Regulation of Human Stem Cells by Functional Food Components: How Vitamins, Minerals and Phytochemicals Influence Mesenchymal Stem Cells’ Fate and Function
Abstract
1. Introduction
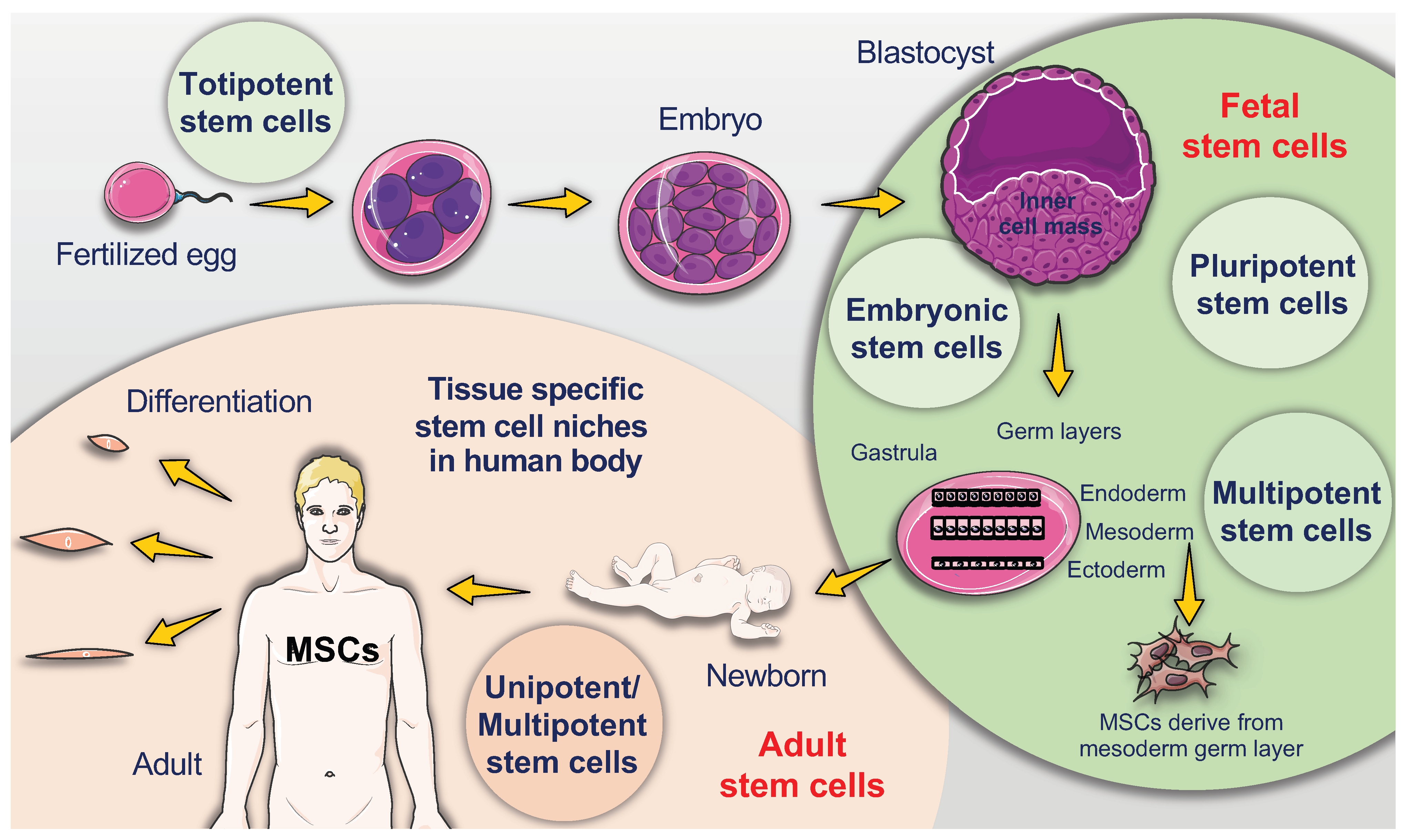
Embryonic Stem Cells vs. Adult Stem Cells
2. Materials and Methods
3. Therapeutic Properties of Mesenchymal Stem Cells
4. Vitamins and Trace Elements Shape MSCs Fate—Regulation of Proliferation, Self-Renewal, and Senescence in Mesenchymal Stem Cells
4.1. The Influence of Vitamins on MSC Fate
4.2. The Role of Trace Elements in MSC Maintenance
| Substance | Effect on MSCs | Concentration | Cell Type/Model | Refs. |
|---|---|---|---|---|
| Vitamin C | ↑ Proliferation, self-renewal, expression of pluripotency genes (Nanog, Oct4, Sox2) ↓ Senescence (reduced SA-β-gal+ cells, p16 suppression) Growth inhibition/apoptosis at higher doses. | 10–250 µM (stimulation), >250 µM (inhibition), 200 µM (anti-aging effect) | Gingival stem cells (GSCs); Rat MSCs | [8,56] |
| Vitamin D | ↑ Expression of pluripotency genes (Nanog, Sox2, Oct4), resistance to dysfunction ↓ Number of senescent cells, p16 expression Proliferation suppression at deficient and excessive doses. | 1 × 10−7 M (with Vit. E) 0 IU/kg or 1000 IU/kg (suppression), 250–500 IU/kg (optimal) | Bone marrow-derived MSCs (BM-MSCs); Rat lung-derived MSCs (in vivo model) | [17,54,58,60,61] |
| Vitamin E | ↓ Apoptosis (via AKT pathway modulation) ↑ Expression of proliferative markers ↓ Proliferation (in combination with Vit. D) | 12 µM (with vit. D) | Human dental pulp stem cells (hDPSCs); Gingiva-derived stem cells (GSCs) | [9,77,78,79] |
| Vitamin B3 | ↑ MSC survival ↓ Delays replicative senescence | 5 mM | Human BM-MSCs | [65] |
| Magnesium (Mg2+) | ↑ Viability, proliferation, adhesion. Inhibition/cytotoxicity at high concentrations. | 1.2–1.8 mM (MgCl2), 5.7–8.7 mM (Mg2+), >20 mM (MgSO4) (inhibition), 32.1 mM (cytotoxicity) | Human MSCs; Rat bone marrow MSCs | |
| Zinc (Zn2+) | ↑ Proliferation, expression of pluripotency genes (Oct4, Sox2, Nanog) and telomerase (TERT). Cytotoxicity at higher concentrations. | 20 µM (optimal) >100 µM (cytotoxicity) | Umbilical cord-derived MSCs (hUC-MSCs) | [10] |
| Selenium (Se) | ↑ Viability ↓ Senescence. Reduced viability at higher concentrations | 50 ng/mL (Se nanoparticles) 0.1 µM (sodium selenite), 300 ng/mL (reduced viability) | Rat bone marrow MSCs | [49,69] |
| Iron (Fe) | ↑ Proliferation (as FeSO4) ↓ Proliferation and senescence (as iron oxide nanoparticles) | 4 × 10−8 M (FeSO4), 100 µg/mL (nanoparticles) | Adipose-derived MSCs; BM-MSCs | [71,72] |
| Calcium (Ca2+) | ↑ Proliferation/survival | 0.01–100 mg/mL (stimulation), 1 mg/mL (optimal), 4–10 mM (stimulation), 6 mM (optimal), >6 mM (inhibition); As calcium phosphate nanoparticle—not specified | In vitro: Human stem cells of the apical papilla (SCAPs); BM-MSCs | [73,75,76,80] |
5. Modulation and Control of MSC Differentiation by Vitamins and Minerals
5.1. Modulation of Osteogenesis
5.2. Regulation of Adipogenesis and Chondrogenesis
5.3. Influence of Inorganic Ions on MSC Differentiation
| Substance | Differentiation | Effect and Mechanism | Concentration | Cell Type/Model | Refs. |
|---|---|---|---|---|---|
| Vitamin C | Osteogenesis | ↑ Restores osteogenic potential in senescent MSCs by activating telomerase. Enhances mineralization and expression of RUNX2, ALP, COL1A1. Essential for collagen synthesis, which in turn upregulates Runx2 | 50 µg/mL | Senescent human BM-MSCs; ASCs | [32,57,85] |
| Adipogenesis | ↓ Suppresses differentiation into adipocytes | 250–500 μM, 150 μM (optimal) | Mouse embryonic mesoderm-derived mesenchymal cells | [81] | |
| Chondrogenesis | ↑ Enhances differentiation and protects chondrocytes from oxidative stress (H2O2) | 50 μg/mL (~170 μM) (ADSCs); 50 μmol/L (GSCs) | Glial-derived stem cells (GSCs); Adipose-derived MSCs (ADSCs) | [8,98] | |
| Vitamin D | Osteogenesis | ↑ Enhances expression of ALP, OCN, RUNX2 via the vitamin D receptor (VDR). Synergizes with BMP-2, TGF-β1, metformin, and dexamethasone. Activates Wnt/β-catenin and BMP2 signaling. Reduces ROS | 20 nM (peak effect) | Bone marrow MSCs (BM-MSCs) | [49,50,61,87,89,92,93] |
| Adipogenesis | ↓ Reduces lipid accumulation. Downregulates adipocyte-specific genes by inhibiting the transcription factor PPARγ2 | 20 nM (strongest inhibition) | Human BM-MSCs | [50,95,96,97] | |
| Vitamin E | Osteogenesis | ↑ Enhances osteogenic commitment, particularly in combination with vitamin D (0.1 μM) | 12 μM (hDPSCs); 6.06 μg/mL α-tocoferol (concentration in human serum used in BM-MSCs culture) | Human dental pulp stem cells (hDPSCs); BM-MSCs | [9,82] |
| Vitamin K2 | Osteogenesis | ↑ Synergistically with vitamin D3 (10 nM) enhances the expression of osteocalcin, osterix, and Runx2 | 10 nM | Diet-induced obese mouse models (primary osteoblasts) | [110] |
| Magnesium | Osteogenesis | ↑ Enhances early differentiation via Notch and Wnt signaling and autophagy. High concentrations may impair late-stage mineralization | 5–10 mM (enhances) <1.3 mM (favors) 0.1 mM (stimulates autophagy) | Rat and human BM-MSCs | [67,68,100,101,102,103] |
| Chondrogenesis | ↑ Supports differentiation and synthesis of cartilage-specific extracellular matrix via integrin-mediated signaling | ~5 mM | BM-MSCs; Synovial MSCs | [104] | |
| Calcium | Osteogenesis | ↑ Upregulates OCN, BSP, ALP expression. Mediated through L-type calcium channels and CaMKII signaling | Not specified—used as calcium phosphate biomaterials surface | Human BM-MSCs; hMSC immunoselected with STRO1 antibody; rabbit BM-MSC | [111,112,113] |
| Zinc | Osteogenesis | ↑ Enhances osteoblast differentiation, matrix formation, and mineralization | Not specified—used as MOF nanoparticles | Adipose-derived MSCs | [109] |
| Selenium | Osteogenesis | ↑ Shifts MSC fate towards osteogenic lineage. Increases ALP activity and osteogenic gene transcription | 25–100 ng/mL (50 ng/mL optimal) | hESC-derived MSCs | [49] |
| Adipogenesis | ↓ Suppresses adipocyte differentiation | 25–100 ng/mL | hESC-derived MSCs | [49] |
6. Vitamins and Micronutrients in MSC-Mediated Tissue Regeneration and Remodeling
6.1. Vitamins Enhancing Tissue Regeneration
6.2. Mineral Compounds in MSC-Mediated Tissue Regeneration
| Substance | Tissue Effect | Mechanism | Dose | Cell Type/Model | Refs. |
|---|---|---|---|---|---|
| Vitamin C | ↑ Enhances bone defect regeneration and tissue repair | Acts as a cofactor for collagen synthesis; supports extracellular matrix (ECM) formation and deposition; improves blood perfusion and neovascularization. | 200 μM (cell culture media component); 50 μg/mL (regeneration/tissue engineering); 0.05–0.2 mM (collagen synthesis cofactor) | In vivo ischemic limb models in mice (SCB-MSCs); ASCs | [13,32,55,71,91,94,114,115,125,126,127,128] |
| Vitamin D | ↑ Promotes bone regeneration, vascular repair, and lung development | Promotes osteogenic differentiation; modulates the immune environment; improves endothelial progenitor cell (EPC) adhesion and migration; alleviates TNF-α-mediated inflammation. | 250 and 500 IU/kg (optimal dietary doses) | Rat models (Perinatal dietary supplementation); lung MSCs | [50,60,116,117] |
| Vitamin E | ↑ Enhances MSC survival and cartilage regeneration | Preconditioning protects MSCs from oxidative stress (H2O2); upregulates TGF-β; downregulates apoptosis genes; reduces VEGF and LDH release; increases proteoglycan content in cartilage. | 100 μM | In vitro preconditioning of MSCs; In vivo: osteoarthritis rat models. | [77,118,119] |
| Vitamin B6 | ↑ Accelerates clearance of MSCs from the body | Led to lower numbers of detectable ucMSCs 4 h post-injection, especially when combined with retinoic acid. | 1 μM | Murine model of hepatitis (mice treated with human ucMSCs) | [120] |
| Magnesium | ↑ Supports bone homeostasis and cartilage regeneration | Enhances MSC adhesion to defects; promotes cartilage matrix synthesis; induces apatite crystal growth on scaffolds for mineralization. | 5 mM MgCl2 (in PBS)— in vivo rabbit model; 1 mM and 10 mM MgCl2 (in PBS)— ex vivo studies; 5 mM and 10 mM MgCl2 (in PBS)— in vitro studies. Not specified (used as Mg-BCP micro-scaffolds prepared in terms of 0.01 Mg/Ca and 1.602 of (Ca+Mg)/P mole ratios) (human AT-MSCs). | In vivo: using a rabbit osteochondral defect model (rabbit synovial MSCs); Ex vivo: with osteochondral tissue (human synovial MSCs); In vitro: chondrogenesis assessment, cell adhesion (human synovial MSCs); In vitro: studies with human AT-MSCs. | [104,121] |
| Zinc | ↑ Promotes tissue repair by enhancing cell homing | Significantly improves MSC migration and adhesion; scaffolds co-doped with copper promote both osteogenesis and angiogenesis. | 5–100 μM (stimulation), 20 μM (optimal), >250 μM (inhibition); Zinc-containing scaffolds: PLLA@MOF containing 0.30 mol of ZnO | In vitro scratch assays (hUC-MSCs); Human adipose tissue-derived MSCs | [10,109] |
| Iron (IONPs) | ↑ Improves wound healing, and reduces liver fibrosis | Enhances migration of MSCs to injury sites, leading to improved angiogenesis. | 15 μg/L nanoparticles of AuFe 3 μg/mL of ps-TNCs Dose not specified— 100 μg of Fe used for labeling 1 × 106 MSCs | Mouse model of skin wounds; Mouse hindlimb ischemia model (angiogenesis) (used human ASCs); Rat models liver fibrosis (rat BM-MSCs) | [122,123,124] |
