Protective Effect of Probiotics Isolated from Traditional Fermented Tea Leaves (Miang) from Northern Thailand and Role of Synbiotics in Ameliorating Experimental Ulcerative Colitis in Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Preparation of Probiotics and Synbiotics
2.3. Animals and Ethics Statement
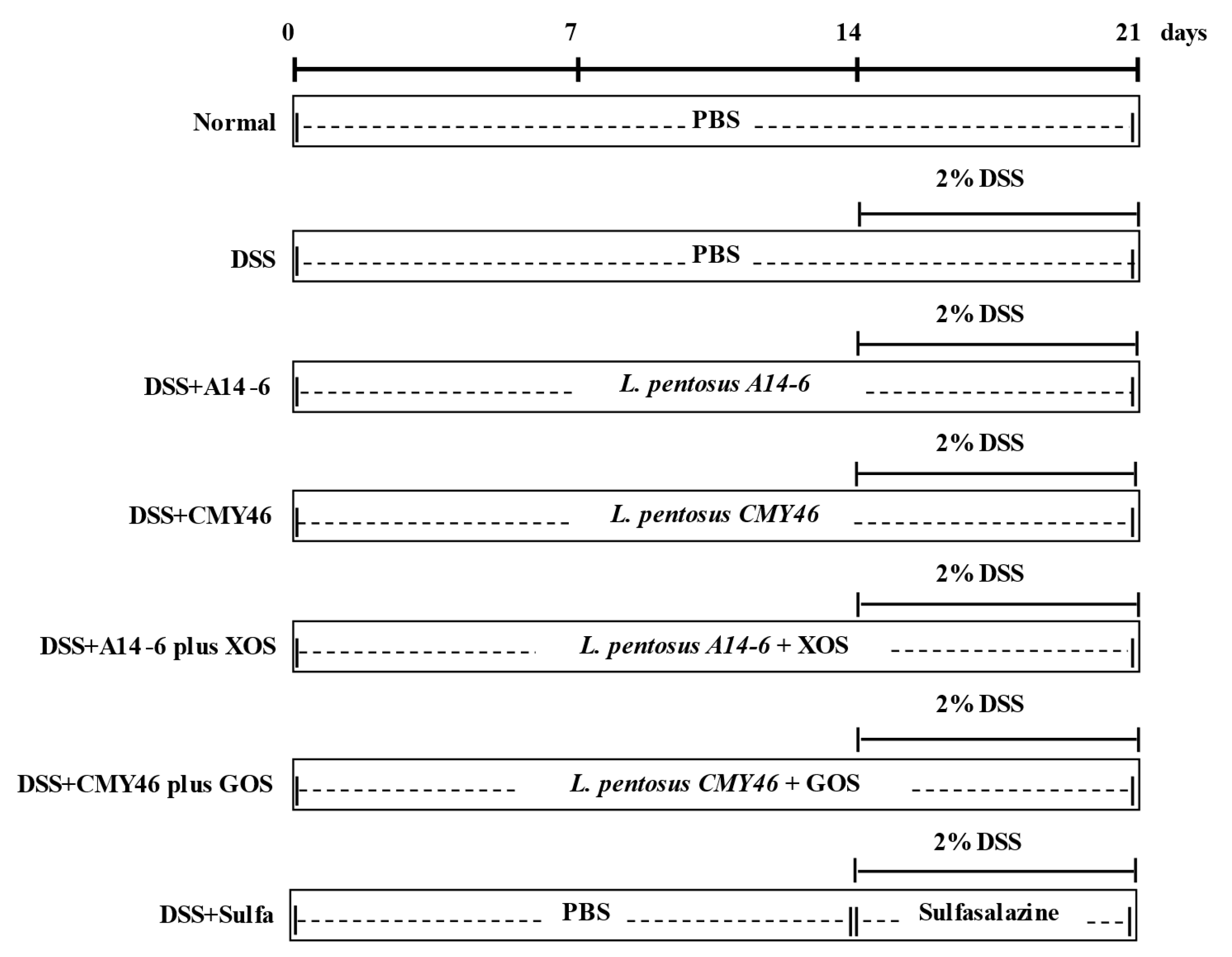
2.4. Experimental Design
2.5. Sample Collection
2.6. Assessment of Colitis Severity
2.7. Histopathologic Analysis
2.8. Determination of Goblet Cells
2.9. Reverse Transcription-Quantitative Polymerase Chain Reaction (qRT-PCR)
2.10. Measurement of Proinflammatory Cytokines in Plasma
2.11. Statistical Analysis
3. Results
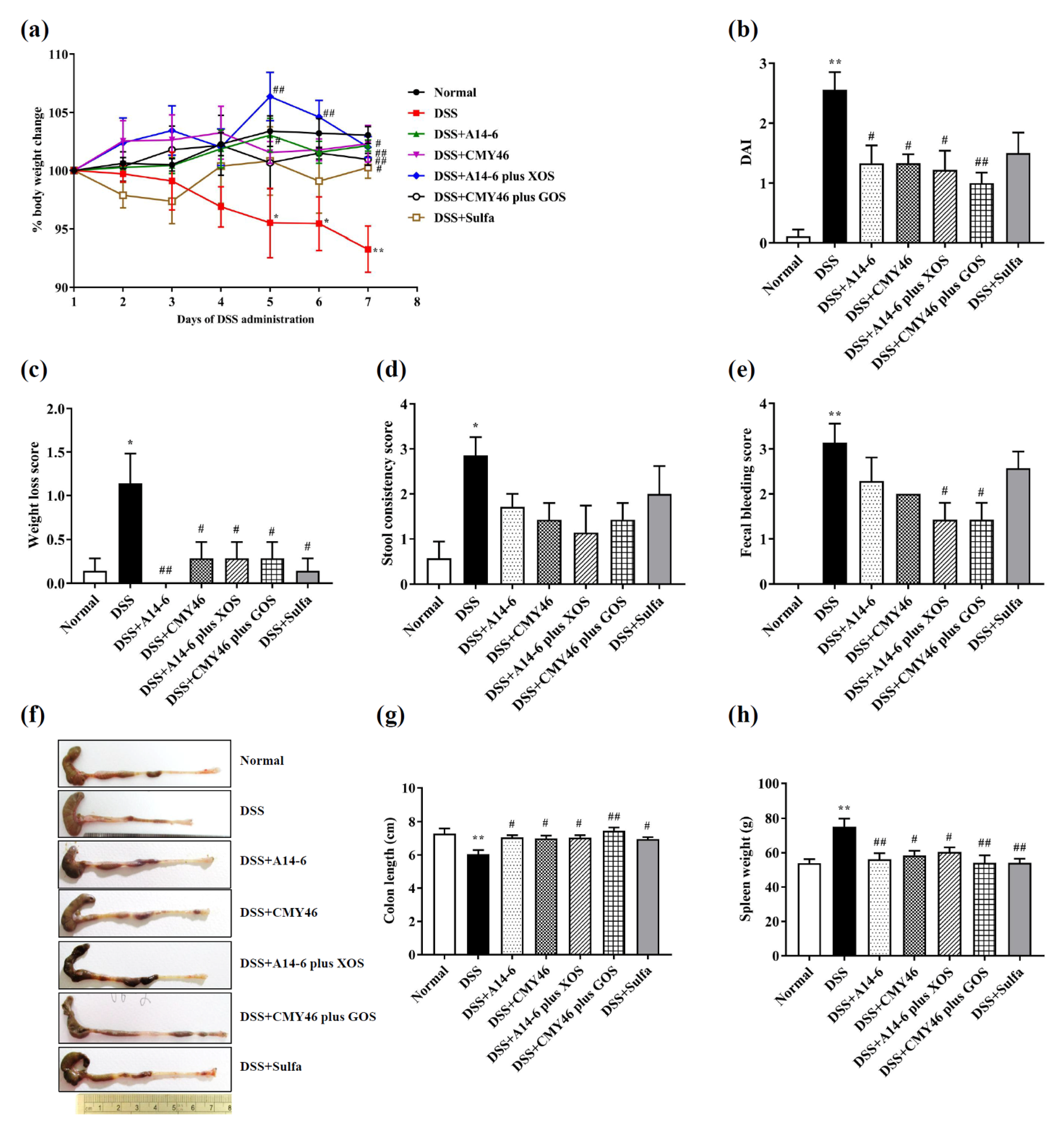
3.1. Probiotics and Synbiotics from Miang Attenuated Clinical Symptoms and Severity of Colitis Induced by DSS
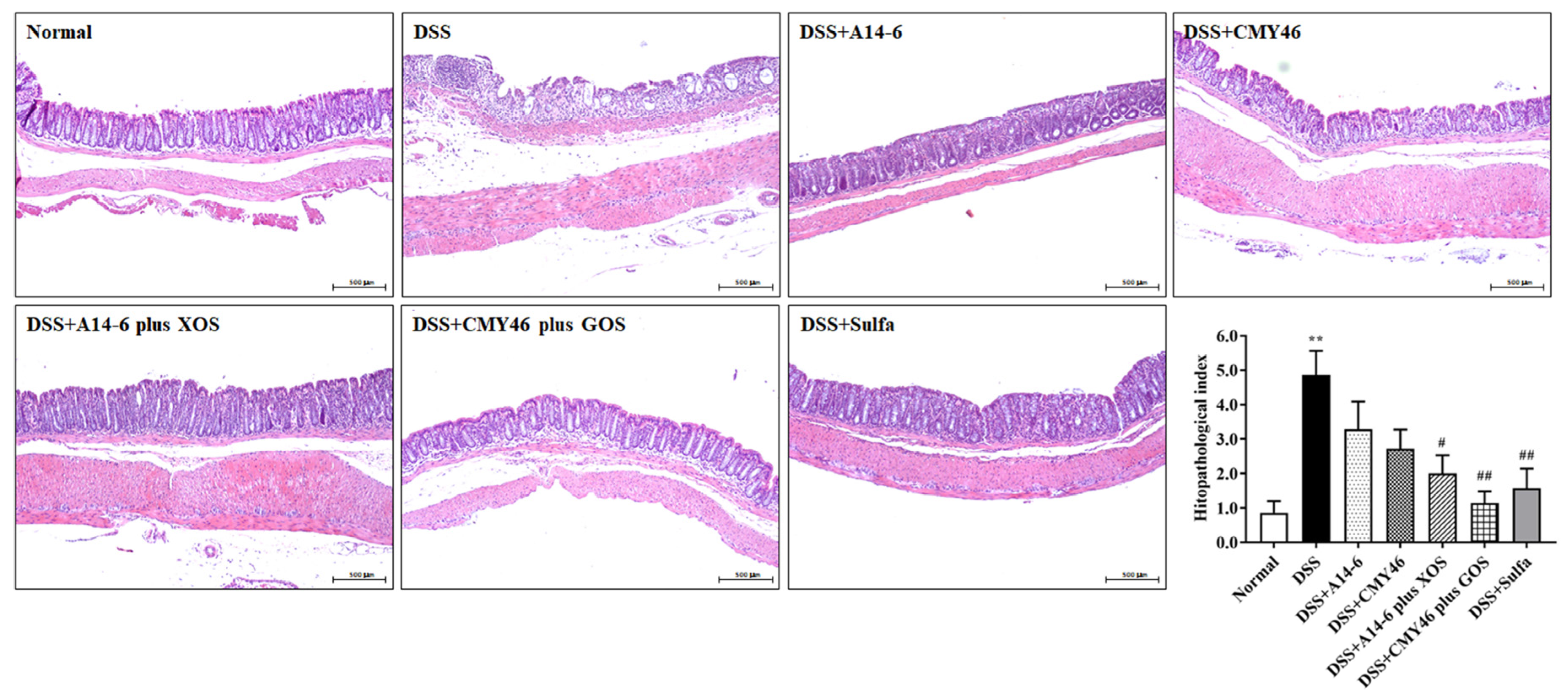
3.2. Effects of Probiotics and Synbiotics from Miang on the Histological Damages in DSS-Induced Colonic Inflammation
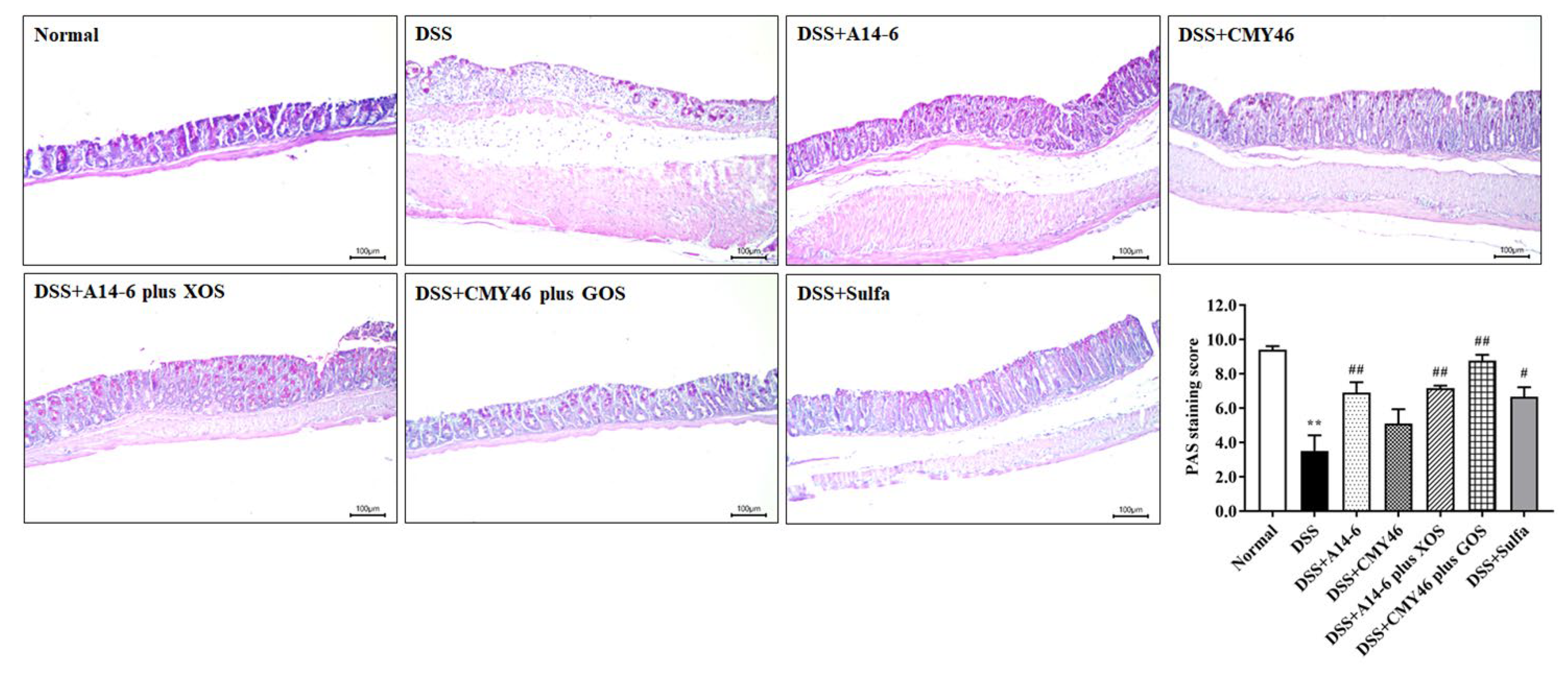
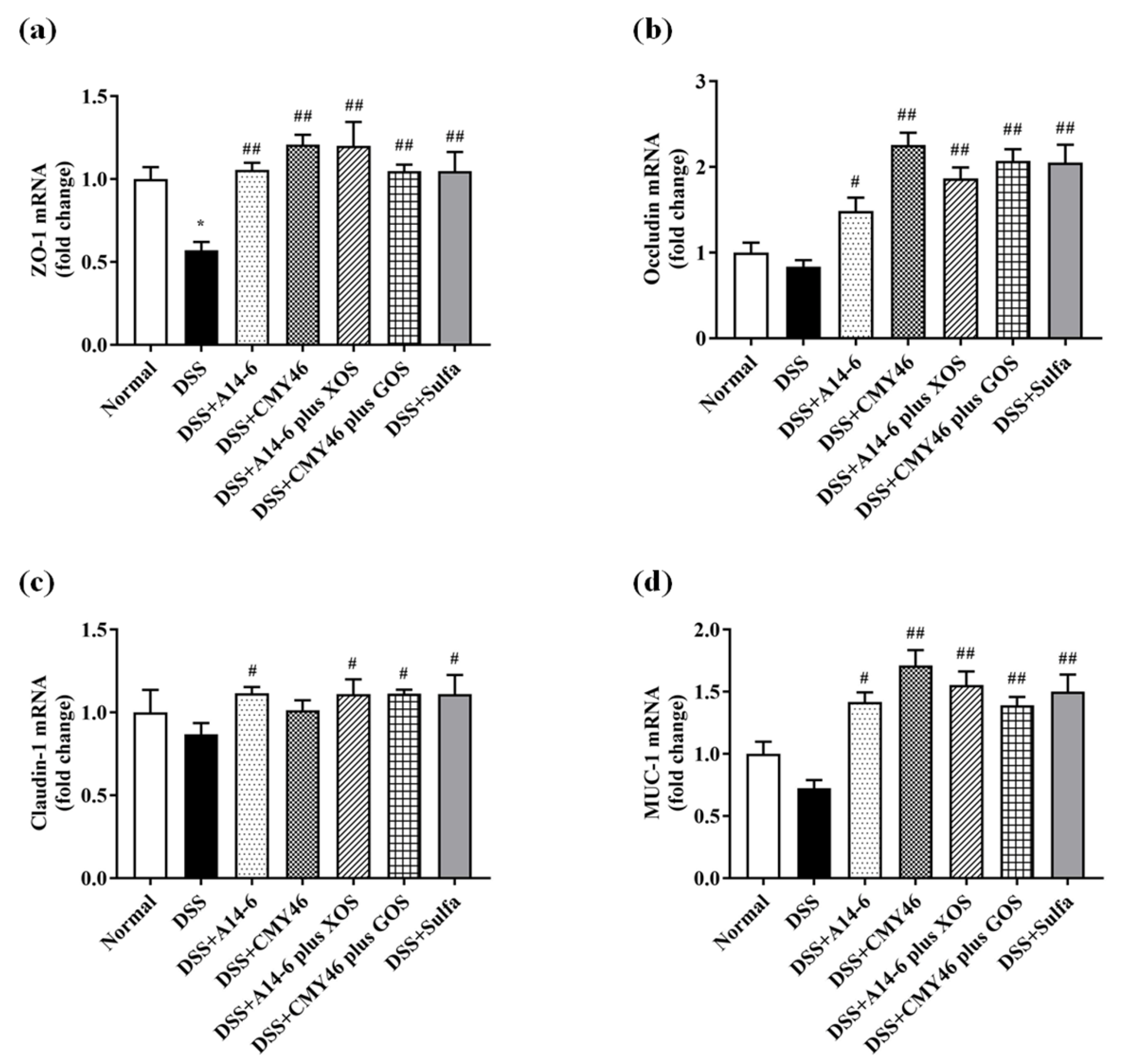
3.3. Probiotics and Synbiotics from Miang Improved the Colonic Barrier Integrity in DSS-Induced Colitis
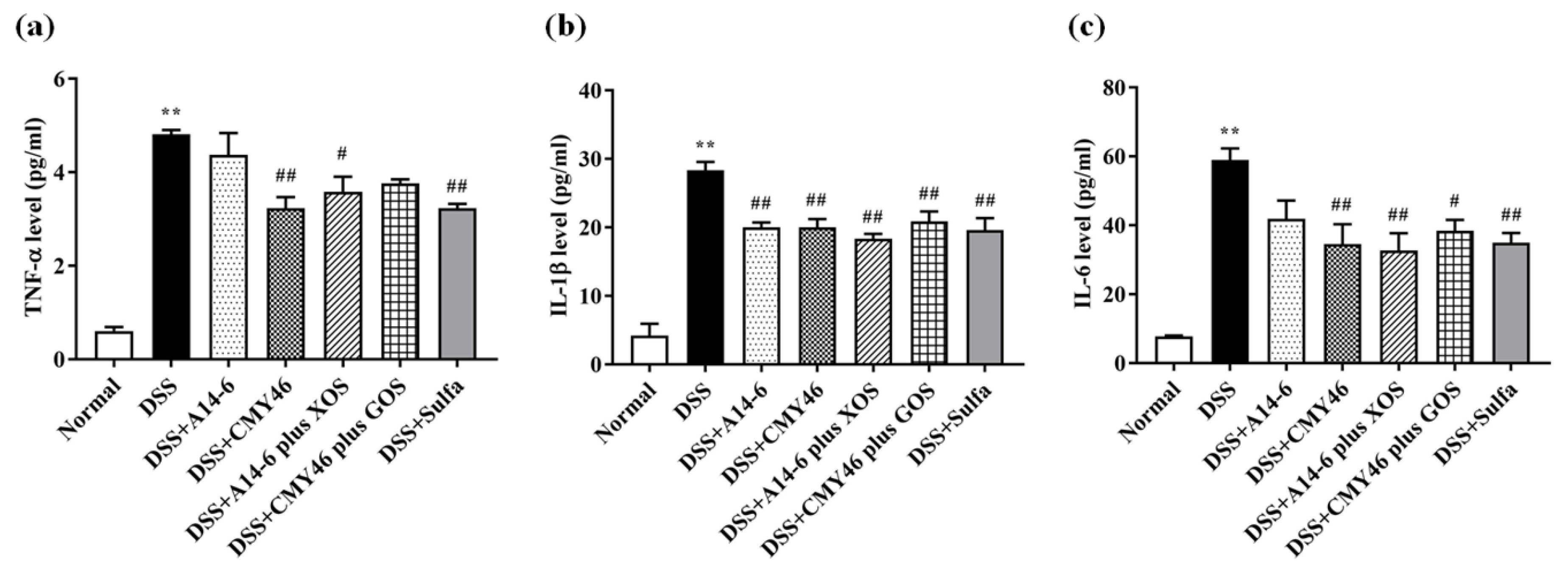
3.4. Probiotics and Synbiotics from Miang Alleviated Expression of Colonic Inflammatory Mediators in DSS-Induced Colitis
3.5. Probiotics and Synbiotics from Miang Attenuated Systemic Inflammation Induced by DSS
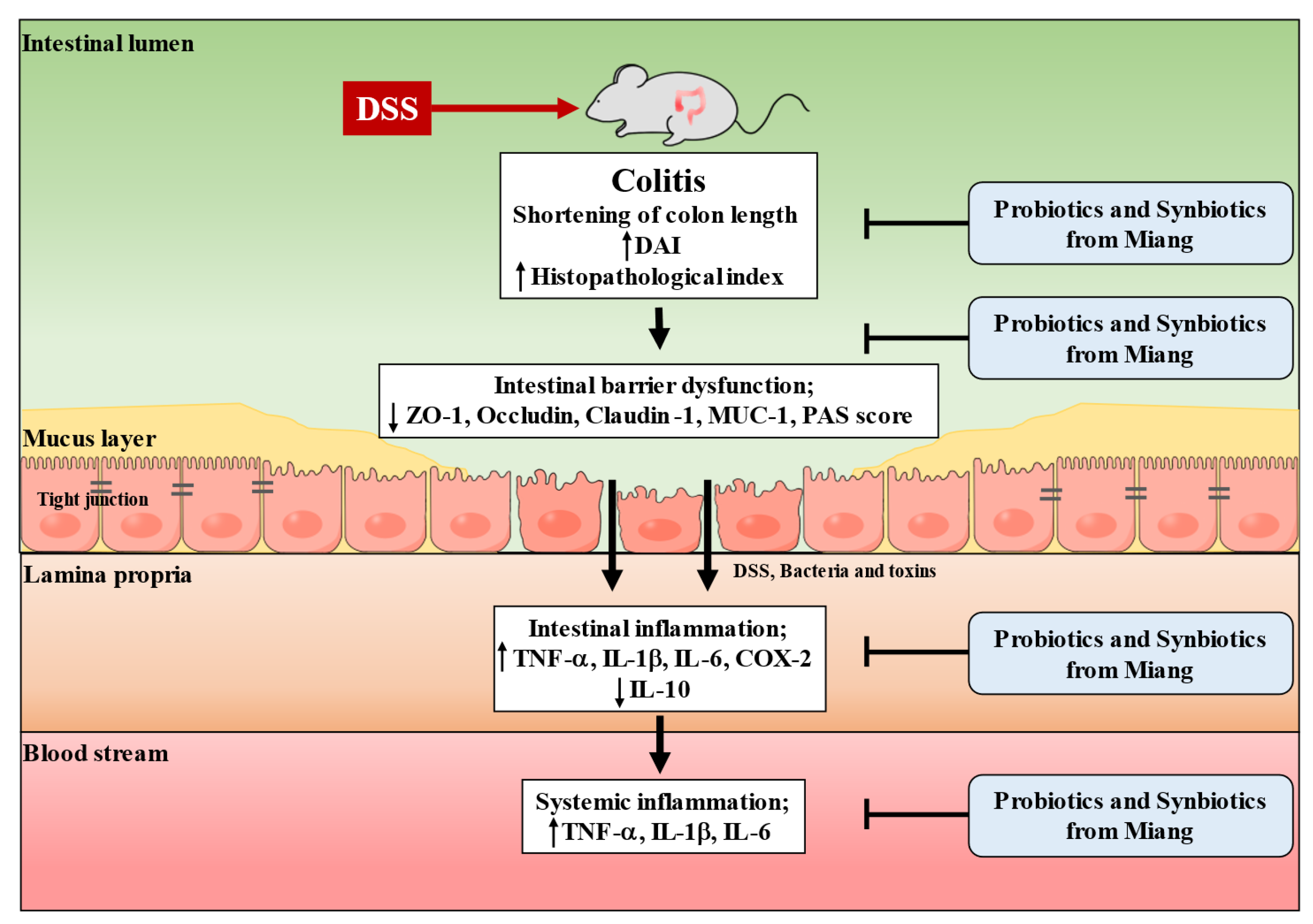
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Feuerstein, J.D.; Moss, A.C.; Farraye, F.A. Ulcerative Colitis. Mayo Clin. Proc. 2019, 94, 1357–1373. [Google Scholar] [CrossRef] [Green Version]
- Collaborators, G.B.D.I.B.D. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990-2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2020, 5, 17–30. [Google Scholar]
- Lechuga, S.; Ivanov, A.I. Disruption of the epithelial barrier during intestinal inflammation: Quest for new molecules and mechanisms. Biochim. Biophys. Acta 2017, 1864, 1183–1194. [Google Scholar] [CrossRef]
