Exploring Vegetarian and Omnivorous Approaches to Cardiovascular Risk and Body Composition
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Ethical Considerations
2.2. Data Collection
2.3. Measurement Outcomes
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO Regional Office for Europe. WHO European Regional Obesity Report 2022; WHO Regional Office for Europe: Copenhagen, Denmark, 2022; ISBN 978-92-890-5773-8. [Google Scholar]
- Cederholm, T.; Barazzoni, R.; Austin, P.; Ballmer, P.; Biolo, G.; Bischoff, S.C.; Compher, C.; Correia, I.; Higashiguchi, T.; Holst, M.; et al. ESPEN Guidelines on Definitions and Terminology of Clinical Nutrition. Clin. Nutr. 2017, 36, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Storz, C.; Heber, S.D.; Rospleszcz, S.; Machann, J.; Sellner, S.; Nikolaou, K.; Lorbeer, R.; Gatidis, S.; Elser, S.; Peters, A.; et al. The Role of Visceral and Subcutaneous Adipose Tissue Measurements and Their Ratio by Magnetic Resonance Imaging in Subjects with Prediabetes, Diabetes and Healthy Controls from a General Population without Cardiovascular Disease. Br. J. Radiol. 2018, 91, 20170808. [Google Scholar] [CrossRef] [PubMed]
- Fox, C.S.; Massaro, J.M.; Hoffmann, U.; Pou, K.M.; Maurovich-Horvat, P.; Liu, C.Y.; Vasan, R.S.; Murabito, J.M.; Meigs, J.B.; Cupples, L.A.; et al. Abdominal Visceral and Subcutaneous Adipose Tissue Compartments: Association with Metabolic Risk Factors in the Framingham Heart Study. Circulation 2007, 116, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Lee, G.; Choi, S.; Oh, Y.H.; Son, J.S.; Park, M.; Park, S.M. Changes in Predicted Lean Body Mass, Appendicular Skeletal Muscle Mass, and Body Fat Mass and Cardiovascular Disease. J. Cachexia Sarcopenia Muscle 2022, 13, 1113–1123. [Google Scholar] [CrossRef] [PubMed]
- Frühbeck, G.; Busetto, L.; Dicker, D.; Yumuk, V.; Goossens, G.H.; Hebebrand, J.; Halford, J.G.C.; Farpour-Lambert, N.J.; Blaak, E.E.; Woodward, E.; et al. The ABCD of Obesity: An EASO Position Statement on a Diagnostic Term with Clinical and Scientific Implications. Obes. Facts 2019, 12, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.; Guo, Z.; Johnson, C.M.; Hensrud, D.D.; Jensen, M.D. Splanchnic Lipolysis in Human Obesity. J. Clin. Investig. 2004, 113, 1582–1588. [Google Scholar] [CrossRef] [PubMed]
- Porter, S.A.; Massaro, J.M.; Hoffmann, U.; Vasan, R.S.; O’Donnel, C.J.; Fox, C.S. Abdominal Subcutaneous Adipose Tissue: A Protective Fat Depot? Diabetes Care 2009, 32, 1068–1075. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, S.M.; Raposo, A.; Saraiva, A.; Zandonadi, R.P. Vegetarian Diet: An Overview through the Perspective of Quality of Life Domains. IJERPH 2021, 18, 4067. [Google Scholar] [CrossRef]
- Fontes, T.; Rodrigues, L.M.; Ferreira-Pêgo, C. Comparison between Different Groups of Vegetarianism and Its Associations with Body Composition: A Literature Review from 2015 to 2021. Nutrients 2022, 14, 1853. [Google Scholar] [CrossRef] [PubMed]
- Pilis, W.; Stec, K.; Zych, M.; Pilis, A. Health benefits and risk associated with adopting a vegetarian diet. Natl. Inst. Public Health Natl. Inst. Hyg. 2014, 65, 9–14. [Google Scholar]
- Arnett, D.K.; Blumenthal, R.S.; Albert, M.A.; Buroker, A.B.; Goldberger, Z.D.; Hahn, E.J.; Himmelfarb, C.D.; Khera, A.; Lloyd-Jones, D.; McEvoy, J.W.; et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019, 140, e596–e646. [Google Scholar] [CrossRef] [PubMed]
- Godfray, H.C.J.; Aveyard, P.; Garnett, T.; Hall, J.W.; Key, T.J.; Lorimer, J.; Pierrehumbert, R.T.; Scarborough, P.; Springmann, M.; Jebb, S.A. Meat Consumption, Health, and the Environment. Science 2018, 361, eaam5324. [Google Scholar] [CrossRef] [PubMed]
- Monteiro Rodrigues, L.; Menezes, R.; Andrade, S.; Fontes, T.; Lopes, S.; Ferreira-Pego, C. Dietary Pattern, Body Composition, Metabolic Markers, and Skin Physiology. Physiology 2023, 38, 5790039. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Nishimura, K.; Barnard, N.D.; Takegami, M.; Watanabe, M.; Sekikawa, A.; Okamura, T.; Miyamoto, Y. Vegetarian Diets and Blood Pressure: A Meta-Analysis. JAMA Intern. Med. 2014, 174, 577. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Ma, G.; Chen, S.; Li, Z.; Xia, E.; Sun, Y.; Yang, F.; Zheng, L.; Feng, X. Comparison of Plasma Triacylglycerol Levels in Vegetarians and Omnivorous: A Meta-Analysis. Nutrition 2013, 29, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Ferreira-Pêgo, C.; Giacomelli Tavares, R.; Lopes, S.; Fontes, T.; Monteiro Rodrigues, L. Body Composition Assessment of Vegetarian-Vegan and Omnivorous Young Women—An Exploratory Study: Body Composition Assessment of Vegetarian-Vegan and Omnivorous Young Women—An Exploratory Study. BBR 2021, 18, 15. [Google Scholar] [CrossRef]
