Interactions between Food and Drugs, and Nutritional Status in Renal Patients: A Narrative Review
Abstract
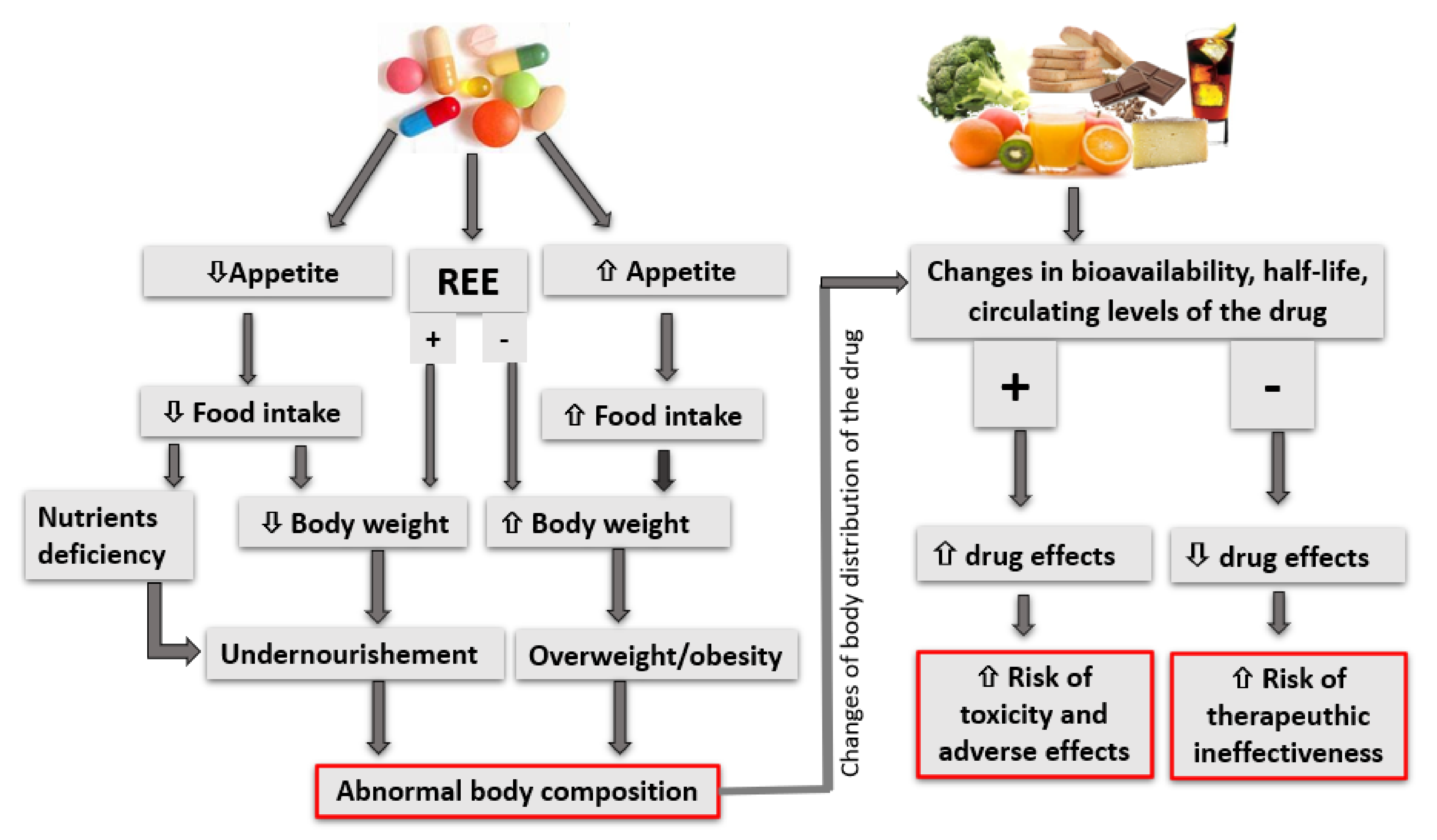
:1. Introduction
2. Interaction between Drugs and Nutritional Status
2.1. Taste and Smell Abnormalities
2.2. Food Intake Changes
2.3. Changes in Resting Energy Expenditure
2.4. Drug-Induced Nutrient Deficiency
3. Interaction between Food and Drugs
- (1)
- Increased concentrations in biological fluids that could enhance drug effect, up to the risk of side effects and toxicity;
- (2)
- Reduced concentrations in biological liquids, and thus reduced effect of the drug, with the risk of total or partial ineffectiveness.
4. Pharmacokinetics Basis of Food–Drug Interactions
5. Changes of Drug Bioavailability
6. Changes Due to Fluids, Protein, Lipid and Fibers Intake
7. Changes of Drug Distribution
8. Changes in Drug Metabolism
9. Changes in Drug Elimination
10. Pharmacodynamics and Pharmacokinetics of Food–Drug Interactions
11. Vegetables Rich in Vitamin K
12. Goitrogenic Foods
13. Fruit or Vegetable Juices
- -
- Statins (i.e., atorvastatin);
- -
- Antihypertensive drugs such as calcium-blocker agents (amlodipine, felodipine, manidipine, nicardipine, nifedipine, nimodipine, nisoldipine, nitrendipine, pranidipine, etc.), angiotensin II receptor antagonists (losartan), and β-blockers (thalnol and acebutolol);
- -
- Immunosuppressant agents (cyclosporin and tacrolimus);
- -
- Antiarrhythmic (amiodarone, quinidine, disopyramide and propafenone);
- -
- Antineoplastic (vinblastine);
- -
- Antibiotics (erythromycin).
14. Alcohol
15. Tea
16. Coffee
17. Milk
18. Macronutrients: Proteins and Lipids
19. Fibers
20. Food Supplements
21. Herbal Products
- -
- Immunosuppressants (cyclosporin, tracrolimus);
- -
- Anticancer drugs;
- -
- Oral contraceptives (ethinyl estradiol, norethindrone, ketodesogestrel);
- -
- Cardiovascular drugs (anticoagulants, statins, beta-blockers);
- -
- Antimicrobials (including anti-HIV, voriconazole);
- -
- Antidepressants and anxiolytics (benzodiazepines and buspirone);
- -
- Anticonvulsant agents;
- -
- Oral hypoglycaemic agents (tolbutamide, glycazide);
- -
- Anaesthetics;
- -
- Respiratory (fexofenadine) and gastrointestinal (omeprazole) agents;
- -
- Anti-migraine drugs (eletriptan);
- -
- Muscle relaxant medications;
- -
- Medications used in drug abusers (i.e., methadone).