- Tatiya-Aphiradee, N.; Chatuphonprasert, W.; Jarukamjorn, K. Immune response and inflammatory pathway of ulcerative colitis. J. Basic Clin. Physiol. Pharmacol. 2018, 30, 1–10. [Google Scholar] [CrossRef]
- Ransford, R.A.; Langman, M.J. Sulphasalazine and mesalazine: Serious adverse reactions re-evaluated on the basis of suspected adverse reaction reports to the Committee on Safety of Medicines. Gut 2002, 51, 536–539. [Google Scholar] [CrossRef]
- Ganji-Arjenaki, M.; Rafieian-Kopaei, M. Probiotics are a good choice in remission of inflammatory bowel diseases: A meta analysis and systematic review. J. Cell. Physiol. 2018, 233, 2091–2103. [Google Scholar] [CrossRef]
- Ishikawa, H.; Matsumoto, S.; Ohashi, Y.; Imaoka, A.; Setoyama, H.; Umesaki, Y.; Tanaka, R.; Otani, T. Beneficial effects of probiotic bifidobacterium and galacto-oligosaccharide in patients with ulcerative colitis: A randomized controlled study. Digestion 2011, 84, 128–133. [Google Scholar] [CrossRef]
- Li, S.C.; Hsu, W.F.; Chang, J.S.; Shih, C.K. Combination of Lactobacillus acidophilus and Bifidobacterium animalis subsp. lactis shows a stronger anti-inflammatory effect than individual strains in HT-29 cells. Nutrients 2019, 11, 969. [Google Scholar]
- Sheng, K.; He, S.; Sun, M.; Zhang, G.; Kong, X.; Wang, J.; Wang, Y. Synbiotic supplementation containing Bifidobacterium infantis and xylooligosaccharides alleviates dextran sulfate sodium-induced ulcerative colitis. Food Funct. 2020, 11, 3964–3974. [Google Scholar] [CrossRef]
- Wang, G.; Huang, S.; Cai, S.; Yu, H.; Wang, Y.; Zeng, X.; Qiao, S. Lactobacillus reuteri ameliorates intestinal inflammation and modulates gut microbiota and metabolic disorders in dextran sulfate sodium-induced colitis in mice. Nutrients 2020, 12, 2298. [Google Scholar] [CrossRef]
- Wang, Y.; Xie, Q.; Zhang, Y.C.; Ma, W.; Ning, K.; Xiang, J.-Y.; Cui, J.; Xiang, H. Combination of probiotics with different functions alleviate DSS-induced colitis by regulating intestinal microbiota, IL-10, and barrier function. Appl. Microbiol. Biotechnol. 2020, 104, 335–349. [Google Scholar] [CrossRef]
