The Role of Magnesium in the Pathogenesis of Metabolic Disorders
Abstract
1. Introduction
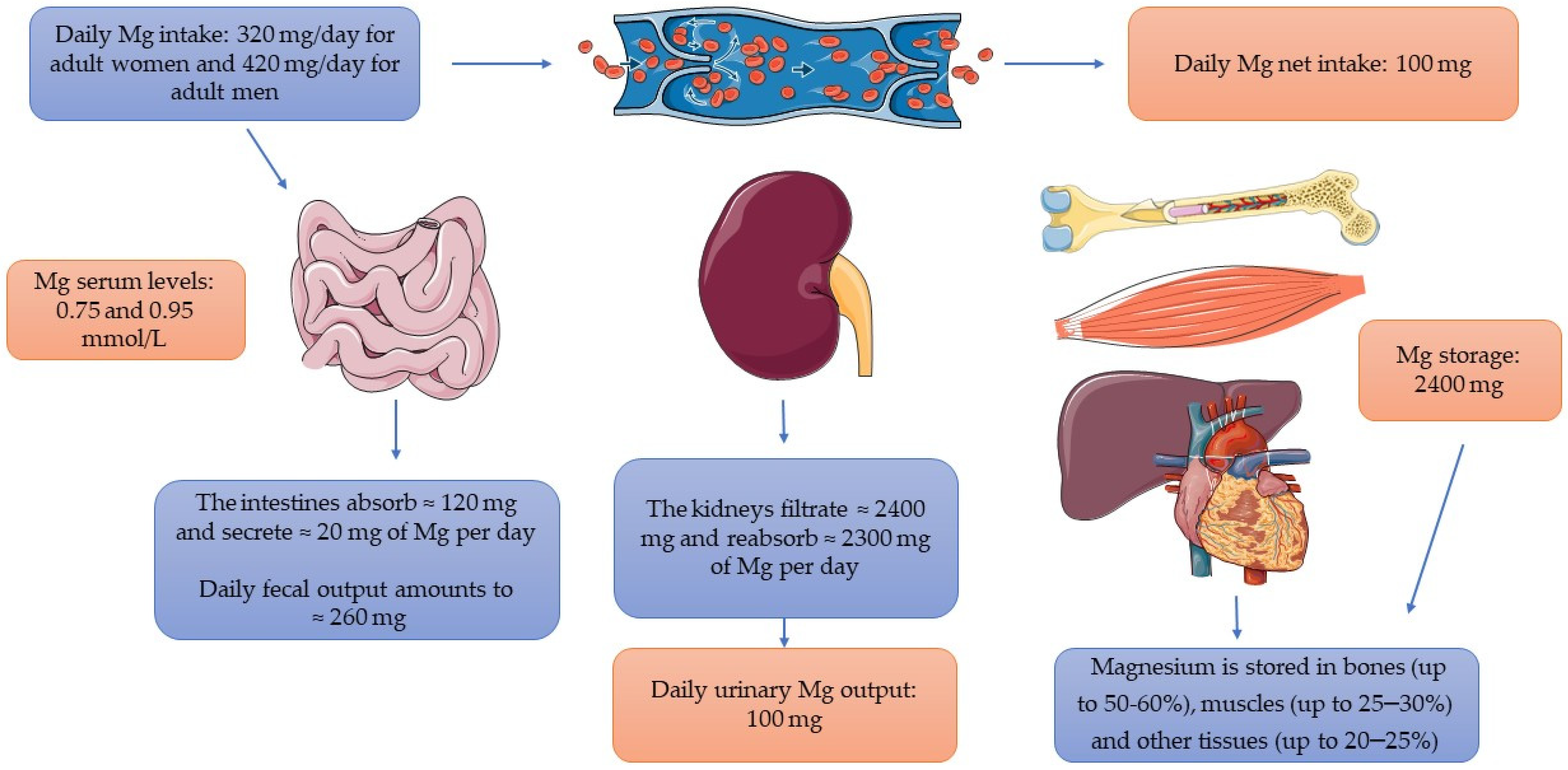
2. The Assessment of Magnesium Status
| Serum Mg Concentration | 0.75–0.95 mmol/L [12] |
| Methods of Mg status evaluation in human body | Mg serum concentration RBC Mg levels Mg urine levels Intravenous or oral Mg loading tests Non-invasive Intracellular Mineral-Electrolyte Analysis Hair mineral analysis test Using isotopic Mg labels Ionized serum Mg concentration Serum Mg/Ca ratio [6,15,16] |
| Mg recommended daily allowance | Children aged 1–3: 80 mg/day Children aged 4–8: 130 mg/day Adolescents aged 9–13: 240 mg/day Girls aged 13–18: 360 mg/day Boys aged 13–18: 410 mg/day Adult women: 320 mg/day Adult men: 420 mg/day [3] |
| Mg deficiency symptoms | Early signs of Mg deficiency: fatigue, weakness, loss of appetite, nausea or vomiting Advanced Mg deficiency: tremor, agitation and muscle fasciculation, cramps, seizures, cardiac arrhythmia, ventricular tachycardia, personality changes or depression [25] |
| Mg food sources | Almonds, bananas, black beans, green vegetables (spinach, broccoli), nuts, oatmeal, seeds, brown rice, unprocessed cereals, soybeans, sweet corn, tofu, and dark chocolate [15,26] |
3. Magnesium Deficiency
4. An Association of Magnesium with Excessive Body Weight
5. Magnesium and Hypertension
6. Magnesium and Diabetes and Metabolic Syndrome
7. Magnesium and Dyslipidemia
8. Magnesium and Inflammation
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schwalfenberg, G.K.; Genuis, S.J. The Importance of Magnesium in Clinical Healthcare. Scientifica 2017, 2017, 4179326. [Google Scholar] [CrossRef] [PubMed]
- Gröber, U.; Schmidt, J.; Kisters, K. Magnesium in Prevention and Therapy. Nutrients 2015, 7, 8199–8226. [Google Scholar] [CrossRef] [PubMed]
- The National Academies Press. Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride; The National Academies Press: Washington, DC, USA, 1997. [Google Scholar]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on Dietary Reference Values for Magnesium: Dietary Reference Values for Magnesium. EFSA J. 2015, 13, 4186. [Google Scholar] [CrossRef]
- Nielsen, F.H.; Johnson, L.A.K. Data from Controlled Metabolic Ward Studies Provide Guidance for the Determination of Status Indicators and Dietary Requirements for Magnesium. Biol. Trace Elem. Res. 2017, 177, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Workinger, J.L.; Doyle, R.P.; Bortz, J. Challenges in the Diagnosis of Magnesium Status. Nutrients 2018, 10, 1202. [Google Scholar] [CrossRef] [PubMed]
- Alawi, A.; Majoni, A.M.; Falhammar, S.W. Magnesium and Human Health: Perspectives and Research Directions. Int. J. Endocrinol. 2018, 2018, 9041694. [Google Scholar] [CrossRef]
- Fang, X.; Wang, K.; Han, D.; He, X.; Wei, J.; Zhao, L.; Imam, M.U.; Ping, Z.; Li, Y.; Xu, Y.; et al. Dietary Magnesium Intake and the Risk of Cardiovascular Disease, Type 2 Diabetes, and All-Cause Mortality: A Dose–Response Meta-Analysis of Prospective Cohort Studies. BMC Med. 2016, 14, 210. [Google Scholar] [CrossRef]
- Gobbo, D.; Imamura, L.C.; Wu, F.; Otto, J.H.Y.; Chiuve, M.C.D.O. Circulating and Dietary Magnesium and Risk of Cardiovascular Disease: A Systematic Review and Meta-Analysis of Prospective Studies. Am. J. Clin. Nutr. 2013, 98, 160–173. [Google Scholar] [CrossRef]
- Piuri, G.; Zocchi, M.; Della Porta, M.; Ficara, V.; Manoni, M.; Zuccotti, G.V.; Pinotti, L.; Maier, J.A.; Cazzola, R. Magnesium in Obesity, Metabolic Syndrome, and Type 2 Diabetes. Nutrients 2021, 13, 320. [Google Scholar] [CrossRef]
- Shechter, M. Magnesium and Cardiovascular System. Magnes. Res. 2010, 23, 60–72. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; O’Keefe, J.H.; Wilson, W. Subclinical Magnesium Deficiency: A Principal Driver of Cardiovascular Disease and a Public Health Crisis. Open Heart 2018, 5, e000668. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, F.H. Guidance for the Determination of Status Indicators and Dietary Requirements for Magnesium. Magnes. Res. 2016, 29, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Costello, R.B.; Elin, R.J.; Rosanoff, A.; Wallace, T.C.; Guerrero-Romero, F.; Hruby, A.; Lutsey, P.L.; Nielsen, F.H.; Rodriguez-Moran, M.; Song, Y.; et al. Perspective: The Case for an Evidence-Based Reference Interval for Serum Magnesium: The Time Has Come. Adv. Nutr. 2016, 7, 977–993. [Google Scholar] [CrossRef] [PubMed]
- Razzaque, M.S. Magnesium: Are We Consuming Enough? Nutrients 2018, 10, 1863. [Google Scholar] [CrossRef]
- Arnaud, M.J. Update on the Assessment of Magnesium Status. Br. J. Nutr. 2008, 99 (Suppl. S3), S24–S36. [Google Scholar] [CrossRef]
- Basso, L.E.; Ubbink, J.B.; Delport, R. Erythrocyte Magnesium Concentration as an Index of Magnesium Status: A Perspective from a Magnesium Supplementation Study. Clin. Chim. Acta 2000, 291, 1–8. [Google Scholar] [CrossRef]
- Zhang, X.; Del Gobbo, L.C.; Hruby, A.; Rosanoff, A.; He, K.; Dai, Q.; Costello, R.B.; Zhang, W.; Song, Y. The Circulating Concentration and 24-h Urine Excretion of Magnesium Dose- and Time-Dependently Respond to Oral Magnesium Supplementation in a Meta-Analysis of Randomized Controlled Trials. J. Nutr. 2016, 146, 595–602. [Google Scholar] [CrossRef]
- Gullestad, L.; Midtvedt, K.; Dolva, L.O.; Norseth, J.; Kjekshus, J. The Magnesium Loading Test: Reference Values in Healthy Subjects. Scand. J. Clin. Lab. Investig. 1994, 54, 23–31. [Google Scholar] [CrossRef]
- Ozono, R.; Oshima, T.; Matsuura, H.; Higashi, Y.; Ishida, T.; Watanabe, M.; Yoshimura, M.; Hiraga, H.; Ono, N.; Kajiyama, G. Systemic Magnesium Deficiency Disclosed by Magnesium Loading Test in Patients with Essential Hypertension. Hypertens. Res. 1995, 18, 39–42. [Google Scholar] [CrossRef]
- Fawcett, W.J.; Haxby, E.J.; Male, D.A. Magnesium: Physiology and Pharmacology. Br. J. Anaesth. 1999, 83, 302–320. [Google Scholar] [CrossRef]
- Hébert, P.; Mehta, N.; Wang, J.; Hindmarsh, T.; Jones, G.; Cardinal, P. Functional Magnesium Deficiency in Critically Ill Patients Identified Using a Magnesium-Loading Test. Crit. Care Med. 1997, 25, 749–755. [Google Scholar] [CrossRef] [PubMed]
- Koch, S.M.; Warters, R.D.; Mehlhorn, U. The Simultaneous Measurement of Ionized and Total Calcium and Ionized and Total Magnesium in Intensive Care Unit Patients. J. Crit. Care 2002, 17, 203–205. [Google Scholar] [CrossRef] [PubMed]
- Rosanoff, A.; Wolf, F.I. A Guided Tour of Presentations at the XIV International Magnesium Symposium. Magnes. Res. 2016, 29, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Gröber, U. Micronutrients: Metabolic Tuning-Prevention-Therapy; MedPharm Scientific Publishers: Stuttgart, Germany, 2009. [Google Scholar]
- Jahnen-Dechent, W.; Ketteler, M. Magnesium Basics. Clin. Kidney J. 2012, 5 (Suppl. S1), i3–i14. [Google Scholar] [CrossRef] [PubMed]
- Olza, J.; Aranceta-Bartrina, J.; González-Gross, M.; Ortega, R.; Serra-Majem, L.; Varela-Moreiras, G.; Gil, Á. Reported Dietary Intake, Disparity between the Reported Consumption and the Level Needed for Adequacy and Food Sources of Calcium, Phosphorus, Magnesium and Vitamin D in the Spanish Population: Findings from the ANIBES Study. Nutrients 2017, 9, 168. [Google Scholar] [CrossRef]
- Rosanoff, A.; Weaver, C.M.; Rude, R.K. Suboptimal Magnesium Status in the United States: Are the Health Consequences Underestimated? Nutr. Rev. 2012, 70, 153–164. [Google Scholar] [CrossRef]
- Cazzola, R.; Della Porta, M.; Manoni, M.; Iotti, S.; Pinotti, L.; Maier, J.A. Going to the Roots of Reduced Magnesium Dietary Intake: A Tradeoff between Climate Changes and Sources. Heliyon 2020, 6, e05390. [Google Scholar] [CrossRef]
- Costello, R.B.; Nielsen, F. Interpreting Magnesium Status to Enhance Clinical Care: Key Indicators. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 504–511. [Google Scholar] [CrossRef]
- Mejía-Rodríguez, F.; Shamah-Levy, T.; Villalpando, S.; García-Guerra, A.; Méndez-Gómez Humarán, I. Iron, Zinc, Copper and Magnesium Deficiencies in Mexican Adults from the National Health and Nutrition Survey 2006. Salud Publica Mex. 2013, 55, 275–284. [Google Scholar] [CrossRef]
- Mousavi, E.; Ghoreishy, S.; Hemmati, S.M.; Mohammadi, A. Association between Magnesium Concentrations and Prediabetes: A Systematic Review and Meta-Analysis. Sci. Rep. 2021, 11, 24388. [Google Scholar] [CrossRef]
