Metabolic, Affective and Neurocognitive Characterization of Metabolic Syndrome Patients with and without Food Addiction. Implications for Weight Progression
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Dietary Assessment
2.3. Biochemical, Anthropometric and Blood Pressure Measurements
2.4. Psychometric Measures
2.5. Cognitive Assessment
2.6. Statistical Analysis
3. Results
3.1. Descriptive for the Sample
3.2. Comparison of Metabolic and Dietary Measures
3.3. Comparison of Psychological and Neuropsychological Measures
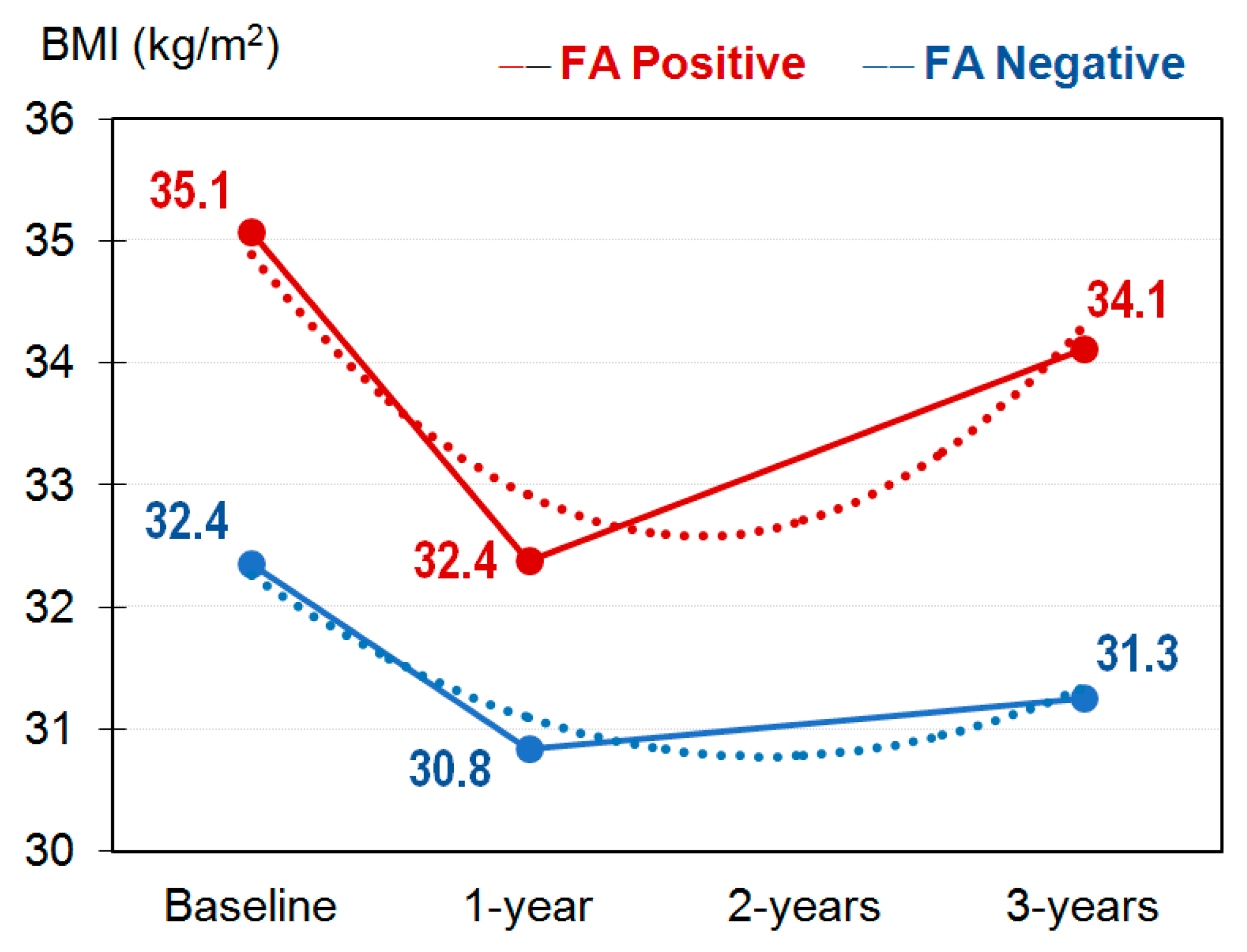
3.4. Evolution of the BMI during the Study
4. Discussion
Limits and Strengths
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schulte, E.M.; Avena, N.M.; Gearhardt, A.N. Which foods may be addictive? The roles of processing, fat content, and glycemic load. PLoS ONE 2015, 10, e0117959. [Google Scholar] [CrossRef] [PubMed]
- Jauch-Chara, K.; Oltmanns, K.M. Obesity—A neuropsychological disease? Systematic review and neuropsychological model. Prog. Neurobiol. 2014, 114, 84–101. [Google Scholar] [CrossRef] [PubMed]
- Gearhardt, A.N.; Corbin, W.R.; Brownell, K.D. Preliminary validation of the Yale Food Addiction Scale. Appetite 2009, 52, 430–436. [Google Scholar] [CrossRef] [Green Version]
- Gearhardt, A.N.; White, M.A.; Masheb, R.M.; Morgan, P.T.; Crosby, R.D.; Grilo, C.M. An examination of the food addiction construct in obese patients with binge eating disorder. Int. J. Eat. Disord. 2012, 45, 657–663. [Google Scholar] [CrossRef] [Green Version]
- Granero, R.; Hilker, I.; Agüera, Z.; Jiménez-Murcia, S.; Sauchelli, S.; Islam, M.A.; Fagundo, A.B.; Sánchez, I.; Riesco, N.; Dieguez, C.; et al. Food addiction in a Spanish sample of eating disorders: DSM-5 diagnostic subtype differentiation and validation data. Eur. Eat. Disord. Rev. 2014, 22, 389–396. [Google Scholar] [CrossRef]
- Guerrero Pérez, F.; Sánchez-González, J.; Sánchez, I.; Jiménez-Murcia, S.; Granero, R.; Simó-Servat, A.; Ruiz, A.; Virgili, N.; López-Urdiales, R.; Montserrat-Gil de Bernabe, M.; et al. Food addiction and preoperative weight loss achievement in patients seeking bariatric surgery. Eur. Eat. Disord. Rev. 2018, 26, 645–656. [Google Scholar] [CrossRef]
- Müller, A.; Leukefeld, C.; Hase, C.; Gruner-Labitzke, K.; Mall, J.W.; Köhler, H.; de Zwaan, M. Food addiction and other addictive behaviours in bariatric surgery candidates. Eur. Eat. Disord. Rev. 2018, 26, 585–596. [Google Scholar] [CrossRef]
