A Randomized Controlled Trial of Fasting and Lifestyle Modification in Patients with Metabolic Syndrome: Effects on Patient-Reported Outcomes
Abstract
:1. Introduction
2. Methods
2.1. Design
2.2. Participants
2.3. Interventions
2.4. Fasting and Lifestyle Modification (F + LM)
2.5. Lifestyle Modification (LM)
2.6. Randomization
2.7. Outcome Measures
- Quality of life on the Medical Outcomes Study 36-Item-Short-Form (SF-36) including eight dimensions of health: physical functioning (10 items), social role functioning (2 items), physical role functioning (4 items), emotional role functioning (3 items), mental health (5 items), vitality (4 items), bodily pain (2 items), and general health perceptions (5 items), and the physical and mental component score [36].
- Anxiety and depression on the Hospital Anxiety and Depression Scale (HADS) [37], a 14-item scale for the assessment of anxiety and depression symptoms.
- Stress on the Cohen Perceived Stress Scale (CPSS) for the assessment of subjective stress within the last week including 14 items about current levels of experienced and perceived stress [38].
- Self-efficacy on the 10-item General Self-Efficacy Scale (GSE) [41]. Self-efficacy is a construct that “reflects an optimistic self-belief that one can master a difficult task or cope with adversity” [41]. Self-efficacy makes it easier to set goals, make efforts, persevere in the face of obstacles, and recover from setbacks. It can be considered a resource for resilience.
- Mindfulness on the 15-item Mindfulness Attention Awareness Scale (MAAS) [42]. Mindfulness is characterized by the following three qualities: (1) intentional, (2) related to the present moment, and (3) nonjudgmental.
- Self-compassion on the 26-Item Self-Compassion Scale (SCS) [43]. Self-compassion refers to a positive basic attitude toward oneself even in difficult life situations. This personality trait has proven to be an effective protective factor that promotes emotional resilience.
2.8. Sample Size Calculation and Statistical Analysis
3. Results
3.1. Patients
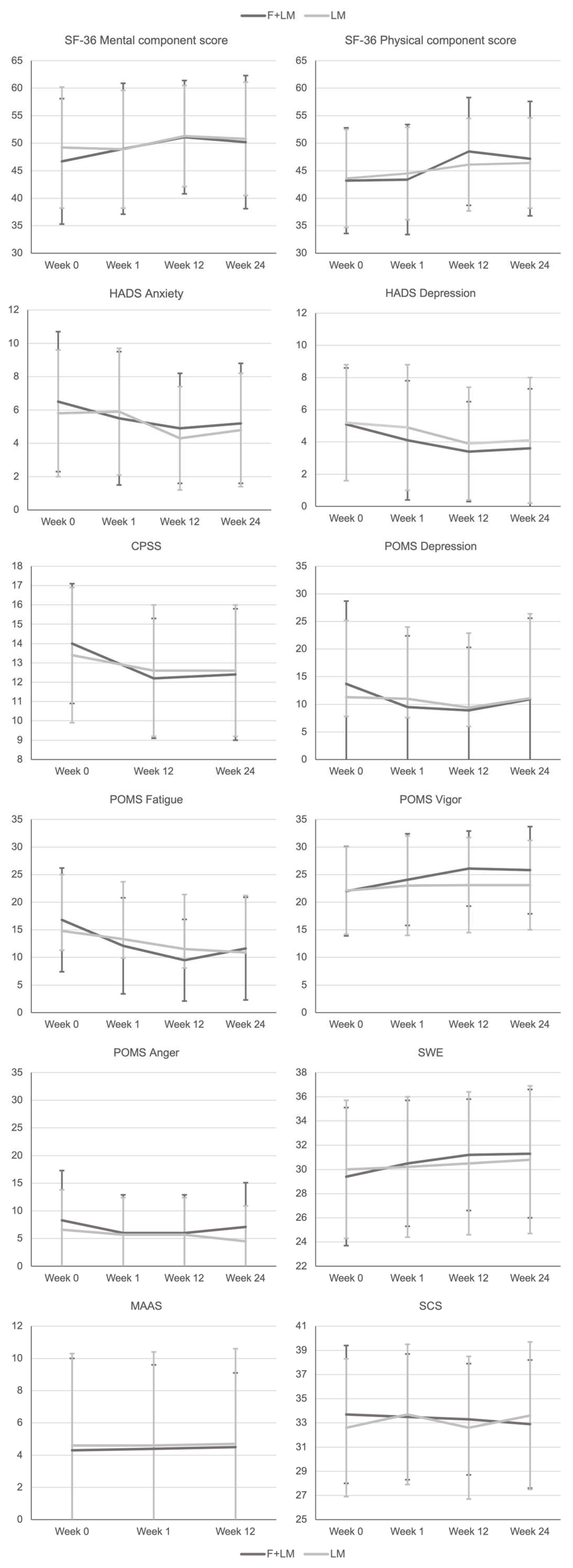
3.2. Outcome Measures
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Eckel, R.H.; Alberti, K.; Grundy, S.M.; Zimmet, P.Z. The metabolic syndrome. Lancet 2010, 375, 181–183. [Google Scholar] [CrossRef] [PubMed]
- Saklayen, M.G. The Global Epidemic of the Metabolic Syndrome. Curr. Hypertens. Rep. 2018, 20, 12. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, A.; Laurent, S.; Cucca, F.; Cockcroft, J.; Cunha, P.G.; Mañas, L.R.; Mattace Raso, F.U.; Muiesan, M.L.; Ryliškytė, L.; Rietzschel, E.; et al. Metabolic syndrome across Europe: Different clusters of risk factors. Eur. J. Prev. Cardiol. 2015, 22, 486–491. [Google Scholar] [CrossRef]
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019: Update From the GBD 2019 Study. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef]
- Rosengren, A.; Hawken, S.; Ounpuu, S.; Sliwa, K.; Zubaid, M.; Almahmeed, W.A.; Blackett, K.N.; Sitthi-amorn, C.; Sato, H.; Yusuf, S. Association of psychosocial risk factors with risk of acute myocardial infarction in 11119 cases and 13648 controls from 52 countries (the INTERHEART study): Case-control study. Lancet 2004, 364, 953–962. [Google Scholar] [CrossRef]
- Kotseva, K.; De Backer, G.; De Bacquer, D.; Ryden, L.; Hoes, A.; Grobbee, D.; Maggioni, A.; Marques-Vidal, P.; Jennings, C.; Abreu, A.; et al. Lifestyle and impact on cardiovascular risk factor control in coronary patients across 27 countries: Results from the European Society of Cardiology ESC-EORP EUROASPIRE V registry. Eur. J. Prev. Cardiol. 2019, 26, 824–835. [Google Scholar] [CrossRef] [PubMed]
- Inoue, K.; Horwich, T.; Bhatnagar, R.; Bhatt, K.; Goldwater, D.; Seeman, T.; Watson, K.E. Urinary Stress Hormones, Hypertension, and Cardiovascular Events: The Multi-Ethnic Study of Atherosclerosis. Hypertension 2021, 78, 1640–1647. [Google Scholar] [CrossRef]
- Marcos-Delgado, A.; López-García, E.; Martínez-González, M.A.; Salas-Salvadó, J.; Corella, D.; Fitó, M.; Romaguera, D.; Vioque, J.; Alonso-Gómez, A.M.; Wärnberg, J.; et al. Health-related quality of life in individuals with metabolic syndrome: A cross-sectional study. Semergen 2020, 46, 524–537. [Google Scholar] [CrossRef]
- Saboya, P.P.; Bodanese, L.C.; Zimmermann, P.R.; Gustavo, A.D.; Assumpção, C.M.; Londero, F. Metabolic syndrome and quality of life: A systematic review. Rev. Lat. Am. Enferm. 2016, 24, e2848. [Google Scholar] [CrossRef]
- Ornish, D.; Brown, S.E.; Scherwitz, L.W.; Billings, J.H.; Armstrong, W.T.; Ports, T.A.; McLanahan, S.M.; Kirkeeide, R.L.; Brand, R.J.; Gould, K.L. Can lifestyle changes reverse coronary heart disease? The Lifestyle Heart Trial. Lancet 1990, 336, 129–133. [Google Scholar]
- Cramer, H.; Lauche, R.; Paul, A.; Langhorst, J.; Michalsen, A.; Dobos, G. MindñBody Medicine in the Secondary Prevention of Coronary Heart Disease. Dtsch Arztebl Int. 2015, 112, 759–767. [Google Scholar] [PubMed] [Green Version]
- Lean, M.E.; Leslie, W.S.; Barnes, A.C.; Brosnahan, N.; Thom, G.; McCombie, L.; Peters, C.; Zhyzhneuskaya, S.; Al-Mrabeh, A.; Hollingsworth, K.G.; et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): An open-label, cluster-randomised trial. Lancet 2018, 391, 541–551. [Google Scholar] [CrossRef] [PubMed]
- Michalsen, A.; Grossman, P.; Lehmann, N.; Knoblauch, N.T.; Paul, A.; Moebus, S.; Budde, T.; Dobos, G.J. Psychological and quality-of-life outcomes from a comprehensive stress reduction and lifestyle program in patients with coronary artery disease: Results of a randomized trial. Psychother. Psychosom. 2005, 74, 344–352. [Google Scholar] [CrossRef]
- Lin, C.H.; Chiang, S.L.; Tzeng, W.C.; Chiang, L.C. Systematic review of impact of lifestyle-modification programs on metabolic risks and patient-reported outcomes in adults with metabolic syndrome. Worldviews Evid.-Based Nurs. 2014, 11, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Dobos, G.; Paul, A. Mind-Body-Medizin—Integrative Konzepte zur Ressourcenstärkung und Lebensstilveränderung; Urban & Fischer Verlag/Elsevier GmbH: Munich, Germany, 2019. [Google Scholar]
- Kabat-Zinn, J. Full Catastrophe Living: Using the Wisdom of Your Body and Mind to Face Stress, Pain, and Illness; Delta Trade Paperback/Bantam Dell: New York, NY, USA, 1990. [Google Scholar]
- Bishop, S.R.; Lau, M.; Shapiro, S.; Carlson, L.; Anderson, N.D.; Carmody, J.; Segal, Z.V.; Abbey, S.; Speca, M.; Velting, D.; et al. Mindfulness: A proposed operational definition. Clin. Psychol. Sci. Pract. 2004, 11, 230–241. [Google Scholar]
- Chu, P.; Gotink, R.A.; Yeh, G.Y.; Goldie, S.J.; Hunink, M.G. The effectiveness of yoga in modifying risk factors for cardiovascular disease and metabolic syndrome: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Prev. Cardiol. 2016, 23, 291–307. [Google Scholar] [CrossRef]
- Cramer, H.; Thoms, M.S.; Anheyer, D.; Lauche, R.; Dobos, G. Yoga in Women with Abdominal Obesity: A Randomized Controlled Trial. Dtsch. Arztebl. Int. 2016, 113, 645–652. [Google Scholar] [CrossRef]
- Chau, J.P.C.; Leung, L.Y.L.; Liu, X.; Lo, S.H.S.; Choi, K.C.; Zhao, J.; Chiang, H.C.Y. Effects of Tai Chi on health outcomes among community-dwelling adults with or at risk of metabolic syndrome: A systematic review. Complement. Ther. Clin. Pract. 2021, 44, 101445. [Google Scholar] [CrossRef]
- Wei, M.; Brandhorst, S.; Shelehchi, M.; Mirzaei, H.; Cheng, C.W.; Budniak, J.; Groshen, S.; Mack, W.J.; Guen, E.; Di Biase, S.; et al. Fasting-mimicking diet and markers/risk factors for aging, diabetes, cancer, and cardiovascular disease. Sci. Transl. Med. 2017, 9, eaai8700. [Google Scholar] [CrossRef]
- Mattson, M.P.; Longo, V.D.; Harvie, M. Impact of intermittent fasting on health and disease processes. Ageing Res. Rev. 2017, 39, 46–58. [Google Scholar] [CrossRef]
- Fanti, M.; Mishra, A.; Longo, V.D.; Brandhorst, S. Time-Restricted Eating, Intermittent Fasting, and Fasting-Mimicking Diets in Weight Loss. Curr. Obes. Rep. 2021, 10, 70–80. [Google Scholar] [CrossRef]
- Longo, V.D.; Mattson, M.P. Fasting: Molecular mechanisms and clinical applications. Cell Metab. 2014, 19, 181–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watkins, E.; Serpell, L. The Psychological Effects of Short-Term Fasting in Healthy Women. Front. Nutr. 2016, 3, 27. [Google Scholar] [CrossRef] [PubMed]
- Hussin, N.M.; Shahar, S.; Teng, N.I.; Ngah, W.Z.; Das, S.K. Efficacy of fasting and calorie restriction (FCR) on mood and depression among ageing men. J. Nutr. Health Aging 2013, 17, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Yang, C.; Wu, R.; Wu, M.; Liu, W.; Dai, Z.; Li, Y. How Experiences Affect Psychological Responses During Supervised Fasting: A Preliminary Study. Front. Psychol. 2021, 12, 651760. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomized trials. Ann. Intern. Med. 2010, 152, 726–732. [Google Scholar] [CrossRef]
- Paul, A.; Lauche, R.; Cramer, H.; Altner, N.; Dobos, G. An Integrative Day-Care Clinic for chronically ill patients: Concept and case presentation. Eur. J. Integr. Med. 2012, 4, E455–E459. [Google Scholar]
- de Lorgeril, M.; Renaud, S.; Mamelle, N.; Salen, P.; Martin, J.L.; Monjaud, I.; Guidollet, J.; Touboul, P.; Delaye, J. Mediterranean alpha-linolenic acid-rich diet in secondary prevention of coronary heart disease. Lancet 1994, 343, 1454–1459. [Google Scholar]
- De Lorgeril, M.; Salen, P.; Martin, J.L.; Mamelle, N.; Monjaud, I.; Touboul, P.; Delaye, J. Effect of a mediterranean type of diet on the rate of cardiovascular complications in patients with coronary artery disease. Insights into the cardioprotective effect of certain nutriments. J. Am. Coll. Cardiol. 1996, 28, 1103–1108. [Google Scholar] [CrossRef]
- Esposito, K.; Marfella, R.; Ciotola, M.; Di Palo, C.; Giugliano, F.; Giugliano, G.; D’Armiento, M.; D’Andrea, F.; Giugliano, D. Effect of a mediterranean-style diet on endothelial dysfunction and markers of vascular inflammation in the metabolic syndrome: A randomized trial. JAMA 2004, 292, 1440–1446. [Google Scholar]
- Kempner, W. Treatment of hypertensive vascular disease with rice diet. Arch. Intern. Med. 1974, 133, 758–790. [Google Scholar] [PubMed]
- Saghaei, M. Random allocation software for parallel group randomized trials. BMC Med. Res. Methodol. 2004, 4, 26. [Google Scholar]
- Maifeld, A.; Bartolomaeus, H.; Löber, U.; Avery, E.G.; Steckhan, N.; Markó, L.; Wilck, N.; Hamad, I.; Šušnjar, U.; Mähler, A.; et al. Fasting alters the gut microbiome reducing blood pressure and body weight in metabolic syndrome patients. Nat. Commun. 2021, 12, 1970. [Google Scholar] [CrossRef]
- Bullinger, M. German translation and psychometric testing of the SF-36 Health Survey: Preliminary results from the IQOLA Project. International Quality of Life Assessment. Soc. Sci. Med. 1995, 41, 1359–1366. [Google Scholar] [CrossRef]
- Herrmann-Lingen, C.; Buss, U.; Snaith, R. Hospital Anxiety and Depression Scale—Deutsche Version (HADS-D); 3., aktualisierte und neu normierte Auflage; Hans Huber: Bern, Switzerland, 2011. [Google Scholar]
- Cohen, S.; Kamarck, T.; Mermelstein, R. A global measure of perceived stress. J. Health Soc. Behav. 1983, 24, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Albani, C.; Blaser, G.; Geyer, M.; Schmutzer, G.; Brahler, E.; Bailer, H.; Grulke, N. The German short version of “Profile of Mood States” (POMS): Psychometric evaluation in a representative sample. Psychother. Psychosom. Med. Psychol. 2005, 55, 324–330. [Google Scholar] [CrossRef] [PubMed]
- McNair, D.; Lorr, M.; Droppleman, L.; Biel, B.; Dangel, S. Profile of mood states states (Dt. Bearbeitung). In Collegium interantionale Psychiatriae Scalarum; Scalarum, C.I.P., Ed.; Beltz Verlag: Weinheim, UK, 1981. [Google Scholar]
