and Specific Nutrients
4. Discussion
5. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Van Horn, L.; McCoin, M.; Kris-Etherton, P.M.; Burke, F.; Carson, J.A.; Champagne, C.M.; Karmally, W.; Sikand, G. The evidence for dietary prevention and treatment of cardiovascular disease. J. Am. Diet. Assoc. 2008, 108, 287–331. [Google Scholar] [CrossRef] [PubMed]
- Cheatham, C.; Nerhammer, A.S.; Asserhøj, M.; Michaelsen, K.F.; Lauritzen, L. Fish oil supplementation during lactation: Effects on cognition and behaviour at 7 years of age. Lipids 2011, 46, 637–645. [Google Scholar] [CrossRef]
- Åberg, M.A.; Åberg, N.; Brisman, J.; Sundberg, R.; Winkvist, A.; Torén, K. Fish intake of Swedish male adolescents is a predictor of cognitive performance. Acta Paediatr. 2009, 98, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Kesse-Guyot, E.; Péneau, S.; Jeandel, C.; Hercberg, S.; Galan, P.; The SU.VI.MAX 2 research group. Thirteen-year prospective study between fish consumption, long-chain n-3 fatty acids intake and cognitive function. J. Nutr. Health Aging 2011, 15, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Timonen, M.; Horrobin, D.; Jokelainen, J.; Laitinen, A.H.; Räsänen, P. Fish consumption and depression: The Northern Finland 1966 birth cohort study. J. Affect. Disord. 2004, 82, 447–452. [Google Scholar] [PubMed]
- Appelton, K.; Woodside, J.V.; Yarnell, J.W.G.; Arveiler, D.; Haas, B.; Amouyel, P.; Montaye, M.; Ferrières, J.; Ruidavets, J.B.; Ducimetiere, P.; et al. Depressed mood and dietary fish intake: Direct relationship or indirect relationship as a result of diet and lifestyle. J. Affect. Disord. 2007, 104, 217–223. [Google Scholar] [CrossRef]
- Hakkarainen, R.; Partonen, T.; Haukka, J.; Virtamo, J.; Albanes, D.; Lönnquist, J. Is low dietary intake of omega 3 fatty acids associated with depression? Am. J. Psychiatry 2004, 161, 567–569. [Google Scholar] [CrossRef] [PubMed]
- Schiepers, O.J.G.; de Groot, R.H.M.; Jolles, J.; van Boxtel, M.P.J. Fish consumption, not fatty acid status, is related to quality of life in a healthy population. Prostaglandins Leukot. Essent Fatty Acids 2010, 83, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.; Frøyland, L.; Hemre, G.I.; Jacobsen, B.K.; Lund, E.; Meltzer, H.M.; Skåre, J.U. A Comprehensive Assessment of Fish and Other Seafood in the Norwegian Diet; Norwegian Scientific Committee for Food Safety: Oslo, Norway, 2007. [Google Scholar]
- Sanchez-Villegas, A.; Henríquez, P.; Figueiras, A.; Ortño, F.; Lahortiga, F.; Martínez- González, M.A. Long chain omega-3 fatty acids intake, fish consumption and mental disorders in the SUN cohort study. Eur. J. Nutr. 2007, 46, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Lansdowne, A.T.G.; Provost, S.C. Vitamin D3 enhances mood in healthy subjects during winter. Psychopharmacology 1998, 135, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Hansen, A.L.; Dahl, L.; Olson, G.; Thornton, D.; Graff, I.E.; Frøyland, L.; Thayer, J.F.; Pallesen, S. Fish consumption, sleep, daily functioning and heart rate variability. J. Clin. Sleep Med. 2014, 10, 567–575. [Google Scholar] [PubMed]
- Mozaffarian, D.; Stein, P.K.; Prineas, R.J.; Siscovick, D.S. Dietary fish ω-3 fatty acid consumption and heart rate variability in US adults. Circulation 2008, 117, 1130–1137. [Google Scholar] [CrossRef] [PubMed]
- Thayer, J.F.; Lane, R.D. A model of neurovisceral integration in emotion regulation and dysregulation. J. Affect. Disord. 2000, 61, 201–216. [Google Scholar] [CrossRef] [PubMed]
