Not All Maca Is Created Equal: A Review of Colors, Nutrition, Phytochemicals, and Clinical Uses
Abstract
1. Introduction
2. Species of Maca
3. The Significant Role of Environmental Factors and Processing Methods
3.1. Environmental Factors
3.2. Processing Methods
4. The Nutrition of Maca
Nutritional Differences between Colors of Maca
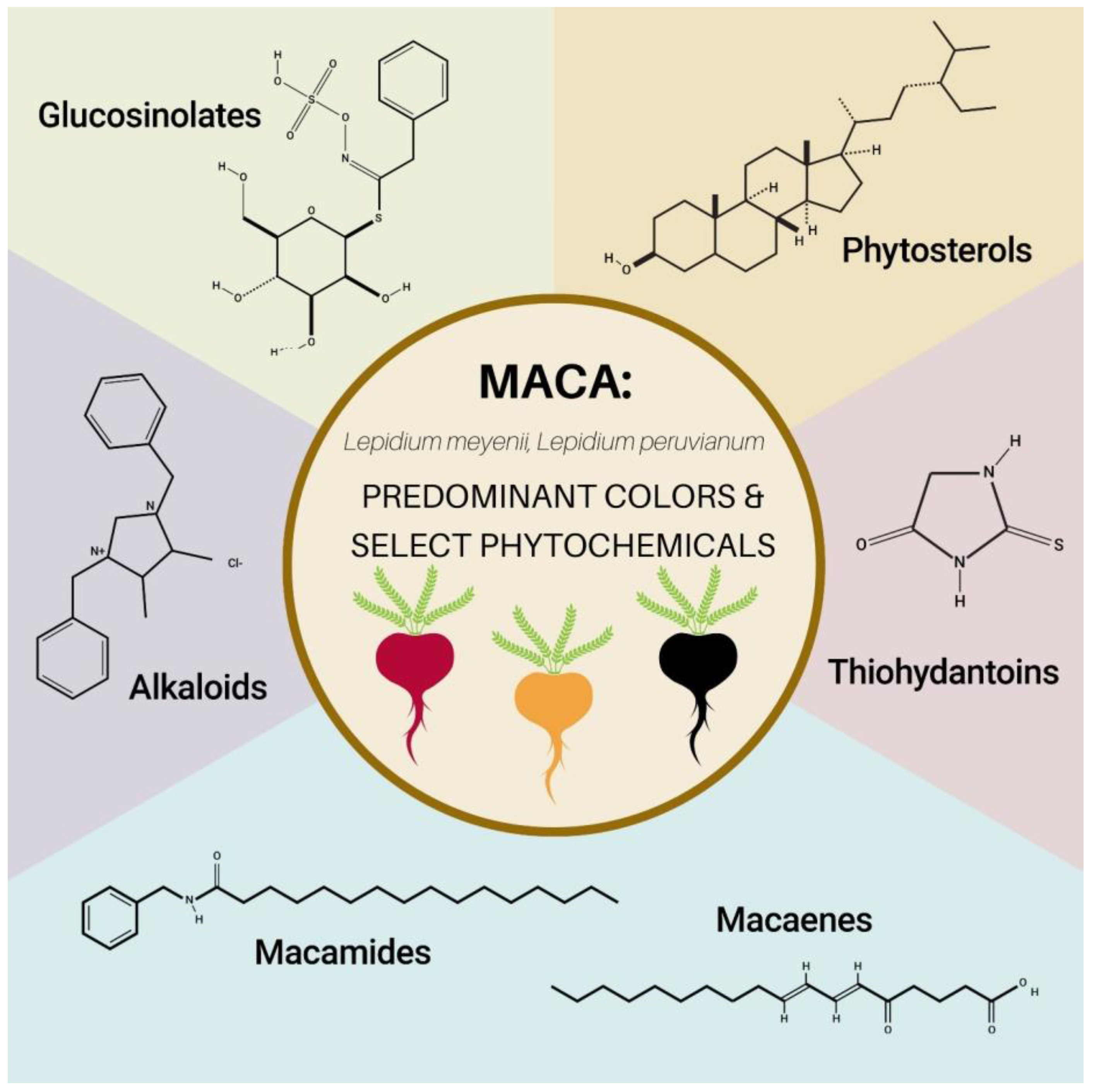
5. Select Maca Phytochemicals
5.1. Glucosinolates
5.2. Alkaloids
5.3. Macamides and Macaenes
5.4. Phytosterols (Plant Sterols)
5.5. Thiohydantins
6. Functions of Maca
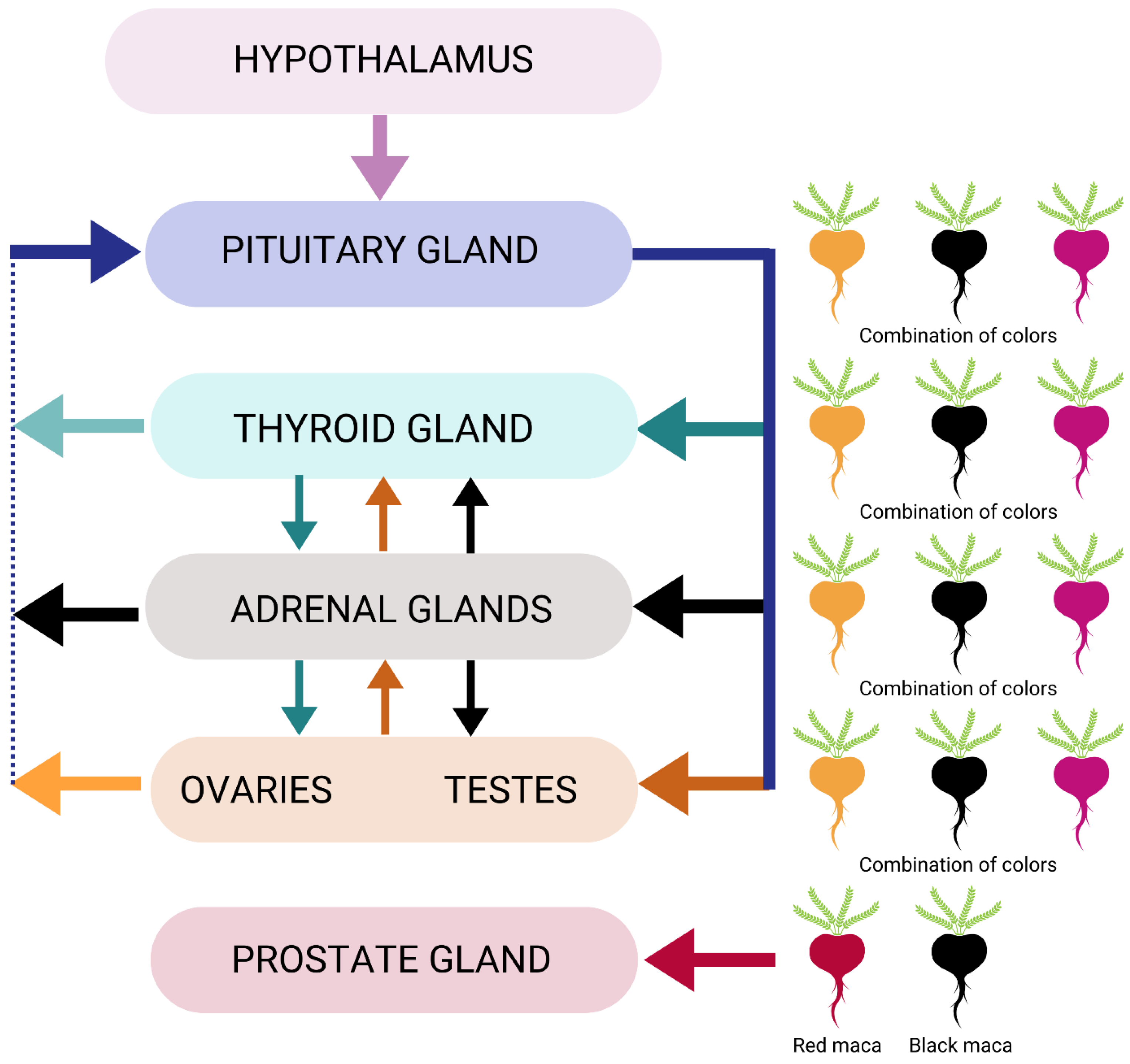
7. Clinical Application: Endocrine System Support
7.1. Adrenal Health
7.2. Ovarian Health
7.3. Testicular and Prostate Health
7.4. Thyroid Health
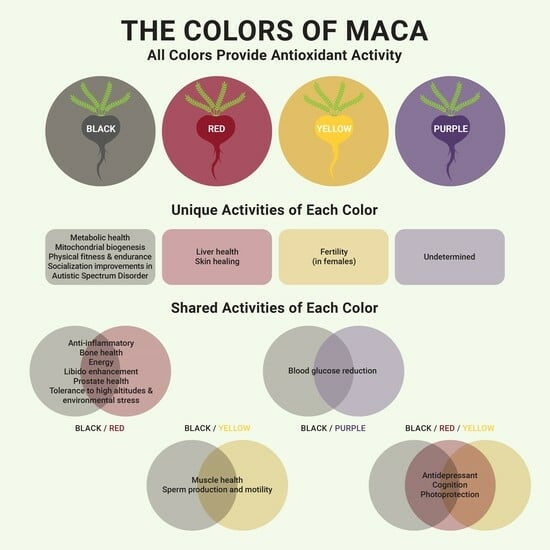
8. Colors (Phenotypes)
9. Black Maca
9.1. Preclinical Studies
9.1.1. Reproductive Health
9.1.2. Brain Health
9.1.3. Metabolic Health
9.1.4. Bone Health
9.1.5. Human Studies (See Table 3)
9.1.6. Clinical Observations
| Study Description | Details on Maca: Species, Product, Location Grown and Cultivation Methods, Form(s), Dose and Route | Overview of Study Results |
|---|---|---|
| 8-week intervention trial [173] in 40 elite athletes: shooting athletes (SA), racket sports athletes (RSA), fin swimming athletes (FSA) | Species: Lepidium meyenii Product: Not stated Location/Cultivation: Not stated Form: 100% concentrated black maca extract Dose and Route: 2300 mg twice daily in the AM and PM | Evaluation of parameters using a digital grip dynamometer and a battery of physical exercises revealed significant increases after black maca supplementation in muscle endurance (p < 0.01 for SA group, p < 0.05 for RSA group), agility (p < 0.05 for SA and RSA groups), muscle strength (p < 0.01 for FSA group), flexibility (p < 0.01 for FSH group), and power (p < 0.05 for RSA group; p < 0.001 for FSA group. After taking black maca for eight weeks, significant differences in lactic acid (mg/dL) (p < 0.05) and total cholesterol (TC, mg/dL) were noted in the SA group (p < 0.01); TC (p < 0.01) and CRP (p < 0.01) in the RSA group; and lactic acid (p < 0.05), TC (p < 0.001), and CRP (p < 0.001) in the FSA group. |
| 4-week study in 10 female athletes [183] Article is published in Korean; abstract only in English, so limited information is available | Species: Unknown Product: Unknown Location/Cultivation: Unknown Form: Unknown Dose and Route: 5 g of maca daily | After four weeks of training and taking black maca, there was a significant decrease in BMI, inflammatory markers, and muscle endurance. |
| 4-week controlled clinical trial [180] in 16 university racket athletes with two groups given black maca: control and resistance exercise (most of article is in Korean) | Species: Not stated Product: Black maca was purchased from Essoco, Seoul, Republic of Korea. Location/Cultivation: Not stated Form: PowderDose and Route: 2.5 g twice daily, taken with water | Statistically significant decrease in ammonia levels (p < 0.05) and increase in muscle endurance and strength (p < 0.05). |
| 12-week, double-blind, randomized, parallel-group, placebo-controlled clinical trial [46] at two hospitals in Korea with 80 men over 40 years old with Aging Males’ Symptoms (AMS) score >/=27; three subjects stopped the trial. | Species: Lepidium meyenii Product: Not stated Location/Cultivation: Not stated Form: Gelatinized powder Dose and Route: 1000 mg, two tablets *, three times daily, total of ~5 g per day, taken before food; each 1000 mg capsule contained 833 mg of maca (* The authors describe the maca as being given in tablet and capsule form.) | AMS, International Index of Erectile Function (IIEF), and International Prostate Symptom Score (IPSS) were significantly improved (p < 0.0001). Androgen Deficiency in the Aging Males was significantly reduced (p < 0.0001), showing an improvement in androgen deficiency symptoms. No changes in T or PSA levels, lipids, body weight, or waist circumference. |
| 12-week randomized controlled trial with 45 men with mild erectile dysfunction (ED), 30–60 years old; [184] reported in [185] | Species: Unknown Product: Unknown Location/Cultivation: Peru Form: Two formats: (1) commercial product, gelatinized and dried, (2) fermented black maca Dose and Route: 5 g in capsules (used for both forms) | Both forms showed significant improvements compared to placebo (p < 0.05). |
| 12-week double-blind placebo-controlled trial [132] in 175 men and women living in low (150 m above sea level, LA) and high altitudes (4340 m above sea level, HA); 18 subjects withdrew | Species: Lepidium meyenii Product: Enterprise A-1 del Perú, Industrial Comercial SAC prepared the finished product. Location/Cultivation: Peru, cultivation methods not stated Form: Spray-dried extract, standardized Dose and Route: 3 g daily | About 50% of participants reported an increase in sexual desire. Effects on mood, energy, and Chronic Mountain Symptom (CMS) scores were better using red maca compared to black maca and placebo. Effects on mood, energy, and CMS scores were better using red maca compared to black maca and placebo. In the red maca group at weeks 8 and 12, 80% of all participants reported increased mood and 90% reported increased energy. Higher quality of life was reported in both red and black maca groups (p < 0.05). Black maca reduced hemoglobin levels in HA participants. In HA, black maca reduced glucose levels in weeks 8 and 12 (p < 0.05), red maca reduced glucose in week 8 (p < 0.01). Systolic blood pressure was reduced in weeks 8 and 12 in the HA group using black maca (p < 0.01). Red maca reduced CMS scores in weeks 4 (p < 0.05), 8 (p < 0.01) and 12 (p < 0.01). Black maca reduced CMS scores in weeks 8 and 12 (p < 0.05). |
10. Red Maca
10.1. Preclinical Studies
10.1.1. Prostate Health
10.1.2. Bone Health
10.1.3. Skin Health
10.1.4. Detoxification
10.1.5. Brain Health
10.1.6. Human Studies (See Table 4)
| Study Description | Details on Maca: Species, Product, Location Grown and Cultivation Methods, Form(s), Dose and Route | Overview of Study Results |
|---|---|---|
| 12-week double-blind placebo-controlled trial [132] in 175 men and women living in low (150 m above sea level, LA) and high altitudes (4340 m above sea level, HA); 18 subjects withdrew | Product: Enterprise A-1 del Perú, Industrial Comercial S.A.C. prepared the finished product. Location/Cultivation: Peru, cultivation methods not stated Form: Spray-dried extract, standardized Dose and Route: 3 g daily | About 50% of participants reported an increase in sexual desire. Effects on mood, energy, and Chronic Mountain Symptom (CMS) scores were better using red maca compared to black maca and placebo. Effects on mood, energy, and CMS scores were better using red maca compared to black maca and placebo. In the red maca group at weeks 8 and 12, 80% of all participants reported increased mood and 90% reported increased energy. Higher quality of life was reported in both red and black maca groups (p < 0.05). Black maca reduced hemoglobin levels in HA participants. In HA, black maca reduced glucose levels in weeks 8 and 12 (p < 0.05), red maca reduced glucose in week 8 (p < 0.01). Systolic blood pressure was reduced in weeks 8 and 12 in the HA group using black maca (p < 0.01). Red maca reduced CMS scores in weeks 4 (p < 0.05), 8 (p < 0.01) and 12 (p < 0.01). Black maca reduced CMS scores in weeks 8 and 12 (p < 0.05). |
11. Yellow Maca
11.1. Preclinical Studies
11.1.1. Musculoskeletal Health
11.1.2. Reproductive Health
11.1.3. Human Studies (See Table 5)
| Study Description | Details on Maca: Species, Product, Location Grown and Cultivation Methods, Form(s), Dose and Route | Overview of Study Results |
|---|---|---|
| 16-week single-center, randomized, double-blind, placebo-controlled trial [193] in 50 infertile men 28–52 years old; 11 dropped out of the study for various reasons including goal achieved (pregnancy of partner), new medical diagnosis, adverse event, and unknown reasons. | Species: Lepidium meyenii Product: Provided by Andean Roots Ltd. Location/Cultivation: Peru, cultivation methods not stated Form: Gelatinized and powdered dried bulb, less than 7% humidity, prepared according to a previous study. Dose and Route: 2.8 g per day (7 capsules of 400 mg each) | In the yellow maca group, sperm count rose 15% (placebo 102%), sperm concentration by 40% (placebo 76%), and there was a significant decrease in free T (27%). Neither group showed a significant improvement in the sperm parameters measured. |
| 12-week, double-blind, randomized, placebo-controlled pilot study [198] in 20 healthy adult men, 20–40 years old, 2 were excluded for oligo-zoospermia | Species: Lepidium meyenii Product: Andean Roots SRL Location/Cultivation: Cerro de Pasco region of Peru 4200–4500 m above sea level, no cultivation methods stated Form: Gelatinized and powdered Dose and Route: 1.75 g per day, 5 capsules containing 350 mg each | Increase in semen parameters after 12 weeks of maca use; increase in sperm count by 20%, sperm concentration by 14%, sperm motility by 14%, semen volume by 9%; morphology of sperm by 21%. No impact on hormone levels. |