7. Modulation of MSC Immunoregulatory Functions by Vitamins and Minerals
Immunomodulation by Micronutrients
| Substance | Effect | Mechanism | Concentration | Cell Type/Model | Refs. |
|---|---|---|---|---|---|
| Vitamin C | ↓ Reduces pro-inflammatory phenotype | Suppresses Senescence-Associated Secretory Phenotype (SASP), reducing TNF-α, IL-1β, IL-6, IL-8. Supports activation of NK cells and cytotoxic T lymphocytes. | 200 µmol/L | SCS-MSCs (cells with prelamin A overexpression—MSC/PLA) | [115] |
| Vitamin D | ↓ Strong anti-inflammatory effects | Reduces secretion of TNF-α, IL-1β, IL-6 by blocking NF-κB signaling. Enhances MSC-mediated suppression of CD4+ T cell proliferation. Suppresses M1 macrophage differentiation. Promotes M2 macrophage differentiation. Inhibits M1 macrophage-mediated MSC migration in a dose-dependent manner. | 100 nM 1.25(OH)2D3 10 nM and 100 nM 100 and 1000 ng/kg, s.c. detrimental effect during proinflammatory stage, neutral effect during regenerative phase 15,000 IU/kg b.w. 3000 IU/kg diet | Human periodontal ligament stem cells (hPDLSCs); In vitro: mouse BM-MSCs; In vivo: studies with mice (bone fracture healing); In vivo: mouse acute model, LPS; In vivo: mouse chronic model, HF diet | [42,130,132] |
| Vitamin B6 | ↑ Enhances immunogenicity and immunomodulatory potential | Increases surface expression of HLA classes I and II. Upregulates PD-L1 and markedly increases IL-1RA mRNA levels. | 1 µM | Human umbilical cord-derived MSCs (ucMSCs) | [120] |
| Retinoic Acid | ↑ Enhances immunosuppression (but can be pro-inflammatory) | Suppresses CD4+ and CD8+ T cell proliferation. Decrease PD-L1 expression (with Vit B6). Significantly increase HLA class I and HLA class II expression. Reduces TNF-α secretion. In another context, it activates the pro-inflammatory NF-κB/NLRP3 axis: promotes the production of the proinflammatory cytokine interleukin-1β. | 1 µM or 10 µM (effect achieved when RA was part of the MC cocktail) ucMSCs pre-treated with 1 µM or 10 µM RA—no effect 1 µM or 10 µM—limited effect (only TNF-α inhibition) 100 nM, 1 µM, 10 µM (proinflammatory effect) | In vitro: human umbilical cord-derived MSCs (ucMSCs) In vivo: liver disease mouse model Ex vivo: liver inflammation co-culture model Human periodontal ligament stem cells (hPDLSCs) | [120,135] |
| Magnesium | ↓ Strong anti-inflammatory and immunosuppressive effects | Reduces IL-1β, IL-6; increases IL-10, PGE2. Modulates NF-κB and STAT3. | 5 mM | Murine MSCs; Macrophages (via conditioned medium) | [136] |
| Zinc | ↓ Exerts anti-inflammatory effects | Upregulates genes for cytokine receptor interactions and IL-17/TNF pathways. Deficiency increases TNF-α, IL-1β, IL-8. | 5 µmol/L | Human umbilical cord-derived MSCs (hUC-MSCs) | [137] |
| Iron (IONPs) | ↓ Enhances anti-inflammatory properties | Shifts cytokine profile to reduce pro-inflammatory factors (IL-2, TNF-α) and increase secretion of anti-inflammatory IL-4, IL-10) cytokines. | 50 μg/mL of Fe3O4@PDA nanoparticles 1 × 106 MSCs labeled with Fe3O4@PDA nanoparticles | In vitro: rat BM-MSCs In vivo: laser burn wound rat model | [138] |
8. Antioxidant Regulation and Redox Homeostasis in MSC Biology
The Role of Vitamins in Redox Regulation
| Substance | Effect/Role | Mechanism | Concentration | Cell Type/Model | Refs. |
|---|---|---|---|---|---|
| Vitamin C | Potent antioxidant, reduces ROS | Scavenges ROS; inhibits ROS production via AKT/mTOR axis. | 200 µmol/L | Adipose-tissue MSCs | [14,32,55,125,155] |
| Vitamin D | Maintains redox balance | Activates VDR to upregulate SOD2. Preserves mitochondrial function. | 100 nmol/L 1,25(OH)2D (ROS reduction model); 1–100 nM (SOD2 dose–response) 0.1 µg/kg 1,25(OH)2D i.p. | In vitro: human and mouse BM-MSCs In vivo: HFD-induced osteoporosis model in mice | [59,61] |
| Vitamin E | Protects from oxidative stress | Prevents lipid peroxidation of cell membranes. Reduces oxidative stress and aging caused by hydrogen peroxide (H2O2). Maintenance of cell membrane integrity. Protects against apoptosis and promotes survival, enhances proliferation. | 50 µM 500 µM 50 µM and 100 µM (in vitro) Rat BM-MSCs pretreated with 100 µM | In vitro: Human DPSCs; porcine ASCs; rat BM-MSCs In vivo: osteoarthritis (OA) rat model | [77,78,145] |
| Selenium (SeNPs) | Protects against oxidative stress | Activates JNK/FOXO3a pathway, increasing SOD and catalase. | 50 ng/mL (optimal) >100 ng/mL (cytotoxic) | hESC-derived MSCs; rBM-MSCs | [49,141,144] |
| Zinc | Enhances antioxidant defense | Modulates Nrf2/Sirt3 pathway to promote antioxidant gene transcription. | 5–100 µM | Human UC-MSCs | [137,151] |
| Iron | Dualistic: adaptive vs. toxic | Low concentrations: mild ROS for adaptive response (HIF-1α stimulation). High concentrations: toxic ROS. | Adaptive: 3 μg/mL and 50 μg/mL. Toxic (high ROS/damage): >3 μg/mL; 15.4 μg/mL; >50 μg/mL | Human ADSCs; Human BM-MSCs; Rat BM-MSCs | [122,152,153,154] |
9. Epigenetic Regulation of Genomic Stability in Mesenchymal Stem Cells by Vitamins and Minerals
| Substance | Role/Effect | Mechanism | Concentration | Cell Type/Model | Refs. |
|---|---|---|---|---|---|
| Vitamin C | Directly modulates DNA and histone demethylation | Serves as a cofactor for TET enzymes, promoting active DNA demethylation (5mC → 5hmC). Supports Jumonji (JHDM) histone demethylases (e.g., of H3K36me2/3), leading to increased c-Myc/Klf4 and repression of p21. | 200–250 µM | Gingival stem cells; mouse ASC | [8,126,159] |
| Vitamin B3 | Regulates histone deacetylation and telomere maintenance | Activates SIRT1, a NAD+-dependent histone deacetylase, which delays senescence and supports differentiation. | 5 mM | Mouse adipose-derived MSCs | [65] |
| Zinc (Zn2+) | Regulates histone deacetylation and telomeric stability | Activates SIRT3 (histone deacetylase). Acts as a structural component for zinc finger transcription factors and chromatin remodelers. Increases TERT expression. | 0.14 µg/mL ZnSO4 | Rat adipose-derived MSCs | [160] |
| Selenium | Supports methylation cycle and chromatin integrity | As a component of selenoproteins, it ensures the availability of S-adenosylmethionine (SAM), the universal methyl donor for DNMTs and HMTs. Reduces DNA damage indicators (micronuclei) by up to 58%. | 100 nM sodium selenite | Human BM-MSCs | [140] |
10. Regulation of MSC Activity by Phytochemicals
| Substance | Effect | Mechanism | Concentration | Cell Type/Model | Refs. |
|---|---|---|---|---|---|
| Epigallocatechin gallate (EGCG)—polyphenol (green tea) | Protects against oxidative stress and cell aging, and increases proliferation and osteogenesis | Activation of the Nrf2 pathway, decrease in p53/p21 acetylation; enhancement of Wnt/β-catenin, increase in cyclin D1 | 10–50 µM | hMSC, BM-MSC (human) | [161,162,165] |
| Curcumin— polyphenol (Curcuma longa) | Protects MSCs from oxidative stress, reduces apoptosis, supports differentiation | Inhibition of ROS and NO production, maintenance of antioxidant enzyme activity | 5–20 µM | hMSCs (human) | [164] |
| Allicin— an organosulfur compound found in garlic | Enhance osteogenesis and bone remodeling | Increases expression of ALP, BGLAP, RANKL, OPG; activates osteoblast cells. Allicin released from bioceramic scaffolds (amount depends on the material). | Allicin released from bioceramic scaffolds (amount depends on the material) | Co-culture of hMSCs + monocytes, in vivo, rat model | [166] |
| Resveratrol— polyphenol (grapes, peanuts, blueberries) | Increases osteoblast proliferation and differentiation | Activation of the ERK1/2 pathway via the estrogen receptor; involvement of p38 MAPK. | 1–10 µM | hBMSC (human) | [167] |
| Coenzyme Q10 (ubiquinone)—lipophilic quinone | Inhibits MSC aging, reduces oxidative stress and expression of aging genes (p53, p21, p16) | Reduction of ROS, inhibition of the Akt/mTOR pathway, regulation of PPARγ expression and antioxidant enzymes. | 1–100µM (D-galactose-induced aging) | hMSC (human) | [172] |
| Melatonin— indoleamine (endogenous, plant-derived) | Increases survival, proliferation, and paracrine activity of MSCs, reduces ER and mitochondrial stress | TGF-β activation, PI3K/Akt, antioxidant and anti-inflammatory effects | 0–10 µM (24 h) | ADMSC, NPMSC (human) | [171,175,176] |
11. Critical Assessment and Translational Challenges in the Application of Bioactive Compounds for Mesenchymal Stem Cell Therapy
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALP | Alkaline phosphatase |
| BMP2 | Bone morphogenetic protein 2 |
| CaMKII | Calcium/calmodulin-dependent protein kinase II |
| CD | Cluster of differentiation |
| COL1A1 | Collagen type I alpha 1 chain |
| ECM | Extracellular matrix |
| EGCG | Epigallocatechin gallate |
| ESC | Embryonic stem cell |
| hBM-MSCs | Human bone marrow-derived mesenchymal stem cells |
| hESC | Human embryonic stem cell |
| hUC-MSCs | Human umbilical cord-derived mesenchymal stem cells |
| hUM-MSCs | Human umbilical cord matrix-derived mesenchymal stem cells |
| IL-1RA | Interleukin-1 receptor antagonist |
| IONPs | Iron oxide nanoparticles |
| LDH | Lactate dehydrogenase |
| MSC | Mesenchymal stem cell |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| Notch | Notch signaling pathway |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| OCN | Osteocalcin |
| PD-L1 | Programmed death-ligand 1 |
