Association between Obesity, Overweight, Elevated Waist Circumference, and Insulin Resistance Markers among Brazilian Adolescent Students
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Sample
2.2. Anthropometric Measures
2.3. Biochemical Assays
2.4. Outcome Definitions
2.5. Statistical Analysis
3. Results
3.1. Description of the Study Population
3.2. Comparison of Association Strengths of Adiposity Variables within Each Insulin Resistance Marker (TyG and TG/HDL)
3.2.1. TyG
3.2.2. TG/HDL
3.3. Comparison between Insulin Resistance Markers Using FIeq
3.3.1. Obesity
3.3.2. Overweight
3.3.3. Elevated WC
3.4. Secondary Analysis with Poisson Regression and the 75th Percentile Value as the Cut-Off Value for Two Insulin Resistance Markers (TyG and TG/HDL)
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Reaven, G.M. Banting Lecture 1988. Role of insulin resistance in human disease. Diabetes 1988, 37, 1595–1607. [Google Scholar]
- Reaven, G. The metabolic syndrome or the insulin resistance syndrome? Different names, different concepts, and different goals. Endocrinol. Metab. Clin. N. Am. 2004, 33, 283–303. [Google Scholar] [CrossRef]
- Kotlyarevska, K.; Wolfgram, P.; Lee, J.M. Is waist circumference a better predictor of insulin resistance than body mass index in U.S. adolescents? J. Adolesc. Health 2011, 49, 330–333. [Google Scholar] [CrossRef]
- Stern, S.E.; Williams, K.; Ferrannini, E.; DeFronzo, R.A.; Bogardus, C.; Stern, M.P. Identification of individuals with insulin resistance using routine clinical measurements. Diabetes 2005, 54, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Krebs, N.F.; Himes, J.H.; Jacobson, D.; Nicklas, T.A.; Guilday, P.; Styne, D. Assessment of child and adolescent overweight and obesity. Pediatrics 2007, 120 (Suppl. 4), S193–S228. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Bacha, F.; Gungor, N.; Arslanian, S.A. Waist circumference is an independent predictor of insulin resistance in black and white youths. J. Pediatr. 2006, 148, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Deusdará, R.; de Souza, A.M.; Szklo, M. Obesity, overweight, elevated waist circumference and Insulin differ in their associations with insulin resistance markers- the ERICA study. Metab.—Clin. Exp. 2022, 128, 155103. [Google Scholar] [CrossRef]
- Michaliszyn, S.F.; Arslanian, S. Techniques to Assess Insulin Action in Youth. In Insulin Resistance—Childhood Precursors of Adult Disease; Zeitler, P.S., Nadeau, K.J., Eds.; Springer: Berlin/Heidelberg, Germany; Nature: Cham, Switerzerland, 2020. [Google Scholar]
- Mohd Nor, N.S.; Lee, S.J.; Bacha, F.; Tfayli, H.; Arslanian, S. Triglyceride glucose index as a surrogate measure of insulin sensitivity in obese adolescents with normoglycemia, prediabetes, and type 2 diabetes mellitus: Comparison with the hyperinsulinemic–euglycemic clamp. Pediatr. Diabetes 2016, 17, 458–465. [Google Scholar] [CrossRef]
- Gungor, N.; Saad, R.; Janosky, J.; Arslanian, S. Validation of surrogate estimates of insulin sensitivity and insulin secretion in children and adolescents. J. Pediatr. 2004, 144, 47–55. [Google Scholar] [CrossRef]
- Antuna-Puente, B.; Disse, E.; Rabasa-Lhoret, R.; Laville, M.; Capeau, J.; Bastard, J.P. How can we measure insulin sensitivity/resistance? Diabetes Metab. 2011, 37, 179–188. [Google Scholar] [CrossRef]
- Sharrett, A.R.; Coady, S.A.; Folsom, A.R.; Couper, D.J.; Heiss, G. Smoking and diabetes differ in their associations with subclinical atherosclerosis and coronary heart disease—The ARIC Study. Atherosclerosis 2004, 172, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Sharrett, A.R.; Ding, J.; Criqui, M.H.; Saad, M.F.; Liu, K.; Polak, J.F.; Folsom, A.R.; Tsai, M.Y.; Burke, G.L.; Szklo, M. Smoking, diabetes, and blood cholesterol differ in their associations with subclinical atherosclerosis: The Multiethnic Study of Atherosclerosis (MESA). Atherosclerosis 2006, 186, 441–447. [Google Scholar] [CrossRef] [PubMed]
- De Vasconcellos, M.T.L.; do Silva, P.L.N.; Szklo, M.; Kuschnir, M.C.C.; Klein, C.H.; Abreu, G.d.; Barufaldi, L.A.; Bloch, K.V. Sampling design for the Study of Cardiovascular Risks in Adolescents (ERICA). Cad. Saude Publica 2015, 31, 921–930. [Google Scholar] [CrossRef] [PubMed]
- Bloch, K.V.; Szklo, M.; Kuschnir, M.C.C.; de Abreu, G.A.; Barufaldi, L.A.; Klein, C.H.; de Vasconcelos, M.T.; da Veiga, G.V.; Figueiredo, V.C.; Dias, A.; et al. The study of cardiovascular risk in adolescents—ERICA: Rationale, design and sample characteristics of a national survey examining cardiovascular risk factor profile in Brazilian adolescents. BMC Public Health 2015, 15, 94. [Google Scholar] [CrossRef]
- Da Silva, T.L.N.; Klein, C.H.; De Moura Souza, A.; Barufaldi, L.A.; De Azevedo Abreu, G.; Kuschnir, M.C.C.; de Vasconcellos, M.T.L.; Bloch, K.V. Response rate in the study of cardiovascular risks in adolescents—ERICA. Rev. Saude Publica 2016, 50, 1s–13s. [Google Scholar] [CrossRef]
- De Onis, M.; Onyango, A.W.; Borghi, E.; Siyam, A.; Siekmann, J. Development of a WHO growth reference for school-aged children and adolescents. Bull. World Health Organ. 2007, 660–667. [Google Scholar] [CrossRef]
- Alberti, S.G.; Zimmet, P. The IDF Consensus Definition of the Metablic Syndrome in Children and Adolescents; IDF Communication: Brussels, Belgium, 2007; ISBN 2-930229-49-7. [Google Scholar]