- Kumar, S.; Shraddha, J.; Sachin, A.; Honmode, A. Impact of Serum Magnesium Levels in Critically Ill Elderly Patients—A Study in a Rural Teaching Hospital. J. Clin. Gerontol. Geriatr. 2016, 7, 104–108. [Google Scholar] [CrossRef]
- de Baaij, J.H.F.; Hoenderop, J.G.J.; Bindels, R.J.M. Regulation of Magnesium Balance: Lessons Learned from Human Genetic Disease. Clin. Kidney J. 2012, 5 (Suppl. S1), i15–i24. [Google Scholar] [CrossRef] [PubMed]
- van der Wijst, J.; Bindels, R.J.M.; Hoenderop, J.G.J. Mg2+ Homeostasis: The Balancing Act of TRPM6: The Balancing Act of TRPM6. Curr. Opin. Nephrol. Hypertens. 2014, 23, 361–369. [Google Scholar] [CrossRef] [PubMed]
- de Baaij, J.H.F.; Hoenderop, J.G.J.; Bindels, R.J.M. Magnesium in Man: Implications for Health and Disease. Physiol. Rev. 2015, 95, 1–46. [Google Scholar] [CrossRef]
- Zittermann, A. Magnesium Deficit? Overlooked Cause of Low Vitamin D Status? BMC Med. 2013, 11, 229. [Google Scholar] [CrossRef]
- Den, X.; Song, Y.; Manson, J.E.; Signorello, L.B.; Zhang, S.M.; Shrubsole, M.J.; Ness, R.M.; Seidner, D.L.; Dai, Q. Magnesium, Vitamin D Status and Mortality: Results from US National Health and Nutrition Examination Survey (NHANES) 2001 to 2006 and NHANES III. BMC Med. 2013, 11, 187. [Google Scholar]
- Vetter, T.; Lohse, M.J. Magnesium and the Parathyroid. Curr. Opin. Nephrol. Hypertens. 2002, 11, 403–410. [Google Scholar] [CrossRef]
- Hansen, B.A.; Bruserud, Ø. Hypomagnesemia in Critically Ill Patients. J. Intensive Care 2018, 6, 21. [Google Scholar] [CrossRef]
- Pham, P.C.T.; Pham, P.A.T.; Pham, S.V.; Pham, P.T.T.; Pham, P.M.T.; Pham, P.T.T. Hypomagnesemia: A Clinical Perspective. Int. J. Nephrol. Renovasc. Dis. 2014, 7, 219–230. [Google Scholar] [CrossRef]
- Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 21 March 2022).
- Nam, G.E.; Park, H.S. Perspective on Diagnostic Criteria for Obesity and Abdominal Obesity in Korean Adults. J. Obes. Metab. Syndr. 2018, 27, 134–142. [Google Scholar] [CrossRef]
- Marra, M.; Sammarco, R.; De Lorenzo, A.; Iellamo, F.; Siervo, M.; Pietrobelli, A.; Donini, L.M.; Santarpia, L.; Cataldi, M.; Pasanisi, F.; et al. Assessment of Body Composition in Health and Disease Using Bioelectrical Impedance Analysis (BIA) and Dual Energy X-ray Absorptiometry (DXA): A Critical Overview. Contrast Media Mol. Imaging 2019, 2019, 3548284. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, P.; Letois, F.; Sultan, A.; Nocca, D.; Mura, T.; Galtier, F. Nutrient Deficiencies in Patients with Obesity Considering Bariatric Surgery: A Cross-Sectional Study. Surg. Obes. Relat. Dis. 2014, 10, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Babapour, M.; Mohammadi, H.; Kazemi, M.; Hadi, A.; Rezazadegan, M.; Askari, G. Associations between Serum Magnesium Concentrations and Polycystic Ovary Syndrome Status: A Systematic Review and Meta-Analysis. Biol. Trace Elem. Res. 2021, 199, 1297–1305. [Google Scholar] [CrossRef] [PubMed]
- Shamnani, G.; Rukadikar, C.; Gupta, V.; Singh, S.; Tiwari, S.; Bhartiy, S.; Sharma, P. Serum Magnesium in Relation with Obesity. Natl. J. Physiol. Pharm. Pharmacol. 2018, 8, 1074–1077. [Google Scholar] [CrossRef]
- Hassan, S.A.U.; Ahmed, I.; Nasrullah, A.; Haq, S.; Ghazanfar, H.; Sheikh, A.B.; Zafar, R.; Askar, G.; Hamid, Z.; Khushdil, A.; et al. Comparison of Serum Magnesium Levels in Overweight and Obese Children and Normal Weight Children. Cureus 2017, 9, e1607. [Google Scholar] [CrossRef]
- Jiang, S.; Ma, X.; Li, M.; Yan, S.; Zhao, H.; Pan, Y.; Wang, C.; Yao, Y.; Jin, L.; Li, B. Association between Dietary Mineral Nutrient Intake, Body Mass Index, and Waist Circumference in U.S. Adults Using Quantile Regression Analysis NHANES 2007–2014. PeerJ 2020, 8, e9127. [Google Scholar] [CrossRef]
- Lu, L.; Chen, C.; Yang, K.; Zhu, J.; Xun, P.; Shikany, J.M.; He, K. Magnesium Intake Is Inversely Associated with Risk of Obesity in a 30-Year Prospective Follow-up Study among American Young Adults. Eur. J. Nutr. 2020, 59, 3745–3753. [Google Scholar] [CrossRef]
- Guerrero-Romero, F.; Morales-Gurrola, G.; Preza-Rodríguez, L.; Gómez-Barrientos, A.; Olivas-Martínez, A.I.; Simental-Mendía, L.E. Magnesium Intake Is Associated with the Metabolically Healthy Obese Phenotype. J. Investig. Med. 2022, 70, 800–804. [Google Scholar] [CrossRef]
- Naseeb, M.; Milliron, B.J.; Bruneau, M.L., Jr.; Sukumar, D.; Foster, G.D.; Smith, S.A.; Volpe, S.L. Dietary Magnesium Intake in Relation to Body Mass Index and Glycemic Indices in Middle School Students from the Healthy Study. Nutr. Health 2021, 27, 211–219. [Google Scholar] [CrossRef]
- Castellanos-Gutiérrez, A.; Sánchez-Pimienta, T.G.; Carriquiry, A.; da Costa, T.H.M.; Ariza, A.C. Higher Dietary Magnesium Intake Is Associated with Lower Body Mass Index, Waist Circumference and Serum Glucose in Mexican Adults. Nutr. J. 2018, 17, 114. [Google Scholar] [CrossRef]
- Dominguez, L.J.; Gea, A.; Ruiz-Estigarribia, L.; Sayón-Orea, C.; Fresán, U.; Barbagallo, M.; Ruiz-Canela, M.; Martinez-Gonzalez, M.A. Low Dietary Magnesium and Overweight/Obesity in a Mediterranean Population: A Detrimental Synergy for the Development of Hypertension. The SUN Project. Nutrients 2020, 13, 125. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.K.; Bae, Y.J. Association of Magnesium Intake with High Blood Pressure in Korean Adults: Korea National Health and Nutrition Examination Survey 2007–2009. PLoS ONE 2015, 10, e0130405. [Google Scholar] [CrossRef] [PubMed]
- Huitrón-Bravo, G.G.; Denova-Gutiérrez, E.; de Jesús Garduño-García, J.; Talavera, J.O.; Herreros, B.; Salmerón, J. Dietary Magnesium Intake and Risk of Hypertension in a Mexican Adult Population: A Cohort Study. BMC Nutr. 2015, 1, 6. [Google Scholar] [CrossRef]
- Chacko, S.A.; Song, Y.; Nathan, L.; Tinker, L.; de Boer, I.H.; Tylavsky, F.; Wallace, R.; Liu, S. Relations of Dietary Magnesium Intake to Biomarkers of Inflammation and Endothelial Dysfunction in an Ethnically Diverse Cohort of Postmenopausal Women. Diabetes Care 2010, 33, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Ma, X.; Liang, H.; Li, H.; Chen, J.; Fang, L.; Yang, Q.; Zhang, Z. Dietary Magnesium Intake Affects the Association between Serum Vitamin D and Type 2 Diabetes: A Cross-Sectional Study. Front. Nutr. 2021, 8, 763076. [Google Scholar] [CrossRef] [PubMed]
- Gant, C.M.; Soedamah-Muthu, S.S.; Binnenmars, S.H.; Bakker, S.J.L.; Navis, G.; Laverman, G.D. Higher Dietary Magnesium Intake and Higher Magnesium Status Are Associated with Lower Prevalence of Coronary Heart Disease in Patients with Type 2 Diabetes. Nutrients 2018, 10, 307. [Google Scholar] [CrossRef]
- Hruby, A.; Guasch-Ferré, M.; Bhupathiraju, S.N.; Manson, J.E.; Willett, W.C.; McKeown, N.M.; Hu, F.B. Magnesium Intake, Quality of Carbohydrates, and Risk of Type 2 Diabetes: Results from Three U.s. Cohorts. Diabetes Care 2017, 40, 1695–1702. [Google Scholar] [CrossRef]
- Konishi, K.; Wada, K.; Tamura, T.; Tsuji, M.; Kawachi, T.; Nagata, C. Dietary Magnesium Intake and the Risk of Diabetes in the Japanese Community: Results from the Takayama Study. Eur. J. Nutr. 2017, 56, 767–774. [Google Scholar] [CrossRef]
- Huang, J.H.; Lu, Y.F.; Cheng, F.C.; Lee, J.N.Y.; Tsai, L.C. Correlation of Magnesium Intake with Metabolic Parameters, Depression and Physical Activity in Elderly Type 2 Diabetes Patients: A Cross-Sectional Study. Nutr. J. 2012, 11, 41. [Google Scholar] [CrossRef]
- Kim, D.J.; Xun, P.; Liu, K.; Loria, C.; Yokota, K.; Jacobs, D.R., Jr.; He, K. Magnesium Intake in Relation to Systemic Inflammation, Insulin Resistance, and the Incidence of Diabetes. Diabetes Care 2010, 33, 2604–2610. [Google Scholar] [CrossRef]
- Jin, H.; Nicodemus-Johnson, J. Gender and Age Stratified Analyses of Nutrient and Dietary Pattern Associations with Circulating Lipid Levels Identify Novel Gender and Age-Specific Correlations. Nutrients 2018, 10, 1760. [Google Scholar] [CrossRef] [PubMed]
- Bain, L.K.M.; Myint, P.K.; Jennings, A.; Lentjes, M.A.H.; Luben, R.N.; Khaw, K.T.; Wareham, N.J.; Welch, A.A. The Relationship between Dietary Magnesium Intake, Stroke and Its Major Risk Factors, Blood Pressure and Cholesterol, in the EPIC-Norfolk Cohort. Int. J. Cardiol. 2015, 196, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Yamori, Y.; for the CARDIAC Study Group; Sagara, M.; Mizushima, S.; Liu, L.; Ikeda, K.; Nara, Y. An Inverse Association between Magnesium in 24-h Urine and Cardiovascular Risk Factors in Middle-Aged Subjects in 50 CARDIAC Study Populations. Hypertens. Res. 2015, 38, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Mirmiran, P.; Shab-Bidar, S.; Hosseini-Esfahani, F.; Asghari, G.; Hosseinpour-Niazi, S.; Azizi, F. Magnesium Intake and Prevalence of Metabolic Syndrome in Adults: Tehran Lipid and Glucose Study. Public Health Nutr. 2012, 15, 693–701. [Google Scholar] [CrossRef]
- Ohira, T.; Peacock, J.M.; Iso, H.; Chambless, L.E.; Rosamond, W.D.; Folsom, A.R. Serum and Dietary Magnesium and Risk of Ischemic Stroke: The Atherosclerosis Risk in Communities Study. Am. J. Epidemiol. 2009, 169, 1437–1444. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Folsom, A.R.; Melnick, S.L.; Eckfeldt, J.H.; Sharrett, A.R.; Nabulsi, A.A.; Hutchinson, R.G.; Metcalf, P.A. Associations of Serum and Dietary Magnesium with Cardiovascular Disease, Hypertension, Diabetes, Insulin, and Carotid Arterial Wall Thickness: The Aric Study. J. Clin. Epidemiol. 1995, 48, 927–940. [Google Scholar] [CrossRef]
- Singh, R.B.; Rastogi, S.S.; Sharma, V.K.; Saharia, R.B.; Kulshretha, S.K. Can Dietary Magnesium Modulate Lipoprotein Metabolism? Magnes. Trace Elem. 1990, 9, 255–264. [Google Scholar] [PubMed]
- Arablou, T.; Aryaeian, N.; Djalali, M.; Shahram, F.; Rasouli, L. Association between Dietary Intake of Some Antioxidant Micronutrients with Some Inflammatory and Antioxidant Markers in Active Rheumatoid Arthritis Patients. Int. J. Vitam. Nutr. Res. 2019, 89, 238–245. [Google Scholar] [CrossRef]
- King, D.E.; Mainous, A.G., 3rd; Geesey, M.E.; Ellis, T. Magnesium Intake and Serum C-Reactive Protein Levels in Children. Magnes. Res. 2007, 20, 32–36. [Google Scholar]
- Song, Y.; Li, T.Y.; van Dam, R.M.; Manson, J.E.; Hu, F.B. Magnesium Intake and Plasma Concentrations of Markers of Systemic Inflammation and Endothelial Dysfunction in Women. Am. J. Clin. Nutr. 2007, 85, 1068–1074. [Google Scholar] [CrossRef]
- Bo, S.; Durazzo, M.; Guidi, S.; Carello, M.; Sacerdote, C.; Silli, B.; Rosato, R.; Cassader, M.; Gentile, L.; Pagano, G. Dietary Magnesium and Fiber Intakes and Inflammatory and Metabolic Indicators in Middle-Aged Subjects from a Population-Based Cohort. Am. J. Clin. Nutr. 2006, 84, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- King, D.E.; Mainous, A.G., III; Geesey, M.E.; Woolson, R.F. Dietary Magnesium and C-Reactive Protein Levels. J. Am. Coll. Nutr. 2005, 24, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Farhanghi, M.A.; Mahboob, S.; Ostadrahimi, A. Obesity Induced Magnesium Deficiency Can Be Treated by Vitamin D Supplementation. J. Pak. Med. Assoc. 2009, 59, 258–261. [Google Scholar] [PubMed]
- Peuhkuri, K.; Vapaatalo, H.; Korpela, R. Even Low-Grade Inflammation Impacts on Small Intestinal Function. World J. Gastroenterol. 2010, 16, 1057–1062. [Google Scholar] [CrossRef]
- Givens, M.H. Studies in calcium and magnesium metabolism, III: The effect of fat and fatty acid derivatives. J. Biol. Chem. 1917, 3, 441–444. [Google Scholar] [CrossRef]
- Drenick, E.J. The influence of ingestion of calcium and other soap-forming substances on fecal fat. Gastroenterology 1961, 41, 242–244. [Google Scholar] [CrossRef]
- Devaux, S.; Adrian, M.; Laurant, P.; Berthelot, A.; Quignard-Boulangé, A. Dietary Magnesium Intake Alters Age-Related Changes in Rat Adipose Tissue Cellularity. Magnes. Res. 2016, 29, 175–183. [Google Scholar] [CrossRef]
- Cinti, S.; Mitchell, G.; Barbatelli, G.; Murano, I.; Ceresi, E.; Faloia, E.; Wang, S.; Fortier, M.; Greenberg, A.S.; Obin, M.S. Adipocyte Death Defines Macrophage Localization and Function in Adipose Tissue of Obese Mice and Humans. J. Lipid Res. 2005, 46, 2347–2355. [Google Scholar] [CrossRef]
- Dibaba, D.T.; Xun, P.; He, K. Dietary Magnesium Intake Is Inversely Associated with Serum C-Reactive Protein Levels: Meta-Analysis and Systematic Review. Eur. J. Clin. Nutr. 2014, 68, 510–516. [Google Scholar] [CrossRef]
- Guerrero-Romero, F.; Bermudez-Peña, C.; Rodríguez-Morán, M. Severe Hypomagnesemia and Low-Grade Inflammation in Metabolic Syndrome. Magnes. Res. 2011, 24, 45–53. [Google Scholar] [CrossRef]
- Mazur, A.; Maier, J.A.M.; Rock, E.; Gueux, E.; Nowacki, W.; Rayssiguier, Y. Magnesium and the Inflammatory Response: Potential Physiopathological Implications. Arch. Biochem. Biophys. 2007, 458, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Tejero-Taldo, M.I.; Kramer, J.H.; Mak, I.T.; Komarov, A.M.; Weglicki, W.B. The Nerve-Heart Connection in the pro-Oxidant Response to Mg-Deficiency. Heart Fail. Rev. 2006, 11, 35–44. [Google Scholar] [CrossRef]
- Calder, P.C. N-3 Polyunsaturated Fatty Acids, Inflammation, and Inflammatory Diseases. Am. J. Clin. Nutr. 2006, 83 (Suppl. S6), 1505S–1519S. [Google Scholar] [CrossRef] [PubMed]
- Tejero-Taldo, M.I.; Chmielinska, J.J.; Gonzalez, G.; Mak, I.T.; Weglicki, W.B. N-Methyl-D-Aspartate Receptor Blockade Inhibits Cardiac Inflammation in the Mg2+ Deficient Rat. J. Pharmacol. Exp. Therapeut. 2004, 311, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Zhu, X.; Manson, J.E.; Song, Y.; Li, X.; Franke, A.A.; Costello, R.B.; Rosanoff, A.; Nian, H.; Fan, L.; et al. Magnesium Status and Supplementation Influence Vitamin D Status and Metabolism: Results from a Randomized Trial. Am. J. Clin. Nutr. 2018, 108, 1249–1258. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, W.; Li, D.; Yin, X.; Zhang, X.; Olsen, N.; Zheng, S.G. Vitamin D and Chronic Diseases. Aging Dis. 2017, 8, 346–353. [Google Scholar] [CrossRef]
- Stokic, E.; Romani, A.; Ilincic, B.; Kupusinac, A.; Stosic, Z.; Isenovic, E.R. Chronic Latent Magnesium Deficiency in Obesity Decreases Positive Effects of Vitamin D on Cardiometabolic Risk Indicators. Curr. Vasc. Pharmacol. 2017, 15, 610–617. [Google Scholar] [CrossRef]
- Askari, M.; Mozaffari, H.; Jafari, A.; Ghanbari, M.; Darooghegi Mofrad, M. The Effects of Magnesium Supplementation on Obesity Measures in Adults: A Systematic Review and Dose-Response Meta-Analysis of Randomized Controlled Trials. Crit. Rev. Food Sci. Nutr. 2021, 61, 2921–2937. [Google Scholar] [CrossRef]
- Rafiee, M.; Ghavami, A.; Rashidian, A.; Hadi, A.; Askari, G. The Effect of Magnesium Supplementation on Anthropometric Indices: A Systematic Review and Dose-Response Meta-Analysis of Clinical Trials. Br. J. Nutr. 2021, 125, 644–656. [Google Scholar] [CrossRef]
- Asbaghi, O.; Hosseini, R.; Boozari, B.; Ghaedi, E.; Kashkooli, S.; Moradi, S. The Effects of Magnesium Supplementation on Blood Pressure and Obesity Measure among Type 2 Diabetes Patient: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Biol. Trace Elem. Res. 2021, 199, 413–424. [Google Scholar] [CrossRef]
- Corre, T.; Arjona, F.J.; Hayward, C.; Youhanna, S.; de Baaij, J.H.F.; Belge, H.; Nägele, N.; Debaix, H.; Blanchard, M.G.; Traglia, M.; et al. Genome-Wide Meta-Analysis Unravels Interactions between Magnesium Homeostasis and Metabolic Phenotypes. J. Am. Soc. Nephrol. 2018, 29, 335–348. [Google Scholar] [CrossRef] [PubMed]
- Dibaba, D.T.; Xun, P.; Song, Y.; Rosanoff, A.; Shechter, M.; He, K. The Effect of Magnesium Supplementation on Blood Pressure in Individuals with Insulin Resistance, Prediabetes, or Noncommunicable Chronic Diseases: A Meta-Analysis of Randomized Controlled Trials. Am. J. Clin. Nutr. 2017, 106, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, Y.; Del Gobbo, L.C.; Rosanoff, A.; Wang, J.; Zhang, W.; Song, Y. Effects of Magnesium Supplementation on Blood Pressure: A Meta-Analysis of Randomized Double-Blind Placebo-Controlled Trials: A Meta-Analysis of Randomized Double-Blind Placebo-Controlled Trials. Hypertension 2016, 68, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Kass, L.; Weekes, J.; Carpenter, L. Effect of Magnesium Supplementation on Blood Pressure: A Meta-Analysis. Eur. J. Clin. Nutr. 2012, 66, 411–418. [Google Scholar] [CrossRef]
- Veronese, N.; Dominguez, L.J.; Pizzol, D.; Demurtas, J.; Smith, L.; Barbagallo, M. Oral Magnesium Supplementation for Treating Glucose Metabolism Parameters in People with or at Risk of Diabetes: A Systematic Review and Meta-Analysis of Double-Blind Randomized Controlled Trials. Nutrients 2021, 13, 4074. [Google Scholar] [CrossRef]
- Verma, H.; Garg, R. Effect of Magnesium Supplementation on Type 2 Diabetes Associated Cardiovascular Risk Factors: A Systematic Review and Meta-Analysis. J. Hum. Nutr. Diet. 2017, 30, 621–633. [Google Scholar] [CrossRef]
- Veronese, N.; Watutantrige-Fernando, S.; Luchini, C.; Solmi, M.; Sartore, G.; Sergi, G.; Manzato, E.; Barbagallo, M.; Maggi, S.; Stubbs, B. Effect of Magnesium Supplementation on Glucose Metabolism in People with or at Risk of Diabetes: A Systematic Review and Meta-Analysis of Double-Blind Randomized Controlled Trials. Eur. J. Clin. Nutr. 2016, 70, 1354–1359. [Google Scholar] [CrossRef]
- Tan, X.; Huang, Y. Magnesium Supplementation for Glycemic Status in Women with Gestational Diabetes: A Systematic Review and Meta-Analysis. Gynecol. Endocrinol. 2022, 38, 202–206. [Google Scholar] [CrossRef]
- Asbaghi, O.; Moradi, S.; Nezamoleslami, S.; Moosavian, S.P.; Hojjati Kermani, M.A.; Lazaridi, A.V.; Miraghajani, M. The Effects of Magnesium Supplementation on Lipid Profile among Type 2 Diabetes Patients: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Biol. Trace Elem. Res. 2021, 199, 861–873. [Google Scholar] [CrossRef]
- Simental-Mendía, L.E.; Simental-Mendía, M.; Sahebkar, A.; Rodríguez-Morán, M.; Guerrero-Romero, F. Effect of Magnesium Supplementation on Lipid Profile: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Eur. J. Clin. Pharmacol. 2017, 73, 525–536. [Google Scholar] [CrossRef]
- Song, Y.; He, K.; Levitan, E.B.; Manson, J.E.; Liu, S. Effects of Oral Magnesium Supplementation on Glycaemic Control in Type 2 Diabetes: A Meta-Analysis of Randomized Double-Blind Controlled Trials: Review Article. Diabet. Med. 2006, 23, 1050–1056. [Google Scholar] [CrossRef]
- Talebi, S.; Miraghajani, M.; Hosseini, R.; Mohammadi, H. The Effect of Oral Magnesium Supplementation on Inflammatory Biomarkers in Adults: A Comprehensive Systematic Review and Dose–Response Meta-Analysis of Randomized Clinical Trials. Biol. Trace Elem. Res. 2022, 200, 1538–1550. [Google Scholar] [CrossRef] [PubMed]
- Veronese, N.; Pizzol, D.; Smith, L.; Dominguez, L.J.; Barbagallo, M. Effect of Magnesium Supplementation on Inflammatory Parameters: A Meta-Analysis of Randomized Controlled Trials. Nutrients 2022, 14, 679. [Google Scholar] [CrossRef] [PubMed]
- Mazidi, M.; Rezaie, P.; Banach, M. Effect of Magnesium Supplements on Serum C-Reactive Protein: A Systematic Review and Meta-Analysis. Arch. Med. Sci. 2018, 14, 707–716. [Google Scholar] [CrossRef]
- Simental-Mendia, L.E.; Sahebkar, A.; Rodriguez-Moran, M.; Zambrano-Galvan, G.; Guerrero-Romero, F. Effect of Magnesium Supplementation on Plasma C-Reactive Protein Concentrations: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Curr. Pharm. Des. 2017, 23, 4678–4686. [Google Scholar] [CrossRef]
- Kjeldsen, S.E. Hypertension and Cardiovascular Risk: General Aspects. Pharmacol. Res. 2018, 129, 95–99. [Google Scholar] [CrossRef]
- Hypertension. Available online: https://www.who.int/news-room/fact-sheets/detail/hypertension (accessed on 22 March 2022).
- Han, H.; Fang, X.; Wei, X.; Liu, Y.; Jin, Z.; Chen, Q.; Fan, Z.; Aaseth, J.; Hiyoshi, A.; He, J.; et al. Dose-Response Relationship between Dietary Magnesium Intake, Serum Magnesium Concentration and Risk of Hypertension: A Systematic Review and Meta-Analysis of Prospective Cohort Studies. Nutr. J. 2017, 16, 26. [Google Scholar] [CrossRef] [PubMed]
- Rotter, I.; Kosik-Bogacka, D.; Dołęgowska, B.; Safranow, K.; Karakiewicz, B.; Laszczyńska, M. Relationship between Serum Magnesium Concentration and Metabolic and Hormonal Disorders in Middle-Aged and Older Men. Magnes. Res. 2015, 28, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Xun, P.; Tang, Q.; Cai, W.; He, K. Circulating Magnesium Levels and Incidence of Coronary Heart Diseases, Hypertension, and Type 2 Diabetes Mellitus: A Meta-Analysis of Prospective Cohort Studies. Nutr. J. 2017, 16, 60. [Google Scholar] [CrossRef]
- Khan, A.M.; Sullivan, L.; McCabe, E.; Levy, D.; Vasan, R.S.; Wang, T.J. Lack of Association between Serum Magnesium and the Risks of Hypertension and Cardiovascular Disease. Am. Heart J. 2010, 160, 715–720. [Google Scholar] [CrossRef]
- Darooghegi Mofrad, M.; Djafarian, K.; Mozaffari, H.; Shab-Bidar, S. Effect of Magnesium Supplementation on Endothelial Function: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Atherosclerosis 2018, 273, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Kisters, K.; Wessels, F.; Küper, H.; Tokmak, F.; Krefting, E.R.; Gremmler, B.; Kosch, M.; Barenbrock, M.; Hausberg, M. Increased Calcium and Decreased Magnesium Concentrations and an Increased Calcium/Magnesium Ratio in Spontaneously Hypertensive Rats versus Wistar-Kyoto Rats: Relation to Arteriosclerosis. Am. J. Hypertens. 2004, 17, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Kisters, K.; Wessels, F.; Tokmak, F.; Krefting, E.R.; Gremmler, B.; Kosch, M.; Hausberg, M. Early-Onset Increased Calcium and Decreased Magnesium Concentrations and an Increased Calcium/Magnesium Ratio in SHR versus WKY. Magnes. Res. 2004, 17, 264–269. [Google Scholar] [PubMed]
- Romani, A.M.P. Beneficial Role of Mg2+ in Prevention and Treatment of Hypertension. Int. J. Hypertens. 2018, 2018, 9013721. [Google Scholar] [CrossRef]
- Goytain, A.; Quamme, G.A. Functional Characterization of Human SLC41A1, a Mg2+ Transporter with Similarity to Prokaryotic MgtE Mg2+ Transporters. Physiol. Genom. 2005, 21, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, R.; Tabata, S.; Shindo, Y.; Hotta, K.; Suzuki, K.; Soga, T.; Oka, K. Mitochondrial Mg2+ Homeostasis Decides Cellular Energy Metabolism and Vulnerability to Stress. Sci. Rep. 2016, 6, 30027. [Google Scholar] [CrossRef]
- Mastrototaro, L.; Smorodchenko, A.; Aschenbach, J.R.; Kolisek, M.; Sponder, G. Solute Carrier 41A3 Encodes for a Mitochondrial Mg2+ Efflux System. Sci. Rep. 2016, 6, 27999. [Google Scholar] [CrossRef]
- Liu, M.; Jeong, E.-M.; Liu, H.; Xie, A.; So, E.Y.; Shi, G.; Jeong, G.E.; Zhou, A.; Dudley, S.C., Jr. Magnesium Supplementation Improves Diabetic Mitochondrial and Cardiac Diastolic Function. JCI Insight 2019, 4, e123182. [Google Scholar] [CrossRef]
- Liu, M.; Dudley, S.C., Jr. Magnesium, Oxidative Stress, Inflammation, and Cardiovascular Disease. Antioxidants 2020, 9, 907. [Google Scholar] [CrossRef]
- Palmer, B.F.; Clegg, D.J. Electrolyte and Acid-Base Disturbances in Patients with Diabetes Mellitus. N. Engl. J. Med. 2015, 373, 548–559. [Google Scholar] [CrossRef]
- Feng, J.; Wang, H.; Jing, Z.; Wang, Y.; Cheng, Y.; Wang, W.; Sun, W. Role of Magnesium in Type 2 Diabetes Mellitus. Biol. Trace Elem. Res. 2020, 196, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Bertinato, J.; Wang, K.; Hayward, S. Serum Magnesium Concentrations in the Canadian Population and Associations with Diabetes, Glycemic Regulation, and Insulin Resistance. Nutrients 2017, 9, 296. [Google Scholar] [CrossRef] [PubMed]
- Kieboom, B.C.T.; Ligthart, S.; Dehghan, A.; Kurstjens, S.; de Baaij, J.H.F.; Franco, O.H.; Hofman, A.; Zietse, R.; Stricker, B.H.; Hoorn, E.J. Serum Magnesium and the Risk of Prediabetes: A Population-Based Cohort Study. Diabetologia 2017, 60, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Han, H.; Li, M.; Liang, C.; Fan, Z.; Aaseth, J.; He, J.; Montgomery, S.; Cao, Y. Dose-Response Relationship between Dietary Magnesium Intake and Risk of Type 2 Diabetes Mellitus: A Systematic Review and Meta-Regression Analysis of Prospective Cohort Studies. Nutrients 2016, 8, 739. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Zeng, L.; Zhao, J.; Wu, Q.; Dong, Y.; Zou, F.; Gan, L.; Wei, Y.; Zhang, W. Association of Magnesium Intake with Type 2 Diabetes and Total Stroke: An Updated Systematic Review and Meta-Analysis. BMJ Open 2020, 10, e032240. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.Y.; Xun, P.; He, K.; Qin, L.Q. Magnesium Intake and Risk of Type 2 Diabetes. Diabetes Care 2011, 34, 2116–2122. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Wolk, A. Magnesium Intake and Risk of Type 2 Diabetes: A Meta-Analysis. J. Intern. Med. 2007, 262, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Sarrafzadegan, N.; Khosravi-Boroujeni, H.; Lotfizadeh, M.; Pourmogaddas, A.; Salehi-Abargouei, A. Magnesium Status and the Metabolic Syndrome: A Systematic Review and Meta-Analysis. Nutrition 2016, 32, 409–417. [Google Scholar] [CrossRef]
- Ju, S.Y.; Choi, W.S.; Ock, S.M.; Kim, C.M.; Kim, D.H. Dietary Magnesium Intake and Metabolic Syndrome in the Adult Population: Dose-Response Meta-Analysis and Meta-Regression. Nutrients 2014, 6, 6005–6019. [Google Scholar] [CrossRef]
- Barbagallo, M.; Dominguez, L.J. Magnesium and Type 2 Diabetes. World J. Diabetes 2015, 6, 1152–1157. [Google Scholar] [CrossRef]
- Ramadass, S.; Basu, S.; Srinivasan, A.R. SERUM Magnesium Levels as an Indicator of Status of Diabetes Mellitus Type 2. Diabetes Metab. Syndr. 2015, 9, 42–45. [Google Scholar] [CrossRef] [PubMed]
- Gommers, L.M.M.; Hoenderop, J.G.J.; Bindels, R.J.M.; de Baaij, J.H.F. Hypomagnesemia in Type 2 Diabetes: A Vicious Circle? Diabetes 2016, 65, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, C.; Agoramoorthy, G.; Hsu, M.J. Exploring the Evolutionary Relationship of Insulin Receptor Substrate Family Using Computational Biology. PLoS ONE 2011, 6, e16580. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Rodelo, C.; Roura-Guiberna, A.; Olivares-Reyes, J.A. Molecular mechanisms of insulin resistance: An update. Gac. Med. Mex. 2017, 153, 214–228. [Google Scholar] [PubMed]
- Crescenzo, R.; Bianco, F.; Mazzoli, A.; Giacco, A.; Liverini, G.; Iossa, S. Mitochondrial Efficiency and Insulin Resistance. Front. Physiol. 2014, 5, 512. [Google Scholar] [CrossRef]
- Kostov, K. Effects of Magnesium Deficiency on Mechanisms of Insulin Resistance in Type 2 Diabetes: Focusing on the Processes of Insulin Secretion and Signaling. Int. J. Mol. Sci. 2019, 20, 1351. [Google Scholar] [CrossRef]
- Günther, T. The Biochemical Function of Mg2+ in Insulin Secretion, Insulin Signal Transduction and Insulin Resistance. Magnes. Res. 2010, 23, 5–18. [Google Scholar] [CrossRef]
- Ashcroft, F.M.; Puljung, M.C.; Vedovato, N. Neonatal Diabetes and the KATP Channel: From Mutation to Therapy. Trends Endocrinol. Metab. 2017, 28, 377–387. [Google Scholar] [CrossRef]
- Libby, P.; Buring, J.E.; Badimon, L.; Hansson, G.K.; Deanfield, J.; Bittencourt, M.S.; Tokgözoğlu, L.; Lewis, E.F. Atherosclerosis. Nat. Rev. Dis. Primers 2019, 5, 56. [Google Scholar] [CrossRef]
- Benjamin, E.J.; Virani, S.S.; Callaway, C.W.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Chiuve, S.E.; Cushman, M.; Delling, F.N.; Deo, R.; et al. Heart Disease and Stroke Statistics-2018 Update: A Report from the American Heart Association. Circulation 2018, 137, e67–e492. [Google Scholar] [CrossRef]
- Gimbrone, M.A., Jr.; García-Cardeña, G. Endothelial Cell Dysfunction and the Pathobiology of Atherosclerosis. Circ. Res. 2016, 118, 620–636. [Google Scholar] [CrossRef] [PubMed]
- Ference, B.A.; Ginsberg, H.N.; Graham, I.; Ray, K.K.; Packard, C.J.; Bruckert, E.; Hegele, R.A.; Krauss, R.M.; Raal, F.J.; Schunkert, H.; et al. Low-Density Lipoproteins Cause Atherosclerotic Cardiovascular Disease. 1. Evidence from Genetic, Epidemiologic, and Clinical Studies. A Consensus Statement from the European Atherosclerosis Society Consensus Panel. Eur. Heart J. 2017, 38, 2459–2472. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Mittal, R.; Ramgobin, D.; Jain, R.; Jain, R. Current Management Guidelines on Hyperlipidemia: The Silent Killer. J. Lipids 2021, 2021, 9883352. [Google Scholar] [CrossRef]
- Guerrero-Romero, F.; Rodríguez-Morán, M. Low Serum Magnesium Levels and Metabolic Syndrome. Acta Diabetol. 2002, 39, 209–213. [Google Scholar] [CrossRef]
- Bersohn, I.; Oelofse, P.J. Correlation of Serum-Magnesium and Serum-Cholesterol Levels in South African Bantu and European Subjects. Lancet 1957, 269, 1020–1021. [Google Scholar] [CrossRef]
- Rodrigues, A.K.; Melo, A.E.; Domingueti, C.P. Association between Reduced Serum Levels of Magnesium and the Presence of Poor Glycemic Control and Complications in Type 1 Diabetes Mellitus: A Systematic Review and Meta-Analysis. Diabetes Metab. Syndr. 2020, 14, 127–134. [Google Scholar] [CrossRef]
- Yu, L.; Zhang, J.; Wang, L.; Li, S.; Zhang, Q.; Xiao, P.; Wang, K.; Zhuang, M.; Jiang, Y. Association between Serum Magnesium and Blood Lipids: Influence of Type 2 Diabetes and Central Obesity. Br. J. Nutr. 2018, 120, 250–258. [Google Scholar] [CrossRef]
- Randell, E.W.; Mathews, M.; Gadag, V.; Zhang, H.; Sun, G. Relationship between Serum Magnesium Values, Lipids and Anthropometric Risk Factors. Atherosclerosis 2008, 196, 413–419. [Google Scholar] [CrossRef]
- Shahbah, D.; El Naga, A.A.; Hassan, T.; Zakaria, M.; Beshir, M.; Al Morshedy, S.; Abdalhady, M.; Kamel, E.; Rahman, D.A.; Kamel, L.; et al. Status of Serum Magnesium in Egyptian Children with Type 1 Diabetes and Its Correlation to Glycemic Control and Lipid Profile. Medicine 2016, 95, e5166. [Google Scholar] [CrossRef]
- Rodríguez-Ortiz, M.E.; Gómez-Delgado, F.; Arenas de Larriva, A.P.; Canalejo, A.; Gómez-Luna, P.; Herencia, C.; López-Moreno, J.; Rodríguez, M.; López-Miranda, J.; Almadén, Y. Serum Magnesium Is Associated with Carotid Atherosclerosis in Patients with High Cardiovascular Risk (CORDIOPREV Study). Sci. Rep. 2019, 9, 8013. [Google Scholar] [CrossRef]
- Khatami, M.R.; Mirchi, E.; Khazaeipour, Z.; Abdollahi, A.; Jahanmardi, A. Association between Serum Magnesium and Risk Factors of Cardiovascular Disease in Hemodialysis Patients. Iran. J. Kidney Dis. 2013, 7, 47–52. [Google Scholar] [PubMed]
- Barragán, R.; Llopis, J.; Portolés, O.; Sorlí, J.V.; Coltell, O.; Rivas-García, L.; Asensio, E.M.; Ortega-Azorín, C.; Corella, D.; Sánchez-González, C. Influence of Demographic and Lifestyle Variables on Plasma Magnesium Concentrations and Their Associations with Cardiovascular Risk Factors in a Mediterranean Population. Nutrients 2020, 12, 1018. [Google Scholar] [CrossRef] [PubMed]
- Na, W.; Chung, B.; Sohn, C. A Relationship between Dietary Patterns and Dyslipidemia in Urban-Dwelling Middle-Aged Korean Men: Using Korean Genome and Epidemiology Study (KoGES). Clin. Nutr. Res. 2019, 8, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Olatunji, L.A.; Oyeyipo, I.P.; Micheal, O.S.; Soladoye, A.O. Effect of Dietary Magnesium on Glucose Tolerance and Plasma Lipid during Oral Contraceptive Administration in Female Rats. Afr. J. Med. Med. Sci. 2008, 37, 135–139. [Google Scholar]
- Rosanoff, A.; Seelig, M.S. Comparison of Mechanism and Functional Effects of Magnesium and Statin Pharmaceuticals. J. Am. Coll. Nutr. 2004, 23, 501S–505S. [Google Scholar] [CrossRef]
- Zhang, Q.; Qian, Z.Y.; Zhou, P.H.; Zhou, X.L.; Zhang, D.L.; He, N.; Zhang, J.; Liu, Y.H.; Gu, Q. Effects of Oral Selenium and Magnesium Co-Supplementation on Lipid Metabolism, Antioxidative Status, Histopathological Lesions, and Related Gene Expression in Rats Fed a High-Fat Diet. Lipids Health Dis. 2018, 17, 165. [Google Scholar] [CrossRef]
- Rashvand, S.; Mobasseri, M.; Tarighat-Esfanjani, A. Effects of Choline and Magnesium Concurrent Supplementation on Coagulation and Lipid Profile in Patients with Type 2 Diabetes Mellitus: A Pilot Clinical Trial. Biol. Trace Elem. Res. 2020, 194, 328–335. [Google Scholar] [CrossRef]
- Jamilian, M.; Samimi, M.; Faraneh, A.E.; Aghadavod, E.; Shahrzad, H.D.; Chamani, M.; Mafi, A.; Asemi, Z. Magnesium Supplementation Affects Gene Expression Related to Insulin and Lipid in Patients with Gestational Diabetes. Magnes. Res. 2017, 30, 71–79. [Google Scholar] [CrossRef]
- Vallim, T.; Salter, A.M. Regulation of Hepatic Gene Expression by Saturated Fatty Acids. Prostaglandins Leukot. Essent. Fat. Acids 2010, 82, 211–218. [Google Scholar] [CrossRef]
- Wegner, M.; Araszkiewicz, A.; Zozulinska-Ziolkiewicz, D.; Wierusz-Wysocka, B.; Piorunska-Mikolajczak, A.; Piorunska-Stolzmann, M. The Relationship between Concentrations of Magnesium and Oxidized Low Density Lipoprotein and the Activity of Platelet Activating Factor Acetylhydrolase in the Serum of Patients with Type 1 Diabetes. Magnes. Res. 2010, 23, 97–104. [Google Scholar]
- Găman, M.A.; Dobrică, E.C.; Cozma, M.A.; Antonie, N.I.; Stănescu, A.M.A.; Găman, A.M.; Diaconu, C.C. Crosstalk of Magnesium and Serum Lipids in Dyslipidemia and Associated Disorders: A Systematic Review. Nutrients 2021, 13, 1411. [Google Scholar] [CrossRef] [PubMed]
- Rifai, N.; Ridker, P.M. Population Distributions of C-Reactive Protein in Apparently Healthy Men and Women in the United States: Implication for Clinical Interpretation. Clin. Chem. 2003, 49, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Giugliano, D. The Metabolic Syndrome and Inflammation: Association or Causation? Nutr. Metab. Cardiovasc. Dis. 2004, 14, 228–232. [Google Scholar] [CrossRef]
- Calder, P.C.; Ahluwalia, N.; Albers, R.; Bosco, N.; Bourdet-Sicard, R.; Haller, D.; Holgate, S.T.; Jönsson, L.S.; Latulippe, M.E.; Marcos, A.; et al. Consideration of Biomarkers to Be Used for Evaluation of Inflammation in Human Nutritional Studies. Br. J. Nutr. 2013, 109 (Suppl. S1), S1–S34. [Google Scholar] [CrossRef]
- Ortega-Gómez, A.; Perretti, M.; Soehnlein, O. Resolution of Inflammation: An Integrated View: Resolution of Inflammation. EMBO Mol. Med. 2013, 5, 661–674. [Google Scholar] [CrossRef]
- Shahi, A.; Aslani, S.; Ataollahi, M.; Mahmoudi, M. The Role of Magnesium in Different Inflammatory Diseases. Inflammopharmacology 2019, 27, 649–661. [Google Scholar] [CrossRef]
- Scanlan, B.J.; Tuft, B.; Elfrey, J.E.; Smith, A.; Zhao, A.; Morimoto, M.; Chmielinska, J.J.; Tejero-Taldo, M.I.; Mak, I.T.; Weglicki, W.B.; et al. Intestinal Inflammation Caused by Magnesium Deficiency Alters Basal and Oxidative Stress-Induced Intestinal Function. Mol. Cell. Biochem. 2007, 306, 59–69. [Google Scholar] [CrossRef]
- Almoznino-Sarafian, D.; Berman, S.; Mor, A.; Shteinshnaider, M.; Gorelik, O.; Tzur, I.; Alon, I.; Modai, D.; Cohen, N. Magnesium and C-Reactive Protein in Heart Failure: An Anti-Inflammatory Effect of Magnesium Administration? Eur. J. Nutr. 2007, 46, 230–237. [Google Scholar] [CrossRef]
- Qu, X.; Jin, F.; Hao, Y.; Li, H.; Tang, T.; Wang, H.; Yan, W.; Dai, K. Magnesium and the Risk of Cardiovascular Events: A Meta-Analysis of Prospective Cohort Studies. PLoS ONE 2013, 8, e57720. [Google Scholar] [CrossRef]
- Oliveira, A.R.S.d.; Cruz, K.J.C.; Severo, J.S.; Morais, J.B.S.; Freitas, T.E.C.d.; Araújo, R.S.; Marreiro, D.D.N. Hypomagnesemia and Its Relation with Chronic Low-Grade Inflammation in Obesity. Rev. Assoc. Med. Bras. 2017, 63, 156–163. [Google Scholar] [CrossRef]
- Rodriguez-Morán, M.; Guerrero-Romero, F. Elevated Concentrations of TNF-Alpha Are Related to Low Serum Magnesium Levels in Obese Subjects. Magnes. Res. 2004, 17, 189–196. [Google Scholar] [PubMed]
- Rayssiguier, Y.; Libako, P.; Nowacki, W.; Rock, E. Magnesium Deficiency and Metabolic Syndrome: Stress and Inflammation May Reflect Calcium Activation. Magnes Res. 2010, 23, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Mahalle, N.; Garg, M.K.; Kulkarni, M.V.; Naik, S.S. Relation of Magnesium with Insulin Resistance and Inflammatory Markers in Subjects with Known Coronary Heart Disease. J. Cariovasc. Dis. Res. 2014, 5, 22–29. [Google Scholar]
- Rodriguez-Morán, M.; Guerrero-Romero, F. Serum Magnesium and C-Reactive Protein Levels. Arch. Dis. Child. 2008, 93, 676–680. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, F.H.; Johnson, L.K.; Zeng, H. Magnesium Supplementation Improves Indicators of Low Magnesium Status and Inflammatory Stress in Adults Older than 51 Years with Poor Quality Sleep. Magnes. Res. 2010, 23, 158–168. [Google Scholar] [CrossRef]
- Almousa, L.A.; Salter, A.M.; Langley-Evans, S.C. Magnesium Deficiency Heightens Lipopolysaccharide-Induced Inflammation and Enhances Monocyte Adhesion in Human Umbilical Vein Endothelial Cells. Magnes. Res. 2018, 31, 39–48. [Google Scholar] [CrossRef]
- Romani, A.M.P. Magnesium in Health and Disease. In Metal Ions in Life Sciences; Springer: Dordrecht, The Netherlands, 2013; pp. 49–79. [Google Scholar]
- King, D.E. Inflammation and Elevation of C-Reactive Protein: Does Magnesium Play a Key Role? Magnes. Res. 2009, 22, 57–59. [Google Scholar] [CrossRef]
- Kawashima, S.; Yokoyama, M. Dysfunction of Endothelial Nitric Oxide Synthase and Atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 998–1005. [Google Scholar] [CrossRef]
- Salvemini, D.; Doyle, T.M.; Cuzzocrea, S. Superoxide, Peroxynitrite and Oxidative/Nitrative Stress in Inflammation. Biochem. Soc. Trans. 2006, 34, 965–970. [Google Scholar] [CrossRef]
- Zheltova, A.A.; Kharitonova, M.V.; Iezhitsa, I.N.; Spasov, A.A. Magnesium Deficiency and Oxidative Stress: An Update. BioMedicine 2016, 6, 20. [Google Scholar] [CrossRef]
- Barbagallo, M.; Belvedere, M.; Dominguez, L.J. Magnesium Homeostasis and Aging. Magnes. Res. 2009, 22, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, M.; Dominguez, L.J.; Tagliamonte, M.R.; Resnick, L.M.; Paolisso, G. Effects of Glutathione on Red Blood Cell Intracellular Magnesium: Relation to Glucose Metabolism: Relation to Glucose Metabolism. Hypertension 1999, 34, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Boparai, R.K.; Kiran, R.; Bansal, D.D. Insinuation of Exacerbated Oxidative Stress in Sucrose-Fed Rats with a Low Dietary Intake of Magnesium: Evidence of Oxidative Damage to Proteins. Free Radic. Res. 2007, 41, 981–989. [Google Scholar] [CrossRef] [PubMed]
- Cazzaniga, A.; Locatelli, L.; Castiglioni, S.; Maier, J. The Contribution of EDF1 to PPARγ Transcriptional Activation in VEGF-Treated Human Endothelial Cells. Int. J. Mol. Sci. 2018, 19, 1830. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, G.; Fiorucci, S. PPARs and Other Nuclear Receptors in Inflammation. Curr. Opin. Pharmacol. 2006, 6, 421–427. [Google Scholar] [CrossRef]
- Locatelli, L.; Fedele, G.; Castiglioni, S.; Maier, J.A. Magnesium Deficiency Induces Lipid Accumulation in Vascular Endothelial Cells via Oxidative Stress-the Potential Contribution of EDF-1 and PPARγ. Int. J. Mol. Sci. 2021, 22, 1050. [Google Scholar] [CrossRef]
- Mukohda, M.; Stump, M.; Ketsawatsomkron, P.; Hu, C.; Quelle, F.W.; Sigmund, C.D. Endothelial PPAR-γ Provides Vascular Protection from IL-1β-Induced Oxidative Stress. Am. J. Physiol. Heart Circ. Physiol. 2016, 310, H39–H48. [Google Scholar] [CrossRef]
- Simental-Mendía, L.E.; Rodríguez-Morán, M.; Guerrero-Romero, F. Oral Magnesium Supplementation Decreases C-Reactive Protein Levels in Subjects with Prediabetes and Hypomagnesemia: A Clinical Randomized Double-Blind Placebo-Controlled Trial. Arch. Med. Res. 2014, 45, 325–330. [Google Scholar] [CrossRef]

| Malabsorption | Crohn’s disease, ulcerative colitis, coeliac disease, short bowel syndrome, Whipple’s disease, chronic diarrhea, pancreatic insufficiency, inflammatory bowel diseases [2] |
| Endocrine disorders | Aldosteronism, hyperparathyroidism, hyperthyroidism, poorly-controlled diabetes [2,26] |
| Renal diseases | Chronic renal failure, dialysis, acute tubular necrosis, postobstructive diuresis, post kidney transplantation, excessive volume expansion, chronic metabolic acidosis [26,41] |
| Redistribution and intracellular shift | Refeeding syndrome, pregnancy, lactation, cardiopulmonary surgeries [41] |
| Medication use | Loop diuretics, aminoglycosides, amphotericin B, cyclosporine and tacrolimus, cisplatin, cetuximab, omeprazole, pentamidine [7,26] |
| Other causes | Inappropriate diet, chronic alcoholism, stress, severe burns [7,26] |
| Metabolic Disorder | Reference-Year | Study Type | Population | Effects |
|---|---|---|---|---|
| Obesity | Guerrero-Romero, F. et al., 2022 [51] | Cross-sectional study | Metabolically healthy obese (MHO) individuals n-124 Metabolically unhealthy obese (MUO) n-123 | The logistic regression analysis adjusted by sex and age showed that Mg intake is significantly associated with the MHO phenotype (OR = 1.17; 95% CI 1.07 to 1.25, p = 0.005) |
| Naseeb, M. et al., 2021 [52] | Randomized, cluster-design study The HEALTHY Study | Ethnically diverse students (10–14 years) n-2181 | Mg intake was related to BMI percentile at baseline and at end of the study (β = −0.05, 95% CI = −0.02 to 0, p = 0.04; β = −0.06, 95% CI = −0.02 to −0.003, p = 0.004); R2 (regression coefficient effect size) = 0.03; R 2 = 0.06) Mg intake was not related to plasma insulin and glucose concentrations | |
| Jiang, S. et al., 2020 [49] | Cross-sectional study National Health and Nutrition Examination Survey (NHANES) 2007–2014 | Adult individuals (≥20 years) n-19,952 | Mg intakes were negatively correlated with BMI (p < 0.05 at the quantiles of 0.1–0.9) and WC (p < 0.05 at 0.1–0.9 quantiles) after adjusting for age and gender | |
| Lu, L. et al., 2020 [50] | Multicenter longitudinal cohort study (30-year follow-up) The Coronary Artery Risk Development in Young Adults (CARDIA) | American young adults (18–30 years) n-5115 | Compared to the lowest quintile (Q1) of Mg intake level, the incidence of obesity was reduced by 51% among participants in the highest quintile (Q5) [HR = 0.49, 95% CI = (0.40, 0.60), p for trend < 0.01] | |
| Castellanos-Gutiérrez, A. et al., 2018 [53] | Population-based multistage probabilistic study Mexican National Health and Nutrition Survey 2012 | Adult individuals (20–65 years) n-1573 | Increase in 10 mg per 1000 kcal/day of Mg was associated with an average decrease in BMI of 0.72% (95% CI: −1.36, − 0.08) and 0.49 cm (95% CI: −0.92, − 0.07) of WC An increase in Mg intake was associated with an average decrease in serum glucose by 0.59% (95% CI: −1.08, − 0.09) | |
| Hypertension | Dominguez, L.J. et al., 2020 [54] | Prospective study The SUN Project (Seguimiento Universidad de Navarra) | Mediterranean population n-14,057 | Dietary Mg intake < 200 mg/day was independently associated with a higher risk of developing high blood pressure, especially in overweight/obese participants |
| Choi, M.-K. et al., 2015 [55] | Cross-sectional studyKorean National Health and Nutritional Examination Survey data (2007–2009) | Adults participants (20 years and over) n-11,685 | No significant association between dietary Mg intake and the risk of HTN in obese women after adjusting relevant factors, the adjusted odds ratio of DBP prevalence in the highest magnesium intake quartile was 0.40 compared with the lowest magnesium intake quartile (95% CI = 0.25–0.63, p for trend = 0.0014) | |
| Huitrón-Bravo, G.G. et al., 2015 [56] | Cohort study Health Workers Cohort Study | Mexican adult subjects n-1378 | Trend of decreasing DBP with rising Mg intake, by tertiles (the coefficients were −0.75 mmHg [95% confidence interval (CI): −1.77, 0.27], −1.27 mmHg (95% CI: −2.73, −0.02; p for trend = 0.01) was found In the fully adjusted model, Mg intake was inversely associated, although not significantly, with the risk of developing hypertension; subjects in the highest tertile of Mg intake had a decreased risk for HTN (odds ratio 0.83, 95% CI: 0.49–1.39, p for trend = 0.48) | |
| Chacko, S. A. et al., 2010 [57] | Cross-sectional study Women’s Health Initiative Observational Study | Postmenopausal women (50–79 years) n-3713 | An inverse relationship between Mg intake and plasma concentrations of soluble vascular cell adhesion molecule 1 (sVCAM-1) and E-selectin An increase of 100 mg Mg per day was inversely associated with sVCAM-1 (−0.04 ± 0.02 ng/mL; p = 0.07) and other markers of inflammation as hs-CRP, IL-6 or TNF-α | |
| Type 2 diabetes | Huang, W. et al., 2021 [58] | Cross-sectional study National Health and Nutrition Examination Survey-NHANES (2007–2014) | Adults participants n-10,249 | The association of serum vitamin D with the incidence of T2D appeared to differ between the low Mg intake group and the high Mg intake group (OR: 0.968, 95%Cl: 0.919–1.02 vs. OR: 0.925, 95%Cl: 0.883–0.97) There was evidence of interaction between vitamin D levels and Mg intake on decreasing the incidence of T2D (p-value for interaction = 0.001) |
| Gant, C. M. et al., 2018 [59] | Cross-sectional study DIAbetes and LifEstyle Cohort Twente-1 | T2DM patients n-450 (mean age 63 ± 9 years) diabetes duration of 11 (7–18) years) | Adjusted coronary heart disease (CHD) prevalence ratios for the highest compared to the lowest quartiles were 0.40 (0.20, 0.79) for Mg intake, 0.63 (0.32, 1.26) for 24 h urinary Mg excretion, and 0.62 (0.32, 1.20) for plasma Mg concentration For every 10 mg increase of Mg intake from vegetables, the prevalence of CHD was, statistically non-significantly, lower [0.75 (0.52, 1.08)] | |
| Hruby, A. et al., 2017 [60] | Prospective cohort study Results From Three U.S. Cohorts: The Nurses’ Health Study (1984–2012), NHS2 (1991–2013) and the Health Professionals’ Follow-Up Study (1986–2012) | Incident cases of T2DM over 28 years of follow-up n-17,130 | In pooled analyses across the three cohorts, those with the highest magnesium intake had 15% lower risk of type 2 diabetes compared with those with the lowest intake (pooled multivariate HR in quintile 5 vs. 1: 0.85 [95% CI 0.80–0.91], p < 0.0001). | |
| Konishi, K. et al., 2017 [61] | Population-based cohort study | Adults participants 13,525 | Compared with women in the low quartile of Mg intake, women in the high quartile were at a significantly reduced risk of diabetes (HR 0.50; 95% CI 0.30–0.84; p-trend 0.005) after adjustments for covariates. In men, there was no association between magnesium intake and the risk of diabetes | |
| Huang, J.H. et al., 2012 [62] | Cross-sectional study | T2DM patients (65 years and above) n-210 | Among type 2 diabetes patients (n-201), 88.6% had Mg intake under the RDA, while 37.1% had measurable hypomagnesemia. Moreover, Mg intake was associated with metabolic syndrome components, i.a., positively with high-density lipoprotein level (HDL, p = 0.005) and negatively with triglycerides (TG), WC, body fat percent and BMI (p < 0.005) | |
| Kim, D.J. et al., 2010 [63] | Prospective cohort study | Adult Americans (18–30 years) n-4497 | The multivariable-adjusted hazard ratio of diabetes for participants in the highest quintile of Mg intake was 0.53 (95% CI, 0.32–0.86; p-trend < 0.01) compared with those in the lowest quintile Mg intake was significantly inversely associated with hs-CRP, IL-6, fibrinogen, and HOMA-IR, and serum magnesium levels were inversely correlated with hs-CRP and HOMA-IR | |
| Dyslipidemia | Jin, H. et al., 2018 [64] | Retrospective National Health and Nutrition Examination Study (NHANES) 2001–2013 | Adult individuals n-12,284 | Dietary Mg was positively correlated with HDL concentration but negatively with the TC/HDL-C ratio in females |
| Bain. L. et al., 2015 [65] | cross-sectional study (European Prospective Investigation into Cancer (EPIC)-Norfolk) | Adult individuals (40–75 years) n-4443 | Inverse relationship between high dietary Mg intake (mean 456 mg/d) and serum TC (p-trend = 0.02 men and 0.04 women) | |
| Yamori, Y. et al., 2015 [66] | Cross-sectional analysis (World Health Organization-coordinated Cardiovascular Diseases and Alimentary Comparison (CARDIAC) Study (1985–1994) | Adult participants (48–56 years) n-4211 | Mg/creatinine (Cre) ratio was inversely associated with BMI, SBP, DBP, and TC (p for linear trend < 0.001 for each) | |
| Mirmiran, P. et al., 2012 [67] | Cross-sectional study Tehran Lipid and Glucose Study 2006–2008 | Tehran healthy adults (18–74 years) n-2504 | Higher dietary Mg was inversely correlated with TG level (p = 0.009), but there was no association with HDL (p = 0.44) Dietary Mg intake was inversely associated with FBG (p = 0.006) and WC (0.006) | |
| Ohira, T. et al., 2009 [68] | Cross-sectional study Atherosclerosis Risk in Communities Study cohort | Adult participants (45–64 years) n-14,221 | Higher Dietary Mg intake was inversely associated with LDL (p = 0.01) and positive correlated with HDL (p = 0.001) | |
| Ma, J. et al., 1995 [69] | Cross-sectional study (Atherosclerosis Risk in Communities (ARIC) Study) | Adult participants (45–64 years) n-15,248 | Dietary Mg intake was inversely associated with fasting serum insulin, HDL, SBP, DBP | |
| Singh, R.B. et al., 1990 [70] | RCTs | High-risk of CDV adults n-430 | Positive changes in TC, LDL, and TG (10.1%) and slight elevation in HDL in the Mg-rich diet group | |
| Low-grade inflammation | Arablou, T. et al., 2019 [71] | Cross-sectional study | Patients with active rheumatoid arthritis n-87 | Significant negative correlations were observed between Mg intake with PGE2 (R = −0.24)], IL-1β (R = −0.23), and IL-2 (R = −0.25) |
| King, D.E. et al., 2007 [72] | Cross-sectional, nationally representative National Health and Nutrition Examination Survey (NHANES 1999–2002) | Children (6–17 years) n-5007 | Insufficient dietary Mg intake (less than 75% the RDA) was associated with higher CRP (OR: 1.58, 95% CI: 1.07-infinity, p < 0.005) | |
| Song, Y. et al., 2007 [73] | Cross-sectional study(Nurses’ Health Study) | Women (43–69 years) n-657 | Mg intake was inversely associated with: CRP (p = 0.003), E-selectin (p = 0.001), and sICAM-1 (p = 0.03) Women in the highest quintile of dietary Mg intake were 24% lower for CRP (p = 0.03) and 14% lower for E-selectin (p for trend = 0.01) than those for women in the lowest quintile | |
| Bo, S. et al., 2006 [74] | Cross-sectional study | Adult subjects n-1653 | Prevalence of hs-CRP ≥ 3 mg/L were 3–4 times as likely in the lowest tertile of magnesium intakes | |
| King, D.E. et al., 2005 [75] | Cross-sectional study (NHANES 1999–2002) | Adult subjects ≥ 17 years) n-5773 | Insufficient dietary Mg intake (less than 50% of the RDA) was associated with higher CRP (95% CI: 1.13, 4.46) |
| Metabolic Disorder | Reference-Year | Study Type | Population | Effects |
|---|---|---|---|---|
