Medical Cannabis Increases Appetite but Not Body Weight in Patients with Inflammatory Bowel Diseases
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Data Collection
2.3. MC Treatment and Dose Calculation
2.4. Appetite, Nutritional Status and Intake
2.5. Statistical Analysis
3. Results
3.1. Study Population Characteristics
3.2. Treatment Dose and Indications at Baseline
3.3. Effect of MC Treatment on Appetite and Dietary Intake
3.4. Adherence to Treatment and Safety
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Park, J.; Jeong, G.H.; Song, M.; Yon, D.K.; Lee, S.W.; Koyanagi, A.; Jacob, L.; Kostev, K.; Dragioti, E.; Radua, J.; et al. The Global, Regional, and National Burden of Inflammatory Bowel Diseases, 1990–2019: A Systematic Analysis for the Global Burden of Disease Study 2019. Dig. Liver Dis. 2023, 55, 1352–1359. [Google Scholar] [CrossRef]
- Wang, R.; Li, Z.; Liu, S.; Zhang, D. Global, Regional and National Burden of Inflammatory Bowel Disease in 204 Countries and Territories from 1990 to 2019: A Systematic Analysis Based on the Global Burden of Disease Study 2019. BMJ Open 2023, 13, e065186. [Google Scholar] [CrossRef]
- Lin, A.; Micic, D. Nutrition Considerations in Inflammatory Bowel Disease. Nutr. Clin. Pract. 2021, 36, 298–311. [Google Scholar] [CrossRef]
- Ünal, N.G.; Oruç, N.; Tomey, O.; Ömer Özütemiz, A. Malnutrition and Sarcopenia Are Prevalent among Inflammatory Bowel Disease Patients with Clinical Remission. Eur. J. Gastroenterol. Hepatol. 2021, 33, 1367–1375. [Google Scholar] [CrossRef]
- Balestrieri, P.; Ribolsi, M.; Guarino, M.P.L.; Emerenziani, S.; Altomare, A.; Cicala, M. Nutritional Aspects in Inflammatory Bowel Diseases. Nutrients 2020, 12, 372. [Google Scholar] [CrossRef]
- Pulley, J.; Todd, A.; Flatley, C.; Begun, J. Malnutrition and Quality of Life among Adult Inflammatory Bowel Disease Patients. JGH Open 2020, 4, 454–460. [Google Scholar] [CrossRef]
- Bischoff, S.C.; Escher, J.; Hébuterne, X.; Kłęk, S.; Krznaric, Z.; Schneider, S.; Shamir, R.; Stardelova, K.; Wierdsma, N.; Wiskin, A.E.; et al. ESPEN Practical Guideline: Clinical Nutrition in Inflammatory Bowel Disease. Clin. Nutr. 2020, 39, 632–653. [Google Scholar] [CrossRef]
- Sriranganathan, D.; Segal, J.P.; Garg, M. Biologics Recommendations in the ECCO Guidelines on Therapeutics in Crohn’s Disease: Medical Treatment. Frontline Gastroenterol. 2022, 13, 168–170. [Google Scholar] [CrossRef]