22. Liquorice
23. Ginseng
24. Spices
25. Black Pepper
26. Turmeric
27. Ginger
28. Chili
29. Cinnamon
30. Conclusive Remarks
- -
- The existence of possible drug–food interactions, with some drugs influencing nutritional status;
- -
- The pharmacological therapy of the patient, comprehensive of supplements and herbal medications;
- -
- The need to educate the patient of a correct diet, in order to maximize the effectiveness and the safety of drug therapy while preserving/correcting nutritional status.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Hanna, R.M.; Ghobry, L.; Wassef, O.; Rhee, C.M.; Kalantar-Zadeh, K. A Practical Approach to Nutrition, Protein-Energy Wasting, Sarcopenia, and Cachexia in Patients with Chronic Kidney Disease. Blood Purif. 2020, 49, 202–211. [Google Scholar] [CrossRef]
- Ackerman, B.H.; Kasbekar, N. Disturbances of Taste and Smell Induced by Drugs. Pharmacotherapy 1997, 17, 482–496. [Google Scholar] [CrossRef]
- Risso, D.; Drayna, D.; Morini, G. Alteration, Reduction and Taste Loss: Main Causes and Potential Implications on Dietary Habits. Nutrients 2020, 12, 3284. [Google Scholar] [CrossRef] [PubMed]
- Ness-Abramof, R.; Apovian, C.M. Drug-Induced Weight Gain. Drugs Today 2005, 41, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Verhaegen, A.A.; Van Gaal, L.F. Drug-Induced Obesity and Its Metabolic Consequences: A Review with a Focus on Mechanisms and Possible Therapeutic Options. J. Endocrinol. Investig. 2017, 40, 1165–1174. [Google Scholar] [CrossRef]
- Zarezadeh, M.; Saedisomeolia, A.; Shekarabi, M.; Khorshidi, M.; Emami, M.R.; Müller, D.J. The Effect of Obesity, Macronutrients, Fasting and Nutritional Status on Drug-Metabolizing Cytochrome P450s: A Systematic Review of Current Evidence on Human Studies. Eur. J. Nutr. 2021, 60, 2905–2921. [Google Scholar] [CrossRef] [PubMed]
- Henkin, R.I. Drug-Induced Taste and Smell Disorders. Incidence, Mechanisms and Management Related Primarily to Treatment of Sensory Receptor Dysfunction. Drug Saf. 1994, 11, 318–377. [Google Scholar] [CrossRef] [PubMed]
- Mayet, A.Y. Loss of Smell (Anosmia) and Taste (Ageusia) in a Patient Treated with Pegylated Interferon Alfa and Ribavirin. Curr. Ther. Res. Clin. Exp. 2007, 68, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Naik, B.S.; Shetty, N.; Maben, E.V.S. Drug-Induced Taste Disorders. Eur. J. Intern. Med. 2010, 21, 240–243. [Google Scholar] [CrossRef] [PubMed]
- Rademacher, W.M.H.; Aziz, Y.; Hielema, A.; Cheung, K.-C.; de Lange, J.; Vissink, A.; Rozema, F.R. Oral Adverse Effects of Drugs: Taste Disorders. Oral Dis. 2020, 26, 213–223. [Google Scholar] [CrossRef]
- Toffanello, E.D.; Inelmen, E.M.; Imoscopi, A.; Perissinotto, E.; Coin, A.; Miotto, F.; Donini, L.M.; Cucinotta, D.; Barbagallo, M.; Manzato, E.; et al. Taste Loss in Hospitalized Multimorbid Elderly Subjects. Clin. Interv. Aging 2013, 8, 167–174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoebel, B.G. Brain Neurotransmitters in Food and Drug Reward. Am. J. Clin. Nutr. 1985, 42, 1133–1150. [Google Scholar] [CrossRef] [PubMed]
- Leibowitz, S.F. Brain Monoamines and Peptides: Role in the Control of Eating Behavior. Fed. Proc. 1986, 45, 1396–1403. [Google Scholar]
- Basdevant, A. Neurotransmitters, Neuropeptides and Eating Behavior. Diabete Metab. 1992, 18, 431–437. [Google Scholar]
- Aaseth, J.; Ellefsen, S.; Alehagen, U.; Sundfør, T.M.; Alexander, J. Diets and Drugs for Weight Loss and Health in Obesity—An Update. Biomed. Pharmacother. 2021, 140, 111789. [Google Scholar] [CrossRef]
- Gadde, K.M.; Xiong, G.L. Bupropion for Weight Reduction. Expert Rev. Neurother. 2007, 7, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Tak, Y.J.; Lee, S.Y. Long-Term Efficacy and Safety of Anti-Obesity Treatment: Where Do We Stand? Curr. Obes. Rep. 2021, 10, 14–30. [Google Scholar] [CrossRef]
- Verhaegen, A.A.; Van Gaal, L.F. Drugs Affecting Body Weight, Body Fat Distribution, and Metabolic Function-Mechanisms and Possible Therapeutic or Preventive Measures: An Update. Curr. Obes. Rep. 2021, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Moreira-Pais, A.; Ferreira, R.; Gil da Costa, R. Platinum-Induced Muscle Wasting in Cancer Chemotherapy: Mechanisms and Potential Targets for Therapeutic Intervention. Life Sci. 2018, 208, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ni, J.; Zhang, L. Cancer Cachexia: Definition, Staging, and Emerging Treatments. Cancer Manag. Res. 2020, 12, 5597–5605. [Google Scholar] [CrossRef]
- Stohs, S.J.; Badmaev, V. A Review of Natural Stimulant and Non-Stimulant Thermogenic Agents. Phytother. Res. 2016, 30, 732–740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marteene, W.; Winckel, K.; Hollingworth, S.; Kisely, S.; Gallagher, E.; Hahn, M.; Ebdrup, B.H.; Firth, J.; Siskind, D. Strategies to Counter Antipsychotic-Associated Weight Gain in Patients with Schizophrenia. Expert Opin. Drug Saf. 2019, 18, 1149–1160. [Google Scholar] [CrossRef]
- Furlong, M.; Deming-Halverson, S.; Sandler, D.P. Chronic Antibiotic Use during Adulthood and Weight Change in the Sister Study. PLoS ONE 2019, 14, e0216959. [Google Scholar] [CrossRef]
- Domecq, J.P.; Prutsky, G.; Leppin, A.; Sonbol, M.B.; Altayar, O.; Undavalli, C.; Wang, Z.; Elraiyah, T.; Brito, J.P.; Mauck, K.F.; et al. Clinical Review: Drugs Commonly Associated with Weight Change: A Systematic Review and Meta-Analysis. J. Clin. Endocrinol. Metab. 2015, 100, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Fernstrom, M.H. Drugs That Cause Weight Gain. Obes. Res. 1995, 3 (Suppl. S4), 435S–439S. [Google Scholar] [CrossRef]