| Obesity | Askari, M. et al., 2021 [91] | 32 RCTs | Adult participants n-2551 Doses: 48–450 mg/d Duration: 6–24 weeks | Mg supplementation resulted in a slight reduction in BMI (WMD: −0.21 kg/m2, 95% CI: −0.41, −0.001, p = 0.048) |
| Rafiee, M. et al., 2021 [92] | 28 RCTs | Adult participants n-2013 | No significant changes in anthropometric indicators after Mg supplementation in the overall analysis In the subgroup Mg supplementation decreases WC in obese individuals (twelve trials, n-997, WMD = −2.09 cm, 95% CI: –4.12, −0.07, p = 0.040; I2 = 0%) | |
| Asbaghi, O. et al., 2021 [93] | 11 RCTs | Patients with T2DM n-673 | Mg supplementation did not significantly change body weight (WMD: −0.01 kg, 95% CI: −0.36 to 0.33), BMI (WMD: −0.07, 95% CI: −0.18 to 0.04) or WC (WMD: 0.12, 95% CI: −1.24 to 1.48) Mg supplementation reduced the SBP (WMD: −5.78 mmHg, 95% CI: −11.37, −0.19) and DBP (WMD: −2.50 mmHg, 95% CI: −4.58, −0.41) in T2DM patients | |
| Hypertension | Dibaba, D.T., 2017 [95] | 11 RCTs | Individuals with insulin resistance, prediabetes or NCDs n-543 Median duration: 3.6 months Doses: 365–450 mg/day | Mg supplementation resulted in a mean reduction in SBP of 4.18 mmHg (standardized mean differences, SMD: −0.20; 95% CI: −0.37, −0.03) and 2.27 mmHg in DBP (SMD: −0.29; 95% CI: −0.46, −0.12) |
| Zhnag, X. et al., 2016 [96] | 34 RCTs | Normotensive and hypertensive adults n-2028 Duration: 3 months Doses: 368 mg/d | Mg supplementation significantly reduces SBP by 2.00 mmHg (95% CI: 0.43, 3.58) and DBP by 1.78 mmHg (95% CI 0.73, 2.82) along with increase in serum Mg concentration by 0.05 mmol/L (95% CI: 0.03, 0.07) | |
| Kass, L. et al., 2012 [97] | 22 RCTs | Adults participants n-1173 Duration: 3 to 24 weeks of follow-up Mean doses: 410 mg/d | A small reduction in SBP (0.32, 95% CI: 0.23, 0.41) and DBP (0.36, 95% CI: 0.27, 0.44) with a greater effect for the intervention in crossover trials (DBP 0.47, SBP 0.51) | |
| Diabetes | Veronese, N. et al., 2021 [98] | 25 RCTs | Diabetic participants (13 studies) n-361 Mg treatment n-359 placebo Duration: median of 12 weeks (range: 4–48) Participants at high risk of developing diabetes (12 studies) n-477 Mg treatment n-480 placebo Duration: median of 14 (range: 4–24) weeks | Treatment with Mg significantly reduced FBG in 325 participants with diabetes compared to 331 taking placebo (n = 11 studies; SMD = −0.426; 95%CI: −0.782 to −0.07; p = 0.02), this finding was characterized by a high heterogeneity (I2 = 79.0%) Mg supplementation did not improve HbA1c in 301 participants compared to 307 participants taking placebo (n = 10 studies; SMD = −0.134; 95%CI: −0.409 to 0.141; p = 0.34; I2 = 63.7%) Mg supplementation significantly improved FBG in 482 subjects at high risk of diabetes compared to 485 randomized to placebo (11 RCTs; SMD = −0.344; 95%CI: −0.655 to −0.03; p < 0.0001; I2 = 81.2%) Similarly, Mg significantly improved 2h OGTT in 3 studies involving 210 participants (SMD = −0.35; 95%CI: −0.62 to −0.07; I2 = 0%) Mg significantly decreases HOMA-IR in 9 studies (340 Mg vs. 344 placebo) (SMD = −0.234; 95%CI: −0.443 to −0.025; p = 0.028; I2 = 43.2%) |
| Verma, H. et al., 2017 [99] | 24 RCTs | Diabetic and non-diabetic individuals n-1694 | Significant improvement in: FBG (WMD) = −4.641 mg dL−1, 95% confidence interval (CI) = −7.602, −1.680, p = 0.002), HDL (WMD = 3.197 mg dL−1, 95% CI = 1.455, 4.938, p < 0.001), LDL (WMD = −10.668 mg dL−1, 95% CI = −19.108, −2.228, p = 0.013), TG(WMD = −15.323 mg dL−1, 95% CI = −28.821, −1.826, p = 0.026) SBP (WMD = −3.056 mmHg, 95% CI = −5.509, −0.603, p = 0.015). During subgroup analysis, a more beneficial effect of magnesium supplementation was observed in diabetic subjects with hypomagnesaemia | |
| Veronese, N. et al., 2016 [100] | 18 RCTs | Individuals with T2DM n-336 Mg treatment n-334 placebo People at high risk of diabetes n-226 Mg treatment n-227 placebo | Mg supplementation influence beneficial on glucose parameters in people with T2DM: reduced FBG (SMD: −0.40; 95% CI: −0.80, −0.00; I2 = 77%-9 studies) Mg supplementation influence beneficial on insulin sensitivity parameters in people at high risk of diabetes: improved FBG after a 2 h oral glucose tolerance test (SMD: −0.35; 95% CI: −0.62, −0.07; I2 = 0%-3 studies) and reductions in HOMA-IR (SMD: −0.57; 95% CI: −1.17, 0.03; I2 = 88%-5 studies) | |
| Dyslipidemia | Tan, X et al., 2022 [101] | 4 RCTs | Gestational diabetes n-ns Duration: 4–26 weeks Doses: 250–500 mg/d | Mg supplementation significantly reduced: FBG (SMD) = −0.99; 95% confidence interval (CI) = −1.28 to −0.70; p < 0.00001), serum insulin (SMD = −0.75; 95% CI = −1.24 to −0.26; p = 0.003), HOMA-IR (SMD = −0.74; 95% CI = −1.10 to −0.39; p < 0.0001) LDL (SMD = −0.39; 95% CI = −0.73 to −0.04; p = 0.03) TC (SMD = −0.62; 95% CI = −0.97 to −0.27; p = 0.0005) and increased quantitative insulin sensitivity check index (SMD = 0.47; 95% CI = 0.12 to 0.82; p = 0.008). |
| Asbaghi, O. et al., 2021 [102] | 12 RCTs | Patients with T2DM n-677 | Significant reduction in serum LDL levels (p = 0.006) with no effect on TG, TC, and LDL Effect of Mg supplementation might be dose- and duration-dependent: Mg supplementation lasting >12 weeks led to decreasing the serum TC (p = 0.002), Mg in dose <300 mg/day significantly decreased the serum LDL (p < 0.001), Mg in dose >300 mg/day markedly increased the serum HDL levels (p = 0.026) Inorganic Mg supplementation (vs. organic Mg) led to improvement in LDL (p < 0.001) and TC (p = 0.003) | |
| Simental-Mendía, L.E. et al., 2017 [103] | 18 RCTs | Diabetic and non-diabetic adults n-1192 Duration: 2–6 months Doses: 300–730 mg/d | No significant effect on: TC (WMD 0.03 mmol/L, 95% CI −0.11, 0.16, p = 0.671), LDL (WMD −0.01 mmol/L, 95% CI −0.13, 0.11, p = 0.903), HDL (WMD 0.03 mmol/L, 95% CI −0.003, 0.06, p = 0.076), TG (WMD −0.10 mmol/L, 95% CI −0.25, 0.04, p = 0.149). | |
| Song, Y. et al., 2006 [104] | 9 RCTs | Patients with T2DM n-370 Duration: 4–16 weeks Median doses: 360 mg/day | Mg supplementation increased HDL levels (WMD: 0.08 mmol/L (95% CI: 0.03, 0.14); p for heterogeneity = 0.36) but had no effect on TC, LDL and TG Lower FBG (WMD: −0.56 mmol/l (95% CI, −1.10 to −0.01); p for heterogeneity = 0.02) with no effect on HbA1c [−0.31% (95% CI, −0.81 to 0.19); p for heterogeneity = 0.10], SBP, DBP. | |
| Low-grade inflammation | Talebi, S. et al., 2022 [105] | 18 RCTs | Adult participants n-927 Duration: 4–26 weeks Doses: 20–500 mg/d | Mg supplementation had no statistically significant effect on serum CRP (WMD,−0.49; 95% CI,−1.72 to 0.75 mg/L; p = 0.44), IL-6 (WMD,−0.03; 95% CI,−0.40 to 0.33 pg/mL; p = 0.86), and TNF-α (WMD, 0.12; 95% CI,−0.08 to 0.31 pg/mL; p = 0.24). |
| Veronese, N. et al., 2022 [106] | 17 RCTs | Adult participants n-889 | Mg supplementation significantly decreased serum CRP (SMD = −0.356; 95% CI: −0.659 to −0.054; p = 0.02), and increased nitric oxide (NO) levels (SMD = 0.321; 95% CI: 0.037 to 0.604; p = 0.026) | |
| Mazidi, M. et al., 2018 [107] | 8 RCTs | Adult participants n-349 Duration: 8 h to 6.5 months Doses: 320–1500 mg/d | Mg supplementation led to reduction in CRP (WMD: −1.33 mg/L; 95% CI: −2.63, −0.02, heterogeneity p < 0.123; I2 = 29.1%) Changes in serum CRP levels were independent of the dosage and duration of Mg supplementation | |
| Simental-Mendía, L.E. et al., 2017 [108] | 11 RCTs | Adults participants n-ns | Mg treatment was found to significantly affect plasma concentrations of CRP in subgroups of populations with baseline plasma CRP > 3 mg/L (WMD: −1.12 mg/L, 95% CI: −2.05, −0.18, p = 0.019) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pelczyńska, M.; Moszak, M.; Bogdański, P. The Role of Magnesium in the Pathogenesis of Metabolic Disorders. Nutrients 2022, 14, 1714. https://doi.org/10.3390/nu14091714
Pelczyńska M, Moszak M, Bogdański P. The Role of Magnesium in the Pathogenesis of Metabolic Disorders. Nutrients. 2022; 14(9):1714. https://doi.org/10.3390/nu14091714
Chicago/Turabian StylePelczyńska, Marta, Małgorzata Moszak, and Paweł Bogdański. 2022. "The Role of Magnesium in the Pathogenesis of Metabolic Disorders" Nutrients 14, no. 9: 1714. https://doi.org/10.3390/nu14091714
APA StylePelczyńska, M., Moszak, M., & Bogdański, P. (2022). The Role of Magnesium in the Pathogenesis of Metabolic Disorders. Nutrients, 14(9), 1714. https://doi.org/10.3390/nu14091714