12. Purple (Violet) Maca
Preclinical Studies
13. Gray Maca
14. Combination of Colors
14.1. Maca-GO®
14.1.1. Preclinical Studies
14.1.2. Human Studies (See Table 6)
14.2. MACAXSTM
Preclinical Study
| Study Description | Details on Maca: Species, Product, Location Grown and Cultivation Methods, Form(s), Dose and Route | Overview of Study Results |
|---|---|---|
| Case report [23] of a 39-year-old Caucasian female with premenstrual syndrome (PMS), dysmenorrhea, and menorrhagia | Species: Lepidium peruvianum Product: FemmenessencePRO® HARMONY (contains Maca-GO®) from Symphony Natural Health, Inc. Location/Cultivation: Not stated, however, previous research indicates Maca-GO® is cultivated, harvested, dried on the plantation site in Junín, Peru [24]. Form: Not stated; however, previous research states Maca-GO® is gelatinized and standardized [27]. Dose and Route: 500 mg twice daily as capsules, taken orally upon waking and mid-afternoon | In four months, the patient experienced a resolution of multiple PMS symptoms, reporting balanced moods, improved satiety, reduced cystic acne, and regulated bowel movements, as well as complete resolution of menstrual pain, cramping, and lighter menstrual cycles. |
| Case report [146] of a 32-year-old post-menopausal woman who had a hysterectomy and oophorectomy | Species: Lepidium peruvianum Product: FemmenessencePRO® (contains Maca-GO®) from Symphony Natural Health, Inc. Location/Cultivation: Not stated, however, previous research indicates Maca-GO® is cultivated, harvested, and dried on the plantation site in Junín, Peru [24]. Form: Not stated; however, previous research states Maca-GO® is gelatinized and standardized [27]. Dose and Route: 1000 mg twice daily, capsules, taken orally upon waking and mid-afternoon | Resolution of hot flushes and anxiety in two months, improved mood and sleep, as measured by the Kupperman’s Menopausal Index (KMI). |
| Case report [22] of a 30-year-old Caucasian female diagnosed with premenstrual syndrome (PMS) and premenstrual dysphoric disorder (PMDD) | Species: Lepidium peruvianum Product: FemmenessencePRO® HARMONY (contains Maca-GO®) from Symphony Natural Health, Inc. Location/Cultivation: Not stated; however, previous research indicates Maca-GO® is cultivated, harvested, dried on the plantation site in Junín, Peru [24]. Form: Not stated; however, previous research states Maca-GO® is gelatinized and standardized [27]. Dose and Route: 500 milligrams once daily to (or and) 1000 mg twice daily. Doses varied throughout care, and were taken orally upon waking and mid-afternoon. | Improvements in premenstrual headaches, fatigue, menstrual cramping and flow, and intense psychiatric symptoms were reported within three months of taking the maca supplement, bio-identical progesterone therapy, and magnesium. Additionally, LH reduced to normal levels, resulting in a normal FSH/LH ratio. |
| Double-blind, placebo-corrected clinical pilot study [24] in 20 Caucasian, healthy, early post-menopausal women, 45–62 years old; 8 subjects did not complete the trial. There were two trials with one lasting three months and the other nine months. Note: The 3-month study included 1 month of placebo, followed by 2 months of Maca-GO®. The 9-month study included 1 month of placebo, followed by 8 months of Maca-GO®. | Species: Lepidium peruvianum Product: Maca-GO® Location/Cultivation: Junín, Peru (4200 and 4500 m above sea level); cultivated, harvested, and dried on the plantation siteof Junín, organic status Form: Gelatinized, standardized, no chemicals used, maca root powder in capsules Dose & Route: 1000 mg twice daily as capsules, oral intake 30 min before morning and evening meals | In 2 and 8 months, there were significant decreases in FSH (p < 0.05) and increases in LH (p < 0.05); At month 8, there were significant increases in E2 and P (p < 0.05); significant reduction in menopausal symptoms was noted in 2 and 8 months (p < 0.05). |
| 4-month, double blind, crossover, randomized pilot trial [27]: 2 months of placebo and 2 months of Maca-GO® in 20 Caucasian, healthy, menstruating women, 41–50 years old; 2 did not complete the study | Species: Lepidium peruvianum Product: Maca-GO® Location/Cultivation: Junín, Peru (4200 and 4500 m above sea level); cultivated, harvested, and dried on the plantation site of Junín, organic status Form: Gelatinized, standardized, no chemicals used, maca root powder in capsules Dose & Route: 1000 mg twice daily as capsules, oral intake 30 min before morning and evening meals | At two months, 95% of women had improvements in menopausal symptoms as assessed by the Kupperman’s Menopausal index. After two months of taking Maca-GO®, significant reduction in body weight, systolic and diastolic blood pressure (p < 0.05), and increased HDL (p < 0.01) and iron (p < 0.05) levels were noted. Significant reductions in menopausal symptoms were reported: hot flushes, excessive sweating, interrupted sleep (all at p < 0.01), nervousness, depression, heart palpitation (all at p < 0.05); significant increases seen in E2, FSH, and P (all at p < 0.01); significant reduction in ACTH (p < 0.05). |
| Two trials [25] conducted at four clinics in 168 Caucasian, healthy, early menopausal women, 49–58 years old Trial 1: 3-month double-blind, randomized, coordinated multi-center, outpatient clinical study in 102 women (88 completed) Trial 2: 4-month double-blind, randomized, coordinated multi-center, outpatient clinical study in 66 women (40 completed) | Species: Lepidium peruvianum Product: Maca-GO® Location/Cultivation: Junín, Peru (4200 and 4500 m above sea level); cultivated, harvested, and dried on the plantation site of Junín, organic status Form: Gelatinized, standardized, no chemicals used, maca root powder in capsules Dose & Route: 1000 mg twice daily as capsules, oral intake 30 min before morning and evening meals | Trial 1: Significant increase in E2 (p < 0.001), decrease in FSH (p < 0.05), significant increase in HDL levels (p < 0.05); significant decrease in menopausal symptoms in one month (p < 0.001) of treatment and further reduction in the second months of treatments, followed by an increase in symptoms when returning to placebo (p < 0.001). Trial 2: Significant decrease in FSH and LH (p < 0.05), significant increase in E2 (p < 0.05), significant reduction in menopausal symptoms (p < 0.001). |
| Trial 1: 4-month double-blind, randomized, outpatient, four months crossover design clinical trial (2 month of treatment and 2 months of placebo) [26] in 22 Caucasian early post-menopausal women, 49–58 years old Trial 2: Inclusion of Trial 1 plus a pilot bone density observation in 12 Caucasian early post-menopausal women, 49–58 years old | Species: Lepidium peruvianum Product: Maca-GO® Location/Cultivation: Junín, Peru (4200 and 4500 m above sea level); cultivated, harvested, and dried on the plantation site of Junín, organic status Form: Gelatinized, standardized, no chemicals used, maca root powder in capsules Dose and Route: 1000 mg twice daily as capsules; oral intake 30 min before morning and evening meals | Trial 1: Significant decrease in BMI and LH levels (p < 0.05), significant increase in E2 (p < 0.05); significant reductions in T3, cortisol, ACTH (p < 0.05); significant increase in serum iron and plasma calcium levels (p < 0.05); significant decrease in menopausal symptoms (p < 0.001) Trial 2: The group on Maca-GO® had an increase in forearm bone density in four months, whereas the placebo group had a reduction in bone density during the same time. Maca-GO® group had an increase in E2 and a decrease in FSH. |
15. Unspecified Colors of Maca
MacaPure M-01 and M-02
Preclinical Study
16. Overview of Clinical Findings on the Colors of Maca
16.1. Plant Terminology and Cultivation
16.2. Phytochemical Content
16.3. Clinical Efficacy Related to Preparation and Administration
16.4. Clinical Efficacy Related to Color
16.5. Safety of Maca for Human Consumption or Therapeutic Use
16.6. Endocrine System Optimization
17. Emerging Ideas for Future Research
17.1. Newer Clinical Research Studies
17.2. Phytochemicals of Interest
17.3. Effects on Neurotransmitters
17.4. Gut Microbiome Modulation
17.5. Gaps in the Literature on Maca
18. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Meissner, H.O.; Mscisz, A.; Kedzia, B.; Pisulewski, P.; Piatkowska, E. Peruvian Maca: Two Scientific Names Lepidium meyenii Walpers and Lepidium Peruvianum Chacon—Are They Phytochemically-Synonymous? Int. J. Biomed. Sci. 2015, 11, 1–15. [Google Scholar] [CrossRef]
- WFO. Genus Lepidium L. Available online: https://wfoplantlist.org/plant-list/taxon/wfo-4000021089-2023-06?page=1 (accessed on 24 November 2023).
