Citrulline, Biomarker of Enterocyte Functional Mass and Dietary Supplement. Metabolism, Transport, and Current Evidence for Clinical Use
Abstract
1. Introduction
2. Results
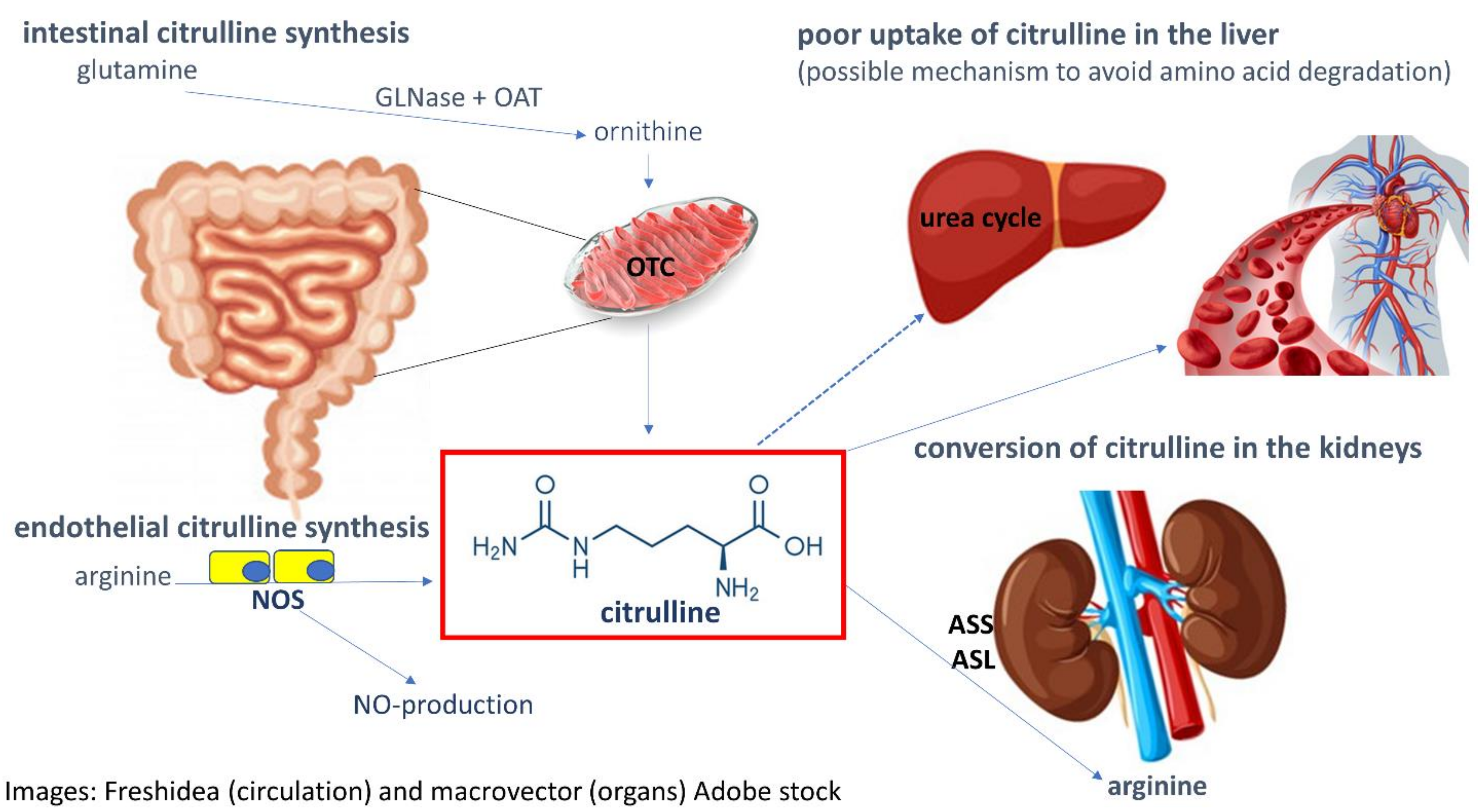
2.1. Citrulline Precursors, Metabolism and Inter-Organ Relationship
2.2. Causes for High and Low Plasma Citrulline Levels
2.3. Citrulline and Cancer
2.4. Intestinal Amino Acid Transporters and Transport Mechanisms—Application to Citrulline
2.5. Brush-Border Membrane Transporters for Citrulline Precursors in Small Intestinal Enterocytes
2.6. Citrulline Transport—Luminal Membrane
2.7. Amino Acid Transport—Basolateral Membrane
2.8. Citrulline Transport—Basolateral Membrane
2.9. Citrulline and Intensive Care Treatment
2.10. Citrulline in Intestinal Development
2.11. Citrulline and Intestinal Microbiota
2.12. Clinical and Therapeutical Implications
2.13. Safety of Oral Citrulline Supplementation
2.14. Oral AA Supplements to Induce Nitric Oxide-Mediated Vasodilation
2.15. Oral L-Citrulline Supplementation to Improve Exercise Performance in Healthy Athletes
2.16. Citrulline Supplementation in Children
2.17. Citrulline Supplementation and Exercise Performance in Sarcopenic Elderly Patients
2.18. Oral Citrulline Supplementation to Improve Non-Alcoholic Fatty Liver Disease
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Study First Author and Year of Publication | N and Patient Characteristics Median (Interquartile Range, IQR) | Intervention | Control | Outcome | Citrulline Level Median (IQR) |
|---|---|---|---|---|---|
| Bourdon, 2012 [37] | 47 Preterm infants <1500 g 29 weeks [27,28,29] | Measurement of citrulline and analysis of potential influencing factors | None | Gastrointestinal (GI) tolerance (citrulline is no predictor of GI tolerance in this age group) | Urinary: 24·7 (14·5–38·6) μmol/mmol creatinine |
| Cox, 2018 [167] | 119 children with sickle cell disease age 8–12, Double blind random order crossover trial | Ready-to-use supplementary food (RUSF) with supplementation of arginine and citrulline (arginine, 0.2 g/kg/day; citrulline, 0.1 g/kg/day) 4 months | RUSF (4 months) and baseline before supplementation washout phases were 4 months | mean height-for-age Z-score and body-mass index-for-age Z-score for both interventions | Baseline 24.53 (23.41–25.70) RUSF with arginine and citrulline 25.52 (22.91–28.41) RUSF alone 21.59 (20.68–22.53) |
| Gosselin, 2014 [170] | 26 children at mean age of 14.9 years undergoing hematopoietic stem cell therapy | Peak mucositis score 2–4 | Peak mucositis score 0–1 | Citrulline was lower when severe mucositis was present. Graft versus host disease preferentially occurred in those patients with very low citrulline levels | Citrulline with severe mucositis 6.7 (95% CI 3.4–13.1) µmol/L-citrulline without severe oral mucositis 11.9 (95% CI 5.8–24.4) µmol/L, p = 0.003 |
| Ioannou, 2010 [171] | 73 children Age 7.6 ± 1.6 years | Citrulline levels according to disease group (celiac disease with or without diet) | Citrulline levels in healthy controls | Patients with celiac disease: 24.5 ± 4.9 μmol/L patients with celiac disease on gluten-free diet: 31.2 ± 6.7 μmol/L patients with gastrointestinal symptoms but normal mucosa: 30.3 ± 4.7 μmol/L healthy controls: 32.4 ± 7.5 μmol/L | |
| Ioannou, 2012 [169] | 41 preterm at a gestational age of 30.8 ± 2 | Preterms with necrotizing enterocolitis (NEC) (n = 17) | Control preterms (n = 24) | Citrulline cut-off distinguishing NEC patient from controls was 17.75 µmol/L (sensitivity 76%, specificity 87%) | At day 7: 16.85 ± 4.2 vs. 20.5 ± 4.5 μmol/L, p < 0.05 |
| Lanyero, 2019 [168] | 430 children aged 21.6 ± 6.5 months | Severe acute malnutrition (n = 400) | Children with normal nutrition (n = 30) | Citrulline levels with severe acute malnutrition were significantly lower compared to normal nutrition | Children with severe acute malnutrition had citrulline levels of 5.14 vs. 27.4 μmol/L in children with normal nutrition |
| Marealle, 2018 [187] | 29 children with sickle cell disease at age 9–11 | Ready-to-use supplementary food with arginine and citrulline (arginine, 0.2 g/kg/day; ci-trulline, 0.1 g/kg/day for 10.7 weeks) | Ready-to-use supplementary food | (Arginine, 0.2 g/kg/day; ci-trulline, 0.1 g/kg/day) | 48.19 ± 62.66 versus 22.18 ± 6.02 μmol/L, p = 0.07 |
| Posod, 2016 [172] | 108 children at 5–7 years of age | Formerly born preterm (n = 79) | Formerly born at term (n = 29) | Amino acid profiles | Elevated in preterm group 31.72 ± 6.8 versus 26.89 ± 7.24 μmol/L, p < 0.01 |
| Raphael, 2011 [188] | 10 children 30.3 ± 30.5 months of age with cisapride use for gastrointestinal rehabilitation | Observational study | - | Amino acid profiles | 14.5 μmol/L (10.5–31.3 μmol/L) |
| Ruiz, 2020 [174] | 15 children at 3–48 months undergoing reconstructive heart surgery | Citrulline supplementation 5 doses a 3 g/m2 each | Placebo administration | Pulmonary artery pressure was lower in intervention group 67 mmHg IQR 63–73 vs. 81 mmHg IQR 75–82, p = 0.025, treated group vs. placebo | 17.93 (10–27) μmol/L |
| Smith, 2006 [162] | 40 children aged 8.5 (4–29) months | 5 doses of oral citrulline 1.9 g/m2 per dose (n = 20) | 5 doses of placebo(n = 20) | If the citrulline level was higher than 37µmol/L, no pulmonary hypertension occurred | 36 [28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48] µmol/L vs. 26 (24–35) µmol/L p = 0.012 |
| Steinbach, 2007 [173] | 122 preterms at a gestational age of 27 (25–28) | Observational study with cholestasis (n = 13) | Without cholestasis (n = 109) | Amino acid profiles | 8.2 [6.6–9.7] vs. 11.4 (7.7–14.3) μmol/L, p = 0.021 |
Appendix B
| First Author and Year | n | Characteristics | Intervention | Control | Outcome | Citrulline Level |
|---|---|---|---|---|---|---|
| Blijlevens, 2004 [80] | 32 | Patients admitted to receive an HLA-matched T-cell-depleted sibling graft, provided they were between 18 and 65 years of age | Aminomix in which a portion of the amino acids were replaced by 200 mL glutamine-dipeptide, L-alanyl-L-glutamine (Dipeptiven, Fresenius-Kabi) | Standard Aminomix (Fresenius-Kabi, Nederland BV‘s-Hertogen- bosch, The Netherlands) parenterally | Significant differences in citrulline, albumin, and CRP apparent on SCT day +21 favoring glutamine-dipeptide supplementation | Citrulline was 18 ± 6 μmol/L with the glutamine-dipeptide and 12 ± 3 μmol/L with the placebo (p = 0.03) |
| Fekkes, 2007 [98] | 40 | Patients participating in a trial of the European Organization for Research and Treatment of Cancer (EORTC 18991), evaluating the efficacy and toxicity of PEG-IFN-a-2b versus controls in high-risk melanoma patients | 8 weeks induction with 6 mg/kg/wk s.c., followed by a five years maintenance with 3 mg/kg/wk s.c | Observation only | The concentrations of citrulline decreased significantly at all time points in the patients treated with PEG-IFN-a during the whole study period. Both compared to baseline and to non-treated controls. No changes in citrulline levels were observed in the non-treated controls | Treatment group Baseline: 26.5 ± 6.0 μmol/L Change in % after 3 Months: −19.4 μmol/L (−31.0/−5.8) (p < 0.05) Control group Baseline: 27.4 ± 7.3 μmol/L Change in % after 3 Months: 5.7 μmol/L (−8.6/22.4) (95% confidence interval) |
| Grilz, 2019 [99] | 957 | Adult patients (aged ≥ 18 years) with a newly diagnosed cancer or a progression of disease after complete or partial remission | / | / | Univariable association between two biomarkers of NET formation, H3Cit and cfDNA, and the risk of mortality in patients with cancer | Only data on citrullinated histone |
| Kim, 2016 [189] | 41 | Patients who received intensity-modulated radiotherapy (IMRT) | Whole pelvic (WP) IMRT | Small field (SF) IMRT | Plasma citrulline levels did not show a serial decrease by radiotherapy volume difference (WP versus SF) and were not relevant to the irradiated doses | WP Baseline: 30.9 ± 13.6 μmol/L At 30 Gy: 38.2 ± 21.8 μmol/L At 60 Gy: 31.6 ± 16.0 μmol/L p-Value: 0.95 SF Baseline: 30.1 ± 10.5 μmol/L At 30 Gy: 24.1 ± 10.5 μmol/L At 60 Gy: 34.4 ± 16.7 μmol/L p-Value: 0.66 |
| Patiroglu, 2015 [190] | 27 | Patients aged 4–17 years receiving chemotherapy regimens with strong mucotoxic effects | Second chemotherapy course given standard oral care (SOC) plus Ankaferd Blood Stopper (ABS) | First chemotherapy course given SOC alone | Stages of oral mucositis were found lower in the second chemotherapy course given SOC plus ABS when compared to first chemotherapy course given SOC alone (p = 0.007) | Control: 44.08 (before) to 23.99 (after) μmol/L (p < 0.001) Intervention: 38.67 (before) to 26.78 (after) μmol/L |
| Vidal-Casarieg [191] | 69 | Patients who needed RT because of pelvic or abdominal malignancies | Glutamine (30 g/d) | Placebo (casein, 30 g/d) | Final citrulline levels were similar between groups. Citrulline concentration was reduced during RT with placebo but remained unchanged with glutamine | Glutamine group: 26.31 (10.29) μmol/L Placebo group: 27.69 (12.31) μmol/L p = 0.639 |
| Vliet, 2009 [91] | 9 | Children newly diagnosed with acute myeloid leucemia | Patients were monitored for the presence of oral (mucosal barrier injury) MBI using two different clinical indices for MBI: the NCI symptomatic scale for oral MBI and the daily mouth score (DMS) | / | A significant decrease in plasma citrulline was seen in the post-samples compared to the pre-samples in all chemotherapy cycles | 1.0–29.4 μmol/L |
Appendix C
| First Author and Year of Publication | n | Characteristics | Intervention | Control | Outcome | Citrulline Level in Μmol/Liter [Interquartile Range] |
|---|---|---|---|---|---|---|
| de Betue, 2011 [192] | 51 | Patients from the Medicosurgical Department of Intensive Care, Erasme University Hospital, who required artificial enteral Nutrition support and were over 18 years of age | Formula enriched with free arginine (6.3 g/L) | Isocaloric and isonitrogenous control solution | The time course of the plasma citrulline concentration was unaffected by the type of enteral solution administered | / |
| de Betue, 2013 [175] | 8 | Critically ill infants with respiratory failure because of viral bronchiolitis | Protein-energy–enriched enteral formulas | Standard infant formula (S-formula) | The intake of a PE-formula in critically ill infants resulted in an increased arginine appearance and NO synthesis, whereas citrulline production and plasma arginine con- centrations were unaffected | / |
| Gills, 2021 [193] | 28 | Recreationally active males | 12 g dextrose + 8 g Citrulline-malate (CM) | Placebo (12 g dextrose) | Acute CM supplementation in recreationally active males provides no ergogenic benefit in aerobic cycling performance followed by an anaerobic cycling test. | / |
| Piton, 2011 [194] | 165 | Ventilated adults with shock | Enteral nutrition | Parenteral nutrition | Plasma citrulline concentration was higher after 3 days of enteral nutrition than after 3 days of parenteral nutrition. This result raises the question of the possibility that enteral nutrition is associated with more rapid restoration of enterocyte mass than parenteral nutrition | 18.7 [13.4; 29.2] enteral nutrition 15.3 [9.8; 21.2] in parenteral nutrition |
| Viana, 2020 [195] | 37 | Critically ill patients (ITT), on mechanical ventilation, with full treatment and a functional gastrointestinal tract | Enteral hydroxymethylbutyrate (HMB) twice 1.5 g/day | Placebo | There was no significant difference in production of essential amino acids and branched-chain amino acids over time (p = 0.1148 and p = 0.1758, respectively) and also no interaction between groups (p = 0.2163 and p = 0.2144, respectively.) However, the HMB group had significant higher conversions over time of arginine to citrulline (nitric oxide synthesis: p = 0.0093) and glutamate to glutamine (p = 0.038). In addition, the HMB group showed over time a higher citrulline production (0.0026) | / |
| Ware, 2013 [196] | 135 | Patients with severe sepsis | Analysis of potential association between citrulline levels and end organ damage | / | Plasma citrulline levels were below normal in all patients and were significantly lower in acute respiratory distress syndrome (ARDS) compared to the no ARDS group. The rate of ARDS was 50% in the lowest citrulline quartile compared to 15% in the highest citrulline quartile (p = 0.002). In multivariable analyses, citrulline levels were associated with ARDS even after adjustment for covariates, including severity of illness | Median 9.2 (5.2–14.4) ARDS: 6 (3.3–10.4) No ARDS 10.1(6.2–16.6) |
Appendix D
| Study First Author and Year of Publication | N and Patient Characteristics Median (Interquartile Range, IQR) | Intervention | Control | Outcome | Citrulline Level Median (IQR) |
|---|---|---|---|---|---|
| Jirka, 2019 [197] | 9 adults with short bowel syndrome | 7 day oral citrulline supplementation 0.18 g/kg/d | 7-day placebo supplementation | Increased citrulline level and no effect on protein metabolism | 25 ± 9 vs. 384 ± 95 μmol/L |
| Van der Velden, 2013 [59] | 106 patients with stem cell transplantation | Time course measurement of citrulline and albumin with different stem cell transplant conditioning regimens | Reduced citrulline may be a suitable marker to monitor GI mucositis and precedes the disease course | Below 10 µmol/L was associated with severe GI mucositis | |
| Picot, 2009 [198] | 26 patients with small bowel disruption and double enterostomy | Assessment before chyme reinfusion | Assessment after start of chyme reinfusion 30 ± 33 days (range 8–186 days) | Assessment citrulline level in each patient | 17.0 ± 10.0 vs. 31.0 ± 12.0 μmol/L; p = 0.0001 |
| Luo, 2007 [77] | 24 patients | Growth hormone treatment (n = 15) | Placebo treatment (n = 9) | Correlation of 0.47, p = 0.028 between bowel length and citrulline levels | 24 ± 2 µmol/L |
| Hyšpler, 2015 [199] | 117 patients undergoing colorectal surgery | Serial measurements of citrulline | 24.9 (19.1–32.0) µmol/L on day 4 after surgery, a value below 20 µmol/L achieved a sensitivity and specificity of 75% and 76% in the diagnosis of prolonged |
References
- Koga, Y. Study report on the constituents of squeezed watermelon. Tokyo Kagaku Kaishi J. Tokyo Chem. Soc. 1914, 35, 519–528. [Google Scholar]
- Fearon, W.R. The carbamido diacetyl reaction: A test for citrulline. Biochem. J. 1939, 33, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Rogers, G.E.; Harding, H.W.; Llewellyn-Smith, I.J. The origin of citrulline-containing proteins in the hair follicle and the chemical nature of trichohyalin, an intracellular precursor. Biochim. et Biophys. Acta (BBA) Protein Struct. 1977, 495, 159–175. [Google Scholar] [CrossRef]
- Vossenaar, E.R.; Zendman, A.J.; van Venrooij, W.J.; Pruijn, G.J. PAD, a growing family of citrullinating enzymes: Genes, features and involvement in disease. BioEssays 2003, 25, 1106–1118. [Google Scholar] [CrossRef]
- Papadia, C.; Osowska, S.; Cynober, L.; Forbes, A. Citrulline in health and disease. Review on human studies. Clin. Nutr. 2018, 37, 1823–1828. [Google Scholar] [CrossRef] [PubMed]
- Bouillanne, O.; Melchior, J.-C.; Faure, C.; Paul, M.; Canoui-Poitrine, F.; Boirie, Y.; Chevenne, D.; Forasassi, C.; Guery, E.; Herbaud, S.; et al. Impact of 3-week citrulline supplementation on postprandial protein metabolism in malnourished older patients: The Ciproage randomized controlled trial. Clin. Nutr. 2018, 38, 564–574. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Morita, M.; Kobayashi, Y.; Kamimura, A. Oral L-citrulline supplementation enhances cycling time trial performance in healthy trained men: Double-blind randomized placebo-controlled 2-way crossover study. J. Int. Soc. Sports Nutr. 2016, 13, 6. [Google Scholar] [CrossRef]
- Crenn, P.; Vahedi, K.; Lavergne-Slove, A.; Cynober, L.; Matuchansky, C.; Messing, B. Plasma citrulline: A marker of enterocyte mass in villous atrophy-associated small bowel disease. Gastroenterol. 2003, 124, 1210–1219. [Google Scholar] [CrossRef]
- Curis, E.; Crenn, P.; Cynober, L. Citrulline and the gut. Curr. Opin. Clin. Nutr. Metab. Care 2007, 10, 620–626. [Google Scholar] [CrossRef]
- Windmueller, G.H.; Spaeth, A.E. Source and fate of circulating citrulline. Am. J. Physiol. -Endocrinol. Metab. 1981, 241, E473–E480. [Google Scholar] [CrossRef]
- Lau, T.; Owen, W.; Yu, Y.M.; Noviski, N.; Lyons, J.; Zurakowski, D.; Tsay, R.; Ajami, A.; Young, V.R.; Castillo, L. Arginine, citrulline, and nitric oxide metabolism in end-stage renal disease patients. J. Clin. Investig. 2000, 105, 1217–1225. [Google Scholar] [CrossRef]
- Rimando, M.A.; Perkins-Veazie, P.M. Determination of citrulline in watermelon rind. J. Chromatogr. A 2005, 1078, 196–200. [Google Scholar] [CrossRef]
- Trexler, E.T.; Persky, A.; Ryan, E.D.; Schwartz, T.; Stoner, L.; Smith-Ryan, A.E. Acute Effects of Citrulline Supplementation on High-Intensity Strength and Power Performance: A Systematic Review and Meta-Analysis. Sports Med. 2019, 49, 707–718. [Google Scholar] [CrossRef] [PubMed]
- Mirenayat, M.S.; Moradi, S.; Mohammadi, H.; Rouhani, M.H. Effect of L-Citrulline Supplementation on Blood Pressure: A Systematic Review and Meta-Analysis of Clinical Trials. Curr. Hypertens. Rep. 2018, 20, 98. [Google Scholar] [CrossRef] [PubMed]
- Wu, G. Intestinal Mucosal Amino Acid Catabolism. J. Nutr. 1998, 128, 1249–1252. [Google Scholar] [CrossRef] [PubMed]
- Fujita, T.; Yanaga, K. Association between glutamine extraction and release of citrulline and glycine by the human small intestine. Life Sci. 2007, 80, 1846–1850. [Google Scholar] [CrossRef]
- van de Poll, M.; Ligthart-Melis, G.C.; Boelens, P.G.; Deutz, N.; Van Leeuwen, P.A.M.; DeJong, C.H.C. Intestinal and hepatic metabolism of glutamine and citrulline in humans. J. Physiol. 2007, 581, 819–827. [Google Scholar] [CrossRef]
- Marini, J.C.; Keller, B.; Didelija, I.C.; Castillo, L.; Lee, B. Enteral arginase II provides ornithine for citrulline synthesis. Am. J. Physiol. Metab. 2011, 300, E188–E194. [Google Scholar] [CrossRef]
- Buijs, N.; Brinkmann, S.J.; Oosterink, J.E.; Luttikhold, J.; Schierbeek, H.; Wisselink, W.; Beishuizen, A.; van Goudoever, J.; Houdijk, A.P.; Van Leeuwen, P.A.; et al. Intravenous glutamine supplementation enhances renal de novo arginine synthesis in humans: A stable isotope study. Am. J. Clin. Nutr. 2014, 100, 1385–1391. [Google Scholar] [CrossRef]
- Tomlinson, C.; Rafii, M.; Ball, R.O.; Pencharz, P.B. Arginine Can Be Synthesized from Enteral Proline in Healthy Adult Humans. J. Nutr. 2011, 141, 1432–1436. [Google Scholar] [CrossRef]
- Curis, E.; Nicolis, I.; Moinard, C.; Osowska, S.; Zerrouk, N.; Bénazeth, S.; Cynober, L. Almost all about citrulline in mammals. Amino Acids 2005, 29, 177–205. [Google Scholar] [CrossRef]
- Rougé, C.; Robert, C.D.; Robins, A.; LE Bacquer, O.; Volteau, C.; De La Cochetière, M.-F.; Darmaun, D. Manipulation of citrulline availability in humans. Am. J. Physiol. Liver Physiol. 2007, 293, G1061–G1067. [Google Scholar] [CrossRef]
- Marini, J.C.; Didelija, I.C.; Castillo, L.; Lee, B. Glutamine: Precursor or nitrogen donor for citrulline synthesis? Am. J. Physiol. Metab. 2010, 299, E69–E79. [Google Scholar] [CrossRef]
- Marini, J.C.; Didelija, I.C.; Castillo, L.; Lee, B. Plasma Arginine and Ornithine Are the Main Citrulline Precursors in Mice Infused with Arginine-Free Diets. J. Nutr. 2010, 140, 1432–1437. [Google Scholar] [CrossRef] [PubMed]
- Marini, J.C.; Agarwal, U.; Didelija, I.C.; Azamian, M.; Stoll, B.; Nagamani, S.C. Plasma Glutamine Is a Minor Precursor for the Synthesis of Citrulline: A Multispecies Study. J. Nutr. 2017, 147, 549–555. [Google Scholar] [CrossRef]
- Ligthart-Melis, C.G.; Deutz, N.E.P. Is glutamine still an important precursor of citrulline? Am. J. Physiol.Endocrinol. Metab. 2011, 301, E264–E266. [Google Scholar] [CrossRef] [PubMed]
- Haines, R.J.; Pendleton, L.C.; Eichler, D.C. Argininosuccinate synthase: At the center of arginine metabolism. Int. J. Biochem. Mol. Boil. 2010, 2, 8–23. [Google Scholar]
- Maric, S.; Flüchter, P.; Guglielmetti, L.C.; Staerkle, R.F.; Sasse, T.; Restin, T.; Schneider, C.; Holland-Cunz, S.G.; Crenn, P.; Vuille-Dit-Bille, R.N. Plasma citrulline correlates with basolateral amino acid transporter LAT4 expression in human small intestine. Clin. Nutr. 2020, 40, 2244–2251. [Google Scholar] [CrossRef]
- Castillo, L.; Sánchez, M.; Vogt, J.; Chapman, T.E.; Derojas-Walker, T.C.; Tannenbaum, S.R.; Ajami, A.M.; Young, V.R. Plasma arginine, citrulline, and ornithine kinetics in adults, with observations on nitric oxide synthesis. Am. J. Physiol. Content 1995, 268, E360–E367. [Google Scholar]
- Nakakariya, M.; Shima, Y.; Shirasaka, Y.; Mitsuoka, K.; Nakanishi, T.; Tamai, I. Organic anion transporter OAT1 is involved in renal handling of citrulline. Am. J. Physiol. Physiol. 2009, 297, F71–F79. [Google Scholar] [CrossRef] [PubMed]
- Mitsuoka, K.; Shirasaka, Y.; Fukushi, A.; Sato, M.; Nakamura, T.; Nakanishi, T.; Tamai, I. Transport characteristics of L-citrulline in renal apical membrane of proximal tubular cells. Biopharm. Drug Dispos. 2009, 30, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Verrey, F.; Singer, D.; Ramadan, T.; Vuille-Dit-Bille, R.; Mariotta, L.; Camargo, S. Kidney amino acid transport. Pflügers Arch. J. Physiol. 2009, 458, 53–60. [Google Scholar] [CrossRef]
- Dhanakoti, S.N.; Brosnan, J.T.; Herzberg, G.R.; Brosnan, M.E. Renal arginine synthesis: Studies in vitro and in vivo. Am. J. Physiol. Metab. 1990, 259, E437–E442. [Google Scholar] [CrossRef] [PubMed]
- Moinard, C.; Nicolis, I.; Neveux, N.; Darquy, S.; Bénazeth, S.; Cynober, L. Dose-ranging effects of citrulline administration on plasma amino acids and hormonal patterns in healthy subjects: The Citrudose pharmacokinetic study. Br. J. Nutr. 2007, 99, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.S. Regulation of enzymes of the urea cycle and arginine metabolism. Annu. Rev. Nutr. 2002, 22, 87–105. [Google Scholar] [CrossRef] [PubMed]
- Cynober, L.; Le Boucher, J.; Vasson, M.-P. Arginine metabolism in mammals. J. Nutr. Biochem. 1995, 6, 402–413. [Google Scholar] [CrossRef]
- Bourdon, A.; Rougé, C.; Legrand, A.; Robert, C.D.; Piloquet, H.; Vodovar, M.; Voyer, M.; Rozé, J.-C.; Darmaun, D. Urinary citrulline in very low birth weight preterm infants receiving intravenous nutrition. Br. J. Nutr. 2011, 108, 1150–1154. [Google Scholar] [CrossRef]
- Osowska, S.; Moinard, C.; Loï, C.; Neveux, N.; Cynober, L. Citrulline increases arginine pools and restores nitrogen balance after massive intestinal resection. Gut 2004, 53, 1781–1786. [Google Scholar] [CrossRef] [PubMed]
- Jourdan, M.; Nair, K.S.; Carter, R.E.; Schimke, J.; Ford, G.C.; Marc, J.; Aussel, C.; Cynober, L. Citrulline stimulates muscle protein synthesis in the post-absorptive state in healthy people fed a low-protein diet—A pilot study. Clin. Nutr. 2014, 34, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Ventura, G.; Moinard, C.; Sinico, F.; Carrière, V.; Lasserre, V.; Cynober, L.; De Bandt, J.P. Evidence for a role of the ileum in the control of nitrogen homeostasis via the regulation of arginine metabolism. Br. J. Nutr. 2011, 106, 227–236. [Google Scholar] [CrossRef][Green Version]
- Castillo, L.; Chapman, T.E.; Sanchez, M.; Yu, Y.M.; Burke, J.F.; Ajami, A.M.; Vogt, J.; Young, V.R. Plasma arginine and citrulline kinetics in adults given adequate and arginine-free diets. Proc. Natl. Acad. Sci. USA 1993, 90, 7749–7753. [Google Scholar] [CrossRef] [PubMed]
- Jonker, R.; Deutz, N.E.; Erbland, M.L.; Anderson, P.J.; Engelen, M.P. Alterations in whole-body arginine metabolism in chronic obstructive pulmonary disease. Am. J. Clin. Nutr. 2016, 103, 1458–1464. [Google Scholar] [CrossRef]
- Marini, J.C.; Agarwal, U.; Robinson, J.L.; Yuan, Y.; Didelija, I.C.; Stoll, B.; Burrin, D. The intestinal-renal axis for arginine synthesis is present and functional in the neonatal pig. Am. J. Physiol. Metab. 2017, 313, E233–E242. [Google Scholar] [CrossRef] [PubMed]
- Ioannou, H.; Avgoustides-Savvopoulou, P.; Diamanti, E.; Tsampoura, Z.; Drosou-Agakidou, V. Age-related serial plasma citrulline levels in preterm neonates. Pediatrics 2008, 121, S137–S138. [Google Scholar] [CrossRef]
- Crenn, P.; Hanachi, M.; Neveux, N.; Cynober, L. Circulating citrulline levels: A biomarker for intestinal functionality assessment. Ann. Biol. Clin. 2011, 69, 513–521. [Google Scholar] [CrossRef]
- Crenn, P.; Messing, B.; Cynober, L. Citrulline as a biomarker of intestinal failure due to enterocyte mass reduction. Clin. Nutr. 2008, 27, 328–339. [Google Scholar] [CrossRef] [PubMed]
- Ratner, S.; Petrack, B. Biosynthesis of urea. III. Further studies on arginine synthesis from citrulline. J. Biol. Chem. 1951, 191, 693–705. [Google Scholar] [CrossRef]
- McMurray, W.C. Biochemical Genetics and Mental Retardation. Can. Med Assoc. J. 1962, 87, 486–490. [Google Scholar]
- Allan, J.; Cusworth, D.; Dent, C.; Wilson, V. A disease, probably hereditary, characterised by severe mental deficiency and a constant gross abnormality of aminoacid metabolism. Lancet 1958, 271, 182–187. [Google Scholar] [CrossRef]
- Chen, G.-F.; Baylis, C. In vivo renal arginine release is impaired throughout development of chronic kidney disease. Am. J. Physiol. Physiol. 2010, 298, F95–F102. [Google Scholar] [CrossRef]
- Saheki, T.; Kobayashi, K. Mitochondrial aspartate glutamate carrier (citrin) deficiency as the cause of adult-onset type II citrullinemia (CTLN2) and idiopathic neonatal hepatitis (NICCD). J. Hum. Genet. 2002, 47, 333–341. [Google Scholar] [CrossRef]
- Booth, C.; Dowling, R.H. Functional compensation after small-bowel resection in man: Demonstration by Direct Measurement. Lancet 1966, 288, 146–147. [Google Scholar] [CrossRef]
- Mifsud, F.; Czernichow, S.; Carette, C.; Levy, R.; Ravaud, P.; Cynober, L.; Neveux, N.; Rives-Lange, C.; Eustache, F.; Coupaye, M.; et al. Behaviour of plasma citrulline after bariatric surgery in the BARIASPERM cohort. Clin. Nutr. 2020, 40, 505–510. [Google Scholar] [CrossRef]
- Seidner, D.L.; Joly, F.; Youssef, N.N. Effect of Teduglutide, a Glucagon-like Peptide 2 Analog, on Citrulline Levels in Patients With Short Bowel Syndrome in Two Phase III Randomized Trials. Clin. Transl. Gastroenterol. 2015, 6, e93. [Google Scholar] [CrossRef] [PubMed]
- Jeppesen, P.B.; Pertkiewicz, M.; Messing, B.; Iyer, K.; Seidner, D.L.; O’Keefe, S.; Forbes, A.; Heinze, H.; Joelsson, B. Teduglutide Reduces Need for Parenteral Support Among Patients with Short Bowel Syndrome with Intestinal Failure. Gastroenterology 2012, 143, 1473–1481.e3. [Google Scholar] [CrossRef] [PubMed]
- Funghini, S.; Thusberg, J.; Spada, M.; Gasperini, S.; Parini, R.; Ventura, L.; Meli, C.; De Cosmo, L.; Sibilio, M.; Mooney, S.; et al. Carbamoyl Phosphate Synthetase 1 deficiency in Italy: Clinical and genetic findings in a heterogeneous cohort. Gene 2011, 493, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Tuchman, M.; Tsai, M.Y.; Holzknecht, R.A.; Brusilow, S.W. Carbamyl Phosphate Synthetase and Ornithine Transcarbamylase Activities in Enzyme-Deficient Human Liver Measured by Radiochromatography and Correlated with Outcome. Pediatr. Res. 1989, 26, 77–82. [Google Scholar] [CrossRef]
- Robinson, J.L.; Smith, V.; Stoll, B.; Agarwal, U.; Premkumar, M.H.; Lau, P.; Cruz, S.M.; Manjarin, R.; Olutoye, O.; Burrin, D.; et al. Prematurity reduces citrulline-arginine-nitric oxide production and precedes the onset of necrotizing enterocolitis in piglets. Am. J. Physiol. Liver Physiol. 2018, 315, G638–G649. [Google Scholar] [CrossRef]
- Van Der Velden, W.J.F.M.; Herbers, A.H.E.; Brüggemann, R.J.M.; Feuth, T.; Donnelly, J.P.; Blijlevens, N.M.A. Citrulline and albumin as biomarkers for gastrointestinal mucositis in recipients of hematopoietic SCT. Bone Marrow Transplant. 2013, 48, 977–981. [Google Scholar] [CrossRef]
- Lutgens, L.; Lambin, P. Biomarkers for radiation-induced small bowel epithelial damage: An emerging role for plasma Citrulline. World J. Gastroenterol. 2007, 13, 3033–3042. [Google Scholar] [CrossRef]
- Fitzgibbons, S.; Ching, Y.A.; Valim, C.; Zhou, J.; Iglesias, J.; Duggan, C.; Jaksic, T. Relationship between serum citrulline levels and progression to parenteral nutrition independence in children with short bowel syndrome. J. Pediatr. Surg. 2009, 44, 928–932. [Google Scholar] [CrossRef] [PubMed]
- Pappas, P.A.; Tzakis, A.G.; Gaynor, J.J.; Carreno, M.R.; Ruiz, P.; Huijing, F.; Kleiner, G.; Rabier, D.; Kato, T.; Levi, D.M.; et al. An Analysis of the Association between Serum Citrulline and Acute Rejection among 26 Recipients of Intestinal Transplant. Arab. Archaeol. Epigr. 2004, 4, 1124–1132. [Google Scholar] [CrossRef]
- Piton, G.; Manzon, C.; Monnet, E.; Cypriani, B.; Barbot, O.; Navellou, J.-C.; Carbonnel, F.; Capellier, G. Plasma citrulline kinetics and prognostic value in critically ill patients. Intensiv. Care Med. 2010, 36, 702–706. [Google Scholar] [CrossRef]
- Batra, A.; Keys, S.C.; Johnson, M.; Wheeler, R.A.; Beattie, R.M. Epidemiology, management and outcome of ultrashort bowel syndrome in infancy. Arch. Dis. Child. Fetal Neonatal Ed. 2017, 102, F551–F556. [Google Scholar] [CrossRef]
- Pironi, L. Definitions of intestinal failure and the short bowel syndrome. Best Pr. Res. Clin. Gastroenterol. 2016, 30, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Pironi, L.; Goulet, O.; Buchman, A.; Messing, B.; Gabe, S.; Candusso, M.; Bond, G.; Gupte, G.; Pertkiewicz, M.; Steiger, E.; et al. Outcome on home parenteral nutrition for benign intestinal failure: A review of the literature and benchmarking with the European prospective survey of ESPEN. Clin. Nutr. 2012, 31, 831–845. [Google Scholar] [CrossRef]
- Choudhury, R.A.; Yoeli, D.; Hoeltzel, G.; Moore, H.B.; Prins, K.; Kovler, M.; Goldstein, S.; Holland-Cunz, S.G.; Adams, M.; Roach, J.; et al. STEP improves long-term survival for pediatric short bowel syndrome patients: A Markov decision analysis. J. Pediatr. Surg. 2020, 55, 1802–1808. [Google Scholar] [CrossRef] [PubMed]
- Dewberry, L.C.; Hilton, S.A.; Vuille-dit-Bille, R.N.; Liechty, K.W. Is Tapering Enteroplasty an Alternative to Resection of Dilated Bowel in Small Intestinal Atresia? J. Surg. Res. 2020, 246, 1–5. [Google Scholar] [CrossRef]
- Mangalat, N.; Teckman, J. Pediatric Intestinal Failure Review. Child. 2018, 5, 100. [Google Scholar] [CrossRef] [PubMed]
- Vantini, I.; Benini, L.; Bonfante, F.; Talamini, G.; Sembenini, C.; Chiarioni, G.; Maragnolli, O.; Benini, F.; Capra, F. Survival rate and prognostic factors in patients with intestinal failure. Dig. Liver Dis. 2004, 36, 46–55. [Google Scholar] [CrossRef]
- Messing, B.; Crenn, P.; Beau, P.; Boutron-Ruault, M.C.; Rambaud, J.-C.; Matuchansky, C. Long-term survival and parenteral nutrition dependence in adult patients with the short bowel syndrome. Gastroenterology 1999, 117, 1043–1050. [Google Scholar] [CrossRef]
- Celik, I.H.; Demirel, G.; Canpolat, F.E.; Dilmen, U. Reduced Plasma Citrulline Levels in Low Birth Weight Infants With Necrotizing Enterocolitis. J. Clin. Lab. Anal. 2013, 27, 328–332. [Google Scholar] [CrossRef]
- Crenn, P.; Coudray–Lucas, C.; Thuillier, F.; Cynober, L.; Messing, B. Postabsorptive plasma citrulline concentration is a marker of absorptive enterocyte mass and intestinal failure in humans. Gastroenterology 2000, 119, 1496–1505. [Google Scholar] [CrossRef]
- Diamanti, A.; Panetta, F.; Gandullia, P.; Morini, F.; Noto, C.; Torre, G.; Lezo, A.; Goffredo, B.; Daniele, A.; Gambarara, M. Plasma citrulline as marker of bowel adaptation in children with short bowel syndrome. Langenbeck’s Arch. Surg. 2011, 396, 1041–1046. [Google Scholar] [CrossRef]
- Rhoads, J.M.; Plunkett, E.; Galanko, J.; Lichtman, S.; Taylor, L.; Maynor, A.; Weiner, T.; Freeman, K.; Guarisco, J.L.; Wu, G.Y. Serum citrulline levels correlate with enteral tolerance and bowel length in infants with short bowel syndrome. J. Pediatr. 2005, 146, 542–547. [Google Scholar] [CrossRef]
- JianFeng, G.; Weiming, Z.; Ning, L.; Fangnan, L.; Li, T.; Nan, L.; Jieshou, L. Serum Citrulline Is a Simple Quantitative Marker for Small Intestinal Enterocytes Mass and Absorption Function in Short Bowel Patients. J. Surg. Res. 2005, 127, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Fernández-Estívariz, C.; Manatunga, A.K.; Bazargan, N.; Gu, L.H.; Jones, D.P.; Klapproth, J.M.; Sitaraman, S.V.; Leader, L.M.; Galloway, J.R.; et al. Are plasma citrulline and glutamine biomarkers of intestinal absorptive function in patients with short bowel syndrome? J. Parenter. Enteral Nutr. 2007, 31, 1–7. [Google Scholar] [CrossRef]
- Jeppesen, P.B.; Gabe, S.M.; Seidner, D.L.; Lee, H.-M.; Olivier, C. Citrulline correlations in short bowel syndrome–intestinal failure by patient stratification: Analysis of 24 weeks of teduglutide treatment from a randomized controlled study. Clin. Nutr. 2020, 39, 2479–2486. [Google Scholar] [CrossRef] [PubMed]
- Barzał, J.A.; Szczylik, C.; Rzepecki, P.; Jaworska, M.; Anuszewska, E. Plasma citrulline level as a biomarker for cancer therapy-induced small bowel mucosal damage. Acta Biochim. Pol. 2014, 61, 615–631. [Google Scholar] [CrossRef] [PubMed]
- Blijlevens, N.M.A.; Lutgens, L.C.H.W.; Schattenberg, A.V.M.B.; Donnelly, J.P. Citrulline: A potentially simple quantitative marker of intestinal epithelial damage following myeloablative therapy. Bone Marrow Transplant. 2004, 34, 193–196. [Google Scholar] [CrossRef]
- Crenn, P.; De Truchis, P.; Neveux, N.; Galpérine, T.; Cynober, L.; Melchior, J.C. Plasma citrulline is a biomarker of enterocyte mass and an indicator of parenteral nutrition in HIV-infected patients. Am. J. Clin. Nutr. 2009, 90, 587–594. [Google Scholar] [CrossRef]
- Pironi, L.; Guidetti, M.; Lauro, A.; Zanfi, C.; Agostini, F.; D’Errico, A.; Altimari, A.; Pinna, A.D. Plasma citrulline after small bowel transplantation: Effect of time from transplantation, acute cellular rejection, and renal failure. Clin. Transplant. 2015, 29, 1039–1046. [Google Scholar] [CrossRef] [PubMed]
- Vecino Lopez, R.; Andrés Moreno, A.M.; Ramos Boluda, E.; Martinez-Ojinaga Nodal, E.; Hernanz Macías, A.; Prieto Bozano, G.; Lopez Santamaria, G.; Tovar Larrucea, J.A. Plasma citrulline concentration as a biomarker of intestinal function in short bowel syndrome and in intestinal transplant. An. Pediatr. 2013, 79, 218–223. [Google Scholar]
- Crenn, P.; Neveux, N.; Chevret, S.; Jaffray, P.; Cynober, L.; Melchior, J.-C.; Annane, D. Plasma l-citrulline concentrations and its relationship with inflammation at the onset of septic shock: A pilot study. J. Crit. Care 2014, 29, 315.e1–315.e6. [Google Scholar] [CrossRef]
- Krief, J.O.; De Tauriers, P.H.; Dumenil, C.; Neveux, N.; Dumoulin, J.; Giraud, V.; Labrune, S.; Tisserand, J.; Julie, C.; Emile, J.-F.; et al. Role of antibiotic use, plasma citrulline and blood microbiome in advanced non-small cell lung cancer patients treated with nivolumab. J. Immunother. Cancer 2019, 7, 176. [Google Scholar] [CrossRef] [PubMed]
- Fragkos, K.C.; Forbes, A. Citrulline as a marker of intestinal function and absorption in clinical settings: A systematic review and meta-analysis. United Eur. Gastroenterol. J. 2018, 6, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.W.; Javid, P.J.; Oh, J.-T.; Andreoli, S.; Kim, H.B.; Fauza, D.; Jaksic, T. Serial Transverse Enteroplasty Enhances Intestinal Function in a Model of Short Bowel Syndrome. Ann. Surg. 2006, 243, 223–228. [Google Scholar] [CrossRef]
- Frongia, G.; Kessler, M.; Weih, S.; Nickkholgh, A.; Mehrabi, A.; Holland-Cunz, S. Comparison of LILT and STEP procedures in children with short bowel syndrome—A systematic review of the literature. J. Pediatr. Surg. 2013, 48, 1794–1805. [Google Scholar] [CrossRef]
- Amiot, A.; Messing, B.; Corcos, O.; Panis, Y.; Joly, F. Determinants of home parenteral nutrition dependence and survival of 268 patients with non-malignant short bowel syndrome. Clin. Nutr. 2012, 32, 368–374. [Google Scholar] [CrossRef]
- Bianchi, A. Intestinal loop lengthening—A technique for increasing small intestinal length. J. Pediatr. Surg. 1980, 15, 145–151. [Google Scholar] [CrossRef]
- van Vliet, M.J.; Tissing, W.J.; Rings, E.H.; Koetse, H.A.; Stellaard, F.; Kamps, W.A.; de Bont, E.S. Citrulline as a marker for chemotherapy induced mucosal barrier injury in pediatric patients. Pediatr. Blood Cancer 2009, 53, 1188–1194. [Google Scholar] [CrossRef] [PubMed]
- Abou-Alfa, G.; Qin, S.; Ryoo, B.-Y.; Lu, S.-N.; Yen, C.-J.; Feng, Y.-H.; Lim, H.; Izzo, F.; Colombo, M.; Sarker, D.; et al. Phase III randomized study of second line ADI-PEG 20 plus best supportive care versus placebo plus best supportive care in patients with advanced hepatocellular carcinoma. Ann. Oncol. 2018, 29, 1402–1408. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.; Leung, Y.; Lo, W.; Tsui, S.M.; Lam, K. Remission of hepatocellular carcinoma with arginine depletion induced by systemic release of endogenous hepatic arginase due to transhepatic arterial embolisation, augmented by high-dose insulin: Arginase as a potential drug candidate for hepatocellular carcinoma. Cancer Lett. 2005, 224, 67–80. [Google Scholar] [CrossRef]
- Szlosarek, P.W.; Steele, J.P.; Nolan, L.; Gilligan, D.; Taylor, P.; Spicer, J.; Lind, M.; Mitra, S.; Shamash, J.; Phillips, M.M.; et al. Arginine Deprivation with Pegylated Arginine Deiminase in Patients with Argininosuccinate Synthetase 1-Deficient Malignant Pleural Mesothelioma: A Randomized Clinical Trial. JAMA Oncol. 2017, 3, 58–66. [Google Scholar] [CrossRef]
- Hall, P.E.; Lewis, R.; Syed, N.; Shaffer, R.; Evanson, J.; Ellis, S.; Williams, M.; Feng, X.; Johnston, A.; Thomson, J.A.; et al. A Phase I Study of Pegylated Arginine Deiminase (Pegargiminase), Cisplatin, and Pemetrexed in Argininosuccinate Synthetase 1-Deficient Recurrent High-grade Glioma. Clin. Cancer Res. 2019, 25, 2708–2716. [Google Scholar] [CrossRef]
- Yao, S.; Janku, F.; Subbiah, V.; Stewart, J.; Patel, S.P.; Kaseb, A.; Westin, S.N.; Naing, A.; Tsimberidou, A.M.; Hong, D.; et al. Phase 1 trial of ADI-PEG20 plus cisplatin in patients with pretreated metastatic melanoma or other advanced solid malignancies. Br. J. Cancer 2021, 124, 1533–1539. [Google Scholar] [CrossRef]
- Blijlevens, N.M.A.; Donnelly, J.P.; Naber, A.H.J.; Schattenberg, A.V.M.B.; Depauw, B.E. A randomised, double-blinded, placebo-controlled, pilot study of parenteral glutamine for allogeneic stem cell transplant patients. Support. Care Cancer 2005, 13, 790–796. [Google Scholar] [CrossRef] [PubMed]
- Fekkes, D.; Bannink, M.; Kruit, W.H.J.; Van Gool, A.R.; Mulder, P.G.H.; Sleijfer, S.; Eggermont, A.M.M.; Stoter, G. Influence of pegylated interferon-α therapy on plasma levels of citrulline and arginine in melanoma patients. Amino Acids 2006, 32, 121–126. [Google Scholar] [CrossRef]
- Grilz, E.; Mauracher, L.; Posch, F.; Königsbrügge, O.; Zöchbauer-Müller, S.; Marosi, C.; Lang, I.; Pabinger, I.; Ay, C. Citrullinated histone H3, a biomarker for neutrophil extracellular trap formation, predicts the risk of mortality in patients with cancer. Br. J. Haematol. 2019, 186, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Broer, S.; Fairweather, S.J. Amino Acid Transport. Across the Mammalian Intestine. Compr. Physiol. 2018, 9, 343–373. [Google Scholar] [PubMed]
- Bröer, S. Amino Acid Transport Across Mammalian Intestinal and Renal Epithelia. Physiol. Rev. 2008, 88, 249–286. [Google Scholar] [CrossRef] [PubMed]
- Bröer, S. Amino Acid Transporters as Disease Modifiers and Drug Targets. SLAS Discov. Adv. Life Sci. R&D 2018, 23, 303–320. [Google Scholar] [CrossRef]
- Fleck, C.; Schwertfeger, M.; Taylor, P.M. Regulation of renal amino acid (AA) transport by hormones, drugs and xenobiotics—A review. Amino Acids 2003, 24, 347–374. [Google Scholar] [CrossRef] [PubMed]
- Rudnick, G.; Krämer, R.; Blakely, R.D.; Murphy, D.L.; Verrey, F. The SLC6 transporters: Perspectives on structure, functions, regulation, and models for transporter dysfunction. Pflügers Arch. Eur. J. Physiol. 2013, 466, 25–42. [Google Scholar] [CrossRef]
- Camargo, S.M.; Singer, D.; Makrides, V.; Huggel, K.; Pos, K.M.; Wagner, C.A.; Kuba, K.; Danilczyk, U.; Skovby, F.; Kleta, R.; et al. Tissue-specific amino acid transporter partners ACE2 and collectrin differentially interact with hartnup mutations. Gastroenterology 2009, 136, 872–882. [Google Scholar] [CrossRef] [PubMed]
- Singer, D.; Camargo, S.M.; Ramadan, T.; Schäfer, M.; Mariotta, L.; Herzog, B.; Huggel, K.; Werner, S.; Penninger, J.M.; Verrey, F.; et al. Defective intestinal amino acid absorption in Ace2 null mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G686–G695. [Google Scholar]
- Camargo, S.M.R.; Vuille-Dit-Bille, R.; Mariotta, L.; Ramadan, T.; Huggel, K.; Singer, D.; Götze, O.; Verrey, F. The Molecular Mechanism of Intestinal Levodopa Absorption and Its Possible Implications for the Treatment of Parkinson’s Disease. J. Pharmacol. Exp. Ther. 2014, 351, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Palacín, M.; Nunes, V.; Font-Llitjós, M.; Jiménez-Vidal, M.; Fort, J.; Gasol, E.; Pineda, M.; Feliubadaló, L.; Chillarón, J.; Zorzano, A. The Genetics of Heteromeric Amino Acid Transporters. Physiology 2005, 20, 112–124. [Google Scholar] [CrossRef]
- Wagner, C.A.; Lang, F.; Broer, S. Function and structure of heterodimeric amino acid transporters. Am. J. Physiol. Physiol. 2001, 281, C1077–C1093. [Google Scholar] [CrossRef]
- Danilczyk, U.; Sarao, R.; Remy, C.; Benabbas, C.; Stange, G.; Richter, A.; Arya, S.; Pospisilik, J.A.; Singer, D.; Camargo, S.; et al. Essential role for collectrin in renal amino acid transport. Nat. Cell Biol. 2006, 444, 1088–1091. [Google Scholar] [CrossRef]
- Camargo, S.M.; Vuille-Dit-Bille, R.N.; Meier, C.F.; Verrey, F. ACE2 and gut amino acid transport. Clin. Sci. 2020, 134, 2823–2833. [Google Scholar] [CrossRef]
- Hamming, I.; Timens, W.; Bulthuis, M.L.; Lely, A.T.; Navis, G.; van Goor, H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004, 203, 631–637. [Google Scholar] [CrossRef]
