Associations of VEGF-A-Related Variants with Adolescent Cardiometabolic and Dietary Parameters
Abstract
1. Introduction
2. Materials and Methods
2.1. The TEENAGE Study
2.2. Statistical Analyses
3. Results
3.1. Population Characteristics
3.2. Associations between the 11 VEGF-A-Related SNPs and the Cardiometabolic Indices
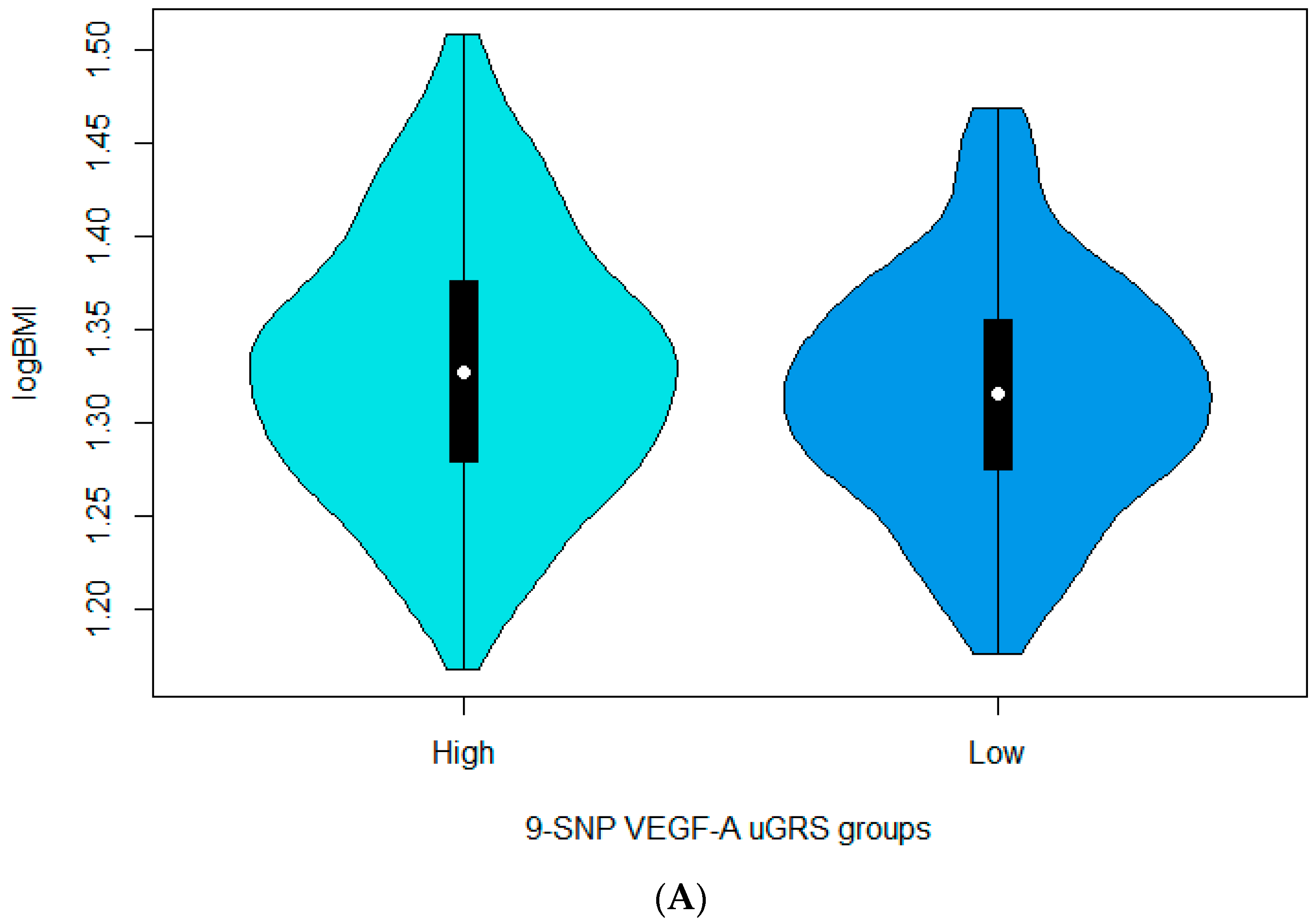
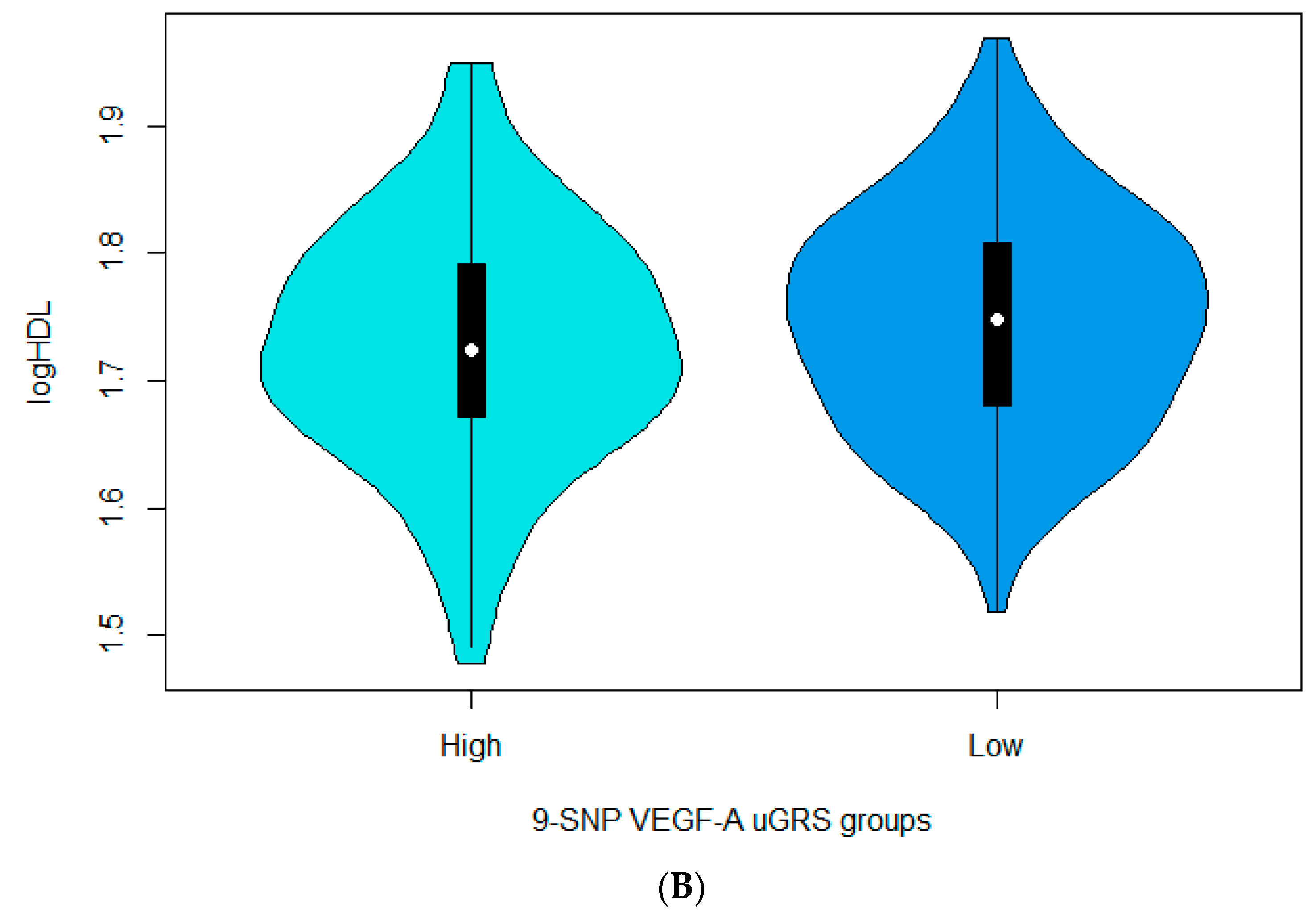
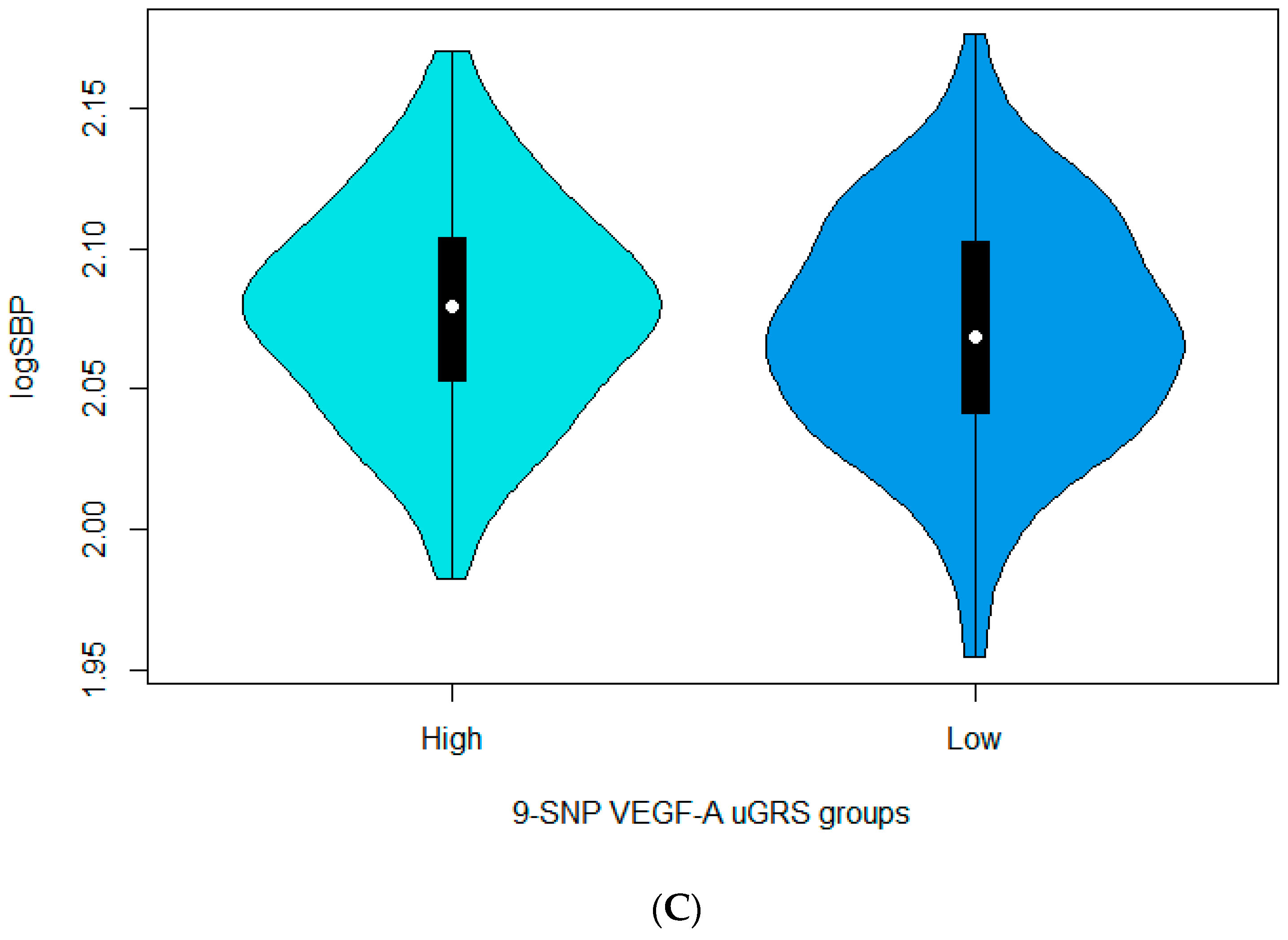
3.3. Associations between the 9-SNP uGRS and the Cardiometabolic Indices
3.4. Interactions between the uGRS and Dietary Patterns
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- di Somma, M.; Vliora, M.; Grillo, E.; Castro, B.; Dakou, E.; Schaafsma, W.; Vanparijs, J.; Corsini, M.; Ravelli, C.; Sakellariou, E.; et al. Role of VEGFs in metabolic disorders. Angiogenesis 2020, 23, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Elias, I.; Franckhauser, S.; Bosch, F. New insights into adipose tissue VEGF-A actions in the control of obesity and insulin resistance. Adipocyte 2013, 2, 109–112. [Google Scholar] [CrossRef]
- Abhinand, C.S.; Raju, R.; Soumya, S.J.; Arya, P.S.; Sudhakaran, P.R. VEGF-A/VEGFR2 signaling network in endothelial cells relevant to angiogenesis. J. Cell Commun. Signal. 2016, 10, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Guangqi, E.; Cao, Y.; Bhattacharya, S.; Dutta, S.; Wang, E.; Mukhopadhyay, D. Endogenous Vascular Endothelial Growth Factor-A (VEGF-A) Maintains Endothelial Cell Homeostasis by Regulating VEGF Receptor-2 Transcription. J. Biol. Chem. 2012, 287, 3029–3041. [Google Scholar] [CrossRef]
- Gennari-Moser, C.; Khankin, E.V.; Escher, G.; Burkhard, F.; Frey, B.M.; Karumanchi, S.A.; Frey, F.J.; Mohaupt, M.G. Vascular endothelial growth factor-A and aldosterone: Relevance to normal pregnancy and preeclampsia. Hypertension 2013, 61, 1111–1117. [Google Scholar] [CrossRef]
- Pi, X.; Xie, L.; Patterson, C. Emerging Roles of Vascular Endothelium in Metabolic Homeostasis. Circ. Res. 2018, 123, 477–494. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhu, X.; Wang, H.; Duan, C.; Cui, H.; Shi, J.; Shi, S.; Yuan, G.; Hu, Y. The Role of VEGF Family in Lipid Metabolism. Curr. Pharm. Biotechnol. 2022, 24, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Staels, W.; Heremans, Y.; Heimberg, H.; De Leu, N. VEGF-A and blood vessels: A beta cell perspective. Diabetologia 2019, 62, 1961–1968. [Google Scholar] [CrossRef]
- Braile, M.; Marcella, S.; Cristinziano, L.; Galdiero, M.R.; Modestino, L.; Ferrara, A.L.; Varricchi, G.; Marone, G.; Loffredo, S. VEGF-A in Cardiomyocytes and Heart Diseases. Int. J. Mol. Sci. 2020, 21, 5294. [Google Scholar] [CrossRef] [PubMed]
- Debette, S.; Visvikis-Siest, S.; Chen, M.-H.; Ndiaye, N.-C.; Song, C.; Destefano, A.; Safa, R.; Nezhad, M.A.; Sawyer, D.; Marteau, J.-B.; et al. Identification of cis- and trans-Acting Genetic Variants Explaining Up to Half the Variation in Circulating Vascular Endothelial Growth Factor Levels. Circ. Res. 2011, 109, 554–563. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Ruggiero, D.; Sorice, R.; Song, C.; Nutile, T.; Smith, A.V.; Concas, M.P.; Traglia, M.; Barbieri, C.; Ndiaye, N.C.; et al. Six Novel Loci Associated with Circulating VEGF Levels Identified by a Meta-analysis of Genome-Wide Association Studies. PLoS Genet. 2016, 12, e1005874. [Google Scholar] [CrossRef]
- Stathopoulou, M.G.; Bonnefond, A.; Ndiaye, N.C.; Azimi-Nezhad, M.; El Shamieh, S.; Saleh, A.; Rancier, M.; Siest, G.; Lamont, J.; Fitzgerald, P.; et al. A common variant highly associated with plasma VEGFA levels also contributes to the variation of both LDL-C and HDL-C. J. Lipid Res. 2013, 54, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Petrelis, A.M.; Stathopoulou, M.G.; Kafyra, M.; Murray, H.; Masson, C.; Lamont, J.; Fitzgerald, P.; Dedoussis, G.; Yen, F.T.; Visvikis-Siest, S. VEGF-A-related genetic variants protect against Alzheimer’s disease. Aging 2022, 14, 2524–2536. [Google Scholar] [CrossRef] [PubMed]