- Greywoode, R.; Cunningham, C.; Hollins, M.; Aroniadis, O. Medical Cannabis Use Patterns and Adverse Effects in Inflammatory Bowel Disease. J. Clin. Gastroenterol. 2022, 57, 824–829. [Google Scholar] [CrossRef]
- Swaminath, A.; Berlin, E.P.; Cheifetz, A.; Hoffenberg, E.; Kinnucan, J.; Wingate, L.; Buchanan, S.; Zmeter, N.; Rubin, D.T. The Role of Cannabis in the Management of Inflammatory Bowel Disease: A Review of Clinical, Scientific, and Regulatory Information. Inflamm. Bowel Dis. 2019, 25, 427–435. [Google Scholar] [CrossRef]
- Kienzl, M.; Storr, M.; Schicho, R. Cannabinoids and Opioids in the Treatment of Inflammatory Bowel Diseases. Clin. Transl. Gastroenterol. 2020, 11, e00120. [Google Scholar] [CrossRef]
- Hasenoehrl, C.; Storr, M.; Schicho, R. Cannabinoids for Treating Inflammatory Bowel Diseases: Where Are We and Where Do We Go? Expert Rev. Gastroenterol. Hepatol. 2017, 11, 329–337. [Google Scholar] [CrossRef]
- Di Marzo, V.; Piscitelli, F. The Endocannabinoid System and Its Modulation by Phytocannabinoids. Neurotherapeutics 2015, 12, 692–698. [Google Scholar] [CrossRef]
- Ligresti, A.; De Petrocellis, L.; Di Marzo, V. From Phytocannabinoids to Cannabinoid Receptors and Endocannabinoids: Pleiotropic Physiological and Pathological Roles Through Complex Pharmacology. Physiol. Rev. 2016, 96, 1593–1659. [Google Scholar] [CrossRef]
- Naftali, T. An Overview of Cannabis Based Treatment in Crohn’s Disease. Expert Rev. Gastroenterol. Hepatol. 2020, 14, 253–257. [Google Scholar] [CrossRef]
- Giorgi, V.; Marotto, D.; Batticciotto, A.; Atzeni, F.; Bongiovanni, S.; Sarzi-Puttini, P. Cannabis and Autoimmunity: Possible Mechanisms of Action. ImmunoTargets Ther. 2021, 10, 261–271. [Google Scholar] [CrossRef]
- Klein, T.W. Cannabinoid-Based Drugs as Anti-Inflammatory Therapeutics. Nat. Rev. Immunol. 2005, 5, 400–411. [Google Scholar] [CrossRef]
- Naftali, T.; Mechulam, R.; Marii, A.; Gabay, G.; Stein, A.; Bronshtain, M.; Laish, I.; Benjaminov, F.; Konikoff, F.M. Low-Dose Cannabidiol Is Safe but Not Effective in the Treatment for Crohn’s Disease, a Randomized Controlled Trial. Dig. Dis. Sci. 2017, 62, 1615–1620. [Google Scholar] [CrossRef]
- Naftali, T.; Bar-Lev Schleider, L.; Almog, S.; Meiri, D.; Konikoff, F.M. Oral CBD-Rich Cannabis Induces Clinical but Not Endoscopic Response in Patients with Crohn’s Disease, a Randomised Controlled Trial. J. Crohns Colitis 2021, 15, 1799–1806. [Google Scholar] [CrossRef]