- Gonzales, G.F. Ethnobiology and Ethnopharmacology of Lepidium meyenii (Maca), a Plant from the Peruvian Highlands. Evid. Based Complement. Altern. Med. 2012, 2012, 193496. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, G.F.; Villaorduña, L.; Gasco, M.; Rubio, J.; Gonzales, C. [Maca (Lepidium meyenii Walp.), a review of its biological properties]. Rev. Peru. Med. Exp. Salud Publica 2014, 31, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, G.F.; Gonzales, C.; Gonzales-Castañeda, C. Lepidium meyenii (Maca): A plant from the highlands of Peru—From tradition to science. Forsch. Komplementmed 2009, 16, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Huarancca Reyes, T.; Esparza, E.; Crestani, G.; Limonchi, F.; Cruz, R.; Salinas, N.; Scartazza, A.; Guglielminetti, L.; Cosio, E. Physiological responses of maca (Lepidium meyenii Walp.) plants to U.V. radiation in its high-altitude mountain ecosystem. Sci. Rep. 2020, 10, 2654. [Google Scholar] [CrossRef]
- Zhang, L.; Li, G.; Wang, S.; Yao, W.; Zhu, F. Physicochemical properties of maca starch. Food Chem. 2017, 218, 56–63. [Google Scholar] [CrossRef]
- Beharry, S.; Heinrich, M. Is the hype around the reproductive health claims of maca (Lepidium meyenii Walp.) justified? J. Ethnopharmacol. 2018, 211, 126–170. [Google Scholar] [CrossRef]
- Sun, Y.; Dai, C.; Shi, S.; Zheng, Y.; Wei, W.; Cai, D. Composition analysis and antioxidant activity of essential oils, lipids and polysaccharides in different phenotypes of Lepidium meyenii. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2018, 1099, 25–33. [Google Scholar] [CrossRef]
- Chen, L.; Li, J.; Fan, L. The Nutritional Composition of Maca in Hypocotyls (Lepidium meyenii Walp.) Cultivated in Different Regions of China. J. Food Qual. 2017, 2017, 3749627. [Google Scholar] [CrossRef]
- He, Y.; Sun, Q.; Zhang, X.; Bao, X.; Wang, Y.; Rasheed, M.; Guo, B. Authentication of the geographical origin of Maca (Lepidium meyenii Walp.) at different regional scales using the stable isotope ratio and mineral elemental fingerprints. Food Chem. 2020, 311, 126058. [Google Scholar] [CrossRef]
- Zhu, H.; Wang, R.; Hua, H.; Qian, H.; Du, P. Deciphering the potential role of Maca compounds prescription influencing gut microbiota in the management of exercise-induced fatigue by integrative genomic analysis. Front. Nutr. 2022, 9, 1004174. [Google Scholar] [CrossRef]
- Smith, T. Maca Madness: Chinese Herb Smugglers Create Chaos in the Peruvian Andes Consequences for the market, consumers, and local farming communities. HerbalGram 2015, 105, 46–55. [Google Scholar]
- ABC-AHP-NCNPR Botanical Adulterants Prevention Program Publishes Maca Root and Root Extract Bulletin. 2018. Available online: http://cms.herbalgram.org/press/2018/ABC_AHP_NCNPR_BAPP_Maca.html (accessed on 14 November 2023).
- Brand, E. The Rise and Fall of Maca in China. HerbalGram 2016, 111, 28–30. [Google Scholar]
- Meissner, H.O.; Xu, L.; Wan, W.; Yi, F. Glucosinolates profiles in Maca phenotypes cultivated in Peru and China (Lepidium peruvianum syn. L. meyenii). Phytochem. Lett. 2019, 31, 208–216. [Google Scholar] [CrossRef]
- Wang, Y.H.; Wang, Y.; McNeil, B.; Harvey, L.M. Maca: An Andean crop with multi-pharmacological functions. Food Res. Int. 2007, 40, 783–792. [Google Scholar] [CrossRef]
- Hooper, P.L.; Hooper, P.L.; Tytell, M.; Vígh, L. Xenohormesis: Health benefits from an eon of plant stress response evolution. Cell Stress. Chaperones 2010, 15, 761–770. [Google Scholar] [CrossRef]
- Carvalho, F.V.; Ribeiro, P.R. Structural diversity, biosynthetic aspects, and LC-HRMS data compilation for the identification of bioactive compounds of Lepidium meyenii. Food Res. Int. 2019, 125, 108615. [Google Scholar] [CrossRef]
- Nieman, K.M.; Zhu, Y.; Tucker, M.; Koecher, K. The Role of Dietary Ingredients in Mental Energy—A Scoping Review of Randomized Controlled Trials. J. Am. Nutr. Assoc. 2024, 43, 167–182. [Google Scholar] [CrossRef]
- National Library of Medicine. “Maca”. PubMed. 2023. Available online: https://pubmed.ncbi.nlm.nih.gov/?term=maca&sort=date (accessed on 14 November 2023).
- Fahoum, M.; Ross, K. An Integrative Approach for Improving and Managing Premenstrual Syndrome (PMS) and Premenstrual Dysphoric Disorder (PMDD): A Case Report. Curr. Res. Cmpl Alt. Med. 2023, 7, 211. [Google Scholar]
- Ross, K. Improvements in Premenstrual Syndrome, Primary Dysmenorrhea, and Menorrhagia with Natural Therapies: A Case Report. Curr. Res. Cmpl Alt. Med. 2023, 7, 207. [Google Scholar]
- Meissner, H.O.; Kapczynski, W.; Mscisz, A.; Lutomski, J. Use of gelatinized maca (Lepidium peruvianum) in early postmenopausal women. Int. J. Biomed. Sci. 2005, 1, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Meissner, H.O.; Mscisz, A.; Reich-Bilinska, H.; Kapczynski, W.; Mrozikiewicz, P.; Bobkiewicz-Kozlowska, T.; Kedzia, B.; Lowicka, A.; Barchia, I. Hormone-Balancing Effect of Pre-Gelatinized Organic Maca (Lepidium peruvianum Chacon): (II) Physiological and Symptomatic Responses of Early-Postmenopausal Women to Standardized doses of Maca in Double Blind, Randomized, Placebo-Controlled, Multi-Centre Clinical Study. Int. J. Biomed. Sci. 2006, 2, 360–374. [Google Scholar] [PubMed]
- Meissner, H.O.; Mscisz, A.; Reich-Bilinska, H.; Mrozikiewicz, P.; Bobkiewicz-Kozlowska, T.; Kedzia, B.; Lowicka, A.; Barchia, I. Hormone-Balancing Effect of Pre-Gelatinized Organic Maca (Lepidium peruvianum Chacon): (III) Clinical responses of early-postmenopausal women to Maca in double blind, randomized, Placebo-controlled, crossover configuration, outpatient study. Int. J. Biomed. Sci. 2006, 2, 375–394. [Google Scholar] [CrossRef]
- Meissner, H.O.; Reich-Bilinska, H.; Mscisz, A.; Kedzia, B. Therapeutic Effects of Pre-Gelatinized Maca (Lepidium peruvianum Chacon) used as a Non-Hormonal Alternative to HRT in Perimenopausal Women—Clinical Pilot Study. Int. J. Biomed. Sci. 2006, 2, 143–159. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yu, L.; Ao, M.; Jin, W. Effect of ethanol extract of Lepidium meyenii Walp. on osteoporosis in ovariectomized rat. J. Ethnopharmacol. 2006, 105, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Gasco, M.; Aguilar, J.; Gonzales, G.F. Effect of chronic treatment with three varieties of Lepidium meyenii (Maca) on reproductive parameters and DNA quantification in adult male rats. Andrologia 2007, 39, 151–158. [Google Scholar] [CrossRef]
- Gonzales, C.; Rubio, J.; Gasco, M.; Nieto, J.; Yucra, S.; Gonzales, G.F. Effect of short-term and long-term treatments with three ecotypes of Lepidium meyenii (MACA) on spermatogenesis in rats. J. Ethnopharmacol. 2006, 103, 448–454. [Google Scholar] [CrossRef]
- Gonzales, G.F.; Cordova, A.; Gonzales, C.; Chung, A.; Vega, K.; Villena, A. Lepidium meyenii (Maca) improved semen parameters in adult men. Asian J. Androl. 2001, 3, 301–303. [Google Scholar]
- Gonzales, G.F.; Gonzales-Castañeda, C.; Gasco, M. A mixture of extracts from Peruvian plants (black maca and yacon) improves sperm count and reduced glycemia in mice with streptozotocin-induced diabetes. Toxicol. Mech. Methods 2013, 23, 509–518. [Google Scholar] [CrossRef]
- Gonzales, G.F.; Nieto, J.; Rubio, J.; Gasco, M. Effect of Black maca (Lepidium meyenii) on one spermatogenic cycle in rats. Andrologia 2006, 38, 166–172. [Google Scholar] [CrossRef]
- Inoue, N.; Farfan, C.; Gonzales, G.F. Effect of butanolic fraction of yellow and black maca (Lepidium meyenii) on the sperm count of adult mice. Andrologia 2016, 48, 915–921. [Google Scholar] [CrossRef]
- Lee, H.W.; Lee, M.S.; Qu, F.; Lee, J.W.; Kim, E. Maca (Lepidium meyenii Walp.) on semen quality parameters: A systematic review and meta-analysis. Front. Pharmacol. 2022, 13, 934740. [Google Scholar] [CrossRef]
- Roberto, O.Y.-J.; Ivan, M.Q.-D.; Daniel, A.-A.; Kelly, S.-M.; Albert, V.-G.; Jazminy, M.-G.; Ronald, Y.-M.; Edmundo, A.V.-C.; Rafael, J.-A.; Pedro Buc, C.; et al. Antidepressant-Like Behavioral and Spatial Memory Effects in Peruvian Red Maca (Lepidium meyenii)-Treated Rats. Pharmacogn. J. 2021, 13, 81–88. [Google Scholar]
- Rubio, J.; Qiong, W.; Liu, X.; Jiang, Z.; Dang, H.; Chen, S.L.; Gonzales, G.F. Aqueous Extract of Black Maca (Lepidium meyenii) on Memory Impairment Induced by Ovariectomy in Mice. Evid. Based Complement. Altern. Med. 2011, 2011, 253958. [Google Scholar] [CrossRef] [PubMed]
- Rubio, J.; Yucra, S.; Gasco, M.; Gonzales, G.F. Dose-response effect of black maca (Lepidium meyenii) in mice with memory impairment induced by ethanol. Toxicol. Mech. Methods 2011, 21, 628–634. [Google Scholar] [CrossRef]
- Tarabasz, D.; Szczeblewski, P.; Laskowski, T.; Płaziński, W.; Baranowska-Wójcik, E.; Szwajgier, D.; Kukula-Koch, W.; Meissner, H.O. The Distribution of Glucosinolates in Different Phenotypes of Lepidium peruvianum and Their Role as Acetyl- and Butyrylcholinesterase Inhibitors—In Silico and In Vitro Studies. Int. J. Mol. Sci. 2022, 23, 4858. [Google Scholar] [CrossRef] [PubMed]
- Dording, C.M.; Schettler, P.J.; Dalton, E.D.; Parkin, S.R.; Walker, R.S.W.; Fehling, K.B.; Fava, M.; Mischoulon, D. A Double-Blind Placebo-Controlled Trial of Maca Root as Treatment for Antidepressant-Induced Sexual Dysfunction in Women. Evid. Based Complement. Altern. Med. 2015, 2015, 949036. [Google Scholar] [CrossRef] [PubMed]
- Gasco, M.; Villegas, L.; Yucra, S.; Rubio, J.; Gonzales, G.F. Dose-response effect of Red Maca (Lepidium meyenii) on benign prostatic hyperplasia induced by testosterone enanthate. Phytomedicine 2007, 14, 460–464. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, C.; Leiva-Revilla, J.; Rubio, J.; Gasco, M.; Gonzales, G.F. Effect of red maca (Lepidium meyenii) on prostate zinc levels in rats with testosterone-induced prostatic hyperplasia. Andrologia 2012, 44 (Suppl. S1), 362–369. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, G.F.; Gasco, M.; Malheiros-Pereira, A.; Gonzales-Castañeda, C. Antagonistic effect of Lepidium meyenii (red maca) on prostatic hyperplasia in adult mice. Andrologia 2008, 40, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, G.F.; Miranda, S.; Nieto, J.; Fernández, G.; Yucra, S.; Rubio, J.; Yi, P.; Gasco, M. Red maca (Lepidium meyenii) reduced prostate size in rats. Reprod. Biol. Endocrinol. 2005, 3, 5. [Google Scholar] [CrossRef]
- Gonzales, G.F.; Vasquez, V.; Rodriguez, D.; Maldonado, C.; Mormontoy, J.; Portella, J.; Pajuelo, M.; Villegas, L.; Gasco, M. Effect of two different extracts of red maca in male rats with testosterone-induced prostatic hyperplasia. Asian J. Androl. 2007, 9, 245–251. [Google Scholar] [CrossRef]
- Shin, D.; Jeon, S.H.; Piao, J.; Park, H.J.; Tian, W.J.; Moon, D.G.; Ahn, S.T.; Jeon, K.H.; Zhu, G.Q.; Park, I.; et al. Efficacy and Safety of Maca (Lepidium meyenii) in Patients with Symptoms of Late-Onset Hypogonadism: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. World J. Mens. Health 2023, 41, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Peng, Z.; Lai, W.; Shao, Y.; Gao, Q.; He, M.; Zhou, W.; Guo, L.; Kang, J.; Jin, X.; et al. The Efficient Synthesis and Anti-Fatigue Activity Evaluation of Macamides: The Unique Bioactive Compounds in Maca. Molecules 2023, 28, 3943. [Google Scholar] [CrossRef] [PubMed]
- Orhan, C.; Gencoglu, H.; Tuzcu, M.; Sahin, N.; Ojalvo, S.P.; Sylla, S.; Komorowski, J.R.; Sahin, K. Maca could improve endurance capacity possibly by increasing mitochondrial biogenesis pathways and antioxidant response in exercised rats. J. Food Biochem. 2022, 46, e14159. [Google Scholar] [CrossRef] [PubMed]
- Stone, M.; Ibarra, A.; Roller, M.; Zangara, A.; Stevenson, E. A pilot investigation into the effect of maca supplementation on physical activity and sexual desire in sportsmen. J. Ethnopharmacol. 2009, 126, 574–576. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, G.F.; Córdova, A.; Vega, K.; Chung, A.; Villena, A.; Góñez, C.; Castillo, S. Effect of Lepidium meyenii (MACA) on sexual desire and its absent relationship with serum testosterone levels in adult healthy men. Andrologia 2002, 34, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Vásquez-Velásquez, C.; Gasco, M.; Fano-Sizgorich, D.; Gonzales, G.F. Inflammatory pathway employed by Red Maca to treat induced benign prostatic hyperplasia in rats. Andrologia 2020, 52, e13516. [Google Scholar] [CrossRef] [PubMed]
- Meissner, H.; (National Institute of Complementary Medicine, Health Research Institute, Western Sydney University, Westmead-Sydney, NSW 2145, Australia). Personal communication, 2023.