| PGE2 | Prostaglandin E2 |
| PPARγ2 | Peroxisome proliferator-activated receptor gamma 2 |
| RA | Retinoic acid |
| ROS | Reactive oxygen species |
| RSVL | Resveratrol |
| RUNX2 | Runt-related transcription factor 2 |
| SIRT1 | Sirtuin 1 |
| SIRT3 | Sirtuin 3 |
| STAT3 | Signal transducer and activator of transcription 3 |
| TET | Ten-eleven translocation |
| TGF-β | Transforming growth factor-beta |
| TNF-α | Tumor necrosis factor-alpha |
| VDR | Vitamin D receptor |
| VEGF | Vascular endothelial growth factor |
| Wnt | Wnt signaling pathway |
References
- Weatherbee, B.A.T.; Cui, T.; Zernicka-Goetz, M. Modeling Human Embryo Development with Embryonic and Extra-Embryonic Stem Cells. Dev. Biol. 2021, 474, 91–99. [Google Scholar] [CrossRef]
- Musiał-Wysocka, A.; Kot, M.; Majka, M. The Pros and Cons of Mesenchymal Stem Cell-Based Therapies. Cell Transplant. 2019, 28, 801–812. [Google Scholar] [CrossRef]
- Musiał-Wysocka, A.; Kot, M.; Sułkowski, M.; Badyra, B.; Majka, M. Molecular and Functional Verification of Wharton’s Jelly Mesenchymal Stem Cells (WJ-MSCs) Pluripotency. Int. J. Mol. Sci. 2019, 20, 1807. [Google Scholar] [CrossRef]
- Huang, Y.; Wu, Q.; Tam, P.K.H. Immunomodulatory Mechanisms of Mesenchymal Stem Cells and Their Potential Clinical Applications. Int. J. Mol. Sci. 2022, 23, 10023. [Google Scholar] [CrossRef]
- Musiał-Wysocka, A.; Kot, M.; Sułkowski, M.; Majka, M. Regenerative Potential of the Product “CardioCell” Derived from the Wharton’s Jelly Mesenchymal Stem Cells for Treating Hindlimb Ischemia. Int. J. Mol. Sci. 2019, 20, 4632. [Google Scholar] [CrossRef] [PubMed]
- Brandhorst, S.; Choi, I.Y.; Wei, M.; Cheng, C.W.; Sedrakyan, S.; Navarrete, G.; Dubeau, L.; Yap, L.P.; Park, R.; Vinciguerra, M.; et al. A Periodic Diet That Mimics Fasting Promotes Multi-System Regeneration, Enhanced Cognitive Performance, and Healthspan. Cell Metab. 2015, 22, 86–99. [Google Scholar] [CrossRef]
- Saud, B.; Malla, R.; Shrestha, K. A Review on the Effect of Plant Extract on Mesenchymal Stem Cell Proliferation and Differentiation. Stem Cells Int. 2019, 2019, 7513404. [Google Scholar] [CrossRef]
- Van Pham, P.; Tran, N.Y.; Phan, N.L.-C.; Vu, N.B.; Phan, N.K. Vitamin C Stimulates Human Gingival Stem Cell Proliferation and Expression of Pluripotent Markers. Vitr. Cell. Dev. Biol.—Anim. 2016, 52, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Escobar, L.M.; Bendahan, Z.; Bayona, A.; Castellanos, J.E.; González, M.-C. Effect of Vitamins D and E on the Proliferation, Viability, and Differentiation of Human Dental Pulp Stem Cells: An In Vitro Study. Int. J. Dent. 2020, 2020, 8860840. [Google Scholar] [CrossRef] [PubMed]
- Sahibdad, I.; Khalid, S.; Chaudhry, G.R.; Salim, A.; Begum, S.; Khan, I. Zinc Enhances the Cell Adhesion, Migration, and Self-Renewal Potential of Human Umbilical Cord Derived Mesenchymal Stem Cells. World J. Stem Cells 2023, 15, 751–767. [Google Scholar] [CrossRef]
- Khillan, J. Vitamin A/Retinol and Maintenance of Pluripotency of Stem Cells. Nutrients 2014, 6, 1209–1222. [Google Scholar] [CrossRef]
- Zipori, D. The Nature of Stem Cells: State Rather than Entity. Nat. Rev. Genet. 2004, 5, 873–878. [Google Scholar] [CrossRef] [PubMed]
- D’Aniello, C.; Cermola, F.; Patriarca, E.J.; Minchiotti, G. Vitamin C in Stem Cell Biology: Impact on Extracellular Matrix Homeostasis and Epigenetics. Stem Cells Int. 2017, 2017, 8936156. [Google Scholar] [CrossRef] [PubMed]
- Lee Chong, T.; Ahearn, E.L.; Cimmino, L. Reprogramming the Epigenome with Vitamin C. Front. Cell Dev. Biol. 2019, 7, 128. [Google Scholar] [CrossRef]
- Hart, D.A. The Use of MSCs, iPSCs, and EVs in the Repair of Human MSK Tissues: Is Ultimate Success Dependent on Developing Excellent Implant Materials as Well as Creating an Optimal Environment for Implantation? What Is the Rationale for These Choices? Int. J. Mol. Sci. 2025, 26, 6250. [Google Scholar] [CrossRef]
- Zong, Q.; Bundkirchen, K.; Neunaber, C.; Noack, S. Are the Properties of Bone Marrow-Derived Mesenchymal Stem Cells Influenced by Overweight and Obesity? Int. J. Mol. Sci. 2023, 24, 4831. [Google Scholar] [CrossRef]
- Godoy-Parejo, C.; Deng, C.; Zhang, Y.; Liu, W.; Chen, G. Roles of Vitamins in Stem Cells. Cell. Mol. Life Sci. 2020, 77, 1771–1791. [Google Scholar] [CrossRef] [PubMed]
- Fekete, N.; Rojewski, M.T.; Lotfi, R.; Schrezenmeier, H. Essential Components for Ex Vivo Proliferation of Mesenchymal Stromal Cells. Tissue Eng. Part C Methods 2014, 20, 129–139. [Google Scholar] [CrossRef]
- Adefegha, S.A. Functional Foods and Nutraceuticals as Dietary Intervention in Chronic Diseases; Novel Perspectives for Health Promotion and Disease Prevention. J. Diet. Suppl. 2018, 15, 977–1009. [Google Scholar] [CrossRef]
- Evans, N.; Swain, R.; Gentleman, E.; Gentleman, M.; Stevens, M. Gene-Expression Analysis Reveals That Embryonic Stem Cells Cultured under Osteogenic Conditions Produce Mineral Non-Specifically Compared to Marrow Stromal Cells or Osteoblasts. Eur. Cell. Mater. 2012, 24, 211–223. [Google Scholar] [CrossRef]
- Bacakova, L.; Zarubova, J.; Travnickova, M.; Musilkova, J.; Pajorova, J.; Slepicka, P.; Kasalkova, N.S.; Svorcik, V.; Kolska, Z.; Motarjemi, H.; et al. Stem Cells: Their Source, Potency and Use in Regenerative Therapies with Focus on Adipose-Derived Stem Cells—A Review. Biotechnol. Adv. 2018, 36, 1111–1126. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Peregrina, K.; Houston, M.; Augenlicht, L.H. Vitamin D and the Nutritional Environment in Functions of Intestinal Stem Cells: Implications for Tumorigenesis and Prevention. J. Steroid Biochem. Mol. Biol. 2020, 198, 105556. [Google Scholar] [CrossRef] [PubMed]
- Bruno, A.; Milillo, C.; Anaclerio, F.; Buccolini, C.; Dell’Elice, A.; Angilletta, I.; Gatta, M.; Ballerini, P.; Antonucci, I. Perinatal Tissue-Derived Stem Cells: An Emerging Therapeutic Strategy for Challenging Neurodegenerative Diseases. Int. J. Mol. Sci. 2024, 25, 976. [Google Scholar] [CrossRef] [PubMed]
- Kassem, M.; Kristiansen, M.; Abdallah, B.M. Mesenchymal Stem Cells: Cell Biology and Potential Use in Therapy. Basic Clin. Pharmacol. Toxicol. 2004, 95, 209–214. [Google Scholar] [CrossRef]
- Meirelles, L.D.S.; Chagastelles, P.C.; Nardi, N.B. Mesenchymal Stem Cells Reside in Virtually All Post-Natal Organs and Tissues. J. Cell Sci. 2006, 119, 2204–2213. [Google Scholar] [CrossRef]
- Jing, W.; Zhiguo, W.; Xia, C.; Kun, L.; Rongan, H.; Yu, A. Research Progress on Exosomes Derived from Human Adipose Mesenchymal Stem Cells. Int. J. Sci. 2019, 8, 114–117. [Google Scholar] [CrossRef]
- Bunnell, B.A. Adipose Tissue-Derived Mesenchymal Stem Cells. Cells 2021, 10, 3433. [Google Scholar] [CrossRef]
- Zhang, P.; Cui, Y.; Li, Z.; Liu, L.; Liu, X.; Ding, X.; Ding, G. Senescence of Dental Pulp Stem Cells: Phenotypes, Underlying Mechanisms and Regulatory Molecules. Hum. Cell 2025, 38, 127. [Google Scholar] [CrossRef]
- Beeravolu, N.; McKee, C.; Alamri, A.; Mikhael, S.; Brown, C.; Perez-Cruet, M.; Chaudhry, G.R. Isolation and Characterization of Mesenchymal Stromal Cells from Human Umbilical Cord and Fetal Placenta. J. Vis. Exp. 2017, 122, 55224. [Google Scholar] [CrossRef]
- Kwiecien, E.; Kot, M.; Czyz, L.; Drabik, L.; Mazurek, A.; Sikorska, M.; Skubera, M.; Tekieli, L.; Majka, M.; Musialek, P. Umbilical Cord Matrix (Wharton Jelly) Mesenchymal Stem Cells in Next-Generation Myocardial Repair and Regeneration: Mechanisms and Pre-Clinical Evidence. Curr. Cardiol. Rev. 2025, 21, e1573403X372908. [Google Scholar] [CrossRef] [PubMed]
- Seyyedin, S.; Ezzatabadipour, M.; Nematollahi-Mahani, S.N. The Role of Various Factors in Neural Differentiation of Human UmbilicalCord Mesenchymal Stem Cells with a Special Focus on the Physical Stimulants. Curr. Stem Cell Res. Ther. 2024, 19, 166–177. [Google Scholar] [CrossRef]
- Shotorbani, B.B.; André, H.; Barzegar, A.; Zarghami, N.; Salehi, R.; Alizadeh, E. Cell Sheet Biofabrication by Co-Administration of Mesenchymal Stem Cells Secretome and Vitamin C on Thermoresponsive Polymer. J. Mater. Sci. Mater. Med. 2018, 29, 170. [Google Scholar] [CrossRef]
- Wang, S.-H.; Lee, S.-P.; Yang, C.-W.; Lo, C.-M. Surface Modification of Biodegradable Mg-Based Scaffolds for Human Mesenchymal Stem Cell Proliferation and Osteogenic Differentiation. Materials 2021, 14, 441. [Google Scholar] [CrossRef]
- Pacheco, C.M.R.; Ferreira, P.E.; Saçaki, C.S.; Tannous, L.A.; Zotarelli-Filho, I.J.; Guarita-Souza, L.C.; Carvalho, K.A.T.D. In Vitro Differentiation Capacity of Human Breastmilk Stem Cells: A Systematic Review. World J. Stem Cells 2019, 11, 1005–1019. [Google Scholar] [CrossRef] [PubMed]
- Rahmani-Moghadam, E.; Zarrin, V.; Mahmoodzadeh, A.; Owrang, M.; Talaei-Khozani, T. Comparison of the Characteristics of Breast Milk-Derived Stem Cells with the Stem Cells Derived from the Other Sources: A Comparative Review. Curr. Stem Cell Res. Ther. 2022, 17, 71–90. [Google Scholar] [CrossRef] [PubMed]
- Kaingade, P.M.; Somasundaram, I.; Nikam, A.B.; Sarang, S.A.; Patel, J.S. Assessment of Growth Factors Secreted by Human Breastmilk Mesenchymal Stem Cells. Breastfeed. Med. 2016, 11, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Abd Allah, S.H.; Shalaby, S.M.; El-Shal, A.S.; El Nabtety, S.M.; Khamis, T.; Abd El Rhman, S.A.; Ghareb, M.A.; Kelani, H.M. Breast Milk MSCs: An Explanation of Tissue Growth and Maturation of Offspring: Breast Milk MSCs. IUBMB Life 2016, 68, 935–942. [Google Scholar] [CrossRef]
