Serum Ionized Calcium May Be Related to White Matter Lesion Volumes in Older Adults: A Pilot Study
Abstract
:1. Introduction
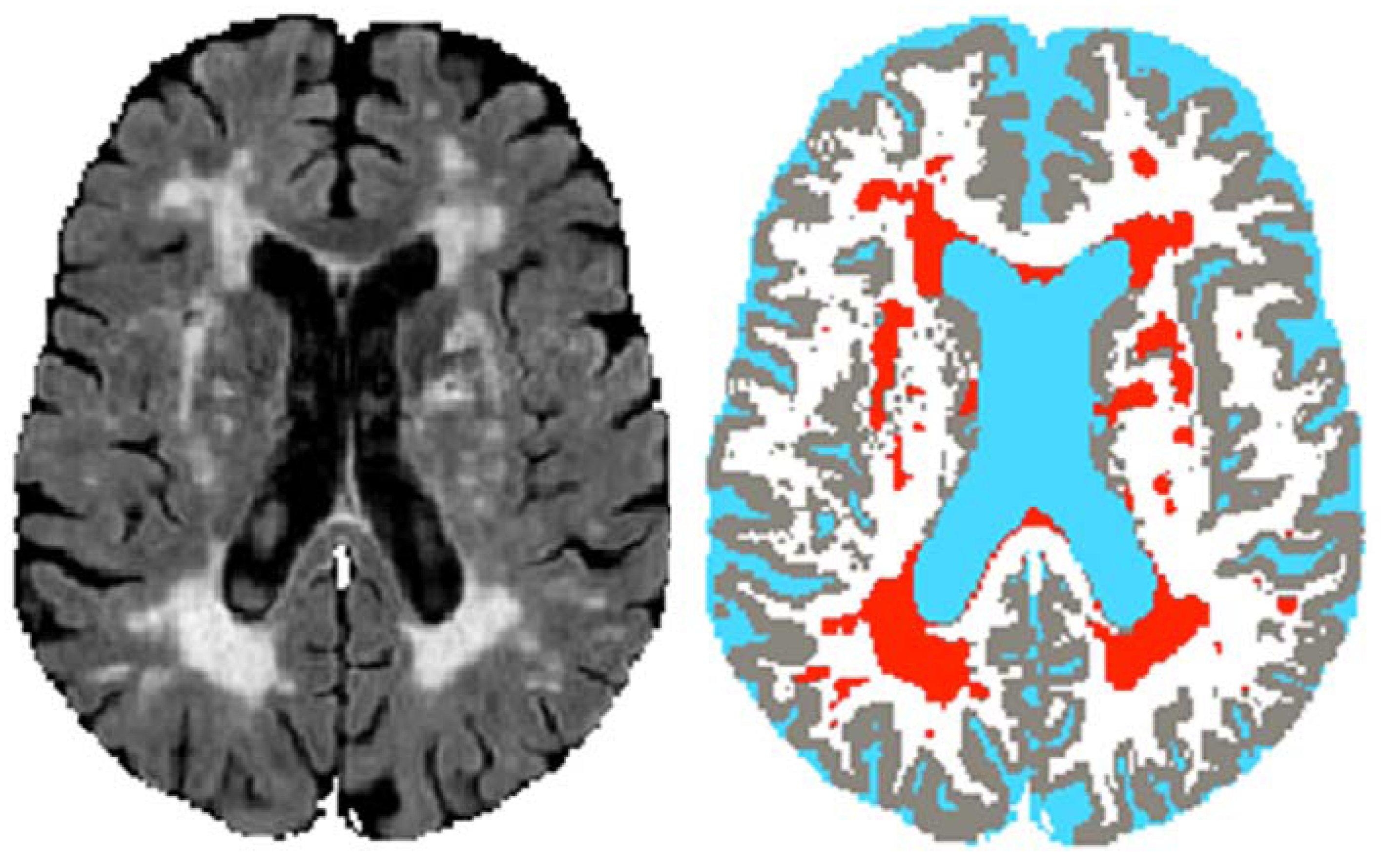
2. Methods
2.1. Sample
2.2. Treatment
2.3. Measures
2.3.1. Laboratory Assessment
2.3.2. White Matter Lesion (WML) Volumes
2.3.3. Covariates
2.4. Statistical Analyses
3. Results
3.1. Participant Characteristics
| Total (n = 42) | Depressed (n = 29) | Non-depressed (n = 13) | p value b | |
|---|---|---|---|---|
| Age in years | 68.4 (6.7) | 68.3 (6.5) | 68.8 (7.5) | 0.8 |
| Sex (female) | 19 (45%) | 15 (52%) | 4 (31%) | 0.2 |
| Race (White) | 39 (93%) | 26 (90%) | 13 (100%) | 0.1 |
| Education in years | 15.8 (1.6) | 15.8 (1.7) | 16.0 (1.5) | 0.7 |
| Hypertension (yes) c | 9 (26%) | 9 (38%) | 0 (0%) | 0.004 |
| Mini-Mental State Examination (total score) | 28.7 (1.3) | 28.8 (1.1) | 28.5 (1.7) | 0.5 |
| Serum ionized calcium (mg/dL) | 5.1 (0.2) | 5.1 (0.2) | 5.1 (0.2) | 0.5 |
| 25-hydroxy-vitamin D (ng/mL) | 34.5 (12.9) | 35.2 (14.7) | 32.9 (7.8) | 0.6 |
| Phosphorus (mg/dL) | 3.5 (0.5) | 3.5 (0.4) | 3.5 (0.6) | 0.7 |
| Parathyroid hormone (pg/mL) | 31.5 (14.2) | 31.8 (13.4) | 30.8 (17.7) | 0.9 |
| WML volume (mL) | 5.4 (3.6) | 5.4 (3.3) | 5.3 (4.2) | 0.9 |
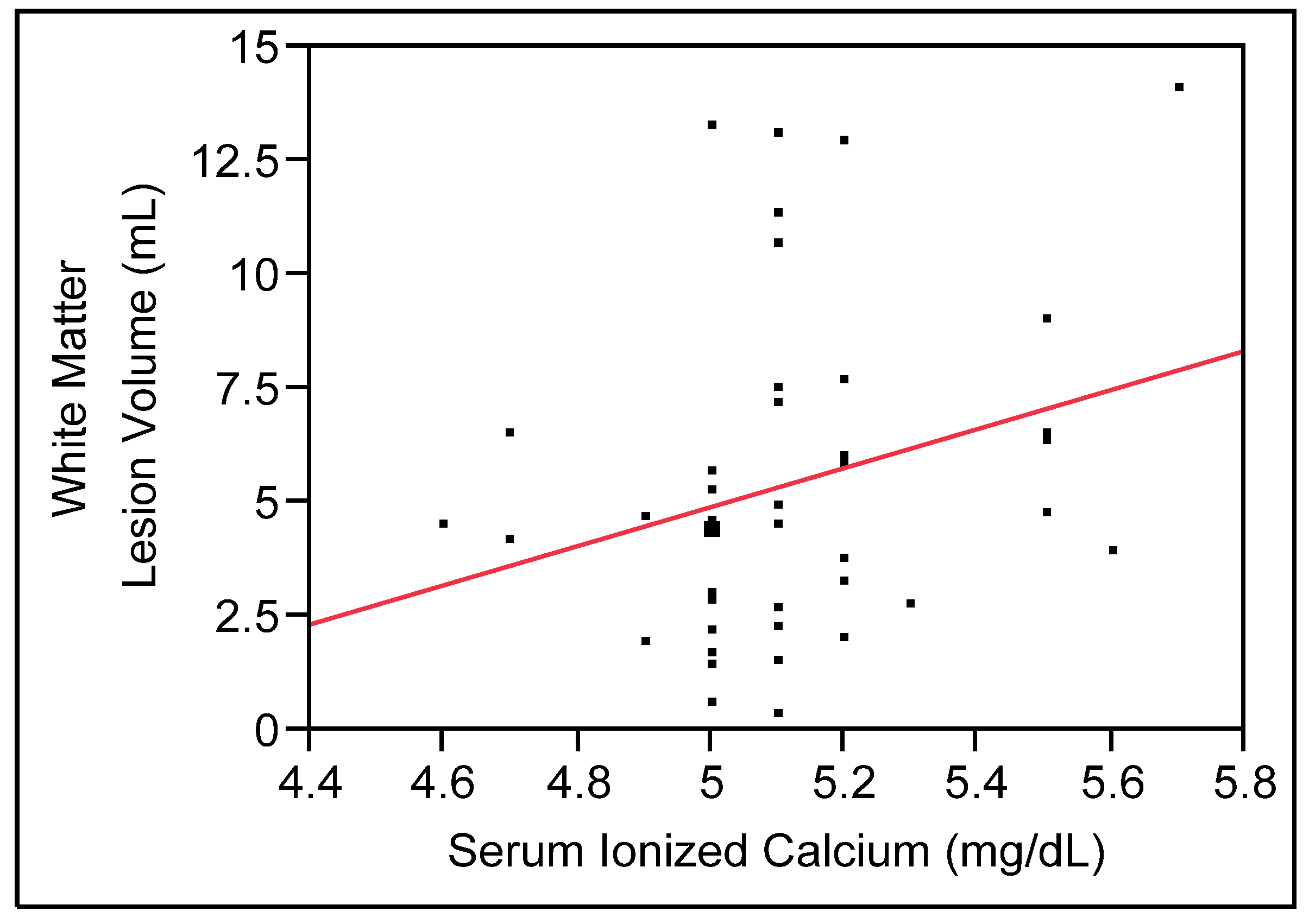
3.2. Serum Ionized Calcium (Serum Cai) and White Matter Lesion (WML) Volume
3.2.1. Main Model
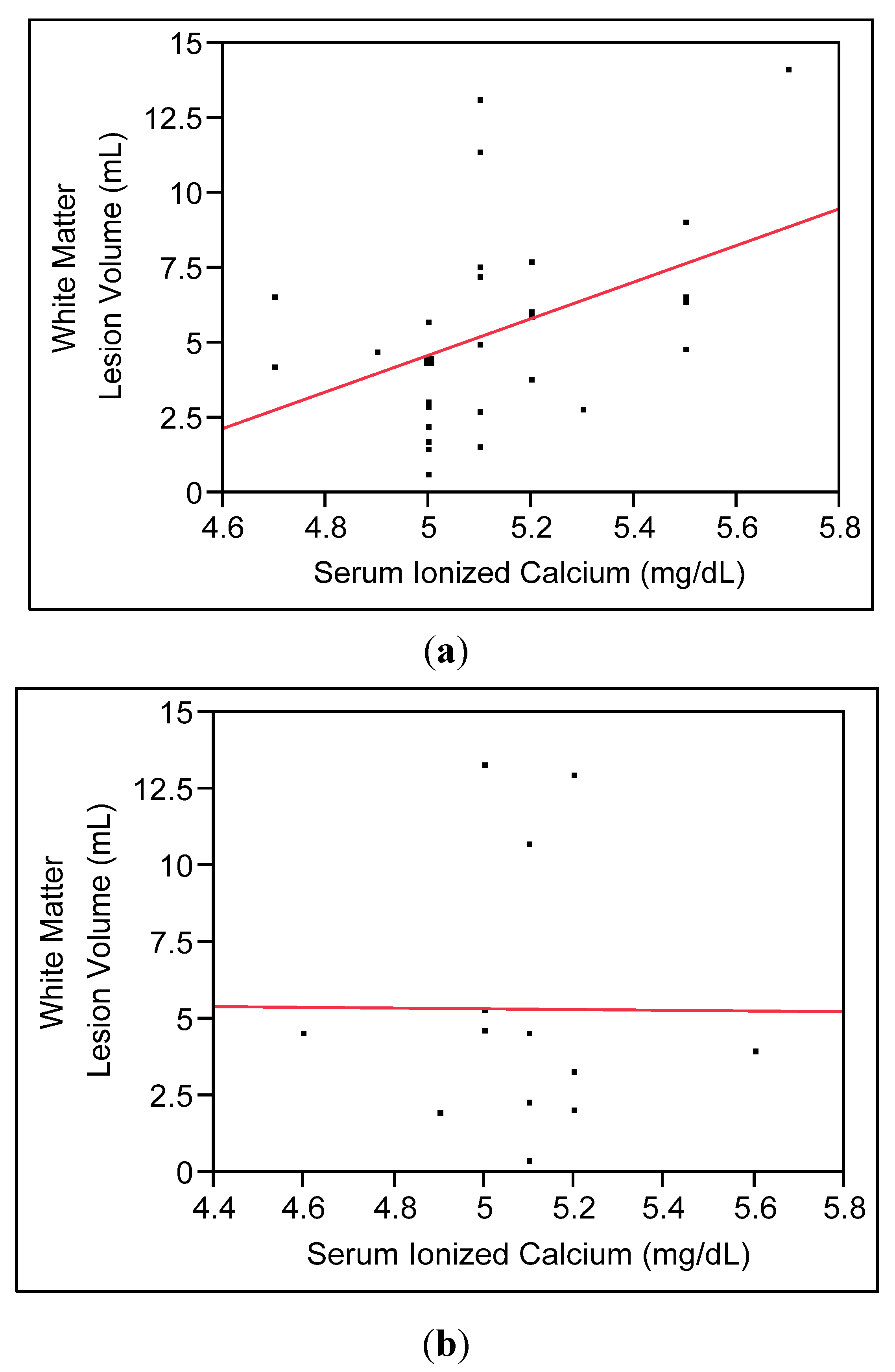
3.2.2. Secondary Analyses
4. Discussion
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Thomas, A.J.; O’Brien, J.T.; Davis, S.; Ballard, C.; Barber, R.; Kalaria, R.N.; Perry, R.H. Ischemic basis for deep white matter hyperintensities in major depression: A neuropathological study. Arch. Gen. Psychiatr. 2002, 59, 785–792. [Google Scholar]
- Godin, O.; Dufouil, C.; Maillard, P.; Delcroix, N.; Mazoyer, B.; Crivello, F.; Alperovitch, A.; Tzourio, C. White matter lesions as a predictor of depression in the elderly: The 3c-dijon study. Biol. Psychiatr. 2008, 63, 663–669. [Google Scholar]
- Steffens, D.C.; Krishnan, K.R.; Crump, C.; Burke, G.L. Cerebrovascular disease and evolution of depressive symptoms in the cardiovascular health study. Stroke 2002, 33, 1636–1644. [Google Scholar] [CrossRef]