- He, Y.-M.; Chen, W.-L.; Kao, T.-W.; Wu, L.-W.; Yang, H.-F.; Peng, T.-C. Association Between Ideal Cardiovascular Health and Vegetarian Dietary Patterns Among Community-Dwelling Individuals. Front. Nutr. 2022, 9, 761982. [Google Scholar] [CrossRef] [PubMed]
- World Medical Association (WMA). Declaration of Helsinki—Ethical Principles for Medical Research Involving Human Subjects. J. Am. Coll. Dent. 2013, 81, 14–18. [Google Scholar]
- Instituto Nacional de Estatística. Nuts 2013|As Novas Unidades Territoriais Para Fins Estatísticos; Instituto Nacional de Estatística: Lisboa, Portugal, 2015. [Google Scholar]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International Physical Activity Questionnaire: 12-Country Reliability and Validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Lopes, C.; Aro, A.; Azevedo, A.; Ramos, E.; Barros, H. Intake and Adipose Tissue Composition of Fatty Acids and Risk of Myocardial Infarction in a Male Portuguese Community Sample. J. Am. Diet. Assoc. 2007, 107, 276–286. [Google Scholar] [CrossRef]
- Keys, A.; Fidanza, F.; Karvonen, M.; Kimura, N.; Taylor, H.L. Indices of relative weight and obesity. J. Chronic Dis. 1972, 25, 329–343. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, R.N.; Koehler, K.M.; Gallagher, D.; Romero, L.; Heymsfield, S.B.; Ross, R.R.; Garry, P.J.; Lindeman, R.D. Epidemiology of Sarcopenia among the Elderly in New Mexico. Am. J. Epidemiol. 1998, 147, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Woolcott, O.O.; Bergman, R.N. Defining Cutoffs to Diagnose Obesity Using the Relative Fat Mass (RFM): Association with Mortality in NHANES 1999–2014. Int. J. Obes. 2020, 44, 1301–1310. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European Consensus on Definition and Diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Cederholm, T.; Jensen, G.L.; Correia, M.I.T.D.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.; et al. GLIM Criteria for the Diagnosis of Malnutrition—A Consensus Report from the Global Clinical Nutrition Community. Clin. Nutr. 2019, 38, 207–217. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Waist Circumference and Waist-Hip Ratio: Report of a WHO Expert Consultation. Geneva, Switzerland, 8–11 December 2008; 2011. Available online: https://iris.who.int/bitstream/handle/10665/44583/9789241501491_eng.pdf?sequence=1 (accessed on 18 June 2024).
- Visseren, F.L.J.; Mach, F.; Smulders, Y.M.; Carballo, D.; Koskinas, K.C.; Bäck, M.; Benetos, A.; Biffi, A.; Boavida, J.-M.; Capodanno, D.; et al. 2021 ESC Guidelines on Cardiovascular Disease Prevention in Clinical Practice. Eur. Heart J. 2021, 42, 3227–3337. [Google Scholar] [CrossRef] [PubMed]
- Magder, S. The Meaning of Blood Pressure. Crit. Care 2018, 22, 257. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Standards of Medical Care in Diabetes—2022 Abridged for Primary Care Providers. Clin. Diabetes 2022, 40, 10–38. [Google Scholar] [CrossRef] [PubMed]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS Guidelines for the Management of Dyslipidaemias: Lipid Modification to Reduce Cardiovascular Risk. Eur. Heart J. 2020, 41, 111–188. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.M.M.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.-C.; James, W.P.T.; Loria, C.M.; Smith, S.C. Harmonizing the Metabolic Syndrome: A Joint Interim Statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar] [CrossRef] [PubMed]
- Hippisley-Cox, J.; Coupland, C.; Brindle, P. Development and Validation of QRISK3 Risk Prediction Algorithms to Estimate Future Risk of Cardiovascular Disease: Prospective Cohort Study. BMJ 2017, 357, j2099. [Google Scholar] [CrossRef] [PubMed]
- Free Software Foundation GNU Lesser General Public License v3.0—GNU Project. Available online: https://www.gnu.org/licenses/lgpl-3.0-standalone.html (accessed on 18 June 2024).