- Hosseini, S.M.; Talaei-khozani, T.; Sani, M.; Owrangi, B. Differentiation of Human Breast-Milk Stem Cells to Neural Stem Cells and Neurons. Neurol. Res. Int. 2014, 2014, 807896. [Google Scholar] [CrossRef]
- Chen, T.; Jiang, Y.; Xu, S.; Cheuk, Y.C.; Wang, J.; Yang, C.; Rong, R. Poly(I:C)-Induced Mesenchymal Stem Cells Protect the Kidney Against Ischemia/Reperfusion Injury via the TLR3/PI3K Pathway. Front. Med. 2021, 8, 755849. [Google Scholar] [CrossRef]
- Johnson, V.; Webb, T.; Norman, A.; Coy, J.; Kurihara, J.; Regan, D.; Dow, S. Activated Mesenchymal Stem Cells Interact with Antibiotics and Host Innate Immune Responses to Control Chronic Bacterial Infections. Sci. Rep. 2017, 7, 9575. [Google Scholar] [CrossRef]
- Nauta, A.J.; Fibbe, W.E. Immunomodulatory Properties of Mesenchymal Stromal Cells. Blood 2007, 110, 3499–3506. [Google Scholar] [CrossRef] [PubMed]
- Wasnik, S.; Rundle, C.H.; Baylink, D.J.; Yazdi, M.S.; Carreon, E.E.; Xu, Y.; Qin, X.; Lau, K.-H.W.; Tang, X. 1,25-Dihydroxyvitamin D Suppresses M1 Macrophages and Promotes M2 Differentiation at Bone Injury Sites. JCI Insight 2018, 3, e98773. [Google Scholar] [CrossRef] [PubMed]
- Hanson, S.E.; Bentz, M.L.; Hematti, P. Mesenchymal Stem Cell Therapy for Nonhealing Cutaneous Wounds. Plast. Reconstr. Surg. 2010, 125, 510–516. [Google Scholar] [CrossRef]
- Chade, A.R. VEGF: Potential Therapy for Renal Regeneration. F1000 Med. Rep. 2012, 4, 1. [Google Scholar] [CrossRef]
- Soetartio, I.M.; Damayanti, T. Mesenchymal Stem Cells and Clinical Application in Chronic Lung Diseases. J. Respirasi 2021, 7, 145. [Google Scholar] [CrossRef]
- Ryan, J.M.; Barry, F.P.; Murphy, J.M.; Mahon, B.P. Mesenchymal Stem Cells Avoid Allogeneic Rejection. J. Inflamm. 2005, 2, 8. [Google Scholar] [CrossRef]
- Department of Rheumatology and Immunology, No.3. People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai, China; Zhao, F.; Yin, J.; Liu, Q.; Xu, S. Effect of Bone Marrow Mesenchymal Stem Cell Transplant on Synovial Proliferation in Rats with Type II Collagen-Induced Arthritis. Exp. Clin. Transplant. 2013, 11, 352–357. [Google Scholar] [CrossRef]
- Babaei, S.; Mirzaei, M.; Maleki, N.; Alizadeh, Z.; Yasari, F.; Novin, M.G. Isolation and Evaluation of Biological Properties and Differentiation Potential of Human Umbilical Cord-Derived Mesenchymal Stem Cells in Covid-19 Positive Patients. LAPLAGE EM Rev. 2021, 7, 542–551. [Google Scholar] [CrossRef]
- Fatima, S.; Alfrayh, R.; Alrashed, M.; Alsobaie, S.; Ahmad, R.; Mahmood, A. Selenium Nanoparticles by Moderating Oxidative Stress Promote Differentiation of Mesenchymal Stem Cells to Osteoblasts. Int. J. Nanomed. 2021, 16, 331–343. [Google Scholar] [CrossRef]
- Borojević, A.; Jauković, A.; Kukolj, T.; Mojsilović, S.; Obradović, H.; Trivanović, D.; Živanović, M.; Zečević, Ž.; Simić, M.; Gobeljić, B.; et al. Vitamin D3 Stimulates Proliferation Capacity, Expression of Pluripotency Markers, and Osteogenesis of Human Bone Marrow Mesenchymal Stromal/Stem Cells, Partly through SIRT1 Signaling. Biomolecules 2022, 12, 323. [Google Scholar] [CrossRef]
- Sharifi, S.; Moghaddam, F.A.; Abedi, A.; Maleki Dizaj, S.; Ahmadian, S.; Abdolahinia, E.D.; Khatibi, S.M.H.; Samiei, M. Phytochemicals Impact on Osteogenic Differentiation of Mesenchymal Stem Cells. BioFactors 2020, 46, 874–893. [Google Scholar] [CrossRef]
- Cao, N.; Liu, Z.; Chen, Z.; Wang, J.; Chen, T.; Zhao, X.; Ma, Y.; Qin, L.; Kang, J.; Wei, B.; et al. Ascorbic Acid Enhances the Cardiac Differentiation of Induced Pluripotent Stem Cells through Promoting the Proliferation of Cardiac Progenitor Cells. Cell Res. 2012, 22, 219–236. [Google Scholar] [CrossRef] [PubMed]
- Potdar, P.D.; D’Souza, S.B. Ascorbic Acid Induces in Vitro Proliferation of Human Subcutaneous Adipose Tissue Derived Mesenchymal Stem Cells with Upregulation of Embryonic Stem Cell Pluripotency Markers Oct4 and SOX 2: Ascorbic Acid Induces Pluripotency. Hum. Cell 2010, 23, 152–155. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.-C.; Lu, C.-L.; Zheng, C.-M.; Liu, W.-C.; Yen, T.-H.; Chen, R.-M.; Lin, Y.-F.; Chao, C.-T.; Lu, K.-C. The Role of Vitamin D in Modulating Mesenchymal Stem Cells and Endothelial Progenitor Cells for Vascular Calcification. Int. J. Mol. Sci. 2020, 21, 2466. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Teng, S.; Ma, C.; Yu, Y.; Wang, P.; Yi, C. Ascorbic Acid Inhibits Senescence in Mesenchymal Stem Cells through ROS and AKT/mTOR Signaling. Cytotechnology 2018, 70, 1301–1313. [Google Scholar] [CrossRef]
- Breu, A.; Sprinzing, B.; Merkl, K.; Bechmann, V.; Kujat, R.; Jenei-Lanzl, Z.; Prantl, L.; Angele, P. Estrogen Reduces Cellular Aging in Human Mesenchymal Stem Cells and Chondrocytes. J. Orthop. Res. 2011, 29, 1563–1571. [Google Scholar] [CrossRef]
- Wang, Y.; Singh, A.; Xu, P.; Pindrus, M.A.; Blasioli, D.J.; Kaplan, D.L. Expansion and Osteogenic Differentiation of Bone Marrow-Derived Mesenchymal Stem Cells on a Vitamin C Functionalized Polymer. Biomaterials 2006, 27, 3265–3273. [Google Scholar] [CrossRef]
- Zhou, S.; Glowacki, J. Chronic Kidney Disease and Vitamin D Metabolism in Human Bone Marrow–Derived MSCs. Ann. N. Y. Acad. Sci. 2017, 1402, 43–55. [Google Scholar] [CrossRef]
- Klotz, B.; Mentrup, B.; Regensburger, M.; Zeck, S.; Schneidereit, J.; Schupp, N.; Linden, C.; Merz, C.; Ebert, R.; Jakob, F. 1,25-Dihydroxyvitamin D3 Treatment Delays Cellular Aging in Human Mesenchymal Stem Cells While Maintaining Their Multipotent Capacity. PLoS ONE 2012, 7, e29959. [Google Scholar] [CrossRef]
- Sakurai, R.; Singh, H.; Wang, Y.; Harb, A.; Gornes, C.; Liu, J.; Rehan, V.K. Effect of Perinatal Vitamin D Deficiency on Lung Mesenchymal Stem Cell Differentiation and Injury Repair Potential. Am. J. Respir. Cell Mol. Biol. 2021, 65, 521–531. [Google Scholar] [CrossRef]
- Chen, J.; Kuang, S.; Cen, J.; Zhang, Y.; Shen, Z.; Qin, W.; Huang, Q.; Wang, Z.; Gao, X.; Huang, F.; et al. Multiomics Profiling Reveals VDR as a Central Regulator of Mesenchymal Stem Cell Senescence with a Known Association with Osteoporosis after High-Fat Diet Exposure. Int. J. Oral Sci. 2024, 16, 41. [Google Scholar] [CrossRef]
- Samant, G.V.; Sylvester, P.W. γ-Tocotrienol Inhibits ErbB3-dependent PI3K/Akt Mitogenic Signalling in Neoplastic Mammary Epithelial Cells. Cell Prolif. 2006, 39, 563–574. [Google Scholar] [CrossRef]
- Depeint, F.; Bruce, W.R.; Shangari, N.; Mehta, R.; O’Brien, P.J. Mitochondrial Function and Toxicity: Role of B Vitamins on the One-Carbon Transfer Pathways. Chem. Biol. Interact. 2006, 163, 113–132. [Google Scholar] [CrossRef]
- Malavolta, M.; Mocchegiani, E. Molecular Basis of Nutrition and Aging; Academic Press: Cambridge, MA, USA, 2016; ISBN 978-0-12-801816-3. [Google Scholar]
- Ok, J.S.; Song, S.B.; Hwang, E.S. Enhancement of Replication and Differentiation Potential of Human Bone Marrow Stem Cells by Nicotinamide Treatment. Int. J. Stem Cells 2018, 11, 13–25. [Google Scholar] [CrossRef]
- Siti Noor Fazliah, M.N.; Yusuf, M.M.; Abdullah, T.K.; Zuhailawati, H. Human Mesenchymal Stem Cells Response to Magnesium-Based Biomaterials. Procedia Chem. 2016, 19, 75–82. [Google Scholar] [CrossRef][Green Version]
- Díaz-Tocados, J.M.; Herencia, C.; Martínez-Moreno, J.M.; Montes De Oca, A.; Rodríguez-Ortiz, M.E.; Vergara, N.; Blanco, A.; Steppan, S.; Almadén, Y.; Rodríguez, M.; et al. Magnesium Chloride Promotes Osteogenesis through Notch Signaling Activation and Expansion of Mesenchymal Stem Cells. Sci. Rep. 2017, 7, 7839. [Google Scholar] [CrossRef]
- Ballouze, R.; Marahat, M.H.; Mohamad, S.; Saidin, N.A.; Kasim, S.R.; Ooi, J.P. Biocompatible Magnesium-Doped Biphasic Calcium Phosphate for Bone Regeneration. J. Biomed. Mater. Res. B Appl. Biomater. 2021, 109, 1426–1435. [Google Scholar] [CrossRef] [PubMed]
- Jayaraman, P.; Gandhimathi, C.; Venugopal, J.R.; Becker, D.L.; Ramakrishna, S.; Srinivasan, D.K. Controlled Release of Drugs in Electrosprayed Nanoparticles for Bone Tissue Engineering. Adv. Drug Deliv. Rev. 2015, 94, 77–95. [Google Scholar] [CrossRef]
- Qi, T.; Weng, J.; Yu, F.; Zhang, W.; Li, G.; Qin, H.; Tan, Z.; Zeng, H. Insights into the Role of Magnesium Ions in Affecting Osteogenic Differentiation of Mesenchymal Stem Cells. Biol. Trace Elem. Res. 2021, 199, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.S.M.; Chan, J.; Shuter, B.; Tan, L.G.; Chong, M.S.K.; Ramachandra, D.L.; Dawe, G.S.; Ding, J.; Teoh, S.H.; Beuf, O.; et al. Microgel Iron Oxide Nanoparticles for Tracking Human Fetal Mesenchymal Stem Cells Through Magnetic Resonance Imaging. Stem Cells 2009, 27, 1921–1931. [Google Scholar] [CrossRef] [PubMed]
- Borriello, A.; Caldarelli, I.; Speranza, M.C.; Scianguetta, S.; Tramontano, A.; Bencivenga, D.; Stampone, E.; Negri, A.; Nobili, B.; Locatelli, F.; et al. Iron Overload Enhances Human Mesenchymal Stromal Cell Growth and Hampers Matrix Calcification. Biochim. Biophys. Acta BBA—Gen. Subj. 2016, 1860, 1211–1223. [Google Scholar] [CrossRef]
- Patil, S.; Paul, S. A Comprehensive Review on the Role of Various Materials in the Osteogenic Differentiation of Mesenchymal Stem Cells with a Special Focus on the Association of Heat Shock Proteins and Nanoparticles. Cells Tissues Organs 2014, 199, 81–102. [Google Scholar] [CrossRef]