- Schwarzer, R.; Jerusalem, M. Generalized Self-Efficacy scale. In Measures in Health Psychology: A User’s Portfolio; Weinman, J., Wright, S., Johnston, M., Eds.; NFER-NELSON: Windsor, UK, 1995. [Google Scholar]
- Michalak, J.; Heidenreich, T.; Ströhle, G.; Nachtigall, C. Die deutsche Version der Mindful Attention and Awareness Scale (MAAS) Psychometrische Befunde zu einem Achtsamkeitsfragebogen. Z. Klin. Psychol. Psychother. 2008, 37, 200–208. [Google Scholar] [CrossRef]
- Hupfeld, J.; Ruffieux, N. Validierung einer deutschen Version der Self-Compassion Scale (SCS-D). Z. Klin. Psychol. Psychother. 2011, 40, 115–123. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar]
- Ornish, D.; Scherwitz, L.W.; Billings, J.H.; Brown, S.E.; Gould, K.L.; Merritt, T.A.; Sparler, S.; Armstrong, W.T.; Ports, T.A.; Kirkeeide, R.L.; et al. Intensive lifestyle changes for reversal of coronary heart disease. JAMA 1998, 280, 2001–2007. [Google Scholar]
- Cramer, H.; Lauche, R.; Haller, H.; Steckhan, N.; Michalsen, A.; Dobos, G. Effects of yoga on cardiovascular disease risk factors: A systematic review and meta-analysis. Int. J. Cardiol. 2014, 173, 170–183. [Google Scholar] [CrossRef]
- Hughes, J.W.; Fresco, D.M.; Myerscough, R.; van Dulmen, M.H.; Carlson, L.E.; Josephson, R. Randomized controlled trial of mindfulness-based stress reduction for prehypertension. Psychosom. Med. 2013, 75, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Ciotola, M.; Giugliano, D. Mediterranean diet and the metabolic syndrome. Mol. Nutr. Food Res. 2007, 51, 1268–1274. [Google Scholar] [CrossRef] [PubMed]
- Rubin, D.B. Multiple Imputation for Nonresponse in Surveys; John Wiley & Sons: New York, NY, USA, 1987. [Google Scholar]
- Schafer, J.L. Analysis of Incomplete Multivariate Data; Chapman and Hall: New York, NY, USA, 1997. [Google Scholar]
- Michalsen, A.; Schlegel, F.; Rodenbeck, A.; Lüdtke, R.; Huether, G.; Teschler, H.; Dobos, G.J. Effects of short-term modified fasting on sleep patterns and daytime vigilance in non-obese subjects: Results of a pilot study. Ann. Nutr. Metab. 2003, 47, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Michalsen, A. Prolonged fasting as a method of mood enhancement in chronic pain syndromes: A review of clinical evidence and mechanisms. Curr. Pain. Headache Rep. 2010, 14, 80–87. [Google Scholar] [CrossRef]
- Fond, G.; Macgregor, A.; Leboyer, M.; Michalsen, A. Fasting in mood disorders: Neurobiology and effectiveness. A review of the literature. Psychiatry Res. 2013, 209, 253–258. [Google Scholar] [CrossRef]
- Maruish, M.; Maruish, M.; Kosinski, M.; Bjorner, J.; Gandek, B.; Turner-Bowker, D.; Ware, J. User’s Manual for the SF-36v2 Survey; Quality Metric Incorporated: Lincoln, RI, USA, 2011. [Google Scholar]
- Lemay, K.R.; Tulloch, H.E.; Pipe, A.L.; Reed, J.L. Establishing the Minimal Clinically Important Difference for the Hospital Anxiety and Depression Scale in Patients With Cardiovascular Disease. J. Cardiopulm. Rehabil. Prev. 2019, 39, E6–E11. [Google Scholar] [CrossRef]
- Cramer, H.; Langhorst, J.; Dobos, G.; Lauche, R. Yoga for metabolic syndrome: A systematic review and meta-analysis. Eur. J. Prev. Cardiol. 2016, 23, 1982–1993. [Google Scholar] [CrossRef]
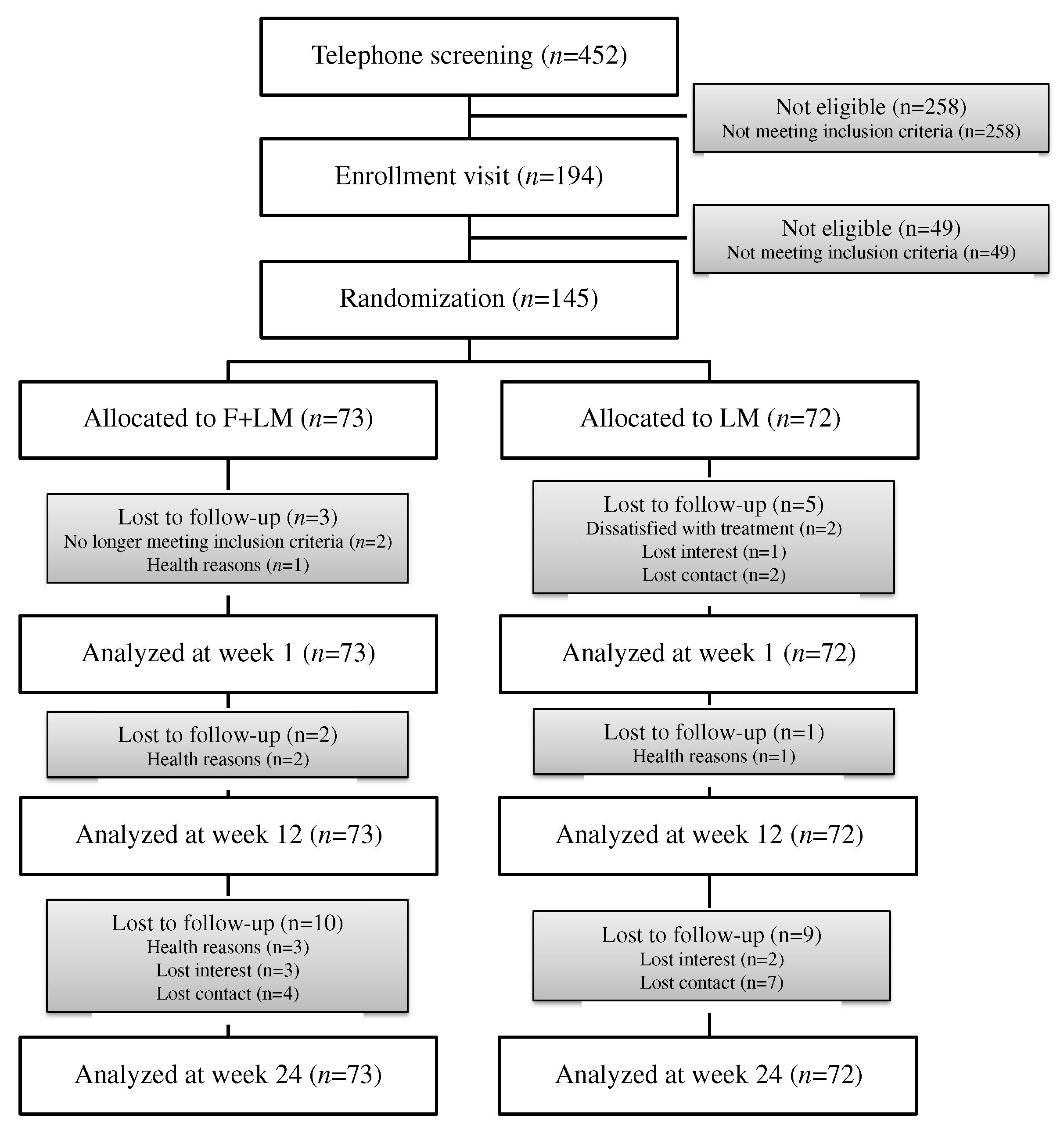
| Total (n = 145) | F + LM (n = 73) | LM (n = 72) | |
|---|---|---|---|
| Sociodemographic characteristics | |||
| Gender female n (%) | 91 (62.8%) | 48 (65.8%) | 43 (59.7%) |
| Age years | 59.7 ± 9.3 | 58.6 ± 10.8 | 60.8 ± 7.5 |
| Marital status n (%) | |||
| Single | 15 (10.3%) | 6 (8.2%) | 9 (12.5%) |
| Married | 98 (67.6%) | 49 (67.1%) | 49 (68.1%) |
| Divorced | 21 (14.5%) | 12 (16.4%) | 9 (12.5%) |
| Widowed | 5 (3.4%) | 3 (4.1%) | 2 (2.8%) |
| Education n (%) | |||
| Secondary modern school (“Hauptschule”) qualification | 25 (17.2%) | 9 (12.3%) | 16 (22.2%) |
| High school (“Realschule”) qualification | 41 (28.3%) | 19 (26.0%) | 22 (30.6%) |
| A level (“Abitur”) | 18 (12.4%) | 12 (16.4%) | 6 (8.3%) |
| University degree | 53 (36.6%) | 28 (38.3%) | 25 (34.7%) |
| Employment n (%) | |||
| Employed full-time | 41 (28.3%) | 20 (27.4%) | 21 (29.2%) |
| Employed part-time | 19 (13.1%) | 11 (15.1%) | 8 (11.1%) |
| Occasionally | 5 (3.4%) | 3 (4.1%) | 2 (2.8%) |
| On sick leave | 3 (2.1%) | 2 (2.7%) | 1 (1.4%) |
| Unemployed | 3 (2.1%) | 2 (2.7%) | 1 (1.4%) |
| Retired age-related | 44 (30.3%) | 20 (27.4%) | 24 (33.3%) |
| Retired health-related | 15 (10.3%) | 7 (9.6%) | 8 (11.1%) |
| Homekeeper | 10 (6.9%) | 6 (8.2%) | 4 (5.6%) |
| Clinical characteristics | |||
| Weight (kg) | 97.0 ± 15.8 | 98.1 ± 16.1 | 95.9 ± 15.5 |
| Body Mass Index (kg/m2) | 33.3 ± 4.5 | 33.7 ± 4.5 | 32.8 ± 4.5 |
| Clinical systolic BP (mmHg) | 140.0 ± 16.8 | 138.9 ± 14.4 | 141.2 ± 19.0 |
| Clinical diastolic BP (mmHg) | 88.9 ± 10.9 | 88.3 ± 10.6 | 89.5 ± 11.2 |
| Waist circumference (cm) | 113.1 ± 10.8 | 114.1 ± 10.5 | 112.1 ± 11.1 |
| HDL cholesterol (mg/dL) | 55.0 ± 17.6 | 53.4 ± 16.0 | 56.6 ± 19.0 |
| Triglyceride (mg/dL) | 181.8 ± 194.0 | 188.0 ± 210.6 | 175.5 ± 111.1 |
| Blood glucose (mg/dL) | 111.7 ± 20.5 | 113.3 ± 18.9 | 110.1 ± 22.0 |
| Week 0 | Week 1 | Week 12 | Week 24 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | Group | Mean ± SD | Mean ± SD | Pwithin | Pbetween | Cohen’s d | Mean ± SD | Pwithin | Pbetween | Cohen’s d | Mean ± SD | Pwithin | Pbetween | Cohen’s d |
| SF-36 Physical functioning | F + LM | 73.3 ± 17.8 | 75.8 ± 18.8 | 0.077 | 0.858 | 0.03 | 83.1 ± 17.4 | <0.001 | 0.324 | 0.15 | 82.9 ± 16.5 | <0.001 | 0.541 | 0.10 |
| LM | 74.2 ± 20.6 | 75.9 ± 21.2 | 0.118 | 81.4 ± 19.1 | <0.001 | 81.2 ± 22.5 | <0.001 | |||||||
| SF-36 Physical role functioning | F + LM | 61.0 ± 40.0 | 58.1 ± 39.0 | 0.453 | 0.244 | 0.21 | 78.0 ± 33.3 | <0.001 | 0.104 | 0.31 | 69.2 ± 37.5 | 0.092 | 0.756 | 0.06 |
| LM | 63.2 ± 41.1 | 65.0 ± 38.9 | 0.595 | 70.5 ± 36.0 | 0.059 | 68.1 ± 38.3 | 0.147 | |||||||
| SF-36 Bodily pain | F + LM | 56.8 ± 22.9 | 57.3 ± 24.0 | 0.812 | 0.119 | 0.28 | 69.6 ± 24.0 | <0.001 | 0.238 | 0.28 | 64.1 ± 24.5 | 0.006 | 0.545 | 0.13 |