- Salami, A.; El Shamieh, S. Association between SNPs of Circulating Vascular Endothelial Growth Factor Levels, Hypercholesterolemia and Metabolic Syndrome. Medicina 2019, 55, 464. [Google Scholar] [CrossRef]
- Kim, Y.R.; Hong, S.-H. The Protective Effects of the VEGF−2578C>A and −1154G>A Polymorphisms Against Hypertension Susceptibility. Genet. Test. Mol. Biomark. 2015, 19, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Ghazizadeh, H.; Esmaeily, H.; Sharifan, P.; Parizadeh, S.M.R.; Ferns, G.A.; Rastegar-Moghaddam, A.; Khedmatgozar, H.; Ghayour-Mobarhan, M.; Avan, A. Interaction between a genetic variant in vascular endothelial growth factor with dietary intakes in association with the main factors of metabolic syndrome. Gene Rep. 2020, 21, 100813. [Google Scholar] [CrossRef]
- Hoseini, Z.; Azimi-Nezhad, M.; Ghayour-Mobarhan, M.; Avan, A.; Eslami, S.; Nematy, M.; Mirhafez, S.R.; Ghazavi, H.; Ferns, G.A.; Safarian, M. VEGF gene polymorphism interactions with dietary trace elements intake in determining the risk of metabolic syndrome. J. Cell. Biochem. 2018, 120, 1398–1406. [Google Scholar] [CrossRef]
- Chedid, P.; Salami, A.; Ibrahim, M.; Visvikis-Siest, S.; El Shamieh, S. The association of vascular endothelial growth factor related SNPs and circulating iron levels might depend on body mass index. Front. Biosci. 2022, 27, 27. [Google Scholar] [CrossRef]
- Kafyra, M.; Kalafati, I.P.; Kumar, S.; Kontoe, M.S.; Masson, C.; Siest, S.; Dedoussis, G.V. Dietary Patterns, Blood Pressure and the Glycemic and Lipidemic Profile of Two Teenage, European Populations. Nutrients 2021, 13, 198. [Google Scholar] [CrossRef]
- Ntalla, I.; Panoutsopoulou, K.; Vlachou, P.; Southam, L.; Rayner, N.W.; Zeggini, E.; Dedoussis, G.V. Replication of Established Common Genetic Variants for Adult BMI and Childhood Obesity in Greek Adolescents: The TEENAGE Study. Ann. Hum. Genet. 2013, 77, 268–274. [Google Scholar] [CrossRef]
- Ntalla, I.; Yannakoulia, M.; Dedoussis, G.V. An Overweight Preventive Score associates with obesity and glycemic traits. Metabolism 2016, 65, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Ntalla, I.; Giannakopoulou, M.; Vlachou, P.; Giannitsopoulou, K.; Gkesou, V.; Makridi, C.; Marougka, M.; Mikou, G.; Ntaoutidou, K.; Prountzou, E.; et al. Body composition and eating behaviours in relation to dieting involvement in a sample of urban Greek adolescents from the TEENAGE (TEENs of Attica: Genes & Environment) study. Public Health Nutr. 2014, 17, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Yannakoulia, M.; Ntalla, I.; Papoutsakis, C.; Farmaki, A.-E.; Dedoussis, G.V. Consumption of Vegetables, Cooked Meals, and Eating Dinner is Negatively Associated with Overweight Status in Children. J. Pediatr. 2010, 157, 815–820. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, S.; Das, S.; Kretzschmar, W.; Delaneau, O.; Wood, A.R.; Teumer, A.; Kang, H.M.; Fuchsberger, C.; Danecek, P.; Sharp, K.; et al. A reference panel of 64,976 haplotypes for genotype imputation. Nat. Genet. 2016, 48, 1279–1283. [Google Scholar] [CrossRef]
- Ihaka, R.; Gentleman, R. R: A Language for Data Analysis and Graphics. J. Comput. Graph. Stat. 1996, 5, 299–314. [Google Scholar] [CrossRef]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.R.; Bender, D.; Maller, J.; Sklar, P.; de Bakker, P.I.W.; Daly, M.J.; et al. PLINK: A Tool Set for Whole-Genome Association and Population-Based Linkage Analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef]
- Yeung, S.L.A.; Lam, H.S.H.S.; Schooling, C.M. Vascular Endothelial Growth Factor and Ischemic Heart Disease Risk: A Mendelian Randomization Study. J. Am. Heart Assoc. 2017, 6, e005619. [Google Scholar] [CrossRef] [PubMed]
- Keller-Baruch, J.; Forgetta, V.; Manousaki, D.; Zhou, S.; Richards, J.B. Genetically Decreased Circulating Vascular Endothelial Growth Factor and Osteoporosis Outcomes: A Mendelian Randomization Study. J. Bone Miner. Res. 2020, 35, 649–656. [Google Scholar] [CrossRef]
- Robinson, E.S.; Khankin, E.V.; Karumanchi, S.A.; Humphreys, B.D. Hypertension Induced by Vascular Endothelial Growth Factor Signaling Pathway Inhibition: Mechanisms and Potential Use as a Biomarker. Semin. Nephrol. 2010, 30, 591–601. [Google Scholar] [CrossRef]