- Concerto, C.; Rodolico, A.; Meo, V.; Chiappetta, D.; Bonelli, M.; Mineo, L.; Saitta, G.; Stuto, S.; Signorelli, M.S.; Petralia, A.; et al. A Systematic Review on the Effect of Nutraceuticals on Antidepressant-Induced Sexual Dysfunctions: From Basic Principles to Clinical Applications. Curr. Issues Mol. Biol. 2022, 44, 3335–3350. [Google Scholar] [CrossRef] [PubMed]
- Dording, C.M.; Fisher, L.; Papakostas, G.; Farabaugh, A.; Sonawalla, S.; Fava, M.; Mischoulon, D. A double-blind, randomized, pilot dose-finding study of maca root (L. meyenii) for the management of SSRI-induced sexual dysfunction. CNS Neurosci. Ther. 2008, 14, 182–191. [Google Scholar] [CrossRef]
- Al Saffar, H.; Xu, J.; O’Brien, J.S.; Kelly, B.D.; Murphy, D.G.; Lawrentschuk, N. US Food and Drug Administration Warning Regarding Finasteride and Suicidal Ideation: What Should Urologists Know? Eur. Urol. Open Sci. 2023, 52, 4–6. [Google Scholar] [CrossRef]
- Gupta, M.A.; Vujcic, B.; Gupta, A.K. Finasteride Use Is Associated with Higher Odds of Obstructive Sleep Apnea: Results from the US Food and Drug Administration Adverse Events Reporting System. Skinmed 2020, 18, 146–150. [Google Scholar]
- Zito, P.M.; Bistas, K.G.; Syed, K. Finasteride. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Stute, P.; Marsden, J.; Salih, N.; Cagnacci, A. Reappraising 21 years of the W.H.I study: Putting the findings in context for clinical practice. Maturitas 2023, 174, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Friedman, J.; Sheeder, J.; Lazorwitz, A.; Polotsky, A.J. Herbal supplement use among reproductive-aged women in an academic infertility practice. F S Rep. 2023, 4, 104–111. [Google Scholar] [CrossRef]
- Todorova, V.; Ivanov, K.; Ivanova, S. Comparison between the Biological Active Compounds in Plants with Adaptogenic Properties (Rhaponticum carthamoides, Lepidium meyenii, Eleutherococcus senticosus and Panax ginseng). Plants 2021, 11, 64. [Google Scholar] [CrossRef]
- Amazon. “Maca”. Available online: https://www.amazon.com/s?k=maca&i=hpc&rh=n%3A3760901%2Cn%3A23675621011&dc&ds=v1%3A7%2BQFEEDfNtXrSoD6O3X1lQjzbxdkGIR086ThGbSvmww&crid=CTXWJJT287C2&qid=1698777909&sprefix=maca%2Caps%2C152&ref=sr_ex_n_1 (accessed on 14 November 2023).
- Xu, Q.; Monagas, M.J.; Kassymbek, Z.K.; Belsky, J.L. Controlling the quality of maca (Lepidium meyenii) dietary supplements: Development of compendial procedures for the determination of intact glucosinolates in maca root powder products. J. Pharm. Biomed. Anal. 2021, 199, 114063. [Google Scholar] [CrossRef] [PubMed]
- The Maca Team. Available online: https://www.themacateam.com/ (accessed on 24 November 2023).
- Anthony’s Goods. Available online: https://anthonysgoods.com/ (accessed on 24 November 2023).
- Clément, C.; Diaz Grados, D.A.; Avula, B.; Khan, I.A.; Mayer, A.C.; Ponce Aguirre, D.D.; Manrique, I.; Kreuzer, M. Influence of colour type and previous cultivation on secondary metabolites in hypocotyls and leaves of maca (Lepidium meyenii Walp.). J. Sci. Food Agric. 2010, 90, 861–869. [Google Scholar] [CrossRef]
- Chain, F.E.; Grau, A.; Martins, J.C.; Catalan, C.A. Macamides from wild ‘Maca’, Lepidium meyenii Walp. (Brassicaceae). Phytochem. Lett. 2014, 8, 145–148. [Google Scholar] [CrossRef]
- Meissner, H.O.; Mscisz, A.; Piatkowska, E.; Baraniak, M.; Mielcarek, S.; Kedzia, B.; Holderna-Kedzia, E.; Pisulewski, P. Peruvian Maca (Lepidium peruvianum): (II) Phytochemical Profiles of Four Prime Maca Phenotypes Grown in Two Geographically-Distant Locations. Int. J. Biomed. Sci. 2016, 12, 9–24. [Google Scholar] [CrossRef]
- Geng, P.; Sun, J.; Chen, P.; Brand, E.; Frame, J.; Meissner, H.; Stewart, J.; Gafner, S.; Clark, S.; Miller, J.; et al. Characterization of Maca (Lepidium meyenii/Lepidium peruvianum) Using a Mass Spectral Fingerprinting, Metabolomic Analysis, and Genetic Sequencing Approach. Planta Med. 2020, 86, 674–685. [Google Scholar] [CrossRef]
- Maca Root. USP-NF/PF Abstract 2023. Available online: https://doi.usp.org/USPNF/USPNF_M4469_10101_01.html (accessed on 6 November 2023).
- Li, J.; Chen, L.; Li, J.; Duan, Z.; Zhu, S.; Fan, L. The Composition Analysis of Maca (Lepidium meyenii Walp.) from Xinjiang and Its Antifatigue Activity. J. Food Qual. 2017, 2017, 2904951. [Google Scholar] [CrossRef]
- Long-Bo, Z.; Qing-Xiu, H.; Li, Z.; Jian, Y.; Min, C.; Li-Ping, K.; Lu-Qi, H. [Quality evaluation method of Lepidium meyenii using UPLC-UV-Q-TOF-MS]. Zhongguo Zhong Yao Za Zhi 2020, 45, 4957–4963. [Google Scholar]
- Meissner, H.O.; Mscisz, A.; Baraniak, M.; Piatkowska, E.; Pisulewski, P.; Mrozikiewicz, M.; Bobkiewicz-Kozlowska, T. Peruvian Maca (Lepidium peruvianum)—III: The Effects of Cultivation Altitude on Phytochemical and Genetic Differences in the Four Prime Maca Phenotypes. Int. J. Biomed. Sci. 2017, 13, 58–73. [Google Scholar] [CrossRef]
- Zhao, J.; Avula, B.; Chan, M.; Clément, C.; Kreuzer, M.; Khan, I.A. Metabolomic differentiation of maca (Lepidium meyenii) accessions cultivated under different conditions using N.M.R and chemometric analysis. Planta Med. 2012, 78, 90–101. [Google Scholar] [CrossRef]
- Chen, S.-X.; Li, K.-K.; Pubu, D.; Jiang, S.-P.; Chen, B.; Chen, L.-R.; Yang, Z.; Ma, C.; Gong, X.-J. Optimization of Ultrasound-Assisted Extraction, HPLC and UHPLC-ESI-Q-TOF-MS/MS Analysis of Main Macamides and Macaenes from Maca (Cultivars of Lepidium meyenii Walp.). Molecules 2017, 22, 2196. [Google Scholar] [CrossRef]
- Li, A.; Duan, S.; Dang, Y.; Zhang, X.; Xia, K.; Liu, S.; Han, X.; Wen, J.; Li, Z.; Wang, X.; et al. Origin identification of Chinese Maca using electronic nose coupled with GC-MS. Sci. Rep. 2019, 9, 12216. [Google Scholar] [CrossRef]
- Hermann, M.; Heller, J. (Eds.) Andean Roots and Tubers: Ahipa, Arracacha, Maca and Yacon. Promoting the Conservation and Use of Underutilized and Neglected Crops. 21; Institute of Plant Genetics and Crop Plant Research, Gatersleben/International Plant Genetic Resources Institute: Rome, Italy, 1997. [Google Scholar]
- Chen, W.; Modi, D.; Picot, A. Soil and Phytomicrobiome for Plant Disease Suppression and Management under Climate Change: A Review. Plants 2023, 12, 2736. [Google Scholar] [CrossRef]
- Ortiz, O. Evolution of agricultural extension and information dissemination in Peru: An historical perspective focusing on potato-related pest control. Agric. Hum. Values 2006, 23, 477–489. [Google Scholar] [CrossRef]
- Orellana Mendoza, E.; Cuadrado, W.; Yallico, L.; Zárate, R.; Quispe-Melgar, H.R.; Limaymanta, C.H.; Sarapura, V.; Bao-Cóndor, D. Heavy metals in soils and edible tissues of Lepidium meyenii (maca) and health risk assessment in areas influenced by mining activity in the Central region of Peru. Toxicol. Rep. 2021, 8, 1461–1470. [Google Scholar] [CrossRef] [PubMed]
- Bodenlos, A.J.; Straczek, J.A. Base-Metal Deposits of the Cordillera Negra Departamento de Ancash, Peru; US Government Printing Office: Washington, DC, USA, 1957; pp. 1–175.
- Zhang, J.; Wang, H.M.; Zhao, Y.L.; Zuo, Z.T.; Wang, Y.Z.; Jin, H. Comparison of Mineral Element Content in a Functional Food Maca (Lepidium meyenii Walp.) from Asia and South America. J. Anal. Methods Chem. 2015, 2015, 530541. [Google Scholar] [CrossRef] [PubMed]
- Shang, R.G.; Yang, P.; Wang, B.Y.; Zhao, Z.L. Transcriptome analysis of maca (Lepidium meyenii) root at different developmental stages. Appl. Plant Sci. 2018, 6, e012062018. [Google Scholar] [CrossRef]
- Wang, S.; Zhu, F. Chemical composition and health effects of maca (Lepidium meyenii). Food Chem. 2019, 288, 422–443. [Google Scholar] [CrossRef] [PubMed]
- Petruzzello, M. Maca. Available online: https://www.britannica.com/plant/maca (accessed on 14 November 2023).