- Kazemi, A.; Soltani, S.; Ghorabi, S.; Keshtkar, A.; Daneshzad, E.; Nasri, F.; Mazloomi, S.M. Effect of probiotic and synbiotic supplementation on inflammatory markers in health and disease status: A systematic review and meta-analysis of clinical trials. Clin. Nutr. 2020, 39, 789–819. [Google Scholar] [CrossRef]
- Jeong, J.-J.; Kim, K.-A.; Jang, S.-E.; Woo, J.-Y.; Han, M.J.; Kim, D.-H. Orally administrated Lactobacillus pentosus var. plantarum C29 ameliorates age-dependent colitis by inhibiting the nuclear factor-kappa B signaling pathway via the regulation of lipopolysaccharide production by gut microbiota. PLoS ONE 2015, 10, e0116533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, S.H.; Chou, L.M.; Chien, Y.W.; Chang, J.S.; Lin, C.I. Prebiotic effects of xylooligosaccharides on the improvement of microbiota balance in human subjects. Gastroenterol. Res. Pract. 2016, 2016, 5789232. [Google Scholar] [CrossRef] [Green Version]
- Chu, H.; Tao, X.; Sun, Z.; Hao, W.; Wei, X. Galactooligosaccharides protects against DSS-induced murine colitis through regulating intestinal flora and inhibiting NF-κB pathway. Life Sci. 2020, 242, 117220. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Wang, J.; Wang, Y.; Peng, B.; Liu, J.; Zhang, B.; Lv, H.; Wang, S. Protection of galacto-oligosaccharide against E. coli O157 colonization through enhancing gut barrier function and modulating gut microbiota. Foods 2020, 9, 1710. [Google Scholar] [CrossRef]
- Holma, R.; Juvonen, P.; Asmawi, M.Z.; Vapaatalo, H.; Korpela, R. Galacto-oligosaccharides stimulate the growth of bifidobacteria but fail to attenuate inflammation in experimental colitis in rats. Scand. J. Gastroenterol. 2002, 37, 1042–1047. [Google Scholar] [CrossRef]
- Christensen, E.G.; Licht, T.R.; Leser, T.D.; Bahl, M.I. Dietary xylo-oligosaccharide stimulates intestinal bifidobacteria and lactobacilli but has limited effect on intestinal integrity in rats. BMC Res. Notes 2014, 7, 660. [Google Scholar] [CrossRef] [Green Version]