- Lahat, A.; Lang, A.; Ben-Horin, S. Impact of Cannabis Treatment on the Quality of Life, Weight and Clinical Disease Activity in Inflammatory Bowel Disease Patients: A Pilot Prospective Study. Digestion 2012, 85, 1–8. [Google Scholar] [CrossRef]
- Dalavaye, N.; Erridge, S.; Nicholas, M.; Pillai, M.; Bapir, L.; Holvey, C.; Coomber, R.; Rucker, J.J.; Hoare, J.; Sodergren, M.H. The Effect of Medical Cannabis in Inflammatory Bowel Disease: Analysis from the UK Medical Cannabis Registry. Expert Rev. Gastroenterol. Hepatol. 2023, 17, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Nduma, B.N.; Mofor, K.A.; Tatang, J.; Ekhator, C.; Ambe, S.; Fonkem, E. The Use of Cannabinoids in the Treatment of Inflammatory Bowel Disease (IBD): A Review of the Literature. Cureus 2023, 15, e36148. [Google Scholar] [CrossRef] [PubMed]
- Hammond, S.; Erridge, S.; Mangal, N.; Pacchetti, B.; Sodergren, M.H. The Effect of Cannabis-Based Medicine in the Treatment of Cachexia: A Systematic Review and Meta-Analysis. Cannabis Cannabinoid Res. 2021, 6, 474–487. [Google Scholar] [CrossRef] [PubMed]
- Häuser, W.; Welsch, P.; Klose, P.; Radbruch, L.; Fitzcharles, M.-A. Efficacy, Tolerability and Safety of Cannabis-Based Medicines for Cancer Pain: A Systematic Review with Meta-Analysis of Randomised Controlled Trials. Schmerz 2019, 33, 424–436. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.; Pek, K.; Chew, J.; Lim, J.P.; Ismail, N.H.; Ding, Y.Y.; Cesari, M.; Lim, W.S. The Simplified Nutritional Appetite Questionnaire (SNAQ) as a Screening Tool for Risk of Malnutrition: Optimal Cutoff, Factor Structure, and Validation in Healthy Community-Dwelling Older Adults. Nutrients 2020, 12, 2885. [Google Scholar] [CrossRef] [PubMed]
- Neufeld, T.; Pfuhlmann, K.; Stock-Schröer, B.; Kairey, L.; Bauer, N.; Häuser, W.; Langhorst, J. Cannabis Use of Patients with Inflammatory Bowel Disease in Germany: A Cross-Sectional Survey. Z. Gastroenterol. 2021, 59, 1068–1077. [Google Scholar] [CrossRef] [PubMed]
- Naftali, T. Cannabis for Inflammatory Bowel Diseases: Should We Follow the Wisdom of the Crowd? Isr. Med. Assoc. J. 2019, 21, 756–758. [Google Scholar]
- Velez-Santiago, A.; Alvarez-Torres, E.; Martinez-Rodriguez, R.; Candal-Rivera, E.; Muniz-Camacho, L.; Ramos-Burgos, L.; Torres, E.A. A Survey of Cannabis Use among Patients with Inflammatory Bowel Disease (IBD). Int. J. Environ. Res. Public Health 2023, 20, 5129. [Google Scholar] [CrossRef]