- Zhao, X.; Zhang, Y.; Long, T.; Wang, S.; Yang, J. Regulation Mechanism of Plant Pigments Biosynthesis: Anthocyanins, Carotenoids, and Betalains. Metabolites 2022, 12, 871. [Google Scholar] [CrossRef] [PubMed]
- van Het Hof, K.H.; West, C.E.; Weststrate, J.A.; Hautvast, J.G. Dietary factors that affect the bioavailability of carotenoids. J. Nutr. 2000, 130, 503–506. [Google Scholar] [CrossRef] [PubMed]
- Meissner, H.O.; Kedzia, B.; Mrozikiewicz, P.M.; Mscisz, A. Short and long-term physiological responses of male and female rats to two dietary levels of pre-gelatinized maca (lepidium peruvianum chacon). Int. J. Biomed. Sci. 2006, 2, 13–28. [Google Scholar] [PubMed]
- Korompokis, K.; Verbeke, K.; Delcour, J.A. Structural factors governing starch digestion and glycemic responses and how they can be modified by enzymatic approaches: A review and a guide. Compr. Rev. Food Sci. Food Saf. 2021, 20, 5965–5991. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Yi, J.; Liu, X. A Low-FODMAP Diet Provides Benefits for Functional Gastrointestinal Symptoms but Not for Improving Stool Consistency and Mucosal Inflammation in IBD: A Systematic Review and Meta-Analysis. Nutrients 2022, 14, 2072. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, J.K.; Salwan, P.; Salwan, S. Various Possible Toxicants Involved in Thyroid Dysfunction: A Review. J. Clin. Diagn. Res. 2016, 10, Fe01–Fe03. [Google Scholar] [CrossRef] [PubMed]
- Petroski, W.; Minich, D.M. Is There Such a Thing as “Anti-Nutrients”? A Narrative Review of Perceived Problematic Plant Compounds. Nutrients 2020, 12, 2929. [Google Scholar] [CrossRef]
- Chang, Q.; Zheng, B.; Zhang, Y.; Zeng, H. A comprehensive review of the factors influencing the formation of retrograded starch. Int. J. Biol. Macromol. 2021, 186, 163–173. [Google Scholar] [CrossRef]
- Okunade, O.A.; Ghawi, S.K.; Methven, L.; Niranjan, K. Thermal and pressure stability of myrosinase enzymes from black mustard (Brassica nigra L. W.D.J. Koch. var. nigra), brown mustard (Brassica juncea L. Czern. var. juncea) and yellow mustard (Sinapsis alba L. subsp. maire) seeds. Food Chem. 2015, 187, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Esparza, E.; Yi, W.; Limonchi, F.; Cosio, E.G. Glucosinolate catabolism during postharvest drying determines the ratio of bioactive macamides to deaminated benzenoids in Lepidium meyenii (maca) root flour. Phytochemistry 2020, 179, 112502. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.; Wei, J.; Chen, R. Evaluation of the Biological Activity of Glucosinolates and Their Enzymolysis Products Obtained from Lepidium meyenii Walp. (Maca). Int. J. Mol. Sci. 2022, 23, 14756. [Google Scholar] [CrossRef] [PubMed]
- Esparza, E.; Hadzich, A.; Kofer, W.; Mithöfer, A.; Cosio, E.G. Bioactive maca (Lepidium meyenii) alkamides are a result of traditional Andean postharvest drying practices. Phytochemistry 2015, 116, 138–148. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, G.F.; Gasco, M.; Lozada-Requena, I. Role of maca (Lepidium meyenii) consumption on serum interleukin-6 levels and health status in populations living in the Peruvian Central Andes over 4000 m of altitude. Plant Foods Hum. Nutr. 2013, 68, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Cao, F.; Zhang, H.; Yan, Y.; Chang, Y.; Ma, J. Extraction of polysaccharides from Maca enhances the treatment effect of 5-FU by regulating CD4(+)T cells. Heliyon 2023, 9, e16495. [Google Scholar] [CrossRef] [PubMed]
- Fei, W.T.; Yue, N.; Li, A.M.; Yu, S.H.; Zhao, D.P.; Zhu, Y.L.; Wang, C.; Zhang, J.J.; Wang, L.Y. Immunomodulatory Effects of Lepidium meyenii Walp. Polysaccharides on an Immunosuppression Model Induced by Cyclophosphamide. J. Immunol. Res. 2022, 2022, 1210890. [Google Scholar] [CrossRef]
- da Silva Leitão Peres, N.; Cabrera Parra Bortoluzzi, L.; Medeiros Marques, L.L.; Formigoni, M.; Fuchs, R.H.B.; Droval, A.A.; Reitz Cardoso, F.A. Medicinal effects of Peruvian maca (Lepidium meyenii): A review. Food Funct. 2020, 11, 83–92. [Google Scholar] [CrossRef]
- Huang, Y.J.; Peng, X.R.; Qiu, M.H. Progress on the Chemical Constituents Derived from Glucosinolates in Maca (Lepidium meyenii). Nat. Prod. Bioprospect 2018, 8, 405–412. [Google Scholar] [CrossRef]
- Chen, Q.; Li, M.; Wang, C.; Li, Z.; Xu, J.; Zheng, Q.; Liu, P.; Zhou, H. Combining Targeted Metabolites Analysis and Transcriptomics to Reveal Chemical Composition Difference and Underlying Transcriptional Regulation in Maca (Lepidium Meyenii Walp.) Ecotypes. Genes 2018, 9, 335. [Google Scholar] [CrossRef]
- Meissner, H.O.; Mscisz, A.; Mrozikiewicz, M.; Baraniak, M.; Mielcarek, S.; Kedzia, B.; Piatkowska, E.; Jólkowska, J.; Pisulewski, P. Peruvian Maca (Lepidium peruvianum): (I) Phytochemical and Genetic Differences in Three Maca Phenotypes. Int. J. Biomed. Sci. 2015, 11, 131–145. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Li, P.; Brantner, A.; Wang, H.; Shu, X.; Yang, J.; Si, N.; Han, L.; Zhao, H.; Bian, B. Chemical profiling analysis of Maca using UHPLC-ESI-Orbitrap MS coupled with UHPLC-ESI-QqQ MS and the neuroprotective study on its active ingredients. Sci. Rep. 2017, 7, 44660. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information. PubChem Compound Summary for CID 656498, [(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl] 2-phenyl-N-sulfooxyethanimidothioate. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Glucotropaeolin. (accessed on 24 November 2023).
- National Center for Biotechnology Information. PubChem Compound Summary for CID 222284, Beta-Sitosterol. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Beta-Sitosterol. (accessed on 24 November 2023).
- National Center for Biotechnology Information. PubChem Compound Summary for CID 404702, Lepidiline A. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Lepidiline-A (accessed on 24 November 2023).
- National Center for Biotechnology Information. PubChem Compound Summary for CID 1274030, 2-Thiohydantoin. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/2-Thiohydantoin (accessed on 24 November 2023).
- National Center for Biotechnology Information. PubChem Compound Summary for CID 11198769, Macamide 1. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Macamide-1. (accessed on 24 November 2023).
- National Center for Biotechnology Information. PubChem Compound Summary for CID 90478988, Macaene. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Macaene (accessed on 24 November 2023).
- Bhat, R. Chapter 12—Glucosinolates. In Nutraceuticals and Health Care; Kour, J., Nayik, G.A., Eds.; Academic Press: Cambridge, MA, USA, 2022; pp. 233–243. [Google Scholar]
- Perez, C.J.; Conceição, R.S.; Ifa, D.R. Chemical profiling and separation of bioactive secondary metabolites in Maca (Lepidium peruvianum) by normal and reverse phase thin layer chromatography coupled to desorption electrospray ionization-mass spectrometry. J. Mass. Spectrom. 2021, 56, e4690. [Google Scholar] [CrossRef]
- Fahey, J.W.; Zalcmann, A.T.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5–51. [Google Scholar] [CrossRef] [PubMed]
- Minich, D.M.; Bland, J.S. A review of the clinical efficacy and safety of cruciferous vegetable phytochemicals. Nutr. Rev. 2007, 65 Pt 1, 259–267. [Google Scholar] [CrossRef]
- Higdon, J.V.; Delage, B.; Williams, D.E.; Dashwood, R.H. Cruciferous vegetables and human cancer risk: Epidemiologic evidence and mechanistic basis. Pharmacol. Res. 2007, 55, 224–236. [Google Scholar] [CrossRef]
- Na, G.; He, C.; Zhang, S.; Tian, S.; Bao, Y.; Shan, Y. Dietary Isothiocyanates: Novel Insights into the Potential for Cancer Prevention and Therapy. Int. J. Mol. Sci. 2023, 24, 1962. [Google Scholar] [CrossRef]
- Connolly, E.L.; Sim, M.; Travica, N.; Marx, W.; Beasy, G.; Lynch, G.S.; Bondonno, C.P.; Lewis, J.R.; Hodgson, J.M.; Blekkenhorst, L.C. Glucosinolates From Cruciferous Vegetables and Their Potential Role in Chronic Disease: Investigating the Preclinical and Clinical Evidence. Front. Pharmacol. 2021, 12, 767975. [Google Scholar] [CrossRef]
- Li, G.A.U.; Quiros, C.F. Glucosinolate contents in maca (Lepidium peruvianum Chacon) seeds, sprouts, mature plants and several derived commercial products. Econ. Bot. 2001, 55, 255–262. [Google Scholar] [CrossRef]
- Williams, D.E. Indoles Derived From Glucobrassicin: Cancer Chemoprevention by Indole-3-Carbinol and 3,3′-Diindolylmethane. Front. Nutr. 2021, 8, 734334. [Google Scholar] [CrossRef]
- Piacente, S.; Carbone, V.; Plaza, A.; Zampelli, A.; Pizza, C. Investigation of the tuber constituents of maca (Lepidium meyenii Walp.). J. Agric. Food Chem. 2002, 50, 5621–5625. [Google Scholar] [CrossRef] [PubMed]
- Cui, B.; Zheng, B.L.; He, K.; Zheng, Q.Y. Imidazole alkaloids from Lepidium meyenii. J. Nat. Prod. 2003, 66, 1101–1103. [Google Scholar] [CrossRef] [PubMed]
- Mlostoń, G.; Kowalczyk, M.; Celeda, M.; Gach-Janczak, K.; Janecka, A.; Jasiński, M. Synthesis and Cytotoxic Activity of Lepidilines A-D: Comparison with Some 4,5-Diphenyl Analogues and Related Imidazole-2-thiones. J. Nat. Prod. 2021, 84, 3071–3079. [Google Scholar] [CrossRef] [PubMed]
- Purnomo, K.A.; Korinek, M.; Tsai, Y.H.; Hu, H.C.; Wang, Y.H.; Backlund, A.; Hwang, T.L.; Chen, B.H.; Wang, S.W.; Wu, C.C.; et al. Decoding Multiple Biofunctions of Maca on Its Anti-allergic, Anti-inflammatory, Anti-thrombotic, and Pro-angiogenic Activities. J. Agric. Food Chem. 2021, 69, 11856–11866. [Google Scholar] [CrossRef] [PubMed]
- Mlostoń, G.; Kowalczyk, M.; Celeda, M.; Jasiński, M.; Denel-Bobrowska, M.; Olejniczak, A.B. Fluorinated Analogues of Lepidilines A and C: Synthesis and Screening of Their Anticancer and Antiviral Activity. Molecules 2022, 27, 3524. [Google Scholar] [CrossRef]
- Xia, C.; Deng, J.; Chen, J.; Zhu, Y.; Song, Y.; Zhang, Y.; Li, H.; Lin, C. Simultaneous determination of macaenes and macamides in maca using an HPLC method and analysis using a chemometric method (HCA) to distinguish maca origin. Rev. Bras. Farmacogn. 2019, 29, 702–709. [Google Scholar] [CrossRef]
- Zheng, B.L.; He, K.; Kim, C.H.; Rogers, L.; Shao, Y.; Huang, Z.Y.; Lu, Y.; Yan, S.J.; Qien, L.C.; Zheng, Q.Y. Effect of a lipidic extract from Lepidium meyenii on sexual behavior in mice and rats. Urology 2000, 55, 598–602. [Google Scholar] [CrossRef]
- Zhu, H.; Wang, R.; Hua, H.; Cheng, Y.; Guo, Y.; Qian, H.; Du, P. The macamide relieves fatigue by acting as inhibitor of inflammatory response in exercising mice: From central to peripheral. Eur. J. Pharmacol. 2022, 917, 174758. [Google Scholar] [CrossRef]
- Hahn, D.; Lee, T.; Lee, S.; Bae, J.-S.; Na, M.; Kim, M. Discovery and Quantitative Analysis of Nuclear Factor Erythroid 2-Related Factor (Nrf2) Activators in Maca (Lepidium meyenii) Using the Synthetic Macamides. Curr. Dev. Nutr. 2020, 4, nzaa045_034. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhu, L.; Li, H.; Xie, W.; Liu, J.; Zhang, Y.; Li, Y.; Wang, C. In vivo and in vitro neuroprotective effects of maca polysaccharide. Front. Biosci. 2022, 27, 8. [Google Scholar] [CrossRef]
- Zhang, K.Y.; Li, C.N.; Zhang, N.X.; Gao, X.C.; Shen, J.M.; Cheng, D.D.; Wang, Y.L.; Zhang, H.; Lv, J.W.; Sun, J.M. UPLC-QE-Orbitrap-Based Cell Metabolomics and Network Pharmacology to Reveal the Mechanism of N-Benzylhexadecanamide Isolated from Maca (Lepidium meyenii Walp.) against Testicular Dysfunction. Molecules 2023, 28, 4064. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, H.; Guo, F.; Si, N.; Brantner, A.; Yang, J.; Han, L.; Wei, X.; Zhao, H.; Bian, B. Integrated Proteomics and Lipidomics Investigation of the Mechanism Underlying the Neuroprotective Effect of N-benzylhexadecanamide. Molecules 2018, 23, 2929. [Google Scholar] [CrossRef] [PubMed]
- Gonzales-Arimborgo, C.; Yupanqui, I.; Montero, E.; Alarcón-Yaquetto, D.E.; Zevallos-Concha, A.; Caballero, L.; Gasco, M.; Zhao, J.; Khan, I.A.; Gonzales, G.F. Acceptability, Safety, and Efficacy of Oral Administration of Extracts of Black or Red Maca (Lepidium meyenii) in Adult Human Subjects: A Randomized, Double-Blind, Placebo-Controlled Study. Pharmaceuticals 2016, 9, 49. [Google Scholar] [CrossRef] [PubMed]