- Barradas, A.M.C.; Fernandes, H.A.M.; Groen, N.; Chai, Y.C.; Schrooten, J.; Van De Peppel, J.; Van Leeuwen, J.P.T.M.; Van Blitterswijk, C.A.; De Boer, J. A Calcium-Induced Signaling Cascade Leading to Osteogenic Differentiation of Human Bone Marrow-Derived Mesenchymal Stromal Cells. Biomaterials 2012, 33, 3205–3215. [Google Scholar] [CrossRef]
- Shie, M.-Y.; Chiang, W.-H.; Chen, I.-W.P.; Liu, W.-Y.; Chen, Y.-W. Synergistic Acceleration in the Osteogenic and Angiogenic Differentiation of Human Mesenchymal Stem Cells by Calcium Silicate–Graphene Composites. Mater. Sci. Eng. C 2017, 73, 726–735. [Google Scholar] [CrossRef]
- Ruparel, N.B.; Teixeira, F.B.; Ferraz, C.C.R.; Diogenes, A. Direct Effect of Intracanal Medicaments on Survival of Stem Cells of the Apical Papilla. J. Endod. 2012, 38, 1372–1375. [Google Scholar] [CrossRef]
- Bhatti, F.U.; Mehmood, A.; Latief, N.; Zahra, S.; Cho, H.; Khan, S.N.; Riazuddin, S. Vitamin E Protects Rat Mesenchymal Stem Cells against Hydrogen Peroxide-Induced Oxidative Stress in Vitro and Improves Their Therapeutic Potential in Surgically-Induced Rat Model of Osteoarthritis. Osteoarthr. Cartil. 2017, 25, 321–331. [Google Scholar] [CrossRef] [PubMed]
- El Alami, M.; Viña-Almunia, J.; Gambini, J.; Mas-Bargues, C.; Siow, R.C.M.; Peñarrocha, M.; Mann, G.E.; Borrás, C.; Viña, J. Activation of P38, P21, and NRF-2 Mediates Decreased Proliferation of Human Dental Pulp Stem Cells Cultured under 21% O2. Stem Cell Rep. 2014, 3, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Kim, M.; Hwa, S.; Ko, Y.; Park, J.-B. Vitamin E Enhances Cell Viability and the Osteogenic Differentiation of Cell Spheroids Made of Gingiva-Derived Stem Cells. Medicina 2023, 59, 736. [Google Scholar] [CrossRef]
- Lee, M.N.; Hwang, H.-S.; Oh, S.-H.; Roshanzadeh, A.; Kim, J.-W.; Song, J.H.; Kim, E.-S.; Koh, J.-T. Elevated Extracellular Calcium Ions Promote Proliferation and Migration of Mesenchymal Stem Cells via Increasing Osteopontin Expression. Exp. Mol. Med. 2018, 50, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Rahman, F.; Bordignon, B.; Culerrier, R.; Peiretti, F.; Spicuglia, S.; Djabali, M.; Landrier, J.F.; Fontes, M. Ascorbic Acid Drives the Differentiation of Mesoderm-derived Embryonic Stem Cells. Involvement of P38 MAPK/CREB and SVCT2 Transporter. Mol. Nutr. Food Res. 2017, 61, 1600506. [Google Scholar] [CrossRef]
- Casado-Díaz, A.; Túnez-Fiñana, I.; Mata-Granados, J.M.; Ruiz-Méndez, M.V.; Dorado, G.; Romero-Sánchez, M.C.; Navarro-Valverde, C.; Quesada-Gómez, J.M. Serum from Postmenopausal Women Treated with a By-Product of Olive-Oil Extraction Process Stimulates Osteoblastogenesis and Inhibits Adipogenesis in Human Mesenchymal Stem-Cells (MSC). Exp. Gerontol. 2017, 90, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Lou, Y.-R.; Toh, T.C.; Tee, Y.H.; Yu, H. 25-Hydroxyvitamin D3 Induces Osteogenic Differentiation of Human Mesenchymal Stem Cells. Sci. Rep. 2017, 7, 42816. [Google Scholar] [CrossRef]
- Weng, Z.; Wang, Y.; Ouchi, T.; Liu, H.; Qiao, X.; Wu, C.; Zhao, Z.; Li, L.; Li, B. Mesenchymal Stem/Stromal Cell Senescence: Hallmarks, Mechanisms, and Combating Strategies. Stem Cells Transl. Med. 2022, 11, 356–371. [Google Scholar] [CrossRef]
- Burgess, K.A.; Herrick, A.L.; Watson, R.E.B. Systemic Sclerosis Skin Is a Primed Microenvironment for Soft Tissue Calcification—A Hypothesis. Rheumatology 2021, 60, 2517–2527. [Google Scholar] [CrossRef]
- González-Casanova, J.E.; Navarro-Marquez, M.; Saez-Tamayo, T.; Angarita, L.; Durán-Agüero, S.; Fuentes-Barría, H.; Bermúdez, V.; Rojas-Gómez, D.M. New Perspectives on the Molecular Action of Metformin in the Context of Cellular Transduction and Adipogenesis. Int. J. Mol. Sci. 2025, 26, 3690. [Google Scholar] [CrossRef]
- Valenti, M.; Dalle Carbonare, L.; Mottes, M. Osteogenic Differentiation in Healthy and Pathological Conditions. Int. J. Mol. Sci. 2016, 18, 41. [Google Scholar] [CrossRef]
- Zhou, S.; LeBoff, M.S.; Glowacki, J. Vitamin D Metabolism and Action in Human Bone Marrow Stromal Cells. Endocrinology 2010, 151, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lee, S.; Kim, K. Bone Tissue Engineering Strategies in Co-Delivery of Bone Morphogenetic Protein-2 and Biochemical Signaling Factors. In Cutting-Edge Enabling Technologies for Regenerative Medicine; Chun, H.J., Park, C.H., Kwon, I.K., Khang, G., Eds.; Advances in Experimental Medicine and Biology; Springer: Singapore, 2018; Volume 1078, pp. 233–244. ISBN 978-981-13-0949-6. [Google Scholar]
- Piek, E.; Sleumer, L.S.; Van Someren, E.P.; Heuver, L.; De Haan, J.R.; De Grijs, I.; Gilissen, C.; Hendriks, J.M.; Van Ravestein-van Os, R.I.; Bauerschmidt, S.; et al. Osteo-Transcriptomics of Human Mesenchymal Stem Cells: Accelerated Gene Expression and Osteoblast Differentiation Induced by Vitamin D Reveals c-MYC as an Enhancer of BMP2-Induced Osteogenesis. Bone 2010, 46, 613–627. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Tang, M.; Weir, M.D.; Detamore, M.S.; Xu, H.H.K. Osteogenic Media and rhBMP-2-Induced Differentiation of Umbilical Cord Mesenchymal Stem Cells Encapsulated in Alginate Microbeads and Integrated in an Injectable Calcium Phosphate-Chitosan Fibrous Scaffold. Tissue Eng. Part A 2011, 17, 969–979. [Google Scholar] [CrossRef]
- Song, I.; Kim, B.-S.; Kim, C.-S.; Im, G.-I. Effects of BMP-2 and Vitamin D3 on the Osteogenic Differentiation of Adipose Stem Cells. Biochem. Biophys. Res. Commun. 2011, 408, 126–131. [Google Scholar] [CrossRef]
- Lu, C.-L.; Shyu, J.-F.; Wu, C.-C.; Hung, C.-F.; Liao, M.-T.; Liu, W.-C.; Zheng, C.-M.; Hou, Y.-C.; Lin, Y.-F.; Lu, K.-C. Association of Anabolic Effect of Calcitriol with Osteoclast-Derived Wnt 10b Secretion. Nutrients 2018, 10, 1164. [Google Scholar] [CrossRef]
- Castano-Izquierdo, H.; Álvarez-Barreto, J.; Dolder, J.V.D.; Jansen, J.A.; Mikos, A.G.; Sikavitsas, V.I. Pre-culture Period of Mesenchymal Stem Cells in Osteogenic Media Influences Their in Vivo Bone Forming Potential. J. Biomed. Mater. Res. A 2007, 82A, 129–138. [Google Scholar] [CrossRef]
- Duque, G.; Rivas, D. Alendronate Has an Anabolic Effect on Bone Through the Differentiation of Mesenchymal Stem Cells. J. Bone Miner. Res. 2007, 22, 1603–1611. [Google Scholar] [CrossRef]
- Muruganandan, S.; Roman, A.A.; Sinal, C.J. Adipocyte Differentiation of Bone Marrow-Derived Mesenchymal Stem Cells: Cross Talk with the Osteoblastogenic Program. Cell. Mol. Life Sci. 2009, 66, 236–253. [Google Scholar] [CrossRef] [PubMed]
- Pesarini, J.R.; Oliveira, R.J.; Pessatto, L.R.; Antoniolli-Silva, A.C.M.B.; Felicidade, I.; Nardi, N.B.; Camassola, M.; Mantovani, M.S.; Ribeiro, L.R. Vitamin D: Correlation with Biochemical and Body Composition Changes in a Southern Brazilian Population and Induction of Cytotoxicity in Mesenchymal Stem Cells Derived from Human Adipose Tissue. Biomed. Pharmacother. 2017, 91, 861–871. [Google Scholar] [CrossRef] [PubMed]
- Barlian, A.; Judawisastra, H.; Alfarafisa, N.M.; Wibowo, U.A.; Rosadi, I. Chondrogenic Differentiation of Adipose-Derived Mesenchymal Stem Cells Induced by L-Ascorbic Acid and Platelet Rich Plasma on Silk Fibroin Scaffold. PeerJ 2018, 6, e5809. [Google Scholar] [CrossRef]
- Chang, Z.; Huo, L.; Li, P.; Wu, Y.; Zhang, P. Ascorbic Acid Provides Protection for Human Chondrocytes against Oxidative Stress. Mol. Med. Rep. 2015, 12, 7086–7092. [Google Scholar] [CrossRef]
- Yoshizawa, S.; Brown, A.; Barchowsky, A.; Sfeir, C. Magnesium Ion Stimulation of Bone Marrow Stromal Cells Enhances Osteogenic Activity, Simulating the Effect of Magnesium Alloy Degradation. Acta Biomater. 2014, 10, 2834–2842. [Google Scholar] [CrossRef] [PubMed]
- Rude, R.K.; Gruber, H.E. Magnesium Deficiency and Osteoporosis: Animal and Human Observations. J. Nutr. Biochem. 2004, 15, 710–716. [Google Scholar] [CrossRef]
- Castiglioni, S.; Cazzaniga, A.; Albisetti, W.; Maier, J. Magnesium and Osteoporosis: Current State of Knowledge and Future Research Directions. Nutrients 2013, 5, 3022–3033. [Google Scholar] [CrossRef]
- Sargenti, A.; Castiglioni, S.; Olivi, E.; Bianchi, F.; Cazzaniga, A.; Farruggia, G.; Cappadone, C.; Merolle, L.; Malucelli, E.; Ventura, C.; et al. Magnesium Deprivation Potentiates Human Mesenchymal Stem Cell Transcriptional Remodeling. Int. J. Mol. Sci. 2018, 19, 1410. [Google Scholar] [CrossRef]
- Shimaya, M.; Muneta, T.; Ichinose, S.; Tsuji, K.; Sekiya, I. Magnesium Enhances Adherence and Cartilage Formation of Synovial Mesenchymal Stem Cells through Integrins. Osteoarthr. Cartil. 2010, 18, 1300–1309. [Google Scholar] [CrossRef]
- Dou, Y.; Li, N.; Zheng, Y.; Ge, Z. Effects of Fluctuant Magnesium Concentration on Phenotype of the Primary Chondrocytes. J. Biomed. Mater. Res. A 2014, 102, 4455–4463. [Google Scholar] [CrossRef] [PubMed]
- Park, K.-S.; Kim, B.-J.; Lih, E.; Park, W.; Lee, S.-H.; Joung, Y.K.; Han, D.K. Versatile Effects of Magnesium Hydroxide Nanoparticles in PLGA Scaffold–Mediated Chondrogenesis. Acta Biomater. 2018, 73, 204–216. [Google Scholar] [CrossRef]
- Shih, Y.-R.V.; Hwang, Y.; Phadke, A.; Kang, H.; Hwang, N.S.; Caro, E.J.; Nguyen, S.; Siu, M.; Theodorakis, E.A.; Gianneschi, N.C.; et al. Calcium Phosphate-Bearing Matrices Induce Osteogenic Differentiation of Stem Cells through Adenosine Signaling. Proc. Natl. Acad. Sci. USA 2014, 111, 990–995. [Google Scholar] [CrossRef]