- Kalucy, R.S. Drug-Induced Weight Gain. Drugs 1980, 19, 268–278. [Google Scholar] [CrossRef]
- Teff, K.L.; Rickels, M.R.; Grudziak, J.; Fuller, C.; Nguyen, H.-L.; Rickels, K. Antipsychotic-Induced Insulin Resistance and Postprandial Hormonal Dysregulation Independent of Weight Gain or Psychiatric Disease. Diabetes 2013, 62, 3232–3240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sitruk-Ware, R.; Nath, A. Characteristics and Metabolic Effects of Estrogen and Progestins Contained in Oral Contraceptive Pills. Best Pract. Res. Clin. Endocrinol. Metab. 2013, 27, 13–24. [Google Scholar] [CrossRef]
- Smith, S.M.; Meyer, M.; Trinkley, K.E. Phentermine/Topiramate for the Treatment of Obesity. Ann. Pharmacother. 2013, 47, 340–349. [Google Scholar] [CrossRef]
- Taramasso, L.; Bonfanti, P.; Ricci, E.; Orofino, G.; Squillace, N.; Menzaghi, B.; De Socio, G.V.; Madeddu, G.; Pellicanò, G.F.; Pagnucco, L.; et al. Factors Associated with Weight Gain in People Treated with Dolutegravir. Open Forum Infect. Dis. 2020, 7, ofaa195. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, S.B.; Fenton, R.A. K+ and the Renin-Angiotensin-Aldosterone System: New Insights into Their Role in Blood Pressure Control and Hypertension Treatment. J. Physiol. 2019, 597, 4451–4464. [Google Scholar] [CrossRef] [PubMed]
- Abbas, A.; Mirza, M.M.; Ganti, A.K.; Tendulkar, K. Renal Toxicities of Targeted Therapies. Target. Oncol. 2015, 10, 487–499. [Google Scholar] [CrossRef] [PubMed]
- Cannon-Babb, M.L.; Schwartz, A.B. Drug-Induced Hyperkalemia. Hosp. Pract. (Off. Ed.) 1986, 21, 99–107, 111, 114–127. [Google Scholar] [PubMed]
- Kovesdy, C.P. Management of Hyperkalaemia in Chronic Kidney Disease. Nat. Rev. Nephrol. 2014, 10, 653–662. [Google Scholar] [CrossRef] [PubMed]
- Palmer, B.F.; Clegg, D.J. Diagnosis and Treatment of Hyperkalemia. Cleve Clin. J. Med. 2017, 84, 934–942. [Google Scholar] [CrossRef]
- Baker, M.; Perazella, M.A. NSAIDs in CKD: Are They Safe? Am. J. Kidney Dis. 2020, 76, 546–557. [Google Scholar] [CrossRef]
- Liamis, G.; Megapanou, E.; Elisaf, M.; Milionis, H. Hyponatremia-Inducing Drugs. Front. Horm. Res. 2019, 52, 167–177. [Google Scholar] [CrossRef]
- Gröber, U. Magnesium and Drugs. Int. J. Mol. Sci. 2019, 20, 2094. [Google Scholar] [CrossRef] [Green Version]
- Liamis, G.; Hoorn, E.J.; Florentin, M.; Milionis, H. An Overview of Diagnosis and Management of Drug-Induced Hypomagnesemia. Pharmacol. Res. Perspect. 2021, 9, e00829. [Google Scholar] [CrossRef]
- Koyyada, A. Long-Term Use of Proton Pump Inhibitors as a Risk Factor for Various Adverse Manifestations. Therapie 2021, 76, 13–21. [Google Scholar] [CrossRef]
- Liamis, G.; Milionis, H.J.; Elisaf, M. A Review of Drug-Induced Hypocalcemia. J. Bone Miner. Metab. 2009, 27, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Reilly, W.; Ilich, J. Prescription Drugs and Nutrient Depletion: How Much Is Known? Adv. Nutr. 2017, 8, 23. [Google Scholar] [CrossRef]
- Linder, L.; Tamboue, C.; Clements, J.N. Drug-Induced Vitamin B12 Deficiency: A Focus on Proton Pump Inhibitors and Histamine-2 Antagonists. J. Pharm. Pract. 2017, 30, 639–642. [Google Scholar] [CrossRef]
- Aroda, V.R.; Edelstein, S.L.; Goldberg, R.B.; Knowler, W.C.; Marcovina, S.M.; Orchard, T.J.; Bray, G.A.; Schade, D.S.; Temprosa, M.G.; White, N.H.; et al. Long-term Metformin Use and Vitamin B12 Deficiency in the Diabetes Prevention Program Outcomes Study. J. Clin. Endocrinol. Metab. 2016, 101, 1754–1761. [Google Scholar] [CrossRef]
- Holick, M.F. The Vitamin D Deficiency Pandemic: Approaches for Diagnosis, Treatment and Prevention. Rev. Endocr. Metab. Disord. 2017, 18, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Ased, S.; Wells, J.; Morrow, L.E.; Malesker, M.A. Clinically Significant Food-Drug Interactions. Consult. Pharm. 2018, 33, 649–657. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Zhu, X.; Chen, Z.; Fan, C.H.; Kwan, H.S.; Wong, C.H.; Shek, K.Y.; Zuo, Z.; Lam, T.N. A Review of Food-Drug Interactions on Oral Drug Absorption. Drugs 2017, 77, 1833–1855. [Google Scholar] [CrossRef] [PubMed]
- Genser, D. Food and Drug Interaction: Consequences for the Nutrition/Health Status. Ann. Nutr. Metab. 2008, 52 (Suppl. S1), 29–32. [Google Scholar] [CrossRef] [PubMed]
- de Lima Toccafondo Vieira, M.; Huang, S.-M. Botanical-Drug Interactions: A Scientific Perspective. Planta Med. 2012, 78, 1400–1415. [Google Scholar] [CrossRef] [Green Version]
- Leite, P.M.; Martins, M.A.P.; Castilho, R.O. Review on Mechanisms and Interactions in Concomitant Use of Herbs and Warfarin Therapy. Biomed. Pharmacother. 2016, 83, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Werba, J.P.; Misaka, S.; Giroli, M.G.; Shimomura, K.; Amato, M.; Simonelli, N.; Vigo, L.; Tremoli, E. Update of Green Tea Interactions with Cardiovascular Drugs and Putative Mechanisms. J. Food Drug Anal. 2018, 26, S72–S77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Custodio, J.M.; Wu, C.-Y.; Benet, L.Z. Predicting Drug Disposition, Absorption/Elimination/Transporter Interplay and the Role of Food on Drug Absorption. Adv. Drug Deliv. Rev. 2008, 60, 717–733. [Google Scholar] [CrossRef] [Green Version]
- Koziolek, M.; Alcaro, S.; Augustijns, P.; Basit, A.W.; Grimm, M.; Hens, B.; Hoad, C.L.; Jedamzik, P.; Madla, C.M.; Maliepaard, M.; et al. The Mechanisms of Pharmacokinetic Food-Drug Interactions—A Perspective from the UNGAP Group. Eur. J. Pharm. Sci. 2019, 134, 31–59. [Google Scholar] [CrossRef]
- Palleria, C.; Di Paolo, A.; Giofrè, C.; Caglioti, C.; Leuzzi, G.; Siniscalchi, A.; De Sarro, G.; Gallelli, L. Pharmacokinetic Drug-Drug Interaction and Their Implication in Clinical Management. J. Res. Med. Sci. 2013, 18, 601–610. [Google Scholar]
- Gu, C.-H.; Li, H.; Levons, J.; Lentz, K.; Gandhi, R.B.; Raghavan, K.; Smith, R.L. Predicting Effect of Food on Extent of Drug Absorption Based on Physicochemical Properties. Pharm. Res. 2007, 24, 1118–1130. [Google Scholar] [CrossRef]
- Golub, A.L.; Frost, R.W.; Betlach, C.J.; Gonzalez, M.A. Physiologic Considerations in Drug Absorption from the Gastrointestinal Tract. J. Allergy Clin. Immunol. 1986, 78, 689–694. [Google Scholar] [CrossRef]
- Cheng, L.; Wong, H. Food Effects on Oral Drug Absorption: Application of Physiologically-Based Pharmacokinetic Modeling as a Predictive Tool. Pharmaceutics 2020, 12, 672. [Google Scholar] [CrossRef] [PubMed]
- Malagelada, J.R.; Go, V.L.; Summerskill, W.H. Different Gastric, Pancreatic, and Biliary Responses to Solid-Liquid or Homogenized Meals. Dig. Dis. Sci. 1979, 24, 101–110. [Google Scholar] [CrossRef]
- Roberts, M.S.; Magnusson, B.M.; Burczynski, F.J.; Weiss, M. Enterohepatic Circulation: Physiological, Pharmacokinetic and Clinical Implications. Clin. Pharmacokinet. 2002, 41, 751–790. [Google Scholar] [CrossRef]
- Honigford, C.R.; Aburub, A.; Fadda, H.M. A Simulated Stomach Duodenum Model Predicting the Effect of Fluid Volume and Prandial Gastric Flow Patterns on Nifedipine Pharmacokinetics From Cosolvent-Based Capsules. J. Pharm. Sci. 2019, 108, 288–294. [Google Scholar] [CrossRef] [Green Version]
- Zeller, F.P.; Ueda, C.T.; Wulf, B.G.; Meyers, D.G. Effect of Caffeine on the Oral Absorption and Disposition of Quinidine. Clin. Pharm. 1984, 3, 72–75. [Google Scholar] [PubMed]
- Yamreudeewong, W.; Henann, N.E.; Fazio, A.; Lower, D.L.; Cassidy, T.G. Drug-Food Interactions in Clinical Practice. J. Fam. Pract. 1995, 40, 376–384. [Google Scholar] [PubMed]
- Contin, M.; Martinelli, P. Pharmacokinetics of Levodopa. J. Neurol. 2010, 257, S253–S261. [Google Scholar] [CrossRef] [PubMed]
- Semb, B.K. Regional Gastric Flow Changes after Meal Stimulation Measured by the Hydrogen Clearance Technique in Conscious Cats. Scand. J. Gastroenterol. 1982, 17, 839–842. [Google Scholar] [CrossRef]
- Fleisher, D.; Li, C.; Zhou, Y.; Pao, L.H.; Karim, A. Drug, Meal and Formulation Interactions Influencing Drug Absorption after Oral Administration. Clinical Implications. Clin. Pharmacokinet. 1999, 36, 233–254. [Google Scholar] [CrossRef]
- Harskamp-van Ginkel, M.W.; Hill, K.D.; Becker, K.C.; Becker, K.; Testoni, D.; Cohen-Wolkowiez, M.; Gonzalez, D.; Barrett, J.S.; Benjamin, D.K.; Siegel, D.A.; et al. Drug Dosing and Pharmacokinetics in Children with Obesity: A Systematic Review. JAMA Pediatr. 2015, 169, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Abernethy, D.R.; Greenblatt, D.J. Drug Disposition in Obese Humans. An Update. Clin. Pharmacokinet. 1986, 11, 199–213. [Google Scholar] [CrossRef]
- Sankaralingam, S.; Kim, R.B.; Padwal, R.S. The Impact of Obesity on the Pharmacology of Medications Used for Cardiovascular Risk Factor Control. Can. J. Cardiol. 2015, 31, 167–176. [Google Scholar] [CrossRef]
- Anderson, B.J.; Holford, N.H. What Is the Best Size Predictor for Dose in the Obese Child? Paediatr. Anaesth. 2017, 27, 1176–1184. [Google Scholar] [CrossRef]
- Osserman, E.F.; Takatsuki, K. The Plasma Proteins in Liver Disease. Med. Clin. N. Am. 1963, 47, 679–710. [Google Scholar] [CrossRef]
- Bienenstock, J.; Poortmans, J. Renal Clearance of 15 Plasma Proteins in Renal Disease. J. Lab. Clin. Med. 1970, 75, 297–306. [Google Scholar] [PubMed]
- Toro-Ramos, T.; Paley, C.; Pi-Sunyer, F.X.; Gallagher, D. Body Composition during Fetal Development and Infancy through the Age of 5 Years. Eur. J. Clin. Nutr. 2015, 69, 1279–1289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reinders, I.; Visser, M.; Schaap, L. Body Weight and Body Composition in Old Age and Their Relationship with Frailty. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 11–15. [Google Scholar] [CrossRef] [Green Version]
- Ronis, M.J.J. Effects of Soy Containing Diet and Isoflavones on Cytochrome P450 Enzyme Expression and Activity. Drug Metab. Rev. 2016, 48, 331–341. [Google Scholar] [CrossRef]
- Mouly, S.; Lloret-Linares, C.; Sellier, P.-O.; Sene, D.; Bergmann, J.-F. Is the Clinical Relevance of Drug-Food and Drug-Herb Interactions Limited to Grapefruit Juice and Saint-John’s Wort? Pharmacol. Res. 2017, 118, 82–92. [Google Scholar] [CrossRef]
- Bushra, R.; Aslam, N.; Khan, A.Y. Food-Drug Interactions. Oman Med. J. 2011, 26, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Remer, T.; Manz, F. Potential Renal Acid Load of Foods and Its Influence on Urine PH. J. Am. Diet. Assoc. 1995, 95, 791–797. [Google Scholar] [CrossRef]
- Choi, J.H.; Ko, C.M. Food and Drug Interactions. J. Lifestyle Med. 2017, 7, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Andrade, C. Fruit Juice, Organic Anion Transporting Polypeptides, and Drug Interactions in Psychiatry. J. Clin. Psychiatry 2014, 75, e1323–e1325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Violi, F.; Lip, G.Y.; Pignatelli, P.; Pastori, D. Interaction between Dietary Vitamin K Intake and Anticoagulation by Vitamin K Antagonists: Is It Really True? A Systematic Review. Medicine 2016, 95, e2895. [Google Scholar] [CrossRef] [PubMed]
- Di Minno, A.; Frigerio, B.; Spadarella, G.; Ravani, A.; Sansaro, D.; Amato, M.; Kitzmiller, J.P.; Pepi, M.; Tremoli, E.; Baldassarre, D. Old and New Oral Anticoagulants: Food, Herbal Medicines and Drug Interactions. Blood Rev. 2017, 31, 193–203. [Google Scholar] [CrossRef]
- Choi, E.J.; Zhang, P.; Kwon, H. Determination of goitrogenic metabolites in the serum of male wistar rat fed structurally different glucosinolates. Toxicol. Res. 2014, 30, 109–116. [Google Scholar] [CrossRef] [Green Version]
- Felker, P.; Bunch, R.; Leung, A.M. Concentrations of thiocyanate and goitrin in human plasma, their precursor concentrations in brassica vegetables, andassociated potential risk for hypothyroidism. Nutr Rev. 2016, 74, 248–258. [Google Scholar] [CrossRef] [Green Version]
- Otun, J.; Sahebkar, A.; Östlundh, L.; Atkin, S.L.; Sathyapalan, T. Systematic Review and Meta-Analysis on the Effect of Soy on Thyroid Function. Sci. Rep. 2019, 9, 3964. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kiani, J.; Imam, S.Z. Medicinal Importance of Grapefruit Juice and Its Interaction with Various Drugs. Nutr. J. 2007, 6, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bailey, D.G.; Malcolm, J.; Arnold, O.; Spence, J.D. Grapefruit Juice-Drug Interactions. Br. J. Clin. Pharmacol. 1998, 46, 101–110. [Google Scholar] [CrossRef] [Green Version]
- Greenblatt, D.J. Analysis of Drug Interactions Involving Fruit Beverages and Organic Anion-Transporting Polypeptides. J. Clin. Pharmacol. 2009, 49, 1403–1407. [Google Scholar] [CrossRef] [PubMed]
- Egashira, K.; Ohtani, H.; Itoh, S.; Koyabu, N.; Tsujimoto, M.; Murakami, H.; Sawada, Y. Inhibitory Effects of Pomelo on the Metabolism of Tacrolimus and the Activities of CYP3A4 and P-Glycoprotein. Drug Metab. Dispos. 2004, 32, 828–833. [Google Scholar] [CrossRef] [Green Version]
- Shirasaka, Y.; Shichiri, M.; Mori, T.; Nakanishi, T.; Tamai, I. Major Active Components in Grapefruit, Orange, and Apple Juices Responsible for OATP2B1-Mediated Drug Interactions. J. Pharm. Sci. 2013, 102, 3418–3426. [Google Scholar] [CrossRef] [PubMed]
- Bailey, D.G. Fruit Juice Inhibition of Uptake Transport: A New Type of Food-Drug Interaction. Br. J. Clin. Pharmacol. 2010, 70, 645–655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanley, M.J.; Masse, G.; Harmatz, J.S.; Cancalon, P.F.; Dolnikowski, G.G.; Court, M.H.; Greenblatt, D.J. Effect of Blueberry Juice on Clearance of Buspirone and Flurbiprofen in Human Volunteers. Br. J. Clin. Pharmacol. 2013, 75, 1041–1052. [Google Scholar] [CrossRef] [Green Version]
- Srinivas, N.R. Is Pomegranate Juice a Potential Perpetrator of Clinical Drug-Drug Interactions? Review of the in Vitro, Preclinical and Clinical Evidence. Eur. J. Drug Metab. Pharmacokinet. 2013, 38, 223–229. [Google Scholar] [CrossRef]
- Chen, M.; Zhou, S.Y.; Fabriaga, E.; Zhang, P.H.; Zhou, Q. Food-drug interactions precipitated by fruit juices other than grapefruit juice: An update review. J. Food Drug Anal. 2018, 26, S61–S71. [Google Scholar] [CrossRef] [PubMed]
- Tsujimoto, M.; Agawa, C.; Ueda, S.; Yamane, T.; Kitayama, H.; Terao, A.; Fukuda, T.; Minegaki, T.; Nishiguchi, K. Inhibitory Effects of Juices Prepared from Individual Vegetables on CYP3A4 Activity in Recombinant CYP3A4 and LS180 Cells. Biol. Pharm. Bull. 2017, 40, 1561–1565. [Google Scholar] [CrossRef]
- Guo, X.; Mei, N. Aloe Vera: A Review of Toxicity and Adverse Clinical Effects. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2016, 34, 77–96. [Google Scholar] [CrossRef]
- Ali, M.Y.; Sina, A.A.; Khandker, S.S.; Neesa, L.; Tanvir, E.M.; Kabir, A.; Khalil, M.I.; Gan, S.H. Nutritional Composition and Bioactive Compounds in Tomatoes and Their Impact on Human Health and Disease: A Review. Foods 2020, 26, 45. [Google Scholar] [CrossRef] [PubMed]
- Sunaga, K.; Ohkawa, K.; Nakamura, K.; Ohkubo, A.; Harada, S.; Tsuda, T. Mechanism-Based Inhibition of Recombinant Human Cytochrome P450 3A4 by Tomato Juice Extract. Biol. Pharm. Bull. 2012, 35, 329–334. [Google Scholar] [CrossRef] [Green Version]
- Briguglio, M.; Hrelia, S.; Malaguti, M.; Serpe, L.; Canaparo, R.; Dell’Osso, B.; Galentino, R.; De Michele, S.; Dina, C.Z.; Porta, M.; et al. Food Bioactive Compounds and Their Interference in Drug Pharmacokinetic/Pharmacodynamic Profiles. Pharmaceutics 2018, 10, 277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Awortwe, C.; Makiwane, M.; Reuter, H.; Muller, C.; Louw, J.; Rosenkranz, B. Critical Evaluation of Causality Assessment of Herb-Drug Interactions in Patients. Br. J. Clin. Pharmacol. 2018, 84, 679–693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guengerich, P.F.; Avadhani, N.G. Roles of Cytochrome P450 in Metabolism of Ethanol and Carcinogens. Adv. Exp. Med. Biol. 2018, 1032, 15–35. [Google Scholar] [CrossRef]
- Leach, J.P.; Mohanraj, R.; Borland, W. Alcohol and drugs in epilepsy: Pathophysiology, presentation, possibilities, and prevention. Epilepsia 2012, 53, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Detampel, P.; Beck, M.; Krähenbühl, S.; Huwyler, J. Drug interaction potential of resveratrol. Drug Metab. Rev. 2012, 44, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Mutschler, J.; Grosshans, M.; Soyka, M.; Rösner, S. Current Findings and Mechanisms of Action of Disulfiram in the Treatment of Alcohol Dependence. Pharmacopsychiatry 2016, 49, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Skinner, M.D.; Lahmek, P.; Pham, H.; Aubin, H.J. Disulfiram efficacy in the treatment of alcohol dependence: A meta-analysis. PLoS ONE 2014, 10, e87366. [Google Scholar] [CrossRef] [Green Version]
- Albassam, A.A.; Markowitz, J.S. An Appraisal of Drug-Drug Interactions with Green Tea (Camellia sinensis). Planta Med. 2017, 83, 496–508. [Google Scholar] [CrossRef] [Green Version]
- Abe, O.; Ono, T.; Sato, H.; Müller, F.; Ogata, H.; Miura, I.; Shikama, Y.; Yabe, H.; Onoue, S.; Fromm, M.F.; et al. Role of (-)-epigallocatechin gallate in the pharmacokinetic interaction between nadolol and green tea in healthy volunteers. Eur. J. Clin. Pharmacol. 2018, 74, 775–783. [Google Scholar] [CrossRef]