- Hajdu, Z.; Nicolussi, S.; Rau, M.; Lorántfy, L.; Forgo, P.; Hohmann, J.; Csupor, D.; Gertsch, J. Identification of endocannabinoid system-modulating N-alkylamides from Heliopsis helianthoides var. scabra and Lepidium meyenii. J. Nat. Prod. 2014, 77, 1663–1669. [Google Scholar] [CrossRef] [PubMed]
- Trautwein, E.A.; Vermeer, M.A.; Hiemstra, H.; Ras, R.T. LDL-Cholesterol Lowering of Plant Sterols and Stanols-Which Factors Influence Their Efficacy? Nutrients 2018, 10, 1262. [Google Scholar] [CrossRef] [PubMed]
- Chacon, G. “Maca” Millenarian Peruvian Food Plant with Highly Nutritional and Medicinal Properties, 1st ed.; Universidad Nacional Mayor de San Marcos: Lima, Peru, 2001. [Google Scholar]
- Hendriks, H.F.; Brink, E.J.; Meijer, G.W.; Princen, H.M.; Ntanios, F.Y. Safety of long-term consumption of plant sterol esters-enriched spread. Eur. J. Clin. Nutr. 2003, 57, 681–692. [Google Scholar] [CrossRef] [PubMed]
- Ayesh, R.; Weststrate, J.A.; Drewitt, P.N.; Hepburn, P.A. Safety evaluation of phytosterol esters. Part 5. Faecal short-chain fatty acid and microflora content, faecal bacterial enzyme activity and serum female sex hormones in healthy normolipidaemic volunteers consuming a controlled diet either with or without a phytosterol ester-enriched margarine. Food Chem. Toxicol. 1999, 37, 1127–1138. [Google Scholar] [PubMed]
- Del Valle Mendoza, J.; Pumarola, T.; Gonzales, L.A.; Del Valle, L.J. Antiviral activity of maca (Lepidium meyenii) against human influenza virus. Asian Pac. J. Trop. Med. 2014, 7s1, S415–S420. [Google Scholar] [CrossRef] [PubMed]
- Gonzales-Castañeda, C.; Gonzales, G.F. Hypocotyls of Lepidium meyenii (maca), a plant of the Peruvian highlands, prevent ultraviolet A-, B-, and C-induced skin damage in rats. Photodermatol. Photoimmunol. Photomed. 2008, 24, 24–31. [Google Scholar] [CrossRef]
- Gonzales, G.F.; Valerio, L.G., Jr. Medicinal plants from Peru: A review of plants as potential agents against cancer. Anticancer. Agents Med. Chem. 2006, 6, 429–444. [Google Scholar] [CrossRef]
- Rubio, J.; Riqueros, M.I.; Gasco, M.; Yucra, S.; Miranda, S.; Gonzales, G.F. Lepidium meyenii (Maca) reversed the lead acetate induced—Damage on reproductive function in male rats. Food Chem. Toxicol. 2006, 44, 1114–1122. [Google Scholar] [CrossRef]
- Yu, Z.; Li, D.; Zhai, S.; Xu, H.; Liu, H.; Ao, M.; Zhao, C.; Jin, W.; Yu, L. Neuroprotective effects of macamide from maca (Lepidium meyenii Walp.) on corticosterone-induced hippocampal impairments through its anti-inflammatory, neurotrophic, and synaptic protection properties. Food Funct. 2021, 12, 9211–9228. [Google Scholar] [CrossRef]
- Shin, B.C.; Lee, M.S.; Yang, E.J.; Lim, H.S.; Ernst, E. Maca (L. meyenii) for improving sexual function: A systematic review. BMC Complement. Altern. Med. 2010, 10, 44. [Google Scholar] [CrossRef]
- Guo, S.S.; Gao, X.F.; Gu, Y.R.; Wan, Z.X.; Lu, A.M.; Qin, Z.H.; Luo, L. Preservation of Cognitive Function by Lepidium meyenii (Maca) Is Associated with Improvement of Mitochondrial Activity and Upregulation of Autophagy-Related Proteins in Middle-Aged Mouse Cortex. Evid. Based Complement. Altern. Med. 2016, 2016, 4394261. [Google Scholar] [CrossRef]
- Lee, M.S.; Shin, B.C.; Yang, E.J.; Lim, H.J.; Ernst, E. Maca (Lepidium meyenii) for treatment of menopausal symptoms: A systematic review. Maturitas 2011, 70, 227–233. [Google Scholar] [CrossRef]
- Ross, K. Nutritional management of surgically induced menopause: A case report. Womens Health 2021, 17, 17455065211031492. [Google Scholar] [CrossRef] [PubMed]
- Nuñez, D.; Olavegoya, P.; Gonzales, G.F.; Gonzales-Castañeda, C. Red Maca (Lepidium meyenii), a Plant from the Peruvian Highlands, Promotes Skin Wound Healing at Sea Level and at High Altitude in Adult Male Mice. High. Alt. Med. Biol. 2017, 18, 372–383. [Google Scholar] [CrossRef]
- Fu, L.; Wei, J.; Gao, Y.; Chen, R. Antioxidant and antitumoral activities of isolated macamide and macaene fractions from Lepidium meyenii (Maca). Talanta 2021, 221, 121635. [Google Scholar] [CrossRef]
- Jin, W.; Chen, X.; Huo, Q.; Cui, Y.; Yu, Z.; Yu, L. Aerial parts of maca (Lepidium meyenii Walp.) as functional vegetables with gastrointestinal prokinetic efficacy in vivo. Food Funct. 2018, 9, 3456–3465. [Google Scholar] [CrossRef] [PubMed]
- Panossian, A.G.; Efferth, T.; Shikov, A.N.; Pozharitskaya, O.N.; Kuchta, K.; Mukherjee, P.K.; Banerjee, S.; Heinrich, M.; Wu, W.; Guo, D.A.; et al. Evolution of the adaptogenic concept from traditional use to medical systems: Pharmacology of stress- and aging-related diseases. Med. Res. Rev. 2021, 41, 630–703. [Google Scholar] [CrossRef] [PubMed]
- Patwardhan, B.; Warude, D.; Pushpangadan, P.; Bhatt, N. Ayurveda and traditional Chinese medicine: A comparative overview. Evid. Based Complement. Altern. Med. 2005, 2, 465–473. [Google Scholar] [CrossRef]
- Chacon, R.G. Phytochemical Study on Lepidium meyenii. Ph.D. Thesis, Universidad Nacional Mayo de San Marcos, Lima, Peru, 1961. [Google Scholar]
- Stojanovska, L.; Law, C.; Lai, B.; Chung, T.; Nelson, K.; Day, S.; Apostolopoulos, V.; Haines, C. Maca reduces blood pressure and depression, in a pilot study in postmenopausal women. Climacteric 2015, 18, 69–78. [Google Scholar] [CrossRef]
- Brooks, N.A.; Wilcox, G.; Walker, K.Z.; Ashton, J.F.; Cox, M.B.; Stojanovska, L. Beneficial effects of Lepidium meyenii (Maca) on psychological symptoms and measures of sexual dysfunction in postmenopausal women are not related to estrogen or androgen content. Menopause 2008, 15, 1157–1162. [Google Scholar] [CrossRef]
- Takewaka, T.; Hara, K. Clinical Effect of Oral Administration of Maca (Lepidium meyenii) Extract on Japanese Peri-Menopausal Women Subjects: A Randomized, Double-Blind, Placebo-Controlled Study. Int. J. Biomed. Sci. 2019, 15, 11–18. [Google Scholar] [CrossRef]
- Grant, M.D.; Marbella, A.; Wang, A.T.; Pines, E.; Hoag, J.; Bonnell, C.; Ziegler, K.M.; Aronson, N. AHRQ Comparative Effectiveness Reviews. In Menopausal Symptoms: Comparative Effectiveness of Therapies; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2015. [Google Scholar]
- David, P.S.; Sobel, T.; Sahni, S.; Mehta, J.; Kling, J.M. Menopausal Hormone Therapy in Older Women: Examining the Current Balance of Evidence. Drugs Aging 2023, 40, 675–683. [Google Scholar] [CrossRef]
- Gilbert, Z.A.; Muller, A.; Leibowitz, J.A.; Kesselman, M.M. Osteoporosis Prevention and Treatment: The Risk of Comorbid Cardiovascular Events in Postmenopausal Women. Cureus 2022, 14, e24117. [Google Scholar] [CrossRef]
- The 2022 Hormone Therapy Position Statement of The North American Menopause Society” Advisory Panel. The 2022 hormone therapy position statement of The North American Menopause Society. Menopause 2022, 29, 767–794. [Google Scholar] [CrossRef]
- Stuenkel, C.A.; Davis, S.R.; Gompel, A.; Lumsden, M.A.; Murad, M.H.; Pinkerton, J.V.; Santen, R.J. Treatment of Symptoms of the Menopause: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2015, 100, 3975–4011. [Google Scholar] [CrossRef] [PubMed]
- National Health Service When to Take Hormone Replacement Therapy. Available online: https://www.nhs.uk/medicines/hormone-replacement-therapy-hrt/when-to-take-hormone-replacement-therapy-hrt/ (accessed on 4 January 2023).
- Lu, J.; Li, K.; Zheng, X.; Liu, R.; Chen, M.; Xian, J.; Tu, S.; Xie, L. Prevalence of menopausal symptoms and attitudes towards menopausal hormone therapy in women aged 40–60 years: A cross-sectional study. BMC Womens Health 2023, 23, 472. [Google Scholar] [CrossRef] [PubMed]
- Srikugan, L.; Sankaralingam, A.; McGowan, B. First case report of testosterone assay-interference in a female taking maca (Lepidium meyenii). BMJ Case Rep. 2011, 2011, bcr0120113781. [Google Scholar] [CrossRef] [PubMed]
- Rubio, J.; Caldas, M.; Dávila, S.; Gasco, M.; Gonzales, G.F. Effect of three different cultivars of Lepidium meyenii (Maca) on learning and depression in ovariectomized mice. BMC Complement. Altern. Med. 2006, 6, 23. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, G.F.; Córdova, A.; Vega, K.; Chung, A.; Villena, A.; Góñez, C. Effect of Lepidium meyenii (Maca), a root with aphrodisiac and fertility-enhancing properties, on serum reproductive hormone levels in adult healthy men. J. Endocrinol. 2003, 176, 163–168. [Google Scholar] [CrossRef]
- Alcalde, A.M.; Rabasa, J. Does Lepidium meyenii (Maca) improve seminal quality? Andrologia 2020, 52, e13755. [Google Scholar] [CrossRef] [PubMed]
- Levano, G.; Quispe, J.; Vargas, D.; García, M.; López, A.; Aguila, L.; Valdivia, M. Effect of Atomized Black Maca (Lepidium meyenii) Supplementation in the Cryopreservation of Alpaca (Vicugna pacos) Epididymal Spermatozoa. Animals 2023, 13, 2054. [Google Scholar] [CrossRef]
- Yucra, S.; Gasco, M.; Rubio, J.; Nieto, J.; Gonzales, G.F. Effect of different fractions from hydroalcoholic extract of Black Maca (Lepidium meyenii) on testicular function in adult male rats. Fertil. Steril. 2008, 89 (Suppl. S5), 1461–1467. [Google Scholar] [CrossRef] [PubMed]
- Zenico, T.; Cicero, A.F.; Valmorri, L.; Mercuriali, M.; Bercovich, E. Subjective effects of Lepidium meyenii (Maca) extract on well-being and sexual performances in patients with mild erectile dysfunction: A randomised, double-blind clinical trial. Andrologia 2009, 41, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Obregon, L. “Maca” Planta Medicinal y Nutritiva Del Peru., 1st ed.; Instituto de Fitoterapia: Lima, Peru, 2001. [Google Scholar]
- Meissner, H.O. The Unique Powers of the Maca Tuber: Scientific Facts behind Traditional Wisdom, 1st ed.; Michaels Verlag & Vertrieb GmbH: Munich, Germany, 2014; pp. 1–432. (In German) [Google Scholar]
- Zevallos-Concha, A.; Nuñez, D.; Gasco, M.; Vasquez, C.; Quispe, M.; Gonzales, G.F. Effect of gamma irradiation on phenol content, antioxidant activity and biological activity of black maca and red maca extracts (Lepidium meyenii walp.). Toxicol. Mech. Methods 2016, 26, 67–73. [Google Scholar] [CrossRef]
- Lee, E.; Park, M.; Kim, B.; Kang, S. Effect of Black Maca Supplementation on Inflammatory Markers and Physical Fitness in Male Elite Athletes. Nutrients 2023, 15, 1618. [Google Scholar] [CrossRef]
- Gonzales, G.F.; Vasquez, V.B.; Gasco, M. The transillumination technique as a method for the assessment of spermatogenesis using medicinal plants: The effect of extracts of black maca (Lepidium meyenii) and camu camu (Myrciaria dubia) on stages of the spermatogenic cycle in male rats. Toxicol. Mech. Methods 2013, 23, 559–565. [Google Scholar] [CrossRef]
- Lembè, D.M.; Gasco, M.; Gonzales, G.F. Synergistic effect of the hydroalcoholic extract from Lepidium meyenii (Brassicaceae) and Fagara tessmannii (Rutaceae) on male sexual organs and hormone level in rats. Pharmacogn. Res. 2014, 6, 80–86. [Google Scholar] [CrossRef]
- Rubio, J.; Dang, H.; Gong, M.; Liu, X.; Chen, S.L.; Gonzales, G.F. Aqueous and hydroalcoholic extracts of Black Maca (Lepidium meyenii) improve scopolamine-induced memory impairment in mice. Food Chem. Toxicol. 2007, 45, 1882–1890. [Google Scholar] [CrossRef]
- Fu, P.; Luo, S.; Liu, Z.; Furuhara, K.; Tsuji, T.; Higashida, H.; Yokoyama, S.; Zhong, J.; Tsuji, C. Oral Supplementation with Maca Improves Social Recognition Deficits in the Valproic Acid Animal Model of Autism Spectrum Disorder. Brain Sci. 2023, 13, 316. [Google Scholar] [CrossRef]
- Wan, W.; Li, H.; Xiang, J.; Yi, F.; Xu, L.; Jiang, B.; Xiao, P. Aqueous Extract of Black Maca Prevents Metabolism Disorder via Regulating the Glycolysis/Gluconeogenesis-TCA Cycle and PPARα Signaling Activation in Golden Hamsters Fed a High-Fat, High-Fructose Diet. Front. Pharmacol. 2018, 9, 333. [Google Scholar] [CrossRef]
- Gonzales, C.; Cárdenas-Valencia, I.; Leiva-Revilla, J.; Anza-Ramirez, C.; Rubio, J.; Gonzales, G.F. Effects of different varieties of Maca (Lepidium meyenii) on bone structure in ovariectomized rats. Forsch. Komplementmed 2010, 17, 137–143. [Google Scholar] [CrossRef]
- Park, M.-H.; Lim, S.-T.; Lee, J.; Kim, B.-J.; Oh, S.-B.; Kang, S. Effects of Resistance Exercise, Black Maca and Combined Treatment on Blood Muscle Fatigue Factors and Muscle Function in Racket Athletes. Exerc. Sci. 2022, 31, 459–468. [Google Scholar] [CrossRef]
- Frame, J.; (Symphony Natural Health Holdings Inc., Craigmuir Chambers, Road Town, Tortola VG1110, British Virgin Islands). Personal communication, 2023.