- Roberts, R.E.; Kaplan, G.A.; Shema, S.J.; Strawbridge, W.J. Prevalence and correlates of depression in an aging cohort: The alameda county study. J. Gerontol. B Psychol. Sci. Soc. Sci. 1997, 52, S252–S258. [Google Scholar]
- Steffens, D.C.; Skoog, I.; Norton, M.C.; Hart, A.D.; Tschanz, J.T.; Plassman, B.L.; Wyse, B.W.; Welsh-Bohmer, K.A.; Breitner, J.C. Prevalence of depression and its treatment in an elderly population: The cache county study. Arch. Gen. Psychiatr. 2000, 57, 601–607. [Google Scholar]
- De Groot, J.C.; de Leeuw, F.E.; Oudkerk, M.; van Gijn, J.; Hofman, A.; Jolles, J.; Breteler, M.M. Periventricular cerebral white matter lesions predict rate of cognitive decline. Ann. Neurol. 2002, 52, 335–341. [Google Scholar]
- Van den Heuvel, D.M.; Ten Dam, V.H.; de Craen, A.J.; Admiraal-Behloul, F.; Olofsen, H.; Bollen, E.L.; Jolles, J.; Murray, H.M.; Blauw, G.J.; Westendorp, R.G.; et al. Increase in periventricular white matter hyperintensities parallels decline in mental processing speed in a non-demented elderly population. J. Neurol. Neurosurg. Psychiatr. 2006, 77, 149–153. [Google Scholar] [CrossRef]
- Steffens, D.C.; Potter, G.G.; McQuoid, D.R.; MacFall, J.R.; Payne, M.E.; Burke, J.R.; Plassman, B.L.; Welsh-Bohmer, K.A. Longitudinal magnetic resonance imaging vascular changes, apolipoprotein e genotype, and development of dementia in the neurocognitive outcomes of depression in the elderly study. Am. J. Geriatr. Psychiatr. 2007, 15, 839–849. [Google Scholar]
- Longstreth, W.T., Jr.; Dulberg, C.; Manolio, T.A.; Lewis, M.R.; Beauchamp, N.J., Jr.; O’Leary, D.; Carr, J.; Furberg, C.D. Incidence, manifestations, and predictors of brain infarcts defined by serial cranial magnetic resonance imaging in the elderly: The cardiovascular health study. Stroke 2002, 33, 2376–2382. [Google Scholar]
- Vermeer, S.E.; Hollander, M.; van Dijk, E.J.; Hofman, A.; Koudstaal, P.J.; Breteler, M.M. Silent brain infarcts and white matter lesions increase stroke risk in the general population: The rotterdam scan study. Stroke 2003, 34, 1126–1129. [Google Scholar] [CrossRef]
