Nutrition of the Critically Ill — A 21st-Century Perspective
Abstract
:1. Morbidity and Death Continue to Rise in Modern Medicine
2. Health Care-Associated Infections (HCAI) Do Not Receive Enough Attention
3. Artificial Nutrition—A Major Contributor to Sepsis
4. Colloid-Associated Morbidity Often Neglected
5. Crystalloids Often Enough after Surgical Procedures
6. Nutrition Made to Prevent Deterioration of Immune Functions
7. Dysbiosis-Associated Over-Reacting Neutrophils
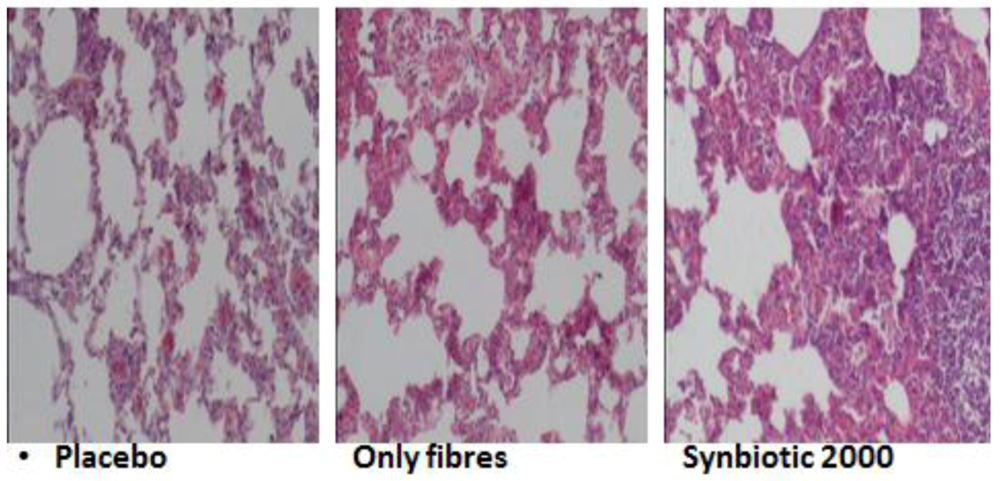
8. Efforts to Reduce Inflammation, Neutrophil Infiltration, and Tissue Destruction
| Treatment group | Average number of neutrophils per viewed fields |
|---|---|
| Synbiotic 2000 | 9.00 ± 0.44 |
| Only LAB | 8.40 ± 0.42 |
| Only the fibers | 31.20 ± 0.98 |
| Placebo | 51.10 ± 0.70 |
| Treatment group | U/g |
|---|---|
| Synbiotic 2000 | 25.62 ± 2.19 |
| Only LAB | 26.75 ± 2.61 |
| Only the fibers | 56.59 ± 1.73 |
| Placebo | 145.53 ± 7.53 |
| Treatment group | nmol/mg |
|---|---|
| Synbiotic 2000 | 0.22 ± 1.31 |
| Only LAB | 0.28 ± 3.55 |
| Only the fibers | 0.48 ± 5.32 |
| Placebo | 0.67 ± 2.94 |
| Treatment group | µmol/g |
|---|---|
| Synbiotic 2000 | 17.16 ± 2.03 |
| Only LAB | 8.91 ± 2.24 |
| Only the fibers | 47.71 ± 3.20 |
| Placebo | 66.22 ± 5.92 |
9. Life-Threatening Systemic Inflammation
10. Numerous Mechanisms to Control Intestinal Homeostasis
11. Personal Experience with Pro- and Synbiotics
11.1. Perioperative Prophylaxis in Elective Surgery
| Synbiotic 2000 | Fibers only | |
|---|---|---|
| Wound infections | 4 | 6 |
| Urinary infection | 1 | 1 |
| Peritonitis | 0 | 5 |
| Pneumonia | 0 | 4 |
| Sepsis | 0 | 2 |
| Cholangitis | 0 | 1 |
| Empyema | 0 | 1 |
| Total 5/40 (12.5%) | Total 16/40 (40%) |
| Isolated microorganisms | Synbiotic 2000 | Fibers only |
|---|---|---|
| Enterobacter cloacae | 2 | 8 |
| Enterococcus faecalis/faecium | 1 | 7 |
| Escherichia coli | 0 | 7 |
| Klebsiella pneumoniae | 2 | 2 |
| Proteus mirabilis | 1 | 1 |
| Staphylococcus aureus | 0 | 2 |
| Total 6 | Total 27 |
| Isolated microorganisms | Synbiotic 2000 | Fibers only |
|---|---|---|
| Pseudomonas aeruginosa | 17 | 24 |
| Staphylococcus aureus | 8 | 11 |
| Staphylococcus epidermidis | 1 | 1 |
| Staphylococcus faecalis | - | 1 |
| Enterobacter cloacae | 4 | - |
| Acinetobacter spp. | 2 | 3 |
| Staphylococcus haemolyticus | - | 1 |
| Serratia spp. | - | 2 |
| Klebsiella spp. | - | 1 |
| Proteus mirabilis | - | 2 |
| Candida albicans | 2 | 6 |
| Aspergillus spp. | - | - |
| Bacillus subtilis | - | 1 |
| Klebsiella spp. | - | 1 |
| Total 34 | Total 54 |
11.2. Perioperative Prophylaxis in Liver Transplantation
| Isolated bacteria | Synbiotic 2000 | Fibers only |
|---|---|---|
| Enterococcus faecalis | 1 | 11 |
| Escherichia coli | 0 | 3 |
| Enterobacter cloacae | 0 | 2 |
| Pseudomonas aeruginosa | 0 | 2 |
| Staphylococcus aureus | 0 | 1 |
| Total 1 | Total 18 |
11.3. Early Treatment in Major Trauma
| Total number of infections | Number of chest infections | |
|---|---|---|
| Alitraq Abbott-Ross (glut + arg) | 16/32 50% | 11/32 34% |
| Nova Source Novartis (+guargum) | 17/29 58% | 12/29 41% |
| Nutricomp peptide Braun (+peptide) | 13/26 50% | 11/26 42% |
| Nutricomp standard (+Synbiotic 2000) | 4/26 15% | 5/26 19% |
| Isolated microorganisms | Synbiotic 2000 | Fibers only |
|---|---|---|
| Acinetobacter baumanni | 21 | 35 |
| Candida albicans | 7 | 17 |
| Pseudomonas aeruginosa | 15 | 14 |
| Staphylococcus epidermidis | 2 | 10 |
| Staphylococcus aureus | 4 | 7 |
| Staphylococcus hominis | - | 2 |
| Enterobacter aerogenes | - | 2 |
| Staphylococcus haemolyticus | - | 1 |
| Serratia spp. | - | 2 |
| Klebsiella spp. | 5 | 12 |
| Proteus spp. | - | 1 |
| Total 54 | Total 103 |
11.4. Early Treatment in Severe Acute Pancreatitis
| Isolated microorganisms | Synbiotic 2000 | Fibers only |
|---|---|---|
| Pseudomonas aeruginosa | 1 | 4 |
| Enterococcus faecalis | 1 | 2 |
| Enterobacter spp. | 1 | 1 |
| Streptococcus spp. | 2 | - |
| Staphylococcusaureus | 1 | 1 |
| Enterococcus faecium | 1 | - |
| Candida spp. | - | 2 |
| Staphylococcus haemolyticus | - | 1 |
| Serratia spp. | - | 2 |
| Klebsiella spp. | - | 1 |
| Escherichia coli | - | 1 |
| Stenotrophomonas maltophilia | - | 1 |
| Citrobacter freundii | - | 1 |
| Total 7 | Total 17 |
11.5. Effects on “Mind Clarity”—Encephalopathy
11.6. Effects in HIV
12. It Is All about Inflammation
13. Special Focus on Fat Metabolism in Liver and Pancreas Disease
14. Inflammation Control—Pharma and/or Probiotics?
15. Single or Multiple Gene Targeting as Treatment?

16. Asian Foods Associated with Less Disease

17. Seven NOs and Three YESes!
- Restrict intake of insulinogenic, IGF1-rich or IGF1-stimulatory and Toll-stimulatory foods, such as refined carbohydrates: cereals, bread, sweets, cookies, rice, pasta, cooked tubers, including potatoes—these are foods which are absorbed high up in the small intestine and are of minimal benefit to microbiota.
- Restrict daily intake of fructose to below 25 grams a day.
- Restrict/eliminate intake of foods heated above 100 °C, known to be rich in the inflammation-inducing molecules AGEs and ALEs, and particularly foods heated above 130 °C, as foods with increased temperature become increasingly rich in pro-inflammatory and carcinogenic substances such as acrylamide and heterocyclic amines, e.g., fried and grilled foods, and toasted and high-temperature baked breads. See also [145,146,147].