- Haskey, N.; Dahl, W.J. Synbiotic therapy: A promising new adjunctive therapy for ulcerative colitis. Nutr. Rev. 2006, 64, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Shinde, T.; Perera, A.P.; Vemuri, R.; Gondalia, S.V.; Beale, D.J.; Karpe, A.V.; Shastri, S.; Basheer, W.; Southam, B.; Eri, R.; et al. Synbiotic supplementation with prebiotic green banana resistant starch and probiotic Bacillus coagulans spores ameliorates gut inflammation in mouse model of inflammatory bowel diseases. Eur. J. Nutr. 2020, 59, 3669–3689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khanongnuch, C.; Unban, K.; Kanpiengjai, A.; Saenjum, C. Recent research advances and ethno-botanical history of miang, a traditional fermented tea (Camellia sinensis var. assamica) of Northern Thailand. J. Ethn. Foods 2017, 4, 135–144. [Google Scholar] [CrossRef]
- Unban, K.; Khatthongngam, N.; Shetty, K.; Khanongnuch, C. Nutritional biotransformation in traditional fermented tea (Miang) from north Thailand and its impact on antioxidant and antimicrobial activities. J. Food Sci. Technol. 2019, 56, 2687–2699. [Google Scholar] [CrossRef] [PubMed]
- Chaikaew, S.; Baipong, S.; Sone, T.; Kanpiengjai, A.; Chui-chai, N.; Asano, K.; Khanongnuch, C. Diversity of lactic acid bacteria from Miang, a traditional fermented tea leaf in northern Thailand and their tannin-tolerant ability in tea extract. J. Microbiol. 2017, 55, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Unban, K.; Chaichana, W.; Baipong, S.; Abdullahi, A.D.; Kanpiengjai, A.; Shetty, K.; Khanongnuch, C. Probiotic and antioxidant properties of lactic acid bacteria isolated from indigenous fermented tea leaves (Miang) of north thailand and promising application in synbiotic formulation. Fermentation 2021, 7, 195. [Google Scholar] [CrossRef]
- Ma, Y.; Hu, C.; Yan, W.; Jiang, H.; Liu, G. Lactobacillus pentosus increases the abundance of Akkermansia and affects the serum metabolome to alleviate DSS-induced colitis in a murine model. Front. Cell. Dev. Biol. 2020, 8, 591408. [Google Scholar] [CrossRef]
- Kanmani, P.; Kim, H. Functional capabilities of probiotic strains on attenuation of intestinal epithelial cell inflammatory response induced by TLR4 stimuli. BioFactors 2019, 45, 223–235. [Google Scholar] [CrossRef]
- Unban, K.; Khatthongngam, N.; Pattananandecha, T.; Saenjum, C.; Shetty, K.; Khanongnuch, C. Microbial community dynamics during the non-filamentous fungi growth-based fermentation process of Miang, a traditional fermented tea of north Thailand and their product characterizations. Front. Microbiol. 2020, 11, 1515. [Google Scholar] [CrossRef]
- Shinde, T.; Perera, A.P.; Vemuri, R.; Gondalia, S.V.; Karpe, A.V.; Beale, D.J.; Shastri, S.; Southam, B.; Eri, R.; Stanley, R. Synbiotic supplementation containing whole plant sugar cane fibre and probiotic spores potentiates protective synergistic effects in mouse model of IBD. Nutrients 2019, 11, 818. [Google Scholar] [CrossRef] [Green Version]
- Oz, H.S.; Chen, T.; de Villiers, W.J.S. Green tea polyphenols and sulfasalazine have parallel anti-inflammatory properties in colitis models. Front. Immunol. 2013, 4, 132. [Google Scholar]
- Xia, Y.; Chen, Y.; Wang, G.; Yang, Y.; Song, X.; Xiong, Z.; Zhang, H.; Lai, P.; Wang, S.; Ai, L. Lactobacillus plantarum AR113 alleviates DSS-induced colitis by regulating the TLR4/MyD88/NF-κB pathway and gut microbiota composition. J. Funct. Foods 2020, 67, 103854. [Google Scholar] [CrossRef]
- Sasaki, S.; Hirata, I.; Maemura, K.; Hamamoto, N.; Murano, M.; Toshina, K.; Katsu, K. Prostaglandin E2 inhibits lesion formation in dextran sodium sulphate-induced colitis in rats and reduces the levels of mucosal inflammatory cytokines. Scand. J. Immunol. 2000, 51, 23–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, Z.; Li, J.; Dai, Y.; Wang, W.; Shi, R.; Wang, Z.; Ding, P.; Lu, Q.; Jiang, H.; Pei, W.; et al. Indigo naturalis alleviates dextran sulfate sodium-induced colitis in rats via altering gut microbiota. Front. Microbiol. 2020, 11, 731. [Google Scholar] [CrossRef] [PubMed]