- Kahleova, H.; Dort, S.; Holubkov, R.; Barnard, N. A Plant-Based High-Carbohydrate, Low-Fat Diet in Overweight Individuals in a 16-Week Randomized Clinical Trial: The Role of Carbohydrates. Nutrients 2018, 10, 1302. [Google Scholar] [CrossRef] [PubMed]
- Kahleova, H.; Hlozkova, A.; Fleeman, R.; Fletcher, K.; Holubkov, R.; Barnard, N.D. Fat Quantity and Quality, as Part of a Low-Fat, Vegan Diet, Are Associated with Changes in Body Composition, Insulin Resistance, and Insulin Secretion. A 16-Week Randomized Controlled Trial. Nutrients 2019, 11, 615. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Dinu, M.; Pagliai, G.; Cesari, F.; Gori, A.M.; Sereni, A.; Becatti, M.; Fiorillo, C.; Marcucci, R.; Casini, A. Low-Calorie Vegetarian Versus Mediterranean Diets for Reducing Body Weight and Improving Cardiovascular Risk Profile: CARDIVEG Study (Cardiovascular Prevention With Vegetarian Diet). Circulation 2018, 137, 1103–1113. [Google Scholar] [CrossRef] [PubMed]
- Shah, B.; Newman, J.D.; Woolf, K.; Ganguzza, L.; Guo, Y.; Allen, N.; Zhong, J.; Fisher, E.A.; Slater, J. Anti-Inflammatory Effects of a Vegan Diet Versus the American Heart Association–Recommended Diet in Coronary Artery Disease Trial. JAHA 2018, 7, e011367. [Google Scholar] [CrossRef]
- Zhang, H.J.; He, J.; Pan, L.L.; Ma, Z.M.; Han, C.K.; Chen, C.S.; Chen, Z.; Han, H.W.; Chen, S.; Sun, Q.; et al. Effects of Moderate and Vigorous Exercise on Nonalcoholic Fatty Liver Disease: A Randomized Clinical Trial. JAMA Intern. Med. 2016, 176, 1074. [Google Scholar] [CrossRef]
- Duan, Y.; Li, F.; Li, Y.; Tang, Y.; Kong, X.; Feng, Z.; Anthony, T.G.; Watford, M.; Hou, Y.; Wu, G.; et al. The Role of Leucine and Its Metabolites in Protein and Energy Metabolism. Amino Acids 2016, 48, 41–51. [Google Scholar] [CrossRef]
- Oussalah, A.; Levy, J.; Berthezène, C.; Alpers, D.H.; Guéant, J.-L. Health Outcomes Associated with Vegetarian Diets: An Umbrella Review of Systematic Reviews and Meta-Analyses. Clin. Nutr. 2020, 39, 3283–3307. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B.; Rosner, B.A. Dietary Fat Intake and the Risk of Coronary Heart Disease in Women. N. Engl. J. Med. 1997, 337, 1491–1499. [Google Scholar] [CrossRef]
- Chandalia, M.; Garg, A.; Lutjohann, D.; Von Bergmann, K.; Grundy, S.M.; Brinkley, L.J. Beneficial Effects of High Dietary Fiber Intake in Patients with Type 2 Diabetes Mellitus. N. Engl. J. Med. 2000, 342, 1392–1398. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.; Kendall, C.W.; Marchie, A.; Jenkins, A.L.; Augustin, L.S.; Ludwig, D.S.; Barnard, N.D.; Anderson, J.W. Type 2 Diabetes and the Vegetarian Diet. Am. J. Clin. Nutr. 2003, 78, 610S–616S. [Google Scholar] [CrossRef] [PubMed]