- To, K.F.; Lo, A.W. Exploring the pathogenesis of severe acute respiratory syndrome (SARS): The tissue distribution of the coronavirus (SARS-CoV) and its putative receptor, angiotensin-converting enzyme 2 (ACE2). J. Pathol. 2004, 203, 740–743. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Walls, A.C.; Park, Y.J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020, 181, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Vig, B.S.; Stouch, T.R.; Timoszyk, J.K.; Quan, Y.; Wall, D.A.; Smith, R.L.; Faria, T.N. Human PEPT1 Pharmacophore Distinguishes between Dipeptide Transport and Binding. J. Med. Chem. 2006, 49, 3636–3644. [Google Scholar] [CrossRef]
- Nässl, A.-M.; Rubio-Aliaga, I.; Fenselau, H.; Marth, M.K.; Kottra, G.; Daniel, H. Amino acid absorption and homeostasis in mice lacking the intestinal peptide transporter PEPT1. Am. J. Physiol. Liver Physiol. 2011, 301, G128–G137. [Google Scholar] [CrossRef] [PubMed]
- Böhmer, C.; Bröer, A.; Munzinger, M.; Kowalczuk, S.; Rasko, J.E.J.; Lang, F.; Bröer, S. Characterization of mouse amino acid transporter B0AT1 (slc6a19). Biochem. J. 2005, 389, 745–751. [Google Scholar] [CrossRef]
- Bröer, S.; Bröer, A. Amino acid homeostasis and signalling in mammalian cells and organisms. Biochem. J. 2017, 474, 1935–1963. [Google Scholar] [CrossRef] [PubMed]
- Vuille-Dit-Bille, R.; Ha-Huy, R.; Stover, J.F. Changes in plasma phenylalanine, isoleucine, leucine, and valine are associated with significant changes in intracranial pressure and jugular venous oxygen saturation in patients with severe traumatic brain injury. Amino Acids 2011, 43, 1287–1296. [Google Scholar] [CrossRef] [PubMed]
- Guetg, A.; Mariotta, L.; Bock, L.; Herzog, B.; Fingerhut, R.; Camargo, S.; Verrey, F. Essential amino acid transporter Lat4 (Slc43a2) is required for mouse development. J. Physiol. 2015, 593, 1273–1289. [Google Scholar] [CrossRef]
- Vilches, C.; Boiadjieva-Knöpfel, E.; Bodoy, S.; Camargo, S.; De Heredia, M.L.; Prat, E.; Ormazabal, A.; Artuch, R.; Zorzano, A.; Verrey, F.; et al. Cooperation of Antiporter LAT2/CD98hc with Uniporter TAT1 for Renal Reabsorption of Neutral Amino Acids. J. Am. Soc. Nephrol. 2018, 29, 1624–1635. [Google Scholar] [CrossRef] [PubMed]
- Boiadjieva, E.; Vilches, C.; Bodoy, S.; Oparija, L.; Jando, J.; Nunes, V.; Verrey, F.; Palacin, M. Cooperation of Basolateral Epithelial Amino Acid Transporters TAT1 and LAT2 Investigated in a Double Knockout Mouse Model. FASEB J. 2015, 29, 969.4. [Google Scholar] [CrossRef]
- Verrey, F.; Christian, M.; Grégoire, R.; Lukas, C.K. Glycoprotein-associated amino acid exchangers: Broadening the range of transport specificity. Pflügers Archiv. 2000, 440, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Vuille-Dit-Bille, R.N.; Camargo, S.M.; Emmenegger, L.; Sasse, T.; Kummer, E.; Jando, J.; Hamie, Q.M.; Meier, C.F.; Hunziker, S.; Forras-Kaufmann, Z.; et al. Human intestine luminal ACE2 and amino acid transporter expression increased by ACE-inhibitors. Amino Acids 2015, 47, 693–705. [Google Scholar] [CrossRef]
- Pineda, M.; Fernández, E.; Torrents, D.; Estevez, R.; López, C.; Camps, M.; Lloberas, J.; Zorzano, A.; Palacín, M. Identification of a Membrane Protein, LAT-2, That Co-expresses with 4F2 Heavy Chain, an L-type Amino Acid Transport Activity with Broad Specificity for Small and Large Zwitterionic Amino Acids. J. Biol. Chem. 1999, 274, 19738–19744. [Google Scholar] [CrossRef] [PubMed]
- Hueso, T.; Ekpe, K.; Mayeur, C.; Gatse, A.; Curt, M.J.-C.; Gricourt, G.; Rodriguez, C.; Burdet, C.; Ulmann, G.; Neut, C.; et al. Impact and consequences of intensive chemotherapy on intestinal barrier and microbiota in acute myeloid leukemia: The role of mucosal strengthening. Gut Microbes 2020, 12. [Google Scholar] [CrossRef]
- Baier, J.; Gänsbauer, M.; Giessler, C.; Arnold, H.; Muske, M.; Schleicher, U.; Lukassen, S.; Ekici, A.B.; Rauh, M.; Daniel, C.; et al. Arginase impedes the resolution of colitis by altering the microbiome and metabolome. J. Clin. Investig. 2020, 130, 5703–5720. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.C.; Cope, J.L.; Hsu, J.W.; Dwarkanath, P.; Karnes, J.M.; Luna, R.A.; Hollister, E.B.; Thame, M.M.; Kurpad, A.V.; Jahoor, F. The Microbiome, Intestinal Function, and Arginine Metabolism of Healthy Indian Women Are Different from Those of American and Jamaican Women. J. Nutr. 2015, 146, 706–713. [Google Scholar] [CrossRef]
- Wegner, N.; Wait, R.; Sroka, A.; Eick, S.; Nguyen, K.-A.; Lundberg, K.; Kinloch, A.; Culshaw, S.; Potempa, J.; Venables, P.J. Peptidylarginine deiminase from Porphyromonas gingivalis citrullinates human fibrinogen and α-enolase: Implications for autoimmunity in rheumatoid arthritis. Arthritis Rheum. 2010, 62, 2662–2672. [Google Scholar] [CrossRef]
- Gabarrini, G.; De Smit, M.; Westra, J.; Brouwer, E.; Vissink, A.; Zhou, K.; Rossen, J.; Stobernack, T.; Van Dijl, J.M.; Van Winkelhoff, A.J. The peptidylarginine deiminase gene is a conserved feature of Porphyromonas gingivalis. Sci. Rep. 2015, 5, 13936. [Google Scholar] [CrossRef] [PubMed]
- Curran, A.M.; Naik, P.; Giles, J.T.; Darrah, E. PAD enzymes in rheumatoid arthritis: Pathogenic effectors and autoimmune targets. Nat. Rev. Rheumatol. 2020, 16, 301–315. [Google Scholar] [CrossRef] [PubMed]
- Allerton, T.D.; Proctor, D.N.; Stephens, J.M.; Dugas, T.R.; Spielmann, G.; Irving, B.A. l-Citrulline Supplementation: Impact on Cardiometabolic Health. Nutrients 2018, 10, 921. [Google Scholar] [CrossRef] [PubMed]
- Hecker, M.; Sessa, W.C.; Harris, H.J.; Anggard, E.E.; Vane, J.R. The metabolism of L-arginine and its significance for the biosynthesis of endothelium-derived relaxing factor: Cultured endothelial cells recycle L-citrulline to L-arginine. Proc. Natl. Acad. Sci. USA 1990, 87, 8612–8616. [Google Scholar] [CrossRef] [PubMed]
- Werner, A.; Koschke, M.; Leuchtner, N.; Luckner-Minden, C.; Habermeier, A.; Rupp, J.; Heinrich, C.; Conradi, R.; Closs, E.I.; Munder, M. Reconstitution of T Cell Proliferation under Arginine Limitation: Activated Human T Cells Take Up Citrulline via L-Type Amino Acid Transporter 1 and Use It to Regenerate Arginine after Induction of Argininosuccinate Synthase Expression. Front. Immunol. 2017, 8, 864. [Google Scholar] [CrossRef]
- Urschel, K.L.; Shoveller, A.K.; Uwiera, R.R.E.; Pencharz, P.B.; Ball, R.O. Citrulline Is an Effective Arginine Precursor in Enterally Fed Neonatal Piglets1,2. J. Nutr. 2006, 136, 1806–1813. [Google Scholar] [CrossRef]
- Morris, S.M. Arginases and arginine deficiency syndromes. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 64–70. [Google Scholar] [CrossRef]
- Gramaglia, I.; Velez, J.; Chang, Y.-S.; Caparros-Wanderley, W.; Combes, V.; Grau, G.; Stins, M.F.; Van Der Heyde, H.C. Citrulline protects mice from experimental cerebral malaria by ameliorating hypoargininemia, urea cycle changes and vascular leak. PLoS ONE 2019, 14, e0213428. [Google Scholar] [CrossRef]
- Kim, I.-Y.; Schutzler, S.E.; Schrader, A.; Spencer, H.J.; Azhar, G.; Deutz, N.E.P.; Wolfe, R.R. Acute ingestion of citrulline stimulates nitric oxide synthesis but does not increase blood flow in healthy young and older adults with heart failure. Am. J. Physiol. Metab. 2015, 309, E915–E924. [Google Scholar] [CrossRef]
- Dikalova, A.; Aschner, J.L.; Kaplowitz, M.R.; Cunningham, G.; Summar, M.; Fike, C.D. Combined L-citrulline and tetrahydrobiopterin therapy improves NO signaling and ameliorates chronic hypoxia-induced pulmonary hypertension in newborn pigs. Am. J. Physiol. Lung Cell. Mol. Physiol. 2020, 318, L762–L772. [Google Scholar] [CrossRef]
- Fike, C.D.; Summar, M.; Aschner, J.L. L-citrulline provides a novel strategy for treating chronic pulmonary hypertension in newborn infants. Acta Paediatr. 2014, 103, 1019–1026. [Google Scholar] [CrossRef] [PubMed]
- Hotta, Y.; Shiota, A.; Kataoka, T.; Motonari, M.; Maeda, Y.; Morita, M.; Kimura, K. Oral L-citrulline supplementation improves erectile function and penile structure in castrated rats. Int. J. Urol. 2014, 21, 608–612. [Google Scholar] [CrossRef] [PubMed]
- Goron, A.; Lamarche, F.; Blanchet, S.; Delangle, P.; Schlattner, U.; Fontaine, E.; Moinard, C. Citrulline stimulates muscle protein synthesis, by reallocating ATP consumption to muscle protein synthesis. J. Cachex Sarcopenia Muscle 2019, 10, 919–928. [Google Scholar] [CrossRef]
- Le Plénier, S.; Walrand, S.; Noirt, R.; Cynober, L.; Moinard, C. Effects of leucine and citrulline versus non-essential amino acids on muscle protein synthesis in fasted rat: A common activation pathway? Amino Acids 2011, 43, 1171–1178. [Google Scholar] [CrossRef]
- Kaore, S.N.; Amane, H.S.; Kaore, N.M. Citrulline: Pharmacological perspectives and its role as an emerging biomarker in future. Fundam. Clin. Pharmacol. 2012, 27, 35–50. [Google Scholar] [CrossRef]