- Pandey, A.K.; Singhi, E.K.; Arroyo, J.P.; Ikizler, T.A.; Gould, E.R.; Brown, J.; Beckman, J.A.; Harrison, D.G.; Moslehi, J. Mechanisms of VEGF (Vascular Endothelial Growth Factor) Inhibitor–Associated Hypertension and Vascular Disease. Hypertension 2018, 71, e1–e8. [Google Scholar] [CrossRef]
- Mäki-Petäjä, K.M.; McGeoch, A.; Yang, L.L.; Hubsch, A.; McEniery, C.M.; Meyer, P.A.; Mir, F.; Gajendragadkar, P.; Ramenatte, N.; Anandappa, G.; et al. Mechanisms Underlying Vascular Endothelial Growth Factor Receptor Inhibition-Induced Hypertension: The HYPAZ Trial. Hypertension 2021, 77, 1591–1599. [Google Scholar] [CrossRef]
- Zorena, K.; Myśliwska, J.; Myśliwiec, M.; Rybarczyk-Kapturska, K.; Malinowska, E.; Wiśniewski, P.; Raczyńska, K. Association between vascular endothelial growth factor and hypertension in children and adolescents type I diabetes mellitus. J. Hum. Hypertens. 2010, 24, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Zafar, M.I.; Mills, K.; Ye, X.; Blakely, B.; Min, J.; Kong, W.; Zhang, N.; Gou, L.; Regmi, A.; Hu, S.Q.; et al. Association between the expression of vascular endothelial growth factors and metabolic syndrome or its components: A systematic review and meta-analysis. Diabetol. Metab. Syndr. 2018, 10, 62. [Google Scholar] [CrossRef] [PubMed]
- Mazidi, M.; Rezaie, P.; Kengne, A.; Stathopoulou, M.G.; Azimi-Nezhad, M.; Siest, S. VEGF, the underlying factor for metabolic syndrome; fact or fiction? Diabetes Metab. Syndr. 2017, 11 (Suppl. S1), S61–S64. [Google Scholar] [CrossRef]
- Herold, J.; Kalucka, J. Angiogenesis in Adipose Tissue: The Interplay Between Adipose and Endothelial Cells. Front. Physiol. 2021, 11, 624903. [Google Scholar] [CrossRef]
- Sun, K.; Asterholm, I.W.; Kusminski, C.M.; Bueno, A.C.; Wang, Z.V.; Pollard, J.W.; Brekken, R.A.; Scherer, P.E. Dichotomous effects of VEGF-A on adipose tissue dysfunction. Proc. Natl. Acad. Sci. USA 2012, 109, 5874–5879. [Google Scholar] [CrossRef]
- Novikova, V.; Gritsinskaya, V.; Petrenko, Y.V.; Gurova, M.; Gurina, O.; Varlamova, O.; Blinov, A.; Strukov, E.; Smirnova, N.; Kuprienko, N.; et al. Level of erythropoietin, sVCAM-1 and VEGF in blood of obese adolescents. Abstracts 2021, 106, A87–A88. [Google Scholar] [CrossRef]
- Loebig, M.; Klement, J.; Schmoller, A.; Betz, S.; Heuck, N.; Schweiger, U.; Peters, A.; Schultes, B.; Oltmanns, K.M. Evidence for a Relationship between VEGF and BMI Independent of Insulin Sensitivity by Glucose Clamp Procedure in a Homogenous Group Healthy Young Men. PLoS ONE 2010, 5, e12610. [Google Scholar] [CrossRef]
- Guzmán-Guzmán, I.P.; Zaragoza-García, O.; Vences-Velázquez, A.; Castro-Alarcón, N.; Muñoz-Valle, J.F.; Parra-Rojas, I. Concentraciones circulantes de MCP-1, VEGF-A, sICAM-1, sVCAM-1, sE-selectina y sVE-cadherina: Su relación con componentes del síndrome metabólico en población joven [Circulating levels of MCP-1, VEGF-A, sICAM-1, sVCAM-1, sE-selectin and sVE-cadherin: Relationship with components of metabolic syndrome in young population]. Med. Clin. 2016, 147, 427–434. [Google Scholar] [CrossRef]
- Dabravolski, S.A.; Khotina, V.A.; Omelchenko, A.V.; Kalmykov, V.A.; Orekhov, A.N. The Role of the VEGF Family in Atherosclerosis Development and Its Potential as Treatment Targets. Int. J. Mol. Sci. 2022, 23, 931. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Schwedhelm, C.; Hoffmann, G.; Knüppel, S.; Iqbal, K.; Andriolo, V.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food Groups and Risk of Hypertension: A Systematic Review and Dose-Response Meta-Analysis of Prospective Studies. Adv. Nutr. Int. Rev. J. 2017, 8, 793–803, Correction in Adv Nutr. 2018, 9, 163–164. [Google Scholar] [CrossRef] [PubMed]
- Hojhabrimanesh, A.; Akhlaghi, M.; Rahmani, E.; Amanat, S.; Atefi, M.; Najafi, M.; Hashemzadeh, M.; Salehi, S.; Faghih, S.; Akhlaghi, M. A Western dietary pattern is associated with higher blood pressure in Iranian adolescents. Eur. J. Nutr. 2017, 56, 399–408. [Google Scholar] [CrossRef]
- Shokri, A.; Pirouzpanah, S.; Foroutan-Ghaznavi, M.; Montazeri, V.; Fakhrjou, A.; Nozad-Charoudeh, H.; Tavoosidana, G. Dietary protein sources and tumoral overexpression of RhoA, VEGF-A and VEGFR2 genes among breast cancer patients. Genes Nutr. 2019, 14, 22. [Google Scholar] [CrossRef] [PubMed]
- Chiarelli, F.; Spagnoli, A.; Basciani, F.; Tumini, S.; Mezzetti, A.; Cipollone, F.; Cuccurullo, F.; Morgese, G.; Verrotti, A. Vascular endothelial growth factor (VEGF) in children, adolescents and young adults with Type 1 diabetes mellitus: Relation to glycaemic control and microvascular complications. Diabet. Med. 2000, 17, 650–656. [Google Scholar] [CrossRef]
- Swann, O.G.; Breslin, M.; Kilpatrick, M.; O’Sullivan, T.A.; Mori, T.A.; Beilin, L.J.; Oddy, W.H. Dietary fibre intake and its association with inflammatory markers in adolescents. Br. J. Nutr. 2021, 125, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.-W.; Lee, Y.-C.; Kuo, C.-S.; Chiang, C.-H.; Chang, H.-H.; Huang, K.-C. Association of Serum Levels of Zinc, Copper, and Iron with Risk of Metabolic Syndrome. Nutrients 2021, 13, 548. [Google Scholar] [CrossRef]
- Wong, M.M.H.; Chan, K.Y.; Lo, K. Manganese Exposure and Metabolic Syndrome: A Systematic Review and Meta-Analysis. Nutrients 2022, 14, 825. [Google Scholar] [CrossRef]
- Ma, J.; Zhou, Y.; Wang, D.; Guo, Y.; Wang, B.; Xu, Y.; Chen, W. Associations between essential metals exposure and metabolic syndrome (MetS): Exploring the mediating role of systemic inflammation in a general Chinese population. Environ. Int. 2020, 140, 105802. [Google Scholar] [CrossRef]
| Consortial Summary Statistics | TEENAGE Cohort | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| SNP | Gene | Chr | Position | Alleles | MAF | Effect Allele | Direction of Effect for VEGF | EAF | Ref. |
| rs114694170 | MEF2C, MEF2C-AS1 | 5 | 5:88884379 | T/C | 0.02 (C) | T | Negative (beta = −0.15) | 0.96 | [6] |
| rs6921438 | SCIRT, LOC100132354 | 6 | 6:43957870 | G/A/C | 0.44 (A) | A | Negative (beta = −0.72) | 0.39 | [6,7] |
| rs1740073 | LINC02537, SCIRT, C6orf223 | 6 | 6:43979661 | T/A/C | 0.20 (T) | T | Positive (beta = 0.09) | 0.35 | [6] |
| rs4416670 | SCIRT | 6 | 6:43982716 | T/A/C | 0.47 (C) | C | Negative (beta = −0.13) | 0.44 | [7] |
| rs6993770 | ZFPM2-AS1,ZFPM2 | 8 | 8:105569300 | A/T | 0.36 (T) | T | Negative (beta = 0.17) | 0.31 | [6,7] |
| rs7043199 | VLDLR-AS1 | 9 | 9:2621145 | T/A | 0.11 (A) | A | Negative (beta = −0.10) | 0.19 | [6] |
| rs10738760 | VLDLR, KCNV2 | 9 | 9:2691186 | A/G | 0.41 (G) | G | Negative (beta = −0.28) | 0.46 | [7] |
| rs2375981 | VLDLR, KCNV2 | 9 | 9:2692583 | C/A/G/T | 0.41 (G) | C | Positive (beta = 0.21) | 0.44 | [6] |
| rs74506613/proxy rs10761741 used | JMJD1C | 10 | 10:63306426 | G/T | 0.37 (T) | T | Positive (beta = 0.08) | 0.47 | [6] |
| rs4782371 | ZFPM1 | 16 | 16:88502423 | T/A/C/G | 0.41 (G) | T | Negative (beta = −0.07) | 0.36 | [6] |
| rs2639990 | ZADH2 | 18 | 18:75203596 | T/C | 0.10 (C) | T | Positive (beta = 0.11) | 0.10 | [6] |
| All | Boys | Girls | |||||
|---|---|---|---|---|---|---|---|
| n | Median (IQR) | n | Median (IQR) | n | Median (IQR) | p-Value * | |
| Age (years) | 766 | 13.30 (1.31) | 349 | 13.36 (1.38) | 417 | 13.26 (1.25) | <0.001 |
| BMI (kg/m2) | 766 | 20.88 (4.38) | 349 | 20.85 (4.45) | 417 | 20.93 (4.37) | 0.517 |
| Triglycerides (mg/dL) | 611 | 56.00 (24) | 283 | 55.00 (25) | 328 | 57.00 (24) | 0.090 |
| Total Cholesterol (mg/dL) | 611 | 157.00 (33) | 283 | 156.49 (25.18) ** | 328 | 157.50 (31) | 0.210 |
| SBP (mmHg) | 743 | 119.00 (16) | 335 | 120.67 (11.93) ** | 408 | 118.00 (15) | 0.001 |
| DBP (mmHg) | 743 | 70.00 (12) | 335 | 71.00 (12) | 408 | 70.00 (12) | 0.825 |
| PP | 743 | 47.00 (13) | 335 | 49.23 (10.61) ** | 408 | 46 (12) | <0.001 |
| LDL (mg/dL) | 611 | 54.00 (16) | 283 | 90.57 (21.78) ** | 328 | 88.40 (26) | 0.651 |
| HDL (mg/dL) | 611 | 89.20 (27) | 283 | 53.00 (16) | 328 | 56.00 (17) | 0.001 |
| Glucose (mg/dL), | 611 | 80.00 (12) | 283 | 81.00 (11) | 328 | 79.00 (12) | <0.05 |
| CRP (mg/dL) | 540 | 0.30 (1) | 254 | 0.45 (1) | 286 | 0.20 (0) | <0.001 |
| Model 1 | Model 2 | Model 3 | ||||
|---|---|---|---|---|---|---|
| Beta | p-Value | Beta | p-Value | Beta | p-Value | |
| LogBMI | ||||||
| rs114694170 | 0.01009 | 0.3424 | 0.01317 | 0.2385 | 0.01239 | 0.2707 |
| rs6921438 | −0.00631 | 0.1131 | −0.0053 | 0.2038 | −0.00475 | 0.2564 |
| rs1740073 | 0.005531 | 0.1785 | 0.003664 | 0.3826 | 0.002784 | 0.5088 |
| rs4416670 | −0.00698 | 0.06125 | −0.00389 | 0.3099 | −0.00363 | 0.3452 |
| rs6993770 | −0.00649 | 0.1252 | −0.00866 | 0.04606 | −0.00858 | 0.0483 |
| rs7043199 | −0.01265 | 0.01352 | −0.01202 | 0.02304 | −0.01185 | 0.02551 |
| rs10738760 | 0.003147 | 0.4208 | 0.002341 | 0.5588 | 0.00203 | 0.6125 |
| rs2375981 | 0.003426 | 0.3883 | 0.002837 | 0.4846 | 0.002472 | 0.5432 |
| rs10761741 | 0.003055 | 0.4467 | 0.003455 | 0.3978 | 0.003062 | 0.4544 |
| rs4782371 | 0.00442 | 0.2833 | 0.003158 | 0.4576 | 0.002953 | 0.4892 |
| rs2639990 | −0.00297 | 0.6463 | −0.00232 | 0.7241 | −0.0021 | 0.7516 |
| logTriglycerides | ||||||
| rs114694170 | 0.008907 | 0.7274 | 0.02828 | 0.2978 | 0.029 | 0.292 |
| rs6921438 | 0.001028 | 0.9184 | 0.01319 | 0.2007 | 0.01328 | 0.2003 |
| rs1740073 | 0.006261 | 0.5473 | 0.002573 | 0.8058 | 0.00253 | 0.8107 |
| rs4416670 | 1.83 × 10−5 | 0.9984 | 0.00513 | 0.5827 | 0.004898 | 0.6018 |
| rs6993770 | 0.006058 | 0.5595 | −0.00307 | 0.7726 | −0.00332 | 0.7567 |
| rs7043199 | −0.01681 | 0.1822 | −0.01787 | 0.1588 | −0.01938 | 0.1304 |
| rs10738760 | −0.02382 | 0.01482 | −0.0201 | 0.04157 | −0.0201 | 0.04306 |
| rs2375981 | −0.01995 | 0.04558 | −0.01675 | 0.09515 | −0.01696 | 0.09375 |
| rs10761741 | 0.004158 | 0.6738 | −0.00254 | 0.7989 | −0.00198 | 0.844 |
| rs4782371 | −0.00071 | 0.9448 | 0.00189 | 0.8571 | 0.001944 | 0.8546 |
| rs2639990 | −0.01428 | 0.3776 | −0.01309 | 0.4196 | −0.0138 | 0.4033 |
| logCholesterol | ||||||
| rs114694170 | −0.00314 | 0.7859 | −0.00783 | 0.5438 | −0.00896 | 0.4916 |
| rs6921438 | −0.00051 | 0.9111 | 0.000254 | 0.9586 | −9.61 × 10−5 | 0.9844 |
| rs1740073 | 0.000767 | 0.8706 | 0.000225 | 0.9639 | −0.00033 | 0.947 |
| rs4416670 | 0.001849 | 0.6564 | 0.004052 | 0.3602 | 0.004303 | 0.3322 |
| rs6993770 | 0.0042 | 0.3709 | 0.002885 | 0.567 | 0.002729 | 0.5901 |
| rs7043199 | −0.00066 | 0.908 | −9.11 × 10−5 | 0.9879 | −0.00107 | 0.8596 |
| rs10738760 | −0.00256 | 0.5642 | −0.00355 | 0.4489 | −0.00351 | 0.4558 |
| rs2375981 | −0.00357 | 0.4299 | −0.00446 | 0.3497 | −0.00424 | 0.3768 |
| rs10761741 | −0.00642 | 0.1503 | −0.00856 | 0.0695 | −0.0087 | 0.06685 |
| rs4782371 | 0.003328 | 0.4736 | 0.001601 | 0.7478 | 0.002173 | 0.6649 |
| rs2639990 | −0.00337 | 0.645 | −0.00521 | 0.4986 | −0.00315 | 0.6864 |
| logSBP | ||||||
| rs114694170 | 0.004856 | 0.4602 | 0.01095 | 0.1322 | 0.01002 | 0.1704 |
| rs6921438 | −0.00528 | 0.03273 | −0.00571 | 0.03214 | −0.00614 | 0.02126 |
| rs1740073 | 0.006211 | 0.01456 | 0.007036 | 0.008435 | 0.007113 | 0.007929 |
| rs4416670 | −0.00707 | 0.002172 | −0.00744 | 0.002407 | −0.00716 | 0.003524 |
| rs6993770 | −0.005 | 0.05437 | −0.00489 | 0.07711 | −0.005 | 0.07093 |
| rs7043199 | 0.007357 | 0.02104 | 0.009594 | 0.004338 | 0.009446 | 0.005093 |
| rs10738760 | −0.00105 | 0.6643 | −0.00018 | 0.9445 | −0.0002 | 0.9368 |
| rs2375981 | −0.00048 | 0.8464 | 0.000475 | 0.8549 | 0.000676 | 0.7948 |
| rs10761741 | 0.004394 | 0.07559 | 0.003574 | 0.1711 | 0.003634 | 0.1643 |
| rs4782371 | −0.0017 | 0.5082 | −0.00148 | 0.5885 | −0.00099 | 0.7192 |
| rs2639990 | −0.00027 | 0.9467 | −0.00181 | 0.6667 | −0.00112 | 0.7913 |
| logDBP | ||||||
| rs114694170 | −0.00538 | 0.5747 | −0.00023 | 0.9829 | −0.00073 | 0.945 |
| rs6921438 | −0.00617 | 0.08685 | −0.00804 | 0.03627 | −0.00845 | 0.0283 |
| rs1740073 | 0.005599 | 0.1311 | 0.006755 | 0.07975 | 0.006983 | 0.07167 |
| rs4416670 | −0.00556 | 0.09872 | −0.00686 | 0.05272 | −0.00661 | 0.06318 |
| rs6993770 | −0.00621 | 0.101 | −0.0043 | 0.281 | −0.00443 | 0.2685 |
| rs7043199 | 0.01191 | 0.01033 | 0.01359 | 0.005051 | 0.0138 | 0.004611 |
| rs10738760 | 6.32 × 10−6 | 0.9986 | 0.001639 | 0.6575 | 0.001642 | 0.6579 |
| rs2375981 | −0.00022 | 0.9508 | 0.001781 | 0.6339 | 0.002048 | 0.5851 |
| rs10761741 | 0.005385 | 0.135 | 0.006435 | 0.08701 | 0.006501 | 0.0848 |
| rs4782371 | 0.000505 | 0.8928 | 0.002055 | 0.6027 | 0.002789 | 0.4824 |
| rs2639990 | 0.004213 | 0.4671 | 0.003025 | 0.6163 | 0.003598 | 0.5553 |
| logPP | ||||||
| rs114694170 | 0.02169 | 0.1799 | 0.03011 | 0.0877 | 0.02892 | 0.1044 |
| rs6921438 | −0.00429 | 0.4814 | −0.00136 | 0.8342 | −0.00166 | 0.7989 |
| rs1740073 | 0.008354 | 0.1826 | 0.008206 | 0.2063 | 0.007979 | 0.223 |
| rs4416670 | −0.01232 | 0.03026 | −0.01075 | 0.07144 | −0.0104 | 0.08316 |
| rs6993770 | −0.0003 | 0.9623 | −0.00313 | 0.6417 | −0.0031 | 0.6466 |
| rs7043199 | −0.00119 | 0.8798 | 0.002393 | 0.77 | 0.001466 | 0.859 |
| rs10738760 | −0.0021 | 0.7244 | −0.00156 | 0.8026 | −0.00142 | 0.8201 |
| rs2375981 | −0.00033 | 0.9559 | −0.00017 | 0.9786 | 9.90 × 10−5 | 0.9875 |
| rs10761741 | 0.005041 | 0.4081 | 0.000931 | 0.8832 | 0.000839 | 0.8954 |
| rs4782371 | −0.00663 | 0.2943 | −0.00846 | 0.2027 | −0.00844 | 0.2076 |
| rs2639990 | −0.00571 | 0.5596 | −0.00865 | 0.3943 | −0.00733 | 0.4765 |
| logGlucose | ||||||
| rs114694170 | 0.01915 | 0.4259 | 0.01844 | 0.488 | 0.01499 | 0.5762 |
| rs6921438 | −0.00684 | 0.4689 | −0.01078 | 0.2855 | −0.01227 | 0.2245 |
| rs1740073 | 0.00942 | 0.3361 | 0.007099 | 0.4879 | 0.006708 | 0.5143 |
| rs4416670 | 0.000832 | 0.9235 | 0.000346 | 0.9698 | 0.000223 | 0.9806 |
| rs6993770 | −0.01043 | 0.2856 | −0.00569 | 0.5839 | −0.00679 | 0.5148 |
| rs7043199 | 0.008424 | 0.4782 | 0.008428 | 0.4973 | 0.006293 | 0.6144 |
| rs10738760 | 0.006866 | 0.457 | 0.003822 | 0.6927 | 0.002642 | 0.7852 |
| rs2375981 | 0.007188 | 0.445 | 0.004344 | 0.6588 | 0.003512 | 0.722 |
| rs10761741 | 0.003465 | 0.7095 | 0.004664 | 0.6322 | 0.006317 | 0.5187 |
| rs4782371 | −0.01497 | 0.1213 | −0.00968 | 0.3456 | −0.00954 | 0.3557 |
| rs2639990 | −0.00127 | 0.9336 | −0.0042 | 0.7913 | −0.00359 | 0.8233 |
| logLDL | ||||||
| rs114694170 | −0.0082 | 0.6443 | −0.02002 | 0.3046 | −0.02187 | 0.2661 |
| rs6921438 | −0.00502 | 0.4711 | −0.00418 | 0.573 | −0.00419 | 0.5718 |
| rs1740073 | 0.000988 | 0.8914 | −0.00091 | 0.9035 | −0.0022 | 0.7704 |
| rs4416670 | 0.001987 | 0.7558 | 0.006226 | 0.3529 | 0.006893 | 0.3039 |
| rs6993770 | −0.00281 | 0.6968 | −0.00581 | 0.4461 | −0.00551 | 0.4718 |
| rs7043199 | 0.006725 | 0.4431 | 0.006013 | 0.5094 | 0.005337 | 0.5605 |
| rs10738760 | −0.01029 | 0.1306 | −0.01186 | 0.09438 | −0.01145 | 0.1071 |
| rs2375981 | −0.01274 | 0.06626 | −0.01425 | 0.04787 | −0.01372 | 0.05769 |
| rs10761741 | −0.00519 | 0.4493 | −0.00794 | 0.2667 | −0.0091 | 0.2047 |
| rs4782371 | 0.01135 | 0.1115 | 0.007783 | 0.3015 | 0.008257 | 0.2758 |
| rs2639990 | −0.00388 | 0.7136 | −0.00713 | 0.517 | −0.00744 | 0.5042 |
| logHDL | ||||||
| rs114694170 | 0.001151 | 0.9449 | −0.00031 | 0.9867 | −0.00111 | 0.9524 |
| rs6921438 | 0.002231 | 0.7332 | 0.00056 | 0.9363 | −0.00014 | 0.9837 |
| rs1740073 | 0.002099 | 0.7572 | 0.005597 | 0.4303 | 0.00606 | 0.3951 |
| rs4416670 | 0.002402 | 0.6887 | 0.000127 | 0.984 | −0.00021 | 0.9737 |
| rs6993770 | 0.01151 | 0.08893 | 0.0148 | 0.03953 | 0.01427 | 0.04781 |
| rs7043199 | −0.00711 | 0.3875 | −0.00429 | 0.6186 | −0.00585 | 0.4992 |
| rs10738760 | 0.01409 | 0.02729 | 0.01249 | 0.06206 | 0.01223 | 0.06815 |
| rs2375981 | 0.01261 | 0.05275 | 0.01139 | 0.09454 | 0.01129 | 0.09822 |
| rs10761741 | −0.01029 | 0.1098 | −0.01098 | 0.1037 | −0.00975 | 0.15 |
| rs4782371 | −0.00762 | 0.2552 | −0.0072 | 0.3117 | −0.0068 | 0.3417 |
| rs2639990 | −0.00388 | 0.7136 | −0.00713 | 0.517 | −0.00744 | 0.5042 |
| logCRP | ||||||
| rs114694170 | −0.0379 | 0.6541 | −0.04237 | 0.6554 | −0.03521 | 0.711 |
| rs6921438 | −0.0418 | 0.1947 | −0.04414 | 0.2017 | −0.04039 | 0.241 |
| rs1740073 | −0.00433 | 0.8972 | −0.0181 | 0.606 | −0.02466 | 0.482 |
| rs4416670 | −0.0194 | 0.511 | −0.01528 | 0.6242 | −0.0162 | 0.6012 |
| rs6993770 | −0.01718 | 0.6107 | −0.00339 | 0.9251 | −0.0048 | 0.8941 |
| rs7043199 | 0.02666 | 0.5029 | 0.003378 | 0.9353 | 0.000455 | 0.9913 |
| rs10738760 | 0.02319 | 0.4658 | 0.02242 | 0.5016 | 0.02371 | 0.4762 |
| rs2375981 | 0.02867 | 0.3747 | 0.02603 | 0.441 | 0.02572 | 0.4462 |
| rs10761741 | 0.0237 | 0.4588 | 0.01415 | 0.6735 | 0.01207 | 0.7179 |
| rs4782371 | −0.04092 | 0.2165 | −0.03658 | 0.3002 | −0.03689 | 0.2958 |
| rs2639990 | −0.05523 | 0.2803 | −0.05647 | 0.2884 | −0.05193 | 0.3325 |
| Model 1 | Model 2 | Model 3 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Estimate | SE | p-Value | Estimate | SE | p-Value | Estimate | SE | p-Value | |
| logBMI | |||||||||
| 9-SNP uGRS for VEGF-A | 0.004445 | 0.001494 | 0.00305 | 0.004349 | 0.001553 | 0.005277 | 0.0040937 | 0.0015678 | 0.009281 |
| logTriglycerides | |||||||||
| 9-SNP uGRS for VEGF-A | 0.005892 | 0.003854 | 0.127 | 0.004260 | 0.003915 | 0.2771 | 0.004650 | 0.003994 | 0.2450 |
| logCholesterol | |||||||||
| 9-SNP uGRS for VEGF-A | −0.0001979 | 0.0017479 | 0.90992 | −0.000716 | 0.001859 | 0.70024 | −0.0007685 | 0.0018917 | 0.68474 |
| logSBP | |||||||||
| 9-SNP uGRS for VEGF-A | 0.002006 | 0.000924 | 0.0303 | 0.0019840 | 0.0009974 | 0.047203 | 0.0020983 | 0.0010045 | 0.037205 |
| logDBP | |||||||||
| 9-SNP uGRS for VEGF-A | 0.001891 | 0.001351 | 0.161963 | 0.002211 | 0.001441 | 0.12569 | 0.002365 | 0.001455 | 0.10458 |
| LogPP | |||||||||
| 9-SNP uGRS for VEGF-A | 0.002425 | 0.002268 | 0.2854 | 0.001599 | 0.002413 | 0.50779 | 0.0015523 | 0.0024439 | 0.52558 |
| LogGlucose | |||||||||
| 9-SNP uGRS for VEGF-A | 0.0009057 | 0.0036448 | 0.804 | 0.001952 | 0.003840 | 0.611 | 0.0028415 | 0.0038989 | 0.4665 |
| logLDL | |||||||||
| 9-SNP uGRS for VEGF-A | 0.003038 | 0.002688 | 0.2589 | 0.002300 | 0.002818 | 0.4148 | 0.001733 | 0.002863 | 0.5454 |
| LogHDL | |||||||||
| 9-SNP uGRS for VEGF-A | −0.005336 | 0.002493 | 0.03279 | −0.004999 | 0.002631 | 0.05812 | −0.004455 | 0.002673 | 0.09630 |
| LogCRP | |||||||||
| 9-SNP uGRS for VEGF-A | 0.001437 | 0.012397 | 0.90778 | −0.0001663 | 0.0131008 | 0.98988 | −0.001631 | 0.013250 | 0.90207 |
| Model 1 * | Model 2 * | |||||
|---|---|---|---|---|---|---|
| Estimate | SE | p-Value | Estimate | SE | p-Value | |
| logBMI | ||||||
| uGRS*Western Breakfast | 0.0006259 | 0.0016544 | 0.70532 | 0.0009623 | 0.0016699 | 0.564684 |
| uGRS*Legumes and Good Fat | 0.0004362 | 0.0014115 | 0.75742 | −0.0002951 | 0.0015027 | 0.844375 |
| uGRS*Homemade Meal | −0.001836 | 0.001302 | 0.15906 | −0.001894 | 0.001326 | 0.153652 |
| uGRS*Chicken and Sugars | −0.001955 | 0.001442 | 0.17566 | −0.001508 | 0.001577 | 0.339236 |
| uGRS*Eggs and Fibers | −0.000687 | 0.001204 | 0.56840 | 0.0004325 | 0.0014616 | 0.767393 |
| logTriglycerides | ||||||
| uGRS*Western Breakfast | −0.003976 | 0.004121 | 0.335 | −0.003394 | 0.004147 | 0.4135 |
| uGRS*Legumes and Good Fat | −0.003084 | 0.003643 | 0.398 | −0.002993 | 0.003701 | 0.4192 |
| uGRS*Homemade Meal | −0.0003673 | 0.0031521 | 0.907 | −0.0004249 | 0.0031042 | 0.8912 |
| uGRS*Chicken and Sugars | −0.000562 | 0.003527 | 0.873 | 0.000446 | 0.003723 | 0.9047 |
| uGRS*Eggs and Fibers | 0.0004714 | 0.0029163 | 0.872 | −8.952 × 10−7 | 3.645 × 10−3 | 0.9998 |
| logCholesterol | ||||||
| uGRS*Western Breakfast | −0.0003120 | 0.0018673 | 0.86737 | −0.0003595 | 0.0019652 | 0.85495 |
| uGRS*Legumes and Good Fat | 4.399 × 10−4 | 1.654 × 10−3 | 0.79038 | 0.0006190 | 0.0017604 | 0.72529 |
| uGRS*Homemade Meal | 0.0022544 | 0.0014247 | 0.11421 | 0.0024594 | 0.0014679 | 0.09455 |
| uGRS*Chicken and Sugars | 0.0005882 | 0.0015997 | 0.71324 | 0.0011419 | 0.0017668 | 0.51840 |
| uGRS*Eggs and Fibers | −0.0024429 | 0.0013171 | 0.064231 | −0.0035654 | 0.0017221 | 0.0390 |
| logSBP | ||||||
| uGRS*Western Breakfast | 0.0019835 | 0.0010171 | 0.05164 | 0.0021791 | 0.0010716 | 0.042500 |
| uGRS*Legumes and Good Fat | 0.0009800 | 0.0008694 | 0.2601 | 0.001112 | 0.000966 | 0.250296 |
| uGRS*Homemade Meal | −0.0004048 | 0.0008249 | 0.6238 | −0.0006534 | 0.0008508 | 0.442827 |
| uGRS*Chicken and Sugars | 0.0003776 | 0.0008987 | 0.6745 | 0.0003459 | 0.0010081 | 0.731659 |