- Rydén, L.; Grant, P.J.; Anker, S.D.; Berne, C.; Cosentino, F.; Danchin, N.; Deaton, C.; Escaned, J.; Hammes, H.P.; Huikuri, H.; et al. The Task Force on Diabetes, Pre-Diabetes, and Cardiovascular Diseases of the European Society of Cardiology (ESC) and Developed in Collaboration with the European Association for the Study of Diabetes (EASD). Rev. Española Cardiol. 2014, 67, 136. [Google Scholar] [CrossRef]
- Yogal, C.; Shakya, S.; Karmarcharya, B.; Koju, R.; Stunes, A.K.; Mosti, M.P.; Gustafsson, M.K.; Åsvold, B.O.; Schei, B.; Syversen, U. Unni Syversen Diabetes Prevalence and Associated Risk Factors among Women in a Rural District of Nepal Using HbA1c as a Diagnostic Tool: A Population-Based Study. IJERPH 2022, 1, 7011. [Google Scholar] [CrossRef] [PubMed]
- Abraham, T.M.; Pedley, A.; Massaro, J.M.; Hoffmann, U.; Fox, C.S. Association Between Visceral and Subcutaneous Adipose Depots and Incident Cardiovascular Disease Risk Factors. Circulation 2015, 132, 1639–1647. [Google Scholar] [CrossRef] [PubMed]
- Neeland, I.J.; Poirier, P.; Després, J.P. Cardiovascular and Metabolic Heterogeneity of Obesity: Clinical Challenges and Implications for Management. Circulation 2018, 137, 1391–1406. [Google Scholar] [CrossRef]
- Saito, K.; Ueno, S.; Nakayama, A.; Nitta, S.; Arai, K.; Hasunuma, T.; Saito, Y. Overall Similarities and a Possible Factor Affecting Plasma Metabolome Profiles Between Venous and Capillary Blood Samples From 20 Healthy Human Males. J. Pharm. Sci. 2019, 108, 3737–3744. [Google Scholar] [CrossRef] [PubMed]
- Stahl, M.; Brandslund, I.; Jørgensen, L.G.M.; Petersen, P.H.; Borch-Johnsen, K.; Olivarius, N.D.F. Can Capillary Whole Blood Glucose and Venous Plasma Glucose Measurements Be Used Interchangeably in Diagnosis of Diabetes Mellitus? Scand. J. Clin. Lab. Investig. 2002, 62, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, L.M.; Haynie, D.L.; Hill, C.; Nansel, T.R.; Li, K.; Liu, D.; Iannotti, R.J.; Simons-Morton, B. Accuracy of Self-Reported Height, Weight, and BMI Over Time in Emerging Adults. Am. J. Prev. Med. 2019, 56, 860–868. [Google Scholar] [CrossRef] [PubMed]
| All Population (n = 176) | Omnivorous (n = 115) | Vegetarian (n = 61) | p-Value | ||
|---|---|---|---|---|---|
| Sex, % (n) | Men | 35.8 (63) | 33.0 (38) | 41.0 (25) | 0.296 b |
| Women | 64.2 (113) | 67.0 (77) | 59.0 (36) | ||
| Age, years | 31.0 (20.0) | 30.0 (22.0) | 33.0 (16.0) | 0.185 a | |
| Height, m | 1.7 (0.1) | 1.7 (0.1) | 1.7 (0.1) | 0.169 a | |
| Body mass, kg | 64.1 (21.3) | 63.3 (21.4) | 65.6 (20.0) | 0.521 a | |
| BMI, kg/m2 | 22.5 (3.9) | 22.4 (4.3) | 22.7 (3.8) | 0.756 a | |
| Waist circumference, cm | Men | 83.0 (12.0) | 83.0 (10.8) | 85.0 (12.7) | 0.855 a |
| Women | 71.0 (13.7) | 70.1 (14.0) | 72.0 (11.9) | 0.205 a | |
