The Natural Product Parthenolide Inhibits Both Angiogenesis and Invasiveness and Improves Gemcitabine Resistance by Suppressing Nuclear Factor κB Activation in Pancreatic Cancer Cell Lines
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Cell Lines and Cell Culture
2.3. Establishment of PaCa Cell Lines Resistant to Gem
2.4. Cytotoxicity Assay
2.5. Immunocytochemical Analysis of NF-κB p65 Localization
2.6. Nuclear Protein Extraction and NF-κB p65 Activity Assays
2.7. Western Blotting of Multidrug Resistance Protein 1 (MRP1)
2.8. ELISA
2.9. Matrigel Invasion Assay
2.10. Tube Formation Assay for Angiogenesis
2.11. Colony Formation Assay
2.12. Statistical Analysis
3. Results
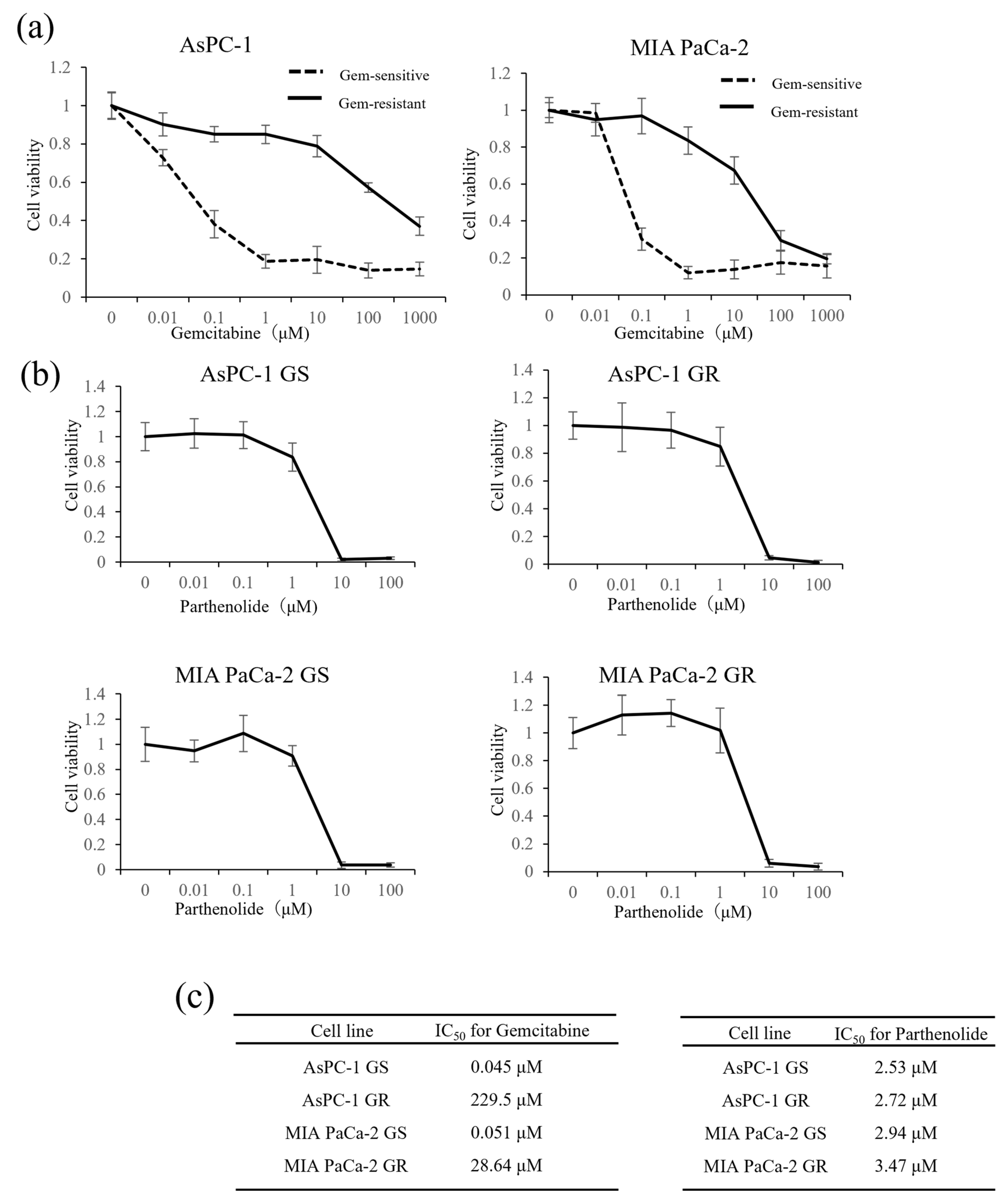
3.1. IC50 Values of Gem and Parthenolide in PaCa Cell Lines
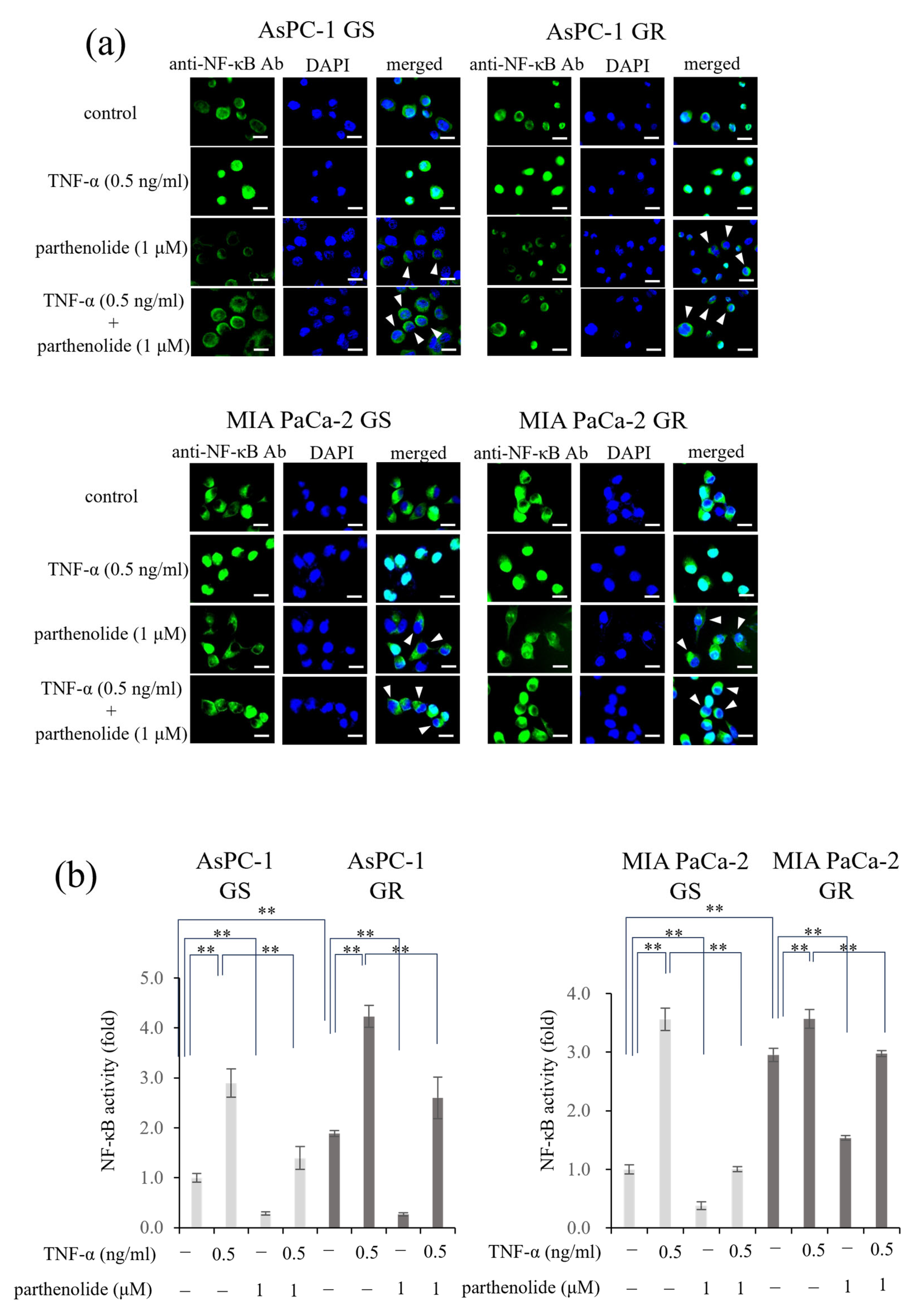