- Kangwan, N.; Kim, Y.-J.; Han, Y.-M.; Jeong, M.; Park, J.-M.; Hahm, K.-B. Concerted actions of ameliorated colitis, aberrant crypt foci inhibition and 15-hydroxyprostaglandin dehydrogenase induction by sonic hedgehog inhibitor led to prevention of colitis-associated cancer. Int. J. Cancer 2016, 138, 1482–1493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kangwan, N.; Pintha, K.; Lekawanvijit, S.; Suttajit, M. Rosmarinic acid enriched fraction from Perilla frutescens leaves strongly protects indomethacin-induced gastric ulcer in rats. BioMed Res. Int. 2019, 2019, 9514703. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chassaing, B.; Aitken, J.D.; Malleshappa, M.; Vijay-Kumar, M. Dextran sulfate sodium (DSS)-induced colitis in mice. Curr. Protoc. Immunol. 2014, 104, 15.25.11–15.25.14. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Kwon, J.E.; Cho, M.-L. Immunological pathogenesis of inflammatory bowel disease. Intest. Res. 2018, 16, 26–42. [Google Scholar] [CrossRef] [Green Version]
- Eichele, D.D.; Kharbanda, K.K. Dextran sodium sulfate colitis murine model: An indispensable tool for advancing our understanding of inflammatory bowel diseases pathogenesis. World J. Gastroenterol. 2017, 23, 6016–6029. [Google Scholar] [CrossRef] [PubMed]
- Perše, M.; Cerar, A. Dextran sodium sulphate colitis mouse model: Traps and tricks. J. Biomed. Biotechnol. 2012, 2012, 718617. [Google Scholar] [CrossRef] [Green Version]
- Shin, M.-R.; Park, H.-J.; Seo, B.-I.; Roh, S.-S. New approach of medicinal herbs and sulfasalazine mixture on ulcerative colitis induced by dextran sodium sulfate. World J. Gastroenterol. 2020, 26, 5272–5286. [Google Scholar] [CrossRef]
- Tanaka, A.; Kanmura, S.; Morinaga, Y.; Kawabata, K.; Arima, S.; Sasaki, F.; Nasu, Y.; Tanoue, S.; Hashimoto, S.; Takeshita, M.; et al. Oral administration of Lactobacillus plantarum 06CC2 prevents experimental colitis in mice via an anti-inflammatory response. Mol. Med. Report. 2020, 21, 1181–1191. [Google Scholar] [CrossRef] [Green Version]
- Fujimori, S.; Gudis, K.; Mitsui, K.; Seo, T.; Yonezawa, M.; Tanaka, S.; Tatsuguchi, A.; Sakamoto, C. A randomized controlled trial on the efficacy of synbiotic versus probiotic or prebiotic treatment to improve the quality of life in patients with ulcerative colitis. Nutrition 2009, 25, 520–525. [Google Scholar] [CrossRef]
- Gersemann, M.; Becker, S.; Kübler, I.; Koslowski, M.; Wang, G.; Herrlinger, K.R.; Griger, J.; Fritz, P.; Fellermann, K.; Schwab, M.; et al. Differences in goblet cell differentiation between Crohn’s disease and ulcerative colitis. Differentiation 2009, 77, 84–94. [Google Scholar] [CrossRef] [PubMed]
- Dicksved, J.; Schreiber, O.; Willing, B.; Petersson, J.; Rang, S.; Phillipson, M.; Holm, L.; Roos, S. Lactobacillus reuteri maintains a functional mucosal barrier during DSS treatment despite mucus layer dysfunction. PLoS ONE 2012, 7, e46399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neurath, M.F. Cytokines in inflammatory bowel disease. Nat. Rev. Immunol. 2014, 14, 329–342. [Google Scholar] [CrossRef]
- Steed, H.; Macfarlane, G.T.; Macfarlane, S. Prebiotics, synbiotics and inflammatory bowel disease. Mol. Nutr. Food Res. 2008, 52, 898–905. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Zhao, T.; Cheng, X.; Zhao, M.; Gong, S.-H.; Zhao, Y.-Q.; Wu, H.-T.; Fan, M.; Zhu, L.-L. Cortical inflammation is increased in a DSS-induced colitis mouse model. Neurosci. Bull. 2018, 34, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Zhang, H.; Tsao, R.; Mine, Y. Lactobacillus pentosus S-PT84 prevents low-grade chronic inflammation-associated metabolic disorders in a lipopolysaccharide and high-fat diet C57/BL6J mouse model. J. Agric. Food Chem. 2020, 68, 4374–4386. [Google Scholar] [CrossRef]

| Gene | Primer Sequences (5′ to 3′) | |
|---|---|---|
| TNF-α | Forward | CGG GCA GGT CTA CTT TGG AG |
| Reverse | ACC CTG AGC CAT AAT CCC CT | |
| IL-1β | Forward | AAA AAA GCC TCG TGC TGT CG |
| Reverse | GTC GTT GCT TGG TTC TCC TTG | |
| IL-6 | Forward | ATC CAG TTG CCT TCT TGG GAC TGA |
| Reverse | TAA GCG TCC GAC TTG TGA AGT GGT | |
| IL-10 | Forward | TAC CTG GTA GAA GTG ATG CC |
| Reverse | CAT CAT GTA TGC TTC TAT GC | |
| COX-2 | Forward | TGA GCA CAG GAT TTG ACC AG |
| Reverse | CCT TGA AGT GGG TCA GGA TG | |
| ZO-1 | Forward | TGG AAT TGC AAT CTC TGG TG |
| Reverse | CTG GCC CTC CTT TTA ACA CA | |
| Occludin | Forward | GCT GTG ATG TGT GTG AGC TG |
| Reverse | GAC GGT CTA CCT GGA GGA AC | |
| Claudin-1 | Forward | TCT ACG AGG GAC TGT GGA TG |
| Reverse | TCA GAT TCA GCA AGG AGT CG | |
| MUC-1 | Forward | CTG TTC ACC ACC ACC ATG AC |
| Reverse | CTT GGA AGG GCA AGA AAA CC | |
| GAPDH | Forward | CAC TCA CGG CAA ATT CAA CGG CAC |
| Reverse | GAC TCC ACG ACA TAC TCA GCA C | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kangwan, N.; Kongkarnka, S.; Boonkerd, N.; Unban, K.; Shetty, K.; Khanongnuch, C. Protective Effect of Probiotics Isolated from Traditional Fermented Tea Leaves (Miang) from Northern Thailand and Role of Synbiotics in Ameliorating Experimental Ulcerative Colitis in Mice. Nutrients 2022, 14, 227. https://doi.org/10.3390/nu14010227
Kangwan N, Kongkarnka S, Boonkerd N, Unban K, Shetty K, Khanongnuch C. Protective Effect of Probiotics Isolated from Traditional Fermented Tea Leaves (Miang) from Northern Thailand and Role of Synbiotics in Ameliorating Experimental Ulcerative Colitis in Mice. Nutrients. 2022; 14(1):227. https://doi.org/10.3390/nu14010227
Chicago/Turabian StyleKangwan, Napapan, Sarawut Kongkarnka, Nitsara Boonkerd, Kridsada Unban, Kalidas Shetty, and Chartchai Khanongnuch. 2022. "Protective Effect of Probiotics Isolated from Traditional Fermented Tea Leaves (Miang) from Northern Thailand and Role of Synbiotics in Ameliorating Experimental Ulcerative Colitis in Mice" Nutrients 14, no. 1: 227. https://doi.org/10.3390/nu14010227
APA StyleKangwan, N., Kongkarnka, S., Boonkerd, N., Unban, K., Shetty, K., & Khanongnuch, C. (2022). Protective Effect of Probiotics Isolated from Traditional Fermented Tea Leaves (Miang) from Northern Thailand and Role of Synbiotics in Ameliorating Experimental Ulcerative Colitis in Mice. Nutrients, 14(1), 227. https://doi.org/10.3390/nu14010227