- Sachdev, P.S.; Wen, W.; Christensen, H.; Jorm, A.F. White matter hyperintensities are related to physical disability and poor motor function. J. Neurol. Neurosurg. Psychiatr. 2005, 76, 362–367. [Google Scholar]
- Corti, M.C.; Baggio, G.; Sartori, L.; Barbato, G.; Manzato, E.; Musacchio, E.; Ferrucci, L.; Cardinali, G.; Donato, D.; Launer, L.J.; et al. White matter lesions and the risk of incident hip fracture in older persons: Results from the progetto veneto anziani study. Arch. Intern. Med. 2007, 167, 1745–1751. [Google Scholar]
- Bokura, H.; Kobayashi, S.; Yamaguchi, S.; Iijima, K.; Nagai, A.; Toyoda, G.; Oguro, H.; Takahashi, K. Silent brain infarction and subcortical white matter lesions increase the risk of stroke and mortality: A prospective cohort study. J. Stroke Cerebrovasc. Dis. 2006, 15, 57–63. [Google Scholar] [CrossRef]
- Jorde, R.; Sundsfjord, J.; Fitzgerald, P.; Bonaa, K.H. Serum calcium and cardiovascular risk factors and diseases: The tromso study. Hypertension 1999, 34, 484–490. [Google Scholar] [CrossRef]
- Leifsson, B.G.; Ahren, B. Serum calcium and survival in a large health screening program. J. Clin. Endocrinol. Metab. 1996, 81, 2149–2153. [Google Scholar] [CrossRef] [Green Version]
- Lind, L.; Skarfors, E.; Berglund, L.; Lithell, H.; Ljunghall, S. Serum calcium: A new, independent, prospective risk factor for myocardial infarction in middle-aged men followed for 18 years. J. Clin. Epidemiol. 1997, 50, 967–973. [Google Scholar] [CrossRef]
- Slinin, Y.; Blackwell, T.; Ishani, A.; Cummings, S.R.; Ensrud, K.E. Serum calcium, phosphorus and cardiovascular events in post-menopausal women. Int. J. Cardiol. 2011, 149, 335–340. [Google Scholar]
- Schram, M.T.; Trompet, S.; Kamper, A.M.; de Craen, A.J.; Hofman, A.; Euser, S.M.; Breteler, M.M.; Westendorp, R.G. Serum calcium and cognitive function in old age. J. Am. Geriatr. Soc. 2007, 55, 1786–1792. [Google Scholar] [CrossRef]
