Chinese Yam and Its Active Components Regulate the Structure of Gut Microbiota and Indole-like Metabolites in Anaerobic Fermentation In Vitro
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of CY and CYPs
2.3. In Vitro Gastrointestinal Digestion of Chinese Yam and Its Components
2.4. Preparation of Fecal Flora Suspension
2.5. In Vitro Colonic Fermentation Model
2.6. Analysis of pH and SCFAs
2.7. Determination of Indole-like Compounds
2.8. Processing and Analysis of 16S rRNA Gene Sequencing
2.9. Statistical Analysis
3. Results
3.1. Establishment of In Vitro Digestion and Fermentation Models of CY and Its Components
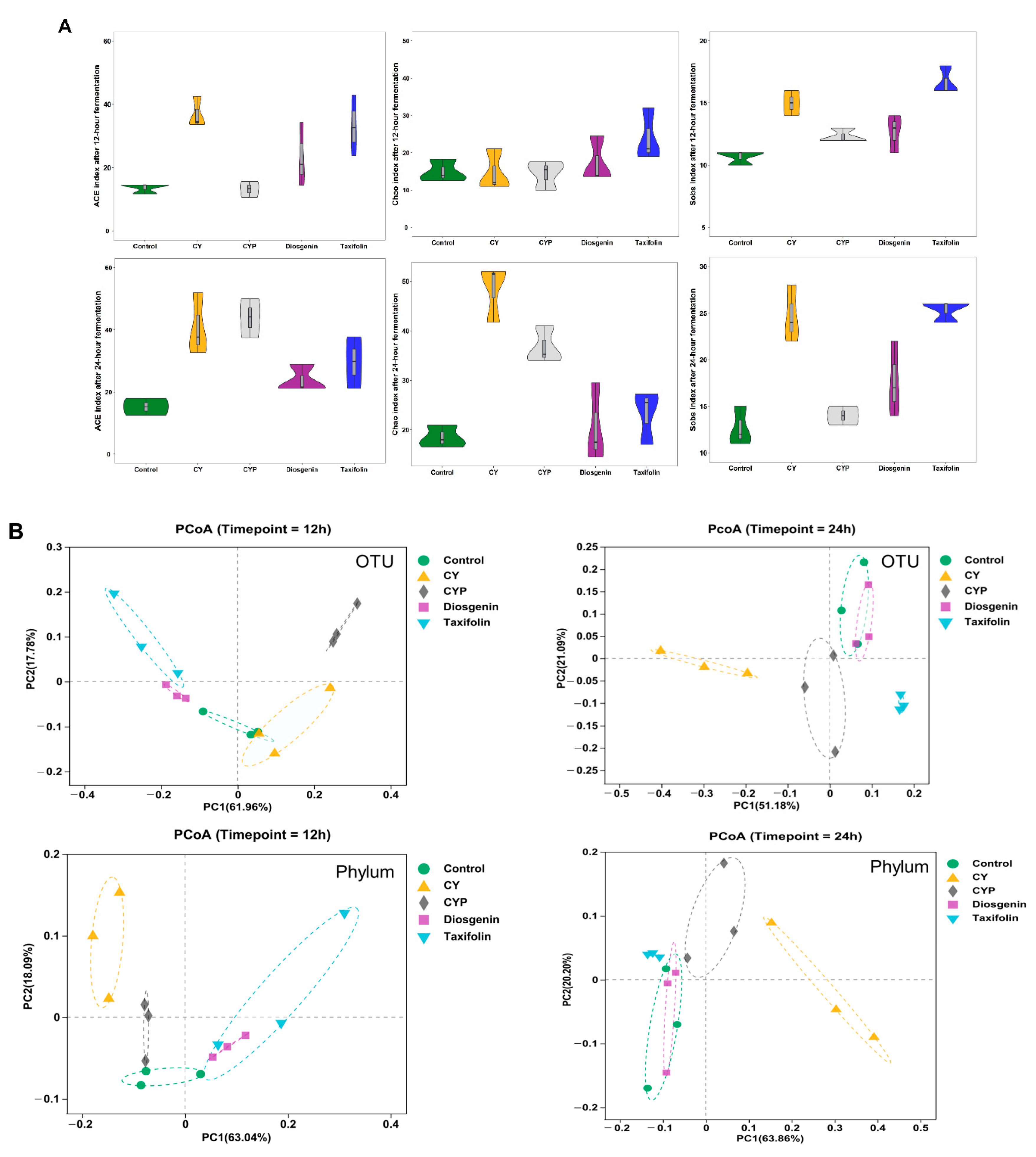
3.2. Improvement in Chinese Yam and Its Components in Terms of Bacterial Richness and Diversity after In Vitro Simulation Trial
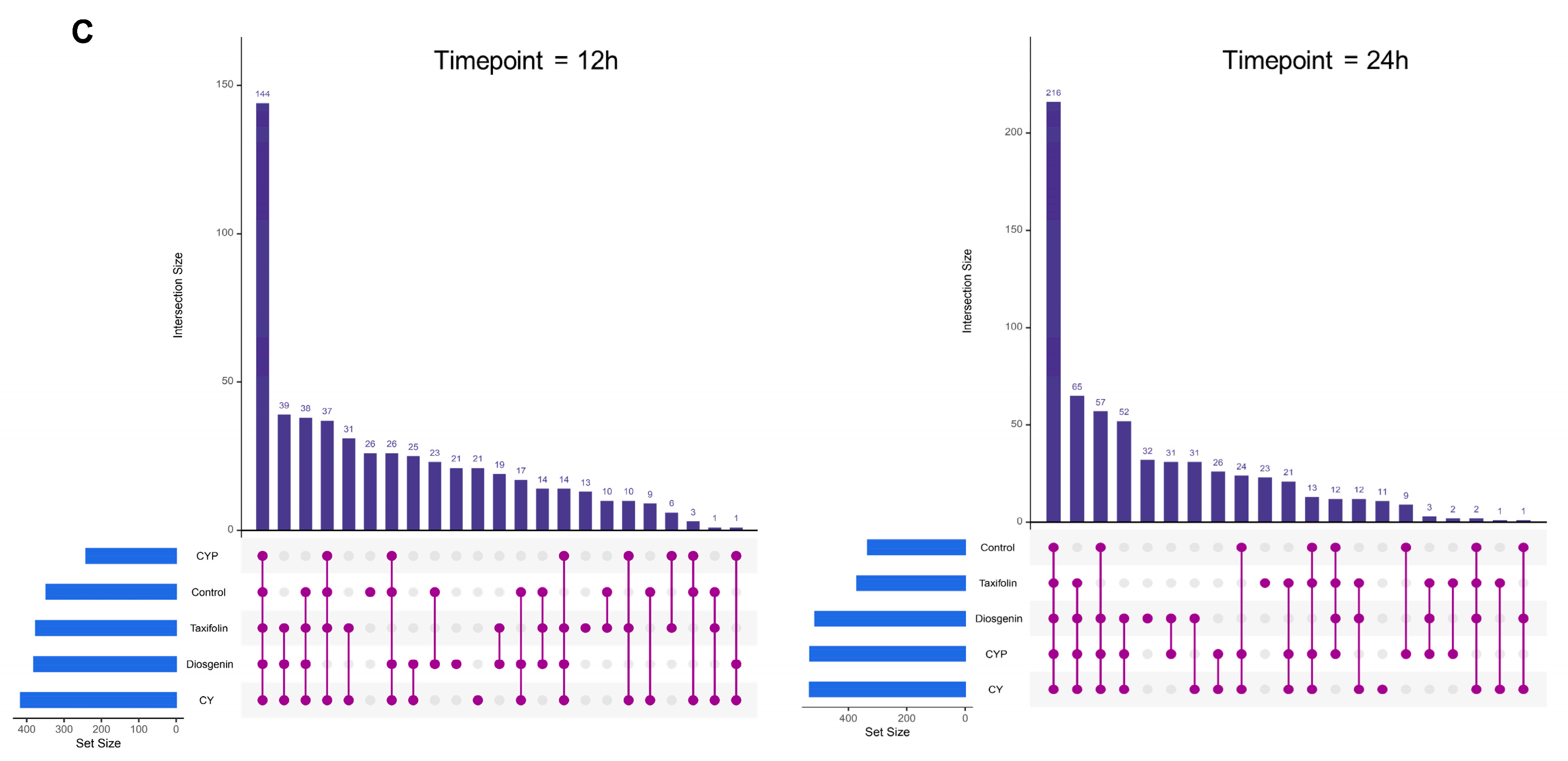
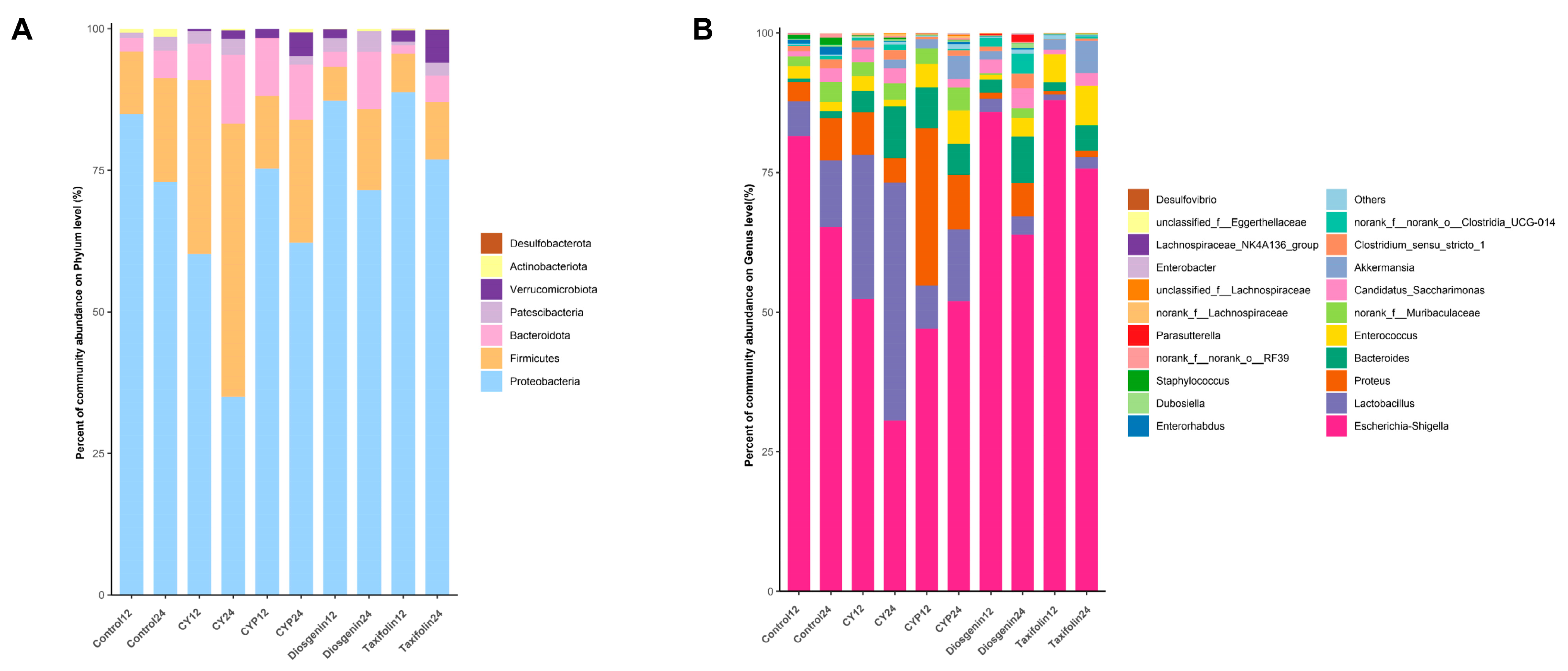
3.3. Effects of CY and Its Active Ingredients on the Composition and Structure of Microbiota In Vitro
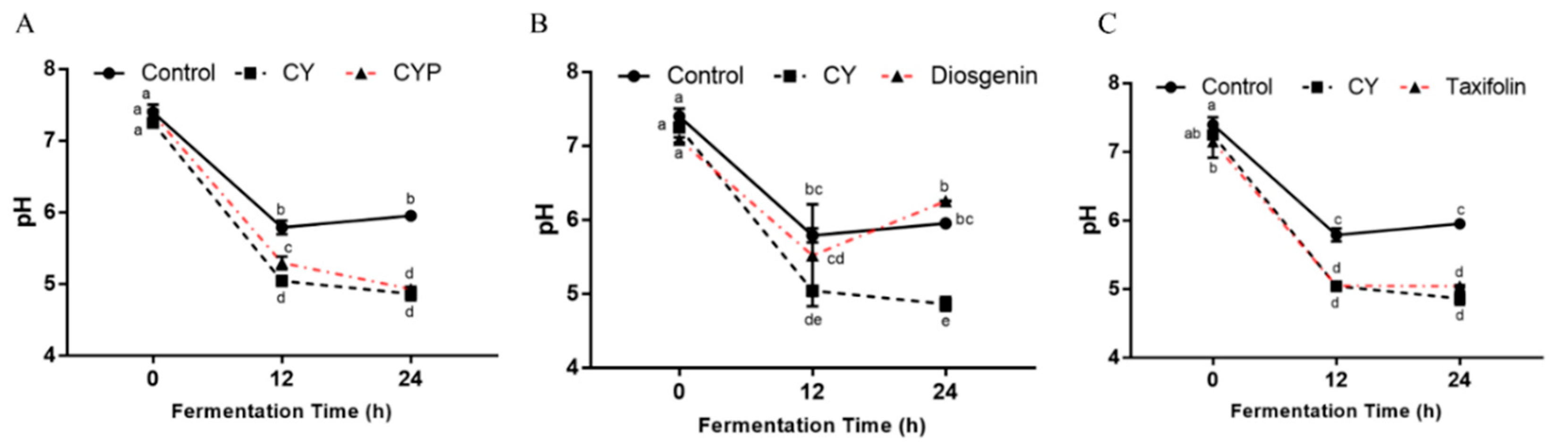
3.4. Effects of CY and Its Active Components on pH and SCFA Production In Vitro
3.5. Effects of CY and Its Active Components on Specific Metabolites during In Vitro Digestive and Fermentation
3.6. Correlation Analysis of Gut Microbiota at the Genus Level and Indole-like Metabolites In Vitro
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
References
- Savage, N. The Complex Relationship between Drugs and the Microbiome. Nature 2020, 577, S10–S11. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.R.; Proctor, L.M.; Surette, M.G.; Suchodolski, J.S. The Microbiome: The Trillions of Microorganisms That Maintain Health and Cause Disease in Humans and Companion Animals. Vet. Pathol. 2016, 53, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Depommier, C.; Derrien, M.; Everard, A.; de Vos, W.M. Akkermansia Muciniphila: Paradigm for next-Generation Beneficial Microorganisms. Nat. Rev. Gastroenterol. Hepatol. 2022, 19, 625–637. [Google Scholar] [CrossRef]
