Does the Time of Day Play a Role in the Acute Effect of p-Synephrine on Fat Oxidation Rate during Exercise in Women? A Randomized, Crossover and Double-Blind Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Design
2.3. Pre-Experimental Procedure
2.4. Experimental Trials
2.5. Menstrual Cycle Phase
2.6. Statistical Analysis
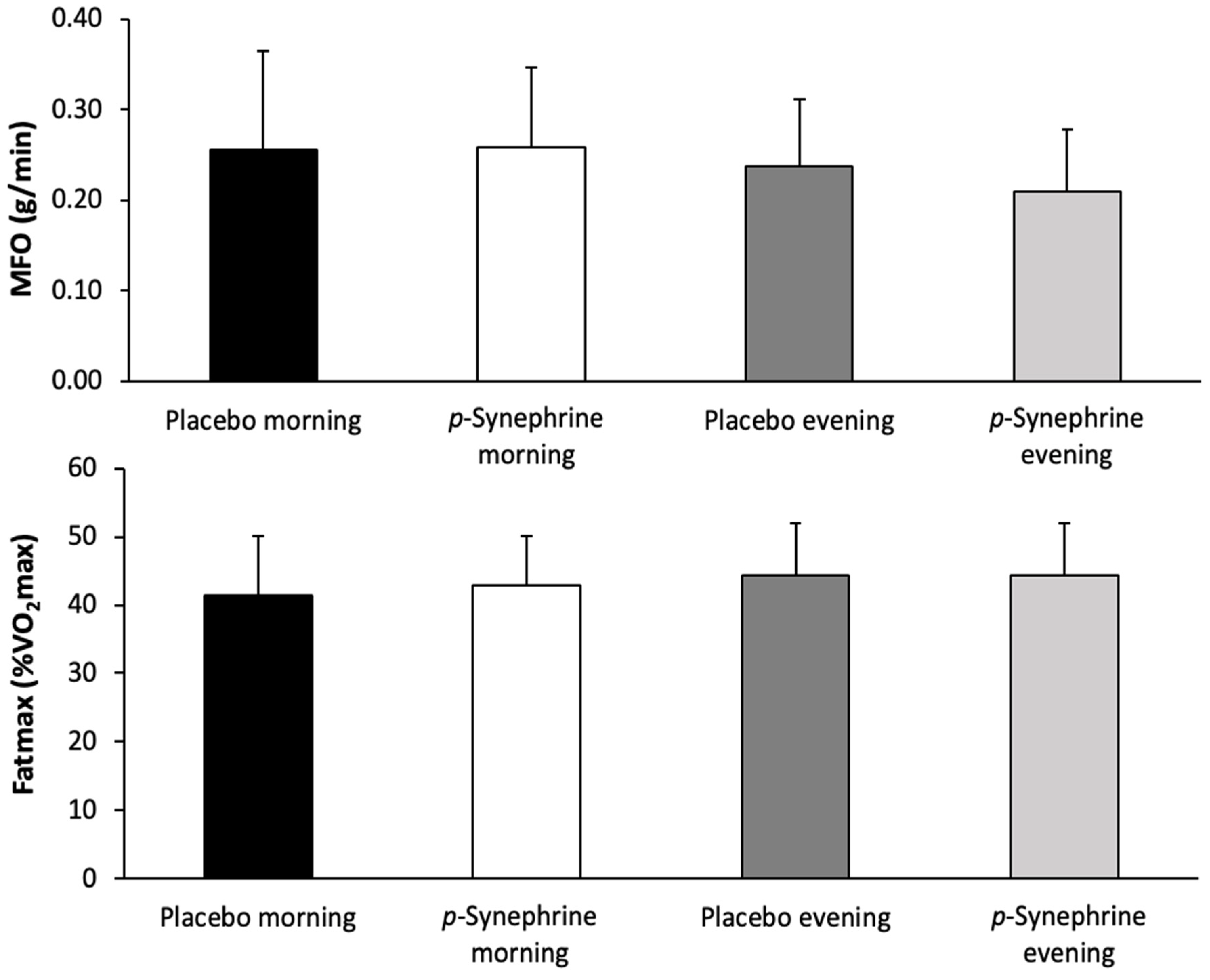
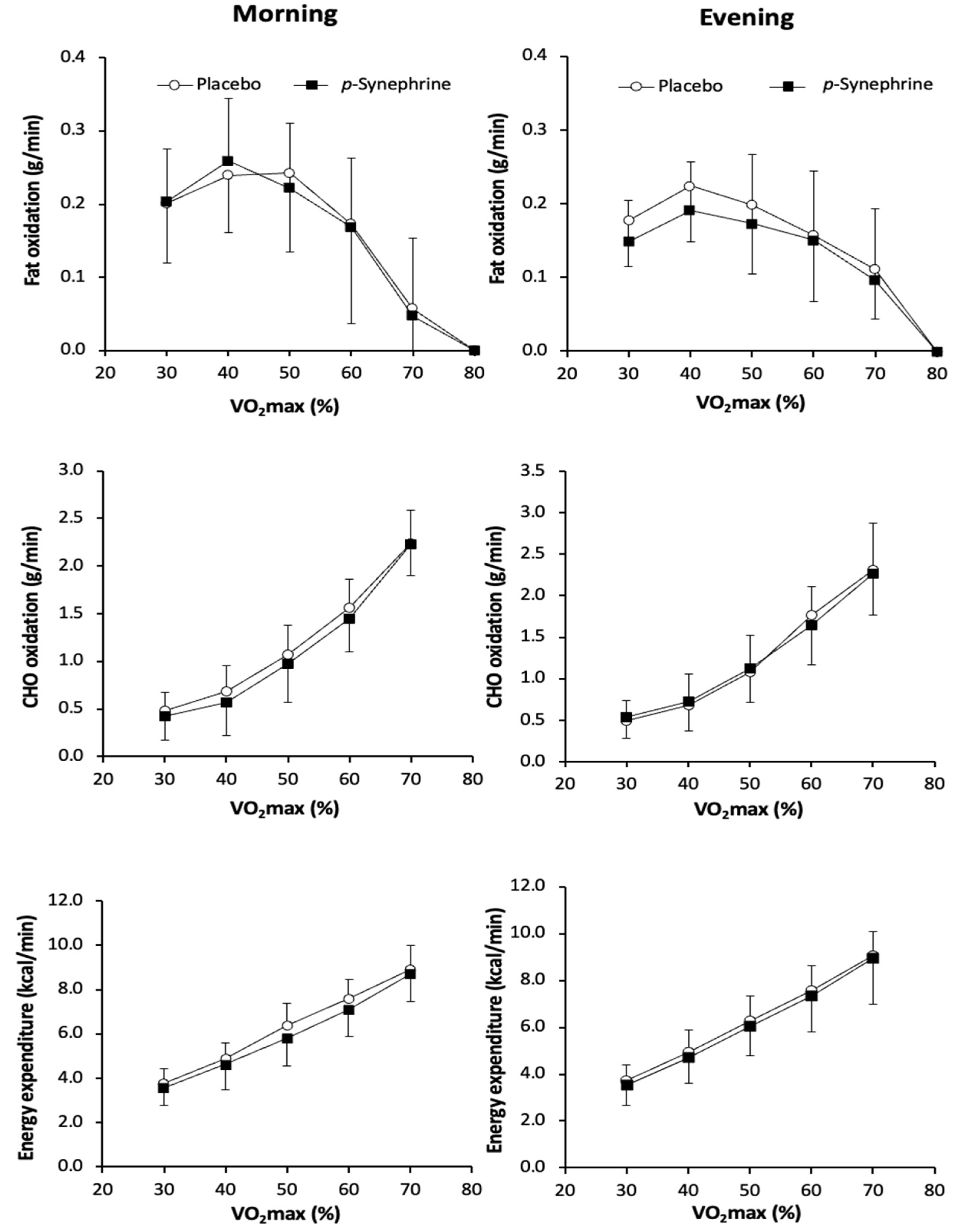
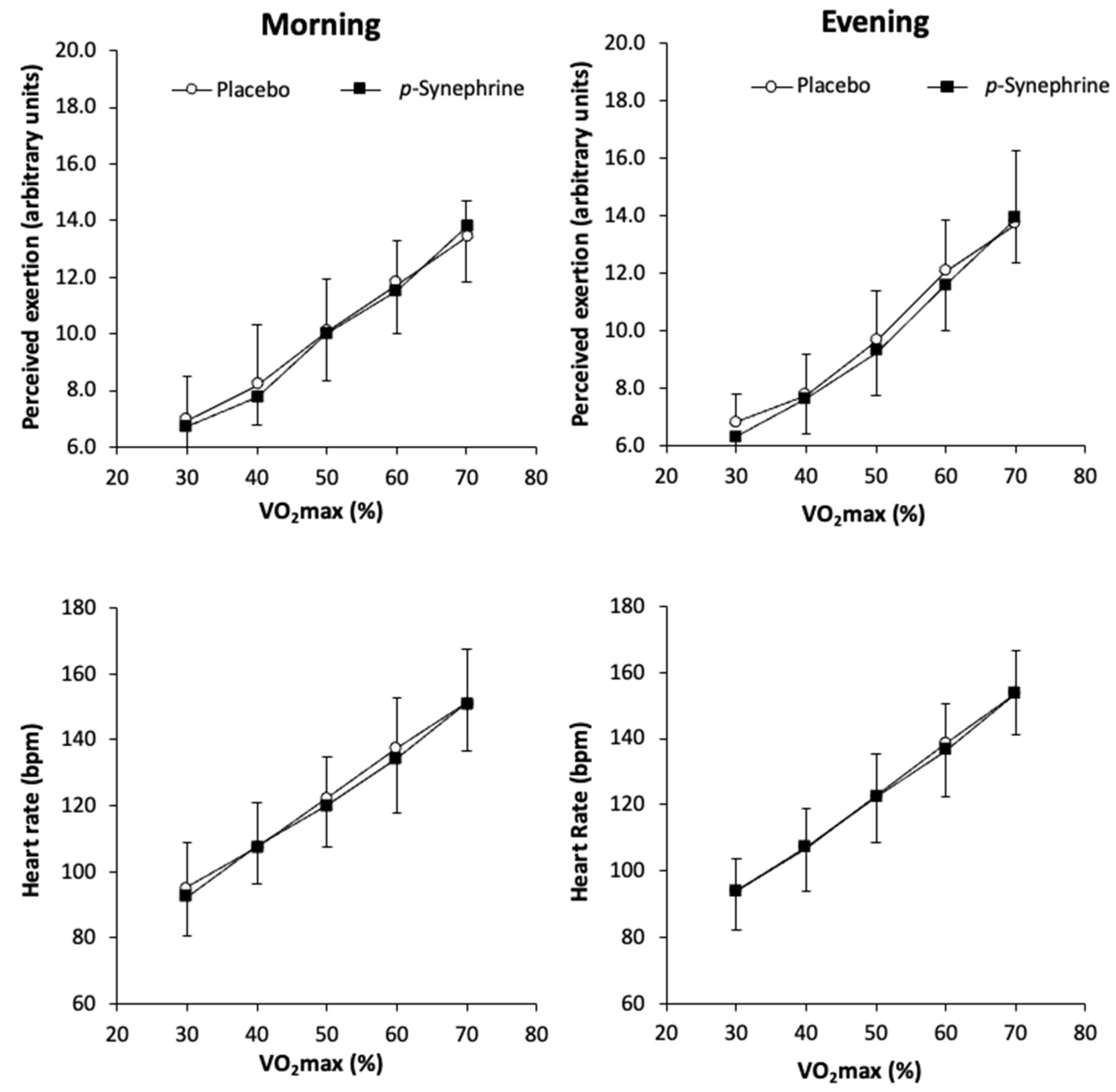
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rossato, L.G.; Costa, V.M.; Limberger, R.P.; Bastos Mde, L.; Remião, F. Synephrine: From trace concentrations to massive consumption in weight-loss. Food Chem. Toxicol. 2011, 49, 8–16. [Google Scholar] [CrossRef]
- Dragull, K.; Breksa, A.P., 3rd; Cain, B. Synephrine content of juice from Satsuma mandarins (Citrus unshiu Marcovitch). J. Agric. Food Chem. 2008, 56, 8874–8878. [Google Scholar] [CrossRef] [PubMed]
- Arbo, M.D.; Larentis, E.R.; Linck, V.M.; Aboy, A.L.; Pimentel, A.L.; Henriques, A.T.; Dallegrave, E.; Garcia, S.C.; Leal, M.B.; Limberger, R.P. Concentrations of p-synephrine in fruits and leaves of Citrus species (Rutaceae) and the acute toxicity testing of Citrus aurantium extract and p-synephrine. Food Chem. Toxicol. 2008, 46, 2770–2775. [Google Scholar] [CrossRef] [PubMed]
- Pellati, F.; Benvenuti, S. Fast high-performance liquid chromatography analysis of phenethylamine alkaloids in Citrus natural products on a pentafluorophenylpropyl stationary phase. J. Chromatogr. A 2007, 1165, 58–66. [Google Scholar] [CrossRef]
- Ruiz-Moreno, C.; Del Coso, J.; Giráldez-Costas, V.; González-García, J.; Gutiérrez-Hellín, J. Effects of p-Synephrine during Exercise: A Brief Narrative Review. Nutrients 2021, 13, 233. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Hellín, J.; Salinero, J.J.; Abían-Vicen, J.; Areces, F.; Lara, B.; Gallo, C.; Puente, C.; Del Coso, J. Acute consumption of p-synephrine does not enhance performance in sprint athletes. Appl. Physiol. Nutr. Metab. 2016, 41, 63–69. [Google Scholar] [CrossRef]
- Gutiérrez-Hellín, J.; Del Coso, J. Acute p-synephrine ingestion increases fat oxidation rate during exercise. Br. J. Clin. Pharmacol. 2016, 82, 362–368. [Google Scholar] [CrossRef]
- Gutiérrez-Hellín, J.; Baltazar-Martins, G.; Rodríguez, I.; Lara, B.; Ruiz-Moreno, C.; Aguilar-Navarro, M.; Del Coso, J. p-Synephrine, the main protoalkaloid of Citrus aurantium, raises fat oxidation during exercise in elite cyclists. Eur. J. Sport Sci. 2021, 21, 1273–1282. [Google Scholar] [CrossRef]
- Gutiérrez-Hellín, J.; Del Coso, J. Dose-Response Effects of p-Synephrine on Fat Oxidation Rate During Exercise of Increasing Intensity. Phytother. Res. 2018, 32, 370–374. [Google Scholar] [CrossRef]
- Gutiérrez-Hellín, J.; Del Coso, J. Effects of p-Synephrine and Caffeine Ingestion on Substrate Oxidation during Exercise. Med. Sci. Sports Exerc. 2018, 50, 1899–1906. [Google Scholar] [CrossRef]
- Gutiérrez-Hellín, J.; Ruiz-Moreno, C.; Del Coso, J. Acute p-synephrine ingestion increases whole-body fat oxidation during 1-h of cycling at Fatmax. Eur. J. Nutr. 2020, 59, 3341–3345. [Google Scholar] [CrossRef]
- Gutiérrez-Hellín, J.; Aguilar-Navarro, M.; Ruiz-Moreno, C.; Muñoz, A.; Amaro-Gahete, F.J.; Posada-Ayala, M.; López-Samanes, Á.; Del Coso, J.; Varillas-Delgado, D. Effect of p-Synephrine on Fat Oxidation Rate during Exercise of Increasing Intensity in Healthy Active Women. Nutrients 2022, 14, 4352. [Google Scholar] [CrossRef]
- Darvakh, H.; Nikbakht, M.; Shakerian, S.; Sadat Mousavian, A. Effect of Circadian Rhythm on Peak of Maximal Fat Oxidation on Non-Athletic Men. Zahedan J. Res. Med. Sci. 2014, 16, 8–11. [Google Scholar]
- Amaro-Gahete, F.J.; Jurado-Fasoli, L.; Triviño, A.R.; Sanchez-Delgado, G.; De-la, O.A.; Helge, J.W.; Ruiz, J.R. Diurnal Variation of Maximal Fat-Oxidation Rate in Trained Male Athletes. Int. J. Sports Physiol. Perform. 2019, 14, 1140–1146. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Maldonado, M.; Jurado-Fasoli, L.; Del Coso, J.; Jonatan, R.R.; Amaro-Gahete, F.J. Caffeine increases maximal fat oxidation during a graded exercise test: Is there a diurnal variation? J. Int. Soc. Sports Nutr. 2021, 18, 5. [Google Scholar] [CrossRef] [PubMed]
- Robles-González, L.; Aguilar-Navarro, M.; López-Samanes, Á.; Ruiz-Moreno, C.; Muñoz, A.; Varillas-Delgado, D.; Gutiérrez-Hellín, J.; Helge, J.W.; Ruiz, J.R.; Amaro-Gahete, F.J. No diurnal variation is present in maximal fat oxidation during exercise in young healthy women: A cross-over study. Eur. J. Sport Sci. 2022, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Horne, J.A.; Ostberg, O. A self-assessment questionnaire to determine morningness-eveningness in human circadian rhythms. Int. J. Chronobiol. 1976, 4, 97–110. [Google Scholar]
- Tanaka, H.; Monahan, K.D.; Seals, D.R. Age-predicted maximal heart rate revisited. J. Am. Coll. Cardiol. 2001, 37, 153–156. [Google Scholar] [CrossRef]
- Ruíz-Moreno, C.; Gutiérrez-Hellín, J.; González-García, J.; GiráLdez-Costas, V.; Brito de Souza, D.; Del Coso, J. Effect of ambient temperature on fat oxidation during an incremental cycling exercise test. Eur. J. Sport Sci. 2021, 21, 1140–1147. [Google Scholar] [CrossRef]
- Jeukendrup, A.E.; Wallis, G.A. Measurement of substrate oxidation during exercise by means of gas exchange measurements. Int. J. Sports Med. 2005, 26 (Suppl. 1), S28–S37. [Google Scholar] [CrossRef]
- Robles-González, L.; Gutiérrez-Hellín, J.; Aguilar-Navarro, M.; Ruiz-Moreno, C.; Muñoz, A.; Del-Coso, J.; Jonatan, R.R.; Amaro-Gahete, F.J. Inter-Day Reliability of Resting Metabolic Rate and Maximal Fat Oxidation during Exercise in Healthy Men Using the Ergostik Gas Analyzer. Nutrients 2021, 13, 4308. [Google Scholar] [CrossRef]
- Amaro-Gahete, F.J.; Sanchez-Delgado, G.; Alcantara, J.M.A.; Martinez-Tellez, B.; Acosta, F.M.; Helge, J.W.; Ruiz, J.R. Impact of data analysis methods for maximal fat oxidation estimation during exercise in sedentary adults. Eur. J. Sport Sci. 2019, 19, 1230–1239. [Google Scholar] [CrossRef]
- Frayn, K.N. Calculation of substrate oxidation rates in vivo from gaseous exchange. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1983, 55, 628–634. [Google Scholar] [CrossRef]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef]
- Salinero, J.J.; Lara, B.; Abian-Vicen, J.; Gonzalez-Millán, C.; Areces, F.; Gallo-Salazar, C.; Ruiz-Vicente, D.; Del Coso, J. The use of energy drinks in sport: Perceived ergogenicity and side effects in male and female athletes. Br. J. Nutr. 2014, 112, 1494–1502. [Google Scholar] [CrossRef]
- Janse de Jonge, X.A. Effects of the menstrual cycle on exercise performance. Sports Med. 2003, 33, 833–851. [Google Scholar] [CrossRef]
- Pokora, I.; Wolowski, L.; Wyderka, P. The effect of a single dose of the Thermo Speed Extreme (Olimp) thermogenic supplement on circulatory functions and body temperatures at rest in male and female subjects. Balt. J. Health Phys. Act. 2019, 11, 11–25. [Google Scholar] [CrossRef]
- Gougeon, R.; Harrigan, K.; Tremblay, J.F.; Hedrei, P.; Lamarche, M.; Morais, J.A. Increase in the thermic effect of food in women by adrenergic amines extracted from citrus aurantium. Obes Res 2005, 13, 1187–1194. [Google Scholar] [CrossRef] [PubMed]
- Mohebbi, H.; Azizi, M.; Tabari, E. Effect of time of day on MFO and Fatmax during exercise in obese and normal weight women. Facta universitatis. Ser. Phys. Educ. Sport 2011, 9, 69–79. [Google Scholar]
- Kim, H.K.; Konishi, M.; Takahashi, M.; Tabata, H.; Endo, N.; Numao, S.; Lee, S.K.; Kim, Y.H.; Suzuki, K.; Sakamoto, S. Effects of Acute Endurance Exercise Performed in the Morning and Evening on Inflammatory Cytokine and Metabolic Hormone Responses. PLoS ONE 2015, 10, e0137567. [Google Scholar] [CrossRef]
- Teo, W.; Newton, M.J.; McGuigan, M.R. Circadian rhythms in exercise performance: Implications for hormonal and muscular adaptation. J. Sports Sci. Med. 2011, 10, 600–606. [Google Scholar]
- Drust, B.; Waterhouse, J.; Atkinson, G.; Edwards, B.; Reilly, T. Circadian rhythms in sports performance—An update. Chronobiol. Int. 2005, 22, 21–44. [Google Scholar] [CrossRef]
- Ayala, V.; Martínez-Bebia, M.; Latorre, J.A.; Gimenez-Blasi, N.; Jimenez-Casquet, M.J.; Conde-Pipo, J.; Bach-Faig, A.; Mariscal-Arcas, M. Influence of circadian rhythms on sports performance. Chronobiol. Int. 2021, 38, 1522–1536. [Google Scholar] [CrossRef] [PubMed]
- Blaak, E. Gender differences in fat metabolism. Curr. Opin. Clin. Nutr. Metab. Care 2001, 4, 499–502. [Google Scholar] [CrossRef] [PubMed]
- Costill, D.L.; Daniels, J.; Evans, W.; Fink, W.; Krahenbuhl, G.; Saltin, B. Skeletal muscle enzymes and fiber composition in male and female track athletes. J. Appl. Physiol. 1976, 40, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Howald, H.; Hoppeler, H.; Claassen, H.; Mathieu, O.; Straub, R. Influences of endurance training on the ultrastructural composition of the different muscle fiber types in humans. Pflugers Arch. 1985, 403, 369–376. [Google Scholar] [CrossRef]
- Horton, T.J.; Pagliassotti, M.J.; Hobbs, K.; Hill, J.O. Fuel metabolism in men and women during and after long-duration exercise. J. Appl. Physiol. 1998, 85, 1823–1832. [Google Scholar] [CrossRef]
- Friedmann, B.; Kindermann, W. Energy metabolism and regulatory hormones in women and men during endurance exercise. Eur. J. Appl. Physiol. Occup. Physiol. 1989, 59, 1–9. [Google Scholar] [CrossRef]
- Oosthuyse, T.; Bosch, A.N. The effect of the menstrual cycle on exercise metabolism: Implications for exercise performance in eumenorrhoeic women. Sports Med. 2010, 40, 207–227. [Google Scholar] [CrossRef]
- D’Eon, T.M.; Sharoff, C.; Chipkin, S.R.; Grow, D.; Ruby, B.C.; Braun, B. Regulation of exercise carbohydrate metabolism by estrogen and progesterone in women. Am. J. Physiol. Endocrinol. Metab. 2002, 283, E1046–E1055. [Google Scholar] [CrossRef]
- Frandsen, J.; Pistoljevic, N.; Quesada, J.P.; Amaro-Gahete, F.J.; Ritz, C.; Larsen, S.; Dela, F.; Helge, J.W. Menstrual cycle phase does not affect whole body peak fat oxidation rate during a graded exercise test. J. Appl. Physiol. 2020, 128, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Stohs, S.J.; Preuss, H.G.; Shara, M. A review of the human clinical studies involving Citrus aurantium (bitter orange) extract and its primary protoalkaloid p-synephrine. Int. J. Med. Sci. 2012, 9, 527–538. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, N.S.; Stohs, S.J.; Magar, C.C.; Kale, A.; Sowmya, B. Bitter orange (Citrus aurantium L.) extract subchronic 90-day safety study in rats. Toxicol. Rep. 2017, 4, 598–613. [Google Scholar] [CrossRef]
- Bordenave, S.; Flavier, S.; Fédou, C.; Brun, J.F.; Mercier, J. Exercise calorimetry in sedentary patients: Procedures based on short 3 min steps underestimate carbohydrate oxidation and overestimate lipid oxidation. Diabetes Metab. 2007, 33, 379–384. [Google Scholar] [CrossRef] [PubMed]
| Variable (Units) | Mean ± SD | Range |
|---|---|---|
| Age (yr) | 26.7 ± 8.7 | 18.0–48.0 |
| Body mass (kg) | 61.6± 9.1 | 47.9–83.5 |
| Body height (cm) | 166.0 ± 7.0 | 152.0–175.0 |
| Fat mass (%) | 24.2 ± 5.8 | 15.0–39.2 |
| VO2max (mL/kg/min) | 39.4 ± 6.6 | 30.4–50.2 |
| Maximal heart rate (beats/min) | 179.0 ± 6.1 | 170–189 |
| Maximal wattage in VO2max test (w) | 207.3 ± 30.7 | 170–275 |
| Variable (Units) | Placebo Morning | p-Synephrine Morning | Placebo Evening | p-Synephrine Evening |
|---|---|---|---|---|
| Nervousness (a.u.) | 1.2 ± 0.6 | 1.6 ± 1.4 | 1.3 ± 0.9 | 1.5 ± 0.7 |
| Vigor (a.u.) | 1.4 ± 0.7 | 1.8 ± 2.3 | 2.1 ± 1.7 | 1.9 ± 1.7 |
| Irritability (a.u.) | 1.2 ± 0.6 | 1.8 ± 1.1 | 1.7 ± 1.7 | 1.2 ± 0.4 |
| Muscle pain (a.u.) | 1.3 ± 0.8 | 2.3 ± 1.5 | 2.1 ± 1.4 | 2.1 ± 1.8 |
| Gastrointestinal distress (a.u.) | 1.2 ± 0.6 | 1.7 ± 1.8 | 1.0 ± 0.3 | 1.2 ± 0.4 |
| Diuresis (a.u.) | 1.5 ± 0.8 | 1.3 ± 0.6 | 2.1 ± 2.1 | 1.6 ± 1.7 |
| Insomnia (a.u.) | 3.2 ± 2.7 | 2.1 ± 2.3 | 2.5 ± 2.9 | 2.6 ± 2.7 |