- Phadke, A.; Shih, Y.V.; Varghese, S. Mineralized Synthetic Matrices as an Instructive Microenvironment for Osteogenic Differentiation of Human Mesenchymal Stem Cells. Macromol. Biosci. 2012, 12, 1022–1032. [Google Scholar] [CrossRef]
- Telgerd, M.D.; Sadeghinia, M.; Birhanu, G.; Daryasari, M.P.; Zandi-Karimi, A.; Sadeghinia, A.; Akbarijavar, H.; Karami, M.H.; Seyedjafari, E. Enhanced Osteogenic Differentiation of Mesenchymal Stem Cells on Metal–Organic Framework Based on Copper, Zinc, and Imidazole Coated Poly-l-lactic Acid Nanofiber Scaffolds. J. Biomed. Mater. Res. A 2019, 107, 1841–1848. [Google Scholar] [CrossRef] [PubMed]
- Poon, C.C.W.; Li, R.W.S.; Seto, S.W.; Kong, S.K.; Ho, H.P.; Hoi, M.P.M.; Lee, S.M.Y.; Ngai, S.M.; Chan, S.W.; Leung, G.P.H.; et al. In Vitro Vitamin K2 and 1α,25-Dihydroxyvitamin D3 Combination Enhances Osteoblasts Anabolism of Diabetic Mice. Eur. J. Pharmacol. 2015, 767, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-McQuire, R.; Green, D.W.; Partridge, K.A.; Oreffo, R.O.C.; Mann, S.; Davis, S.A. Coating of Human Mesenchymal Cells in 3D Culture with Bioinorganic Nanoparticles Promotes Osteoblastic Differentiation and Gene Transfection. Adv. Mater. 2007, 19, 2236–2240. [Google Scholar] [CrossRef]
- Hu, Q.; Tan, Z.; Liu, Y.; Tao, J.; Cai, Y.; Zhang, M.; Pan, H.; Xu, X.; Tang, R. Effect of Crystallinity of Calcium Phosphate Nanoparticles on Adhesion, Proliferation, and Differentiation of Bone Marrow Mesenchymal Stem Cells. J. Mater. Chem. 2007, 17, 4690. [Google Scholar] [CrossRef]
- Müller, P.; Bulnheim, U.; Diener, A.; Lüthen, F.; Teller, M.; Klinkenberg, E.; Neumann, H.; Nebe, B.; Liebold, A.; Steinhoff, G.; et al. Calcium Phosphate Surfaces Promote Osteogenic Differentiation of Mesenchymal Stem Cells. J. Cell. Mol. Med. 2008, 12, 281–291. [Google Scholar] [CrossRef]
- Sarkar, N.; Morton, H.; Bose, S. Effects of Vitamin C on Osteoblast Proliferation and Osteosarcoma Inhibition Using Plasma Coated Hydroxyapatite on Titanium Implants. Surf. Coat. Technol. 2020, 394, 125793. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.-N.; Zhang, L.; Wang, T.; Zhang, H.-Y.; Yang, Z.-J.; Yuan, F.-F.; Wang, Y.; Li, S.-W.; Jiang, X.-X.; Xie, X.-H. Vitamin C Treatment Rescues Prelamin A-Induced Premature Senescence of Subchondral Bone Mesenchymal Stem Cells. Stem Cells Int. 2020, 2020, 3150716. [Google Scholar] [CrossRef] [PubMed]
- Rathinavelu, S.; Guidry-Elizondo, C.; Banu, J. Molecular Modulation of Osteoblasts and Osteoclasts in Type 2 Diabetes. J. Diabetes Res. 2018, 2018, 6354787. [Google Scholar] [CrossRef] [PubMed]
- Schröder-Heurich, B.; Hardenberg, S.; Brodowski, L.; Kipke, B.; Meyer, N.; Borns, K.; Kaisenberg, C.S.; Brinkmann, H.; Claus, P.; Versen-Höynck, F. Vitamin D Improves Endothelial Barrier Integrity and Counteracts Inflammatory Effects on Endothelial Progenitor Cells. FASEB J. 2019, 33, 9142–9153. [Google Scholar] [CrossRef]
- Hu, C.; Li, L. Preconditioning Influences Mesenchymal Stem Cell Properties in Vitro and in Vivo. J. Cell. Mol. Med. 2018, 22, 1428–1442. [Google Scholar] [CrossRef]
- Kornicka, K.; Babiarczuk, B.; Krzak, J.; Marycz, K. The Effect of a Sol–Gel Derived Silica Coating Doped with Vitamin E on Oxidative Stress and Senescence of Human Adipose-Derived Mesenchymal Stem Cells (AMSCs). RSC Adv. 2016, 6, 29524–29537. [Google Scholar] [CrossRef]
- De Witte, S.F.H.; Merino, A.M.; Franquesa, M.; Strini, T.; Van Zoggel, J.A.A.; Korevaar, S.S.; Luk, F.; Gargesha, M.; O’Flynn, L.; Roy, D.; et al. Cytokine Treatment Optimises the Immunotherapeutic Effects of Umbilical Cord-Derived MSC for Treatment of Inflammatory Liver Disease. Stem Cell Res. Ther. 2017, 8, 140. [Google Scholar] [CrossRef]
- Kim, D.-H.; Shin, K.-K.; Jung, J.S.; Chun, H.H.; Park, S.S.; Lee, J.K.; Park, H.-C.; Yoon, S.-Y. The Role of Magnesium Ion Substituted Biphasic Calcium Phosphate Spherical Micro-Scaffolds in Osteogenic Differentiation of Human Adipose Tissue-Derived Mesenchymal Stem Cells. J. Nanosci. Nanotechnol. 2015, 15, 5520–5523. [Google Scholar] [CrossRef]
- Kim, Y.H.; Jung, E.; Im, G.-B.; Kim, Y.-J.; Kim, S.-W.; Jeong, G.-J.; Jang, Y.C.; Park, K.M.; Kim, D.-I.; Yu, T.; et al. Regulation of Intracellular Transition Metal Ion Level with a pH-Sensitive Inorganic Nanocluster to Improve Therapeutic Angiogenesis by Enriching Conditioned Medium Retrieved from Human Adipose Derived Stem Cells. Nano Converg. 2020, 7, 34. [Google Scholar] [CrossRef]
- Khalifa, Y.H.; Mourad, G.M.; Stephanos, W.M.; Omar, S.A.; Mehanna, R.A. Bone Marrow-Derived Mesenchymal Stem Cell Potential Regression of Dysplasia Associating Experimental Liver Fibrosis in Albino Rats. BioMed Res. Int. 2019, 2019, 5376165. [Google Scholar] [CrossRef] [PubMed]
- Im, G.-B.; Kim, Y.H.; Kim, Y.-J.; Kim, S.-W.; Jung, E.; Jeong, G.-J.; Wang, K.; Kim, J.; Kim, D.-I.; Kim, T.-H.; et al. Enhancing the Wound Healing Effect of Conditioned Medium Collected from Mesenchymal Stem Cells with High Passage Number Using Bioreducible Nanoparticles. Int. J. Mol. Sci. 2019, 20, 4835. [Google Scholar] [CrossRef] [PubMed]
- Cimmino, L.; Neel, B.G.; Aifantis, I. Vitamin C in Stem Cell Reprogramming and Cancer. Trends Cell Biol. 2018, 28, 698–708. [Google Scholar] [CrossRef]
- Wei, C.; Liu, X.; Tao, J.; Wu, R.; Zhang, P.; Bian, Y.; Li, Y.; Fang, F.; Zhang, Y. Effects of Vitamin C on Characteristics Retaining of in Vitro-Cultured Mouse Adipose-Derived Stem Cells. Vitr. Cell. Dev. Biol.–Anim. 2014, 50, 75–86. [Google Scholar] [CrossRef]
- Serpooshan, V.; Julien, M.; Nguyen, O.; Wang, H.; Li, A.; Muja, N.; Henderson, J.E.; Nazhat, S.N. Reduced Hydraulic Permeability of Three-Dimensional Collagen Scaffolds Attenuates Gel Contraction and Promotes the Growth and Differentiation of Mesenchymal Stem Cells. Acta Biomater. 2010, 6, 3978–3987. [Google Scholar] [CrossRef]
- Virdee, S.S.; Bashir, N.; Camilleri, J.; Cooper, P.R.; Tomson, P.L. Exploiting Dentine Matrix Proteins in Cell-Free Approaches for Periradicular Tissue Engineering. Tissue Eng. Part B Rev. 2022, 28, 707–732. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.; Maggini, S. Vitamin C and Immune Function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef]
- Behm, C.; Blufstein, A.; Gahn, J.; Kubin, B.; Nemec, M.; Moritz, A.; Rausch-Fan, X.; Andrukhov, O. 1,25(OH)2D3 Differently Affects Immunomodulatory Activities of Mesenchymal Stem Cells Depending on the Presence of TNF-α, IL-1β and IFN-γ. J. Clin. Med. 2019, 8, 2211. [Google Scholar] [CrossRef]
- Marcotorchino, J.; Gouranton, E.; Romier, B.; Tourniaire, F.; Astier, J.; Malezet, C.; Amiot, M.; Landrier, J. Vitamin D Reduces the Inflammatory Response and Restores Glucose Uptake in Adipocytes. Mol. Nutr. Food Res. 2012, 56, 1771–1782. [Google Scholar] [CrossRef]
- Karkeni, E.; Marcotorchino, J.; Tourniaire, F.; Astier, J.; Peiretti, F.; Darmon, P.; Landrier, J.-F. Vitamin D Limits Chemokine Expression in Adipocytes and Macrophage Migration In Vitro and in Male Mice. Endocrinology 2015, 156, 1782–1793. [Google Scholar] [CrossRef]
- Di Tinco, R.; Bertani, G.; Pisciotta, A.; Bertoni, L.; Pignatti, E.; Maccaferri, M.; Bertacchini, J.; Sena, P.; Vallarola, A.; Tupler, R.; et al. Role of PD-L1 in Licensing Immunoregulatory Function of Dental Pulp Mesenchymal Stem Cells. Stem Cell Res. Ther. 2021, 12, 598. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, P.; Yan, M.; Bauche, A.; Smeets, R.; Müller, C.E.; Koch-Nolte, F.; Haag, F.; Fliegert, R.; Kluwe, L.; Schulze Zur Wiesch, J.; et al. Human Dental Pulp Cells Modulate CD8+ T Cell Proliferation and Efficiently Degrade Extracellular ATP to Adenosine in Vitro. Cell. Immunol. 2022, 380, 104589. [Google Scholar] [CrossRef]
- Guo, L.; Zhang, Y.; Liu, H.; Cheng, Q.; Yang, S.; Yang, D. All-Trans Retinoic Acid Inhibits the Osteogenesis of Periodontal Ligament Stem Cells by Promoting IL-1β Production via NF-κB Signaling. Int. Immunopharmacol. 2022, 108, 108757. [Google Scholar] [CrossRef]
- Da Silva Lima, F.; Da Rocha Romero, A.B.; Hastreiter, A.; Nogueira-Pedro, A.; Makiyama, E.; Colli, C.; Fock, R.A. An Insight into the Role of Magnesium in the Immunomodulatory Properties of Mesenchymal Stem Cells. J. Nutr. Biochem. 2018, 55, 200–208. [Google Scholar] [CrossRef]
- Xiaodan, L.; Yifan, L.; Xiuying, L.; Qiang, Z.; Zihang, W.; Xuguang, M.; Ruobing, W.; Xiaofang, Z.; Xu, L.; Yan, L.; et al. Anti-Oxidative Effect of Zinc in Human Umbilical Cord Mesenchymal Stem Cells. Biophys. Rep. 2021, 7, 142–151. [Google Scholar] [CrossRef]
- Li, X.; Wei, Z.; Li, B.; Li, J.; Lv, H.; Wu, L.; Zhang, H.; Yang, B.; Zhu, M.; Jiang, J. In Vivo Migration of Fe3O4 @polydopamine Nanoparticle-Labeled Mesenchymal Stem Cells to Burn Injury Sites and Their Therapeutic Effects in a Rat Model. Biomater. Sci. 2019, 7, 2861–2872. [Google Scholar] [CrossRef]
- Wei, F.; Qu, C.; Song, T.; Ding, G.; Fan, Z.; Liu, D.; Liu, Y.; Zhang, C.; Shi, S.; Wang, S. Vitamin C Treatment Promotes Mesenchymal Stem Cell Sheet Formation and Tissue Regeneration by Elevating Telomerase Activity. J. Cell. Physiol. 2012, 227, 3216–3224. [Google Scholar] [CrossRef]
- Ebert, R.; Ulmer, M.; Zeck, S.; Meissner-Weigl, J.; Schneider, D.; Stopper, H.; Schupp, N.; Kassem, M.; Jakob, F. Selenium Supplementation Restores the Antioxidative Capacity and Prevents Cell Damage in Bone Marrow Stromal Cells In Vitro. Stem Cells 2006, 24, 1226–1235. [Google Scholar] [CrossRef]