- Vijay, A.; Valdes, A.M. Role of the Gut Microbiome in Chronic Diseases: A Narrative Review. Eur. J. Clin. Nutr. 2021, 76, 489–501. [Google Scholar] [CrossRef] [PubMed]
- Van Craeyveld, V.; Swennen, K.; Dornez, E.; Van De Wiele, T.; Marzorati, M.; Verstraete, W.; Delaedt, Y.; Onagbesan, O.; Decuypere, E.; Buyse, J.; et al. Structurally Different Wheat-Derived Arabinoxylooligosaccharides Have Different Prebiotic and Fermentation Properties in Rats. J. Nutr. 2008, 138, 2348–2355. [Google Scholar] [CrossRef] [PubMed]
- Warren, F.J.; Fukuma, N.M.; Mikkelsen, D.; Flanagan, B.M.; Williams, B.A.; Lisle, A.T.; Cuív, P.Ó.; Morrison, M.; Gidley, M.J. Food Starch Structure Impacts Gut Microbiome Composition. mSphere 2018, 3, e00086-18. [Google Scholar] [CrossRef] [PubMed]
- Gill, P.A.; van Zelm, M.C.; Muir, J.G.; Gibson, P.R. Review Article: Short Chain Fatty Acids as Potential Therapeutic Agents in Human Gastrointestinal and Inflammatory Disorders. Aliment. Pharmacol. Ther. 2018, 48, 15–34. [Google Scholar] [CrossRef] [PubMed]
- Serino, M. SCFAs—The Thin Microbial Metabolic Line between Good and Bad. Nat. Rev. Endocrinol. 2019, 15, 318–319. [Google Scholar] [CrossRef]
- Neis, E.P.J.G.; Dejong, C.H.C.; Rensen, S.S. The Role of Microbial Amino Acid Metabolism in Host Metabolism. Nutrients 2015, 7, 2930. [Google Scholar] [CrossRef]
- Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut Microbiota Functions: Metabolism of Nutrients and Other Food Components. Eur. J. Nutr. 2017, 57, 1–24. [Google Scholar] [CrossRef]
- Stoeva, M.K.; Garcia-So, J.; Justice, N.; Myers, J.; Tyagi, S.; Nemchek, M.; McMurdie, P.J.; Kolterman, O.; Eid, J. Butyrate-Producing Human Gut Symbiont, Clostridium butyricum, and Its Role in Health and Disease. Gut Microbes 2021, 13, 1907272. [Google Scholar] [CrossRef] [PubMed]