3.2. PaCa Increases p65 Activity Induced by Gem Resistance, While Parthenolide Inhibits p65 Activity by Blocking the Nuclear Translocation of p65
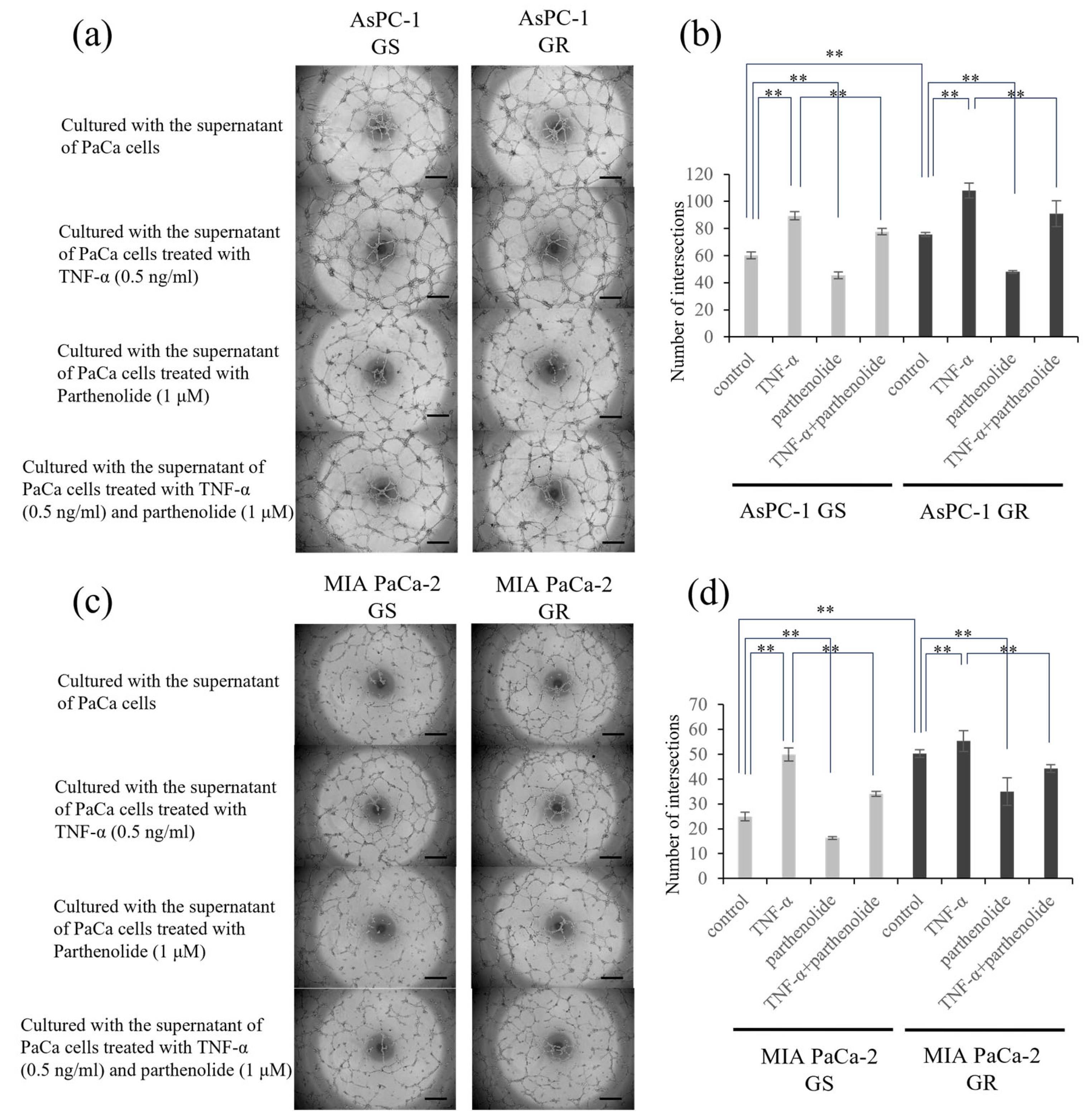
3.3. The Invasion Ability of PaCa Is Increased by Gem Resistance, but Suppressed by Parthenolide
3.4. Angiogenesis of PaCa Is Increased by Gem Resistance, but Suppressed by Parthenolide
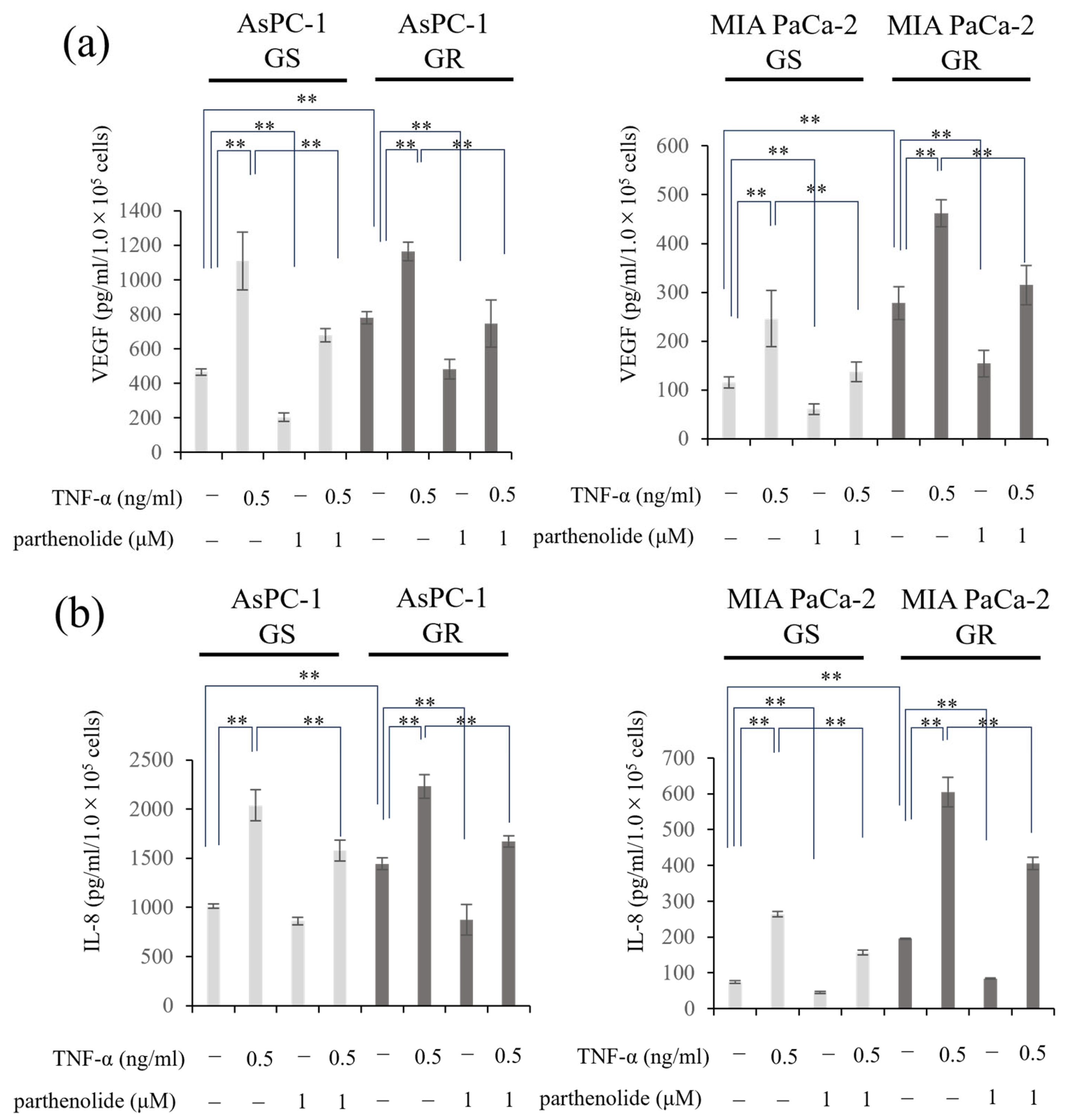
3.5. Parthenolide Suppresses the Secretion of IL-8 and VEGF from PaCa Cells