- Restrict exposure to microbe-derived high inflammation-inducing endotoxins, especially found in meat hung for several days, hard cheeses, pork and ice creams.
- Restrict, eventually eliminate, intake of foods rich in proteotoxins such as casein, gluten and zein.
- Restrict intake of chemicals, including pharmaceutical drugs, to only what is absolutely necessary, as most chemicals are likely to be detrimental to microbiota.
- Increase dramatically the intake of fresh and raw greens, fresh spices and vegetables, rich in antioxidants, fibers, minerals and nutrients, but also inflammation-controlling factors such as curcumin, resveratrol and many others—see Figure 3.
- Increase/favor intake of ancient antioxidant-rich, high-fiber, low-calorie-containing grains such as buckwheat, amaranth, chia, lupin, millet, quinoa, sorghum, taro, teff, etc., and also the intake of beans, peas, chickpeas, lentils, nuts and almonds. See Table 12.
- Supplement large doses of vitamin D and omega fatty acids, both of special importance for control of inflammation and for function of microbiota. If ill—also supplement pro-/synbiotics, but only brands with documented clinical effects.
18. Nutrition of the Sick Made to Mimic Healthy Foods for the Healthy
19. Source of Protein Is Critical
20. Muscle Protein Synthesis Varies with Type of Protein Supplied
21. Plant-Based Proteins May Have Advantages
| Seed | Protein | Fat | Total starch | Dietary fiber |
|---|---|---|---|---|
| Amaranth | 16.5 | 5.7 | 61.5 | 20.6 |
| Quinoa | 14.5 | 5.2 | 64.2 | 14.2 |
| Buckwheat | 12.5 | 2.1 | 58.9 | 29.5 |
| Wheat | 12.0 | 2.5 | 63.0 | 17.4 |
22. Pseudocereals—A Promising Alternative Protein Source
23. Source of Fats for Clinical Nutrition
24. Omega-3-Based Emulsions Receive Increasing Interest
25. Olive Oil-Enriched Emulsions May Have Benefits
26. A Great Need for New Enteral Nutrition Formulas
27. Avocado: A Unique Fruit Rich in Healthy Oils, but Also Antioxidants
28. Red Palm Oil: Rich in Vitamin A, MUFAs and Unsaturated Fats
29. Red Palm Oil (and Coconut Oil): Effective against Inflammation
30. Medium-Chain Fatty Acids Have Anti-Infectious Effects and Do Not Produce Resistance!
31. Urgent Need for New Eco-Biological Formulas
| Nutrients | Beets 100 g | Beet greens 100 g Difference % | ||
|---|---|---|---|---|
| Calories | 43.00 | 22.00 | −51 | |
| Protein (g) | 1.61 | 2.20 | +35 | |
| Fat total (g) | 0.17 | 0.13 | −76 | |
| Carbohydrate (g) | 9.56 | 4.33 | −45 | |
| Fiber Total (g) | 2.80 | 3.70 | +32 | |
| Sugar Total (g) | 6.76 | 0.50 | −740 | |
| Calcium (mg) | 16.00 | 117.00 | +731 | |
| Iron (mg) | 0.80 | 2.57 | +321 | |
| Magnesium (mg) | 23.00 | 70.00 | +304 | |
| Potassium (mg) | 325.00 | 762.00 | +235 | |
| Sodium (mg) | 78.00 | 226.00 | +290 | |
| Copper (mg) | 0.08 | 0.19 | +238 | |
| Selenium (mg) | 0.70 | 0.90 | +29 | |
| Vitamin C (mg) | 4.90 | 30.00 | +500 | |
| Riboflavin (mg) | 0.04 | 0.22 | +612 | |
| Vitamin A (IU) | 33.00 | 6326.00 | +19,169 | |
| Vitamin E (mg) | 0.04 | 1.50 | +3750 | |
| Vitamin K (mcg) | 0.20 | 400.00 | +200,000 | |
32. Restoring Microbiota—Key to Success?
Conflict of Interest
References
- Kumar, G.; Kumar, N.; Taneja, A.; Kaleekal, T.; Tarima, S.; McGinley, E.; Jimenez, E.; Mohan, A.; Khan, R.A.; Whittle, J.; Jacobs, E.; Nanchal, R. Nationwide trends of severe sepsis in the twenty fi rst century (2000-2007). Chest 2011, 140, 1223–1231. [Google Scholar]
- Hall, M.J.; Williams, S.J.; DeFrances, C.J.; Golosinsky, A. Inpatient care for septicemia or sepsis: A challenge for patients and hospitals. Available online: http://www.cdc.gov/nchs/data/databriefs/db62.pdf (accessed on 16 November 2012).
- Vincent, J.L. Increasing awareness of sepsis: World Sepsis Day. Crit. Care 2012, 16, 152. [Google Scholar] [CrossRef]
- Guggenbichler, J.P.; Assadian, O.; Boeswald, M.; Kramer, A. Incidence and clinical implication of nosocomial infections associated with implantable biomaterials—catheters, ventilator-associated pneumonia, urinary tract infections. GMS Krankenhhyg. Interdiszip. 2011, 6, 1. [Google Scholar]
- Weiser, T.G.; Regenbogen, S.E.; Thompson, K.D.; Haynes, A.B.; Lipsitz, S.R.; Berry, W.R.; Gawande, A.A. An estimation of the global volume of surgery: A modelling strategy based on available data. Lancet 2008, 372, 139–144. [Google Scholar]
- Pearse, R.M.; Moreno, R.P.; Bauer, P.; Pelosi, P.; Metnitz, P.; Spies, C.; Vallet, B.; Vincent, J.L.; Hoeft, A.; Rhodes, A.; et al. Mortality after surgery in Europe: A 7 day cohort study. Lancet 2012, 380, 1059–1065. [Google Scholar]
- Khuri, S.F.; Henderson, W.G.; DePalma, R.G.; Mosca, C.; Healey, N.A.; Kumbhani, D.J. Determinants of long-term survival after major surgery and the adverse eff ect of postoperative complications. Ann. Surg. 2005, 242, 326–341. [Google Scholar]
- Pearse, R.M.; Harrison, D.A.; James, P.; Watson, D.; Hinds, C.; Rhodes, A.; Grounds, R.M.; Bennett, E.D. Identification and characterisation of the high-risk surgical population in the United Kingdom. Crit. Care 2006, 10, R81. [Google Scholar] [CrossRef]
- Jhanji, S.; Thomas, B.; Ely, A.; Watson, D.; Hinds, C.J.; Pearse, R.M. Mortality and utilisation of critical care resources amongst high-risk surgical patients in a large NHS trust. Anaesthesia 2008, 63, 695–700. [Google Scholar] [CrossRef]
- Findlay, G.; Goodwin, A.; Protopappa, K.; Smith, N.; Mason, M. Knowing the Risk: A Review of the Peri-Operative Care of Surgical Patients; National Confidential Enquiry into Patient Outcome and Death: London, UK, 2011. [Google Scholar]
- Head, J.; Ferrie, J.E.; Alexanderson, K.; Westerlund, H.; Vahtera, J.; Kivimaki, M. Diagnosis-specific sickness absence as a predictor of mortality: the Whitehall II prospective cohort study. BMJ 2008, 337, a1469. [Google Scholar]
- Jencks, S.F.; Williams, M.V.; Coleman, E.A. Rehospitalizations among patients in the Medicare fee-for-service program. N. Engl. J. Med. 2009, 360, 1418–1428. [Google Scholar] [CrossRef]
- Derogar, M.; Orsini, N.; Sadr-Azodi, O.; Lagergren, P. Influence of major postoperative complications on health-related quality of life among long-term survivors of esophageal cancer surgery. J. Clin. Oncol. 2012, 30, 1615–1619. [Google Scholar] [CrossRef]
- Dombrovskiy, V.Y.; Martin, A.A.; Sunderram, J.; Paz, H.L. Facing the challenge: Decreasing case fatality rates in severe sepsis despite increasing hospitalizations. Crit. Care Med. 2005, 33, 2555–2562. [Google Scholar] [CrossRef]