- Luster, A.D.; Alon, R.; von Andrian, U.H. Immune Cell Migration in Inflammation: Present and Future Therapeutic Targets. Nat. Immunol. 2005, 6, 1182–1190. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Su, X.; Chen, X.; Zhao, H.; Bo, C.; Xu, J.; Bai, H.; Ning, K. Biological Ingredient Analysis of Traditional Chinese Medicine Preparation Based on High-Throughput Sequencing: The Story for Liuwei Dihuang Wan. Sci. Rep. 2014, 4, 5147. [Google Scholar] [CrossRef] [PubMed]
- Epping, J.; Laibach, N. An Underutilized Orphan Tuber Crop—Chinese Yam: A Review. Planta 2020, 252, 58. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Liang, X. Food Therapy and Medical Diet Therapy of Traditional Chinese Medicine. Clin. Nutr. Exp. 2018, 18, 1–5. [Google Scholar] [CrossRef]
- Li, M.; Chen, L.X.; Chen, S.R.; Deng, Y.; Zhao, J.; Wang, Y.; Li, S.P. Non-Starch Polysaccharide from Chinese Yam Activated RAW 264.7 Macrophages through the Toll-like Receptor 4 (TLR4)-NF-ΚB Signaling Pathway. J. Funct. Foods 2017, 37, 491–500. [Google Scholar] [CrossRef]
- Dong, M.; Meng, Z.; Kuerban, K.; Qi, F.; Liu, J.; Wei, Y.; Wang, Q.; Jiang, S.; Feng, M.; Ye, L. Diosgenin Promotes Antitumor Immunity and PD-1 Antibody Efficacy against Melanoma by Regulating Intestinal Microbiota. Cell Death Dis. 2018, 9, 1039. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Liu, T.; Si, Y.; Cao, B.; Zhang, Y.; Zheng, X.; Feng, W. Integrated Metabolomics and 16S RRNA Sequencing to Investigate the Regulation of Chinese Yam on Antibiotic-Induced Intestinal Dysbiosis in Rats. Artif. Cells Nanomed. Biotechnol. 2019, 47, 3382–3390. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Ji, S.; Xu, T.; Zhong, Y.; Xu, M.; Liu, Y.; Li, M.; Fan, B.; Wang, F.; Xiao, J.; et al. Chinese Yam (Dioscorea): Nutritional Value, Beneficial Effects, and Food and Pharmaceutical Applications. Trends Food Sci. Technol. 2023, 134, 29–40. [Google Scholar] [CrossRef]