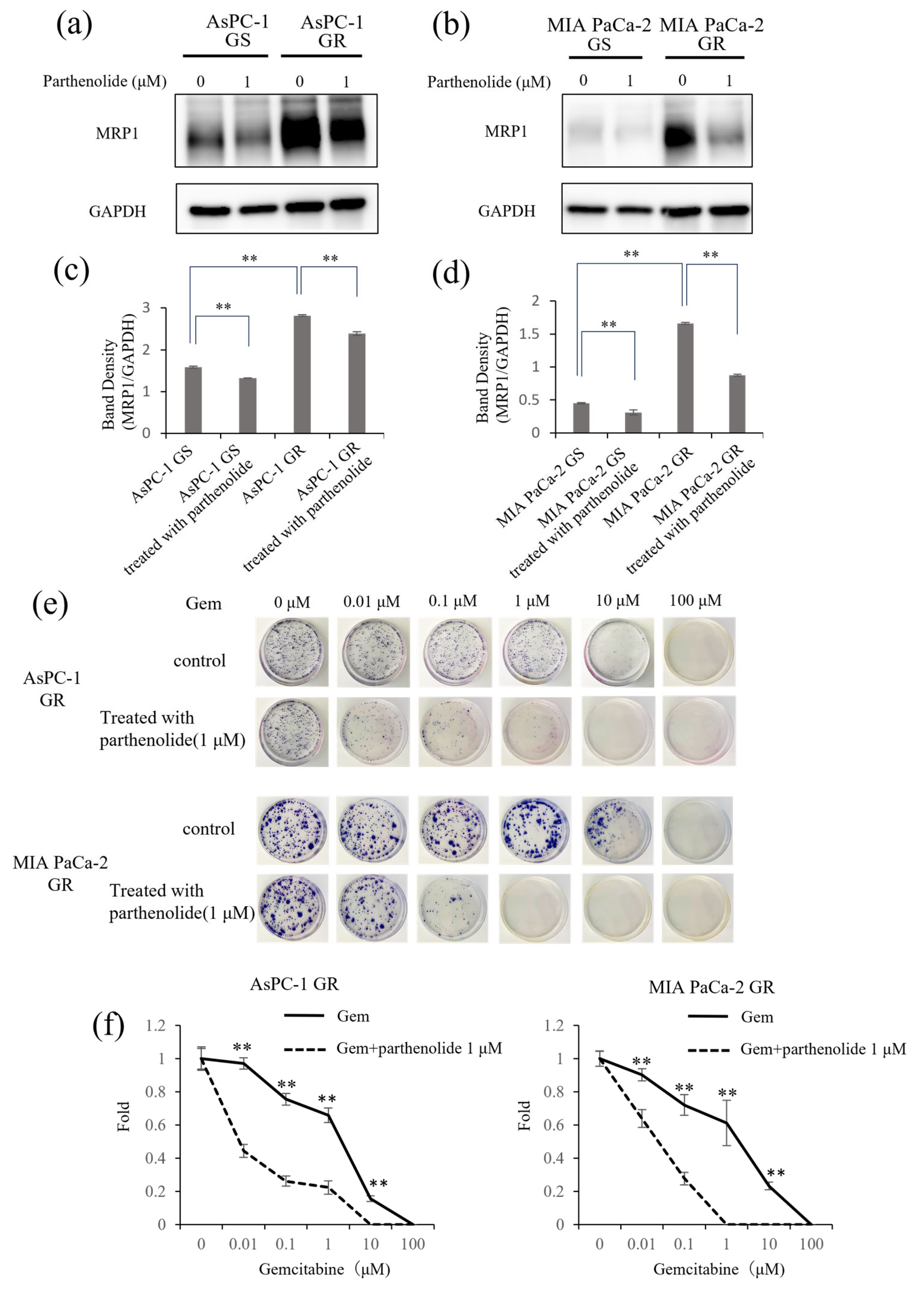
3.6. Parthenolide Suppresses the Expression of the Multidrug-Resistance-Related Protein MRP1 and Improves the Sensitivity to Gem
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- National Cancer Center Japan, Center for Cancer Control and Information Services. 2023. Available online: https://ganjoho.jp/reg_stat/statistics/stat/summary.html (accessed on 17 February 2024).