- Dombrovskiy, V.Y.; Martin, A.A.; Sunderram, J.; Paz, H.L. Rapid increase in hospitalization and mortality rates for severe sepsis in the United States: A trend analysis from 1993 to 2003. Crit. Care Med. 2007, 35, 1244–1250. [Google Scholar] [CrossRef]
- Vogel, T.R.; Dombrovskiy, V.Y.; Carson, J.L.; Graham, A.M.; Lowry, S.F. Postoperative sepsis in the United States. Ann. Surg. 2010, 252, 1065–1071. [Google Scholar] [CrossRef]
- Arias, E.; Smith, B.L. Deaths: Preliminary data for 2001. Natl. Vital Stat. Rep. 2003, 51, 1–44. [Google Scholar] [Green Version]
- Angus, D.C.; Linde-Zwirble, W.T.; Lidicker, J.; Clermont, G.; Carcillo, J.; Pinsky, M.R. Epidemiology of severe sepsis in the United States: Analysis of incidence, outcome and associated costs of care. Crit. Care Med. 2001, 29, 1303–1310. [Google Scholar] [CrossRef]
- Elias, A.C.; Matsuo, T.; Grion, C.M.; Cardoso, L.T.; Verri, P.H. Incidence and risk factors forsepsisin surgical patients: A cohort study. J. Crit. Care 2012, 27, 159–166. [Google Scholar]
- Beghetto, M.G.; Victorino, J.; Teixeira, L.; de Azevedo, M.J. Parenteral nutrition as a risk factor or central venous catheter-related infection. JPEN J. Parenter. Enteral Nutr. 2005, 29, 367–373. [Google Scholar] [CrossRef]
- Wren, S.M.; Ahmed, N.; Jamal, A.; Safadi, B.Y. Preoperative oral antibiotics in colorectal surgery increase the rate of Clostridium difficile colitis. Arch. Surg. 2005, 140, 752–756. [Google Scholar] [CrossRef]
- Bucher, P.; Gervaz, P.; Soravia, C.; Mermillod, B.; Erne, M.; Morel, P. Randomized clinical trial of mechanical bowel preparation versus no preparation before elective left-sided colorectal surgery. Br. J. Surg. 2005, 92, 409–414. [Google Scholar] [CrossRef]
- Bucher, P.; Gervaz, P.; Egger, J.F.; Soravia, C.; Morel, P. Morphologic alterations associated with mechanical bowel preparation before elective colorectal surgery: A randomized trial. Dis. Colon Rectum 2006, 49, 109–112. [Google Scholar]
- Wunsch, H.; Linde-Zwirble, W.T.; Angus, D.C.; Hartman, M.E.; Milbrandt, E.B.; Kahn, J.M. The epidemiology of mechanical ventilation use in the United States. Crit. Care Med. 2010, 38, 1947–1953. [Google Scholar]
- Haskel, Y.; Xu, D.; Lu, Q.; Deitch, E. Elemental diet-induced bacterial translocation can be hormonally modulated. Ann. Surg. 1993, 217, 634–642. [Google Scholar] [CrossRef]
- Deitch, E.; Xu, D.; Naruhn, M.B.; Deitch, D.C.; Lu, Q.; Mario, A.A. Elemental diet and IV-TPN-induced bacterial translocation is associated with loss of intestinal mucosal barrier function against bacteria. Ann. Surg. 1995, 221, 299–307. [Google Scholar] [CrossRef]
- Herndon, D.N.; Barrow, R.E.; Stein, M.; Linares, H.; Rutan, T.C.; Rutan, R.; Abston, S. Increased mortality with intravenous supplemental feeding in severely burned patients. J. Burn Care. Rehabil. 1989, 10, 309–313. [Google Scholar] [CrossRef]
- Lange, H. Multiorgan dysfunction syndrome: how water might contribute to its progression. J. Cell. Mol. Med. 2002, 6, 653–660. [Google Scholar] [CrossRef]
- Lobo, D.N.; Bostock, K.A.; Neal, K.R.; Perkins, A.C.; Rowlands, B.J.; Allison, S.P. Effect of salt and water balance on recovery of gastrointestinal function after elective colonic resection: a randomised controlled trial. Lancet 2002, 359, 1812–1818. [Google Scholar] [CrossRef]
- Macafee, D.A.L.; Allison, S.P.; Lobo, D.N. Some interactions between gastrointestinal function and fluid and electrolyte homeostasis. Curr. Opin. Clin. Nutr. Metab. Care 2005, 8, 197–203. [Google Scholar] [CrossRef]
- Wan, J.M.F.; Teo, T.C.; Babayan, V.K.; Blackburn, G.L. Invited comment: Lipids and the development of immune dysfunction and infection. JPEN J. Parenter. Enteral Nutr. 1988, 12, 43–52. [Google Scholar] [CrossRef]
- Van der Poll, T.; Coyle, S.M.; Levi, M.; Boermeester, M.A.; Braxton, C.C.; Jansen, P.; Hack, C.E.; Lowry, S.F. Fat emulsion infusion potentiates coagulation activation during human endotoxemia. Thrombosis Heamostasis 1996, 75, 83–86. [Google Scholar]
- Lin, B.F.; Huang, C.C.; Chiang, B.L.; Jeng, S.J. Dietary fat influences Ia antigen expression, cytokines and prostaglandin E2 production in immune cells in autoimmune-prone NZBxNZW F1 mice. Br. J. Nutr. 1996, 75, 711–722. [Google Scholar] [CrossRef]
- Umpierrez, G.E.; Isaacs, S.D.; Bazargan, N.; You, X.; Thaler, L.M.; Kitabchi, A.E. Hyperglycemia: An independent marker of in-hospital mortality in patients with undiagnosed diabetes. J. Clin. Endocrinol. Metab. 2002, 87, 978–962. [Google Scholar] [CrossRef]
- Mesotten, D.; Van den Berghe, G. Clinical potential of insulin therapy in critically ill patients. Drugs 2003, 63, 625–636. [Google Scholar] [CrossRef]
- Mesotten, D.; van den Berghe, G. Glycemic targets and approaches to management of the patient with critical illness. Curr. Diab. Rep. 2012, 12, 101–107. [Google Scholar] [CrossRef]
- Bulger, E.M. 7.5% saline and 7.5% saline/6% dextran for hypovolemic shock. J. Trauma 2011, 70, S27–S29. [Google Scholar] [CrossRef]
- Reinhart, K.; Perner, A.; Sprung, C.L.; Jaeschke, R.; Schortgen, F.; Johan Groeneveld, A.B.; Beale, R.; Hartog, C.S. European Society of Intensive Care Medicine. Consensus statement of the ESICM task force on colloid volume therapy in critically ill patients. Intensive Care Med. 2012, 38, 368–383. [Google Scholar]
- Elmér, O.; Göransson, G.; Saku, M.; Bengmark, S. Influence of physiological saline, dextran 70, hydroxyethyl starch, degraded gelatin, and fat emulsion solutions on screen filtration pressure. Eur. Surg. Res. 1977, 9, 85–95. [Google Scholar]
- Zoucas, E.; Göransson, G.; Bengmark, S. Colloid-induced changes in bleeding following liver resection in the rat. Res. Exp. Med. (Berl.) 1984, 184, 251–258. [Google Scholar] [CrossRef]
- Andersson, R.; Alwmark, A.; Bengmark, S. Influence of dextran on pneumococcal septicemia in splenic artery-ligated or splenectomized rats. Res. Exp. Med. (Berl.) 1987, 187, 423–427. [Google Scholar] [CrossRef]
- Andersson, R.; Tranberg, K.G.; Alwmark, A.; Bengmark, S. Factors influencing the outcome of E. coli peritonitis in rats. Acta Chir. Scand. 1989, 155, 155–157. [Google Scholar]
- Perner, A.; Haase, N.; Guttormsen, A.B.; Tenhunen, J.; Klemenzson, G.; Åneman, A.; Madsen, K.R.; Møller, M.H.; Elkjær, J.M.; Poulsen, L.M.; et al. Hydroxyethyl Starch 130/0.42 versus Hydroxyethyl Starch 130/0.42 versus Ringer’s Acetate in Severe Sepsis. N. Engl. J. Med. 2012, 367, 124–134. [Google Scholar] [CrossRef]
- Sandström, R.; Drott, C.; Hyltander, A.; Arfvidsson, B.; Schersten, T.; Wickström, I.; Lundholm, K. The effect of postoperative intravenous feeding (TPN) on outcome following major surgery evaluated in a randomized study. Ann. Surg. 1993, 217, 185–195. [Google Scholar] [CrossRef]