- Ma, F.; Wang, D.; Zhang, Y.; Li, M.; Qing, W.; Tikkanen-Kaukanen, C.; Liu, X.; Bell, A.E. Characterisation of the Mucilage Polysaccharides from Dioscorea Opposita Thunb. with Enzymatic Hydrolysis. Food Chem. 2018, 245, 13–21. [Google Scholar] [CrossRef]
- Wu, S.; Chen, X.; Cai, R.; Chen, X.; Zhang, J.; Xie, J.; Shen, M. Sulfated Chinese Yam Polysaccharides Alleviate LPS-Induced Acute Inflammation in Mice through Modulating Intestinal Microbiota. Foods 2023, 12, 1772. [Google Scholar] [CrossRef] [PubMed]
- Lepage, C.; Léger, D.Y.; Bertrand, J.; Martin, F.; Beneytout, J.L.; Liagre, B. Diosgenin Induces Death Receptor-5 through Activation of P38 Pathway and Promotes TRAIL-Induced Apoptosis in Colon Cancer Cells. Cancer Lett. 2011, 301, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Rojas, J.; Buitrago, A. Antioxidant Activity of Phenolic Compounds Biosynthesized by Plants and Its Relationship with Prevention of Neurodegenerative Diseases; Elsevier Inc.: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Su, H.; Wang, W.J.; Zheng, G.D.; Yin, Z.P.; Li, J.E.; Chen, L.L.; Zhang, Q.F. The Anti-Obesity and Gut Microbiota Modulating Effects of Taxifolin in C57BL/6J Mice Fed with a High-Fat Diet. J. Sci. Food Agric. 2022, 102, 1598–1608. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, Y.; Hu, X.; Feng, F.; Cai, L.; Chen, F. Assessing the Effects of Ginger Extract on Polyphenol Profiles and the Subsequent Impact on the Fecal Microbiota by Simulating Digestion and Fermentation In Vitro. Nutrients 2020, 12, 3194. [Google Scholar] [CrossRef]