- Rahimi, B.; Panahi, M.; Lotfi, H.; Khalili, M.; Salehi, A.; Saraygord-Afshari, N.; Alizadeh, E. Sodium Selenite Preserves rBM-MSCs’ Stemness, Differentiation Potential, and Immunophenotype and Protects Them against Oxidative Stress via Activation of the Nrf2 Signaling Pathway. BMC Complement. Med. Ther. 2023, 23, 131. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Jeong, E.-M.; Liu, H.; Xie, A.; So, E.Y.; Shi, G.; Jeong, G.E.; Zhou, A.; Dudley, S.C. Magnesium Supplementation Improves Diabetic Mitochondrial and Cardiac Diastolic Function. JCI Insight 2019, 4, e123182. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Liu, H.; Feng, F.; Xie, A.; Kang, G.; Zhao, Y.; Hou, C.R.; Zhou, X.; Dudley, S.C. Magnesium Deficiency Causes a Reversible, Metabolic, Diastolic Cardiomyopathy. J. Am. Heart Assoc. 2021, 10, e020205. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Bian, W.; Liu, S.; Huang, K. Selenium Protects Bone Marrow Stromal Cells Against Hydrogen Peroxide-Induced Inhibition of Osteoblastic Differentiation by Suppressing Oxidative Stress and ERK Signaling Pathway. Biol. Trace Elem. Res. 2012, 150, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, F.U.R.; Kim, S.J.; Yi, A.-K.; Hasty, K.A.; Cho, H. Cytoprotective Role of Vitamin E in Porcine Adipose-Tissue-Derived Mesenchymal Stem Cells against Hydrogen-Peroxide-Induced Oxidative Stress. Cell Tissue Res. 2018, 374, 111–120. [Google Scholar] [CrossRef]
- Khorraminejad-Shirazi, M.; Sani, M.; Talaei-Khozani, T.; Dorvash, M.; Mirzaei, M.; Faghihi, M.A.; Monabati, A.; Attar, A. AICAR and Nicotinamide Treatment Synergistically Augment the Proliferation and Attenuate Senescence-Associated Changes in Mesenchymal Stromal Cells. Stem Cell Res. Ther. 2020, 11, 45. [Google Scholar] [CrossRef]
- Cruciani, S.; Garroni, G.; Pala, R.; Cossu, M.L.; Ginesu, G.C.; Ventura, C.; Maioli, M. Metformin and Vitamin D Modulate Inflammation and Autophagy during Adipose-Derived Stem Cell Differentiation. Int. J. Mol. Sci. 2021, 22, 6686. [Google Scholar] [CrossRef]
- Cruciani, S.; Garroni, G.; Pala, R.; Coradduzza, D.; Cossu, M.L.; Ginesu, G.C.; Capobianco, G.; Dessole, S.; Ventura, C.; Maioli, M. Metformin and Vitamin D Modulate Adipose-Derived Stem Cell Differentiation towards the Beige Phenotype. Adipocyte 2022, 11, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Le Pelletier, L.; Mantecon, M.; Gorwood, J.; Auclair, M.; Foresti, R.; Motterlini, R.; Laforge, M.; Atlan, M.; Fève, B.; Capeau, J.; et al. Metformin Alleviates Stress-Induced Cellular Senescence of Aging Human Adipose Stromal Cells and the Ensuing Adipocyte Dysfunction. eLife 2021, 10, e62635. [Google Scholar] [CrossRef]
- Casati, L.; Pagani, F.; Limonta, P.; Vanetti, C.; Stancari, G.; Sibilia, V. Beneficial Effects of δ-Tocotrienol against Oxidative Stress in Osteoblastic Cells: Studies on the Mechanisms of Action. Eur. J. Nutr. 2020, 59, 1975–1987. [Google Scholar] [CrossRef]
- Moon, M.-Y.; Kim, H.J.; Choi, B.Y.; Sohn, M.; Chung, T.N.; Suh, S.W. Zinc Promotes Adipose-Derived Mesenchymal Stem Cell Proliferation and Differentiation towards a Neuronal Fate. Stem Cells Int. 2018, 2018, 5736535. [Google Scholar] [CrossRef]
- Novotna, B.; Jendelova, P.; Kapcalova, M.; Rossner, P.; Turnovcova, K.; Bagryantseva, Y.; Babic, M.; Horak, D.; Sykova, E. Oxidative Damage to Biological Macromolecules in Human Bone Marrow Mesenchymal Stromal Cells Labeled with Various Types of Iron Oxide Nanoparticles. Toxicol. Lett. 2012, 210, 53–63. [Google Scholar] [CrossRef]
- Li, X.; Wei, Z.; Lv, H.; Wu, L.; Cui, Y.; Yao, H.; Li, J.; Zhang, H.; Yang, B.; Jiang, J. Iron Oxide Nanoparticles Promote the Migration of Mesenchymal Stem Cells to Injury Sites. Int. J. Nanomed. 2019, 14, 573–589. [Google Scholar] [CrossRef]
- Zhang, C.; Liu, Z.; Zhang, Y.; Ma, L.; Song, E.; Song, Y. “Iron Free” Zinc Oxide Nanoparticles with Ion-Leaking Properties Disrupt Intracellular ROS and Iron Homeostasis to Induce Ferroptosis. Cell Death Dis. 2020, 11, 183. [Google Scholar] [CrossRef]
- Bourebaba, L.; Michalak, I.; Röcken, M.; Marycz, K. Cladophora Glomerata Methanolic Extract Decreases Oxidative Stress and Improves Viability and Mitochondrial Potential in Equine Adipose Derived Mesenchymal Stem Cells (ASCs). Biomed. Pharmacother. 2019, 111, 6–18. [Google Scholar] [CrossRef]
- Jung, J.-W.; Lee, S.; Seo, M.-S.; Park, S.-B.; Kurtz, A.; Kang, S.-K.; Kang, K.-S. Histone Deacetylase Controls Adult Stem Cell Aging by Balancing the Expression of Polycomb Genes and Jumonji Domain Containing 3. Cell. Mol. Life Sci. 2010, 67, 1165–1176. [Google Scholar] [CrossRef]
- Takaya, J.; Iharada, A.; Okihana, H.; Kaneko, K. Magnesium Deficiency in Pregnant Rats Alters Methylation of Specific Cytosines in the Hepatic Hydroxysteroid Dehydrogenase-2 Promoter of the Offspring. Epigenetics 2011, 6, 573–578. [Google Scholar] [CrossRef]
- Newell-Price, J.; Clark, A.J.L.; King, P. DNA Methylation and Silencing of Gene Expression. Trends Endocrinol. Metab. 2000, 11, 142–148. [Google Scholar] [CrossRef]
- Thienpont, B.; Steinbacher, J.; Zhao, H.; D’Anna, F.; Kuchnio, A.; Ploumakis, A.; Ghesquière, B.; Van Dyck, L.; Boeckx, B.; Schoonjans, L.; et al. Tumour Hypoxia Causes DNA Hypermethylation by Reducing TET Activity. Nature 2016, 537, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Fathi, E.; Farahzadi, R.; Rahbarghazi, R.; Samadi Kafil, H.; Yolmeh, R. Rat Adipose-Derived Mesenchymal Stem Cells Aging Reduction by Zinc Sulfate under Extremely Low Frequency Electromagnetic Field Exposure Is Associated with Increased Telomerase Reverse Transcriptase Gene Expression. Vet. Res. Forum Int. Q. J. 2017, 8, 89–96. [Google Scholar]
- Shin, J.-H.; Jeon, H.-J.; Park, J.; Chang, M.-S. Epigallocatechin-3-Gallate Prevents Oxidative Stress-Induced Cellular Senescence in Human Mesenchymal Stem Cells via Nrf2. Int. J. Mol. Med. 2016, 38, 1075–1082. [Google Scholar] [CrossRef]
- Khan, I.M.; Gul, H.; Khan, S.; Nassar, N.; Khalid, A.; Swelum, A.A.; Wang, Z. Green Tea Polyphenol Epigallocatechin-3-Gallate Mediates an Antioxidant Response via Nrf2 Pathway in Heat-Stressed Poultry: A Review. Poult. Sci. 2025, 104, 105071. [Google Scholar] [CrossRef] [PubMed]
- Valle-Prieto, A.; Conget, P.A. Human Mesenchymal Stem Cells Efficiently Manage Oxidative Stress. Stem Cells Dev. 2010, 19, 1885–1893. [Google Scholar] [CrossRef] [PubMed]
- Yagi, H.; Tan, J.; Tuan, R.S. Polyphenols Suppress Hydrogen Peroxide-induced Oxidative Stress in Human Bone-marrow Derived Mesenchymal Stem Cells. J. Cell. Biochem. 2013, 114, 1163–1173. [Google Scholar] [CrossRef]
- Wang, D.; Wang, Y.; Xu, S.; Wang, F.; Wang, B.; Han, K.; Sun, D.; Li, L. Epigallocatechin-3-gallate Protects against Hydrogen Peroxide-Induced Inhibition of Osteogenic Differentiation of Human Bone Marrow-Derived Mesenchymal Stem Cells. Stem Cells Int. 2016, 2016, 7532798. [Google Scholar] [CrossRef]
- Bose, S.; Robertson, S.F.; Vu, A.A. Garlic Extract Enhances Bioceramic Bone Scaffolds through Upregulating ALP & BGLAP Expression in hMSC-Monocyte Co-Culture. Biomater. Adv. 2023, 154, 213622. [Google Scholar] [CrossRef]
- Dai, Z.; Li, Y.; Quarles, L.D.; Song, T.; Pan, W.; Zhou, H.; Xiao, Z. Resveratrol Enhances Proliferation and Osteoblastic Differentiation in Human Mesenchymal Stem Cells via ER-Dependent ERK1/2 Activation. Phytomedicine 2007, 14, 806–814. [Google Scholar] [CrossRef]
- Quiles, J.L.; Ochoa, J.J.; Ramirez-Tortosa, M.C.; Linde, J.; Bompadre, S.; Battino, M.; Narbona, E.; Maldonado, J.; Mataix, J. Coenzyme Q Concentration and Total Antioxidant Capacity of Human Milk at Different Stages of Lactation in Mothers of Preterm and Full-Term Infants. Free Radic. Res. 2006, 40, 199–206. [Google Scholar] [CrossRef]
- National Institute of Child Health and Human Development. Coenzyme Q10. In Drugs and Lactation Database (LactMed®); National Institute of Child Health and Human Development: Bethesda, MD, USA, 2006. [Google Scholar]
- Tang, P.H.; Miles, M.V.; Steele, P.; Davidson, B.S.; Geraghty, S.R.; Morrow, A.L. Determination of Coenzyme Q10 in Human Breast Milk by High-performance Liquid Chromatography. Biomed. Chromatogr. 2006, 20, 1336–1343. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.-N.; Wang, Z.-Y.; Cheng, X.-F.; Huang, Z.-Z.; Han, Y.-L.; Cui, Y.-Z.; Liu, B.; Tian, W. Melatonin Alleviates Oxidative Stress-Induced Injury to Nucleus Pulposus-Derived Mesenchymal Stem Cells through Activating PI3K/Akt Pathway. J. Orthop. Transl. 2023, 43, 66–84. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Yan, B.; Yu, S.; Zhang, C.; Wang, B.; Wang, Y.; Wang, J.; Yuan, Z.; Zhang, L.; Pan, J. Coenzyme Q10 Inhibits the Aging of Mesenchymal Stem Cells Induced by D-Galactose through Akt/mTOR Signaling. Oxid. Med. Cell. Longev. 2015, 2015, 867293. [Google Scholar] [CrossRef]
- Xue, R.; Yang, J.; Wu, J.; Meng, Q.; Hao, J. Coenzyme Q10 Inhibits the Activation of Pancreatic Stellate Cells through PI3K/AKT/mTOR Signaling Pathway. Oncotarget 2017, 8, 92300–92311. [Google Scholar] [CrossRef]
- Meng, X.; Li, Y.; Li, S.; Zhou, Y.; Gan, R.-Y.; Xu, D.-P.; Li, H.-B. Dietary Sources and Bioactivities of Melatonin. Nutrients 2017, 9, 367. [Google Scholar] [CrossRef]
- Feng, Z.-Y.; Yang, S.-D.; Wang, T.; Guo, S. Effect of Melatonin for Regulating Mesenchymal Stromal Cells and Derived Extracellular Vesicles. Front. Cell Dev. Biol. 2021, 9, 717913. [Google Scholar] [CrossRef]