- Ouelaa, W.; Jegatheesan, P.; M’Bouyou-Boungou, J.; Vicente, C.; Nakib, S.; Nubret, E.; De Bandt, J.P. Citrulline decreases hepatic endotoxin-induced injury in fructose-induced non-alcoholic liver disease: An ex vivo study in the isolated perfused rat liver. Br. J. Nutr. 2017, 117, 1487–1494. [Google Scholar] [CrossRef]
- Ginguay, A.; Regazzetti, A.; Laprevote, O.; Moinard, C.; De Bandt, J.-P.; Cynober, L.; Billard, J.-M.; Allinquant, B.; Dutar, P. Citrulline prevents age-related LTP decline in old rats. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Jegatheesan, P.; Beutheu, S.; Ventura, G.; Sarfati, G.; Nubret, E.; Kapel, N.; Waligora-Dupriet, A.-J.; Bergheim, I.; Cynober, L.; De-Bandt, J.-P. Effect of specific amino acids on hepatic lipid metabolism in fructose-induced non-alcoholic fatty liver disease. Clin. Nutr. 2015, 35, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Grimble, G.K. Adverse Gastrointestinal Effects of Arginine and Related Amino Acids. J. Nutr. 2007, 137, 1693S–1701S. [Google Scholar] [CrossRef]
- Lassala, A.; Bazer, F.W.; Cudd, T.A.; Li, P.; Li, X.; Satterfield, M.C.; Spencer, T.E.; Wu, G. Intravenous Administration of L-Citrulline to Pregnant Ewes Is More Effective Than L-Arginine for Increasing Arginine Availability in the Fetus. J. Nutr. 2009, 139, 660–665. [Google Scholar] [CrossRef]
- Tran, N.; Amarger, V.; Bourdon, A.; Misbert, E.; Grit, I.; Winer, N.; Darmaun, D. Maternal citrulline supplementation en-hances placental function and fetal growth in a rat model of IUGR: Involvement of insulin-like growth factor 2 and angio-genic factors. J. Matern. Fetal Neonatal Med. 2017, 30, 1906–1911. [Google Scholar] [CrossRef]
- Bourdon, A.; Parnet, P.; Nowak, C.; Tran, N.-T.; Winer, N.; Darmaun, D. L-Citrulline Supplementation Enhances Fetal Growth and Protein Synthesis in Rats with Intrauterine Growth Restriction. J. Nutr. 2016, 146, 532–541. [Google Scholar] [CrossRef] [PubMed]
- Powers, R. L-citrulline administration increases the arginine /ADMA ratio, decxrases blood pressure and improves vascular function in obese pregnant women. Prenancy Hypertens. Int. J. Women’s Cardiovasc. Health. 2015, 5, 4. [Google Scholar]
- Häberle, J.; Boddaert, N.; Burlina, A.; Chakrapani, A.; Dixon, M.; Huemer, M.; Karall, D.; Martinelli, D.; Crespo, P.S.; Santer, R.; et al. Suggested guidelines for the diagnosis and management of urea cycle disorders. Orphanet J. Rare Dis. 2012, 7, 32. [Google Scholar] [CrossRef] [PubMed]
- Barr, F.E.; Tirona, R.G.; Taylor, M.B.; Rice, G.; Arnold, J.; Cunningham, G.; Smith, H.A.; Campbell, A.; Canter, J.A.; Christian, K.G.; et al. Pharmacokinetics and safety of intravenously administered citrulline in children undergoing congenital heart surgery: Potential therapy for postoperative pulmonary hypertension. J. Thorac. Cardiovasc. Surg. 2007, 134, 319–326. [Google Scholar] [CrossRef]
- Schwedhelm, E.; Maas, R.; Freese, R.; Jung, D.; Lukacs, Z.; Jambrecina, A.; Spickler, W.; Schulze, F.; Böger, R.H. Pharmacokinetic and pharmacodynamic properties of oral L-citrulline and L-arginine: Impact on nitric oxide metabolism. Br. J. Clin. Pharmacol. 2008, 65, 51–59. [Google Scholar] [CrossRef]
- Khalaf, D.; Krüger, M.; Wehland, M.; Infanger, M.; Grimm, D. The Effects of Oral l-Arginine and l-Citrulline Supplementation on Blood Pressure. Nutrients 2019, 11, 1679. [Google Scholar] [CrossRef]
- Rashid, J.; Kumar, S.S.; Job, K.M.; Liu, X.; Fike, C.D.; Sherwin, C.M.T. Therapeutic Potential of Citrulline as an Arginine Supplement: A Clinical Pharmacology Review. Pediatr. Drugs 2020, 22, 279–293. [Google Scholar] [CrossRef]
- Xuan, C.; Lun, L.-M.; Zhao, J.-X.; Wang, H.-W.; Wang, J.; Ning, C.-P.; Liu, Z.; Zhang, B.-B.; He, G.-W. L-citrulline for protection of endothelial function from ADMA–induced injury in porcine coronary artery. Sci. Rep. 2015, 5, srep10987. [Google Scholar] [CrossRef]
- Hayashi, T.; Juliet, P.A.R.; Matsui-Hirai, H.; Miyazaki, A.; Fukatsu, A.; Funami, J.; Iguchi, A.; Ignarro, L.J. L-citrulline and L-arginine supplementation retards the progression of high-cholesterol-diet-induced atherosclerosis in rabbits. Proc. Natl. Acad. Sci. USA 2005, 102, 13681–13686. [Google Scholar] [CrossRef] [PubMed]
- Barkhidarian, B.; Khorshidi, M.; Shab-Bidar, S.; Hashemi, B. Effects of L-citrulline supplementation on blood pressure: A systematic review and meta-analysis. Avicenna J. Phytomed. 2019, 9, 10–20. [Google Scholar] [PubMed]
- Smith, H.A.; Canter, J.A.; Christian, K.G.; Drinkwater, D.C.; Scholl, F.G.; Christman, B.W.; Rice, G.D.; Barr, F.E.; Summar, M. Nitric oxide precursors and congenital heart surgery: A randomized controlled trial of oral citrulline. J. Thorac. Cardiovasc. Surg. 2006, 132, 58–65. [Google Scholar] [CrossRef]
- Holguin, F.; Grasemann, H.; Sharma, S.; Winnica, D.; Wasil, K.; Smith, V.; Cruse, M.H.; Perez, N.; Coleman, E.; Scialla, T.J.; et al. L-Citrulline increases nitric oxide and improves control in obese asthmatics. JCI Insight 2019, 4. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.; Blackwell, J.R.; Lord, T.; Vanhatalo, A.; Winyard, P.; Jones, A.M. l-Citrulline supplementation improves O2 uptake kinetics and high-intensity exercise performance in humans. J. Appl. Physiol. 2015, 119, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Cutrufello, P.T.; Gadomski, S.J.; Zavorsky, G.S. The effect of l-citrulline and watermelon juice supplementation on anaerobic and aerobic exercise performance. J. Sports Sci. 2015, 33, 1459–1466. [Google Scholar] [CrossRef]
- Marealle, A.I.; Siervo, S.M.; Wassel, L.; Bluck, A.M.; Prentice, O.; Minzi, P.; Sasi, A.; Kamuhabwa, D.; Soka, J.; Makani, S.E. A Pilot Study of a Non-Invasive Oral Nitrate Stable Isotopic Method Suggests That Arginine and Citrulline Supplementation Increases Whole-Body No Production in Tanzanian Children with Sickle Cell Disease. Nitric Oxide. 2018, 74, 532–541. [Google Scholar] [CrossRef]
- Cox, S.E.; Ellins, E.A.; Marealle, A.I.; Newton, C.R.; Soka, D.; Sasi, P.; di Tanna, G.L.; Johnson, W.; Makani, J.; Prentice, A.M.; et al. Ready-to-Use Food Supplement, with or without Arginine and Citrulline, with Daily Chloroquine in Tanzanian Children with Sickle-Cell Disease: A Double-Blind, Random Order Crossover Trial. Lancet Haematol 2018, 5, e147–e160. [Google Scholar] [CrossRef]
- Lanyero, B.; Grenov, B.; Barungi, N.N.; Namusoke, H.; Michaelsen, K.F.; Mupere, E.; Mølgaard, C.; Jiang, P.; Frøkiær, H.; Wiese, M.; et al. Correlates of Gut Function in Children Hospitalized for Severe Acute Malnutrition, a Cross-sectional Study in Uganda. J. Pediatr. Gastroenterol. Nutr. 2019, 69, 292–298. [Google Scholar] [CrossRef]
- Ioannou, H.P.; Diamanti, E.; Piretzi, K.; Drossou-Agakidou, V.; Augoustides-Savvopoulou, P. Plasma citrulline levels in preterm neonates with necrotizing enterocolitis. Early Hum. Dev. 2012, 88, 563–566. [Google Scholar] [CrossRef] [PubMed]
- Gosselin, K.B.; Feldman, H.A.; Sonis, A.; Bechard, L.J.; Kellogg, M.; Gura, K.; Venick, R.; Gordon, C.M.; Guinan, E.C.; Duggan, C. Serum Citrulline as a Biomarker of Gastrointestinal Function During Hematopoietic Cell Transplantation in Children. J. Pediatr. Gastroenterol. Nutr. 2014, 58, 709–714. [Google Scholar] [CrossRef]
- Ioannou, H.P.; Fotoulaki, M.; Pavlitou, A.; Efstratiou, I.; Augoustides-Savvopoulou, P. Plasma citrulline levels in paediatric patients with celiac disease and the effect of a gluten-free diet. Eur. J. Gastroenterol. Hepatol. 2011, 23, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Posod, A.; Komazec, I.O.; Kager, K.; Peglow, U.P.; Griesmaier, E.; Schermer, E.; Würtinger, P.; Baumgartner, D.; Kiechl-Kohlendorfer, U. Former Very Preterm Infants Show an Unfavorable Cardiovascular Risk Profile at a Preschool Age. PLoS ONE 2016, 11, e0168162. [Google Scholar] [CrossRef] [PubMed]
- Steinbach, M.; Clark, R.H.; Kelleher, A.S.; Flores, C.; White, R.; Chace, D.H.; Spitzer, A.R.; For the Pediatrix Amino-Acid Study Group. Demographic and nutritional factors associated with prolonged cholestatic jaundice in the premature infant. J. Perinatol. 2007, 28, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Silvera Ruiz, S.; Grosso, C.L.; Tablada, M.; Cabrera, M.; de Kremer, R.D.; Juaneda, E.; Laróvere, L.E. Efficacy of oral citrulline supplementation to decrease the risk of pulmonary hypertension after congenital heart disease surgery. Rev. Fac. Cienc. Med. 2020, 77, 249–253. [Google Scholar]