- Moon, J.S.; Li, L.; Bang, J.; Han, N.S. Application of in Vitro Gut Fermentation Models to Food Components: A Review. Food Sci. Biotechnol. 2016, 25, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Burillo, S.; Molino, S.; Navajas-Porras, B.; Valverde-Moya, Á.J.; Hinojosa-Nogueira, D.; López-Maldonado, A.; Pastoriza, S.; Rufián-Henares, J.Á. An In Vitro Batch Fermentation Protocol for Studying the Contribution of Food to Gut Microbiota Composition and Functionality. Nat. Protoc. 2021, 16, 3186–3209. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Xie, M.; Wan, P.; Chen, D.; Ye, H.; Chen, L.; Zeng, X.; Liu, Z. Digestion under Saliva, Simulated Gastric and Small Intestinal Conditions and Fermentation In Vitro by Human Intestinal Microbiota of Polysaccharides from Fuzhuan Brick Tea. Food Chem. 2018, 244, 331–339. [Google Scholar] [CrossRef]
- Blaak, E.E.; Canfora, E.E.; Theis, S.; Frost, G.; Groen, A.K.; Mithieux, G.; Nauta, A.; Scott, K.; Stahl, B.; van Harsselaar, J.; et al. Short Chain Fatty Acids in Human Gut and Metabolic Health. Benef. Microbes 2020, 11, 411–455. [Google Scholar] [CrossRef]
- Deleu, S.; Machiels, K.; Raes, J.; Verbeke, K.; Vermeire, S. Short Chain Fatty Acids and Its Producing Organisms: An Overlooked Therapy for IBD. eBioMedicine 2021, 66, 103293. [Google Scholar] [CrossRef]
- Agus, A.; Planchais, J.; Sokol, H. Gut Microbiota Regulation of Tryptophan Metabolism in Health and Disease. Cell Host Microbe 2018, 23, 716–724. [Google Scholar] [CrossRef]
- Hezaveh, K.; Shinde, R.S.; Klö, A.; Philpott, D.J.; Brooks, D.G.; Mcgaha, T.L. Tryptophan-Derived Microbial Metabolites Activate the Aryl Hydrocarbon Receptor in Tumor-Associated Macrophages to Suppress Anti-Tumor Immunity. Immunity 2022, 55, 324–340. [Google Scholar] [CrossRef] [PubMed]
- Abedi, E.; Hashemi, S.M.B. Lactic Acid Production—Producing Microorganisms and Substrates Sources-State of Art. Heliyon 2020, 6, e04974. [Google Scholar] [CrossRef]