| Academic qualifications, % (n) | High school | 34.7 (61) | 33.9 (39) | 36.1 (22) | 0.895 b |
| BSc degree | 38.6 (68) | 38.3 (44) | 39.3 (24) | ||
| MSc and PhD | 26.7 (47) | 27.8 (32) | 24.6 (15) | ||
| Time following the dietary pattern, % (n) | Between 1 to 5 years | 14.2 (25) | 0.0 (0) | 41.0 (25) | <0.001 b |
| Between 5 to 10 years | 14.2 (25) | 0.0 (0) | 41.0 (25) | ||
| More than 10 years | 71.6 (126) | 100.0 (115) | 18.0 (11) | ||
| Smoking habits, % (n) | Non-Smoking | 77.8 (137) | 77.4 (89) | 78.7 (48) | 0.656 b |
| Former smoker | 11.4 (20) | 10.4 (12) | 13.1 (8) | ||
| Smoker | 10.8 (19) | 12.2 (14) | 8.2 (5) | ||
| Levels physical activity, % (n) | Low | 25.0 (44) | 27.0 (31) | 21.3 (13) | 0.014 b |
| Moderate | 48.9 (86) | 53.9 (62) | 39.3 (24) | ||
| High | 26.1 (46) | 19.1 (22) | 39.3 (24) | ||
| Family monthly income, % (n) | <1000 € | 10.2 (18) | 7.0 (8) | 16.4 (10) | 0.127 b |
| 1000–3000 € | 59.1 (104) | 60.0 (69) | 57.4 (35) | ||
| >3000 € | 30.7 (54) | 33.0 (38) | 26.2 (16) | ||
| All Population (n = 176) | p-Value | Men (n = 63) | p-Value | Women (n = 113) | p-Value | ||||
|---|---|---|---|---|---|---|---|---|---|
| Omnivorous (n = 115) | Vegetarian (n = 61) | Omnivorous (n = 38) | Vegetarian (n = 25) | Omnivorous (n = 77) | Vegetarian (n = 36) | ||||
| Energy, kcal | 2392.11 (758.33) | 2066.40 (602.13) | 0.004 a | 2537.58 (1512.23) | 1991.36 (572.76) | 0.024 b | 2338.50 (706.31) | 2119.11 (666.89) | 0.120 a |
| Proteins, % TEV | 18.00 (5.46) | 13.07 (6.59) | <0.001 b | 18.61 (6.64) | 12.56 (6.72) | 0.002 b | 17.76 (5.36) | 13.88 (6.81) | <0.001 b |
| Carbohydrates, % TEV | 42.24 (11.97) | 50.20 (18.09) | <0.001 b | 44.84 (11.73) | 53.34 (10.20) | 0.004 a | 43.35 (8.97) | 47.57 (12.20) | 0.041 a |
| Complex, % TEV | 15.62 (4.81) | 17.91 (5.12) | 0.004 a | 16.11 (6.64) | 18.08 (6.16) | 0.013 b | 14.81 (4.62) | 18.35 (7.69) | 0.077 b |
| Sugars, % TEV | 16.20 (6.16) | 17.49 (8.33) | 0.044 b | 15.49 (7.32) | 18.35 (10.70) | 0.095 b | 16.48 (5.69) | 17.29 (6.48) | 0.247 b |
| Total fat, % TEV | 39.72 (7.71) | 38.38 (9.43) | 0.311 a | 38.95 (7.96) | 35.93 (8.57) | 0.158 a | 40.10 (7.60) | 40.08 (9.73) | 0.989 a |
| Saturated fat, % TEV | 9.43 (2.47) | 7.84 (2.76) | <0.001 a | 9.45 (3.93) | 7.11 (4.88) | 0.013 b | 9.66 (2.41) | 8.11 (4.51) | 0.004 b |
| Monounsaturated fat, % TEV | 20.44 (5.35) | 20.84 (6.40) | 0.665 a | 19.75 (4.82) | 19.28 (5.63) | 0.727 a | 20.78 (5.59) | 21.91 (6.75) | 0.351 a |
| Polyunsaturated fat, % TEV | 6.63 (1.60) | 6.96 (2.43) | 0.443 b | 6.70 (1.61) | 6.37 (1.73) | 0.653 b | 6.71 (1.20) | 7.06 (1.67) | 0.202 a |
| Cholesterol, mg | 349.25 (221.77) | 155.29 (292.58) | <0.001 b | 350.03 (436.30) | 45.00 (265.26) | <0.001 b | 349.03 (156.96) | 229.06 (291.25) | <0.001 b |