- Dyba, T.; Randi, G.; Bray, F.; Martos, C.; Giusti, F.; Nicholson, N.; Gavin, A.; Flego, M.; Neamtiu, L.; Dimitrova, N.; et al. The European cancer burden in 2020: Incidence and mortality estimates for 40 countries and 25 major cancers. Eur. J. Cancer 2021, 157, 308–347. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Chen, H.; Lu, M.; Zhang, Y.; Lu, B.; You, L.; Zhang, T.; Dai, M.; Zhao, Y. Advances in the epidemiology of pancreatic cancer: Trends, risk factors, screening, and prognosis. Cancer Lett. 2021, 520, 1–11. [Google Scholar] [CrossRef]
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting cancer incidence and deaths to 2030: The unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014, 74, 2913–2921. [Google Scholar] [CrossRef] [PubMed]
- Katsuda, M.; Miyazawa, M.; Ojima, T.; Katanuma, A.; Hakamada, K.; Sudo, K.; Asahara, S.; Endo, I.; Ueno, M.; Hara, K.; et al. A double-blind randomized comparative clinical trial to evaluate the safety and efficacy of dendritic cell vaccine loaded with WT1 peptides (TLP0-001) in combination with S-1 in patients with advanced pancreatic cancer refractory to standard chemotherapy. Trials 2019, 20, 242. [Google Scholar] [CrossRef] [PubMed]
- Ioka, T.; Ueno, M.; Ueno, H.; Park, J.O.; Chang, H.M.; Sasahira, N.; Kanai, M.; Chung, I.J.; Ikeda, M.; Nakamori, S.; et al. TAS-118 (S-1 plus leucovorin) versus S-1 in patients with gemcitabine-refractory advanced pancreatic cancer: A randomised, open-label, phase 3 study (GRAPE trial). Eur. J. Cancer 2019, 106, 78–88. [Google Scholar] [CrossRef]
- Oettle, H.; Riess, H.; Stieler, J.M.; Heil, G.; Schwaner, I.; Seraphin, J.; Görner, M.; Mölle, M.; Greten, T.F.; Lakner, V.; et al. Second-line oxaliplatin, folinic acid, and fluorouracil versus folinic acid and fluorouracil alone for gemcitabine-refractory pancreatic cancer: Outcomes from the CONKO-003 trial. J. Clin. Oncol. 2014, 32, 2423–2429. [Google Scholar] [CrossRef] [PubMed]
- Yoo, C.; Hwang, J.Y.; Kim, J.E.; Kim, T.W.; Lee, J.S.; Park, D.H.; Lee, S.S.; Seo, D.W.; Lee, S.K.; Kim, M.H.; et al. A randomised phase II study of modified FOLFIRI.3 vs modified FOLFOX as second-line therapy in patients with gemcitabine-refractory advanced pancreatic cancer. Br. J. Cancer 2009, 101, 1658–1663. [Google Scholar] [CrossRef] [PubMed]
- Burris, H.A., 3rd; Moore, M.J.; Andersen, J.; Green, M.R.; Rothenberg, M.L.; Modiano, M.R.; Cripps, M.C.; Portenoy, R.K.; Storniolo, A.M.; Tarassoff, P.; et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: A randomized trial. J. Clin. Oncol. 1997, 15, 2403–2413. [Google Scholar] [CrossRef]
- Mizrahi, J.D.; Surana, R.; Valle, J.W.; Shroff, R.T. Pancreatic cancer. Lancet 2020, 395, 2008–2020. [Google Scholar] [CrossRef]
- Motoi, F.; Kosuge, T.; Ueno, H.; Yamaue, H.; Satoi, S.; Sho, M.; Honda, G.; Matsumoto, I.; Wada, K.; Furuse, J.; et al. Randomized phase II/III trial of neoadjuvant chemotherapy with gemcitabine and S-1 versus upfront surgery for resectable pancreatic cancer (Prep-02/JSAP05). Jpn. J. Clin. Oncol. 2019, 49, 190–194. [Google Scholar] [CrossRef]
- Imafuji, H.; Matsuo, Y.; Ueda, G.; Omi, K.; Hayashi, Y.; Saito, K.; Tsuboi, K.; Morimoto, M.; Koide, S.; Ogawa, R.; et al. Acquisition of gemcitabine resistance enhances angiogenesis via upregulation of IL-8 production in pancreatic cancer. Oncol. Rep. 2019, 41, 3508–3516. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, M.; Matsuo, Y.; Koide, S.; Tsuboi, K.; Shamoto, T.; Sato, T.; Saito, K.; Takahashi, H.; Takeyama, H. Enhancement of the CXCL12/CXCR4 axis due to acquisition of gemcitabine resistance in pancreatic cancer: Effect of CXCR4 antagonists. BMC Cancer 2016, 16, 305. [Google Scholar] [CrossRef]
- Melisi, D.; Chiao, P.J. NF-kappa B as a target for cancer therapy. Expert Opin. Ther. Targets 2007, 11, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Lin, Z.N.; Yang, C.F.; Shi, X.; Ong, C.N.; Shen, H.M. Suppressed NF-kappaB and sustained JNK activation contribute to the sensitization effect of parthenolide to TNF-alpha-induced apoptosis in human cancer cells. Carcinogenesis 2004, 25, 2191–2199. [Google Scholar] [CrossRef] [PubMed]