- Bolland, M.J.; Wang, T.K.; van Pelt, N.C.; Horne, A.M.; Mason, B.H.; Ames, R.W.; Grey, A.B.; Ruygrok, P.N.; Gamble, G.D.; Reid, I.R. Abdominal aortic calcification on vertebral morphometry images predicts incident myocardial infarction. J. Bone Miner. Res. 2010, 25, 505–512. [Google Scholar]
- Ishizaka, N.; Ishizaka, Y.; Takahashi, E.; Tooda, E.; Hashimoto, H.; Nagai, R.; Yamakado, M. Serum calcium concentration and carotid artery plaque: A population-based study. J. Cardiol. 2002, 39, 151–157. [Google Scholar]
- Rubin, M.R.; Rundek, T.; McMahon, D.J.; Lee, H.S.; Sacco, R.L.; Silverberg, S.J. Carotid artery plaque thickness is associated with increased serum calcium levels: The northern manhattan study. Atherosclerosis 2007, 194, 426–432. [Google Scholar] [CrossRef]
- De Leeuw, F.E.; de Groot, J.C.; Oudkerk, M.; Witteman, J.C.; Hofman, A.; van Gijn, J.; Breteler, M.M. Aortic atherosclerosis at middle age predicts cerebral white matter lesions in the elderly. Stroke 2000, 31, 425–429. [Google Scholar] [CrossRef]
- Rosano, C.; Naydeck, B.; Kuller, L.H.; Longstreth, W.T., Jr.; Newman, A.B. Coronary artery calcium: Associations with brain magnetic resonance imaging abnormalities and cognitive status. J. Am. Geriatr. Soc. 2005, 53, 609–615. [Google Scholar] [CrossRef]
- Vidal, J.S.; Sigurdsson, S.; Jonsdottir, M.K.; Eiriksdottir, G.; Thorgeirsson, G.; Kjartansson, O.; Garcia, M.E.; van Buchem, M.A.; Harris, T.B.; Gudnason, V.; et al. Coronary artery calcium, brain function and structure: The ages-reykjavik study. Stroke 2010, 41, 891–897. [Google Scholar] [CrossRef]
- Green, J.H.; Booth, C.; Bunning, R. Postprandial metabolic responses to milk enriched with milk calcium are different from responses to milk enriched with calcium carbonate. Asia Pac. J. Clin. Nutr. 2003, 12, 109–119. [Google Scholar]
- Karp, H.J.; Ketola, M.E.; Lamberg-Allardt, C.J. Acute effects of calcium carbonate, calcium citrate and potassium citrate on markers of calcium and bone metabolism in young women. Br. J. Nutr. 2009, 102, 1341–1347. [Google Scholar] [CrossRef]
- Oshima, T.; Young, E.W. Systemic and cellular calcium metabolism and hypertension. Semin. Nephrol. 1995, 15, 496–503. [Google Scholar]