- Raaska, K.; Raitasuo, V.; Laitila, J.; Neuvonen, P.J. Effect of Caffeine-Containing versus Decaffeinated Coffee on Serum Clozapine Concentrations in Hospitalised Patients. Basic Clin. Pharmacol. Toxicol. 2004, 94, 13–18. [Google Scholar] [CrossRef]
- Wright, K.P.; Myers, B.L.; Plenzler, S.C.; Drake, C.L.; Badia, P. Acute Effects of Bright Light and Caffeine on Nighttime Melatonin and Temperature Levels in Women Taking and Not Taking Oral Contraceptives. Brain Res. 2000, 873, 310–317. [Google Scholar] [CrossRef]
- Nehlig, A. Interindividual Differences in Caffeine Metabolism and Factors Driving Caffeine Consumption. Pharmacol. Rev. 2018, 70, 384–411. [Google Scholar] [CrossRef] [Green Version]
- Choi, S.O.; Jung, S.-H.; Um, S.-Y.; Jung, S.-J.; Kim, J.-I.; Chung, S.Y. Food-Effect Bioavailability and Fed Bioequivalence Studies. J. Korean Pharm. Sci. 2004, 34, 223–228. [Google Scholar] [CrossRef]
- Koziolek, M.; Carrière, F.; Porter, C.J.H. Lipids in the Stomach—Implications for the Evaluation of Food Effects on Oral Drug Absorption. Pharm. Res. 2018, 35, 55. [Google Scholar] [CrossRef]
- González Canga, A.; Fernández Martínez, N.; Sahagún Prieto, A.M.; García Vieitez, J.J.; Díez Liébana, M.J.; Díez Láiz, R.; Sierra Vega, M. Dietary Fiber and Its Interaction with Drugs. Nutr. Hosp. 2010, 25, 535–539. [Google Scholar]
- Betz, J.M.; Rimmer, C.A.; Saldanha, L.G.; Phillips, M.M.; Andrews, K.W.; Wise, S.A.; Wood, L.J.; Kuszak, A.J.; Gusev, P.A.; Pehrsson, P.R. Challenges in Developing Analytically Validated Laboratory-Derived Dietary Supplement Databases. J. Nutr. 2018, 1, 1406S–1412S. [Google Scholar] [CrossRef]
- Ronis, M.J.J.; Pedersen, K.B.; Watt, J. Adverse Effects of Nutraceuticals and Dietary Supplements. Annu. Rev. Pharmacol. Toxicol. 2018, 58, 583–601. [Google Scholar] [CrossRef] [PubMed]
- Tirona, R.G.; Bailey, D.G. Herbal Product-Drug Interactions Mediated by Induction. Br. J. Clin. Pharmacol. 2006, 61, 677–681. [Google Scholar] [CrossRef] [Green Version]
- Wierzejska, R.E. Dietary Supplements-For Whom? The Current State of Knowledge about the Health Effects of Selected Supplement Use. Int. J. Environ. Res. Public Health 2021, 24, 8897. [Google Scholar] [CrossRef]
- Chang, T.K.H. Activation of Pregnane X Receptor (PXR) and Constitutive Androstane Receptor (CAR) by Herbal Medicines. AAPS J. 2009, 11, 590–601. [Google Scholar] [CrossRef]
- Na, D.H.; Ji, H.Y.; Park, E.J.; Kim, M.S.; Liu, K.H.; Lee, H.S. Evaluation of metabolism-mediated herb–drug interactions. Arch. Pharm. Res. 2011, 34, 1829–1842. [Google Scholar] [CrossRef] [PubMed]
- Hermann, R.; von Richter, O. Clinical evidence of herbal drugs as perpetrators of pharmacokinetic drug interactions. Planta Med. 2012, 78, 1458–1477. [Google Scholar] [CrossRef] [Green Version]
- National Kidney Foundation of Hawaii. Use of Herbal Supplements in Chronic Kidney Disease. Available online: www.kidneyhi.org/use-of-herbal-supplements-in-chronic-kidney-disease (accessed on 21 November 2021).
- Borrelli, F.; Izzo, A.A. Herb-Drug Interactions with St John’s Wort (Hypericum Perforatum): An Update on Clinical Observations. AAPS J. 2009, 11, 710–727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Unger, M. Pharmacokinetic Drug Interactions Involving Ginkgo Biloba. Drug Metab. Rev. 2013, 45, 353–385. [Google Scholar] [CrossRef] [PubMed]
- Crawford, C.; Boyd, C.; Avula, B.; Wang, Y.H.; Khan, I.A.; Deuster, P.A. A Public Health Issue: Dietary Supplements Promoted for Brain Health and Cognitive Performance. J. Altern. Complement. Med. 2020, 26, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.S.; Yu, J.T.; Tan, C.C.; Wang, H.F.; Meng, X.F.; Wang, C.; Jiang, T.; Zhu, X.C.; Tan, L. Efficacy and adverse effects of ginkgo biloba for cognitive impairment and dementia: A systematic review and meta-analysis. J. Alzheimers Dis. 2015, 43, 589–603. [Google Scholar] [CrossRef] [Green Version]
- Roland, P.D.; Nergård, C.S. Ginkgo biloba—Effekt, bivirkninger og interaksjoner [Ginkgo biloba—Effect, adverse events and drug interaction]. Tidsskr Nor Laegeforen 2012, 30, 956–959, Erratum in Tidsskr Nor Laegeforen 2012, 132, 1319. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ke, J.; Li, M.T.; Huo, Y.J.; Cheng, Y.Q.; Guo, S.F.; Wu, Y.; Zhang, L.; Ma, J.; Liu, A.J.; Han, Y. The Synergistic Effect of Ginkgo biloba Extract 50 and Aspirin Against Platelet Aggregation. Drug Des Devel Ther. 2021, 14, 3543–3560. [Google Scholar] [CrossRef]
- Williams, B.; Watanabe, C.M.; Schultz, P.G.; Rimbach, G.; Krucker, T. Age-related effects of Ginkgo biloba extract on synaptic plasticity and excitability. Neurobiol. Aging 2004, 25, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Kehr, J.; Yoshitake, S.; Ijiri, S.; Koch, E.; Noldner, M.; Yoshitake, T. Ginkgo biloba leaf extract (EGb 761(R)) and its specific acylated flavonol constituents increase dopamine and acetylcholine levels in the rat medial prefrontal cortex: Possible implications for the cognitive enhancing properties of EGb 761(R). Int. Psychogeriatr. 2012, 24, S25–S34. [Google Scholar] [CrossRef]
- Yoshitake, T.; Yoshitake, S.; Kehr, J. The Ginkgo biloba extract EGb 761(R) and its main constituent flavonoids and ginkgolides increase extracellular dopamine levels in the rat prefrontal cortex. Br. J. Pharmacol. 2010, 159, 659–668. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fugh-Berman, A. Herb-Drug Interactions. Lancet 2000, 355, 134–138. [Google Scholar] [CrossRef]