- Symphony Natural Health Institute. Lepidium Peruvianum. 2023. Available online: http://www.lepidiumperuvianum.org (accessed on 14 November 2023).
- Choi, J.W. Effect of Intake Black Maca on Inflammatory Factors in Female Athletics. J. Sport. Dan. Sci. 2021, 1, 39–47. [Google Scholar] [CrossRef]
- Kim, S. Effects of maca (Lepidium meyenii Walp.) for sperm mobility and erectile function in adult men: A randomized placebo controlled clinical trial. Chung-Ang Univ. Hosp. Seoul 2011. [Google Scholar] [CrossRef]
- Lee, M.S.; Lee, H.W.; You, S.; Ha, K.T. The use of maca (Lepidium meyenii) to improve semen quality: A systematic review. Maturitas 2016, 92, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Fano, D.; Vásquez-Velásquez, C.; Gonzales-Castañeda, C.; Guajardo-Correa, E.; Orihuela, P.A.; Gonzales, G.F. N-Butanol and Aqueous Fractions of Red Maca Methanolic Extract Exerts Opposite Effects on Androgen and Oestrogens Receptors (Alpha and Beta) in Rats with Testosterone-Induced Benign Prostatic Hyperplasia. Evid. Based Complement. Altern. Med. 2017, 2017, 9124240. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, A.; Gupta, V.B. Various treatment options for benign prostatic hyperplasia: A current update. J. Midlife Health 2012, 3, 10–19. [Google Scholar]
- Leiva-Revilla, J.; Guerra-Castañon, F.; Olcese-Mori, P.; Lozada, I.; Rubio, J.; Gonzales, C.; Gonzales, G.F. [Effect of red maca (Lepidium meyenii) on INF-gamma levels in ovariectomized rats]. Rev. Peru. Med. Exp. Salud Publica 2014, 31, 683–688. [Google Scholar]
- Díaz, P.; Cardenas, H.; Orihuela, P.A. Red Maca (Lepidium meyenii) did not affect cell viability despite increased androgen receptor and prostate-specific antigen gene expression in the human prostate cancer cell line LNCaP. Andrologia 2016, 48, 922–926. [Google Scholar] [CrossRef]
- Ybañez-Julca, R.O.; Asunción-Alvarez, D.; Palacios, J.; Nwokocha, C.R. Maca extracts and estrogen replacement therapy in ovariectomized rats exposed at high altitude. Reprod. Med. Biol. 2021, 20, 88–95. [Google Scholar] [CrossRef]
- Gonzales-Castañeda, C.; Rivera, V.; Chirinos, A.L.; Evelson, P.; Gonzales, G.F. Photoprotection against the UVB-induced oxidative stress and epidermal damage in mice using leaves of three different varieties of Lepidium meyenii (maca). Int. J. Dermatol. 2011, 50, 928–938. [Google Scholar] [CrossRef]
- Ybañez-Julca, R.O.; Palacios, J.; Asunción-Alvarez, D.; Quispe-Díaz, I.; Nwokocha, C.R.; de Albuquerque, R. Lepidium meyenii Walp. (red maca) Supplementation Prevents Acrylamide-Induced Oxidative Stress and Liver Toxicity in Rats: Phytochemical Composition by UHPLC-ESI-MS/MS. Plant Foods Hum. Nutr. 2022, 77, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Melnikovova, I.; Russo, D.; Fait, T.; Kolarova, M.; Tauchen, J.; Kushniruk, N.; Falabella, R.; Milella, L.; Fernández Cusimamani, E. Evaluation of the effect of Lepidium meyenii Walpers in infertile patients: A randomized, double-blind, placebo-controlled trial. Phytother. Res. 2021, 35, 6359–6368. [Google Scholar] [CrossRef] [PubMed]
- Tafuri, S.; Cocchia, N.; Carotenuto, D.; Vassetti, A.; Staropoli, A.; Mastellone, V.; Peretti, V.; Ciotola, F.; Albarella, S.; Del Prete, C.; et al. Chemical Analysis of Lepidium meyenii (Maca) and Its Effects on Redox Status and on Reproductive Biology in Stallions. Molecules 2019, 24, 1981. [Google Scholar] [CrossRef] [PubMed]
- Yi, D.; Yoshikawa, M.; Sugimoto, T.; Tomoo, K.; Okada, Y.; Hashimoto, T. Effects of Maca on Muscle Hypertrophy in C2C12 Skeletal Muscle Cells. Int. J. Mol. Sci. 2022, 23, 6825. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Luna, A.C.; Salazar, S.; Aspajo, N.J.; Rubio, J.; Gasco, M.; Gonzales, G.F. Lepidium meyenii (Maca) increases litter size in normal adult female mice. Reprod. Biol. Endocrinol. 2005, 3, 16. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Salazar, L.; Gonzales, G.F. Aqueous extract of yellow maca (Lepidium meyenii) improves sperm count in experimental animals but response depends on hypocotyl size, pH and routes of administration. Andrologia 2018, 50, e12929. [Google Scholar] [CrossRef] [PubMed]
- Melnikovova, I.; Fait, T.; Kolarova, M.; Fernandez, E.C.; Milella, L. Effect of Lepidium meyenii Walp. on Semen Parameters and Serum Hormone Levels in Healthy Adult Men: A Double-Blind, Randomized, Placebo-Controlled Pilot Study. Evid. Based Complement. Altern. Med. 2015, 2015, 324369. [Google Scholar] [CrossRef] [PubMed]
- Bendokas, V.; Stanys, V.; Mažeikienė, I.; Trumbeckaite, S.; Baniene, R.; Liobikas, J. Anthocyanins: From the Field to the Antioxidants in the Body. Antioxidants 2020, 9, 819. [Google Scholar] [CrossRef] [PubMed]
- Zhong, H.; Xu, J.; Yang, M.; Hussain, M.; Liu, X.; Feng, F.; Guan, R. Protective Effect of Anthocyanins against Neurodegenerative Diseases through the Microbial-Intestinal-Brain Axis: A Critical Review. Nutrients 2023, 15, 496. [Google Scholar] [CrossRef] [PubMed]
- Strathearn, K.E.; Yousef, G.G.; Grace, M.H.; Roy, S.L.; Tambe, M.A.; Ferruzzi, M.G.; Wu, Q.L.; Simon, J.E.; Lila, M.A.; Rochet, J.C. Neuroprotective effects of anthocyanin- and proanthocyanidin-rich extracts in cellular models of Parkinson׳s disease. Brain Res. 2014, 1555, 60–77. [Google Scholar] [CrossRef] [PubMed]
- Haskell-Ramsay, C.F.; Stuart, R.C.; Okello, E.J.; Watson, A.W. Cognitive and mood improvements following acute supplementation with purple grape juice in healthy young adults. Eur. J. Nutr. 2017, 56, 2621–2631. [Google Scholar] [CrossRef] [PubMed]
- Rashid, K.; Wachira, F.N.; Nyabuga, J.N.; Wanyonyi, B.; Murilla, G.; Isaac, A.O. Kenyan purple tea anthocyanins ability to cross the blood brain barrier and reinforce brain antioxidant capacity in mice. Nutr. Neurosci. 2014, 17, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Shan, Q.; Lu, J.; Zheng, Y.; Li, J.; Zhou, Z.; Hu, B.; Zhang, Z.; Fan, S.; Mao, Z.; Wang, Y.J.; et al. Purple sweet potato color ameliorates cognition deficits and attenuates oxidative damage and inflammation in aging mouse brain induced by d-galactose. J. Biomed. Biotechnol. 2009, 2009, 564737. [Google Scholar] [CrossRef] [PubMed]
- Dani, C.; Pasquali, M.A.; Oliveira, M.R.; Umezu, F.M.; Salvador, M.; Henriques, J.A.; Moreira, J.C. Protective effects of purple grape juice on carbon tetrachloride-induced oxidative stress in brains of adult Wistar rats. J. Med. Food 2008, 11, 55–61. [Google Scholar] [CrossRef]
- Adnyana, I.M.O.; Sudewi, R.; Samatra, P.; Suprapta, S. Balinese Cultivar of Purple Sweet Potato Improved Neurological Score and BDNF and Reduced Caspase-Independent Apoptosis among Wistar Rats with Ischemic Stroke. Open Access Maced. J. Med. Sci. 2019, 7, 38–44. [Google Scholar] [CrossRef]
- Wood, E.; Hein, S.; Mesnage, R.; Fernandes, F.; Abhayaratne, N.; Xu, Y.; Zhang, Z.; Bell, L.; Williams, C.; Rodriguez-Mateos, A. Wild blueberry (poly)phenols can improve vascular function and cognitive performance in healthy older individuals: A double-blind randomized controlled trial. Am. J. Clin. Nutr. 2023, 117, 1306–1319. [Google Scholar] [CrossRef]
- Rodrigo-Gonzalo, M.J.; González-Manzano, S.; Pablos-Hernández, M.C.; Méndez-Sánchez, R.; Ayuda Duran, B.; González-Sánchez, J.; Barbero-Iglesias, F.; González-Paramás, A.M.; Recio-Rodríguez, J.I. Effects of a Raisin Supplement on Cognitive Performance, Quality of Life, and Functional Activities in Healthy Older Adults-Randomized Clinical Trial. Nutrients 2023, 15, 2811. [Google Scholar] [CrossRef]
- Lopresti, A.L.; Smith, S.J.; Pouchieu, C.; Pourtau, L.; Gaudout, D.; Pallet, V.; Drummond, P.D. Effects of a polyphenol-rich grape and blueberry extract (Memophenol™) on cognitive function in older adults with mild cognitive impairment: A randomized, double-blind, placebo-controlled study. Front. Psychol. 2023, 14, 1144231. [Google Scholar] [CrossRef]
- Minich, D.M. A Review of the Science of Colorful, Plant-Based Food and Practical Strategies for “Eating the Rainbow”. J. Nutr. Metab. 2019, 2019, 2125070. [Google Scholar] [CrossRef]
- Qiu, C.; Zhu, T.; Lan, L.; Zeng, Q.; Du, Z. Analysis of Maceaene and Macamide Contents of Petroleum Ether Extract of Black, Yellow, and Purple Lepidium Meyenii (Maca) and Their Antioxidant Effect on Diabetes Mellitus Rat Model. Braz. Arch. Biol. Technol. 2016, 59. [Google Scholar] [CrossRef]
- Ishida, M.; Hara, M.; Fukino, N.; Kakizaki, T.; Morimitsu, Y. Glucosinolate metabolism, functionality and breeding for the improvement of Brassicaceae vegetables. Breed. Sci. 2014, 64, 48–59. [Google Scholar] [CrossRef]
- Meissner, H.O.; Mrozikiewicz, P.; Bobkiewicz-Kozlowska, T.; Mscisz, A.; Kedzia, B.; Lowicka, A.; Reich-Bilinska, H.; Kapczynski, W.; Barchia, I. Hormone-Balancing Effect of Pre-Gelatinized Organic Maca (Lepidium peruvianum Chacon): (I) Biochemical and Pharmacodynamic Study on Maca using Clinical Laboratory Model on Ovariectomized Rats. Int. J. Biomed. Sci. 2006, 2, 260–272. [Google Scholar] [CrossRef]
- Ross, K.; Minich, D.M. Maca-GO®: The Science, Publications, and Clinical Applications for Hormone Health, 2nd ed.; Symphony Natural Health, Inc.: West Valley City, UT, USA, 2023; Available online: https://symphonynaturalhealthpro.com/pages/clinical-research (accessed on 15 May 2023).
