Dietary Intake of Toxic Heavy Metals with Major Groups of Food Products—Results of Analytical Determinations
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents (Chemicals)
2.2. Sampling
2.3. Analytical Determination of Cd, Pb, Hg and Ni Using ICP-OES
2.4. Statistical Analysis
3. Results and Discussion
3.1. Dietary Intake of Cd, Pb, Hg and Ni with Major Groups of Food Products
3.1.1. Contribution of Major Food Items to an Overall Dietary Intake of Cd
3.1.2. Contribution of Major Food Items to an Overall Dietary Intake of Pb
3.1.3. Contribution of Major Food Items to an Overall Dietary Intake of Hg
3.1.4. Contribution of Major Food Items to an Overall Dietary Intake of Ni
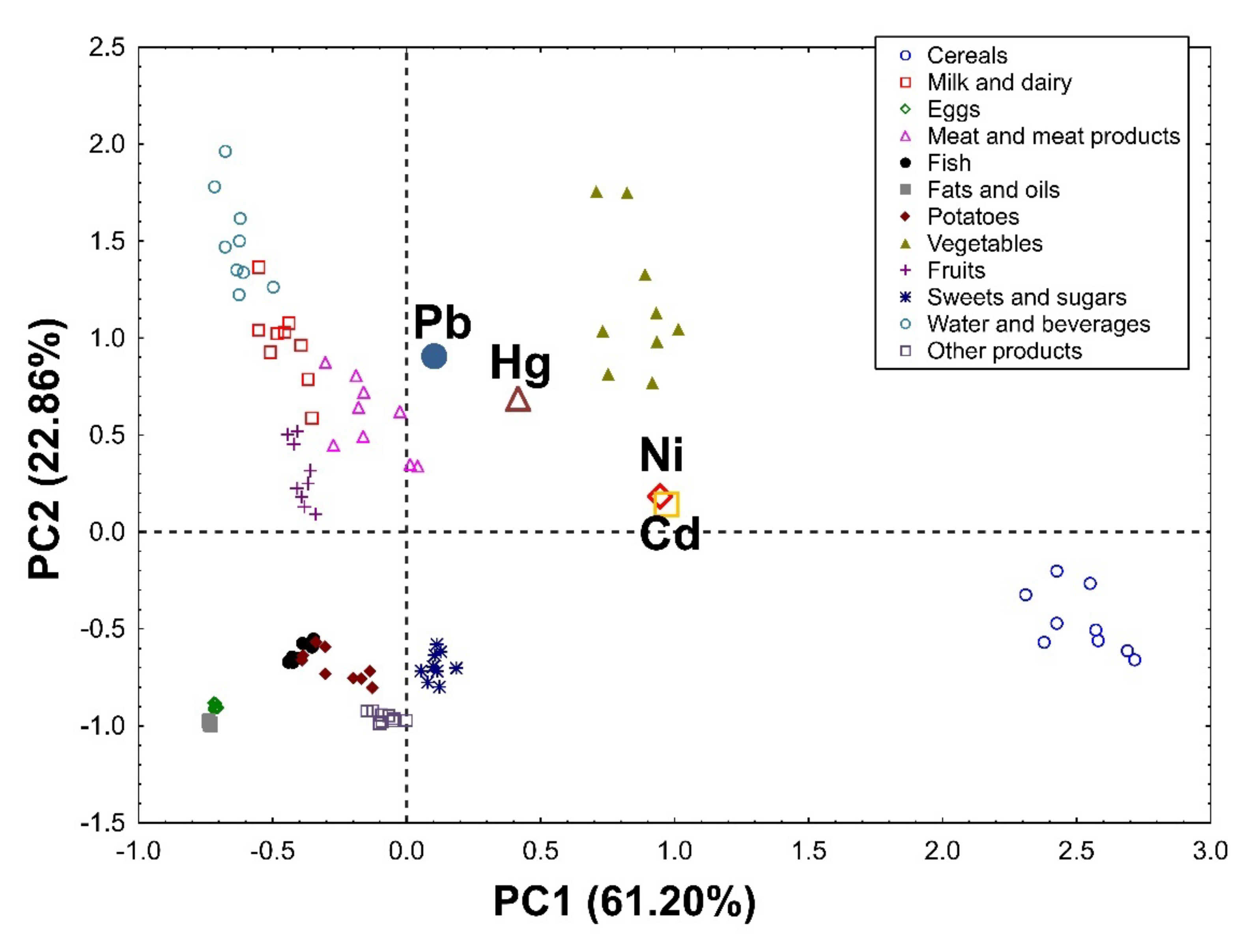
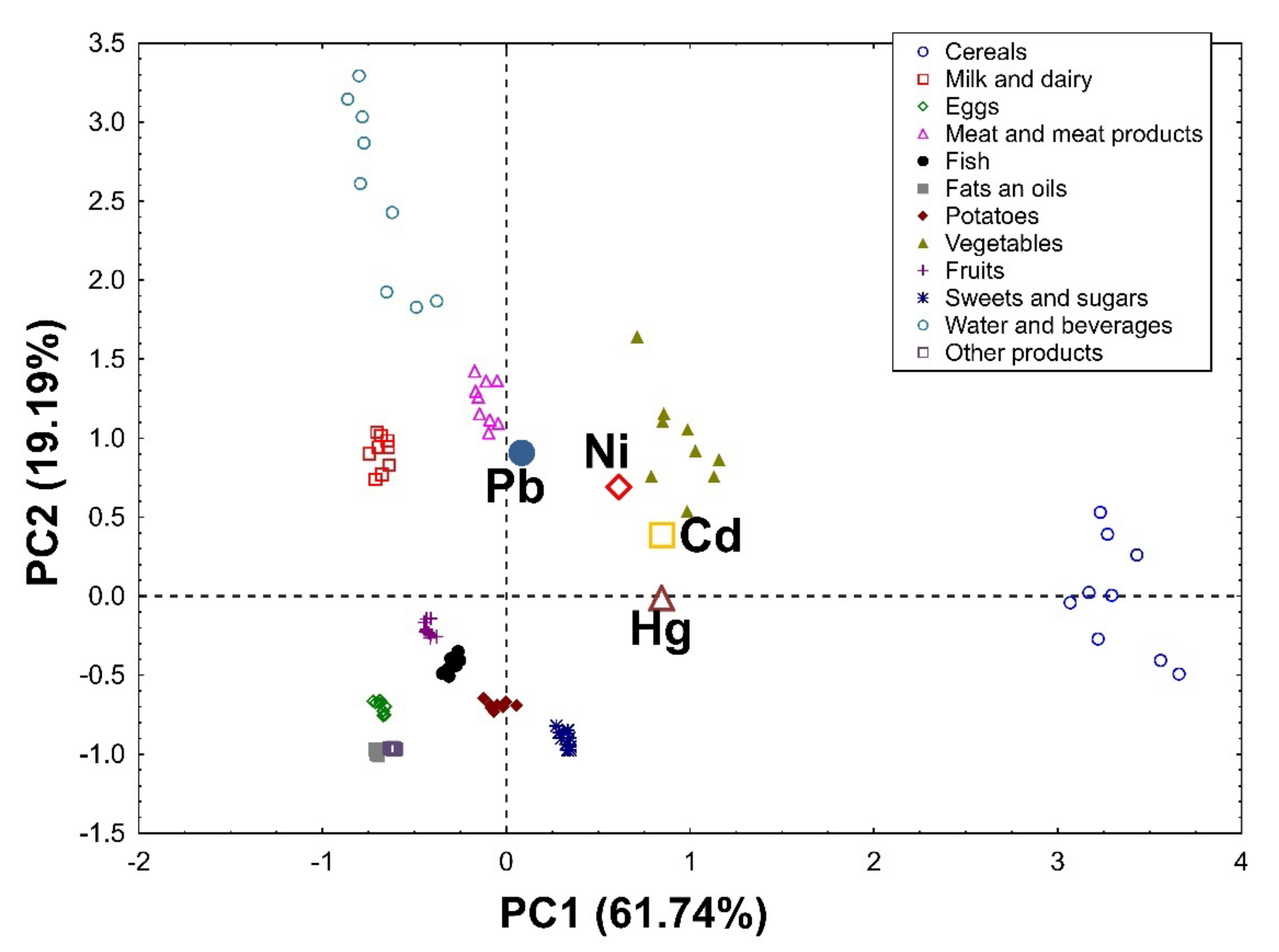
3.1.5. Principal Component Analysis (PCA)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, Y.; Michalak, M.; Agellon, L.B. Importance of nutrients and nutrient metabolism on human health. Yale J. Biol. Med. 2018, 91, 95–103. [Google Scholar] [PubMed]
- Marzec, Z.; Koch, W.; Marzec, A.; Żukiewicz-Sobczak, W. Dietary exposure to cadmium, lead and nickel among students from south-east Poland. Ann. Agric. Environ. Med. 2014, 21, 825–828. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bocio, A.; Nadal, M.; Domingo, J.L. Human exposure to metals through the diet in Tarragona, Spain: Temporal trend. Biol. Trace Elem. Res. 2005, 104, 193–202. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, 364, 5–24. [Google Scholar]
- European Commission. Commission Regulation (EC) No 420/2011 Amending Regulation (EC) No 1881/2006 setting maximum levels for certain contaminants in foodstuffs, 29 April 2011. Off. J. Eur. Union 2011, 111, 3–6. [Google Scholar]
- Arnich, N.; Sirot, V.; Riviere, G.; Jean, J.; Noel, L.; Guerin, T.; Leblanc, J.C. Dietary exposure to trace elements and health risk assessment in the 2nd French total diet study. Food Chem. Toxicol. 2012, 50, 2432–2449. [Google Scholar] [CrossRef]
- Coelho, S.D.; Pastorinho, M.R.; Itai, T.; Isobe, T.; Kunisue, T.; Nogueira, A.J.A.; Tanabe, S.; Sousa, A.C.A. Lead in duplicate diet samples from an academic community. Sci. Total Environ. 2016, 573, 603–607. [Google Scholar] [CrossRef]
- Koch, W.; Karim, M.R.; Marzec, Z.; Miyataka, H.; Himeno, S.; Asakawa, Y. Dietary intake of metals by the young adult population of Eastern Poland: Results from a market basket study. J. Trace Elem. Med. Biol. 2016, 35, 36–42. [Google Scholar] [CrossRef]
- Marini, M.; Angouria-Tsorochidou, E.; Caro, D.; Thomsen, M. Daily intake of heavy metals and minerals in food—A case study of four Danish dietary profiles. J. Clean. Prod. 2021, 280, 124279. [Google Scholar] [CrossRef]
- Brown, L.; Poudyal, H.; Panchal, S.K. Functional foods as potential therapeutic options for metabolic syndrome. Obes. Rev. 2015, 16, 914–941. [Google Scholar] [CrossRef]
- Konstantinidi, M.; Koutelidakis, A.E. Functional foods and bioactive compounds: A Review of Its Possible Role on Weight Management and Obesity’s Metabolic Consequences. Medicines 2019, 6, 94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carrera-Bastos, P.; Fontes-Villalba, M.; O’Keefe, J.H.; Lindeberg, S.; Cordain, L. The western diet and lifestyle and diseases of civilization. Res. Rep. Clin. Cardiol. 2011, 2, 15–35. [Google Scholar] [CrossRef] [Green Version]
- Glibowski, P.; Ćwiklińska, M.; Białasz, A.; Koch, W.; Marzec, Z. Fast consumption increases the risk of overweight and obesity. Rocz. Panstw. Zakl. Hig. 2020, 71, 27–31. [Google Scholar] [CrossRef]
- Hurst, Y.; Fukuda, H. Effects of changes in eating speed on obesity in patients with diabetes: A secondary analysis of longitudinal health check-up data. BMJ Open 2018, 8, e019589. [Google Scholar] [CrossRef] [PubMed]
- Rippin, H.L.; Hutchinson, J.; Jewell, J.; Breda, J.J.; Cade, J.E. Adult Nutrient Intakes from Current National Dietary Surveys of European Populations. Nutrients 2017, 9, 1288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Merten, C.; Ferrari, P.; Bakker, M.; Boss, A.; Hearty, A.; Leclercq, C.; Lindtner, O.; Tlustos, C.; Verger, P.; Volatier, J.-L. Methodological characteristics of the national dietary surveys carried out in the European Union as included in the European Food Safety Authority (EFSA) Comprehensive European Food Consumption Database. Food Addit. Contam. Part A 2011, 28, 975–995. [Google Scholar] [CrossRef] [Green Version]
- Hutchinson, J.; Rippin, H.; Jewell, J.; Breda, J.; Cade, J.E. Comparison of high and low trans fatty acid consumers: Analyses of UK National Diet and Nutrition Surveys before and after product reformulation. Public Health Nutr. 2018, 21, 465–479. [Google Scholar] [CrossRef]
- Marzec, Z.; Schlegel-Zawadzka, M. Exposure to cadmium, lead and mercury in the adult population from Eastern Poland, 1990–2002. Food Addit. Contam. 2004, 21, 963–970. [Google Scholar] [CrossRef]
- Domingo, J.L.; Perelló, G.; Bordonaba, J.G. Dietary Intake of Metals by the Population of Tarragona County (Catalonia, Spain): Results from a Duplicate Diet Study. Biol. Trace Elem. Res. 2012, 146, 420–425. [Google Scholar] [CrossRef]
- Koch, W.; Czop, M.; Nawrocka, A.; Wiącek, D. Contribution of Major Groups of Food Products to the Daily Intake of Selected Elements—Results from Analytical Determinations Supported by Chemometric Analysis. Nutrients 2020, 12, 3412. [Google Scholar] [CrossRef]
- Dodd, K.W.; Guenther, P.M.; Freedman, L.S.; Subar, A.F.; Kipnis, V.; Midthune, D.; Tooze, J.A.; Krebs-Smith, S.M. Statistical Methods for Estimating Usual Intake of Nutrients and Foods: A Review of the Theory. J. Am. Diet. Assoc. 2006, 106, 1640–1650. [Google Scholar] [CrossRef] [PubMed]
- Thompson, F.E.; Kirkpatrick, S.I.; Subar, A.F.; Reedy, J.; Schap, T.E.; Wilson, M.M.; Krebs-Smith, S.M. The National Cancer Institute’s Dietary Assessment Primer: A Resource for Diet Research. J. Acad. Nutr. Diet. 2015, 115, 1986–1995. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holmes, B.; Nelson, M. The strengths and weaknesses of dietary survey methods in materially deprived households in England: A discussion paper. Public Health Nutr. 2009, 12, 1157–1164. [Google Scholar] [CrossRef] [PubMed]
- Rippin, H.L.; Hutchinson, J.; Evans, C.E.L.; Jewell, J.; Breda, J.J.; Cade, J.E. National nutrition surveys in Europe: A review on the current status in the 53 countries of the WHO European region. Food Nutr. Res. 2018, 62, 1362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mejia, E.G.; Ramirez-Mares, M.V.; Puangpraphant, S. Bioactive components of tea: Cancer, inflammation and behavior. Brain Behav. Immun. 2009, 23, 721–731. [Google Scholar] [CrossRef] [PubMed]
- Koch, W. Black tea infusion as a major source of polyphenols in the daily food rations of students. Bromat Chem. Toksykol. 2016, 49, 308–312. [Google Scholar]
- Brzezicha-Cirocka, J.; Grembecka, M.; Szefer, P. Analytical Assessment of Bio- and Toxic Elements Distribution in Pu-erh and Fruit Teas in View of Chemometric Approach. Biol. Trace Elem. Res. 2016, 174, 240–250. [Google Scholar] [CrossRef] [Green Version]
- Koch, W.; Kukula-Koch, W.; Czop, M.; Baj, T.; Kocki, J.; Bawiec, P.; Casasnovas, R.O.; Głowniak-Lipa, A.; Głowniak, K. Analytical Assessment of Bioelements in Various Types of Black Teas from Different Geographical Origins in View of Chemometric Approach. Molecules 2021, 26, 6017. [Google Scholar] [CrossRef]
- Dambiec, M.; Polechońska, L.; Klink, A. Levels of Essential and Non-Essential Elements in Black Teas Commercialized in Poland and Their Transfer to Tea Infusion. J. Food Compost. Anal. 2013, 31, 62–66. [Google Scholar] [CrossRef]
- Brzezicha-Cirocka, J.; Grembecka, M.; Ciesielski, T.; Flaten, T.P.; Szefer, P. Evaluation of Macro- and Microelement Levels in Black Tea in View of Its Geographical Origin. Biol. Trace Elem. Res. 2017, 176, 429–441. [Google Scholar] [CrossRef] [Green Version]
- Milani, R.; Morgano, M.A.; Cadore, S. Trace Elements in Camelia sinensis Marketed in Southern Brazil: Extraction from Tea Leaves to Beverages and Dietary Exposure. LWT-Food Sci. Technol. 2016, 68, 491–498. [Google Scholar] [CrossRef]
- Sousa, A.G.; Da Costa, T.H.M. Assessment of Nutrient and Food Group Intakes across Sex, Physical Activity, and Body Mass Index in an Urban Brazilian Population. Nutrients 2018, 10, 1714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fenech, M.; Baghurst, P.; Luderer, W.; Turner, J.; Record, S.; Ceppi, M.; Bonassi, S. Low intake of calcium, folate, nicotinic acid, vitamin E, retinol, beta-carotene and high intake of pantothenic acid, biotin and riboflavin are significantly associated with increased genome instability--results from a dietary intake and micronucleus index survey in South Australia. Carcinogenesis 2005, 26, 991–999. [Google Scholar] [CrossRef] [Green Version]
- Barbaresko, J.; Koch, M.; Schulze, M.B.; Nothlings, U. Dietary pattern analysis and biomarkers of low-grade inflammation: A systematic literature review. Nutr. Rev. 2013, 71, 511–527. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, C.E.; Keast, D.R.; Fulgoni, V.L.; Nicklas, T.A. Food sources of energy and nutrients among adults in the US: NHANES 2003–2006. Nutrients 2012, 4, 2097–2120. [Google Scholar] [CrossRef] [Green Version]
- Kolb, H.; Mandrup-Poulsen, T. The global diabetes epidemic as a consequence of lifestyle-induced low-grade inflammation. Diabetologia 2010, 53, 10–20. [Google Scholar] [CrossRef] [Green Version]
- Dobrzański, Z.; Kołacz, R.; Bodak, E. Metale ciężkie w środowisku zwierząt. Med. Wet. 1996, 52, 570–574. [Google Scholar]
- Thévenod, F.; Lee, W.K. Toxicology of cadmium and its damage to mammalian organs. Met. Ions Life Sci. 2013, 11, 415–490. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Cadmium in food—Scientific opinion of the Panel on Contaminants in the Food Chain (Question No EFSA-Q-2007-138). EFSA J. 2009, 980, 1–139. [Google Scholar]