| Variable (Units) | Placebo Morning | p-Synephrine Morning | Placebo Evening | p-Syneprhine Evening |
|---|---|---|---|---|
| Urine p-synephrine concentration (µg/L) | 102.22 ± 22.23 | 23,531 ± 24,074 | 65.31 ± 25.99 | 17,106 ± 16,252 |
| Urine 4-hydroxymandelic acid concentration (µg/L) | 0.40 ± 0.01 | 3.03 ± 5.24 | 0.65 ± 0.31 | 1.31 ± 1.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gutiérrez-Hellín, J.; Del Coso, J.; Aguilar-Navarro, M.; Varillas-Delgado, D.; Ruiz-Moreno, C.; López-Samanés, Á.; Amaro-Gahete, F.J.; Muñoz, A. Does the Time of Day Play a Role in the Acute Effect of p-Synephrine on Fat Oxidation Rate during Exercise in Women? A Randomized, Crossover and Double-Blind Study. Nutrients 2022, 14, 5030. https://doi.org/10.3390/nu14235030
Gutiérrez-Hellín J, Del Coso J, Aguilar-Navarro M, Varillas-Delgado D, Ruiz-Moreno C, López-Samanés Á, Amaro-Gahete FJ, Muñoz A. Does the Time of Day Play a Role in the Acute Effect of p-Synephrine on Fat Oxidation Rate during Exercise in Women? A Randomized, Crossover and Double-Blind Study. Nutrients. 2022; 14(23):5030. https://doi.org/10.3390/nu14235030
Chicago/Turabian StyleGutiérrez-Hellín, Jorge, Juan Del Coso, Millán Aguilar-Navarro, David Varillas-Delgado, Carlos Ruiz-Moreno, Álvaro López-Samanés, Francisco J. Amaro-Gahete, and Alejandro Muñoz. 2022. "Does the Time of Day Play a Role in the Acute Effect of p-Synephrine on Fat Oxidation Rate during Exercise in Women? A Randomized, Crossover and Double-Blind Study" Nutrients 14, no. 23: 5030. https://doi.org/10.3390/nu14235030
APA StyleGutiérrez-Hellín, J., Del Coso, J., Aguilar-Navarro, M., Varillas-Delgado, D., Ruiz-Moreno, C., López-Samanés, Á., Amaro-Gahete, F. J., & Muñoz, A. (2022). Does the Time of Day Play a Role in the Acute Effect of p-Synephrine on Fat Oxidation Rate during Exercise in Women? A Randomized, Crossover and Double-Blind Study. Nutrients, 14(23), 5030. https://doi.org/10.3390/nu14235030