- Block, G.; Port, F.K. Calcium phosphate metabolism and cardiovascular disease in patients with chronic kidney disease. Semin. Dial. 2003, 16, 140–147. [Google Scholar] [CrossRef]
- World Health Organization (WHO), The Global Burden of Disease: 2004 Update; WHO: Geneva, Switzerland, 2004.
- Carman, J.S.; Wyatt, R.J. Alterations in Cerebrospinal Fluid and Serum Total Calcium with Changes in Psychiatric State. In Neuroregulators and Psychiatric Disorders; Usdin, E., Hamburg, D.A., Barchas, J., Eds.; Oxford University Press: New York, NY, USA, 1977; pp. 488–494. [Google Scholar]
- Jimerson, D.C.; Post, R.M.; Carman, J.S.; van Kammen, D.P.; Wood, J.H.; Goodwin, F.K.; Bunney, W.E., Jr. Csf calcium: Clinical correlates in affective illness and schizophrenia. Biol. Psychiatr. 1979, 14, 37–51. [Google Scholar]
- Konopka, L.M.; Cooper, R.; Crayton, J.W. Serotonin-induced increases in platelet cytosolic calcium concentration in depressed, schizophrenic, and substance abuse patients. Biol. Psychiatr. 1996, 39, 708–713. [Google Scholar] [CrossRef]
- Trulson, M.E.; Arasteh, K.; Ray, D.W. Effects of elevated calcium on learned helplessness and brain serotonin metabolism in rats. Pharmacol. Biochem. Behav. 1986, 24, 445–448. [Google Scholar] [CrossRef]
- Saade, S.; Balleine, B.W.; Minor, T.R. The l-type calcium channel blocker nimodipine mitigates “learned helplessness” in rats. Pharmacol. Biochem. Behav. 2003, 74, 269–278. [Google Scholar] [CrossRef]
- Taragano, F.E.; Allegri, R.; Vicario, A.; Bagnatti, P.; Lyketsos, C.G. A double blind, randomized clinical trial assessing the efficacy and safety of augmenting standard antidepressant therapy with nimodipine in the treatment of “vascular depression”. Int. J. Geriatr. Psychiatr. 2001, 16, 254–260. [Google Scholar] [CrossRef]
- Steffens, D.C.; Welsh-Bohmer, K.A.; Burke, J.R.; Plassman, B.L.; Beyer, J.L.; Gersing, K.R.; Potter, G.G. Methodology and preliminary results from the neurocognitive outcomes of depression in the elderly study. J. Geriatr. Psychiatr. Neurol. 2004, 17, 202–211. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Chang, C.C.; Yu, S.C.; McQuoid, D.R.; Messer, D.F.; Taylor, W.D.; Singh, K.; Boyd, B.D.; Krishnan, K.R.; MacFall, J.R.; Steffens, D.C.; et al. Reduction of dorsolateral prefrontal cortex gray matter in late-life depression. Psychiatr. Res. 2011, 193, 1–6. [Google Scholar] [CrossRef]