- Cureau, F.V.; Bloch, K.V.; Henz, A.; Schaan, C.W.; Klein, C.H.; de Oliveira, C.L.; Giannini, D.T.; de Leon, E.B.; de Abreu, G.A.; Telo, G.H.; et al. Challenges for conducting blood collection and biochemical analysis in a large multicenter school-based study with adolescents: Lessons from ERICA in Brazil. Cad. Saude Publica 2017, 33, e00122816. [Google Scholar] [CrossRef]
- Simental-MendíaI, L.E.; Rodríguez-Moran, M.; Guerrero-Romero, F. The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab. Syndr. Relat. Disord. 2008, 6, 299–304. [Google Scholar] [CrossRef]
- McLaughlin, T.; Abbasi, F.; Cheal, K.; Chu, J.; Lamendola, C.; Reaven, G. Use of metabolic markers to identify overweight individuals who are insulin resistant. Ann. Intern. Med. 2003, 139, 802–809. [Google Scholar] [CrossRef]
- Lawlor, D.A.; Benfield, L.; Logue, J.; Tilling, K.; Howe, L.D.; Fraser, A.; Cherry, L.; Watt, P.; Ness, A.R.; Smith, G.D.; et al. Association between general and central adiposity in childhood, and change in these, with cardiovascular risk factors in adolescence: Prospective cohort study. BMJ 2010, 341, 1205. [Google Scholar] [CrossRef]
- Arslanian, S.; Suprasongsin, C. Insulin Sensitivity, Lipids, and Body Composition in Childhood: Is “Syndrome X” Present? J. Clin. Endocrinol. Metab. 1996, 81, 1058–1062. [Google Scholar] [CrossRef] [PubMed]
- Janssen, I.; Heymsfield, S.B.; Allison, D.B.; Kotler, D.P.; Ross, R. Body mass index and waist circumference independently contribute to the prediction of nonabdominal, abdominal subcutaneous, and visceral fat. Am. J. Clin. Nutr. 2002, 75, 683–688. [Google Scholar] [CrossRef] [PubMed]
- Rothman, K.J. BMI-related errors in the measurement of obesity. Int. J. Obes. 2008, 32, S56–S59. [Google Scholar] [CrossRef]
- Martin-Calvo, N.; Moreno-Galarraga, L.; Martinez-Gonzalez, M.A. Association between body mass index, waist-to-height ratio and adiposity in children: A systematic review and meta-analysis. Nutrients 2016, 8, 512. [Google Scholar] [CrossRef] [PubMed]
- Widhalm, K.; Schonegger, K.; Pietrobelli, A.; Faith, M.S.; Heymsfield, S.B. BMI: Does it really reflect body fat mass? J. Pediatr. 1999, 134, 522–523. [Google Scholar] [CrossRef]
- Boeke, C.E.; Oken, E.; Kleinman, K.P.; Rifas-Shiman, S.L.; Taveras, E.M.; Gillman, M.W. Correlations among adiposity measures in school-aged children. BMC Pediatr. 2013, 13, 99. [Google Scholar] [CrossRef]
- Du, T.; Sun, X.; Huo, R.; Yu, X. Visceral adiposity index, hypertriglyceridemic waist and risk of diabetes: The China Health and Nutrition Survey 2009. Int. J. Obes. 2014, 38, 840–847. [Google Scholar] [CrossRef]