- Forbes, A.; Escher, J.; Hébuterne, X.; Kłęk, S.; Krznaric, Z.; Schneider, S.; Shamir, R.; Stardelova, K.; Wierdsma, N.; Wiskin, A.E.; et al. ESPEN Guideline: Clinical Nutrition in Inflammatory Bowel Disease. Clin. Nutr. 2017, 36, 321–347. [Google Scholar] [CrossRef]
- Colombel, J.-F.; Shin, A.; Gibson, P.R. Functional Gastrointestinal Symptoms in Patients with Inflammatory Bowel Disease: A Clinical Challenge. Clin. Gastroenterol. Hepatol. 2019, 17, 380–390.e1. [Google Scholar] [CrossRef]
- Carcamo, L.; Miranda, P.; Zúñiga, A.; Alexander, E.; Molina, M.E.; Urrejola, G.; Larach, T.; Miguieles, R.; Bellolio, F. Ileal Pouch-Anal Anastomosis in Ulcerative Colitis: Outcomes, Functional Results, and Quality of Life in Patients with More than 10-Year Follow-Up. Int. J. Color. Dis. 2020, 35, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Fraguas-Sánchez, A.I.; Torres-Suárez, A.I. Medical Use of Cannabinoids. Drugs 2018, 78, 1665–1703. [Google Scholar] [CrossRef] [PubMed]
- Le Strat, Y.; Le Foll, B. Obesity and Cannabis Use: Results from 2 Representative National Surveys. Am. J. Epidemiol. 2011, 174, 929–933. [Google Scholar] [CrossRef] [PubMed]
- Lent, M.R.; Visek, M.; Syracuse, P.; Dugosh, K.L.; Festinger, D.S. Weight Stability in Adults with Obesity Initiating Medical Marijuana Treatment for Other Medical Conditions. J. Cannabis Res. 2022, 4, 48. [Google Scholar] [CrossRef] [PubMed]
- Beulaygue, I.C.; French, M.T. Got Munchies? Estimating the Relationship between Marijuana Use and Body Mass Index. J. Ment. Health Policy Econ. 2016, 19, 123–140. [Google Scholar] [PubMed]
- Alshaarawy, O.; Anthony, J.C. Are Cannabis Users Less Likely to Gain Weight? Results from a National 3-Year Prospective Study. Int. J. Epidemiol. 2019, 48, 1695–1700. [Google Scholar] [CrossRef] [PubMed]
- Bodine, M.; Kemp, A.K. Medical Cannabis Use in Oncology. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Reuter, S.E.; Martin, J.H. Pharmacokinetics of Cannabis in Cancer Cachexia-Anorexia Syndrome. Clin. Pharmacokinet. 2016, 55, 807–812. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, Y.; Tong, M.; Pan, H.; Li, D. Medical Cannabinoids for Cancer Cachexia: A Systematic Review and Meta-Analysis. BioMed Res. Int. 2019, 2019, e2864384. [Google Scholar] [CrossRef]