- Thayer, J.F.; Lane, R.D. The role of vagal function in the risk for cardiovascular disease and mortality. Biol. Psychol. 2007, 74, 224–242. [Google Scholar] [CrossRef] [PubMed]
- Thayer, J.F.; Friedman, B.H.; Borkovec, T.D. Autonomic characteristics of General anxiety disorder and worry. Biol. Psychiatry 1996, 39, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Hovland, A.; Pallesen, S.; Hammar, Å.; Hansen, A.L.; Thayer, J.F.; Sivertsen, B.; Tarvainen, M.; Nordhus, I.H. Subjective sleep quality in relation to inhibition and heart rate variability in patients with panic disorder. J. Affect. Disord. 2013, 150, 152–155. [Google Scholar] [CrossRef] [PubMed]
- Pittig, A.; Arch, J.J.; Lam, C.W.R.; Craske, M.G. Heart rate and heart rate variability in panic, social anxiety, obsessive-compulsive, and generalized anxiety disorders at baseline and in response to relaxation and hyperventilation. Int. J. Psychophysiol. 2013, 87, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, J.; Quintana, D.S.; Abbott, M.J.-A.; Kemp, A.H. Anxiety disorders are associated with reduced heart rate variability: A meta-analysis. Front. Psychiatry 2014, 5. [Google Scholar] [CrossRef]
- Porges, S.W. Autonomic regulation and attention. In Attention and Information Processing in Infants and Adults; Campbell, B.A., Hayne, H., Richardson, R., Eds.; Erlbaum: Hillside, NJ, USA, 1992; pp. 201–223. [Google Scholar]
- Thayer, J.F.; Åhs, F.; Fredrikson, M.; Sollers, J.J.; Wager, R.D. A meta-analysis of heart rate variability and neuroimagine studies: Implications for heart rate variability as a maker of stress and health. Neurosci. Biobehav. Rev. 2012, 36, 747–756. [Google Scholar] [CrossRef] [PubMed]
- Erkkilä, A.; Schwab, U.S.; De Mello, V.D.E.; Lappalainen, T.; Mussalo, H.; Lehto, S.; Kemi, V.; Lamberg-Allardt, C.; Uustupa, M.I.J. Effects of fatty and lean fish intake on blood pressure in subjects with coronary heart disease using multiple medications. Eur. J. Nutr. 2008, 47, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Hare, R.D. The Hare Psychopathy Checklist-Revised; Multi-Health Systems: Toronto, Canada, 1991. [Google Scholar]
- Brage, S.; Brage, N.; Franks, P.W.; Ekelund, U.; Wareham, N.J. Reliability and validity of the combined heart rate and movement sensor Actiheart. Eur. J. Clin. Nutr. 2005, 59, 561–570. [Google Scholar] [CrossRef]
- Spielberger, C.D.; Gorsuch, R.L.; Lushene, P.R.; Vagg, G.A. Manual for the State-Trait Anxiety Inventory; Consulting Psychologists Press: Palo Alto, CA, USA, 1983. [Google Scholar]
- Corruble, E.; Durrbach, A.; Charpentier, B.; Lang, P.; Amisi, S.; Dezamis, A.; Barry, C.; Falissard, B. Progressive increase of anxiety and depression in patients waiting for a kidney transplantation. Behav. Med. 2010, 36, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, M.; Birnbaum, L.; Denison, M.; De Vito, M.; Farland, W.; Feeley, M.; Fiedler, H.; Hakansson, H.; Hanberg, A.; Haws, L.; et al. The 2005 World Health Organization Reevaluation of human and mammalian toxic equivalency factors for dioxins and dioxin-like compounds. Toxicol. Sci. 2006, 93, 223–241. [Google Scholar] [CrossRef] [PubMed]