- Serger, E.; Luengo-Gutierrez, L.; Chadwick, J.S.; Kong, G.; Zhou, L.; Crawford, G.; Danzi, M.C.; Myridakis, A.; Brandis, A.; Bello, A.T.; et al. The Gut Metabolite Indole-3 Propionate Promotes Nerve Regeneration and Repair. Nature 2022, 607, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Zhou, Y.; Zhang, R.; Chen, Y.; Wang, F.; Zhang, M. Gut Microbial Fermentation Promotes the Intestinal Anti-Inflammatory Activity of Chinese Yam Polysaccharides. Food Chem. 2023, 402, 134003. [Google Scholar] [CrossRef] [PubMed]
- Pan, T.; Pei, Z.; Fang, Z.; Wang, H.; Zhu, J.; Zhang, H.; Zhao, J.; Chen, W.; Lu, W. Uncovering the Specificity and Predictability of Tryptophan Metabolism in Lactic Acid Bacteria with Genomics and Metabolomics. Front. Cell. Infect. Microbiol. 2023, 13, 1154346. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Hu, W.; Wu, S.; Zhu, Z.; Lu, R.; Yang, G.; Qin, C.; Yang, L.; Nie, G. Chinese Yam Peel Enhances the Immunity of the Common Carp (Cyprinus carpio L.) by Improving the Gut Defence Barrier and Modulating the Intestinal Microflora. Fish Shellfish Immunol. 2019, 95, 528–537. [Google Scholar] [CrossRef]
- Rastogi, S.; Singh, A. Gut Microbiome and Human Health: Exploring How the Probiotic Genus Lactobacillus Modulate Immune Responses. Front. Pharmacol. 2022, 13, 1042189. [Google Scholar] [CrossRef] [PubMed]
- Roager, H.M.; Licht, T.R. Microbial Tryptophan Catabolites in Health and Disease. Nat. Commun. 2018, 9, 3294. [Google Scholar] [CrossRef]
- Zhang, J.; Zhu, S.; Ma, N.; Johnston, L.J.; Wu, C.; Ma, X. Metabolites of Microbiota Response to Tryptophan and Intestinal Mucosal Immunity: A Therapeutic Target to Control Intestinal Inflammation. Med. Res. Rev. 2021, 41, 1061–1088. [Google Scholar] [CrossRef]



| Additives | Content in CY (Dry Basis, %) | Amount (mg) |
|---|---|---|
| CY | 100 | 400 |
| CYPs | 12.54 | 139 |
| Diosgenin | 0.9 | 6.4 |
| Taxifolin | 0.41 | 1.64 |
| SCFA | Treatment | Fermentation Time (h) | ||
|---|---|---|---|---|
| 0 | 12 | 24 | ||