- Robins, L.N.; Helzer, J.E.; Croughan, J.; Ratcliff, K.S. National institute of mental health diagnostic interview schedule. Its history, characteristics, and validity. Arch. Gen. Psychiatr. 1981, 38, 381–389. [Google Scholar]
- Payne, M.E.; Anderson, J.J.; Steffens, D.C. Calcium and vitamin d intakes may be positively associated with brain lesions in depressed and nondepressed elders. Nutr. Res. 2008, 28, 285–292. [Google Scholar] [CrossRef]
- Bolland, M.J.; Barber, P.A.; Doughty, R.N.; Mason, B.; Horne, A.; Ames, R.; Gamble, G.D.; Grey, A.; Reid, I.R. Vascular events in healthy older women receiving calcium supplementation: Randomised controlled trial. BMJ 2008, 336, 262–266. [Google Scholar] [CrossRef]
- Pentti, K.; Tuppurainen, M.T.; Honkanen, R.; Sandini, L.; Kroger, H.; Alhava, E.; Saarikoski, S. Use of calcium supplements and the risk of coronary heart disease in 52–62-year-old women: The kuopio osteoporosis risk factor and prevention study. Maturitas 2009, 63, 73–78. [Google Scholar] [CrossRef]
- Bolland, M.J.; Avenell, A.; Baron, J.A.; Grey, A.; MacLennan, G.S.; Gamble, G.D.; Reid, I.R. Effect of calcium supplements on risk of myocardial infarction and cardiovascular events: Meta-analysis. BMJ 2010, 341, c3691. [Google Scholar]
- Li, K.; Kaaks, R.; Linseisen, J.; Rohrmann, S. Associations of dietary calcium intake and calcium supplementation with myocardial infarction and stroke risk and overall cardiovascular mortality in the heidelberg cohort of the european prospective investigation into cancer and nutrition study (epic-heidelberg). Heart 2012, 98, 920–925. [Google Scholar] [CrossRef]
- Wang, T.K.; Bolland, M.J.; van Pelt, N.C.; Horne, A.M.; Mason, B.H.; Ames, R.W.; Grey, A.B.; Ruygrok, P.N.; Gamble, G.D.; Reid, I.R. Relationships between vascular calcification, calcium metabolism, bone density, and fractures. J. Bone Miner. Res. 2010, 25, 2501–2509. [Google Scholar]