- Bickerton, A.S.T.; Roberts, R.; Fielding, B.A.; Hodson, L.; Blaak, E.E.; Wagenmakers, A.J.M.; Gilbert, M.; Karpe, F.; Frayn, K.N. Preferential uptake of dietary fatty acids in adipose tissue and muscle in the postprandial period. Diabetes 2007, 56, 168–176. [Google Scholar] [CrossRef]
- Garg, A.; Helderman, J.H.; Koffler, M.; Ayuso, R.; Rosenstock, J.; Raskin, P. Relationship between lipoprotein levels and in vivo insulin action in normal young white men. Metabolism 1988, 37, 982–987. [Google Scholar] [CrossRef]
- Vasques, A.C.J.; Novaes, F.S.; da Saúde de Oliveira, M.; Matos Souza, J.R.; Yamanaka, A.; Pareja, J.C.; Tambascia, M.A.; Saad, M.J.A.; Geloneze, B. TyG index performs better than HOMA in a Brazilian population: A hyperglycemic clamp validated study. Diabetes Res. Clin. Pract. 2011, 93, E98–E100. [Google Scholar] [CrossRef]
- McLaughlin, T.; Lamendola, C.; Liu, A.; Abbasi, F. Preferential fat deposition in subcutaneous versus visceral depots is associated with insulin sensitivity. J. Clin. Endocrinol. Metab. 2011, 96, E1756. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Janssen, I.; Katzmarzyk, P.T.; Ross, R. Waist circumference and not body mass index explains obesity-related health risk. Am. J. Clin. Nutr. 2004, 79, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, M.A. Association between cardiovascular risk factors and triglyceride to high-density lipoprotein ratio: A single-center experience. Arch. Med. Sci.—Atheroscler. Dis. 2019, 4, 196–200. [Google Scholar] [CrossRef]
- Ren, X.; Chen, Z.A.; Zheng, S.; Han, T.; Li, Y.; Liu, W.; Hu, Y. Association between triglyceride to HDL-C Ratio (TG/HDL-C) and insulin resistance in chinese patients with newly diagnosed type 2 diabetes mellitus. PLoS ONE 2016, 11, e0154345. [Google Scholar] [CrossRef]
- Lee, S.H.; Kwon, H.S.; Park, Y.M.; Ha, H.S.; Jeong, S.H.; Yang, H.K.; Lee, J.H.; Yim, H.W.; Kang, M.I.; Lee, W.C.; et al. Predicting the development of diabetes using the product of triglycerides and glucose: The chungju metabolic disease cohort (CMC) study. PLoS ONE 2014, 9, e90430. [Google Scholar] [CrossRef]
- Iwani, N.A.K.Z.; Jalaludin, M.Y.; Zin, R.M.W.M.; Fuziah, M.Z.; Hong, J.Y.H.; Abqariyah, Y.; Mokhtar, A.H.; Wan Nazaimoon, W.M. Triglyceride to HDL-C Ratio is Associated with Insulin Resistance in Overweight and Obese Children. Sci. Rep. 2017, 7, 40055. [Google Scholar] [CrossRef]
- Musso, C.; Graffigna, M.; Soutelo, J.; Honfi, M.; Ledesma, L.; Miksztowicz, V.; Pazos, M.; Migliano, M.; Schreier, L.E.; Berg, G.A. Cardiometabolic risk factors as apolipoprotein B, triglyceride/HDL-cholesterol ratio and C-reactive protein, in adolescents with and without obesity: Cross-sectional study in middle class suburban children. Pediatr. Diabetes 2011, 12, 229–234. [Google Scholar] [CrossRef]
- Rader, D.J. Effect of Insulin Resistance, Dyslipidemia, and Intra-abdominal Adiposity on the Development of Cardiovascular Disease and Diabetes Mellitus. Am. J. Med. 2007, 120, S12–S18. [Google Scholar] [CrossRef]
- Freedman, D.S.; Serdula, M.K.; Srinivasan, S.R.; Berenson, G.S. Relation of circumferences and skinfold thicknesses to lipid and insulin concentrations in children and adolescents: The Bogalusa Heart Study. Am. J. Clin. Nutr. 1999, 69, 308–317. [Google Scholar] [CrossRef]
- Brenner, D.R.; Tepylo, K.; Eny, K.M.; Cahill, L.E.; El-Sohemy, A. Comparison of body mass index and waist circumference as predictors of cardiometabolic health in a population of young Canadian adults. Diabetol. Metab. Syndr. 2010, 2, 28. [Google Scholar] [CrossRef]
- Cao, J.; Zhang, L.; Li, J.; Sun, L.; Liu, S.; Zhang, J.; Zhao, H. Pubertal maturation and weight status are associated with dyslipidemia among children and adolescents in Northwest China. Sci. Rep. 2020, 10, 16344. [Google Scholar] [CrossRef] [PubMed]
- Loomba-Albrecht, L.A.; Styne, D.M. Effect of puberty on body composition. Curr. Opin. Endocrinol. Diabetes Obes. 2009, 16, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Gröber-Grätz, D.; Widhalm, K.; De Zwaan, M.; Reinehr, T.; Blüher, S.; Schwab, K.O.; Wiegand, S.; Holl, R.W. Body mass index or waist circumference: Which is the better predictor for hypertension and dyslipidemia in overweight/obese children and adolescents? association of cardiovascular risk related to body mass index or waist circumference. Horm. Res. Paediatr. 2013, 80, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Fox, C.S.; Hickson, D.M.A.; May, W.D.; Hairston, K.G.; Carr, J.J.; Taylor, H.A. Impact of Abdominal Visceral and Subcutaneous Adipose Tissue on Cardiometabolic Risk Factors: The Jackson Heart Study. J. Clin. Endocrinol. Metab. 2010, 95, 5419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Variables | n | |||
| Continuous | Median | 1°Q | 3°Q | |
| Age (years) | 37,815 | 15 | 13 | 16 |
| TG/HDL δ | 37,706 | 0.65 | 0.47 | 0.91 |
| TyG Φ | 37,559 | 8.0 | 7.73 | 8.28 |
| Insulin (mU/L) | 37,760 | 8.3 | 5.8 | 11.7 |
| Categorical | (%) | 95% CI | ||
| Female | 22,682 | 50.2 | - | - |
| Smoking (≥1 cigarette smoked in the last 30 days) | 1406 | 4.2 | 3.8 | 4.7 |
| Alcohol consumption (≥1 drink in the last 30 days) | 7685 | 21.6 | 20.3 | 23.0 |
| Sedentary behavior γ | 14,133 | 40.5 | 38.9 | 42.1 |
| Physical inactivity (<420 min per week) | 24,713 | 62.7 | 61.7 | 63.8 |
| Public Schools | 27,990 | 77.8 | 72.4 | 82.3 |
| Tanner Stage | ||||
| Stage 1 | 172 | 0.5 | 0.4 | 0.6 |
| Stage 2 | 1917 | 5.6 | 4.9 | 6.2 |
| Stage 3 | 6651 | 16.9 | 16.0 | 17.9 |
| Stage 4 | 14,889 | 40.0 | 38.5 | 41.5 |
| Stage 5 | 14,162 | 37.0 | 35.7 | 38.3 |
| Nutritional status | ||||
| Normal a | 27,073 | 71.0 | 69.4 | 72.5 |
| Overweight b | 6635 | 17.5 | 44.0 | 49.2 |
| Obesity c | 3097 | 9.2 | 8.5 | 10.0 |
| Waist circumference (cm) | ||||
| Normal | 33,373 | 87.4 | 86.3 | 88.4 |
| Elevated d | 4386 | 12.6 | 11.6 | 13.7 |
| Girls | ||||||||
| 12–14 years | 15–17 years | |||||||
| TyG Φ | TG/HDL δ | TyG Φ | TG/HDL δ | |||||
| Unadjusted | Adjusted | Unadjusted | Adjusted | Unadjusted | Adjusted | Unadjusted | Adjusted | |
| Overweight b,f | 0.01 (−0.04, 0.05) | 0.02 (−0.02, 0.06) | 0.03 (−0.02, 0.09) | 0.04 (−0.02, 0.09) | 0.01 (−0.04, 0.07) | 0.01 (−0.04, 0.06) | 0.01 (−0.03, 0.06) | 0.01 (−0.04, 0.05) |
| Obesity c,f | 0.19 * (0.11, 0.27) | 0.19 * (0.11, 0.27) | 0.17 * (0.03, 0.31) | 0.18 * (0.03, 0.32) | 0.00 (−0.10, 0.10) | 0.00 (−0.11, 0.10) | 0.04 (−0.06, 0.15) | 0.04 (−0.07, 0.15) |
| Elevated WC d,g | 0.02 (−0.06, 0.11) | 0.02 (−0.06, 0.11) | 0.16 * (0.04, 0.28) | 0.16 * (0.04, 0.28) | 0.08 * (0.01, 0.14) | 0.07 * (0.01, 0.14) | 0.13 * (0.06, 0.20) | 0.12 * (0.05, 0.19) |
| Insulin | 0.01 * (0.01, 0.02) | 0.01 * (0.01, 0.02) | 0.01 * (0.01, 0.02) | 0.01 * (0.01, 0.02) | 0.01 * (0.01, 0.01) | 0.01 * (0.01, 0.01) | 0.01 * (0.00, 0.01) | 0.01 * (0.00, 0.01) |
| Boys | ||||||||
| 12–14 years | 15–17 years | |||||||
| TyG Φ | TG/HDL δ | TyG Φ | TG/HDL δ | |||||
| Unadjusted | Adjusted | Unadjusted | Adjusted | Unadjusted | Adjusted | Unadjusted | Adjusted | |
| Overweight b,f | 0.15 * (0.10, 0.21) | 0.14 * (0.09, 0.19) | 0.18 * (0.12, 0.25) | 0.18 * (0.12, 0.24) | 0.01 (−0.06, 0.09) | 0.01 (−0.06, 0.09) | 0.03 (−0.04, 0.10) | 0.03 (−0.04, 0.10) |
| Obesity c,f | 0.21 * (0.12, 0.30) | 0.19 * (0.10, 0.27) | 0.33 * (0.22, 0.44) | 0.33 * (0.22, 0.43) | −0.03 (−0.21, 0.14) | −0.03 (−0.19, 0.14) | −0.02 (−0.31, 0.26) | −0.01 (−0.29, 0.26) |
| Elevated WC d,g | 0.10 * (0.00, 0.20) | 0.10 * (0.00, 0.21) | 0.10 (−0.02, 0.22) | 0.10 (−0.02, 0.22) | 0.34 * (0.16, 0.53) | 0.34 * (0.16, 0.51) | 0.58 * (0.29, 0.87) | 0.57 * (0.29, 0.85) |
| Insulin | 0.01 * (0.01, 0.02) | 0.02 * (0.01, 0.02) | 0.01 * (0.01, 0.02) | 0.01 * (0.01, 0.02) | 0.02 * (0.02, 0.03) | 0.02 * (0.02, 0.03) | 0.02 * (0.01, 0.03) | 0.02 * (0.01, 0.03) |
| Girls | ||||||||
| 12–14 years | 15–17 years | |||||||
| TyG Φ | TG/HDL δ | TyG Φ | TG/HDL δ | |||||
| Unadjusted | Adjusted | Unadjusted | Adjusted | Unadjusted | Adjusted | Unadjusted | Adjusted | |
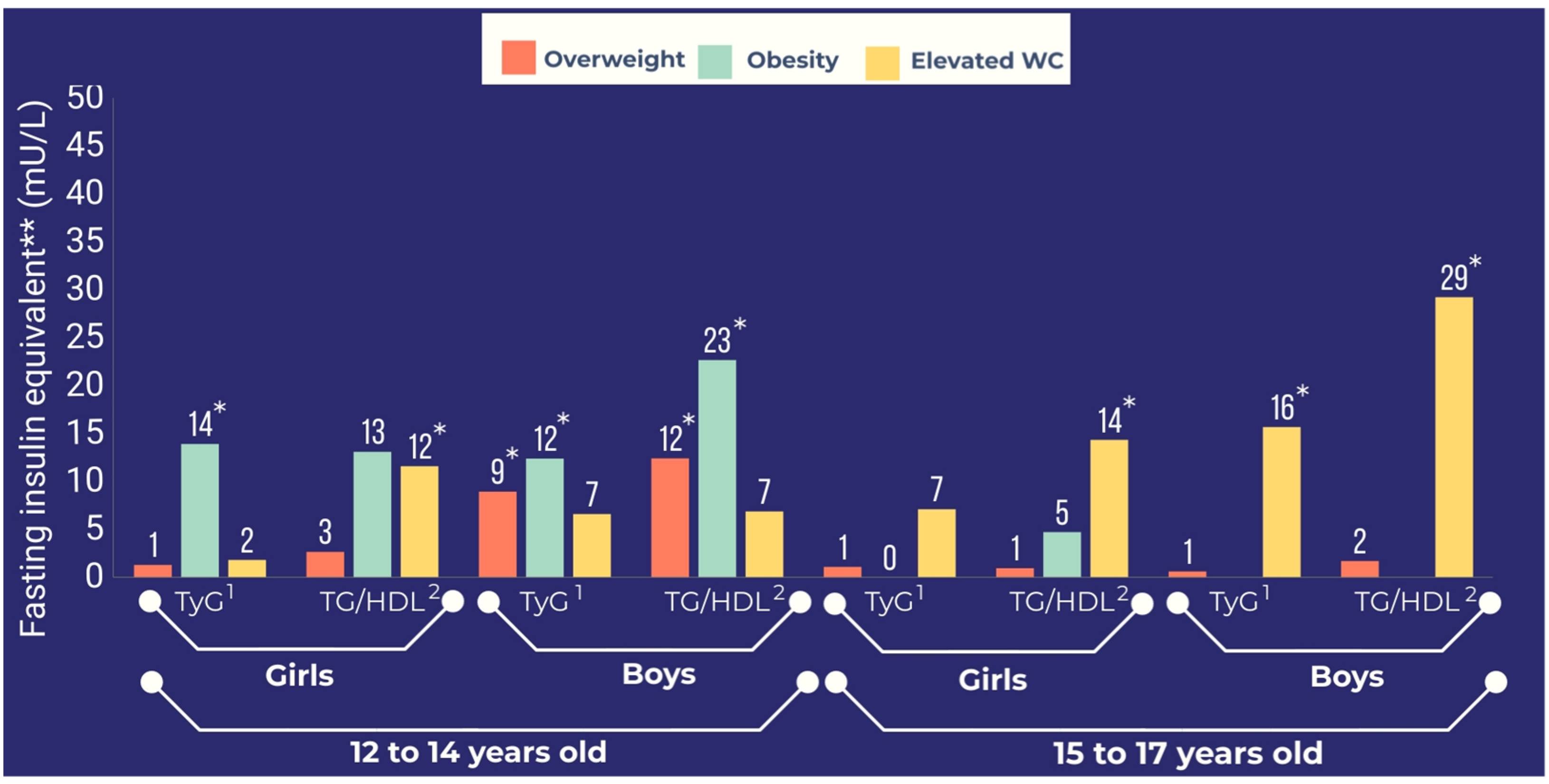
| FIeq for overweight | 0.72 (−3.49, 4.94) | 1.33 (−2.03, 4.70) | 2.35 (−3.14, 7.83) | 2.70 (−2.34, 7.74) | 1.49 (−4.34, 7.31) | 1.13 (−4.48, 6.74) | 1.38 (−3.99, 6.74) | 0.99 (−4.32, 6.31) |
| FIeq for obesity | 13.82 * (5.89, 21.76) | 13.94 * (5.70, 22.19) | 12.86 (−3.66, 29.39) | 13.12 (−4.35, 30.60) | −0.20 (−10.99, 10.59) | −0.41 (−11.18, 10.36) | 5.15 * (−7.38, 17.68) | 4.76 (−7.94, 17.46) |
| FIeq for elevated WC | 1.87 (−6.04, 9.78) | 1.84 (−5.73, 9.43) | 11.62 * (1.35, 21.90) | 11.62 * (1.35, 21.89) | 7.70 (−1.66, 17.06) | 7.15 (−1.68, 15.97) | 14.75 * (0.92, 28.58) | 14.37 * (0.64, 28.11) |
| Boys | ||||||||
| 12–14 years | 15–17 years | |||||||
| TyG Φ | TG/HDL δ | TyG Φ | TG/HDL δ | |||||
| Unadjusted | Adjusted | Unadjusted | Adjusted | Unadjusted | Adjusted | Unadjusted | Adjusted | |
| FIeq for overweight | 10.32 * (5.42, 15.21) | 8.98 * (4.89, 13.07) | 12.42 * (5.54, 19.31) | 12.44 * (6.22, 18.67) | 0.64 (−3.45, 4.73) | 0.66 (−3.68, 4.99) | 1.62 (−2.54, 5.79) | 1.74 (−2.71, 6.19) |
| FIeq for obesity | 14.05 * (6.09, 22.00) | 12.42 * (5.21, 19.63) | 22.29 * (8.94, 35.64) | 22.70 * (9.20, 36.19) | −1.32 (−11.38, 8.74) | −1.08 (−10.76, 8.60) | −0.99 (−18.60, 16.62) | −0.55 (−17.61, 16.51) |
| FIeq for elevated WC | 6.89 (−2.17, 15.95) | 6.33 (−2.58, 15.84) | 6.78 (−4.07, 17.62) | 6.91 (−4.35, 18.18) | 15.71 * (3.25, 28.17) | 15.70 * (3.70, 27.71) | 29.28 * (7.33, 51.23) | 29.26 * (7.87, 50.66) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deusdará, R.; de Moura Souza, A.; Szklo, M. Association between Obesity, Overweight, Elevated Waist Circumference, and Insulin Resistance Markers among Brazilian Adolescent Students. Nutrients 2022, 14, 3487. https://doi.org/10.3390/nu14173487
Deusdará R, de Moura Souza A, Szklo M. Association between Obesity, Overweight, Elevated Waist Circumference, and Insulin Resistance Markers among Brazilian Adolescent Students. Nutrients. 2022; 14(17):3487. https://doi.org/10.3390/nu14173487
Chicago/Turabian StyleDeusdará, Rodolfo, Amanda de Moura Souza, and Moyses Szklo. 2022. "Association between Obesity, Overweight, Elevated Waist Circumference, and Insulin Resistance Markers among Brazilian Adolescent Students" Nutrients 14, no. 17: 3487. https://doi.org/10.3390/nu14173487
APA StyleDeusdará, R., de Moura Souza, A., & Szklo, M. (2022). Association between Obesity, Overweight, Elevated Waist Circumference, and Insulin Resistance Markers among Brazilian Adolescent Students. Nutrients, 14(17), 3487. https://doi.org/10.3390/nu14173487