| LM | 62.0 ± 26.8 | 63.0 ± 25.6 | 0.654 | 65.0 ± 29.2 | 0.337 | 69.1 ± 27.7 | 0.008 | |||||||
| SF-36 General health perceptions | F + LM | 58.3 ± 16.8 | 63.5 ± 17.1 | 0.001 | 0.238 | 0.27 | 70.1 ± 17.7 | <0.001 | 0.370 | 0.20 | 69.3 ± 16.8 | <0.001 | 0.527 | 0.14 |
| LM | 58.1 ± 16.7 | 59.5 ± 18.4 | 0.367 | 65.9 ± 19.0 | <0.001 | 66.1 ± 19.1 | <0.001 | |||||||
| SF-36 Vitality | F + LM | 55.0 ± 19.7 | 58.4 ± 19.6 | 0.014 | 0.695 | 0.05 | 67.7 ± 17.0 | <0.001 | 0.121 | 0.25 | 63.5 ± 20.2 | <0.001 | 0.678 | 0.08 |
| LM | 56.8 ± 21.4 | 58.4 ± 21.0 | 0.192 | 63.5 ± 21.0 | <0.001 | 61.8 ± 23.7 | 0.004 | |||||||
| SF-36 Social role functioning | F + LM | 70.4 ± 27.8 | 75.9 ± 27.7 | 0.023 | 0.722 | 0.07 | 83.7 ± 23.2 | <0.001 | 0.176 | 0.25 | 81.0 ± 24.0 | <0.001 | 0.922 | 0.02 |
| LM | 80.7 ± 23.4 | 79.4 ± 25.3 | 0.514 | 82.2 ± 24.6 | 0.493 | 83.3 ± 21.0 | 0.275 | |||||||
| SF-36 Emotional role functioning | F + LM | 68.9 ± 37.8 | 70.9 ± 39.1 | 0.605 | 0.495 | 0.13 | 77.3 ± 31.9 | 0.030 | 0.504 | 0.12 | 77.1 ± 33.7 | 0.012 | 0.957 | 0.01 |
| LM | 72.2 ± 39.6 | 71.1 ± 39.6 | 0.722 | 80.4 ± 34.4 | 0.036 | 80.5 ± 34.2 | 0.017 | |||||||
| SF-36 Mental health | F + LM | 66.9 ± 18.8 | 71.5 ± 19.5 | 0.002 | 0.444 | 0.12 | 75.6 ± 16.8 | <0.001 | 0.128 | 0.23 | 73.0 ± 20.2 | <0.001 | 0.887 | 0.03 |
| LM | 70.7 ± 17.8 | 71.4 ± 18.2 | 0.536 | 75.5 ± 15.2 | 0.001 | 74.0 ± 18.2 | 0.027 | |||||||
| SF-36 Physical component score | F + LM | 43.2 ± 8.9 | 43.4 ± 8.4 | 0.739 | 0.131 | 0.24 | 48.5 ± 8.4 | <0.001 | 0.177 | 0.26 | 47.2 ± 8.2 | <0.001 | 0.990 | 0.01 |
| LM | 43.6 ± 9.6 | 44.5 ± 10.0 | 0.223 | 46.1 ± 9.8 | 0.006 | 46.4 ± 10.4 | 0.002 | |||||||
| SF-36 Mental component score | F + LM | 46.7 ± 11.4 | 49.0 ± 11.9 | 0.005 | 0.129 | 0.22 | 51.1 ± 10.3 | <0.001 | 0.096 | 0.24 | 50.2 ± 12.1 | <0.001 | 0.594 | 0.10 |
| LM | 49.2 ± 11.0 | 48.9 ± 10.7 | 0.580 | 51.3 ± 9.2 | 0.017 | 50.8 ± 10.3 | 0.065 | |||||||
| HADS Anxiety | F + LM | 6.5 ± 4.2 | 5.5 ± 4.0 | <0.001 | 0.303 | 0.16 | 4.9 ± 3.3 | <0.001 | 0.227 | 0.18 | 5.2 ± 3.6 | <0.001 | 0.206 | 0.21 |
| LM | 5.8 ± 3.8 | 5.9 ± 3.8 | 0.751 | 4.3 ± 3.1 | <0.001 | 4.8 ± 3.4 | 0.004 | |||||||
| HADS Depression | F + LM | 5.1 ± 3.5 | 4.1 ± 3.7 | <0.001 | 0.386 | 0.15 | 3.4 ± 3.1 | <0.001 | 0.432 | 0.13 | 3.6 ± 3.7 | <0.001 | 0.740 | 0.07 |
| LM | 5.2 ± 3.6 | 4.9 ± 3.9 | 0.367 | 3.9 ± 3.5 | <0.001 | 4.1 ± 3.9 | 0.004 | |||||||
| CPSS | F + LM | 14.0 ± 3.1 | NA | NA | NA | NA | 12.2 ± 3.1 | <0.001 | 0.070 | 0.33 | 12.4 ± 3.4 | <0.001 | 0.064 | 0.37 |
| LM | 13.4 ± 3.5 | NA | NA | 12.6 ± 3.4 | 0.005 | 12.6 ± 3.4 | 0.020 | |||||||
| POMS Depression | F + LM | 13.7 ± 15.0 | 9.5 ± 12.9 | <0.001 | 0.049 | 0.30 | 8.9 ± 11.4 | <0.001 | 0.397 | 0.11 | 10.9 ± 14.7 | 0.010 | 0.504 | 0.12 |
| LM | 11.3 ± 13.9 | 11.0 ± 13.0 | 0.747 | 9.4 ± 13.5 | 0.065 | 11.1 ± 15.3 | 0.856 | |||||||