- Langford, R.M.; Mares, J.; Novotna, A.; Vachova, M.; Novakova, I.; Notcutt, W.; Ratcliffe, S. A Double-Blind, Randomized, Placebo-Controlled, Parallel-Group Study of THC/CBD Oromucosal Spray in Combination with the Existing Treatment Regimen, in the Relief of Central Neuropathic Pain in Patients with Multiple Sclerosis. J. Neurol. 2013, 260, 984–997. [Google Scholar] [CrossRef]
- Bedi, G.; Foltin, R.W.; Gunderson, E.W.; Rabkin, J.; Hart, C.L.; Comer, S.D.; Vosburg, S.K.; Haney, M. Efficacy and Tolerability of High-Dose Dronabinol Maintenance in HIV-Positive Marijuana Smokers: A Controlled Laboratory Study. Psychopharmacology 2010, 212, 675–686. [Google Scholar] [CrossRef]
- Andries, A.; Frystyk, J.; Flyvbjerg, A.; Støving, R.K. Changes in IGF-I, Urinary Free Cortisol and Adipokines during Dronabinol Therapy in Anorexia Nervosa: Results from a Randomised, Controlled Trial. Growth Horm. IGF Res. 2015, 25, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Whiting, P.F.; Wolff, R.F.; Deshpande, S.; Di Nisio, M.; Duffy, S.; Hernandez, A.V.; Keurentjes, J.C.; Lang, S.; Misso, K.; Ryder, S.; et al. Cannabinoids for Medical Use: A Systematic Review and Meta-Analysis. JAMA 2015, 313, 2456–2473. [Google Scholar] [CrossRef] [PubMed]
- Simon, L.; Baldwin, C.; Kalea, A.Z.; Slee, A. Cannabinoid Interventions for Improving Cachexia Outcomes in Cancer: A Systematic Review and Meta-Analysis. J. Cachexia Sarcopenia Muscle 2022, 13, 23–41. [Google Scholar] [CrossRef] [PubMed]
- Zeraatkar, D.; Cooper, M.A.; Agarwal, A.; Vernooij, R.W.M.; Leung, G.; Loniewski, K.; Dookie, J.E.; Ahmed, M.M.; Hong, B.Y.; Hong, C.; et al. Long-Term and Serious Harms of Medical Cannabis and Cannabinoids for Chronic Pain: A Systematic Review of Non-Randomised Studies. BMJ Open 2022, 12, e054282. [Google Scholar] [CrossRef] [PubMed]
- Nugent, S.M.; Morasco, B.J.; O’Neil, M.E.; Freeman, M.; Low, A.; Kondo, K.; Elven, C.; Zakher, B.; Motu’apuaka, M.; Paynter, R.; et al. The Effects of Cannabis among Adults with Chronic Pain and an Overview of General Harms: A Systematic Review. Ann. Intern. Med. 2017, 167, 319–331. [Google Scholar] [CrossRef] [PubMed]
- Harrold, J.A.; Elliott, J.C.; King, P.J.; Widdowson, P.S.; Williams, G. Down-Regulation of Cannabinoid-1 (CB-1) Receptors in Specific Extrahypothalamic Regions of Rats with Dietary Obesity: A Role for Endogenous Cannabinoids in Driving Appetite for Palatable Food? Brain Res. 2002, 952, 232–238. [Google Scholar] [CrossRef]