- Posadzki, P.; Watson, L.K.; Ernst, E. Adverse effects of herbal medicines: An overview of systematic reviews. Clin. Med. 2013, 13, 7–12. [Google Scholar] [CrossRef]
- Izzo, A.A.; Ernst, E. Interactions between herbal medicines and prescribed drugs: A systematic review. Drugs 2001, 61, 2163–2175. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.J.; Son, D.H.; Chung, T.H.; Lee, Y.J. A Review of the Pharmacological Efficacy and Safety of Licorice Root from Corroborative Clinical Trial Findings. J. Med. Food 2020, 23, 12–20. [Google Scholar] [CrossRef]
- Makino, T.; Okajima, K.; Uebayashi, R.; Ohtake, N.; Inoue, K.; Mizukami, H. 3-Monoglucuronyl-glycyrrhretinic acid is a substrate of organic anion transporters expressed in tubular epithelial cells and plays important roles in licorice-induced pseudoaldosteronism by inhibiting 11β-hydroxysteroid dehydrogenase 2. J. Pharmacol. Exp. Ther. 2012, 342, 297–304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pastorino, G.; Cornara, L.; Soares, S.; Rodrigues, F.; Oliveira, M.B.P.P. Liquorice (Glycyrrhiza glabra): A phytochemical and pharmacological review. Phytother. Res. 2018, 32, 2323–2339. [Google Scholar] [CrossRef]
- Oh, M.R.; Park, S.H.; Kim, S.Y.; Back, H.I.; Kim, M.G.; Jeon, J.Y.; Ha, K.C.; Na, W.T.; Cha, Y.S.; Park, B.H.; et al. Postprandial glucose-lowering effects of fermented red ginseng in subjects with impaired fasting glucose or type 2 diabetes: A randomized, double-blind, placebo-controlled clinical trial. BMC Complement. Altern. Med. 2014, 14, 237. [Google Scholar] [CrossRef] [Green Version]
- Teng, C.M.; Kuo, S.C.; Ko, F.N.; Lee, J.C.; Lee, L.G.; Chen, S.C.; Huang, T.F. Antiplatelet actions of panaxynol and ginsenosides isolated from ginseng. Biochim. Biophys. Acta 1989, 990, 315–320. [Google Scholar] [CrossRef]
- Ramanathan, M.R.; Penzak, S.R. Pharmacokinetic Drug Interactions with Panax Ginseng. Eur. J. Drug Metab. Pharmacokinet. 2017, 42, 545–557. [Google Scholar] [CrossRef]
- Malati, C.Y.; Robertson, S.M.; Hunt, J.D.; Chairez, C.; Alfaro, R.M.; Kovacs, J.A.; Penzak, S.R. Influence of Panax ginseng on cytochrome P450 (CYP)3A and P-glycoprotein (P-gp) activity in healthy participants. J. Clin. Pharmacol. 2012, 52, 932–939. [Google Scholar] [CrossRef] [Green Version]
- Choi, M.K.; Song, I.S. Interactions of ginseng with therapeutic drugs. Arch. Pharm. Res. 2019, 42, 862–878. [Google Scholar] [CrossRef] [PubMed]
- Xin, H.W.; Wu, X.C.; Li, Q.; Yu, A.R.; Zhu, M.; Shen, Y.; Su, D.; Xiong, L. Effects of Schisandra sphenanthera extract on the pharmacokinetics of tacrolimus in healthy volunteers. Br. J. Clin. Pharmacol. 2007, 64, 469–475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seong, S.J.; Kang, W.Y.; Heo, J.K.; Jo, J.; Choi, W.G.; Liu, K.H.; Lee, S.; Choi, M.K.; Han, Y.H.; Lee, H.S.; et al. A comprehensive in vivo and in vitro assessment of the drug interaction potential of red ginseng. Clin. Ther. 2018, 40, 1322–1337. [Google Scholar] [CrossRef]
- Bhardwaj, R.K.; Glaeser, H.; Becquemont, L.; Klotz, U.; Gupta, S.K.; Fromm, M.F. Piperine, a Major Constituent of Black Pepper, Inhibits Human P-Glycoprotein and CYP3A4. J. Pharmacol. Exp. Ther. 2002, 302, 645–650. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, H.-K. The Effects of Black Pepper on the Intestinal Absorption and Hepatic Metabolism of Drugs. Expert Opin. Drug Metab. Toxicol. 2011, 7, 721–729. [Google Scholar] [CrossRef]
- Gurley, B.J.; Fifer, E.K.; Gardner, Z. Pharmacokinetic herb-drug interactions (part 2): Drug interactions involving popular botanical dietary supplements and their clinical relevance. Planta Med. 2012, 78, 1490–1514. [Google Scholar] [CrossRef] [PubMed]
- Bahramsoltani, R.; Rahimi, R.; Farzaei, M.H. Pharmacokinetic Interactions of Curcuminoids with Conventional Drugs: A Review. J. Ethnopharmacol. 2017, 209, 1–12. [Google Scholar] [CrossRef]
- Unlu, A.; Nayir, E.; Dogukan Kalenderoglu, M.; Kirca, O.; Ozdogan, M. Curcumin (Turmeric) and cancer. J. BUON 2016, 21, 1050–1060. [Google Scholar] [PubMed]
- Soleimani, V.; Sahebkar, A.; Hosseinzadeh, H. Turmeric (Curcuma longa) and its major constituent (curcumin) as nontoxic and safe substances: Review. Phytother. Res. 2018, 32, 985–995. [Google Scholar] [CrossRef]
- Gupta, S.C.; Sung, B.; Kim, J.H.; Prasad, S.; Li, S.; Aggarwal, B.B. Multitargeting by turmeric, the golden spice: From kitchen to clinic. Mol. Nutr. Food Res. 2013, 57, 1510–1528. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Choi, H.-K.; Huang, L. State of Panax Ginseng Research: A Global Analysis. Molecules 2017, 22, 1518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baskaran, P.; Covington, K.; Bennis, J.; Mohandass, A.; Lehmann, T.; Thyagarajan, B. Binding Efficacy and Thermogenic Efficiency of Pungent and Nonpungent Analogs of Capsaicin. Molecules 2018, 23, 3198. [Google Scholar] [CrossRef] [Green Version]
- Varghese, S.; Kubatka, P.; Rodrigo, L.; Gazdikova, K.; Caprnda, M.; Fedotova, J.; Zulli, A.; Kruzliak, P.; Büsselber, D. Chili pepper as a body weight-loss food. Int. J. Food Sci. Nutr. 2017, 68, 392–401. [Google Scholar] [CrossRef]
- Fattori, V.; Hohmann, M.S.; Rossaneis, A.C.; Pinho-Ribeiro, F.A.; Verri, W.A. Capsaicin: Current Understanding of Its Mechanisms and Therapy of Pain and Other Pre-Clinical and Clinical Uses. Molecules 2016, 28, 844. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frydas, S.; Varvara, G.; Murmura, G.; Saggini, A.; Caraffa, A.; Antinolfi, P.; Tete’, S.; Tripodi, D.; Conti, F.; Cianchetti, E.; et al. Impact of capsaicin on mast cell inflammation. Int. J. Immunopathol. Pharmacol. 2013, 26, 597–600. [Google Scholar] [CrossRef] [Green Version]
- Espiritu, M.J.; Chen, J.; Yadav, J.; Larkin, M.; Pelletier, R.D.; Chan, J.M.; Gc, J.B.; Natesan, S.; Harrelson, J.P. Mechanisms of Herb-Drug Interactions Involving Cinnamon and CYP2A6: Focus on Time-Dependent Inhibition by Cinnamaldehyde and 2-Methoxycinnamaldehyde. Drug Metab. Dispos. 2020, 48, 1028–1043. [Google Scholar] [CrossRef]
- Singh, N.; Rao, A.S.; Nandal, A.; Kumar, S.; Yadav, S.S.; Ganaie, S.A.; Narasimhan, B. Phytochemical and pharmacological review of Cinnamomum verum J. Presl-a versatile spice used in food and nutrition. Food Chem. 2021, 15, 127773. [Google Scholar] [CrossRef] [PubMed]
- Seibel, R.; Schneider, R.H.; Gottlieb, M.G.V. Effects of Spices (Saffron, Rosemary, Cinnamon, Turmeric and Ginger) in Alzheimer’s Disease. Curr. Alzheimer Res. 2021, 18, 347–357. [Google Scholar] [CrossRef] [PubMed]
| Amitriptyline, imipramine (tricyclic drugs) and mirtazapine (an atypical antidepressant) |
| Duloxetine and venlafaxine (serotonin and norepinephrine reuptake inhibitor, SNRI) |
| Citalopram and fluvoxamine (selective serotonin reuptake inhibitor, SSRI) |
| Isoniazide, iproniazide, nialamide, phenelzine (monoamine oxydase inhibitors, MAOI). |
| Clozapine, olanzapine, quetiapine and risperidone (Second-generation neuroleptics); |
| Valproic acid, lamotrigine, gabapentin, pregabalin and vigabatrin (Antiepileptic drugs) |
| Oxandrolone and prednisone (Androgenic steroids) |
| ⇧ Body Weight | ⇩ Body Weight | ||
|---|---|---|---|
| Antidepressant | mirtazapine, fluvoxamine, phenelzine, citalopram amitriptyline, doxepin, imipramine, trimipramine | Anticancer | |
| Neuroleptics | clozapine, olanzapine, risperidone, quetiapine, haloperidol | Drugs to treat obesity and diabetes | orlistat, metformin, liraglutide, GLP1 agonists |
| Corticosteroids | cortisone, prednisone, oxalandrone | Amphetamines and derivatives | phenifuramine, phentermine |
| Benzodiazepines | alprazolam, diazepam, clonazepam | Antipsychotics | phenothiazines |
| Estrogen progestin | levonorgestrel | Psychostimulants | methylphenidate, glucagon |
| Antiepileptics | valproic acid, gabapentin | Immunomodulators | lenalinimide, aldesleukin, interferon alfa |
| Antidiabetics | glibenclamide, glicazide, repaglinide, pioglitazone | Antihypertensives | captopril, diltiazide, nifedipine, reserpine |
| Interferon alfa | Nasal decongestants | oxymetazoline, nafazoline | |
| Penicillins | ampicillin amoxicillin | Orexin antagonists | suvorexant, lemborexant |
| Antiretrovirals | indinavir, ritonavir | ||
| Antituberculous | isoniazide | ||
| Food | Drug or Drug Classes | Interactions |
|---|---|---|
| Grapefruit, juice and fruit | Amiodarone, amlodipine, antihistamines, atorvastatin, carbamazepine, carvedilol, cyclosporine, diazepan, disopyramide, erythromycin, ethinylestradiol, losartan, repaglinide, sertraline, simvastatin, stomach acid-blocking drugs, tacrolimus, thyroid replacement drugs, triazolam, verapamil | ⇧ Effect of drugs metabolized by cytochrome P450 |
| Hypericum or St. John’s wort | Anticoagulants, antidepressants, anti-proteases, beta-blockers, cyclosporine, oral contraceptives, digoxin, immunosuppressants, statins, theophylline | ⇧ Drugs metabolism lowering therapeutic effectiveness |
| Alcoholic beverages | Anxiolytics, antidepressants, antiepileptics, antihistamines, opioids | Concurrent consumption ⇧ sedative effect or can cause paradoxical effects |
| Caffeine | Anxiolytics, antidepressants, medications for insomnia and urinary incontinence | ⇩ The action |
| Anticoagulants, clozapine, MAOIs, NSAIDs, theophylline | ⇧ The effect | |
| Green leafy vegetables | Anticoagulants (warfarin, acenocoumarol) | Source of vitamin K Alteration of drug effects with sudden changes in their intake |
| Liquorice | Antiarrhythmics, antihypertensives, diuretics, digoxin, | Salt and water retention, hypokalemia |
| Ginkgo Biloba | Warfarin | ⇧ risk of bleeding |
| Milk and dairy | Antibiotics (ciprofloxacin, tetracycline) | ⇩ Absorption |
| Orange, kiwi, papaya, and apple juice | Antihistamines, celiprolol, ciprofloxacin, fexofenadine | ⇩ Absorption |
| Hard cheeses | IMAO, linezolid | Source of tyramine, risk sudden increase in blood pressure |
| High protein intake | Levodopa | ⇩ Absorption |
| Leagy vegetables, soy, cabbage | Levotiroxina | Inhibition of iodine incorporation |
| Ginseng | Antidepressant | Induction of manic episodes |
| Garlic, blueberry juice | Warfarin | ⇧ Risk of bleeding |
| Chocolate | Sertraline, paroxetine, fluoxetine, fluvoxamine, citalopram | ⇧ Effect |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Alessandro, C.; Benedetti, A.; Di Paolo, A.; Giannese, D.; Cupisti, A. Interactions between Food and Drugs, and Nutritional Status in Renal Patients: A Narrative Review. Nutrients 2022, 14, 212. https://doi.org/10.3390/nu14010212
D’Alessandro C, Benedetti A, Di Paolo A, Giannese D, Cupisti A. Interactions between Food and Drugs, and Nutritional Status in Renal Patients: A Narrative Review. Nutrients. 2022; 14(1):212. https://doi.org/10.3390/nu14010212
Chicago/Turabian StyleD’Alessandro, Claudia, Alessia Benedetti, Antonello Di Paolo, Domenico Giannese, and Adamasco Cupisti. 2022. "Interactions between Food and Drugs, and Nutritional Status in Renal Patients: A Narrative Review" Nutrients 14, no. 1: 212. https://doi.org/10.3390/nu14010212
APA StyleD’Alessandro, C., Benedetti, A., Di Paolo, A., Giannese, D., & Cupisti, A. (2022). Interactions between Food and Drugs, and Nutritional Status in Renal Patients: A Narrative Review. Nutrients, 14(1), 212. https://doi.org/10.3390/nu14010212