- Aloi, M.; Rania, M.; Rodríguez Muñoz, R.C.; Jiménez Murcia, S.; Fernández-Aranda, F.; De Fazio, P.; Segura-Garcia, C. Validation of the Italian version of the Yale Food Addiction Scale 2.0 (I-YFAS 2.0) in a sample of undergraduate students. Eat. Weight. Disord. 2017, 22, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Gearhardt, A.N.; Grilo, C.M.; DiLeone, R.J.; Brownell, K.D.; Potenza, M.N. Can food be addictive? Public health and policy implications. Addiction 2011, 106, 1208–1212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiménez-Murcia, S.; Agüera, Z.; Paslakis, G.; Munguia, L.; Granero, R.; Sánchez-González, J.; Sánchez, I.; Riesco, N.; Gearhardt, A.N.; Dieguez, C.; et al. Food addiction in eating disorders and obesity: Analysis of clusters and implications for treatment. Nutrients 2019, 11, 2633. [Google Scholar] [CrossRef] [Green Version]
- Meule, A.; Müller, A.; Gearhardt, A.N.; Blechert, J. German version of the Yale Food Addiction Scale 2.0: Prevalence and correlates of ‘food addiction’ in students and obese individuals. Appetite 2017, 115, 54–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schulte, E.M.; Gearhardt, A.N. Associations of Food Addiction in a Sample Recruited to Be Nationally Representative of the United States. Eur. Eat. Disord. Rev. 2018, 26, 112–119. [Google Scholar] [CrossRef]
- Som, M.; Val-Laillet, D.; Constant, A.; Moirand, R.; Thibault, R. Prevalence of food addiction (FA) diagnosed by the yale food addiction scale version 2.0 (YFAS 2.0): Prospective study in a referral centre for obesity surgery. Clin. Nutr. 2018, 37, S264. [Google Scholar] [CrossRef]
- Kiyici, S.; Koca, N.; Sigirli, D.; Aslan, B.B.; Guclu, M.; Kisakol, G. Food Addiction Correlates with Psychosocial Functioning More Than Metabolic Parameters in Patients with Obesity. Metab. Syndr. Relat. Disord. 2020, 18, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Ayaz, A.; Nergiz-Unal, R.; Dedebayraktar, D.; Akyol, A.; Pekcan, A.G.; Besler, H.T.; Buyuktuncer, Z. How does food addiction influence dietary intake profile? PLoS ONE 2018, 13, e0195541. [Google Scholar] [CrossRef] [Green Version]
- Mallorquí-Bagué, N.; Lozano-Madrid, M.; Vintró-Alcaraz, C.; Forcano, L.; Díaz-López, A.; Galera, A.; Fernández-Carrión, R.; Granero, R.; Jiménez-Murcia, S.; Corella, D.; et al. Effects of a psychosocial intervention at one-year follow-up in a PREDIMED-plus sample with obesity and metabolic syndrome. Sci. Rep. 2021, 11, 9144. [Google Scholar] [CrossRef]
- De Vries, S.-K.; Meule, A. Food Addiction and Bulimia Nervosa: New Data Based on the Yale Food Addiction Scale 2.0. Eur. Eat. Disord. Rev. 2016, 24, 518–522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Granero, R.; Jiménez-Murcia, S.; Gerhardt, A.N.; Agüera, Z.; Aymamí, N.; Gómez-Peña, M.; Lozano-Madrid, M.; