A Multi-Ingredient Supplement Protects against Obesity and Infertility in Western Diet-Fed Mice
Abstract
1. Introduction
2. Methods
2.1. Ethics Approval
2.2. Animals, Housing, Food Consumption and Body Weights
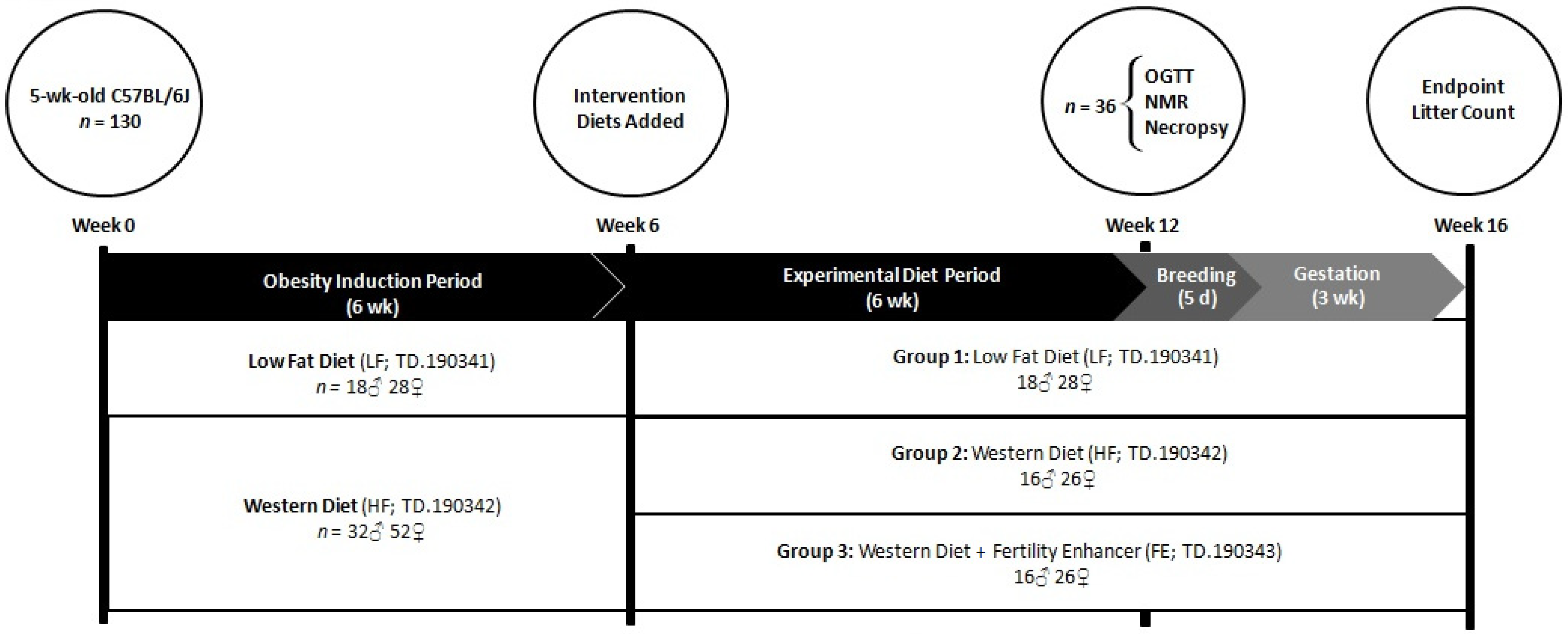
2.3. Study Design
2.4. Diets
2.5. Oral Glucose Tolerance Test (OGTT)
2.6. Time-Domain NMR Whole-Body Composition
2.7. Necropsy
2.8. Paraffin Embedding
2.9. Liver Pathology
2.10. Ovarian Pathology
2.11. Testicular Pathology
Terminal Deoxynucleotidyl Transferase (TdT)-Mediated dUTP Nick-End Labeling (TUNEL)
2.12. RNA Isolation
2.13. cDNA Synthesis
2.14. Quantitative RT-PCR
2.15. Perigonadal WAT Immunoblotting
2.16. Statistical Analyses
3. Results
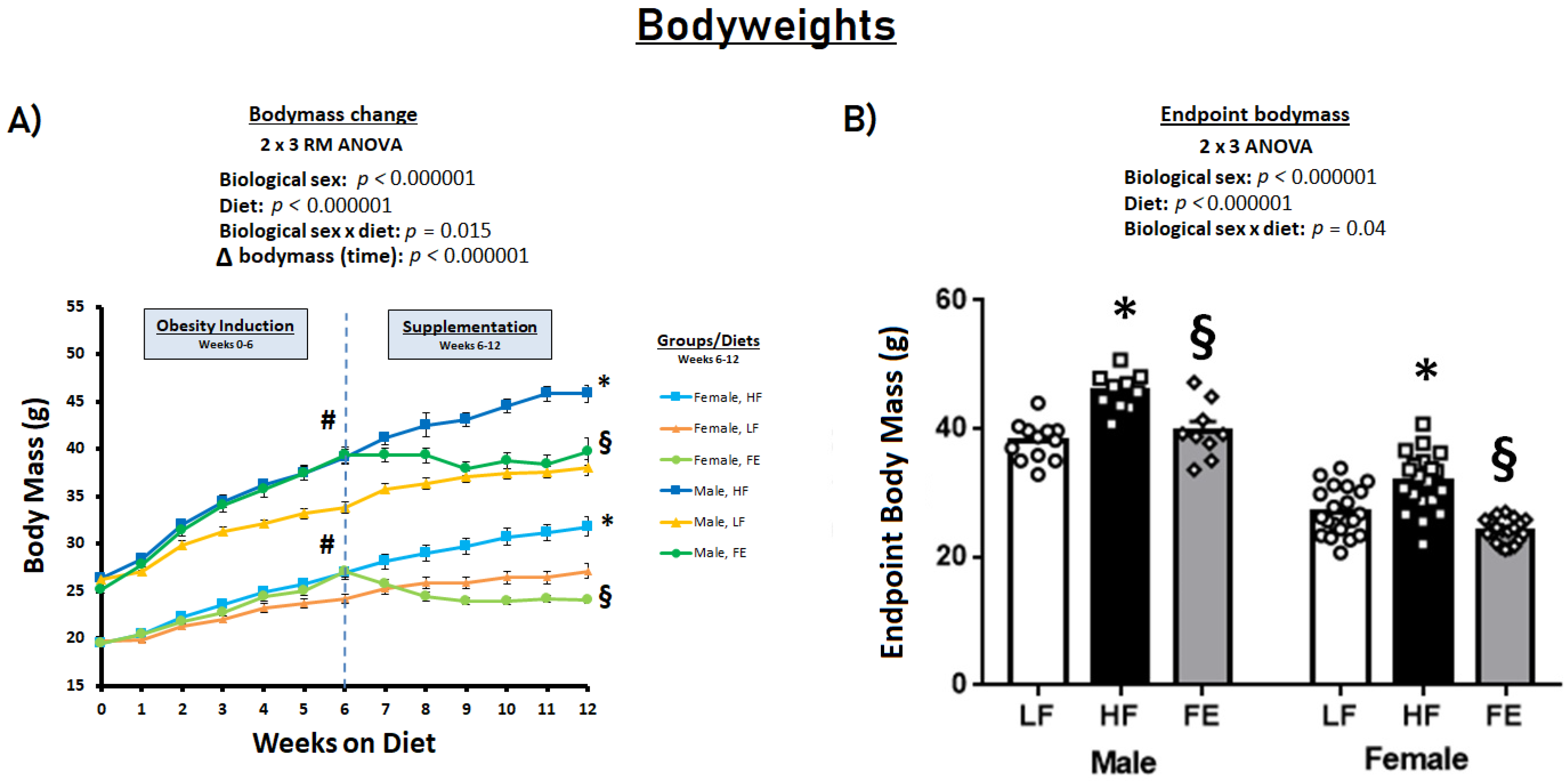
3.1. Food Intakes, Body Weights, and Body Conditions
3.2. Blood Glucose
3.3. Body Composition, WAT Expansion, and WAT Biochemistry
3.4. WAT PGC-1α mRNA Levels and Total OXPHOS Expression
3.5. Liver Pathology
3.5.1. Hepatosteatosis
3.5.2. Liver mRNA Levels
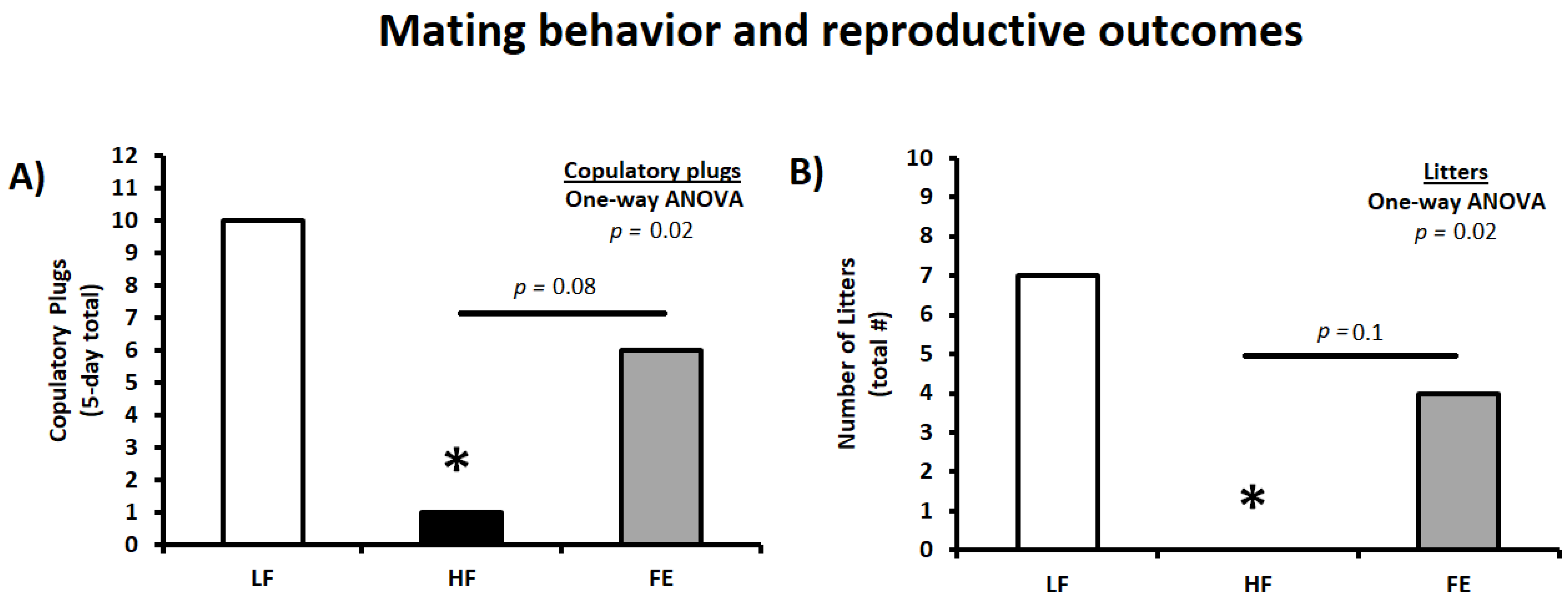
3.6. Mating Behavior and Reproductive Outcomes
3.7. Gonadal Pathology
3.7.1. H&E and TUNEL Staining
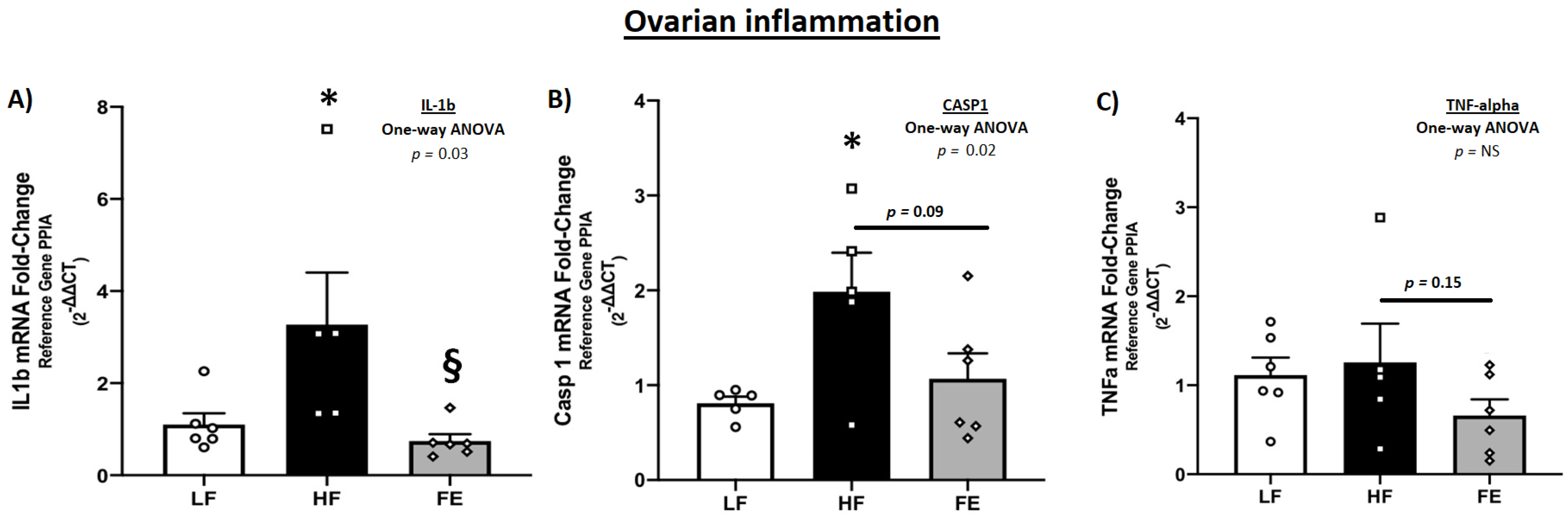
3.7.2. Gonadal mRNA Levels
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eaton, S.B.; Konner, M.; Shostak, M. Stone agers in the fast lane: Chronic degenerative diseases in evolutionary perspective. Am. J. Med. 1988, 84, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Kopp, W. How Western Diet and Lifestyle Drive the Pandemic of Obesity And Civilization Diseases. Diabetes Metab. Syndr. Obes. Targets Ther. 2019, 12, 2221–2236. [Google Scholar] [CrossRef]
- Balistreri, C.R.; Caruso, C.; Candore, G. The role of adipose tissue and adipokines in obesity-related inflammatory diseases. Mediat. Inflamm. 2010, 2010, 802078. [Google Scholar] [CrossRef] [PubMed]
- Koliaki, C.; Roden, M. Alterations of Mitochondrial Function and Insulin Sensitivity in Human Obesity and Diabetes Mellitus. Annu. Rev. Nutr. 2016, 36, 337–367. [Google Scholar] [CrossRef] [PubMed]
- Riazi, K.; Azhari, H.; Charette, J.H.; Underwood, F.E.; King, J.A.; Afshar, E.E.; Swain, M.G.; Congly, S.E.; Kaplan, G.G.; Shaheen, A.-A. The prevalence and incidence of NAFLD worldwide: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2022, 7, 851–861. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef]
- Dobbins, M.; Decorby, K.; Choi, B.C. The Association between Obesity and Cancer Risk: A Meta-Analysis of Observational Studies from 1985 to 2011. ISRN Prev. Med. 2013, 2013, 680536. [Google Scholar] [CrossRef]
- Al-Goblan, A.S.; Al-Alfi, M.A.; Khan, M.Z. Mechanism linking diabetes mellitus and obesity. Diabetes Metab. Syndr. Obes. 2014, 7, 587–591. [Google Scholar] [CrossRef]
- Powell-Wiley, T.M.; Poirier, P.; Burke, L.E.; Després, J.-P.; Gordon-Larsen, P.; Lavie, C.J.; Lear, S.A.; Ndumele, C.E.; Neeland, I.J.; Sanders, P.; et al. Obesity and Cardiovascular Disease: A Scientific Statement From the American Heart Association. Circulation 2021, 143, e984–e1010. [Google Scholar] [CrossRef]
- Abdelaal, M.; le Roux, C.W.; Docherty, N.G. Morbidity and mortality associated with obesity. Ann. Transl. Med. 2017, 5, 161. [Google Scholar] [CrossRef]
- Practice Committee of the American Society for Reproductive Medicine. Obesity and reproduction: A committee opinion. Fertil. Steril. 2015, 104, 1116–1126. [Google Scholar] [CrossRef]
- Bushnik, T.; Cook, J.L.; Yuzpe, A.A.; Tough, S.; Collins, J. Estimating the prevalence of infertility in Canada. Hum. Reprod. 2012, 27, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Boivin, J.; Bunting, L.; Collins, J.A.; Nygren, K.G. International estimates of infertility prevalence and treatment-seeking: Potential need and demand for infertility medical care. Hum. Reprod. 2007, 22, 1506–1512. [Google Scholar] [CrossRef]
- Ramlau-Hansen, C.H.; Thulstrup, A.M.; Nohr, E.A.; Bonde, J.P.; Sorensen, T.I.A.; Olsen, J. Subfecundity in overweight and obese couples. Hum. Reprod. 2007, 22, 1634–1637. [Google Scholar] [CrossRef] [PubMed]
- Smits, R.M.; Mackenzie-Proctor, R.; Fleischer, K.; Showell, M.G. Antioxidants in fertility: Impact on male and female reproductive outcomes. Fertil. Steril. 2018, 110, 578–580. [Google Scholar] [CrossRef]
- Pasquali, R.; Patton, L.; Gambineri, A. Obesity and infertility. Curr. Opin. Endocrinol. Diabetes Obes. 2007, 14, 482–487. [Google Scholar] [CrossRef]
- Zain, M.M.; Norman, R.J. Impact of Obesity on Female Fertility and Fertility Treatment. Women's Health 2008, 4, 183–194. [Google Scholar] [CrossRef]
- Hammoud, A.O.; Gibson, M.; Peterson, C.M.; Hamilton, B.D.; Carrell, D.T. Obesity and Male Reproductive Potential. J. Androl. 2006, 27, 619–626. [Google Scholar] [CrossRef]
- Sermondade, N.; Faure, C.; Fezeu, L.; Shayeb, A.G.; Bonde, J.P.; Jensen, T.K.; Van Wely, M.; Cao, J.; Martini, A.C.; Eskandar, M.; et al. BMI in relation to sperm count: An updated systematic review and collaborative meta-analysis. Hum. Reprod. Update 2012, 19, 221–231. [Google Scholar] [CrossRef]
- Aune, D.; Saugstad, O.D.; Henriksen, T.; Tonstad, S. Maternal Body Mass Index and the Risk of Fetal Death, Stillbirth, and Infant Death: A Systematic Review and Meta-analysis. JAMA 2014, 311, 1536–1546. [Google Scholar] [CrossRef]
- Luke, B.; Brown, M.B.; Stern, J.E.; Missmer, S.A.; Fujimoto, V.Y.; Leach, R.; Group, A.S.W. Female obesity adversely affects assisted reproductive technology (ART) pregnancy and live birth rates. Hum. Reprod. 2011, 26, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Jensen, T.; Scheike, T.; Keiding, N.; Schaumburg, I.; Grandjean, P. Fecundability in relation to body mass and menstrual cycle patterns. Epidemiology 1999, 10, 422–428. [Google Scholar] [CrossRef]
- Sermondade, N.; Huberlant, S.; Bourhis-Lefebvre, V.; Arbo, E.; Gallot, V.; Colombani, M.; Fréour, T. Female obesity is negatively associated with live birth rate following IVF: A systematic review and meta-analysis. Hum. Reprod. Update 2019, 25, 439–451. [Google Scholar] [CrossRef] [PubMed]
- Zumoff, B. Hormonal Abnormalities in Obesity. Acta Med. Scand. 1987, 222, 153–160. [Google Scholar] [CrossRef]
- van der Steeg, J.W.; Steures, P.; Eijkemans, M.J.C.; Habbema, J.D.F.; Hompes, P.G.A.; Burggraaff, J.M.; Oosterhuis, G.J.E.; Bossuyt, P.M.M.; van der Veen, F.; Mol, B.W.J. Obesity affects spontaneous pregnancy chances in subfertile, ovulatory women. Hum. Reprod. 2008, 23, 324–328. [Google Scholar] [CrossRef]
- Koning, A.M.H.; Kuchenbecker, W.K.H.; Groen, H.; Hoek, A.; Land, J.A.; Khan, K.S.; Mol, B.W.J. Economic consequences of overweight and obesity in infertility: A framework for evaluating the costs and outcomes of fertility care. Hum. Reprod. Update 2010, 16, 246–254. [Google Scholar] [CrossRef]
- Maheshwari, A. Overweight and obesity in infertility: Cost and consequences. Hum. Reprod. Update 2010, 16, 229–230. [Google Scholar] [CrossRef]
- LeBlanc, E.L.; Patnode, C.D.; Webber, E.M.; Redmond, N.; Rushkin, M.; O’Connor, E.A. U.S. Preventive Services Task Force Evidence Syntheses, formerly Systematic Evidence Reviews. In Behavioral and Pharmacotherapy Weight Loss Interventions to Prevent Obesity-Related Morbidity and Mortality in Adults: An Updated Systematic Review for the U.S. Preventive Services Task Force; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2018. [Google Scholar]
- Heck, A.M.; Yanovski, J.A.; Calis, K.A. Orlistat, a new lipase inhibitor for the management of obesity. Pharmacotherapy 2000, 20, 270–279. [Google Scholar] [CrossRef]
- Allison, D.B.; Gadde, K.M.; Garvey, W.T.; Peterson, C.A.; Schwiers, M.L.; Najarian, T.; Tam, P.Y.; Troupin, B.; Day, W.W. Controlled-release phentermine/topiramate in severely obese adults: A randomized controlled trial (EQUIP). Obesity 2012, 20, 330–342. [Google Scholar] [CrossRef]
- Wilding, J.P.H.; Batterham, R.L.; Calanna, S.; Davies, M.; Van Gaal, L.F.; Lingvay, I.; McGowan, B.M.; Rosenstock, J.; Tran, M.T.D.; Wadden, T.A.; et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N. Engl. J. Med. 2021, 384, 989–1002. [Google Scholar] [CrossRef]
- Källén, B.A. Antiobesity drugs in early pregnancy and congenital malformations in the offspring. Obes. Res. Clin. Pract. 2014, 8, e571–e576. [Google Scholar] [CrossRef]
- D'Adesky, N.; Ghosh, S. Phentermine Use During First and Second Trimesters Associated with Fetal Stroke. Cureus 2019, 11, e6170. [Google Scholar] [CrossRef]
- Kang, J.G.; Park, C.Y. Anti-Obesity Drugs: A Review about Their Effects and Safety. Diabetes Metab. J. 2012, 36, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Abenhaim, H.A.; Alrowaily, N.; Czuzoj-Shulman, N.; Spence, A.R.; Klam, S.L. Pregnancy outcomes in women with bariatric surgery as compared with morbidly obese women. J. Matern.-Fetal Neonatal Med. 2016, 29, 3596–3601. [Google Scholar] [CrossRef]
- Falcone, V.; Stopp, T.; Feichtinger, M.; Kiss, H.; Eppel, W.; Husslein, P.W.; Prager, G.; Göbl, C.S. Pregnancy after bariatric surgery: A narrative literature review and discussion of impact on pregnancy management and outcome. BMC Pregnancy Childbirth 2018, 18, 507. [Google Scholar] [CrossRef]
- Parker, M.H.; Berghella, V.; Nijjar, J.B. Bariatric surgery and associated adverse pregnancy outcomes among obese women. J. Matern.-Fetal Neonatal Med. 2016, 29, 1747–1750. [Google Scholar] [CrossRef]
- Alamri, S.H.; Abdeen, G.N. Maternal Nutritional Status and Pregnancy Outcomes Post-bariatric Surgery. Obes. Surg. 2022, 32, 1325–1340. [Google Scholar] [CrossRef]
- Hinman, S.K.; Smith, K.B.; Quillen, D.M.; Smith, M.S. Exercise in Pregnancy: A Clinical Review. Sports Health 2015, 7, 527–531. [Google Scholar] [CrossRef]
- Thangaratinam, S.; Rogozińska, E.; Jolly, K.; Glinkowski, S.; Roseboom, T.; Tomlinson, J.W.; Kunz, R.; Mol, B.W.; Coomarasamy, A.; Khan, K.S. Effects of interventions in pregnancy on maternal weight and obstetric outcomes: Meta-analysis of randomised evidence. BMJ Br. Med. J. 2012, 344, e2088. [Google Scholar] [CrossRef]
- Yang, C.; Yang, S.; Zheng, W.; Zu, R.; Ran, S.; Wu, H.; Ren, B.; Lv, N.; Kuang, Y.; Li, M.; et al. Effect of a 60-day weight reduction intervention prior to IVF/ICSI on perinatal outcomes in overweight or obese infertile women. Front. Endocrinol. 2022, 13, 1062790. [Google Scholar] [CrossRef]
- Bullen, B.A.; Skrinar, G.S.; Beitins, I.Z.; von Mering, G.; Turnbull, B.A.; McArthur, J.W. Induction of Menstrual Disorders by Strenuous Exercise in Untrained Women. N. Engl. J. Med. 1985, 312, 1349–1353. [Google Scholar] [CrossRef] [PubMed]
- Pasch, L.A.; Holley, S.R.; Bleil, M.E.; Shehab, D.; Katz, P.P.; Adler, N.E. Addressing the needs of fertility treatment patients and their partners: Are they informed of and do they receive mental health services? Fertil. Steril. 2016, 106, 209–215.e2. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, J.T.; Coates, P.M.; Smith, M.J. Dietary Supplements: Regulatory Challenges and Research Resources. Nutrients 2018, 10, 41. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Risi, R.; Masi, D.; Caputi, A.; Balena, A.; Rossini, G.; Tuccinardi, D.; Mariani, S.; Basciani, S.; Manfrini, S.; et al. Current Evidence to Propose Different Food Supplements for Weight Loss: A Comprehensive Review. Nutrients 2020, 12, 2873. [Google Scholar] [CrossRef] [PubMed]
- Stohs, S.J.; Badmaev, V. A Review of Natural Stimulant and Non-stimulant Thermogenic Agents. Phytother. Res. PTR 2016, 30, 732–740. [Google Scholar] [CrossRef] [PubMed]
- Abdali, D.; Samson, S.E.; Grover, A.K. How effective are antioxidant supplements in obesity and diabetes? Med. Princ. Pract. Int. J. Kuwait Univ. Health Sci. Cent. 2015, 24, 201–215. [Google Scholar] [CrossRef]
- Tun, S.; Spainhower, C.J.; Cottrill, C.L.; Lakhani, H.V.; Pillai, S.S.; Dilip, A.; Chaudhry, H.; Shapiro, J.I.; Sodhi, K. Therapeutic Efficacy of Antioxidants in Ameliorating Obesity Phenotype and Associated Comorbidities. Front. Comorbidities. Front. Pharmacol. 2020, 11, 1234. [Google Scholar] [CrossRef]
- Dongiovanni, P.; Lanti, C.; Riso, P.; Valenti, L. Nutritional therapy for nonalcoholic fatty liver disease. J. Nutr. Biochem. 2016, 29, 1–11. [Google Scholar] [CrossRef]
- Unuofin, J.O.; Lebelo, S.L. Antioxidant Effects and Mechanisms of Medicinal Plants and Their Bioactive Compounds for the Prevention and Treatment of Type 2 Diabetes: An Updated Review. Oxid. Med. Cell. Longev. 2020, 2020, 1356893. [Google Scholar] [CrossRef]
- Garolla, A.; Petre, G.C.; Francini-Pesenti, F.; De Toni, L.; Vitagliano, A.; Di Nisio, A.; Foresta, C. Dietary Supplements for Male Infertility: A Critical Evaluation of Their Composition. Nutrients 2020, 12, 1472. [Google Scholar] [CrossRef]
- Vitagliano, A.; Petre, G.C.; Francini-Pesenti, F.; De Toni, L.; Di Nisio, A.; Grande, G.; Foresta, C.; Garolla, A. Dietary Supplements for Female Infertility: A Critical Review of Their Composition. Nutrients 2021, 13, 3552. [Google Scholar] [CrossRef] [PubMed]
- WHO. Antenatal Care Recommendations for a Positive Pregnancy Experience. Nutritional Interventions Update: Multiple Micronutrient Supplements during Pregnancy; WHO: Geneva, Switzerland, 2020; pp. 1–68. [Google Scholar]
- Pena-Rosas, J.P.; De-Regil, L.M.; Garcia-Casal, M.N.; Dowswell, T. Daily oral iron supplementation during pregnancy. Cochrane Database Syst. Rev. 2015, 12, CD004736. [Google Scholar] [CrossRef] [PubMed]
- De-Regil, L.M.; Pena-Rosas, J.P.; Fernandez-Gaxiola, A.C.; Rayco-Solon, P. Effects and safety of periconceptional oral folate supplementation for preventing birth defects. Cochrane Database Syst. Rev. 2015, 2015, CD007950. [Google Scholar] [CrossRef] [PubMed]
- Smits, R.M.; Mackenzie-Proctor, R.; Yazdani, A.; Stankiewicz, M.T.; Jordan, V.; Showell, M.G. Antioxidants for male subfertility. Cochrane Database Syst. Rev. 2020, 2020, CD007411. [Google Scholar] [CrossRef] [PubMed]
- Middleton, P.; Gomersall, J.C.; Gould, J.F.; Shepherd, E.; Olsen, S.F.; Makrides, M. Omega 3 fatty acid addition during pregnancy. Cochrane Database Syst. Rev. 2018, 2018, CD003402. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, H.; Davies-Tuck, M.; Ellery, S.J.; Grieger, J.A.; Wallace, E.M.; Snow, R.J.; Walker, D.W.; Clifton, V.L. Maternal creatine in pregnancy: A retrospective cohort study. BJOG Int. J. Obstet. Gynaecol. 2016, 123, 1830–1838. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, H.; Ellery, S.; Ireland, Z.; LaRosa, D.; Snow, R.; Walker, D.W. Creatine supplementation during pregnancy: Summary of experimental studies suggesting a treatment to improve fetal and neonatal morbidity and reduce mortality in high-risk human pregnancy. BMC Pregnancy Childbirth 2014, 14, 150. [Google Scholar] [CrossRef]
- Hsu, C.-N.; Tain, Y.-L. Impact of Arginine Nutrition and Metabolism during Pregnancy on Offspring Outcomes. Nutrients 2019, 11, 1452. [Google Scholar] [CrossRef]
- Wikoff, D.; Welsh, B.T.; Henderson, R.; Brorby, G.P.; Britt, J.; Myers, E.; Goldberger, J.; Lieberman, H.R.; O’Brien, C.; Peck, J.; et al. Systematic review of the potential adverse effects of caffeine consumption in healthy adults, pregnant women, adolescents, and children. Food Chem. Toxicol. 2017, 109, 585–648. [Google Scholar] [CrossRef]
- Showell, M.G.; Mackenzie-Proctor, R.; Jordan, V.; Hart, R.J. Antioxidants for female subfertility. Cochrane Database Syst. Rev. 2020, 2020, CD007807. [Google Scholar] [CrossRef]
- Nilsson, M.I.; Mikhail, A.; Lan, L.; Di Carlo, A.; Hamilton, B.; Barnard, K.; Hettinga, B.P.; Hatcher, E.; Tarnopolsky, M.G.; Nederveen, J.P.; et al. A Five-Ingredient Nutritional Supplement and Home-Based Resistance Exercise Improve Lean Mass and Strength in Free-Living Elderly. Nutrients 2020, 12, 2391. [Google Scholar] [CrossRef] [PubMed]
- Bell, K.E.; Snijders, T.; Zulyniak, M.; Kumbhare, D.; Parise, G.; Chabowski, A.; Phillips, S.M. A whey protein-based multi-ingredient nutritional supplement stimulates gains in lean body mass and strength in healthy older men: A randomized controlled trial. PLoS ONE 2017, 12, e0181387. [Google Scholar] [CrossRef] [PubMed]
- Nederveen, J.P.; Manta, K.; Bujak, A.L.; Simone, A.C.; Fuda, M.R.; Nilsson, M.I.; Hettinga, B.P.; Hughes, M.C.; Perry, C.G.R.; Tarnopolsky, M.A. A Novel Multi-Ingredient Supplement Activates a Browning Program in White Adipose Tissue and Mitigates Weight Gain in High-Fat Diet-Fed Mice. Nutrients 2021, 13, 3726. [Google Scholar] [CrossRef]
- Andrikopoulos, S.; Blair, A.R.; Deluca, N.; Fam, B.C.; Proietto, J. Evaluating the glucose tolerance test in mice. Am. J. Physiol.-Endocrinol. Metab. 2008, 295, E1323–E1332. [Google Scholar] [CrossRef]
- Halldorsdottir, S.; Carmody, J.; Boozer, C.N.; Leduc, C.A.; Leibel, R.L. Reproducibility and accuracy of body composition assessments in mice by dual energy x-ray absorptiometry and time domain nuclear magnetic resonance. Int. J. Body Compos. Res. 2009, 7, 147–154. [Google Scholar]
- Kim, M.-S.; Park, J.-Y.; Namkoong, C.; Jang, P.-G.; Ryu, J.-W.; Song, H.-S.; Yun, J.-Y.; Namgoong, I.-S.; Ha, J.; Park, I.-S.; et al. Anti-obesity effects of α-lipoic acid mediated by suppression of hypothalamic AMP-activated protein kinase. Nat. Med. 2004, 10, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Li, Y.; Ma, Z.; Jing, J.; Zhang, Z.; Liu, Y.; Ding, Z. Obesity induces morphological and functional changes in female reproductive system through increases in NF-κB and MAPK signaling in mice. Reprod. Biol. Endocrinol. 2021, 19, 148. [Google Scholar] [CrossRef] [PubMed]
- Nteeba, J.; Ganesan, S.; Keating, A.F. Progressive Obesity Alters Ovarian Folliculogenesis with Impacts on Pro-Inflammatory and Steroidogenic Signaling in Female Mice1. Biol. Reprod. 2014, 91, 86. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Luo, L.L.; Xu, J.J.; Xu, M.Y.; Zhang, X.M.; Zhou, X.L.; Liu, W.J.; Fu, Y.C. Obesity accelerates ovarian follicle development and follicle loss in rats. Metab. Clin. Exp. 2014, 63, 94–103. [Google Scholar] [CrossRef]
- Jungheim, E.S.; Schoeller, E.L.; Marquard, K.L.; Louden, E.D.; Schaffer, J.E.; Moley, K.H. Diet-Induced Obesity Model: Abnormal Oocytes and Persistent Growth Abnormalities in the Offspring. Endocrinology 2010, 151, 4039–4046. [Google Scholar] [CrossRef]
- Hibi, M.; Takase, H.; Iwasaki, M.; Osaki, N.; Katsuragi, Y. Efficacy of tea catechin-rich beverages to reduce abdominal adiposity and metabolic syndrome risks in obese and overweight subjects: A pooled analysis of 6 human trials. Nutr. Res. 2018, 55, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Jurgens, T.M.; Whelan, A.M.; Killian, L.; Doucette, S.; Kirk, S.; Foy, E. Green tea for weight loss and weight maintenance in overweight or obese adults. Cochrane Database Syst. Rev. 2012, 2012, CD008650. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; Lim, W.; Kim, S.; Khil, H.; Cheon, E.; An, S.; Hong, S.; Lee, D.H.; Kang, S.-S.; Oh, H.; et al. Coffee Intake and Obesity: A Meta-Analysis. Nutrients 2019, 11, 1274. [Google Scholar] [CrossRef] [PubMed]
- Tajik, N.; Tajik, M.; Mack, I.; Enck, P. The potential effects of chlorogenic acid, the main phenolic components in coffee, on health: A comprehensive review of the literature. Eur. J. Nutr. 2017, 56, 2215–2244. [Google Scholar] [CrossRef] [PubMed]
- Arroyave-Ospina, J.C.; Wu, Z.; Geng, Y.; Moshage, H. Role of Oxidative Stress in the Pathogenesis of Non-Alcoholic Fatty Liver Disease: Implications for Prevention and Therapy. Antioxidants 2021, 10, 174. [Google Scholar] [CrossRef]
- Dludla, P.V.; Orlando, P.; Silvestri, S.; Marcheggiani, F.; Cirilli, I.; Nyambuya, T.M.; Mxinwa, V.; Mokgalaboni, K.; Nkambule, B.B.; Johnson, R.; et al. Coenzyme Q(10) Supplementation Improves Adipokine Levels and Alleviates Inflammation and Lipid Peroxidation in Conditions of Metabolic Syndrome: A Meta-Analysis of Randomized Controlled Trials. Int. J. Mol. Sci. 2020, 21, 3247. [Google Scholar] [CrossRef]
- Dludla, P.V.; Nyambuya, T.M.; Orlando, P.; Silvestri, S.; Mxinwa, V.; Mokgalaboni, K.; Nkambule, B.B.; Louw, J.; Muller, C.J.F.; Tiano, L. The impact of coenzyme Q(10) on metabolic and cardiovascular disease profiles in diabetic patients: A systematic review and meta-analysis of randomized controlled trials. Endocrinol. Diabetes Metab. 2020, 3, e00118. [Google Scholar] [CrossRef]
- Bobe, G.; Michels, A.J.; Zhang, W.-J.; Purnell, J.Q.; Woffendin, C.; Pereira, C.; Vita, J.A.; Thomas, N.O.; Traber, M.G.; Frei, B.; et al. A Randomized Controlled Trial of Long-Term Alpha Lipoic Acid Supplementation Promotes Weight Loss in Overweight or Obese Adults without Altering Baseline Elevated Plasma Triglyceride Concentrations. J. Nutr. 2020, 150, 2336–2345. [Google Scholar] [CrossRef]
- Vajdi, M.; Abbasalizad Farhangi, M. Alpha-lipoic acid supplementation significantly reduces the risk of obesity in an updated systematic review and dose response meta-analysis of randomised placebo-controlled clinical trials. Int. J. Clin. Pract. 2020, 74, e13493. [Google Scholar] [CrossRef]
- Kucukgoncu, S.; Zhou, E.; Lucas, K.B.; Tek, C. Alpha-lipoic acid (ALA) as a supplementation for weight loss: Results from a meta-analysis of randomized controlled trials. Obes. Rev. 2017, 18, 594–601. [Google Scholar] [CrossRef]
- Namazi, N.; Larijani, B.; Azadbakht, L. Alpha-lipoic acid supplement in obesity treatment: A systematic review and meta-analysis of clinical trials. Clin. Nutr. 2017, 37, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, V.; Khalili, M.; Eghtesadi, S.; Dehghani, S.; Jazayeri, S.; Aghababaee, S.K.; Sabour, H.; Saberi, H.; Eghtesadi, M.; Gohari, M.R. The effect of alpha-lipoic acid (ALA) supplementation on cardiovascular risk factors in men with chronic spinal cord injury: A clinical trial. Spinal Cord 2015, 53, 621–624. [Google Scholar] [CrossRef] [PubMed]
- Devaraj, S.; Leonard, S.; Traber, M.G.; Jialal, I. Gamma-tocopherol supplementation alone and in combination with alpha-tocopherol alters biomarkers of oxidative stress and inflammation in subjects with metabolic syndrome. Free. Radic. Biol. Med. 2008, 44, 1203–1208. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.K.; Chin, K.-Y.; Suhaimi, F.H.; Ahmad, F.; Ima-Nirwana, S. Vitamin E As a Potential Interventional Treatment for Metabolic Syndrome: Evidence from Animal and Human Studies. Front. Pharmacol. 2017, 8, 444. [Google Scholar] [CrossRef]
- Godard, M.P.; Johnson, B.A.; Richmond, S.R. Body Composition and Hormonal Adaptations Associated with Forskolin Consumption in Overweight and Obese Men. Obes. Res. 2005, 13, 1335–1343. [Google Scholar] [CrossRef]
- Loftus, H.L.; Astell, K.J.; Mathai, M.L.; Su, X.Q. Coleus forskohlii Extract Supplementation in Conjunction with a Hypocaloric Diet Reduces the Risk Factors of Metabolic Syndrome in Overweight and Obese Subjects: A Randomized Controlled Trial. Nutrients 2015, 7, 9508–9522. [Google Scholar] [CrossRef]
- Sim, K.A.; Dezarnaulds, G.M.; Denyer, G.S.; Skilton, M.R.; Caterson, I.D. Weight loss improves reproductive outcomes in obese women undergoing fertility treatment: A randomized controlled trial. Clin. Obes. 2014, 4, 61–68. [Google Scholar] [CrossRef]
- Sim, K.A.; Partridge, S.R.; Sainsbury, A. Does weight loss in overweight or obese women improve fertility treatment outcomes? A systematic review. Obes. Rev. 2014, 15, 839–850. [Google Scholar] [CrossRef]
- Rumbold, A.; Ota, E.; Nagata, C.; Shahrook, S.; Crowther, C.A. Vitamin C supplementation in pregnancy. Cochrane Database Syst. Rev. 2015, 2015, CD004072. [Google Scholar] [CrossRef]
- Rumbold, A.; Ota, E.; Hori, H.; Miyazaki, C.; Crowther, C.A. Vitamin E supplementation in pregnancy. Cochrane Database Syst. Rev. 2015, 2015, CD004069. [Google Scholar] [CrossRef]
- Palacios, C.; Trak-Fellermeier, M.A.; Martinez, R.X.; Lopez-Perez, L.; Lips, P.; Salisi, J.A.; John, J.C.; Pena-Rosas, J.P. Regimens of vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2019, 2019, CD013446. [Google Scholar] [CrossRef] [PubMed]
- Vadillo-Ortega, F.; Perichart-Perera, O.; Espino, S.; Avila-Vergara, M.A.; Ibarra, I.; Ahued, R.; Godines, M.; Parry, S.; Macones, G.; Strauss, J.F. Effect of supplementation during pregnancy with L-arginine and antioxidant vitamins in medical food on pre-eclampsia in high risk population: Randomised controlled trial. BMJ 2011, 342, d2901. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Kalman, D.S.; Antonio, J.; Ziegenfuss, T.N.; Wildman, R.; Collins, R.; Candow, D.G.; Kleiner, S.M.; Almada, A.L.; Lopez, H.L. International Society of Sports Nutrition position stand: Safety and efficacy of creatine supplementation in exercise, sport, and medicine. J. Int. Soc. Sports Nutr. 2017, 14, 18. [Google Scholar] [CrossRef]
- De Guingand, D.L.; Ellery, S.J.; Davies-Tuck, M.L.; Dickinson, H. Creatine and pregnancy outcomes, a prospective cohort study in low-risk pregnant women: Study protocol. BMJ Open 2019, 9, e026756. [Google Scholar] [CrossRef] [PubMed]
- Kuchakulla, M.; Narasimman, M.; Soni, Y.; Leong, J.Y.; Patel, P.; Ramasamy, R. A systematic review and evidence-based analysis of ingredients in popular male testosterone and erectile dysfunction supplements. Int. J. Impot. Res. 2020, 33, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, F.; La Vignera, S.; Cannarella, R.; Aversa, A.; Calogero, A.E.; Condorelli, R.A. Evaluation of Sperm Mitochondrial Function: A Key Organelle for Sperm Motility. J. Clin. Med. 2020, 9, 363. [Google Scholar] [CrossRef]
- Buzadzic, B.; Vucetic, M.; Jankovic, A.; Stancic, A.; Korac, A.; Korac, B.; Otasevic, V. New insights into male (in)fertility: The importance of NO. Br. J. Pharmacol. 2015, 172, 1455–1467. [Google Scholar] [CrossRef]
- Umehara, T.; Kawai, T.; Goto, M.; Richards, J.S.; Shimada, M. Creatine enhances the duration of sperm capacitation: A novel factor for improving in vitro fertilization with small numbers of sperm. Hum. Reprod. 2018, 33, 1117–1129. [Google Scholar] [CrossRef]
- Sullivan, E.M.; Pennington, E.R.; Green, W.D.; Beck, M.A.; Brown, D.A.; Shaikh, S.R. Mechanisms by Which Dietary Fatty Acids Regulate Mitochondrial Structure-Function in Health and Disease. Adv. Nutr. 2018, 9, 247–262. [Google Scholar] [CrossRef]
- Cobb, L.K.; McAdams-DeMarco, M.A.; Gudzune, K.A.; Anderson, C.A.; Demerath, E.; Woodward, M.; Selvin, E.; Coresh, J. Changes in Body Mass Index and Obesity Risk in Married Couples Over 25 Years: The ARIC Cohort Study. Am. J. Epidemiol. 2016, 183, 435–443. [Google Scholar] [CrossRef] [PubMed]







| MALE | FEMALE | |||||
|---|---|---|---|---|---|---|
| LF | HF | FE | LF | HF | FE | |
| Food Intake | ||||||
| g/day | 3.4 ± 0.07 | 3.1 ± 0.05 * | 2.5 ± 0.17 § | 3.0 ± 0.02 | 2.6 ± 0.04 * | 2.2 ± 0.03 § |
| kcal/day | 12.3 ± 0.24 | 14.1 ± 0.22 * | 11.2 ± 0.74 § | 10.6 ± 0.08 | 12.0 ± 0.20 * | 9.8 ± 0.15 § |
| Body Weight | ||||||
| BW (g) | 38.0 ± 0.9 | 45.8 ± 0.9 * | 39.7 ± 1.5 § | 27.1 ± 0.8 | 31.8 ± 1.0 * | 24.1 ± 0.4 § |
| Body Composition (NMR) | ||||||
| Fat mass (g) | 11.8 ± 1.0 | 23.0 ± 0.7 * | 17.0 ± 1.7 § | 5.0 ± 1.3 | 13.2 ± 1.7 * | 4.1 ± 1.1 § |
| Fat mass (%) | 34.3 ± 2.3 | 52.2 ± 1.2 * | 46.5 ± 3.1 § | 19.7 ± 3.7 | 42.4 ± 3.7 * | 16.4 ± 3.6 § |
| Body Composition Index | 1.93 ± 0.15 | 1.10 ± 0.04 * | 1.31 ± 0.12 § | 4.05 ± 0.61 | 1.50 ± 0.19 * | 5.47 ± 1.2 § |
| Tissue Weights | ||||||
| Perirenal WAT (mg) | 438.1 ± 86.4 | 882.6 ± 34.8 * | 602.5 ± 137.0 § | 229.4 ± 102.5 | 465.1 ± 105.8 * | 115.1 ± 65.2 § |
| Mesenteric WAT (mg) | 403.8 ± 52.8 | 963.25 ± 99.4 * | 606.8 ± 129.9 § | 194.8 ± 57.1 | 315.0 ± 47.3 * | 92.6 ± 31.8 § |
| Perigonadal WAT (mg) | 1538.6 ± 130.7 | 2670.3 ± 48.3 * | 2532.7 ± 233.9 | 475.0 ± 117.8 | 1290.2 ± 191.2 * | 301.7 ± 81.4 § |
| Inguinal WAT (mg) | 313.4 ± 31.2 | 462.8 ± 55.0 * | 331.8 ± 48.8 § | 168.9 ± 42.0 | 478.7 ± 54.9 * | 99.8 ± 17.9 § |
| Total WAT (mg) | 3367 ± 376 | 6223 ± 297 * | 5092 ± 560 § | 1335 ± 399 | 3186 ± 499 * | 762 ± 245 § |
| Liver (mg) | 1328.9 ± 80.4 | 3176.4 ± 153.5 * | 1354.6 ± 83.2 § | 951.6 ± 89.0 | 1553.1 ± 124.2 * | 1014.4 ± 17.8 § |
| Blood Glucose | ||||||
| Fasting (mmol/L) | 9.9 ± 1.2 | 12.5 ± 0.6 * | 11.0 ± 0.8 | 9.20 ± 0.6 | 10.6 ± 0.3 | 7.1 ± 0.5 § |
| AUC | 1819.9 ± 128.3 | 2084.3 ± 168.8 | 2144.5 ± 128.5 | 1683.9 ± 157.3 | 2028.4 ± 123.5 | 1674.4 ± 83.1 |
| Mating and Reproductive Outcomes | LF | HF | FE |
|---|---|---|---|
| Copulatory plugs (5-day total) | 10 | 1 * | 6 |
| Copulatory plugs (% total females) | 50% | 6% | 30% |
| Number of litters (total) | 7 | 0 * | 4 |
| Copulatory success (%; Litters/Plugs) | 70% | 0% | 67% |
| MALE | FEMALE | |||||
|---|---|---|---|---|---|---|
| LF (n = 5) | HF (n = 5) | FE (n = 5) | LF (n = 3) | HF (n = 3) | FE (n = 3) | |
| Gonadal pathology | ||||||
| Testicular pathology | ||||||
| Mass (mg) | 189.5 ± 3.1 | 242.0 ± 9.5 * | 199.1 ± 8.4 § | NA | NA | NA |
| Seminiferous tubule diameter (AU; pixels) | 571.1 ± 10.0 | 604.7 ± 12.6 | 568.5 ± 24.0 | NA | NA | NA |
| Seminiferous epithelia (pathology score) | 0.9 ± 0.5 | 0.7 ± 0.2 | 0.5 ± 0.2 | NA | NA | NA |
| Seminiferous tubules with apoptosis (%) | 29.5 ± 1.7 | 38.1 ± 17.8 | 28.6 ± 5.4 | NA | NA | NA |
| Ovarian pathology | ||||||
| Mass (mg) | NA | NA | NA | 16.0 ± 1.0 | 19.4 ± 0.9 | 17.4 ± 1.0 |
| Volume (mm3) | NA | NA | NA | 0.046 ± 0.013 | 0.054 ± 0.010 | 0.040 ± 0.007 |
| Viable follicles (#) | NA | NA | NA | 61.7 ± 10.3 | 55.7 ± 19.9 | 55.7 ± 17.6 |
| Viable follicles (#/mm3) | NA | NA | NA | 1574.4 ± 465.6 | 986.1 ± 296.5 | 1344.5 ± 301.7 |
| Growing follicles (#) | NA | NA | NA | 29.0 ± 2.9 | 23.0 ± 7.9 | 29.0 ± 8.7 |
| Growing follicles (#/mm3) | NA | NA | NA | 754.5 ± 209.2 | 402.9 ± 73.9 | 691.6 ± 134.6 |
| Atretic follicles (#) | NA | NA | NA | 18.0 ± 1.5 | 26.0 ± 1.0 * | 12.0 ± 1.0 § |
| Atretic follicles (#/mm3) | NA | NA | NA | 461.6 ± 128.5 | 512.8 ± 85.0 | 317.6 ± 52.1 |
| Apoptotic granulosa cells (#/ovary) | NA | NA | NA | 424.0 ± 109.1 | 606.5 ± 321.1 | 364.2 ± 81.6 |
| Apoptotic granulosa cells (#/follicle) | NA | NA | NA | 15.1 ± 5.7 | 15.6 ± 7.5 | 14.5 ± 2.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nilsson, M.I.; May, L.; Roik, L.J.; Fuda, M.R.; Luo, A.; Hettinga, B.P.; Bujak, A.L.; Tarnopolsky, M.A. A Multi-Ingredient Supplement Protects against Obesity and Infertility in Western Diet-Fed Mice. Nutrients 2023, 15, 611. https://doi.org/10.3390/nu15030611
Nilsson MI, May L, Roik LJ, Fuda MR, Luo A, Hettinga BP, Bujak AL, Tarnopolsky MA. A Multi-Ingredient Supplement Protects against Obesity and Infertility in Western Diet-Fed Mice. Nutrients. 2023; 15(3):611. https://doi.org/10.3390/nu15030611
Chicago/Turabian StyleNilsson, Mats I., Linda May, Liza J. Roik, Matthew R. Fuda, Ashely Luo, Bart P. Hettinga, Adam L. Bujak, and Mark A. Tarnopolsky. 2023. "A Multi-Ingredient Supplement Protects against Obesity and Infertility in Western Diet-Fed Mice" Nutrients 15, no. 3: 611. https://doi.org/10.3390/nu15030611
APA StyleNilsson, M. I., May, L., Roik, L. J., Fuda, M. R., Luo, A., Hettinga, B. P., Bujak, A. L., & Tarnopolsky, M. A. (2023). A Multi-Ingredient Supplement Protects against Obesity and Infertility in Western Diet-Fed Mice. Nutrients, 15(3), 611. https://doi.org/10.3390/nu15030611