- European Food Safety Authority (EFSA). Opinion of the Scientific Panel on contaminants in the food chain [CONTAM] related to mercury and methylmercury in food (Question NoEFSA-Q-2003-030). EFSA J. 2004, 2, 34. [Google Scholar] [CrossRef]
- EFSA CONTAM Panel (EFSA Panel on Contaminants in the Food Chain); Schrenk, D.; Bignami, M.; Bodin, L.; Chipman, J.K.; del Mazo, J.; Grasl-Kraupp, B.; Hogstrand, C.; Hoogenboom, L.R.; Leblanc, J.-C.; et al. Scientific Opinion on the update of the risk assessment of nickel in food and drinking water. EFSA J. 2020, 18, 6268. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the FoodChain (CONTAM). Scientific opinion on lead in food. EFSA J. 2010, 8, 1570, (Question No EFSA-Q-2007-137). Adopted on March 2010. [Google Scholar] [CrossRef]
- Nasreddine, L.; Nashalian, O.; Naja, F.; Itani, L.; Parent-Massin, D.; Nabhani-Zeidan, M.; Hwalla, N. Dietary exposure to essential and toxic trace elements from a Total diet study in an adult Lebanese urban population. Food Chem. Toxicol. 2010, 48, 1262–1269. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Pan, L.; Liu, G.; Zhang, H.; Zhang, J.; Jiang, J.; Xiao, Y.; Bai, W.; Jiao, R.; Huang, W. Dietary exposure to cadmium of Shenzhen adult residents from a total diet study. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2018, 35, 706–714. [Google Scholar] [CrossRef] [PubMed]
- Itoh, H.; Iwasaki, M.; Sawada, N.; Takachi, R.; Kasuga, Y.; Yokoyama, S.; Onuma, H.; Nishimura, H.; Kusama, R.; Yokoyama, K.; et al. Dietary cadmium intake and breast cancer risk in Japanese women: A case-control study. Int. J. Hyg. Environ. Health 2014, 217, 70–77. [Google Scholar] [CrossRef]
- Minh, N.G.; Hough, R.L.; Thuy, L.T.; Nyberg, Y.; Mai, L.B.; Vinh, N.C.; Khai, N.M.; Oborn, I. Assessing dietary exposure to cadmium in a metal recycling community in Vietnam: Age and gender aspects. Sci. Total Environ. 2012, 416, 164–171. [Google Scholar] [CrossRef]
- Satarug, S.; Swaddiwudhipong, W.; Ruangyuttikarn, W.; Nishijo, M.; Ruiz, P. Modeling cadmium exposures in low- and high-exposure areas in Thailand. Environ. Health Perspect. 2013, 121, 531–536. [Google Scholar] [CrossRef] [Green Version]
- Eriksen, K.T.; Halkjaer, J.; Sorensen, M.; Meliker, J.R.; McElroy, J.A.; Tjonneland, A.; Raaschou-Nielsen, O. Dietary cadmium intake and risk of breast, endometrial and ovarian cancer in Danish postmenopausal women: A prospective cohort study. PLoS ONE 2014, 9, e100815. [Google Scholar] [CrossRef] [Green Version]
- Vacchi-Suzzi, C.; Eriksen, K.T.; Levine, K.; McElroy, J.; Tjonneland, A.; Raaschou-Nielsen, O.; Harrington, J.M.; Meliker, J.R. Dietary Intake Estimates and Urinary Cadmium Levels in Danish Postmenopausal Women. PLoS ONE 2015, 10, e0138784. [Google Scholar] [CrossRef] [Green Version]
- Sand, S.; Becker, W. Assessment of dietary cadmium exposure in Sweden and population health concern including scenario analysis. Food Chem. Toxicol. 2012, 50, 536–544. [Google Scholar] [CrossRef]
- Škrbić, B.; Živančev, J.; Mrmoš, N. Concentrations of arsenic, cadmium and lead in selected foodstuffs from Serbian market basket: Estimated intake by the population from the Serbia. Food Chem. Toxicol. 2013, 58, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Vesey, D.A.; Gobe, G.C. Current health risk assessment practice for dietary cadmium: Data from different countries. Food Chem. Toxicol. 2017, 106 Pt A, 430–445. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Cadmium dietary exposure in the European population. EFSA J. 2012, 10, 2551. [Google Scholar] [CrossRef]