- Heslin, M.J.; Latkany, L.; Leung, D.; Brooks, A.D.; Hochwald, S.N.; Pisters, P.W.; Shike, M.; Brennan, M.F. A prospective randomized trial of early enteral feeding after resection of upper gastrointestinal malignancy. Ann. Surg. 1997, 226, 567–580. [Google Scholar] [CrossRef]
- Haskel, Y.; Xu, D.; Lu, Q.; Deitch, E. Bombesin protects against bacterial translocation induced by three commercially available liquid enteral diets: a prospective, randomized, multigroup trial. Crit. Care Med. 1994, 22, 108–113. [Google Scholar]
- Haskel, Y.; Xu, D.; Lu, Q.; Deitch, E. The modulatory role of gut hormones in elemental diet and intravenous total parenteral nutrition-induced bacterial translocation in rats. JPEN J. Parenter. Enteral Nutr. 1994, 18, 159–166. [Google Scholar]
- Slotwinski, R.; Olszewski, W.L.; Slotkowski, M.; Lech, G.; Zaleska, M.; Slotwinska, S.; Krasnodebski, W.I. Can the interleukin-1 receptor antagonist (IL-1ra) be a marker of anti-inflammatory response to enteral immunonutrition in malnourished patients after pancreaticoduodenectomy? J. Pancreas 2007, 8, 759–769. [Google Scholar]
- Oz, H.S.; Chen, T.S.; Neuman, M. Nutrition intervention: A strategy against systemic inflammatory syndrome. JPEN J. Parenter. Enteral Nutr. 2009, 33, 380–389. [Google Scholar] [CrossRef]
- Kaido, T.; Ogura, Y.; Ogawa, K.; Hata, K.; Yoshizawa, A.; Yagi, S.; Uemoto, S. Effects of post-transplant enteral nutrition with an immunomodulating diet containing hydrolyzed whey peptide after liver transplantation. World J. Surg. 2012, 36, 1666–1671. [Google Scholar] [CrossRef]
- Menges, T.; Engel, J.; Welters, I.; Wagner, R.M.; Little, S.; Ruwoldt, R.; Wollbrueck, M.; Hempelmann, G. Changes in blood lymphocyte populations after multiple trauma. Crit. Care Med. 1999, 27, 733–740. [Google Scholar]
- Zahorec, R. Ratio of neutrophil to lymphocyte counts—rapid and simple parameter of systemic inflammation and stress in critically ill. Bratisl. Lek. Listy 2001, 102, 5–14. [Google Scholar]
- Kalff, C.; Carlos, T.M.; Schraut, W.H.; Billiar, T.R.; Simmons, R.L.; Bauer, A.J. Surgically induced leukocytic infiltrates within the rat intestinal muscularis mediate postoperative ileus. Gastroenterology 1999, 117, 378–387. [Google Scholar] [CrossRef]
- De Jonge, W.J.; Van den Wungaard, R.M.; The, F.O.; Ter Bek, M.L.; Bennink, R.J.; Tytgat, G.N.J. Postoperative ileus is maintained by intestinal immune infiltrates that activate inhibitory neural pathways in mice. Gastroenterology 2003, 125, 1137–1147. [Google Scholar]
- Kubes, P.; Hunter, J.; Granger, D.N. Ischemia/reperfusion induced feline intestinal dysfunction: importance of granulocyte recruitment. Gastroenterology 1992, 103, 807–812. [Google Scholar]
- Wei, L.; Wei, H.; Frenkel, K. Sensitivity to tumor promotion of SENCAR and C57BL/6J mice correlates with oxidative events and DNA damage. Carcinogenesis 1993, 14, 841–847. [Google Scholar] [CrossRef]
- Steinberg, K.P.; Milberg, J.A.; Martin, T.A.; Maunder, R.J.; Cockrill, B.A.; Hudson, L.D. Evolution of bronchoalveolar cell populations in the adult respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 1994, 150, 113–122. [Google Scholar]
- Sookhai, S.; Wang, J.H.; McCourt, M.; Di Wu, Q.; Kirwan, H.D.; Redmond, H.P. A novel mechanism for attenuating neutrophil-mediated lung injury in vivo. Surg. Forum 1999, 50, 205–208. [Google Scholar]
- Ho, J.S.; Buchweitz, J.P.; Roth, R.A.; Ganey, P.E. Identification of factors from rat neutrophil responsible for cytotoxicity to isolated hepatocytes. J. Leukoc. Biol. 1996, 59, 716–724. [Google Scholar]
- Lowell, C.A.; Bertin, G. Resistance to endotoxic shock and reduced neutrophil migration in mice deficient for the Src-family kinases Hck and Fgr. Proc. Natl. Acad. Sci. USA 1998, 95, 7580–7584. [Google Scholar] [CrossRef]
- Goris, R.J.; Boekholtz, W.K.; van Bebber, I.P.; Nuytinck, J.K.; Schillings, P.H. Multiple-organ failure and sepsis without bacteria. An experimental model. Arch. Surg. 1986, 121, 897–901. [Google Scholar] [CrossRef]
- Wilson, M.R.; Choudhury, S.; Takata, M. Pulmonary inflammation induced by high-stretch ventilation is mediated by tumor necrosis factor signaling in mice. Am. J. Physiol. Lung Cell Mol. Physiol. 2005, 288, L599–L607. [Google Scholar]
- Rassias, A.J.; Marrin, C.A.S.; Arruda, J.; Whalen, P.K.; Beach, M.; Yeager, M.P. Insulin infusion improves neutrophil function in diabetic cardiac surgery patients. Anaesth. Analg. 1999, 88, 1011–1016. [Google Scholar]
- O´Brien, G.; Shields, C.J.; Winter, D.C.; Dillin, J.P.; Kirwan, W.O.; Redmont, H.P. Cyclooxygenase-2 plays a central role in the genesis of pancreatitis and associated lung injury. Hepatobil. Pancreat. Dis. Int. 2005, 4, 126–129. [Google Scholar]
- Bengmark, S. Control of systemic inflammation and chronic disease-the use of turmeric and curcumenoids. In Nutrigenomics and Proteonomics in Health and Disease. Food Factors and Gene Interaction; Mine, Y., Miyashita, K., Shahidi, F., Eds.; Wiley-Blackwell: Chichester, West Sussex, UK, 2009; pp. 161–180. [Google Scholar]
- Lee, J.C.; Kinniry, P.A.; Arguiri, E.; Serota, M.; Kanterakis, S.; Chatterjee, S.; Solomides, C.C.; Javvadi, P.; Koumenis, C.; Cengel, K.A.; Christofidou-Solomidou, M. Dietary curcumin increases antioxidant defenses in lung, ameliorates radiation-induced pulmonary fibrosis, and improves survival in mice. Radiat. Res. 2010, 173, 590–601. [Google Scholar] [CrossRef]
- Landi-Librandi, A.P.; Caleiro Seixas Azzolini, A.E.; de Oliveira, C.A.; Lucisano-Valim, Y.M. Inhibitory activity of liposomal flavonoids during oxidative metabolism of human neutrophils upon stimulation with immune complexes and phorbol ester. Drug Deliv. 2012, 19, 177–187. [Google Scholar] [CrossRef]
- Bengmark, S. Synbiotics in human Medicine. In Therapeutic Microbiology: Probiotics and Related Strategies; Versalovic, J., Wilson, M., Eds.; ASM Press: Washington, DC, USA, 2008; pp. 307–321. [Google Scholar]
- Bengmark, S. Gut microbiota, immune development and function. Pharmacol. Res. 2012. [Google Scholar] [CrossRef]
- Ilkgul, O.; Aydede, H.; Erhan, Y.; Surucuoglu, S.; Gazi, H.; Vatansever, S.; Taneli, F.; Ulman, C.; Kose, C.; Bengmark, S. Subcutaneous administration of live lactobacillus prevents sepsis-induced lung organ failure in rats. Br. J. Int. Care 2005, 15, 52–57. [Google Scholar]
- Tok, D.; Ilkgul, O.; Bengmark, S.; Aydede, H.; Erhan, Y.; Taneli, F.; Ulman, C.; Vatansever, S.; Kose, C.; Ok, G. Pretreatment with pro- and synbiotics reduces peritonitis-induced acute lung injury in rats. J. Trauma 2007, 62, 880–885. [Google Scholar] [CrossRef]
- Shimizu, K.; Ogura, H.; Goto, M.; Asahara, T.; Nomoto, K.; Morotomi, M.; Yoshiya, K.; Matsushima, A.; Sumi, Y.; Kuwagata, Y.; Tanaka, H.; Shimazu, T.; Sugimoto, H. Altered gut flora and environment in patients with severe SIRS. J. Trauma 2006, 60, 126–133. [Google Scholar] [CrossRef]
- Shimizu, K.; Ogura, H.; Asahara, T.; Nomoto, K.; Morotomi, M.; Nakahori, Y.; Osuka, A.; Yamano, S.; Goto, M.; Matsushima, A.; Tasaki, O.; Kuwagata, Y.; Sugimoto, H. Gastrointestinal dysmotility is associated with altered gut flora and septic mortality in patients with severe systemic inflammatory response syndrome: A preliminary study. Neurogastroenterol. Motil. 2011, 23, 330–335. [Google Scholar] [CrossRef]
- Shimizu, K.; Ogura, H.; Goto, M.; Asahara, T.; Nomoto, K.; Morotomi, M.; Matsushima, A.; Tasaki, O.; Fujita, K.; Hosotsubo, H.; Kuwagata, Y.; Tanaka, H.; Shimazu, T.; Sugimoto, H. Synbiotics decrease the incidence of septic complications in patients with severe SIRS: A preliminary report. Dig. Dis. Sci. 2009, 54, 1071–1078. [Google Scholar] [CrossRef]
- Neish, A. Microbes in gastrointestinal health and disease. Gastroenterology 2009, 136, 65–80. [Google Scholar] [CrossRef]
- Frank, D.N.; Pace, N.R. Gastrointestinal microbiology enters the metagenomics era. Curr. Opin. Gastroenterol. 2008, 24, 4–10. [Google Scholar] [CrossRef]
- Arumugam, M.; Raes, J.; Pelletier, E.; Le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; Batto, J.M.; et al. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar]
- Ursell, L.K.; Clemente, J.C.; Rideout, J.R.; Gevers, D.; Caporaso, J.G.; Knight, R. The interpersonal and intrapersonal diversity of human-associated microbiota in key body sites. J. Allergy Clin. Immunol. 2012, 129, 1204–1208. [Google Scholar] [CrossRef]
- Clemente, J.C.; Ursell, L.K.; Parfrey, L.W.; Knight, R. The impact of the gut microbiota on human health: an integrative view. Cell 2012, 148, 1258–1270. [Google Scholar] [CrossRef]
- Ley, R.E.; Hamady, M.; Lozupone, C.; Turnbaugh, P.J.; Ramey, R.R.; Bircher, J.S.; Schlegel, M.L.; Tucker, T.A.; Schrenzel, M.D.; Knight, R.; Gordon, J.I. Evolution of mammals and their gut microbes. Science 2008, 320, 1647–1651. [Google Scholar]
- Muegge, B.D.; Kuczynski, J.; Knights, D.; Clemente, J.C.; Gonzalez, A.; Fontana, L.; Henrissat, B.; Knight, R.; Gordon, J.I. Diet drives convergence in gut microbiome functions across mammalian phylogeny and within humans. Science 2011, 332, 970–974. [Google Scholar]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R.; et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef]
- Matafome, P.; Santos-Silva, D.; Crisóstomo, J.; Rodrigues, T.; Rodrigues, L.; Sena, C.M.; Pereira, P.; Seiça, R. Methylglyoxal causes structural and functional alterations in adipose tissue independently of obesity. Arch. Physiol. Biochem. 2012, 118, 58–68. [Google Scholar] [CrossRef]
- Sekirov, I.; Russel, S.L.; Antunes, C.M.; Finlay, B.B. Gut microbiota in health and disease. Physiol. Rev. 2010, 90, 859–904. [Google Scholar] [CrossRef]
- Tap, J.; Mondot, S.; Levenez, F.; Pelletier, E.; Caron, C.; Furet, J.P.; Ugarte, E.; Muñoz-Tamayo, R.; Paslier, D.L.; Nalin, R.; Dore, J.; Leclerc, M. Towards the human intestinal microbiota phylogenetic core. Environ. Microbiol. 2009, 11, 2574–2584. [Google Scholar] [CrossRef]
- Håkansson, Å.; Molin, G. Gut microbiota and inflammation. Nutrients 2011, 3, 637–382. [Google Scholar] [CrossRef]
- Stearns, J.C.; Lynch, M.D.J.; Senadheera, D.B.; Tenenbaum, H.C.; Goldberg, M.B.; Cvitkovitch, D.G.; Croitoru, K.; Moreno-Hagelsieb, G.; Neufeld, J.D. Bacterial biogeography of the human digestive tract. Sci. Rep. 2011, 1, 170. [Google Scholar]
- Ekberg, H. Colorectal liver cancer, resection and regional chemotherapy. In Bulletin from Department of Surgery; Lund University: Lund, Sweden, 1986; pp. 1–76. [Google Scholar]
- Ekberg, H.; Tranberg, K.G.; Andersson, R.; Jeppsson, B.; Bengmark, S. Major liver resection: Perioperative course and management. Surgery 1986, 100, 1–8. [Google Scholar]
- Gustafsson, B.E. The physiological importance of the colonic microflora. Scand. J. Gastroenterol. 1982, 77, S117–S131. [Google Scholar]
- Gilliland, S.E.; Speck, M.L. Antagonistic action of Lactobacillus acidophilus towards intestinal and food-borne pathogens in associative cultures. J. Food Prot. 1977, 40, 820–823. [Google Scholar]
- Johansson, M.L.; Molin, G.; Jeppsson, B.; Nobaek, S.; Ahrné, S.; Bengmark, S. Administration of different Lactobacillus strains in fermented oatmeal soup: in vivo colonization of human intestinal mucosa and effect on the indigenous flora. Appl. Environ. Microbiol. 1993, 59, 15–20. [Google Scholar]
- Molin, G.; Andersson, R.; Ahrné, S.; Lönner, C.; Marklinder, I.; Johansson, M.L.; Jeppsson, B.; Bengmark, S. Effect of fermented oatmeal soup on the cholesterol level and the Lactobacillus colonization of rat intestinal mucosa. Antonie Van Leeuwenhoek 1992, 61, 167–173. [Google Scholar] [CrossRef]
- Molin, G.; Jeppsson, B.; Johansson, M.L.; Ahrné, S.; Nobaek, S.; Ståhl, M.; Bengmark, S. Numerical taxonomy of Lactobacillus spp. associated with healthy and diseased mucosa of the human intestines. J. Appl. Bacteriol. 1993, 74, 314–323. [Google Scholar] [CrossRef]
- Lan, J.G.; Yamagisawa, N. Isolation, selection and characteristics of Lactobacillus paracasei ssp. paracasei isolate F19. Microb. Ecol. Health Dis. 2002, 3, S4–S6. [Google Scholar]
- Kruszewska, K.; Lan, J.; Lorca, G.; Yanagisawa, N.; Marklinder, I.; Ljungh, Å. Selection of lactic acid bacteria as probiotic strains by in vitro tests. Microecol. Ther. 2002, 29, 37–51. [Google Scholar]
- Rayes, N.; Hansen, S.; Seehöfer, D.; Müller, A.R.; Serke, S.; Bengmark, S.; Neuhaus, P. Early enteral supply of fiber and Lactobacilli versus conventional nutrition: a controlled trial in patients with major abdominal surgery. Nutrition 2002, 18, 609–615. [Google Scholar] [CrossRef]
- Rayes, N.; Seehöfer, D.; Theruvath, T.; Mogl, M.; Langrehr, J.M.; Nüssler, N.C.; Bengmark, S.; Neuhaus, P. Effect of enteral nutrition and synbiotics on bacterial infection rates after pylorus-preserving pancreatoduodenectomy: a randomized, double-blind trial. Ann. Surg. 2007, 246, 36–41. [Google Scholar] [CrossRef]
- Han, C.M. Personal communication, Medical School, Zhejiang University: Hangzhou, China, 2012.