- Ravinet Trillou, C.; Delgorge, C.; Menet, C.; Arnone, M.; Soubrié, P. CB1 Cannabinoid Receptor Knockout in Mice Leads to Leanness, Resistance to Diet-Induced Obesity and Enhanced Leptin Sensitivity. Int. J. Obes. 2004, 28, 640–648. [Google Scholar] [CrossRef]
- Maykut, M.O. Health Consequences of Acute and Chronic Marihuana Use. Prog. Neuropsychopharmacol. Biol. Psychiatry 1985, 9, 209–238. [Google Scholar] [CrossRef]
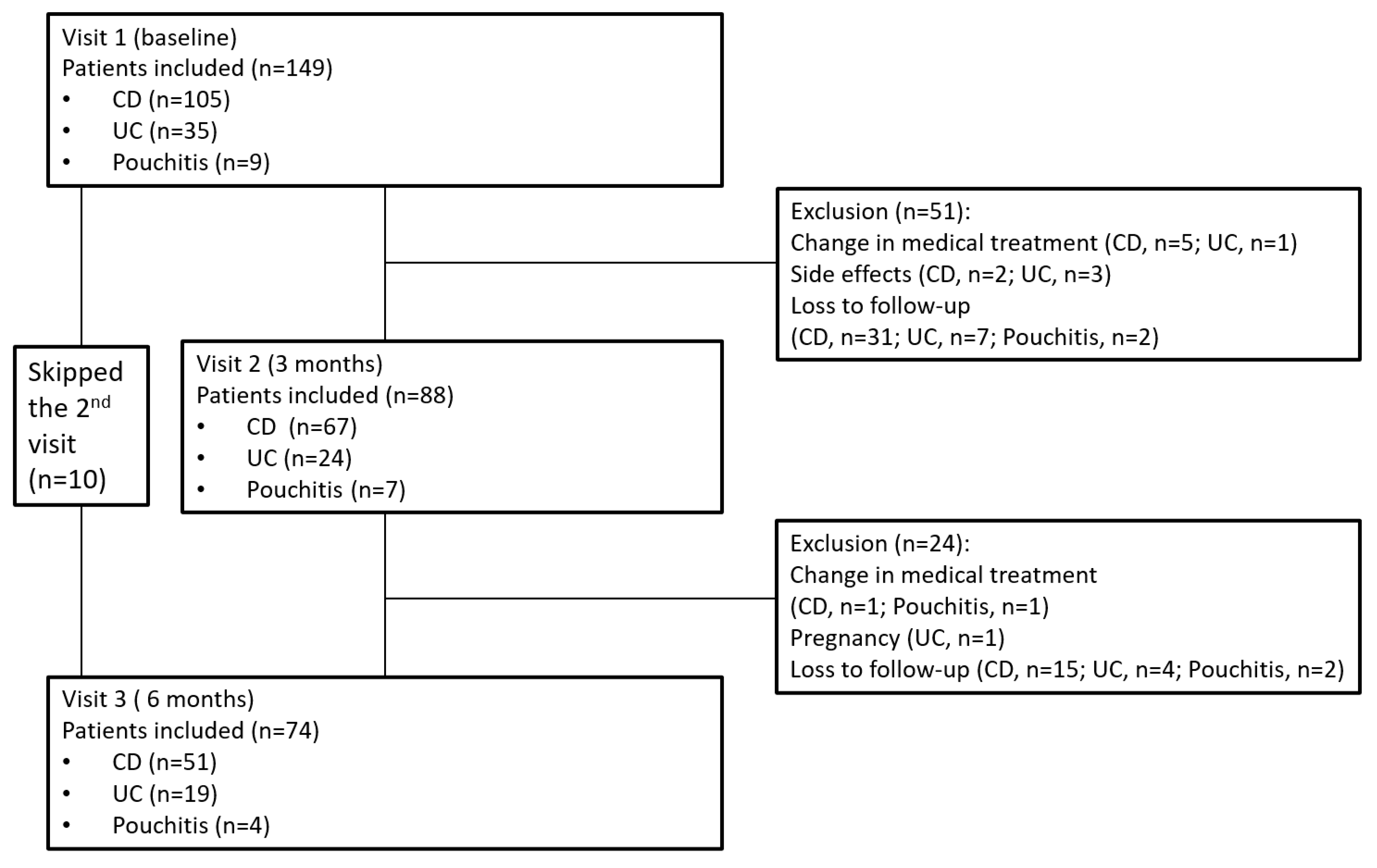
| Total Study Population (N = 149) | MC for Increasing Appetite/Improving Nutritional Status (n = 50) | MC for Other Indications (n = 99) | Pv | |
|---|---|---|---|---|
| Demographic characteristics | ||||
| Age (years, mean ± sd) | 39.0 ± 14.1 | 37.2 ± 14.4 | 40.0 ± 14.0 | 0.255 |
| Gender–female %, (n) | 42.3 (63) | 44.0 (22) | 41.4 (41) | 0.763 |
| Past smoking %, (n) | 28.9 (43) | 22.0 (11) | 32.3 (32) | 0.358 |
| Current smoking %, (n) | 26.2 (39) | 26.0 (13) | 26.3 (26) | |
| Previous Cannabis use %, (n) | 37.6 (56) | 42.0 (21) | 35.4 (35) | 0.541 |
| BMI at baseline (kg/m2, mean ± sd) | 23.0 ± 3.9 | 21.6 ± 3.8 | 23.8 ± 3.8 | 0.002 |
| Underweight (BMI ≤ 18.5 kg/m2) %, (n) | 10.1 (15) | 18.0 (9) | 6.1 (6) | 0.022 |
| Unintentional weight loss %, (n) | 30.9 (46) | 52.0 (26) | 20.2 (20) | <0.001 |
| Percent weight change 3 months prior to enrollment (%, mean ± sd) (n = 81) | −4.8 ± 15.5 | −6.5 ± 15.9 | −3.6 ± 15.3 | 0.409 |
| IBD characteristics | ||||
| Disease type %, (n) | ||||
| CD | 72.5 (108) | 68.0 (34) | 74.7 (74) | 0.006 |
| UC | 21.5 (32) | 20.0 (10) | 22.2 (22) | |
| Pouchitis | 6.0 (9) | 12.0 (6) | 3.0 (3) | |
| Disease duration (years, mean ± sd) | 12.5 ± 10.9 | 12.5 ± 10.5 | 12.5 ± 11.2 | 0.996 |
| Extra-intestinal manifestations %, (n) | 60.4 (90) | 66.0 (33) | 57.6 (57) | 0.609 |
| Active clinical disease %, (n) | 74.5 (111) | 82.0 (41) | 70.7 (70) | 0.213 |
| Biologic therapy experience %, (n) | ||||
| Naive | 18.9 (25) | 8.9 (4) | 24.1 (21) | 0.016 |
| Past therapy | 15.9 (21) | 17.8 (8) | 14.9 (13) | |
| Current therapy | 65.7 (86) | 73.4 (33) | 60.9 (53) | |
| Prescribed cannabis treatment characteristics | ||||
| Treatment dose (gr/month) (mean ± sd) | 23.2 ± 7.4 | 25.7 ± 8.1 | 22.0 ± 6.8 | 0.005 |
| THC dose (gr/month) (mean ± sd) | 1.7 ± 1.3 | 2.09 ± 1.5 | 1.5 ± 1.1 | 0.014 |