| Dietary fiber, g | 30.85 (17.39) | 35.32 (22.32) | 0.027 b | 34.54 (20.49) | 36.09 (21.92) | 0.465 b | 29.61 (17.36) | 34.06 (22.15) | 0.039 b |
| Alcohol, g | 3.99 (5.97) | 3.11 (4.66) | 0.152 a | 5.07 (7.93) | 2.12 (2.98) | 0.019 a | 3.46 (4.70) | 3.80 (5.46) | 0.627 a |
| Only consumers, g | 3.18 (7.27) | 2.58 (3.55) | 0.285 b | 3.40 (7.47) | 2.70 (2.86) | 0.127 b | 2.71 (5.76) | 2.52 (4.60) | 0.829 b |
| Caffeine, mg | 41.35 (69.55) | 34.83 (78.53) | 0.443 b | 40.44 (79.53) | 25.81 (79.38) | 0.305 b | 41.35 (62.28) | 41.11 (76.78) | 0.961 b |
| Only consumers, mg | 41.35 (69.55) | 34.89 (77.79) | 0.551 b | 40.44 (79.53) | 25.81 (79.38) | 0.305 b | 41.35 (62.28) | 46.24 (76.79) | 0.770 b |
| All Population (n = 176) | p-Value | Adjusted p-Value | Men (n = 63) | p-Value | Adjusted p-Value | Women (n = 113) | p-Value | Adjusted p-Value | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Omnivorous (n = 115) | Vegetarian (n = 61) | Omnivorous (n = 38) | Vegetarian (n = 25) | Omnivorous (n = 77) | Vegetarian (n = 36) | |||||||
| Fat mass, % | 29.23 (8.94) | 28.38 (8.24) | 0.534 a | 0.306 | 21.89 (6.80) | 22.38 (6.28) | 0.773 a | 0.096 | 32.85 (7.56) | 32.52 (6.77) | 0.827 a | 0.961 |
| VAT, cm3 | 210.00 (583.00) | 261.00 (482.00) | 0.397 b | 0.945 | 323.00 (737.00) | 532.00 (495.00) | 0.550 b | 0.084 | 173.00 (377.00) | 205.50 (323.00) | 0.753 b | 0.180 |
| SAT, cm3 | 893.00 (935.00) | 1039.00 (842.00) | 0.522 b | 0.074 | 875.50 (915.00) | 947.00 (811.00) | 0.911 b | 0.056 | 898.00 (983.00) | 1053.00 (910.00) | 0.373 b | 0.441 |
| VAT/SAT Ratio | 0.26 (0.31) | 0.29 (0.29) | 0.530 b | 0.183 | 0.40 (0.54) | 0.46 (0.53) | 0.347 b | 0.425 | 0.23 (0.26) | 0.23 (0.20) | 0.573 b | 0.199 |
| Fat-free mass, % | 70.77 (8.94) | 71.63 (8.24) | 0.535 a | 0.305 | 78.11 (6.81) | 77.61 (6.29) | 0.770 a | 0.181 | 67.14 (7.55) | 67.47 (6.77) | 0.827 a | 0.956 |
| Bone mass, % | 3.77 (0.54) | 3.78 (0.58) | 0.603 b | 0.573 | 3.87 (0.40) | 3.98 (0.47) | 0.300 a | 0.394 | 3.77 (0.60) | 3.61 (0.52) | 0.166 b | 0.191 |
| Lean mass, % | 67.01 (8.60) | 67.84 (7.94) | 0.532 a | 0.306 | 74.25 (6.60) | 73.63 (5.98) | 0.709 a | 0.013 | 63.44 (7.12) | 63.83 (6.55) | 0.785 a | 0.888 |
| ALMI, Kg/m2 | 6.68 (2.20) | 6.70 (1.71) | 0.743 b | 0.306 | 8.40 (0.88) | 7.93 (0.99) | 0.055 a | 0.006 | 6.21 (0.89) | 6.27 (0.72) | 0.759 a | 0.650 |
| All Population (n = 176) | Omnivorous (n = 115) | Vegetarian (n = 61) | p-Value | Adjusted p-Value | |
|---|---|---|---|---|---|
| Hemoglobin A1c, % | 5.40 (0.60) | 5.40 (0.50) | 5.30 (0.80) | 0.613 b | 0.548 |
| Triglycerides, mg/dL | 102.00 (74.00) | 107.00 (75.00) | 99.00 (67.00) | 0.715 b | 0.740 |
| Fasting glycemia, mg/dL | 91.00 (13.00) | 91.00 (14.00) | 92.00 (11.00) | 0.682 b | 0.829 |