- Niu, J.; Li, Z.; Peng, B.; Chiao, P.J. Identification of an autoregulatory feedback pathway involving interleukin-1alpha in induction of constitutive NF-kappaB activation in pancreatic cancer cells. J. Biol. Chem. 2004, 279, 16452–16462. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, Y.; Ahmad, A.; Banerjee, S.; Azmi, A.S.; Kong, D.; Sarkar, F.H. Pancreatic cancer: Understanding and overcoming chemoresistance. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Pramanik, K.C.; Makena, M.R.; Bhowmick, K.; Pandey, M.K. Advancement of NF-κB Signaling Pathway: A Novel Target in Pancreatic Cancer. Int. J. Mol. Sci. 2018, 19, 3890. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, A.S. Control of oncogenesis and cancer therapy resistance by the transcription factor NF-kappaB. J Clin. Investig. 2001, 107, 241–246. [Google Scholar] [CrossRef]
- Bork, P.M.; Schmitz, M.L.; Kuhnt, M.; Escher, C.; Heinrich, M. Sesquiterpene lactone containing Mexican Indian medicinal plants and pure sesquiterpene lactones as potent inhibitors of transcription factor NF-kappaB. FEBS Lett. 1997, 402, 85–90. [Google Scholar] [CrossRef]
- Di Cesare Mannelli, L.; Tenci, B.; Zanardelli, M.; Maidecchi, A.; Lugli, A.; Mattoli, L.; Ghelardini, C. Widespread pain reliever profile of a flower extract of Tanacetum parthenium. Phytomedicine 2015, 22, 752–758. [Google Scholar] [CrossRef] [PubMed]
- Shah, V.O.; Ferguson, J.E.; Hunsaker, L.A.; Deck, L.M.; Vander Jagt, D.L. Natural products inhibit LPS-induced activation of pro-inflammatory cytokines in peripheral blood mononuclear cells. Nat. Prod. Res. 2010, 24, 1177–1188. [Google Scholar] [CrossRef] [PubMed]
- Juliana, C.; Fernandes-Alnemri, T.; Wu, J.; Datta, P.; Solorzano, L.; Yu, J.W.; Meng, R.; Quong, A.A.; Latz, E.; Scott, C.P.; et al. Anti-inflammatory compounds parthenolide and Bay 11-7082 are direct inhibitors of the inflammasome. J. Biol. Chem. 2010, 285, 9792–9802. [Google Scholar] [CrossRef] [PubMed]
- García-Piñeres, A.J.; Lindenmeyer, M.T.; Merfort, I. Role of cysteine residues of p65/NF-kappaB on the inhibition by the sesquiterpene lactone parthenolide and N-ethyl maleimide, and on its transactivating potential. Life Sci. 2004, 75, 841–856. [Google Scholar] [CrossRef]
- Liu, X.; Gao, Z.; Wang, X.; Shen, Y. Parthenolide targets NF-κB (P50) to inhibit HIF-1α-mediated metabolic reprogramming of HCC. Aging 2022, 14, 8346–8356. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Huang, R.; Li, M.; Zhu, Z.; Chen, Z.; Cui, L.; Luo, H.; Luo, L. Parthenolide inhibits the growth of non-small cell lung cancer by targeting epidermal growth factor receptor. Cancer Cell Int. 2020, 20, 561. [Google Scholar] [CrossRef]
- Kim, S.L.; Trang, K.T.; Kim, S.H.; Kim, I.H.; Lee, S.O.; Lee, S.T.; Kim, D.G.; Kim, S.W. Parthenolide suppresses tumor growth in a xenograft model of colorectal cancer cells by inducing mitochondrial dysfunction and apoptosis. Int. J. Oncol. 2012, 41, 1547–1553. [Google Scholar] [CrossRef]
- Shanmugam, R.; Kusumanchi, P.; Cheng, L.; Crooks, P.; Neelakantan, S.; Matthews, W.; Nakshatri, H.; Sweeney, C.J. A water-soluble parthenolide analogue suppresses in vivo prostate cancer growth by targeting NFkappaB and generating reactive oxygen species. Prostate 2010, 70, 1074–1086. [Google Scholar] [CrossRef]
- Guzman, M.L.; Jordan, C.T. Feverfew: Weeding out the root of leukaemia. Expert Opin. Biol. Ther. 2005, 5, 1147–1152. [Google Scholar] [CrossRef]
- Huang, L.; Liu, F.; Liu, X.; Niu, L.; Sun, L.; Fang, F.; Ma, K.; Hu, P. Parthenolide inhibits the proliferation and migration of cervical cancer cells via FAK/GSK3β pathway. Cancer Chemother. Pharmacol. 2023, 93, 203–213. [Google Scholar] [CrossRef]
- Omi, K.; Matsuo, Y.; Ueda, G.; Aoyama, Y.; Kato, T.; Hayashi, Y.; Imafuji, H.; Saito, K.; Tsuboi, K.; Morimoto, M.; et al. Escin inhibits angiogenesis by suppressing interleukin-8 and vascular endothelial growth factor production by blocking nuclear factor-κB activation in pancreatic cancer cell lines. Oncol. Rep. 2021, 45, 1–9. [Google Scholar] [CrossRef]