- de Betue, C.T.; Joosten, K.F.; Deutz, N.E.; Vreugdenhil, A.C.; van Waardenburg, D.A. Arginine appearance and nitric oxide synthesis in critically ill infants can be increased with a protein-energy-enriched enteral formula. Am. J. Clin. Nutr. 2013, 98, 907–916. [Google Scholar] [CrossRef]
- Roubenoff, R. Sarcopenia: A major modifiable cause of frailty in the elderly. J. Nutr. Heal. Aging 2000, 4, 140–142. [Google Scholar]
- Manini, T.M.; Clark, B.C. Dynapenia and Aging: An Update. J. Gerontol. Ser. A 2011, 67, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.-P.; Rolland, Y.; Schneider, S.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef]
- Short, K.R.; Bigelow, M.L.; Kahl, J.; Singh, R.; Coenen-Schimke, J.; Raghavakaimal, S.; Nair, K.S. Decline in skeletal muscle mitochondrial function with aging in humans. Proc. Natl. Acad. Sci. USA 2005, 102, 5618–5623. [Google Scholar] [CrossRef] [PubMed]
- Lanza, I.; Short, D.K.; Short, K.; Raghavakaimal, S.; Basu, R.; Joyner, M.J.; McConnell, J.P.; Nair, K.S. Endurance Exercise as a Countermeasure for Aging. Diabetes 2008, 57, 2933–2942. [Google Scholar] [CrossRef]
- Faure, C.; Raynaud-Simon, A.; Ferry, A.; Daugé, V.; Cynober, L.; Aussel, C.; Moinard, C. Leucine and citrulline modulate muscle function in malnourished aged rats. Amino Acids 2011, 42, 1425–1433. [Google Scholar] [CrossRef]
- Thibault, R.; Flet, L.; Vavasseur, F.; Lemerle, M.; Ferchaud-Roucher, V.; Picot, D.; Darmaun, D. Oral citrulline does not affect whole body protein metabolism in healthy human volunteers: Results of a prospective, randomized, double-blind, cross-over study. Clin. Nutr. 2011, 30, 807–811. [Google Scholar] [CrossRef]
- Jirka, A.; Layec, S.; Picot, D.; Bernon-Ferreira, S.; Grasset, N.; Flet, L.; Thibault, R.; Darmaun, D. Effect of oral citrulline supplementation on whole body protein metabolism in adult patients with short bowel syndrome: A pilot, randomized, double-blind, cross-over study. Clin. Nutr. 2019, 38, 2599–2606. [Google Scholar] [CrossRef]
- Weimann, A.; Braga, M.; Carli, F.; Higashiguchi, T.; Hübner, M.; Klek, S.; Laviano, A.; Ljungqvist, O.; Lobo, D.N.; Martindale, R.; et al. ESPEN guideline: Clinical nutrition in surgery. Clin. Nutr. 2017, 36, 623–650. [Google Scholar] [CrossRef]
- Scheede-Bergdahl, C.; Minnella, E.M.; Carli, F. Multi-modal prehabilitation: Addressing the why, when, what, how, who and where next? Anaesthesia 2019, 74, 20–26. [Google Scholar] [CrossRef]
- Darabi, Z.; Darand, M.; Yari, Z.; Hedayati, M.; Faghihi, A.; Agah, S.; Hekmatdoost, A. Inflammatory markers response to citrulline supplementation in patients with non-alcoholic fatty liver disease: A randomized, double blind, placebo-controlled, clinical trial. BMC Res. Notes 2019, 12, 1–5. [Google Scholar] [CrossRef]
- Marealle, A.; Makani, J.; Prentice, A.; Kamuhabwa, A.; Sasi, P.; Minzi, O.; Siervo, M.; Cox, S.; Wassell, S. Systemic Nitric Oxide (NO) Production is Increased in Children with Sickle Cell Disease (SCD) Receiving Fortified Supplementary Food. FASEB J. 2015, 29, LB276. [Google Scholar] [CrossRef]
- Raphael, B.P.; Nurko, S.; Jiang, H.; Hart, K.; Kamin, D.S.; Jaksic, T.; Duggan, C. Cisapride Improves Enteral Tolerance in Pediatric Short-bowel Syndrome With Dysmotility. J. Pediatr. Gastroenterol. Nutr. 2011, 52, 590–594. [Google Scholar] [CrossRef]
- Kim, Y.J.; Park, J.H.; Yun, I.H.; Kim, Y.S. A prospective comparison of acute intestinal toxicity following whole pelvic versus small field intensity-modulated radiotherapy for prostate cancer. Onco Targets Ther. 2016, 9, 1319–1325. [Google Scholar] [CrossRef]
- Patiroglu, T.; Sahin, N.; Unal, E.; Kendirci, M.; Ozdemir, M.A.; Karakukcu, M. Effectiveness of ankaferd blood stopper in prophylaxis and treatment of oral mucositis seen in childhood cancers and correlation with plasma citrulline levels. Pediatric Blood Cancer 2015, 62, S336–S337. [Google Scholar]
- Vidal-Casariego, A.; Calleja-Fernández, A.; De Urbina-González, J.J.O.; Cano-Rodríguez, I.; Cordido, F.; Ballesteros-Pomar, M.D. Efficacy of glutamine in the prevention of acute radiation enteritis: A randomized controlled trial. J. Parenter. Enter. Nutr. 2014, 38, 205–213. [Google Scholar] [CrossRef]
- de Betue, C.T.; van Waardenburg, D.A.; Deutz, N.E.; van Eijk, H.M.; van Goudoever, J.B.; Luiking, Y.C.; Zimmermann, L.J.; Joosten, K.F. Increased protein-energy intake promotes anabolism in critically ill infants with viral bronchiolitis: A double-blind randomised controlled trial. Arch. Dis. Child. 2011, 96, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Gills, J.L.; Glenn, J.M.; Gray, M.; Romer, B.; Lu, H. Acute citrulline-malate supplementation is ineffective during aerobic cycling and subsequent anaerobic performance in recreationally active males. Eur. J. Sport Sci. 2020, 21, 77–83. [Google Scholar] [CrossRef]
- Piton, G.; Manzon, C.; Cypriani, B.; Carbonnel, F.; Capellier, G. Acute intestinal failure in critically ill patients: Is plasma citrulline the right marker? Intensive Care Med. 2011, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Viana, M.; Becce, F.; Schmidt, S.; Bagnoud, G.; Berger, M.; Deutz, N. Beta-hydroxy-beta-methylbutyrate (HMB) modifies amino acid metabolism in critically ill patients. a RCT. Clin. Nutr. ESPEN 2020, 40, 428. [Google Scholar] [CrossRef]
- Ware, L.B.; Magarik, J.A.; Wickersham, N.; Cunningham, G.; Rice, T.W.; Christman, B.W.; Wheeler, A.P.; Bernard, G.R.; Summar, M.L. Low plasma citrulline levels are associated with acute respiratory distress syndrome in patients with severe sepsis. Crit. Care 2013, 17, R10. [Google Scholar] [CrossRef] [PubMed]
- Jirka, A.; Layec, S.; Picot, D.; Bernon-Ferreira, S.; Darmaun, D. SUN-P234: Effect of Citrulline Supplementation on Protein Metabolism in Patients with Short Bowel Syndrome: A Stable Isotope Study. Clin. Nutr. 2016, 35, S131. [Google Scholar] [CrossRef]
- Picot, D.; Garin, L.; Trivin, F.; Kossovsky, M.P.; Darmaun, D.; Thibault, R. Plasma citrulline is a marker of absorptive small bowel length in patients with transient enterostomy and acute intestinal failure. Clin. Nutr. 2010, 29, 235–242. [Google Scholar] [CrossRef]
- Hyšpler, R.; Tichá, A.; Kaska, M.; Žaloudková, L.; Plíšková, L.; Havel, E.; Zadak, Z. Markers of Perioperative Bowel Complications in Colorectal Surgery Patients. Dis. Markers 2015, 2015, 428535. [Google Scholar] [CrossRef]
| Elevated Citrulline | Decreased Citrulline |
|---|---|
| |
| |
|
| Physiological Function | Potential Medical Use |
|---|---|
| Counteracts arginine deficiency such as during conditions of increased arginase activity (hemolysis, liver damage) [137] Protects against cerebral malaria [138] |
| Reduces blood pressures in hypertension [133] Vasodilator for pulmonary hypertension [140,141] Improvement of erectile dysfunction [142] |
| Improved T cell function [135] |
| Counteracts sarcopenia state [6,144] |
| improves the capacity of neuronal networks during aging [147] Attenuates fructose-induced non-alcoholic fatty liver disease [148] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maric, S.; Restin, T.; Muff, J.L.; Camargo, S.M.; Guglielmetti, L.C.; Holland-Cunz, S.G.; Crenn, P.; Vuille-dit-Bille, R.N. Citrulline, Biomarker of Enterocyte Functional Mass and Dietary Supplement. Metabolism, Transport, and Current Evidence for Clinical Use. Nutrients 2021, 13, 2794. https://doi.org/10.3390/nu13082794
Maric S, Restin T, Muff JL, Camargo SM, Guglielmetti LC, Holland-Cunz SG, Crenn P, Vuille-dit-Bille RN. Citrulline, Biomarker of Enterocyte Functional Mass and Dietary Supplement. Metabolism, Transport, and Current Evidence for Clinical Use. Nutrients. 2021; 13(8):2794. https://doi.org/10.3390/nu13082794
Chicago/Turabian StyleMaric, Stefano, Tanja Restin, Julian Louis Muff, Simone Mafalda Camargo, Laura Chiara Guglielmetti, Stefan Gerhard Holland-Cunz, Pascal Crenn, and Raphael Nicolas Vuille-dit-Bille. 2021. "Citrulline, Biomarker of Enterocyte Functional Mass and Dietary Supplement. Metabolism, Transport, and Current Evidence for Clinical Use" Nutrients 13, no. 8: 2794. https://doi.org/10.3390/nu13082794
APA StyleMaric, S., Restin, T., Muff, J. L., Camargo, S. M., Guglielmetti, L. C., Holland-Cunz, S. G., Crenn, P., & Vuille-dit-Bille, R. N. (2021). Citrulline, Biomarker of Enterocyte Functional Mass and Dietary Supplement. Metabolism, Transport, and Current Evidence for Clinical Use. Nutrients, 13(8), 2794. https://doi.org/10.3390/nu13082794