| Total cholesterol, mg/dL | 175.49 (35.48) | 183.83 (38.47) | 159.77 (21.79) | <0.001 a | <0.001 |
| cHDL, mg/dL | 55.50 (20.00) | 57.00 (22.00) | 53.00 (19.00) | 0.114 b | 0.222 |
| cLDL, mg/dL | 89.00 (36.00) | 93.00 (46.00) | 83.00 (28.00) | 0.002 b | <0.001 |
| cVLDL, mg/dL | 21.00 (15.00) | 21.00 (15.00) | 20.00 (13.00) | 0.646 b | 0.510 |
| Total cholesterol/cHDL, mg/dL | 3.04 (1.16) | 3.00 (1.29) | 3.13 (0.89) | 0.637 b | 0.159 |
| Non-HDL cholesterol, mg/dL | 112.00 (43.00) | 116.00 (51.00) | 106.00 (28.00) | <0.001 b | <0.001 |
| SBP, mmHg | 110.00 (17.00) | 110.00 (19.00) | 110.00 (16.00) | 0.667 b | 0.742 |
| DBP, mmHg | 74.00 (14.00) | 74.00 (14.00) | 73.00 (14.00) | 0.311 b | 0.382 |
| MAP, mmHg | 86.33 (13.67) | 87.00 (14.33) | 84.67 (13.83) | 0.356 b | 0.479 |
| Heart Rate, bpm | 67.00 (12.00) | 67.00 (11.00) | 67.00 (14.00) | 0.494 b | 0.784 |
| Cardiovascular disease risk * | |||||
| 10-year QRISK3 score, % | 0.70 (1.75) | 0.90 (2.93) | 0.40 (0.80) | 0.077 b | 0.184 |
| Relative risk | 0.80 (0.40) | 0.80 (0.43) | 0.70 (0.30) | 0.137 b | 0.990 |
| Men (n = 63) | p-Value | Adjusted p-Value | Women (n = 113) | p-Value | Adjusted p-Value | |||
|---|---|---|---|---|---|---|---|---|
| Omnivorous (n = 38) | Vegetarian (n = 25) | Omnivorous (n = 77) | Vegetarian (n = 36) | |||||
| Body Composition, % (n) | ||||||||
| Fat mass excess | 13.20 (5) | 8.00 (2) | 0.693 a | 0.683 1 | 19.50 (15) | 11.10 (4) | 0.268 b | 0.217 1 |
| Lean mass deficit | 7.90 (3) | 16.00 (4) | 0.421 a | 0.404 1 | 16.90 (13) | 11.10 (4) | 0.424 b | 0.685 1 |
| Anthropometrics, % (n) | ||||||||
| Increased WC | 7.90 (3) | 12.00 (3) | 0.799 c | 0.193 1 | 9.10 (7) | 13.9 (5) | 0.517 b | 0.970 1 |
| Substantially increased WC | 2.60 (1) | 0.00 (0) | 18.2 (14) | 11.1 (4) | ||||
| Metabolic markers and vital signs % (n) | ||||||||
| Elevated hemoglobin A1c | 23.70 (9) | 16.00 (4) | 0.461 b | 0.637 2 | 20.80 (16) | 47.20 (17) | 0.004 b | 0.009 2 |
| Elevated triglycerides | 26.30 (10) | 28.00 (7) | 0.883 b | 0.624 2 | 20.80 (16) | 16.70 (6) | 0.607 b | 0.745 2 |
| Elevated fasting glycemia | 39.5 (15) | 20.00 (5) | 0.104 b | 0.344 2 | 20.80 (16) | 19.40 (7) | 0.870 b | 0.874 2 |
| Elevated total cholesterol | 68.40 (26) | 52.00 (13) | 0.189 b | 0.497 2 | 79.20 (61) | 61.10 (22) | 0.042 b | 0.024 2 |
| Reduced cHDL | 31.60 (12) | 8.00 (2) | 0.028 b | 0.036 2 | 10.40 (8) | 30.60 (11) | 0.008 b | 0.002 2 |
| Elevated cLDL | 50.00 (19) | 20.00 (5) | 0.016 b | 0.007 2 | 40.30 (31) | 22.20 (8) | 0.060 b | 0.017 2 |
| Elevated SBP | 23.70 (9) | 20.00 (5) | 0.731 b | 0.987 2 | 7.80 (6) | 0.00 (0) | 0.174 a | 0.153 2 |
| Elevated DBP | 26.30 (10) | 20.00 (5) | 0.565 b | 0.675 2 | 14.30 (11) | 8.30 (3) | 0.543 a | 0.776 2 |