- Rayes, N.; Seehöfer, D.; Hansen, S.; Boucsein, K.; Müller, A.R.; Serke, S.; Bengmark, S.; Neuhaus, P. Early enteral supply of lactobacillus and fiber versus selective bowel decontamination: A controlled trial in liver transplant recipients. Transplantation 2002, 74, 123–127. [Google Scholar] [CrossRef]
- Rayes, N.; Seehöfer, D.; Theruvath, T.; Schiller, R.A.; Langrehr, J.M.; Jonas, S.; Bengmark, S.; Neuhaus, P. Supply of pre- and probiotics reduces bacterial infection rates after liver transplantation—a randomized, double-blind trial. Am. J. Transplant. 2005, 5, 125–130. [Google Scholar] [CrossRef]
- Spindler-Vesel, A.; Bengmark, S.; Vovk, I.; Cerovic, O.; Kompan, L. Synbiotics, pre-biotics, glutamine, or peptide in early enteral nutrition: a randomized study in trauma patients. JPEN J. Parenter. Enteral Nutr. 2007, 31, 119–126. [Google Scholar] [CrossRef]
- Kotzampassi, K.; Giamerellos-Bourboulis, E.J.; Voudouris, A.; Kazamias, P.; Eleftheriadis, E. Benefits of Synbiotic 2000 Forte in critically ill trauma patients-early results of a randomized controlled trial. World J. Surg. 2006, 30, 1848–1855. [Google Scholar] [CrossRef]
- Giamarellos-Bourboulis, E.J.; Bengmark, S.; Kanellakopoulou, K.; Kotzampassi, K. Pro- and synbiotics to control inflammation and infection in patients with multiple injuries. J. Trauma 2009, 67, 815–818. [Google Scholar] [CrossRef]
- Koutelidakis, I.M.; Bezirtzoglou, E.; Giamarellos-Bourboulis, E.J.; Grosomanidis, V.; Kotzampassi, K. Impact of synbiotics on the intestinal flora of critically ill patients with multiple injuries. Int. J. Antimicrob. Agents 2010, 36, 90–91. [Google Scholar] [CrossRef]
- Oláh, A.; Belágyi, T.; Issekutz, Á.; Gamal, M.E.; Bengmark, S. Early enteral nutrition with specific lactobacillus and fibre reduces sepsis in severe acute pancreatitis. Br. J. Surg. 2002, 89, 1103–1107. [Google Scholar] [CrossRef]
- Oláh, A.; Belágyi, T.; Pótó, L.; Romics, L., Jr.; Bengmark, S. Synbiotic control of inflammation and infection in severe acute pancreatitis, a prospective randomized double blind study. Hepatogastroenterology 2007, 54, 590–594. [Google Scholar]
- Plaudis, H.; Pupelis, G.; Zeiza, K.; Boka, V. Early Low Volume Oral Synbiotic/Prebiotic Supplemented Enteral Stimulation of the Gut in Patients with Severe Acute Pancreatitis: A Prospective Feasibility Study. Acta Chir. Belg. 2012, 112, 131–138. [Google Scholar]
- Butterworth, R.F. Hepatic encephalopathy: A central neuroinflammator1 disorder? Hepatology 2011, 53, 1372–1376. [Google Scholar] [CrossRef]
- Eutamene, H.; Bueno, L. Role of probiotics in correcting abnormalities of colonic flora induced by stress. Gut 2007, 56, 1495–1497. [Google Scholar] [CrossRef]
- Ait-Belgnaoui, A.; Durand, H.; Cartier, C.; Chaumaz, G.; Eutamene, H.; Ferrier, L.; Houdeau, E.; Fioramonti, J.; Bueno, L.; Theodorou, V. Prevention of gut leakiness by a probiotic treatment leads to attenuated HPA response to an acute psychological stress in rats. Psychoneuroendocrinoloy 2012, 37, 1885–1895. [Google Scholar] [CrossRef]
- Liu, Q.; Duan, Z.P.; Ha, D.K.; Bengmark, S.; Kurtovic, J.; Riordan, S.M. Synbiotic modulation of gut flora: effect on minimal hepatic encephalopathy in patients with cirrhosis. Hepatology 2004, 39, 1441–1449. [Google Scholar] [CrossRef]
- Riordan, S.M.; Skinner, N.A.; McIver, C.J.; Liu, Q.; Bengmark, S.; Bihari, D.; Visvanathan, K. Synbiotic-associated improvement in liver function in cirrhotic patients: Relation to changes in circulating cytokine messenger RNA and protein levels. Microb. Ecol. Health Dis. 2007, 19, 7–16. [Google Scholar] [CrossRef]
- Bengmark, S. Bio-ecological control of chronic liver disease and encephalopathy. Metab. Brain Dis. 2009, 24, 223–236. [Google Scholar] [CrossRef]
- Brenchley, J.M.; Price, D.A.; Schacker, T.W.; Asher, T.E.; Silvestri, G.; Rao, S.; Kazzaz, Z.; Bornstein, E.; Lambotte, O.; Altmann, D.; et al. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat. Med. 2006, 12, 1365–1371. [Google Scholar]
- Gori, A.; Tincati, C.; Rizzardini, G.; Torti, C.; Quirino, T.; Haarman, M.; Amor, K.B.; van Schaik, J.; Vriesema, A.; Knol, J.; et al. Early impairment of gut function and gut flora supporting a role for alteration of gastrointestinal mucosa in human immunodeficiency virus pathogenesis. J. Clin. Microbiol. 2008, 46, 757. [Google Scholar] [CrossRef]
- Ellis, C.L.; Ma, C.M.; Mann, S.K.; Li, S.C.; Wu, J.; Knight, T.H.; Yotter, T.; Hayes, T.L.; Maniar, A.H.; Troia-Cancio, P.V.; et al. Molecular characterization of stool microbiota in HIV-infected subjects by panbacterial and order-level 16S Ribosomal DNA (rDNA) quantification and correlations with immune activation. J. Acquir. Immune Defic. Syndr. 2011, 57, 363–370. [Google Scholar] [CrossRef]
- Cunningham-Rundles, S.; Ahrné, S.; Johann-Liang, R.; Abuav, R.; Dunn-Navarra, A.M.; Grassey, C.; Bengmark, S.; Cervia, J.S. Effect of probiotic bacteria on microbial host defense, growth, and immune function in human immunodeficiency virus type-1 infection. Nutrients 2011, 3, 1042–1070. [Google Scholar] [CrossRef]
- Hummelen, R.; Changalucha, J.; Butamanya, N.L.; Cook, A.; Habbema, J.D.; Reid, G. Lactobacillus rhamnosus GR-1 and L. reuteri RC-14 to prevent or cure bacterial vaginosis among women with HIV. Int. J. Gynaecol. Obstet. 2010, 111, 245–248. [Google Scholar] [CrossRef]
- Hummelen, R.; Changalucha, J.; Butamanya, N.L.; Koyama, T.E.; Cook, A.; Habbema, J.D.; Reid, G. Effect of 25 weeks probiotic supplementation on immune function of HIV patients. Gut Microbes 2011, 2, 80–85. [Google Scholar] [CrossRef]
- Hummelen, R.; Hemsworth, J.; Changalucha, J.; Butamanya, N.L.; Hekmat, S.; Habbema, J.D.; Reid, G. Effect of micronutrient and probiotic fortified yogurt on immune-function of anti-retroviral therapy naive HIV patients. Nutrients 2011, 3, 897–909. [Google Scholar] [CrossRef]
- Schunter, M.; Chu, H.; Hayes, T.L.; McConnell, D.L.; Crawford, S.S.; Luciw, P.A.; Bengmark, S.; Asmuth, D.M.; Brown, J.; Bevins, C.L.; et al. Randomized Pilot Trial of a Synbiotic Dietary Supplement in Chronic HIV-1 Infection. BMC Complement Altern. Med. 2012, 12, 84. [Google Scholar]
- Calvano, S.E.; Xiao, W.; Richards, D.R.; Felciano, R.M.; Baker, H.V.; Cho, R.J.; Chen, R.O.; Brownstein, B.H.; Cobb, J.P.; Tschoeke, S.K.; et al. A network-based analysis of systemic inflammation in humans. Nature 2005, 13, 1032–1037. [Google Scholar]
- Loza, M.J.; McCall, C.E.; Li, L.; Isaacs, W.B.; Xu, J.; Chang, B.L. Assembly of inflammation-related genes for pathway-focused genetic analysis. PLoS One 2007, 2, e1035. [Google Scholar] [CrossRef]
- Seok, J.; Xiao, W.; Moldawer, L.L.; Davis, R.W.; Covert, M.W. A dynamic network of transcription in LPS-treated human subjects. BMC Syst. Biol. 2009, 3, 78. [Google Scholar] [CrossRef]
- Burcelin, R.; Serino, M.; Chabo, C.; Blasco-Baque, V.; Amar, J. Gut microbiota and diabetes: from pathogenesis to therapeutic perspective. Acta Diabetol. 2011, 48, 257–273. [Google Scholar] [CrossRef]
- Festi, D.; Schiumerini, R.; Birtolo, C.; Marzi, L.; Montrone, L.; Scaioli, E.; Di Biase, A.R.; Colecchia, A. Gut microbiota and its pathophysiology in disease paradigms. Dig. Dis. 2011, 29, 518–524. [Google Scholar] [CrossRef]
- Leavy, O. Inflammation: Trauma kicks up a storm. Nat. Rev. Immunol. 2011, 12, 3. [Google Scholar]