| uGRS*Eggs and Fibers | −0.0011855 | 0.0007341 | 0.1068 | −0.0018073 | 0.0009354 | 0.053889 |
| logDBP | ||||||
| uGRS*Western Breakfast | 0.0060753 | 0.0014736 | 4.28 × 10−5 | 0.005687 | 0.001537 | 0.000239 |
| uGRS*Legumes and Good Fat | 0.0009039 | 0.0012713 | 0.477344 | 0.001483 | 0.001396 | 0.28856 |
| uGRS*Homemade Meal | −0.0008981 | 0.0012064 | 0.45691 | −0.001097 | 0.001229 | 0.37234 |
| uGRS*Chicken and Sugars | 1.822 × 10−5 | 1.316 × 10−3 | 0.988960 | 0.001229 | 0.001457 | 0.39932 |
| uGRS*Eggs and Fibers | 0.0001876 | 0.0010752 | 0.86156 | −0.0009524 | 0.0013559 | 0.48273 |
| logPP | ||||||
| uGRS*Western Breakfast | −0.004375 | 0.002501 | 0.08081 | −0.003179 | 0.002602 | 0.22237 |
| uGRS*Legumes and Good Fat | 0.0006745 | 0.0021355 | 0.75221 | 0.0001765 | 0.0023393 | 0.93989 |
| uGRS*Homemade Meal | 0.0001585 | 0.0020281 | 0.93772 | −5.986 × 10−5 | 2.067 × 10−3 | 0.97691 |
| uGRS*Chicken and Sugars | 0.0006736 | 0.0022094 | 0.76055 | −0.001662 | 0.002442 | 0.49637 |
| uGRS*Eggs and Fibers | −0.003235 | 0.001801 | 0.07296 | −0.002587 | 0.002269 | 0.2548 |
| logGlucose | ||||||
| uGRS*Western Breakfast | −0.0002371 | 0.0038992 | 0.952 | −0.0006882 | 0.0040671 | 0.866 |
| uGRS*Legumes and Good Fat | −0.004075 | 0.003441 | 0.237 | −0.002575 | 0.003628 | 0.478 |
| uGRS*Homemade Meal | −0.0035228 | 0.0029773 | 0.237 | −0.003946 | 0.003039 | 0.195 |
| uGRS*Chicken and Sugars | 0.003550 | 0.003317 | 0.285 | 0.003922 | 0.003634 | 0.281 |
| uGRS*Eggs and Fibers | 5.869 × 10−3 | 2.745 × 10−3 | 0.0330 | 0.008830 | 0.003550 | 0.0132 |
| logLDL | ||||||
| uGRS*Western Breakfast | −0.0003845 | 0.0028733 | 0.8936 | −0.0008217 | 0.0029791 | 0.7828 |
| uGRS*Legumes and Good Fat | 0.001102 | 0.002545 | 0.6652 | 0.001857 | 0.002669 | 0.4870 |
| uGRS*Homemade Meal | 0.002229 | 0.002194 | 0.3103 | 0.002617 | 0.002230 | 0.2412 |
| uGRS*Chicken and Sugars | 0.0024563 | 0.0024563 | 0.9468 | 0.0008795 | 0.0026757 | 0.7425 |
| uGRS*Eggs and Fibers | −0.004027 | 0.002024 | 0.0472 | −0.005950 | 0.002606 | 0.0229 |
| logHDL | ||||||
| uGRS*Western Breakfast | 0.0007058 | 0.0026675 | 0.79145 | 0.001002 | 0.002789 | 0.71958 |
| uGRS*Legumes and Good Fat | 0.0004628 | 0.0023529 | 0.84413 | −7.341 × 10−5 | 2.485 × 10−3 | 0.97644 |
| uGRS*Homemade Meal | 0.003719 | 0.002032 | 0.06787 | 0.003693 | 0.002080 | 0.07649 |
| uGRS*Chicken and Sugars | 0.001880 | 0.002275 | 0.40903 | 0.002321 | 0.002496 | 0.3529 |
| uGRS*Eggs and Fibers | −0.0003372 | 0.0018861 | 0.85819 | −0.0007087 | 0.0024472 | 0.77227 |
| logCRP | ||||||
| uGRS*Western Breakfast | −0.009797 | 0.013082 | 0.45430 | −0.0072781 | 0.0136345 | 0.59379 |
| uGRS*Legumes and Good Fat | 0.002883 | 0.011393 | 0.80035 | −0.0031947 | 0.0119986 | 0.79019 |
| uGRS*Homemade Meal | 0.010795 | 0.009823 | 0.27239 | 0.011024 | 0.010010 | 0.27144 |
| uGRS*Chicken and Sugars | 0.004140 | 0.010979 | 0.70632 | −0.0006592 | 0.0120081 | 0.95625 |
| uGRS*Eggs and Fibers | −0.006220 | 0.008995 | 0.48963 | 0.0010038 | 0.011644 | 0.93135 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kafyra, M.; Kalafati, I.P.; Gavra, I.; Siest, S.; Dedoussis, G.V. Associations of VEGF-A-Related Variants with Adolescent Cardiometabolic and Dietary Parameters. Nutrients 2023, 15, 1884. https://doi.org/10.3390/nu15081884
Kafyra M, Kalafati IP, Gavra I, Siest S, Dedoussis GV. Associations of VEGF-A-Related Variants with Adolescent Cardiometabolic and Dietary Parameters. Nutrients. 2023; 15(8):1884. https://doi.org/10.3390/nu15081884
Chicago/Turabian StyleKafyra, Maria, Ioanna Panagiota Kalafati, Ioanna Gavra, Sophie Siest, and George V. Dedoussis. 2023. "Associations of VEGF-A-Related Variants with Adolescent Cardiometabolic and Dietary Parameters" Nutrients 15, no. 8: 1884. https://doi.org/10.3390/nu15081884
APA StyleKafyra, M., Kalafati, I. P., Gavra, I., Siest, S., & Dedoussis, G. V. (2023). Associations of VEGF-A-Related Variants with Adolescent Cardiometabolic and Dietary Parameters. Nutrients, 15(8), 1884. https://doi.org/10.3390/nu15081884