- Llobet, J.M.; Falco, G.; Casas, C.; Teixido, A.; Domingo, J.L. Concentrations of arsenic, cadmium, mercury, and lead in common foods and estimated daily intake by children, adolescents, adults, and seniors of Catalonia, Spain. J. Agric. Food Chem. 2003, 51, 838–842. [Google Scholar] [CrossRef]
- Munoz, O.; Zamorano, P.; Garcia, O.; Bastias, J.M. Arsenic, cadmium, mercury, sodium, and potassium concentrations in common foods and estimated daily intake of the population in Valdivia (Chile) using a total diet study. Food Chem. Toxicol. 2017, 109 Pt 2, 1125–1134. [Google Scholar] [CrossRef]
- Chiodo, L.M.; Covington, C.; Sokol, R.J.; Hannigan, J.H.; Jannise, J.; Ager, J.; Greenwald, M.; Delaney-Black, V. Blood lead levels and specific attention effects in young children. Neurotoxicol. Teratol. 2007, 29, 538–546. [Google Scholar] [CrossRef]
- ATSDR. Public Health Service Agency for Toxic Substances and Disease Registry. U.S. Department of Health and Human Services, Toxicological Profile for LEAD (Update). 2020. Available online: http://www.atsdr.cdc.gov/toxprofiles/tp13.pdf (accessed on 18 March 2022).
- Turconi, G.; Minoia, C.; Ronchi, A.; Roggi, C. Dietary exposure estimates of twenty-one trace elements from a Total Diet Study carried out in Pavia, Northern Italy. Br. J. Nutr. 2009, 101, 1200–1208. [Google Scholar] [CrossRef] [Green Version]
- Raghunath, R.; Tripathi, R.M.; Suseela, B.; Bhalke, S.; Shukla, V.K.; Puranik, V.D. Dietary intake of metals by Mumbai adult population. Sci. Total Environ. 2006, 356, 62–68. [Google Scholar] [CrossRef]
- Wilhelm, M.; Wittsiepe, J.; Schrey, P.; Feldmann, C.; Idel, H. Dietary intake of lead by children and adults from Germany measured by the duplicate method. Int. J. Hyg. Environ. Health 2003, 206, 493–503. [Google Scholar] [CrossRef]
- Aung, N.N.; Yoshinaga, J.; Takahashi, J.I. Dietary intake of toxic and essential trace elements by the children and parents living in Tokyo Metropolitan Area, Japan. Food Addit. Contam. 2006, 23, 883–894. [Google Scholar] [CrossRef]
- Liu, P.; Wang, C.N.; Song, X.Y.; Wu, Y.N. Dietary intake of lead and cadmium by children and adults—Result calculated from dietary recall and available lead/cadmium level in food in comparison to result from food duplicate diet method. Int. J. Hyg. Environ. Health 2010, 213, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Cuadrado, C.; Kumpulainen, J.; Moreiras, O. Contaminants and nutrients in total diets in Spain. Eur. J. Clin. Nutr. 1995, 49, 767–778. [Google Scholar] [PubMed]
- European Food Safety Authority. Lead dietary exposure in the European population. EFSA J. 2012, 10, 2831. [Google Scholar] [CrossRef]
- JECFA (Joint FAO/WHO Expert Committee on Food Additives). Summary and Conclusions of the Sixty-First Meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA). 2003, pp. 18–22. Available online: http://www.who.int/pcs/jecfa/Summary61.pdf (accessed on 18 March 2022).
- WHO. Safety Evaluation of Certain Food Additives and Contaminants WHO Food Additive Series No. 52. Methylmercury. 2004. Available online: http://www.inchem.org/documents/jecfa/jecmono/v52je23.htm (accessed on 18 March 2022).