| CBD dose (gr/month) (mean ± sd) | 2.4 ± 0.9 | 2.4 ± 0.8 | 2.3 ± 0.9 | 0.495 |
| THC/CBD dose ratio (median, range) | 0.6 (0.03–5.0) | 0.8 (0.03–5.0) | 0.5 (0.03–5.0) | 0.202 |
| Baseline | 3 Months | 6 Months | Pv-Time | |
|---|---|---|---|---|
| BMI (kg/m2, mean(se)) (n = 146) | 23.1 (2.5) | 22.7 (2.5) | 22.9 (2.5) | 0.825 |
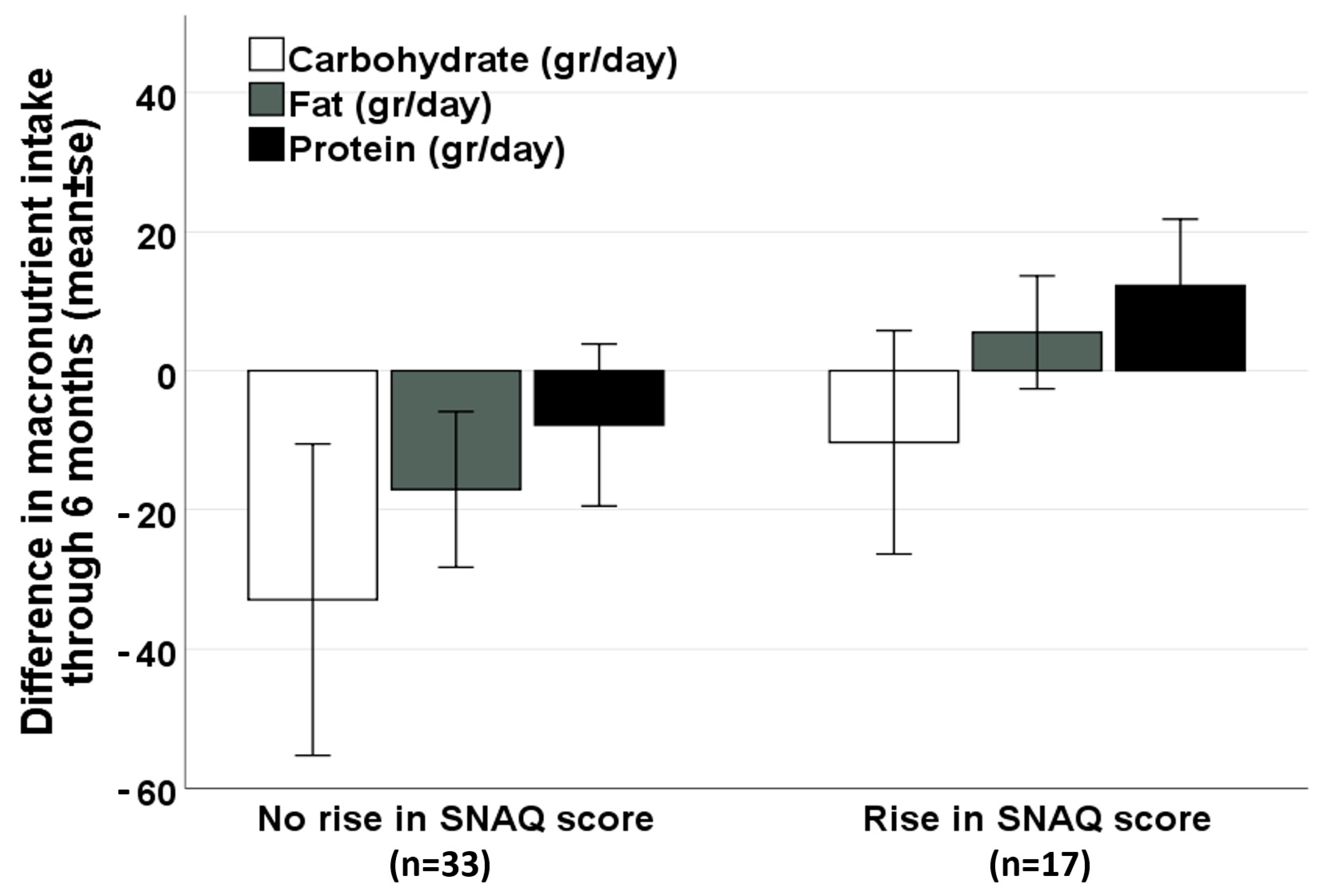
| SNAQ appetite score (mean (se)) (n = 101) | 25.6 (0.4) | 27.2 (0.6) | 26.8 (0.5) | 0.064 |
| Total energy (kcal/day, mean (se)) (n = 60) | 2193 (86) | 2025 (91) | 2068 (99) | 0.627 |
| Protein (% of kcal, mean (se)) (n = 60) | 0.1 (0.006) | 0.1 (0.006) | 0.2 (0.007) | 0.089 |
| Carbohydrates (% of kcal, mean (se)) (n = 60) | 0.4 (0.010) | 0.4 (0.010) | 0.4 (0.011) | 0.808 |
| Fat (% of kcal, mean (se)) (n = 60) | 0.3 (0.007) | 0.3 (0.007) | 0.3 (0.008) | 0.993 |
| SNAQ Appetite Score (Mean (se)) | |||||||
|---|---|---|---|---|---|---|---|
| Baseline | 3 Months | 6 Months | Pv-Time | Pv-Treatment | Pv-Interaction | ||
| MC dose (gr/month) | High dose | 23.1 (1.3) | 25.1 (1.6) | 24.5 (1.4) | 0.297 | 0.026 | 0.950 |
| Low dose | 25.6 (0.8) | 28.2 (1.0) | 26.3 (4.4) | 0.604 | |||
| THC dose (gr/month) | High dose | 24.5 (0.9) | 26.3 (1.1) | 26.3 (1.0) | 0.367 | 0.371 | 0.581 |
| Low dose | 23.8 (1.8) | 26.6 (2.1) | 23.8 (1.8) | 0.425 | |||
| CBD dose (gr/month) | High dose | 23.1 (1.0) | 25.6 (1.2) | 23.8 (1.1) | 0.304 | 0.004 | 0.895 |
| Low dose | 26.1 (1.1) | 28.0 (1.5) | 27.4 (1.1) | 0.601 | |||
| THC/CBD dose ratio | High dose | 25.0 (1.0) | 26.9 (1.2) | 26.8 (1.0) | 0.391 | 0.050 | 0.633 |
| Low dose | 23.4 (1.2) | 25.8 (1.5) | 23.4 (1.3) | 0.422 | |||
| Prior cannabis use | MC Experienced | 25.5 (0.9) | 27.5 (1.2) | 27.9 (1.0) | 0.205 | 0.001 | 0.606 |
| MC naïve | 22.0 (2.1) | 23.4 (2.5) | 19.6 (2.5) | 0.574 | |||
| Total energy (kcal/day, mean (se)) | |||||||
| Baseline | 3 months | 6 months | Pv-time | Pv-treatment | Pv-interaction | ||
| MC dose (gr/month) | High dose | 2065 (509) | 1531 (512) | 2064 (516) | 0.114 | 0.040 | 0.252 |
| Low dose | 2331 (351) | 2561 (376) | 2213 (351) | 0.759 | |||
| THC dose (gr/month) | High dose | 2035 (580) | 1649 (582) | 1877 (580) | 0.388 | <0.001 | 0.381 |
| Low dose | 2503 (378) | 2897 (429) | 2768 (429) | 0.780 | |||
| CBD dose (gr/month) | High dose | 2089 (236) | 1861 (245) | 2135 (279) | 0.715 | 0.312 | 0.772 |
| Low dose | 2349 (332) | 2314 (367) | 2158 (306) | 0.904 | |||