- London, N.R.; Zhu, W.; Bozza, F.A.; Smith, M.C.; Greif, D.M.; Sorensen, L.K.; Chen, L.; Kaminoh, Y.; Chan, A.C.; Passi, S.F.; et al. Targeting Robo4-dependent Slit signaling to survive the cytokine storm in sepsis and influenza. Sci. Transl. Med. 2010, 2, 23–ra19. [Google Scholar] [CrossRef]
- Thomas, E.L.; Saeed, N.; Hajnal, J.V.; Goldstone, A.P.; Frost, G.; Bell, J.D. Magnetic resonance imaging of total body fat. J. Appl. Physiol. 1998, 85, 1778–1785. [Google Scholar]
- Alessi, M.C.; Peiretti, F.; Morange, P.; Henry, M.; Nalbone, G.; Juhan-Vague, I. Production of plasminogen activator inhibitor 1 by human adipose tissue: Possible link between visceral fat accumulation and vascular disease. Diabetes 1997, 46, 860–867. [Google Scholar]
- Almersjö, O.; Bengmark, S.; Engevik, L.; Kerstell, J.; Svanborg, A. Serum lipids after extensiveliver resectionin man. Acta Hepatosplenol. 1968, 15, 1–12. [Google Scholar]
- Bengmark, S. Liver steatosis and liver resection. Digestion 1969, 2, 304–311. [Google Scholar] [CrossRef]
- Henao-Mejia, J.; Elinav, E.; Jin, C.; Hao, L.; Mehal, W.Z.; Strowig, T.; Thaiss, C.A.; Kau, A.L.; Eisenbarth, S.C.; Jurczak, M.J.; et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature 2012, 482, 179–185. [Google Scholar]
- Bengmark, S.; Olsson, R.; Svanborg, A. Fat infiltration after partial hepatectomy, changes in glycerides, cholesterol and phospholipids in the residual liver of the rat. Acta Hepatosplenol. 1964, 11, 276–285. [Google Scholar]
- Girish, C.; Pradhan, S.C. Hepatoprotective activities of picroliv, curcumin, and ellagic acid compared to silymarin on carbon-tetrachloride-induced liver toxicity in mice. J. Pharmacol. Pharmacother. 2012, 3, 149–155. [Google Scholar]
- Xu, R.Y.; Wan, Y.P.; Fang, Q.Y.; Lu, W.; Cai, W. Supplementation with probiotics modifies gut flora and attenuates liver fat accumulation in rat nonalcoholic fatty liver disease model. J. Clin. Biochem. Nutr. 2012, 50, 72–77. [Google Scholar]
- Seehofer, D.; Schirmeier, A.; Bengmark, S.; Cho, S.Y.; Koch, M.; Lederer, A.; Rayes, N.; Menger, M.D.; Neuhaus, P.; Nüssler, A.K. Curcumin attenuates oxidative stress and inflammatory response in the early phase after partial hepatectomy with simultaneous intraabdominal infection in rats. J. Surg. Res. 2010, 159, 497–502. [Google Scholar] [CrossRef]
- Kuo, J.J.; Chang, H.H.; Tsai, T.H.; Lee, T.Y. Positive effect of curcumin on inflammation and mitochondrial dysfunction in obese mice with liver steatosis. Int. J. Mol. Med. 2012, 30, 673–679. [Google Scholar]
- Müller-Ladner, U.; Pap, T.; Gay, R.E.; Neidhart, M.; Gay, S. Mechanisms of disease: the molecular and cellular basis of joint destruction in rheumatoid arthritis. Nat. Clin. Pract. Rheumatol. 2005, 1, 102–110. [Google Scholar] [CrossRef]
- Simmonds, R.E.; Foxwell, B.M. Signalling, inflammation and arthritis NF-κB and its relevance to arthritis and inflammation. Rheumatology 2008, 47, 584–590. [Google Scholar] [CrossRef]
- Perrier, C.; Rutgeerts, P. Cytokine blockade in inflammatory bowel diseases. Immunotherapy 2011, 3, 1341–1352. [Google Scholar] [CrossRef]
- Mancini, G.B.; Baker, S.; Bergeron, J.; Fitchett, D.; Frohlich, J.; Genest, J.; Gupta, M.; Hegele, R.A.; Ng, D.; Pope, J. Diagnosis, prevention, and management of statin adverse effects and intolerance: proceedings of a Canadian Working Group Consensus Conference. Can. J. Cardiol. 2011, 27, 635–662. [Google Scholar]
- Aggarwal, B.B.; Sethi, G.; Baladandayuthapani, V.; Krishnan, S.; Shishodia, S. Targeting cell signaling pathways for drug discovery: an old lock needs a new key. J. Cell. Biochem. 2007, 102, 580–592. [Google Scholar] [CrossRef]
- Bengmark, S. Advanced glycation and lipoxidation end products—amplifiers of inflammation: The role of food. JPEN J. Parenter. Enteral Nutr. 2007, 31, 430–440. [Google Scholar] [CrossRef]
- Bengmark, S. ALE RAGE and disease—a foods perspective. In Handbook of Prebiotic and Probiotic Ingredients: Health Benefits and Food Applications; Cho, S.S., Finocchiaro, T., Eds.; CRC Press, Taylor and Francis Group: Boca Raton, FL, USA, 2010; pp. 139–162. [Google Scholar]
- Bengmark, S. Modified Amino Acid-Based Molecules: Accumulation and Health Implications. In Amino Acids in Human Nutrition and Health; Mello, J.F.D., Ed.; CABI: Wallingford, UK, 2011; pp. 382–405. [Google Scholar]
- Vanschoonbeek, K.; Lansink, M.; van Laere, K.M.; Senden, J.M.; Verdijk, L.B.; van Loon, L.J. Slowly digestible carbohydrate sources can be used to attenuate the postprandial glycemic response to the ingestion of diabetes-specific enteral formulas. Diabetes Educ. 2009, 35, 631–640. [Google Scholar] [CrossRef]
- Martindale, R.G.; DeLegge, M.; McClave, S.; Monroe, C.; Smith, V.; Kiraly, L. Nutrition delivery for obese ICU patients: Delivery issues, lack of guidelines, and missed opportunities. JPEN J. Parenter. Enteral Nutr. 2011, 35, S80–S87. [Google Scholar] [CrossRef]
- Miner-Williams, W.; Deglaire, A.; Benamouzig, R.; Fuller, M.F.; Tomé, D.; Moughan, P.J. Endogenous proteins in terminal ileal digesta of adult subjects fed acasein-based diet. Am. J. Clin. Nutr. 2012, 96, 508–515. [Google Scholar] [CrossRef]
- Veldhorst, M.A.; Nieuwenhuizen, A.G.; Hochstenbach-Waelen, A.; Westerterp, K.R.; Engelen, M.P.; Deutz, N.E.; Westerterp-Plantenga, M.S. A breakfast with alpha-lactalbumin, gelatin, or gelatin + TRP lowers energy intake at lunch compared with a breakfast with casein, soy, whey, or whey-GMP. Clin. Nutr. 2009, 28, 147–155. [Google Scholar] [CrossRef]
- Ang, M.; Müller, A.S.; Wagenlehner, F.; Pilatz, A.; Linn, T. Combining protein and carbohydrate increases postprandial insulin levels but does not improve glucose response in patients with type 2 diabetes. Metabolism 2012, 61, 1696–1702. [Google Scholar] [CrossRef]
- Liu, X.; Murali, S.G.; Holst, J.J.; Ney, D.M. Wheyprotein potentiates the intestinotrophic action of glucagon-like peptide-2 in parenterally fed rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 297, R1554–R1562. [Google Scholar] [CrossRef]
- Tang, J.E.; Moore, D.R.; Kujbida, G.W.; Tarnopolsky, M.A.; Phillips, S.M. Ingestion of whey hydrolysate, casein, or soy protein isolate: effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. J. Appl. Physiol. 2009, 107, 987–992. [Google Scholar] [CrossRef]
- Morifuji, M.; Sakai, K.; Sugiura, K. Dietary whey protein modulates liver glycogen level and glycoregulatory enzyme activities in exercise-trained rats. Exp. Biol. Med. (Maywood) 2005, 230, 23–30. [Google Scholar]
- Ebaid, H.; Salem, A.; Sayed, A.; Metwalli, A. Whey proteinenhances normal inflammatory responses during cutaneous wound healing in diabetic rats. Lipids Health Dis. 2011, 10, 235. [Google Scholar] [CrossRef]
- Hoppe, C.; Mølgaard, C.; Dalum, C.; Vaag, A.; Michaelsen, K.F. Differential effects of casein versus whey on fasting plasma levels of insulin, IGF-1 and IGF-1/IGFBP-3: results from a randomized 7-day supplementation study in prepubertal boys. Eur. J. Clin. Nutr. 2009, 63, 1076–1083. [Google Scholar] [CrossRef]
- Graf, S.; Egert, S.; Heer, M. Effects of whey protein supplements on metabolism: Evidence from human intervention studies. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 569–580. [Google Scholar] [CrossRef]