- Bots, M.L.; van Swieten, J.C.; Breteler, M.M.; de Jong, P.T.; van Gijn, J.; Hofman, A.; Grobbee, D.E. Cerebral white matter lesions and atherosclerosis in the rotterdam study. Lancet 1993, 341, 1232–1237. [Google Scholar] [CrossRef]
- Babiarz, L.S.; Yousem, D.M.; Wasserman, B.A.; Wu, C.; Bilker, W.; Beauchamp, N.J., Jr. Cavernous carotid artery calcification and white matter ischemia. Am. J. Neuroradiol. 2003, 24, 872–877. [Google Scholar]
- Chung, P.W.; Park, K.Y.; Moon, H.S.; Kim, Y.B.; Youn, Y.C.; Byun, J.S.; Kwon, O.S. Intracranial internal carotid artery calcification: A representative for cerebral artery calcification and association with white matter hyperintensities. Cerebrovasc. Dis. 2010, 30, 65–71. [Google Scholar] [CrossRef]
- Erbay, S.; Han, R.; Baccei, S.; Krakov, W.; Zou, K.H.; Bhadelia, R.; Polak, J. Intracranial carotid artery calcification on head ct and its association with ischemic changes on brain mri in patients presenting with stroke-like symptoms: Retrospective analysis. Neuroradiology 2007, 49, 27–33. [Google Scholar] [CrossRef]
- Fanning, N.F.; Walters, T.D.; Fox, A.J.; Symons, S.P. Association between calcification of the cervical carotid artery bifurcation and white matter ischemia. AJNR Am. J. Neuroradiol. 2006, 27, 378–383. [Google Scholar]
- Khatri, M.; Wright, C.B.; Nickolas, T.L.; Yoshita, M.; Paik, M.C.; Kranwinkel, G.; Sacco, R.L.; DeCarli, C. Chronic kidney disease is associated with white matter hyperintensity volume: The northern manhattan study (nomas). Stroke 2007, 38, 3121–3126. [Google Scholar] [CrossRef]
- Kim, B.J.; Lee, S.H.; Kim, C.K.; Ryu, W.S.; Kwon, H.M.; Choi, S.Y.; Yoon, B.W. Advanced coronary artery calcification and cerebral small vessel diseases in the healthy elderly. Circ. J. 2011, 75, 451–456. [Google Scholar] [CrossRef]
- Demuro, A.; Mina, E.; Kayed, R.; Milton, S.C.; Parker, I.; Glabe, C.G. Calcium dysregulation and membrane disruption as a ubiquitous neurotoxic mechanism of soluble amyloid oligomers. J. Biol. Chem. 2005, 280, 17294–17300. [Google Scholar]
- Foster, T.C.; Kumar, A. Calcium dysregulation in the aging brain. Neuroscientist 2002, 8, 297–301. [Google Scholar] [CrossRef]