| THC/CBD dose ratio | High dose | 2202 (233) | 1736 (240) | 2048 (233) | 0.376 | 0.082 | 0.268 |
| Low dose | 2206 (344) | 2711 (391) | 2376 (391) | 0.628 | |||
| Prior cannabis use | MC Experienced | 2381 (264) | 2301 (283) | 2180 (264) | 0.951 | 0.029 | 0.734 |
| MC naïve | 1341 (221) | 1048 (221) | 1948 (221) | 0.865 | |||
| BMI (kg/m2, mean (se)) | |||||||
| Baseline | 3 months | 6 months | Pv-time | Pv-treatment | Pv-interaction | ||
| MC dose (gr/month) | High dose | 20.4 (0.4) | 20.5 (0.5) | 20.3 (0.5) | 0.755 | <0.001 | 0.857 |
| Low dose | 22.1 (0.5) | 22.6 (0.6) | 22.5 (0.6) | 0.981 | |||
| THC dose (gr/month) | High dose | 21.1 (2.0) | 21.3 (2.1) | 21.5 (2.1) | 0.833 | 0.548 | 0.867 |
| Low dose | 21.5 (1.0) | 22.0 (1.1) | 21.5 (1.1) | 0.870 | |||
| CBD dose (gr/month) | High dose | 20.7 (2.1) | 21.5 (2.1) | 21.1 (2.1) | 0.988 | 0.139 | 0.819 |
| Low dose | 21.9 (0.5) | 21.8 (0.7) | 21.9 (0.6) | 0.679 | |||
| THC/CBD dose ratio | High dose | 21.4 (0.5) | 21.5 (0.6) | 21.6 (0.6) | 0.790 | 0.830 | 0.933 |
| Low dose | 21.1 (0.5) | 21.7 (0.6) | 21.4 (0.6) | 0.959 | |||
| Prior cannabis use | MC Experienced | 21.1 (0.5) | 21.7 (0.7) | 21.3 (0.6) | 0.975 | 0.922 | 0.970 |
| MC naïve | 21.9 (0.8) | 21.7 (1.0) | 21.6 (1.0) | 0.812 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fliss Isakov, N.; Seidenberg, C.; Meiri, D.; Yackobovitch-Gavan, M.; Maharshak, N.; Hirsch, A. Medical Cannabis Increases Appetite but Not Body Weight in Patients with Inflammatory Bowel Diseases. Nutrients 2024, 16, 78. https://doi.org/10.3390/nu16010078
Fliss Isakov N, Seidenberg C, Meiri D, Yackobovitch-Gavan M, Maharshak N, Hirsch A. Medical Cannabis Increases Appetite but Not Body Weight in Patients with Inflammatory Bowel Diseases. Nutrients. 2024; 16(1):78. https://doi.org/10.3390/nu16010078
Chicago/Turabian StyleFliss Isakov, Naomi, Chen Seidenberg, David Meiri, Michal Yackobovitch-Gavan, Nitsan Maharshak, and Ayal Hirsch. 2024. "Medical Cannabis Increases Appetite but Not Body Weight in Patients with Inflammatory Bowel Diseases" Nutrients 16, no. 1: 78. https://doi.org/10.3390/nu16010078
APA StyleFliss Isakov, N., Seidenberg, C., Meiri, D., Yackobovitch-Gavan, M., Maharshak, N., & Hirsch, A. (2024). Medical Cannabis Increases Appetite but Not Body Weight in Patients with Inflammatory Bowel Diseases. Nutrients, 16(1), 78. https://doi.org/10.3390/nu16010078