| POMS Fatigue | F + LM | 16.8 ± 9.4 | 12.1 ± 8.7 | <0.001 | 0.014 | 0.50 | 9.5 ± 7.4 | <0.001 | 0.206 | 0.23 | 11.6 ± 9.3 | <0.001 | 0.549 | 0.12 |
| LM | 14.8 ± 10.2 | 13.3 ± 10.4 | 0.096 | 11.5 ± 9.9 | 0.001 | 10.9 ± 10.3 | <0.001 | |||||||
| POMS Vigor | F + LM | 22.0 ± 8.1 | 24.1 ± 8.3 | 0.010 | 0.629 | 0.10 | 26.1 ± 6.8 | <0.001 | 0.033 | 0.45 | 25.8 ± 7.9 | <0.001 | 0.147 | 0.30 |
| LM | 22.1 ± 7.9 | 23.0 ± 9.0 | 0.264 | 23.1 ± 8.6 | 0.278 | 23.1 ± 8.1 | 0.260 | |||||||
| POMS Anger | F + LM | 8.3 ± 9.0 | 6.0 ± 6.9 | 0.001 | 0.089 | 0.27 | 6.0 ± 6.9 | 0.001 | 0.412 | 0.14 | 7.1 ± 8.0 | 0.087 | 0.192 | 0.23 |
| LM | 6.6 ± 7.2 | 5.7 ± 6.7 | 0.138 | 5.7 ± 6.7 | 0.279 | 4.5 ± 6.4 | 0.010 | |||||||
| GSE | F + LM | 29.4 ± 5.7 | 30.5 ± 5.2 | 0.020 | 0.244 | 0.17 | 31.2 ± 4.6 | <0.001 | 0.206 | 0.19 | 31.3 ± 5.3 | <0.001 | 0.262 | 0.19 |
| LM | 30.0 ± 5.7 | 30.2 ± 5.8 | 0.553 | 30.5 ± 5.9 | 0.170 | 30.8 ± 6.1 | 0.061 | |||||||
| MAAS | F + LM | 4.3 ± 0.9 | NA | NA | NA | NA | 4.4 ± 0.8 | 0.030 | 0.696 | 0.06 | 4.5 ± 0.8 | 0.001 | 0.265 | 0.18 |
| LM | 4.6 ± 0.7 | NA | NA | 4.6 ± 0.7 | 0.183 | 4.7 ± 0.7 | 0.169 | |||||||
| SCS | F + LM | 33.7 ± 5.5 | 33.5 ± 4.9 | 0.702 | 0.618 | 0.09 | 33.3 ± 4.5 | 0.503 | 0.698 | 0.06 | 32.9 ± 5.0 | 0.060 | 0.007 | 0.41 |
| LM | 32.6 ± 6.2 | 33.7 ± 4.5 | 0.110 | 32.6 ± 5.0 | 0.978 | 33.6 ± 4.6 | 0.057 | |||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeitler, M.; Lauche, R.; Hohmann, C.; Choi, K.-E.; Schneider, N.; Steckhan, N.; Rathjens, F.; Anheyer, D.; Paul, A.; von Scheidt, C.; et al. A Randomized Controlled Trial of Fasting and Lifestyle Modification in Patients with Metabolic Syndrome: Effects on Patient-Reported Outcomes. Nutrients 2022, 14, 3559. https://doi.org/10.3390/nu14173559
Jeitler M, Lauche R, Hohmann C, Choi K-E, Schneider N, Steckhan N, Rathjens F, Anheyer D, Paul A, von Scheidt C, et al. A Randomized Controlled Trial of Fasting and Lifestyle Modification in Patients with Metabolic Syndrome: Effects on Patient-Reported Outcomes. Nutrients. 2022; 14(17):3559. https://doi.org/10.3390/nu14173559
Chicago/Turabian StyleJeitler, Michael, Romy Lauche, Christoph Hohmann, Kyung-Eun (Anna) Choi, Nadia Schneider, Nico Steckhan, Florian Rathjens, Dennis Anheyer, Anna Paul, Christel von Scheidt, and et al. 2022. "A Randomized Controlled Trial of Fasting and Lifestyle Modification in Patients with Metabolic Syndrome: Effects on Patient-Reported Outcomes" Nutrients 14, no. 17: 3559. https://doi.org/10.3390/nu14173559
APA StyleJeitler, M., Lauche, R., Hohmann, C., Choi, K.-E., Schneider, N., Steckhan, N., Rathjens, F., Anheyer, D., Paul, A., von Scheidt, C., Ostermann, T., Schneider, E., Koppold-Liebscher, D., Kessler, C. S., Dobos, G., Michalsen, A., & Cramer, H. (2022). A Randomized Controlled Trial of Fasting and Lifestyle Modification in Patients with Metabolic Syndrome: Effects on Patient-Reported Outcomes. Nutrients, 14(17), 3559. https://doi.org/10.3390/nu14173559





_Choi.png)