- Joborn, C.; Hetta, J.; Niklasson, F.; Rastad, J.; Wide, L.; Agren, H.; Akerstrom, G.; Ljunghall, S. Cerebrospinal fluid calcium, parathyroid hormone, and monoamine and purine metabolites and the blood-brain barrier function in primary hyperparathyroidism. Psychoneuroendocrinology 1991, 16, 311–322. [Google Scholar] [CrossRef]
- Tiemeier, H.; van Dijck, W.; Hofman, A.; Witteman, J.C.; Stijnen, T.; Breteler, M.M. Relationship between atherosclerosis and late-life depression: The rotterdam study. Arch. Gen. Psychiatr. 2004, 61, 369–376. [Google Scholar]
- Chen, C.S.; Chen, C.C.; Kuo, Y.T.; Chiang, I.C.; Ko, C.H.; Lin, H.F. Carotid intima-media thickness in late-onset major depressive disorder. Int. J. Geriatr. Psychiatr. 2006, 21, 36–42. [Google Scholar] [CrossRef]
- Muneyyirci-Delale, O.; Nacharaju, V.L.; Dalloul, M.; Altura, B.M.; Altura, B.T. Serum ionized magnesium and calcium in women after menopause: Inverse relation of estrogen with ionized magnesium. Fertil. Steril. 1999, 71, 869–872. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Payne, M.E.; Pierce, C.W.; McQuoid, D.R.; Steffens, D.C.; Anderson, J.J.B. Serum Ionized Calcium May Be Related to White Matter Lesion Volumes in Older Adults: A Pilot Study. Nutrients 2013, 5, 2192-2205. https://doi.org/10.3390/nu5062192
Payne ME, Pierce CW, McQuoid DR, Steffens DC, Anderson JJB. Serum Ionized Calcium May Be Related to White Matter Lesion Volumes in Older Adults: A Pilot Study. Nutrients. 2013; 5(6):2192-2205. https://doi.org/10.3390/nu5062192
Chicago/Turabian StylePayne, Martha E., Cortnee W. Pierce, Douglas R. McQuoid, David C. Steffens, and John J. B. Anderson. 2013. "Serum Ionized Calcium May Be Related to White Matter Lesion Volumes in Older Adults: A Pilot Study" Nutrients 5, no. 6: 2192-2205. https://doi.org/10.3390/nu5062192
APA StylePayne, M. E., Pierce, C. W., McQuoid, D. R., Steffens, D. C., & Anderson, J. J. B. (2013). Serum Ionized Calcium May Be Related to White Matter Lesion Volumes in Older Adults: A Pilot Study. Nutrients, 5(6), 2192-2205. https://doi.org/10.3390/nu5062192