- Ohta, Y.; Yoshida, K.; Kamiya, S.; Kawate, N.; Takahashi, M.; Inaba, T.; Hatoya, S.; Morii, H.; Takahashi, K.; Ito, M.; et al. Feeding hydroalcoholic extract powder of Lepidium meyenii (maca) increases serum testosterone concentration and enhances steroidogenic ability of Leydig cells in male rats. Andrologia 2016, 48, 347–354. [Google Scholar] [CrossRef]
- Bower-Cargill, C.; Yarandi, N.; Petróczi, A. A systematic review of the versatile effects of the Peruvian Maca Root (Lepidium meyenii) on sexual dysfunction, menopausal symptoms and related conditions. Phytomedicine Plus 2022, 2, 100326. [Google Scholar] [CrossRef]
- Quelca Tancara, B.; Solares Espinoza, M.; Cortez, J.; Velez, G.; Salcedo, Y.; Salinas, A.M.; Carvajal, R. Effect of the Lepidium meyenii (Maca) on the spermatogenesis and the spermatic quality of subjects with diagnosis of infertility: Study of cases. Biofarbo 2010, 18, 61–70. [Google Scholar]
- Poveda, C.; Rodriguez, R.; Chu, E.E.; Aparicio, L.E.; Gonzales, I.G.; Moreno, C.J. A placebo-controlled double-blind randomized trial of the effect of oral supplementation with spermotrend, maca extract (Lepidium meyenii) or l-carnitine in semen parameters of infertile men in IFFS-ASRM. In Proceedings of the International Federation of Fertility Societies 21st World Congress on Fertility and Sterility and the 69th Annual Meeting of the American Society for Reproductive Medicine, Boston, MA, USA, 12–17 October 2013; p. S440. [Google Scholar]
- Honma, A.; Fujiwara, Y.; Takei, S.; Kino, T. The improvements of daily fatigue in women following the intake of maca (Lepidium meyenii) extract containing benzyl glucosinolate. Funct. Foods Health Dis. 2022, 12, 175–187. [Google Scholar] [CrossRef]



| Scientific Functions | Proposed Health Benefits |
|---|---|
|
|
| Color | Number of Clinical Studies Reported |
|---|---|
| Black | 6 |
| Red | 1 * |
| Yellow | 2 |
| Purple | 0 |
| Gray | 0 |
| Combination of colors: Black, yellow, red/purple | 7 |
| Not stated or not known ** | 15 |
| Study Description | Details on Maca: Species, Product, Location Grown and Cultivation Methods, Form(s), Dose and Route | Overview of Study Results |
|---|---|---|
| 12-week double-blind, randomized, parallel-group, dose-finding pilot study [54] in 20 subjects with major depressive disorder on anti-depressant medication(s) for a minimum of 8 weeks | Species: Lepidium peruvianum Product: Maca was purchased from A Healthy Alternative (ahealthya.com) in Long Island, New York. Location/Cultivation: Not stated Form: Not stated Dose and Route: 1.5 g per day (low dose), 3.0 g per day (high dose); both as capsules for oral intake. | Both groups had significant improvement in sexual dysfunction assessments (ASEX: Arizona Sexual Experience Scale; MGH-SFQ: Massachusetts General Hospital Sexual Function Questionnaire). For the group taking the higher dose (3 g/day), an increased improvement in sexual function was observed compared to the low-dose group, as measured by ASEX (p = 0.028) and MGH-SFQ (p = 0.017), in addition to a significant improvement in libido (p < 0.05), with increased sexual attempts (p = 0.048) and enjoyment (p = 0.019). A small but significant decrease in depressive symptoms (p = 0.047) was also observed. |
| 4-month intervention trial [31] in 9 men, 24–44 years old | Species: Lepidium meyenii Product: Maca Gelatinizada La Molina tablets provided by the Laboratorios Hersil (Lima, Peru), which could also be purchased in the pharmacy. Location/Cultivation: Peru; cultivation methods unknown [abstract only] Form: Unknown [abstract only] Dose and Route: Six men received 1.5 g/day and three men received 3 g/day; each tablet provided 500 mg. | Maca treatment resulted in improvements in sperm markers (seminal volume (p < 0.05), total sperm count per ejaculum (p < 0.05), motile sperm count (p < 0.05), sperm motility (p < 0.05)); serum hormone levels (LH, FSH, prolactin, T, E2) were not affected. |
| Population-based, cross-sectional study [97], comparing individuals who consumed maca as a food source to those who did not. Subjects included 50 men and women, 35–69 years old, living in Junín, Peru (4100 m above sea level), with 27 of them as regular maca consumers. | Species: Lepidium meyenii Product: Not applicable, since it was examining maca as a dietary source. Location/Cultivation: Peru, cultivation methods not stated. Form: 96% consumed maca as a juice, prepared at home Dose and Route: Average time consuming maca was 25.8 ± 3.2 years. | Compared to non-consumer, the regular consumers of maca had statistically significant changes: higher serum E2 (p < 0.01) and health scores (p < 0.01), lower systolic blood pressure (p < 0.05), serum T levels (p < 0.05), T/E2 ratio (p < 0.05), serum IL-6 (p < 0.05), and CMS scores (p < 0.01), in addition to performing better on the lower limb strength test (p < 0.01). The lowest IL-6 levels were observed when maca was consumed in greater quantities (15.9 ± 3.08 g daily) and for the longer durations (17.8 ± 4.48 years for 41.9 ± 4.48 days per year). |
| 12-week, prospective, randomized, double-blind, placebo-controlled parallel trial [166] in 69 males (20–40 yrs old) diagnosed with mild astheno-zoospermia and/or mild oligozoospermia; 2 were lost to follow-up. | Species: Lepidium meyenii Product: Not stated Location/Cultivation: Not stated Form: Gelatinized maca, powder Dose and Route: 2 g per day (1 g capsule twice daily) | Significant increase (p < 0.011) in sperm concentration (31.69%); non-statistically significant increases in sperm motility (10.48%) and morphology of spermatozoa (6.33%). |
| 12-week randomized, single-center, double-blind, placebo-controlled, cross-over study [153] (6 weeks maca and 6 weeks placebo) with 34 post-menopausal Chinese women (from Hong Kong); 46–59 years old; N = 29 completed the trial | Species: Lepidium meyenii Product: Maca Power, Healthy-choices, Murwillumbah, NSW, Australia Location/Cultivation: Peru, cultivation methods not stated Form: Not stated Dose and Route: 3.3 g daily, 7 capsules total intake per day, with 4 capsules following breakfast, 3 capsules following dinner | After 6 weeks of treatment, the maca group showed statistically significant changes in systolic (p = 0.05) and diastolic (p = 0.01) blood pressure reductions, increased (10.8%) general well-being (p < 0.05), increased (13.5%) mental health (p < 0.05), increased (16.4%) social functioning (p < 0.05), increased (13%) mental component score (p < 0.05); significant reduction (30%) of menopausal symptoms (p = 0.04) including reduction in anxiety (p = 0.02), depression (p < 0.01), and somatic scores (p < 0.03); no differences in E2, FSH, LH, SHBG, glucose, lipid profile, serum cytokines, or vasomotor symptoms. |
| 12-week, double-blind, randomized clinical trial [169] in 50 Caucasian men (36 years old ± 5 years) with mild erectile dysfunction (ED) | Species: Lepidium meyenii Product: Provided by Ibersan Srl, Forli, Italy Location/Cultivation: Peru, cultivation methods not stated Form: Pulverized, dehydrated tablets Dose and Route: 2.4 g per day (1.2 g twice daily) as tablets | While both the placebo group and maca group reported significant improvements, the maca group experienced more significant effects after 12 weeks of treatment per the International Index of Erectile Function (IIEF-5) score (p < 0.05) and in psychological performance-related Satisfaction Profile (SAT-P) score (p < 0.05). Only maca-treated patients experienced a statistically significant improvement in physical and social performance-related SAT-P score compared with the baseline (p < 0.05). |
| 12-week, randomized, double-blind, placebo-controlled, crossover trial [154] (6 weeks of maca and 6 weeks of placebo) with 16 post-menopausal women, 50–60 years old; 2 did not complete the trial. | Species: Lepidium meyenii Product: Maca Power, Incan Food, Murwillumbah, NSW, Australia Location/Cultivation: Peru, cultivation methods not stated Form: Dried maca powder Dose and Route: 3.5 g one time per day, powder taken in breakfast cereal, soup, or a milk shake | Statistically significant reduction (30%, p < 0.05) in symptoms, including decreased anxiety and depression and improved sexual function (p < 0.05); no differences were seen in serum concentrations of E2, FSH, LH, and SHBG. Post hoc analyses revealed a significant increase in E2 would only be detected at a 30% increase. |
| 12-week double-blind, placebo-controlled, randomized, parallel trial [165] in 56 men, 21–56 years old | Species: Lepidium meyenii Product: Maca Gelatinizada from Laboratorios Hersil Location/Cultivation: La Molina-Lima, Peru; cultivation methods not stated. Form: Dehydrated, gelatinized root Dose and Route: Three groups: (1) 500 mg three times daily, every eight hours. (2) 1000 mg three times daily, every eight hours. (3) 1500 mg every day in the morning | Results of the three groups did not differ.Sexual desire increased in all three groups at weeks 8 and 12. At week 2, a significant increase in 17-alpha hydroxyprogesterone was observed in the group treated with 3000 mg, but after assessing as a percentage, there was no significant difference. No other hormones (testosterone, prolactin, FSH, LH, or E2) were significantly increased or decreased. |
| 12-week double blind placebo-controlled, randomized, parallel trial [50] in 57 healthy men, 21–56 years old | Species: Lepidium meyenii Product: Maca Gelatinizada from Laboratorios Hersil Location/Cultivation: La Molina-Lima, Peru; cultivation methods not stated. Form: Dehydrated, gelatinized root Dose and Route: 1500 mg (three 500 milligram capsules per day) or 3000 mg (six capsules of 500 mg per day) | At weeks 8 and 12, the group treated with maca had statistically significant increases (40%, p < 0.008 and 42.2%, p < 0.006, respectively) in sexual desire; no differences in the effect were noted between the dosing groups. No changes in hormone levels (T or E2) or mood (depression or anxiety) were observed. |
| 14-day, randomized, double-blind, cross-over study [49] with a 1-week washout period in 8 experienced endurance- trained male cyclists, aged 30 ± 7 years | Species: Lepidium meyenii Product: Not stated Location/Cultivation: Cerro de Pasco region of Peru, cultivation methods not stated Form: Gelatinized extract Dose and Route: 2000 mg daily (5 capsules containing 400 mg each) | Maca extract significantly improved 40 km cycling time performance compared to the baseline test (p = 0.01) but not compared to the placebo trial after supplementation (p > 0.05). Maca extract significantly improved the self-rated sexual desire score compared to the baseline test (p = 0.01) and compared to the placebo trial after supplementation (p = 0.03). |
| 12-week study [217] in 10 infertile males, 25–50 years old Article is published in Spanish; abstract in English. | Species: Lepidium meyenii Product: Not stated Location/Cultivation: Not stated Form: Not stated Dose and Route: 3000 mg per day | Improvements in sperm motility (p < 0.001), vitality (p < 0.01), and morphology (p < 0.01), immature germ cell (p < 0.01); no changes in hormone levels. |
| Case report [163] of a Caucasian female in her thirties | Species: Lepidium meyenii Product: Not stated Location/Cultivation: Not stated Form: Extract Dose and Route: 1 teaspoon of maca powder added to milk one time daily | Maca consumption increased plasma T levels without features of virilization. Testosterone immunoassays use direct immunoassays which are prone to interference from androgenic compounds resulting in falsely elevated T levels. Maca may cause T immunoassay interference in females. |
| 90-day, prospective, randomized, double-blind, placebo-controlled study [218] in 60 infertile, healthy men | Species: Lepidium meyenii Product: Supplement provided by Nature Way Products, Inc. Location/Cultivation: Not stated [abstract only] Form: Not stated [abstract only] Dose and Route: 1 g twice daily | Maca improved sperm motility (p < 0.05). |
| 4-week randomized, double-blind, placebo-controlled, parallel-group comparison study [219] in 60 healthy Japanese women 20–65 years old, with daily fatigue; 3 subjects withdrew from the study, 2 were excluded. | Species: Lepidium meyenii Product: Provided by Kinos Inc. Tokyo, Japan Location/Cultivation: Not stated Form: Concentrated extract containing 9.6 mg/day of benzyl glucosinolate Dose and Route: 200 milligram capsule, twice daily; undesignated time taken | Significant decrease in fatigue (p < 0.001) in women 20–45 years old, as measured by visual analogue scale (VAS). |
| 8-week, double-blind, placebo-controlled, parallel clinical study [155] in 42 healthy peri-menopausal Japanese women, aged 40–59 years old; 1 subject did not complete the study | Species: Lepidium meyenii Product: Maca-BG1.2TM purchased from CPX PERU S.A.C. Location/Cultivation: Peru, cultivation methods not stated Form: An extract made through washing, slicing, and oven-drying, hydrous ethanol, and evaporation to prepare powdered named Maca-BG1.2TM containing at least 1.2% benzyl glucosinolate. Dose and Route: 300 mg per day, (two 150 milligram capsules daily) after morning meal | E2 level showed a 2.2-fold increase though not of significance; further analysis revealed serum E2 level in the peri-menopausal subjects with regular menstruation cycle displayed a mostly significant change. Marginal decrease in insomnia and hot flushes. Women with mild symptoms achieved the greatest improvements in reducing menopausal symptoms. |
| Phenotype | Health Benefits |
|---|---|
| Black |
|
| Red |
|
| Yellow |
|
| Purple |
|
| Gray |
|
| Combination of colors: Maca-GO® |
|
| Combination of colors: MACAXSTM |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Minich, D.M.; Ross, K.; Frame, J.; Fahoum, M.; Warner, W.; Meissner, H.O. Not All Maca Is Created Equal: A Review of Colors, Nutrition, Phytochemicals, and Clinical Uses. Nutrients 2024, 16, 530. https://doi.org/10.3390/nu16040530
Minich DM, Ross K, Frame J, Fahoum M, Warner W, Meissner HO. Not All Maca Is Created Equal: A Review of Colors, Nutrition, Phytochemicals, and Clinical Uses. Nutrients. 2024; 16(4):530. https://doi.org/10.3390/nu16040530
Chicago/Turabian StyleMinich, Deanna M., Kim Ross, James Frame, Mona Fahoum, Wendy Warner, and Henry O. Meissner. 2024. "Not All Maca Is Created Equal: A Review of Colors, Nutrition, Phytochemicals, and Clinical Uses" Nutrients 16, no. 4: 530. https://doi.org/10.3390/nu16040530
APA StyleMinich, D. M., Ross, K., Frame, J., Fahoum, M., Warner, W., & Meissner, H. O. (2024). Not All Maca Is Created Equal: A Review of Colors, Nutrition, Phytochemicals, and Clinical Uses. Nutrients, 16(4), 530. https://doi.org/10.3390/nu16040530