- Murase, H.; Matsuo, Y.; Denda, Y.; Nonoyama, K.; Kato, T.; Aoyama, Y.; Hayashi, Y.; Imafuji, H.; Saito, K.; Morimoto, M.; et al. Upregulation of integrin-linked kinase enhances tumor progression in gemcitabine-resistant pancreatic cancer. Oncol. Rep. 2023, 50, 1–13. [Google Scholar] [CrossRef]
- Aggarwal, B.B. Nuclear factor-kappaB: The enemy within. Cancer Cell 2004, 6, 203–208. [Google Scholar] [CrossRef]
- Deka, K.; Li, Y. Transcriptional Regulation during Aberrant Activation of NF-κB Signalling in Cancer. Cells 2023, 12, 788. [Google Scholar] [CrossRef] [PubMed]
- Moynagh, P.N. The NF-kappaB pathway. J. Cell Sci. 2005, 118, 4589–4592. [Google Scholar] [CrossRef] [PubMed]
- Hoesel, B.; Schmid, J.A. The complexity of NF-κB signaling in inflammation and cancer. Mol. Cancer 2013, 12, 86. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, Y.; Ochi, N.; Sawai, H.; Yasuda, A.; Takahashi, H.; Funahashi, H.; Takeyama, H.; Tong, Z.; Guha, S. CXCL8/IL-8 and CXCL12/SDF-1alpha co-operatively promote invasiveness and angiogenesis in pancreatic cancer. Int. J. Cancer 2009, 124, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Kontos, C.D.; Annex, B.H. Angiogenesis. Curr. Atheroscler. Rep. 1999, 1, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Le, X.; Abbruzzese, J.L.; Peng, Z.; Qian, C.N.; Tang, H.; Xiong, Q.; Wang, B.; Li, X.C.; Xie, K. Constitutive Sp1 activity is essential for differential constitutive expression of vascular endothelial growth factor in human pancreatic adenocarcinoma. Cancer Res. 2001, 61, 4143–4154. [Google Scholar]
- Brown, L.F.; Berse, B.; Jackman, R.W.; Tognazzi, K.; Manseau, E.J.; Senger, D.R.; Dvorak, H.F. Expression of vascular permeability factor (vascular endothelial growth factor) and its receptors in adenocarcinomas of the gastrointestinal tract. Cancer Res. 1993, 53, 4727–4735. [Google Scholar]
- Sawai, H.; Funahashi, H.; Okada, Y.; Matsuo, Y.; Sakamoto, M.; Yamamoto, M.; Takeyama, H.; Manabe, T. Interleukin-1alpha enhances IL-8 secretion through p38 mitogen-activated protein kinase and reactive oxygen species signaling in human pancreatic cancer cells. Med. Sci. Monit. 2005, 11, Br343–Br350. [Google Scholar]
- Matsuo, Y.; Raimondo, M.; Woodward, T.A.; Wallace, M.B.; Gill, K.R.; Tong, Z.; Burdick, M.D.; Yang, Z.; Strieter, R.M.; Hoffman, R.M.; et al. CXC-chemokine/CXCR2 biological axis promotes angiogenesis in vitro and in vivo in pancreatic cancer. Int. J. Cancer 2009, 125, 1027–1037. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, Y.; Sawai, H.; Ochi, N.; Yasuda, A.; Sakamoto, M.; Takahashi, H.; Funahashi, H.; Takeyama, H.; Guha, S. Proteasome inhibitor MG132 inhibits angiogenesis in pancreatic cancer by blocking NF-kappaB activity. Dig. Dis. Sci. 2010, 55, 1167–1176. [Google Scholar] [CrossRef] [PubMed]
- Benckert, C.; Thelen, A.; Cramer, T.; Weichert, W.; Gaebelein, G.; Gessner, R.; Jonas, S. Impact of microvessel density on lymph node metastasis and survival after curative resection of pancreatic cancer. Surg. Today 2012, 42, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Amin, Z.; Theis, B.; Russell, R.C.; House, C.; Novelli, M.; Lees, W.R. Diagnosing pancreatic cancer: The role of percutaneous biopsy and CT. Clin. Radiol. 2006, 61, 996–1002. [Google Scholar] [CrossRef] [PubMed]
- Stipa, F.; Lucandri, G.; Limiti, M.R.; Bartolucci, P.; Cavallini, M.; Di Carlo, V.; D’Amato, A.; Ribotta, G.; Stipa, S. Angiogenesis as a prognostic indicator in pancreatic ductal adenocarcinoma. Anticancer Res. 2002, 22, 445–449. [Google Scholar] [PubMed]
- Annese, T.; Tamma, R.; Ruggieri, S.; Ribatti, D. Angiogenesis in Pancreatic Cancer: Pre-Clinical and Clinical Studies. Cancers 2019, 11, 381. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Ji, S.; Zhang, B.; Liu, J.; Qin, Y.; Xu, J.; Yu, X. Role of angiogenesis in pancreatic cancer biology and therapy. Biomed. Pharmacother. 2018, 108, 1135–1140. [Google Scholar] [CrossRef]
- Saito, K.; Matsuo, Y.; Imafuji, H.; Okubo, T.; Maeda, Y.; Sato, T.; Shamoto, T.; Tsuboi, K.; Morimoto, M.; Takahashi, H.; et al. Xanthohumol inhibits angiogenesis by suppressing nuclear factor-κB activation in pancreatic cancer. Cancer Sci. 2018, 109, 132–140. [Google Scholar] [CrossRef]