- NRC (National Research Council); Committee on the Toxicological Effects of Methylmercury. Toxicological Effects of Methylmercury; National Academy Press: Washington, DC, USA, 2000. [Google Scholar]
- Kuras, R.; Janasik, B.; Stanislawska, M.; Kozlowska, L.; Wasowicz, W. Assessment of Mercury Intake from Fish Meals Based on Intervention Research in the Polish Subpopulation. Biol. Trace Elem. Res. 2017, 179, 23–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wojciechowska-Mazurek, M.; Starska, K.; Brulińska-Ostrowska, E.; Plewa, M.; Karłowski, K. Monitoring studies of fish, seafood and their processed products related to the content of elements noxious to health. Bromat. Chem. Toksykol. 2008, 41, 525–529. [Google Scholar]
- Wojciechowska-Mazurek, M.; Starska, K.; Mania, M.; Brulińska-Ostrowska, E.; Biernat, U.; Karłowski, K. Monitoring of contamination of foodstuffs with elements noxious to human health, part II. Mineral, waters, soft drinks, fruits, nuts, rice, soybeans, fish and seafood. Rocz. Panstw. Zakl. Hig. 2010, 61, 27–35. [Google Scholar]
- Spodniewska, A.; Barski, D. Concentration of some metals in the muscles of fish from selected lakes of Warmia and Mazury region (Poland). Acta Vet. Brno 2013, 82, 067–071. [Google Scholar] [CrossRef]
- Larsen, E.H.; Andersen, N.L.; Møller, A.; Petersen, A.; Mortensen, G.K.; Petersen, J. Monitoring the content and intake of trace elements from food in Denmark. Food Addit. Contam. 2002, 19, 33–46. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Evaluation of Certain Food Additives and Contaminants (Forty-First Report of the Joint FAO/WHO Expert Committee on Food Additives); WHO Tech. Report Series No. 837; World Health Organization: Geneva, Switzerland, 1993. [Google Scholar]
- Bartos, A.; Majak, I.; Leszczyńska, J. Uptake and assimilability of nickel in the course of systemic allergy: Implications for elimination diet. Food Res. Int. 2014, 55, 412–417. [Google Scholar] [CrossRef]
- Suomi, J.; Valsta, L.; Suominen, K.; Tuominen, P. Risk assessment on the dietary heavy metal exposure and aluminium exposure of Finnish adults. Finn. Food Auth. Res. Rep. 2020, 2020, 78. [Google Scholar]
- Cubadda, F.; Iacoponi, F.; Ferraris, F.; D’Amato, M.; Aureli, F.; Raggi, A.; Sette, S.; Turrini, A.; Mantovani, A. Dietary exposure of the Italian population to nickel: The national Total Diet Study. Food Chem. Toxicol. 2020, 146, 111813. [Google Scholar] [CrossRef] [PubMed]
- FERA (The Food and Environment Research Agency). Total Diet Study of Metals and Other Elements in Food. Report for the UK Food Standards Agency (FS102081). 2015; pp. 1–69. Available online: https://www.food.gov.uk/sites/default/files/media/document/total-diet-study-of-metals-and-other-elements-in-food_0.pdf (accessed on 18 March 2022).
| Food Group | Women | Men | ||||
|---|---|---|---|---|---|---|
| Amount (g) | Energy (kcal) | Composition (%) | Amount (g) | Energy (kcal) | Composition (%) | |
| Cereals | 224.1 ± 20.8 | 653.6 ± 79.2 | baking 51.5 cereal flakes 14.7 grits 12.5 rice 11.5 pasta 9.80 | 284.8 ± 25.3 | 770.1 ± 84.2 | baking 66.6 rice 12.9 pasta 9.40 cereal flakes 6.35 grits 4.75 |
| Milk and dairy | 276.9 ± 25.7 | 303.9 ± 35.4 | milk 45.1 cheese 30.4 yogurts 23.2 sour cream 1.3 | 232.3 ± 22.4 | 327.1 ± 29.8 | cheese 47.0 milk 31.2 yogurts 18.9 sour cream 2.90 |
| Eggs | 18.2 ± 2.11 | 21.4 ± 1.92 | scrambled eggs 48.1 boiled eggs 38.7 omlette 13.2 | 45.5 ± 5.17 | 53.1 ± 7.12 | scrambled eggs 60.9 boiled eggs 33.2 omlette 5.90 |