| Acetic acid | Control | 65.62 ± 4.06 a | 115.74 ± 7.55 b | 77.15 ± 3.85 a |
| CY | 82.88 ± 4.46 a | 145.75 ± 10.52 c | 123.69 ± 4.66 b | |
| Propionic acid | Control | 12.44 ± 1.27 a | 15.05 ± 0.48 ab | 13.68 ± 0.49 a |
| CY | 12.25 ± 0.33 a | 18.03 ± 0.75 c | 17.04 ± 1.47 bc | |
| Butyric acid | Control | 10.24 ± 0.27 a | 13.95 ± 0.65 b | 14.41 ± 1.30 bc |
| CY | 9.81 ± 0.34 a | 17.51 ± 1.31 c | 26.55 ± 1.69 d | |
| Iso-butyric acid | Control | 1.73 ± 0.09 a | 3.98 ± 0.42 b | 4.13 ± 0.48 b |
| CY | 1.59 ± 0.13 a | 4.72 ± 0.70 b | 3.75 ± 0.36 b | |
| Valeric acid | Control | 6.45 ± 0.27 c | 6.18 ± 0.32 bc | 5.11 ± 0.13 a |
| CY | 6.35 ± 0.17 c | 7.34 ± 0.14 d | 5.38 ± 0.48 ab | |
| Iso-valeric acid | Control | 6.18 ± 0.31 ab | 7.21 ± 0.67 bc | 6.29 ± 0.19 ab |
| CY | 6.27 ± 0.41 ab | 8.28 ± 0.63 c | 5.62 ± 0.30 a | |
| Total SCFA | Control | 102.66 ± 5.30 a | 162.10 ± 8.11 b | 120.77 ± 3.79 ab |
| CY | 119.15 ± 4.44 a | 201.63 ± 11.87 c | 182.02 ± 5.69 bc | |
| Treatment | Fermentation Time (h) | ||
|---|---|---|---|
| 0 | 12 | 24 | |
| Control | 894.84 ± 54.89 a | 1051.73 ± 9.10 d* | 1027.06 ± 4.11 bc*# |
| CY | 978.08 ± 17.14 a | 1018.75 ± 8.87 c | 1053.33 ± 7.13 c*# |
| CYPs | 974.50 ± 42.88 a | 995.75 ± 9.22 bc | 989.96 ± 21.89 ab |
| Diosgenin | 926.07 ± 30.39 a | 981.19 ± 10.29 b | 1026.71 ± 16.33 bc*# |
| Taxifolin | 906.03 ± 16.18 a | 932.96 ± 3.00 a* | 956.01 ± 3.51 a* |
| Treatment | Fermentation Time (h) | ||
|---|---|---|---|
| 0 | 12 | 24 | |
| Control | 177.75 ± 11.77 a | 209.06 ± 6.19 a | 241.09 ± 11.42 a* |
| CY | 199.28 ± 12.12 a | 212.75 ± 17.16 a | 324.9 ± 23.38 b*# |
| CYPs | 162.34 ± 14.73 a | 173.37 ± 8.8 a | 176.39 ± 11.27 a |
| Diosgenin | 154.44 ± 18.69 a | 183.58 ± 18.14 a | 208.64 ± 29.98 a |
| Taxifolin | 163.79 ± 9.98 a | 173.72 ± 10.25 a | 236.65 ± 13.83 a*# |
| Treatment | Fermentation Time (h) | ||
|---|---|---|---|