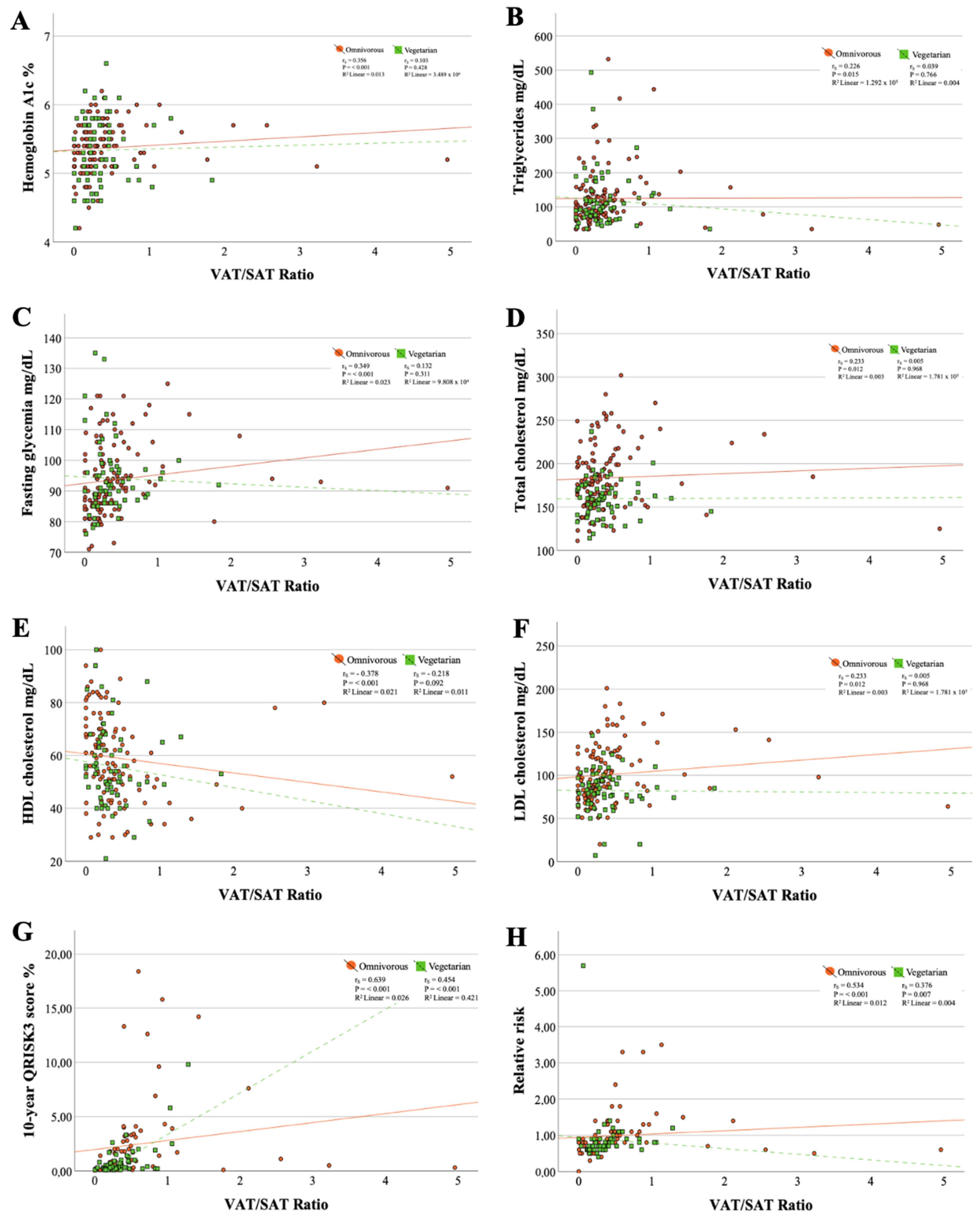
| Fat Mass, % | VAT, cm3 | SAT, cm3 | ||||
|---|---|---|---|---|---|---|
| Correlation | p-Value a,b | Correlation | p-Value a | Correlation | p-Value a,b | |
| Omnivorous population | ||||||
| Hemoglobin A1c, % | 0.058 | 0.537 a | 0.298 | <0.001 | 0.078 | 0.410 a |
| Triglycerides, mg/dL | 0.230 | 0.013 a | 0.369 | <0.001 | 0.349 | <0.001 a |
| Fasting glycemia, mg/dL | 0.169 | 0.071 a | 0.414 | <0.001 | 0.275 | 0.003 a |
| Total cholesterol, mg/dL | 0.393 | <0.001 b | 0.317 | <0.001 | 0.271 | 0.003 a |
| HDL cholesterol, mg/dL | 0.067 | 0.478 b | −0.411 | <0.001 | −0.250 | 0.007 a |
| LDL cholesterol, mg/dL | 0.291 | 0.002 a | 0.429 | <0.001 | 0.324 | <0.001 a |
| MAP, mmHg | 0.027 | 0.772 a | 0.329 | <0.001 | 0.172 | 0.066 a |
| 10-year QRISK3 score, % | 0.259 | 0.026 a | 0.687 | <0.001 | 0.380 | <0.001 a |
| Relative risk | 0.193 | 0.100 a | 0.606 | <0.001 | 0.395 | <0.001 a |
| Vegetarian population | ||||||
| Hemoglobin A1c, % | 0.328 | 0.010 b | 0.176 | 0.175 | 0.177 | 0.173 b |
| Triglycerides, mg/dL | −0.029 | 0.825 a | 0.055 | 0.672 | 0.061 | 0.640 a |
| Fasting glycemia, mg/dL | 0.017 | 0.897 a | 0.169 | 0.193 | 0.061 | 0.641 a |
| Total cholesterol, mg/dL | 0.104 | 0.425 b | 0.120 | 0.357 | 0.079 | 0.543 b |
| HDL cholesterol, mg/dL | −0.156 | 0.229 a | −0.250 | 0.052 | −0.261 | 0.043 a |
| LDL cholesterol, mg/dL | 0.292 | 0.022 b | 0.192 | 0.138 | 0.266 | 0.038 b |
| MAP, mmHg | 0.025 | 0.302 a | 0.510 | 0.018 | 0.194 | 0.133 a |
| 10-year QRISK3 score, % | 0.203 | 0.154 a | 0.583 | < 0.001 | 0.357 | 0.010 a |
| Relative risk | 0.136 | 0.341 a | 0.510 | < 0.001 | 0.380 | 0.006 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fontes, T.; Lopes, S.; Menezes, R.; Esgalhado, M.; Monteiro Rodrigues, L.; Ferreira-Pêgo, C. Exploring Vegetarian and Omnivorous Approaches to Cardiovascular Risk and Body Composition. Nutrients 2024, 16, 2013. https://doi.org/10.3390/nu16132013
Fontes T, Lopes S, Menezes R, Esgalhado M, Monteiro Rodrigues L, Ferreira-Pêgo C. Exploring Vegetarian and Omnivorous Approaches to Cardiovascular Risk and Body Composition. Nutrients. 2024; 16(13):2013. https://doi.org/10.3390/nu16132013
Chicago/Turabian StyleFontes, Tatiana, Sofia Lopes, Regina Menezes, Marta Esgalhado, Luís Monteiro Rodrigues, and Cíntia Ferreira-Pêgo. 2024. "Exploring Vegetarian and Omnivorous Approaches to Cardiovascular Risk and Body Composition" Nutrients 16, no. 13: 2013. https://doi.org/10.3390/nu16132013
APA StyleFontes, T., Lopes, S., Menezes, R., Esgalhado, M., Monteiro Rodrigues, L., & Ferreira-Pêgo, C. (2024). Exploring Vegetarian and Omnivorous Approaches to Cardiovascular Risk and Body Composition. Nutrients, 16(13), 2013. https://doi.org/10.3390/nu16132013