| Meat and meat products | 191.7 ± 20.8 | 406.5 ± 47.2 | pork 39.0 poultry 37.6 beef 23.4 | 283.5 ± 31.4 | 621.5 ± 56.4 | pork 42.0 beef 30.4 poultry 27.6 |
| Fish | 17.5 ± 2.14 | 21.4 ± 2.72 | tuna 44.2 cod 38.6 salmon 12.0 herring 4.0 pike 1.2 | 20.0 ± 3.18 | 36.7 ± 4.12 | mackerel 47.0 salmon 35.3 tuna 8.90 sardine 8.80 |
| Fats and oils | 14.0 ± 1.34 | 116.7 ± 11.4 | butter 35.2 rapeseed oil 32.5 sunflower oil 15.7 olive oil 11.1 margarine 3.29 linseed oil 2.21 | 25.1 ± 2.91 | 209.7 ± 18.4 | rapeseed oil 38.2 butter 32.6 sunflower oil 20.2 lard 6.90 olive oil 2.10 |
| Potatoes | 50.1 ± 6.55 | 38.6 ± 4.22 | 82.5 ± 9.52 | 63.5 ± 7.48 | ||
| Vegetables | 384.1 ± 45.2 | 90.9 ± 12.5 | tomatoes 30.1 pepper 19.4 carrot 10.9 cabbage 9.95 beetroot 7.85 cauliflower 6.82 broccoli 6.64 lettuce 2.17 celery 1.16 others 5 | 343.8 ± 38.4 | 72.6 ± 9.15 | tomatoes 32.2 carrot 22.3 pepper 15.6 cucumber 11.2 onion 4.05 cabbage 3.58 broccoli 2.50 pumpkin 1.30 leek 1.27 lettuce 1.05 others 5 |
| Fruits | 227.2 ± 35.6 | 140.5 ± 18.8 | apples 34.2 bananas 33.6 oranges 7.65 mandarins 5.92 pears 4.14 peaches 3.42 raspberries 3.05 watermelons 1.86 kiwi 1.48 raisins 1.32 nectarines 1.25 others 1.44 | 192.2 ± 26.2 | 114.4 ± 17.8 | apples 40.5 bananas 28.9 oranges 10.1 mandarins 5.92 pears 5.56 grapefruit 3.10 grapes 1.55 red currants 1.85 dried dates 1.36 others 1.16 |
| Sweets and sugars | 62.4 ± 10.2 | 236.9 ± 31.8 | cakes and cookies 53.4 bars 14.3 sugar 13.4 chocolate 12.5 honey 1.70 others 4.70 | 69.1 ± 12.4 | 251 ± 40.8 | cakes and cookies 60.5 sugar 23.4 bars 10.6 chocolate 4.80 honey 0.70 |
| Water and beverages | 638.7 ± 75.4 | 33.8 ± 4.52 | black tea (infusion) 56.8 soups (only water) 18.8 fruit juices 11.1 coffee (infusion) 11.0 sweet beverages 2.30 | 713.1 ± 100.2 | 52.3 ± 6.18 | black tea (infusion) 53.4 coffee (infusion) 17.0 soups (only water) 13.2 fruit juices 11.8 sweet beverages 3.27 bear 1.33 |
| Other products | 34.5 ± 5.52 | 139 ± 15.8 | jams (no added sugar) 32.4 sauces 22.8 hooves 16.9 seeds (pumpkin/sunflower) 10.6 nuts 5.50 snacks 4.24 pudding 2.56 almonds 2.11 tomato concentrate 1.94 cocoa 0.95 | 18.4 ± 3.11 | 98.7 ± 10.2 | sauces 56.3 jams (no added sugar) 22.0 pancakes 13.4 almonds 2.78 nuts 2.25 pudding 1.68 cocoa 1.10 others 0.49 |
| Total | 2139 ± 264 | 2203 ± 331 | 2310 ± 351 | 2671 ± 355 | ||
| Parameter | Cd | Pb | Hg | Ni |
|---|---|---|---|---|
| Reference value (mg/kg) | 0.30 ± 0.02 | 0.50 ± 0.04 | 0.20 ± 0.01 | 0.50 ± 0.03 |
| Determined value (mg/kg) | 0.27 | 0.53 | 0.16 | 0.52 |
| 0.31 | 0.48 | 0.18 | 0.51 | |
| 0.33 | 0.46 | 0.16 | 0.57 | |
| 0.29 | 0.52 | 0.21 | 0.49 | |
| 0.27 | 0.52 | 0.23 | 0.55 | |
| 0.28 | 0.54 | 0.17 | 0.5 | |
| Average | 0.29 | 0.51 | 0.19 | 0.54 |
| SD | 0.02 | 0.03 | 0.03 | 0.03 |
| RSD (%) | 8.28 | 6.08 | 13.68 | 5.58 |
| Recovery (%) | 96.67 | 102.0 | 95.0 | 106.0 |
| LOD * (µg/kg) | 0.8 | 4.5 | 1.8 | 1.2 |
| LOQ ** (µg/kg) | 2.1 | 14.8 | 6.0 | 4.0 |
| Food Group | Element | |||||||
|---|---|---|---|---|---|---|---|---|
| Cd (µg) | Pb (µg) | Hg (µg) | Ni (µg) | |||||
| Women | Men | Women | Men | Women | Men | Women | Men | |
| Cereals | 3.20 ± 0.25 g 2.82–3.46 | 5.13 ± 0.25 f 4.71–5.44 | 5.28 ± 0.94 c 4.12–6.76 | 9.80 ± 1.97 cd 7.26–12.91 | 2.99 ± 0.23 g 2.71–3.42 | 3.57 ± 0.24 i 3.21–3.84 | 85.73 ± 6.34 g 77.89–95.25 | 87.57 ± 6.78 g 74.21–94.87 |
| Milk and dairy | 0.45 ± 0.07 bc 0.36–0.55 | <LOQ | 8.26 ± 1.18 d 6.33–10.19 | 7.72 ± 0.48 d 7.05–8.34 | 3.59 ± 0.26 h 3.11–3.91 | 3.27 ± 0.26 h 2.81–3.55 | 3.37 ± 0.35 ab 2.75–3.85 | 3.07 ± 0.41 ab 2.55–3.58 |