| 0 | 12 | 24 | |
| Control | 21.46 ± 0.62 a | 21.00 ± 0.31 bc | 21.80 ± 1.07 ab |
| CY | 20.26 ± 0.96 a | 20.05 ± 0.63 ab | 23.09 ± 0.14 b*# |
| CYPs | 20.88 ± 1.13 a | 19.19 ± 0.41 a | 20.50 ± 0.29 a |
| Diosgenin | 22.09 ± 0.64 a | 21.20 ± 0.11 bc | 22.95 ± 0.18 b# |
| Taxifolin | 21.77 ± 0.48 a | 22.22 ± 0.72 c | 22.77 ± 0.73 b |
| Treatment | Fermentation Time (h) | ||
|---|---|---|---|
| 0 | 12 | 24 | |
| Control | 98.69 ± 0.38 ab | 98.06 ± 3.96 a | 95.09 ± 1.55 a |
| CY | 101.86 ± 1.62 b | 85.70 ± 7.36 a | 86.25 ± 5.25 a |
| CYPs | 95.31 ± 1.63 a | 93.13 ± 1.86 a | 93.09 ± 1.70 a |
| Diosgenin | 95.31 ± 1.37 a | 96.43 ± 0.19 a | 93.67 ± 0.25 a |
| Taxifolin | 94.93 ± 2.03 a | 91.29± 0.56 a | 88.48 ± 1.28 a* |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cui, Y.; Zhou, Y.; Li, Y.; Wang, J.; Li, D.; Chen, F. Chinese Yam and Its Active Components Regulate the Structure of Gut Microbiota and Indole-like Metabolites in Anaerobic Fermentation In Vitro. Nutrients 2023, 15, 5112. https://doi.org/10.3390/nu15245112
Cui Y, Zhou Y, Li Y, Wang J, Li D, Chen F. Chinese Yam and Its Active Components Regulate the Structure of Gut Microbiota and Indole-like Metabolites in Anaerobic Fermentation In Vitro. Nutrients. 2023; 15(24):5112. https://doi.org/10.3390/nu15245112
Chicago/Turabian StyleCui, Yifan, Yingzhuo Zhou, Yan Li, Jieying Wang, Daotong Li, and Fang Chen. 2023. "Chinese Yam and Its Active Components Regulate the Structure of Gut Microbiota and Indole-like Metabolites in Anaerobic Fermentation In Vitro" Nutrients 15, no. 24: 5112. https://doi.org/10.3390/nu15245112
APA StyleCui, Y., Zhou, Y., Li, Y., Wang, J., Li, D., & Chen, F. (2023). Chinese Yam and Its Active Components Regulate the Structure of Gut Microbiota and Indole-like Metabolites in Anaerobic Fermentation In Vitro. Nutrients, 15(24), 5112. https://doi.org/10.3390/nu15245112