- Malik, M. Task force of the European society of cardiology and the North American society of pacing and electrophysiology. Eur. Heart J. 1996, 17, 354–381. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. A power primer. Psychol. Bull. 1992, 112, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Gominak, S.C.; Stumpf, W.E. The world epidemic of sleep disorders is linked to vitamin D deficiency. Med. Hypotheses 2012, 79, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Spielberger, C.D. The effects of anxiety on complex learning and academic achievement. In Anxiety and Behavior; Spielberger, C.D., Ed.; Academic Press: New York, NY, USA, 1966; pp. 361–398. [Google Scholar]
- Endler, N.S.; Kocovski, N.L. State and trait anxiety revisited. J. Anxiety Disord. 2001, 15, 231–245. [Google Scholar] [CrossRef] [PubMed]
- Wells, R.; Outhred, T.; Heathers, J.A.J.; Quintana, D.S.; Kemp, A.H. Matter over mind: A randomized controlled trial of Single biofeedback training on performance anxiety and heart rate variability in musicians. PLoS One 2012, 7, e46597. [Google Scholar] [CrossRef] [PubMed]
- Partonen, T. Vitamin D and serotonin in winter. Med. Hypotheses 1998, 51, 267–268. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Chen, T.C.; Lu, Z.; Sauter, E. Vitamin D and skin physiology: A D-lightful story. J. Bone Miner. Res. 2007, 22 (Suppl. 2), V28–V33. [Google Scholar] [CrossRef]
- Hansen, A.L.; Dahl, L.; Bakke, L.; Frøyland, L.; Thayer, J.F. Fish Consumption and Heart Rate Variability. Preliminary results. J. Psychophysiol. 2010, 24, 41–47. [Google Scholar] [CrossRef]
- Stumpf, W.E.; Privette, T.H. Light, vitamin D and psychiatry: Role of 1,25-dihydroxyvitamin D3 (soltriol) in etiology and therapy of seasonal affective disorder and other mental processes. Psychopharmacology 1989, 97, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Hibbeln, J.R.; Ferguson, T.A.; Blasbalg, T.L. Omega-3 fatty acid deficiencies in neurodevelopment, aggression, and autonomic dysregulation: Opportunities for intervention. Int. Rev. Psychiatry 2006, 18, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Kellett, D.O.; Ramage, A.G.; Jordan, D. Central 5-HT7 receptors are critical for reflex activation of cardiac vagal drive in anaesthetized rats. J. Physiol. 2005, 563, 319–331. [Google Scholar] [CrossRef] [PubMed]
- Seafood Data. Available online: http://nifes.no/en/seafood-data-2/ (accessed on 20 November 2014).
- Endler, N.S.; Cox, B.J.; Parker, J.D.A.; Bagby, R.M. Self-reports of depression and State-Trait Anxiety: Evidence for differential assessment. J. Pers. Soc. Psychol. 1992, 63, 832–838. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hansen, A.L.; Olson, G.; Dahl, L.; Thornton, D.; Grung, B.; Graff, I.E.; Frøyland, L.; Thayer, J.F. Reduced Anxiety in Forensic Inpatients after a Long-Term Intervention with Atlantic Salmon. Nutrients 2014, 6, 5405-5418. https://doi.org/10.3390/nu6125405
Hansen AL, Olson G, Dahl L, Thornton D, Grung B, Graff IE, Frøyland L, Thayer JF. Reduced Anxiety in Forensic Inpatients after a Long-Term Intervention with Atlantic Salmon. Nutrients. 2014; 6(12):5405-5418. https://doi.org/10.3390/nu6125405
Chicago/Turabian StyleHansen, Anita L., Gina Olson, Lisbeth Dahl, David Thornton, Bjørn Grung, Ingvild E. Graff, Livar Frøyland, and Julian F. Thayer. 2014. "Reduced Anxiety in Forensic Inpatients after a Long-Term Intervention with Atlantic Salmon" Nutrients 6, no. 12: 5405-5418. https://doi.org/10.3390/nu6125405
APA StyleHansen, A. L., Olson, G., Dahl, L., Thornton, D., Grung, B., Graff, I. E., Frøyland, L., & Thayer, J. F. (2014). Reduced Anxiety in Forensic Inpatients after a Long-Term Intervention with Atlantic Salmon. Nutrients, 6(12), 5405-5418. https://doi.org/10.3390/nu6125405