- Sebens, S.; Arlt, A.; Schäfer, H. NF-kappaB as a molecular target in the therapy of pancreatic carcinoma. Recent Results Cancer Res. 2008, 177, 151–164. [Google Scholar]
- Jia, Z. Role of integrin-linked kinase in drug resistance of lung cancer. Onco. Targets Ther. 2015, 8, 1561–1565. [Google Scholar] [CrossRef][Green Version]
- Abolhoda, A.; Wilson, A.E.; Ross, H.; Danenberg, P.V.; Burt, M.; Scotto, K.W. Rapid activation of MDR1 gene expression in human metastatic sarcoma after in vivo exposure to doxorubicin. Clin. Cancer Res. 1999, 5, 3352–3356. [Google Scholar]
- Vasiliou, V.; Vasiliou, K.; Nebert, D.W. Human ATP-binding cassette (ABC) transporter family. Hum. Genom. 2009, 3, 281–290. [Google Scholar] [CrossRef]
- Adamska, A.; Falasca, M. ATP-binding cassette transporters in progression and clinical outcome of pancreatic cancer: What is the way forward? World J. Gastroenterol. 2018, 24, 3222–3238. [Google Scholar] [CrossRef]
- Chang, X.B. A molecular understanding of ATP-dependent solute transport by multidrug resistance-associated protein MRP1. Cancer Metastasis Rev. 2007, 26, 15–37. [Google Scholar] [CrossRef]
- Mohelnikova-Duchonova, B.; Brynychova, V.; Oliverius, M.; Honsova, E.; Kala, Z.; Muckova, K.; Soucek, P. Differences in transcript levels of ABC transporters between pancreatic adenocarcinoma and nonneoplastic tissues. Pancreas 2013, 42, 707–716. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.; Du, J.; Gu, J.; Deng, J.; Guo, Y.; Tao, B.; Jin, C.; Fu, D.; Li, J. Gemcitabine-Loaded Albumin Nanoparticle Exerts An Antitumor Effect on Gemcitabine-Resistant Pancreatic Cancer Cells Induced by MDR1 and MRP1 Overexpression in Vitro. Front. Surg. 2022, 9, 890412. [Google Scholar] [CrossRef]
- Lin, S.; Hoffmann, K.; Xiao, Z.; Jin, N.; Galli, U.; Mohr, E.; Büchler, M.W.; Schemmer, P. MEK inhibition induced downregulation of MRP1 and MRP3 expression in experimental hepatocellular carcinoma. Cancer Cell Int. 2013, 13, 3. [Google Scholar] [CrossRef] [PubMed]
- Wattanawongdon, W.; Hahnvajanawong, C.; Namwat, N.; Kanchanawat, S.; Boonmars, T.; Jearanaikoon, P.; Leelayuwat, C.; Techasen, A.; Seubwai, W. Establishment and characterization of gemcitabine-resistant human cholangiocarcinoma cell lines with multidrug resistance and enhanced invasiveness. Int. J. Oncol. 2015, 47, 398–410. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Denda, Y.; Matsuo, Y.; Sugita, S.; Eguchi, Y.; Nonoyama, K.; Murase, H.; Kato, T.; Imafuji, H.; Saito, K.; Morimoto, M.; et al. The Natural Product Parthenolide Inhibits Both Angiogenesis and Invasiveness and Improves Gemcitabine Resistance by Suppressing Nuclear Factor κB Activation in Pancreatic Cancer Cell Lines. Nutrients 2024, 16, 705. https://doi.org/10.3390/nu16050705
Denda Y, Matsuo Y, Sugita S, Eguchi Y, Nonoyama K, Murase H, Kato T, Imafuji H, Saito K, Morimoto M, et al. The Natural Product Parthenolide Inhibits Both Angiogenesis and Invasiveness and Improves Gemcitabine Resistance by Suppressing Nuclear Factor κB Activation in Pancreatic Cancer Cell Lines. Nutrients. 2024; 16(5):705. https://doi.org/10.3390/nu16050705
Chicago/Turabian StyleDenda, Yuki, Yoichi Matsuo, Saburo Sugita, Yuki Eguchi, Keisuke Nonoyama, Hiromichi Murase, Tomokatsu Kato, Hiroyuki Imafuji, Kenta Saito, Mamoru Morimoto, and et al. 2024. "The Natural Product Parthenolide Inhibits Both Angiogenesis and Invasiveness and Improves Gemcitabine Resistance by Suppressing Nuclear Factor κB Activation in Pancreatic Cancer Cell Lines" Nutrients 16, no. 5: 705. https://doi.org/10.3390/nu16050705
APA StyleDenda, Y., Matsuo, Y., Sugita, S., Eguchi, Y., Nonoyama, K., Murase, H., Kato, T., Imafuji, H., Saito, K., Morimoto, M., Ogawa, R., Takahashi, H., Mitsui, A., Kimura, M., & Takiguchi, S. (2024). The Natural Product Parthenolide Inhibits Both Angiogenesis and Invasiveness and Improves Gemcitabine Resistance by Suppressing Nuclear Factor κB Activation in Pancreatic Cancer Cell Lines. Nutrients, 16(5), 705. https://doi.org/10.3390/nu16050705