- Li, B.; Cheng, X.; Aierken, A.; Du, J.; He, W.; Zhang, M.; Tan, N.; Kou, Z.; Peng, S.; Jia, W.; et al. Melatonin Promotes the Therapeutic Effect of Mesenchymal Stem Cells on Type 2 Diabetes Mellitus by Regulating TGF-β Pathway. Front. Cell Dev. Biol. 2021, 9, 722365. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Harikumar, K.B. Potential Therapeutic Effects of Curcumin, the Anti-Inflammatory Agent, against Neurodegenerative, Cardiovascular, Pulmonary, Metabolic, Autoimmune and Neoplastic Diseases. Int. J. Biochem. Cell Biol. 2009, 41, 40–59. [Google Scholar] [CrossRef]
- Han, X.; Shen, T.; Lou, H. Dietary Polyphenols and Their Biological Significance. Int. J. Mol. Sci. 2007, 8, 950–988. [Google Scholar] [CrossRef]
- Mandel, S.; Amit, T.; Reznichenko, L.; Weinreb, O.; Youdim, M.B.H. Green Tea Catechins as Brain-permeable, Natural Iron Chelators--antioxidants for the Treatment of Neurodegenerative Disorders. Mol. Nutr. Food Res. 2006, 50, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Gorabi, A.M.; Kiaie, N.; Hajighasemi, S.; Jamialahmadi, T.; Majeed, M.; Sahebkar, A. The Effect of Curcumin on the Differentiation of Mesenchymal Stem Cells into Mesodermal Lineage. Molecules 2019, 24, 4029. [Google Scholar] [CrossRef]
- Rauf, A.; Imran, M.; Butt, M.S.; Nadeem, M.; Peters, D.G.; Mubarak, M.S. Resveratrol as an Anti-Cancer Agent: A Review. Crit. Rev. Food Sci. Nutr. 2018, 58, 1428–1447. [Google Scholar] [CrossRef]
- Scalbert, A.; Johnson, I.T.; Saltmarsh, M. Polyphenols: Antioxidants and Beyond. Am. J. Clin. Nutr. 2005, 81, 215S–217S. [Google Scholar] [CrossRef] [PubMed]
- Ding, G.; Zhao, J.; Jiang, D. Allicin Inhibits Oxidative Stress-Induced Mitochondrial Dysfunction and Apoptosis by Promoting PI3K/AKT and CREB/ERK Signaling in Osteoblast Cells. Exp. Ther. Med. 2016, 11, 2553–2560. [Google Scholar] [CrossRef]
- Hanga-Farcaș, A.; Miere (Groza), F.; Filip, G.A.; Clichici, S.; Fritea, L.; Vicaș, L.G.; Marian, E.; Pallag, A.; Jurca, T.; Filip, S.M.; et al. Phytochemical Compounds Involved in the Bone Regeneration Process and Their Innovative Administration: A Systematic Review. Plants 2023, 12, 2055. [Google Scholar] [CrossRef]
- Hennessy, Á.; Walton, J.; Flynn, A. The Impact of Voluntary Food Fortification on Micronutrient Intakes and Status in European Countries: A Review. Proc. Nutr. Soc. 2013, 72, 433–440. [Google Scholar] [CrossRef]
- Wagner, K.-H.; Blauensteiner, D.; Schmid, I.; Elmadfa, I. The Role of Fortified Foods—Situation in Austria. Forum Nutr. 2005, 57, 84–90. [Google Scholar] [CrossRef]
- Segon, B.; Lam, L.; Chan, H.Y.; Andersen, S.; Brown, T.; Kenway, D.; Bauer, J. Vitamin Requirements during Stem Cell Transplantation: A Systematic Review. Support. Care Cancer 2022, 30, 10391–10405. [Google Scholar] [CrossRef]
- Pedersen, L.L.; Sørum, M.E.; Nissen, A.; Gerbek, T.; Kok, K.; Sørensen, K.; Fridh, M.K.; Mølgaard, C.; Müller, K.G. Dietary Micronutrient Intake in Long-Term Survivors of Pediatric Hematopoietic Stem Cell Transplantation. Nutrients 2025, 17, 1663. [Google Scholar] [CrossRef] [PubMed]
- Dhivya, S.; Keshav Narayan, A.; Logith Kumar, R.; Viji Chandran, S.; Vairamani, M.; Selvamurugan, N. Proliferation and Differentiation of Mesenchymal Stem Cells on Scaffolds Containing Chitosan, Calcium Polyphosphate and Pigeonite for Bone Tissue Engineering. Cell Prolif. 2018, 51, e12408. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; Xu, H.; Wang, C.; Qin, H.; An, Z. Magnesium Enhances the Chondrogenic Differentiation of Mesenchymal Stem Cells by Inhibiting Activated Macrophage-Induced Inflammation. Sci. Rep. 2018, 8, 3406. [Google Scholar] [CrossRef]
- Li, M.; Jiang, Y.; Hou, Q.; Zhao, Y.; Zhong, L.; Fu, X. Potential Pre-Activation Strategies for Improving Therapeutic Efficacy of Mesenchymal Stem Cells: Current Status and Future Prospects. Stem Cell Res. Ther. 2022, 13, 146. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.; Heo, J.; Ju, H.; Shin, J.-W.; Kim, Y.; Lee, S.; Yu, H.Y.; Ryu, C.-M.; Yun, H.; Song, S.; et al. Glutathione Dynamics Determine the Therapeutic Efficacy of Mesenchymal Stem Cells for Graft-versus-Host Disease via CREB1-NRF2 Pathway. Sci. Adv. 2020, 6, eaba1334. [Google Scholar] [CrossRef]
- Khader, A.; Arinzeh, T.L. Biodegradable Zinc Oxide Composite Scaffolds Promote Osteochondral Differentiation of Mesenchymal Stem Cells. Biotechnol. Bioeng. 2020, 117, 194–209. [Google Scholar] [CrossRef]
- Watanabe, J.; Yamada, M.; Niibe, K.; Zhang, M.; Kondo, T.; Ishibashi, M.; Egusa, H. Preconditioning of Bone Marrow-Derived Mesenchymal Stem Cells with N-Acetyl-L-Cysteine Enhances Bone Regeneration via Reinforced Resistance to Oxidative Stress. Biomaterials 2018, 185, 25–38. [Google Scholar] [CrossRef]
- Park, K.H.; Choi, Y.; Yoon, D.S.; Lee, K.-M.; Kim, D.; Lee, J.W. Zinc Promotes Osteoblast Differentiation in Human Mesenchymal Stem Cells Via Activation of the cAMP-PKA-CREB Signaling Pathway. Stem Cells Dev. 2018, 27, 1125–1135. [Google Scholar] [CrossRef]
- Fujisawa, K.; Hara, K.; Takami, T.; Okada, S.; Matsumoto, T.; Yamamoto, N.; Sakaida, I. Evaluation of the Effects of Ascorbic Acid on Metabolism of Human Mesenchymal Stem Cells. Stem Cell Res. Ther. 2018, 9, 93. [Google Scholar] [CrossRef]
- EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA); Turck, D.; Bohn, T.; Cámara, M.; Castenmiller, J.; de Henauw, S.; Hirsch-Ernst, K.; Jos, A.; Maciuk, A.; Mangelsdorf, I.; et al. Guidance for Establishing and Applying Tolerable Upper Intake Levels for Vitamins and Essential Minerals. EFSA J. 2024, 22, e9052. [Google Scholar] [CrossRef]
- EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA); Turck, D.; Bohn, T.; Castenmiller, J.; de Henauw, S.; Hirsch-Ernst, K.; Knutsen, H.K.; Maciuk, A.; Mangelsdorf, I.; McArdle, H.J.; et al. Scientific Opinion on the Tolerable Upper Intake Level for Preformed Vitamin A and Β-carotene. EFSA J. 2024, 22, e8814. [Google Scholar] [CrossRef]
- Tang, X.-H.; Gudas, L.J. Retinoids, Retinoic Acid Receptors, and Cancer. Annu. Rev. Pathol. Mech. Dis. 2011, 6, 345–364. [Google Scholar] [CrossRef]
- Toledo-Guzmán, M.E.; Hernández, M.I.; Gómez-Gallegos, Á.A.; Ortiz-Sánchez, E. ALDH as a Stem Cell Marker in Solid Tumors. Curr. Stem Cell Res. Ther. 2019, 14, 375–388. [Google Scholar] [CrossRef]
- Chen, H.; Shi, Y.; Sun, L.; Ni, S. Electrospun Composite Nanofibers with All-Trans Retinoic Acid and MWCNTs-OH against Cancer Stem Cells. Life Sci. 2020, 378, 118152. [Google Scholar] [CrossRef]
- Wang, S.; Liu, J.; Wu, H.; Jiang, A.; Zhao, K.; Yan, K.; Wu, W.; Han, H.; Zhang, Y.; Yang, W.; et al. All-Trans Retinoic Acid (ATRA) Inhibits Insufficient Radiofrequency Ablation (IRFA)-Induced Enrichment of Tumor-Initiating Cells in Hepatocellular Carcinoma. Chin. J. Cancer Res. 2021, 33, 694–707. [Google Scholar] [CrossRef] [PubMed]
- Shao, X.; Xiang, S.; Chen, Y.; Zhang, N.; Cao, J.; Zhu, H.; Yang, B.; Zhou, Q.; Ying, M.; He, Q. Inhibition of M2-like Macrophages by All-Trans Retinoic Acid Prevents Cancer Initiation and Stemness in Osteosarcoma Cells. Acta Pharmacol. Sin. 2019, 40, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Chiodi, I.; Mondello, C. Life Style Factors, Tumor Cell Plasticity and Cancer Stem Cells. Mutat. Res. Mutat. Res. 2020, 784, 108308. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.; Wang, L.; Huang, H.; Li, X.; Wang, P.; Mi, K.; Cheng, J.; Liu, H.; Gu, C.; Huang, L.; et al. All-Trans Retinoic Acid Reduces Cancer Stem Cell-like Cell-Mediated Resistance to Gefitinib in NSCLC Adenocarcinoma Cells. BMC Cancer 2020, 20, 315. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.C.; Kang, H.J.; Kim, Y.S.; Choi, E.C. All-Trans-Retinoic Acid Inhibits Growth of Head and Neck Cancer Stem Cells by Suppression of Wnt/β-Catenin Pathway. Eur. J. Cancer 2012, 48, 3310–3318. [Google Scholar] [CrossRef]
- Nguyen, P.H.; Giraud, J.; Staedel, C.; Chambonnier, L.; Dubus, P.; Chevret, E.; Bœuf, H.; Gauthereau, X.; Rousseau, B.; Fevre, M.; et al. All-Trans Retinoic Acid Targets Gastric Cancer Stem Cells and Inhibits Patient-Derived Gastric Carcinoma Tumor Growth. Oncogene 2016, 35, 5619–5628. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kot, M.; Bronowicka-Adamska, P.; Tyszka-Czochara, M. Regulation of Human Stem Cells by Functional Food Components: How Vitamins, Minerals and Phytochemicals Influence Mesenchymal Stem Cells’ Fate and Function. Nutrients 2025, 17, 3548. https://doi.org/10.3390/nu17223548
Kot M, Bronowicka-Adamska P, Tyszka-Czochara M. Regulation of Human Stem Cells by Functional Food Components: How Vitamins, Minerals and Phytochemicals Influence Mesenchymal Stem Cells’ Fate and Function. Nutrients. 2025; 17(22):3548. https://doi.org/10.3390/nu17223548
Chicago/Turabian StyleKot, Marta, Patrycja Bronowicka-Adamska, and Malgorzata Tyszka-Czochara. 2025. "Regulation of Human Stem Cells by Functional Food Components: How Vitamins, Minerals and Phytochemicals Influence Mesenchymal Stem Cells’ Fate and Function" Nutrients 17, no. 22: 3548. https://doi.org/10.3390/nu17223548
APA StyleKot, M., Bronowicka-Adamska, P., & Tyszka-Czochara, M. (2025). Regulation of Human Stem Cells by Functional Food Components: How Vitamins, Minerals and Phytochemicals Influence Mesenchymal Stem Cells’ Fate and Function. Nutrients, 17(22), 3548. https://doi.org/10.3390/nu17223548