- Yang, L.; Chen, J.H.; Xu, T.; Zhou, A.S.; Yang, H.K. Rice protein improves oxidative stress by regulating glutathione metabolism and attenuating oxidative damage to lipids and proteins in rats. Life Sci. 2012, 91, 389–394. [Google Scholar] [CrossRef]
- Alvarez-Jubete, L.; Arendt, E.K.; Gallagher, E. Nutritive value and chemical composition of pseudocereals as gluten-free ingredients. Int. J. Food Sci. Nutr. 2009, 60, S240–S257. [Google Scholar] [CrossRef]
- Chintagari, N.R.; Liu, L. GABA receptor ameliorates ventilator-induced lung injury in rats by improving alveolar fluid clearance. Crit. Care 2012, 16, R55. [Google Scholar] [CrossRef]
- Schep, L.J.; Knudsen, K.; Slaughter, R.J.; Vale, J.A.; Mégarbane, B. The clinical toxicology of γ-hydroxybutyrate, γ-butyrolactone and 1,4-butanediol. Clin. Toxicol. 2012, 5, 458–470. [Google Scholar]
- Coda, R.; Rizzello, C.G.; Gobbetti, M. Use of sourdough fermentation and pseudo-cereals and leguminous flours for the making of a functional bread enriched of gamma-aminobutyric acid (GABA). Int. J. Food Microbiol. 2010, 137, 236–245. [Google Scholar] [CrossRef]
- Coda, R.; Di Cagno, R.; Rizzello, C.G.; Nionelli, L.; Edema, M.O.; Gobbetti, M. Utilization of African grains for sourdough bread making. J. Food Sci. 2011, 76, M329–M335. [Google Scholar] [CrossRef]
- Han, Y.Y.; Lai, S.L.; Ko, W.J.; Chou, C.H.; Lai, H.S. Effects of fish oil on inflammatory modulation in surgical intensive care unit patients. Nutr. Clin. Pract. 2012, 27, 91–98. [Google Scholar] [CrossRef]
- Versleijen, M.W.; Roelofs, H.M.; Rombouts, C.; Hermans, P.W.; Noakes, P.S.; Calder, P.C.; Wanten, G.J. Short-term infusion of a fish oil-based lipid emulsion modulates fatty acid status, but notimmunefunction or (anti)oxidant balance: a randomized cross-over study. Eur. J. Clin. Invest. 2012, 42, 290–302. [Google Scholar] [CrossRef]
- Levy, B.D. Resolvins and protectins: Natural pharmacophores for resolution biology. Prostaglandins Leukot. Essent. Fatty Acids 2010, 82, 327–332. [Google Scholar] [CrossRef]
- Ott, J.; Hiesgen, C.; Mayer, K. Lipids in critical care medicine. Prostaglandins Leukot. Essent. Fatty Acids 2011, 85, 267–273. [Google Scholar] [CrossRef]
- van der Meij, B.S.; van Bokhorst-de van der Schueren, M.A.; Langius, J.A.; Brouwer, I.A.; van Leeuwen, P.A. n-3 PUFAs in cancer, surgery, and critical care: A systematic review on clinical effects, incorporation, and washout of oral or enteral compared with parenteral supplementation. Am. J. Clin. Nutr. 2011, 94, 1248–1265. [Google Scholar] [CrossRef]
- Rice, T.W.; Wheeler, A.P.; Thompson, B.T.; deBoisblanc, B.P.; Steingrub, J.; Rock, P. NIH NHLBI Acute Respiratory Distress Syndrome Network of Investigators. Enteral omega-3 fatty acid, gamma-linolenic acid, and antioxidant supplementation in acute lung injury. JAMA 2011, 306, 1574–1581. [Google Scholar] [CrossRef]
- Wang, J.; Yu, J.C.; Kang, W.M.; Ma, Z.Q. Superiority of a fish oil-enriched emulsion to medium-chain triacylglycerols/long-chain triacylglycerols in gastrointestinal surgery patients: A randomized clinical trial. Nutrtion 2012, 28, 623–629. [Google Scholar]
- Umpierrez, G.E.; Spiegelman, R.; Zhao, V.; Smiley, D.D.; Pinzon, I.; Griffith, D.P.; Peng, L.; Morris, T.; Luo, M.; Garcia, H.; Thomas, C.; Newton, C.A.; Ziegler, T.R. A double-blind, randomized clinical trial comparing soybean oil-based versus olive oil-based lipid emulsions in adult medical-surgical intensive care unit patients requiring parenteral nutrition. Crit. Care Med. 2012, 40, 1792–1798. [Google Scholar]
- Siqueira, J.; Smiley, D.; Newton, C.; Le, N.A.; Gosmanov, A.R.; Spiegelman, R.; Peng, L.; Osteen, S.J.; Jones, D.P.; Quyyumi, A.A.; Ziegler, T.R.; Umpierrez, G.E. Substitution of standard soybean oil with olive oil-based lipid emulsion in parenteral nutrition: comparison of vascular, metabolic, and inflammatory effects. J. Clin. Endocrinol. Metab. 2011, 96, 3207–3216. [Google Scholar]
- Unlu, N.Z.; Bohn, T.; Clinton, S.K.; Schwartz, S.J. Carotenoid absorption from salad and salsa by humans is enhanced by the addition of avocado or avocado oil. J. Nutr. 2005, 135, 431–436. [Google Scholar]
- Boileau, C.; Martel-Pelletier, J.; Caron, J.; Msika, P.; Guillou, G.B.; Baudouin, C. Protective effects of total fraction of avocado/soybean unsaponifiables on the structural changes in experimental dog osteoarthritis: inhibition of nitric oxide synthase and matrix metalloproteinase-13. Arthritis Res. Ther. 2009, 11, R41. [Google Scholar] [CrossRef]
- Oguntibeju, O.O.; Esterhuyse, A.J.; Truter, E.J. Red palm oil: nutritional, physiological and therapeutic roles in improving human wellbeing and quality of life. Br. J. Biomed. Sci. 2009, 66, 216–222. [Google Scholar]
- Sen, C.K.; Rink, C.; Khanna, S. Palm oil-derived natural vitamin E alpha-tocotrienol in brain health and disease. J. Am. Coll. Nutr. 2010, 29, S314–S323. [Google Scholar]
- Takeuchi, H.; Sekine, S.; Kojima, K.; Aoyama, T. The application of medium-chainfatty acids: edible oil with a suppressing effect on body fat accumulation. Asia Pac. J. Clin. Nutr. 2008, 17, S320–S323. [Google Scholar]
- Wu, S.J.; Liu, P.L.; Ng, L.T. Tocotrienol-rich fraction ofpalm oilexhibits anti-inflammatory property by suppressing the expression of inflammatory mediators in human monocytic cells. Mol. Nutr. Food Res. 2008, 52, 921–929. [Google Scholar] [CrossRef]
- Esterhuyse, A.J.; Du Toit, E.F.; Van Rooyen, J. Dietary Red Palm Oil supplementation protects against the consequences of global ischaemia in the isolated perfused rat heart. Asia Pac. J. Clin. Nutr. 2005, 14, 1–7. [Google Scholar]
- Esterhuyse, A.J.; Du Toit, E.F.; Benadè, A.J.S.; van Rooyen, J. Dietary Red Palm Oil improves reperfusion cardiac function in the isolated perfused rat heart of animals fed a high cholesterol diet. Prostaglandins Leukot. Essent. Fatty Acids 2005, 72, 153–161. [Google Scholar] [CrossRef]
- van Rooyen, J.; Esterhuyse, A.J.; Engelbrecht, A.M.; du Toit, E.F. Health benefits of a natural carotenoid rich oil: a proposed mechanism of protection against ischaemia/reperfusion injury. Asia Pac. J. Clin. Nutr. 2008, 17, S316–S319. [Google Scholar]
- Desbois, A.P.; Smith, V.J. Antibacterial free fatty acids: Activities, mechanisms of action and biotechnological potential. Appl. Microbiol. Biotechnol. 2010, 85, 1629–1642. [Google Scholar] [CrossRef]
- Descotes, J. Immunotoxicology: Role in the safety assessment of drugs. Drug Saf. 2005, 28, 127–136. [Google Scholar] [CrossRef]
- Boutenko, V. Green for Life; North Atlantic Books: Berkeley, CA, USA, 2010. [Google Scholar]
- Iapichino, G.; Callegari, M.L.; Marzorati, S.; Cigada, M.; Corbella, D.; Ferrari, S.; Morelli, L. Impact of antibiotics on the gut microbiota of critically ill patients. J. Med. Microbiol. 2008, 57, 1007–1014. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bengmark, S. Nutrition of the Critically Ill — A 21st-Century Perspective. Nutrients 2013, 5, 162-207. https://doi.org/10.3390/nu5010162
Bengmark S. Nutrition of the Critically Ill — A 21st-Century Perspective. Nutrients. 2013; 5(1):162-207. https://doi.org/10.3390/nu5010162
Chicago/Turabian StyleBengmark, Stig. 2013. "Nutrition of the Critically Ill — A 21st-Century Perspective" Nutrients 5, no. 1: 162-207. https://doi.org/10.3390/nu5010162
APA StyleBengmark, S. (2013). Nutrition of the Critically Ill — A 21st-Century Perspective. Nutrients, 5(1), 162-207. https://doi.org/10.3390/nu5010162