| Eggs | 0.06 ± 0.01 a 0.05–0.07 | 0.12 ± 0.01 a 0.10–0.13 | 0.87 ± 0.07 a 0.76–0.95 | 1.72 ± 0.31 ab 1.31–2.21 | 0.22 ± 0.02 a 0.19–0.25 | 0.51 ± 0.09 cd 0.34–0.62 | 0.50 ± 0.06 a 0.40–0.59 | 1.30 ± 0.10 a 1.18–1.50 |
| Meat and meat products | 1.06 ± 0.17 e 0.77–1.28 | 1.17 ± 0.09 d 1.03–1.33 | 8.16 ± 1.10 d 6.55–9.45 | 11.03 ± 0.78 c 9.84–12.07 | 2.59 ± 0.25 f 2.21–2.94 | 3.48 ± 0.27 hi 3.05–3.92 | 10.0 ± 0.96 c 8.55–11.62 | 10.52 ± 0.98 d 9.11–12.11 |
| Fish | 0.29 ± 0.02 b 0.26–0.34 | 0.38 ± 0.03 bc 0.34–0.43 | 0.40 ± 0.06 a 0.32–0.48 | 0.65 ± 0.08 a 0.53–0.76 | 1.94 ± 0.22 d 1.71–2.34 | 2.67 ± 0.21 g 2.40–3.0 | 0.67 ± 0.08 a 0.56–0.80 | 0.38 ± 0.04 a 0.31–0.43 |
| Fats and oils | <LOQ | <LOQ | 0.29 ± 0.04 a 0.22–0.35 | 0.97 ± 0.10 a 0.81–1.12 | 0.14 ± 0.02 a 0.11–0.17 | 0.15 ± 0.02 a 0.12–0.18 | 0.16 ± 0.01 a 0.14–0.17 | 1.58 ± 0.24 ab 1.22–1.94 |
| Potatoes | 0.98 ± 0.21 e 0.69–1.26 | 1.32 ± 0.13 d 1.18–1.54 | 2.98 ± 0.35 b 2.56–3.55 | 2.99 ± 0.24 b 2.70–3.28 | 0.50 ± 0.05 bc 0.42–0.57 | 0.82 ± 0.06 e 0.72–0.87 | 4.88 ± 0.35 b 4.35–5.27 | 5.89 ± 0.49 bc 5.18–6.59 |
| Vegetables | 1.84 ± 0.26 f 1.53–2.11 | 2.73 ± 0.26 e 2.41–3.09 | 11.65 ± 2.06 e 9.32–15.0 | 11.88 ± 1.96 c 8.51–15.56 | 3.87 ± 0.26 i 3.52–4.20 | 3.42 ± 0.26 hi 3.03–3.85 | 43.43 ± 3.18 f 40.18–49.58 | 29.18 ± 2.19 e 26.14–32.18 |
| Fruits | 0.26 ± 0.03 ab 0.22–0.30 | 0.22 ± 0.03 ab 0.18–0.29 | 5.77 ± 0.90 c 4.50–7.04 | 3.12 ± 0.20 b 2.74–3.32 | 2.32 ± 0.15 e 2.08–2.55 | 1.89 ± 0.12 f 1.70–2.10 | 12.42 ± 0.52 c 11.45–12.92 | 7.77 ± 0.76 cd 6.75–9.14 |
| Sweets and sugars | 1.17 ± 0.08 e 1.04–1.28 | 1.20 ± 0.09 d 1.10–1.35 | 3.13 ± 0.48 b 2.32–3.84 | 1.98 ± 0.24 ab 1.62–2.35 | 0.75 ± 0.07 b 0.65–0.85 | 0.76 ± 0.09 de 0.64–0.86 | 18.48 ± 1.15 d 16.32–19.61 | 25.63 ± 1.84 e 23.12–28.16 |
| Water and beverages | 0.59 ± 0.09 cd 0.44–0.69 | 0.54 ± 0.07 c 0.45–0.67 | 16.37 ± 1.59 f 14.33–19.36 | 23.25 ± 3.52 e 18.87–27.99 | 0.54 ± 0.04 b 0.49–0.63 | 0.45 ± 0.07 bc 0.36–0.58 | 26.27 ± 2.35 e 22.14–29.15 | 41.09 ± 5.90 f 30.17–47.87 |
| Other products | 0.70 ± 0.07 d 0.62–0.84 | 0.15 ± 0.02 a 0.12–0.18 | 1.59 ± 0.11 ab 1.42–1.74 | 0.63 ± 0.03 a 0.58–0.65 | 0.27 ± 0.03 ac 0.23–0.32 | 0.21 ± 0.02 ab 0.18–0.23 | 18.85 ± 1.61 d 16.11–20.78 | 2.88 ± 0.34 ab 2.32–3.24 |
| Total | 10.59 ± 0.74 9.74–11.67 | 12.97 ± 0.36 12.40–13.49 | 64.82 ± 2.79 61.29–69.57 | 75.12 ± 4.58 69.31–82.11 | 19.73 ± 0.67 18.43–20.54 | 21.19 ± 0.75 20.37–22.33 | 224.76 ± 9.64 210.23–236.68 | 216.86 ± 8.20 205.82–227.41 |
| PTWI/TWI/TDI (µg) [8,39,40,41,42] | 20.7 | 26.4 | NA * | NA * | 33 (THg) | 42.3 (THg) | 754 | 962 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koch, W.; Czop, M.; Iłowiecka, K.; Nawrocka, A.; Wiącek, D. Dietary Intake of Toxic Heavy Metals with Major Groups of Food Products—Results of Analytical Determinations. Nutrients 2022, 14, 1626. https://doi.org/10.3390/nu14081626
Koch W, Czop M, Iłowiecka K, Nawrocka A, Wiącek D. Dietary Intake of Toxic Heavy Metals with Major Groups of Food Products—Results of Analytical Determinations. Nutrients. 2022; 14(8):1626. https://doi.org/10.3390/nu14081626
Chicago/Turabian StyleKoch, Wojciech, Marcin Czop, Katarzyna Iłowiecka, Agnieszka Nawrocka, and Dariusz Wiącek. 2022. "Dietary Intake of Toxic Heavy Metals with Major Groups of Food Products—Results of Analytical Determinations" Nutrients 14, no. 8: 1626. https://doi.org/10.3390/nu14081626
APA StyleKoch, W., Czop, M., Iłowiecka, K., Nawrocka, A., & Wiącek, D. (2022). Dietary Intake of Toxic Heavy Metals with Major Groups of Food Products—Results of Analytical Determinations. Nutrients, 14(8), 1626. https://doi.org/10.3390/nu14081626









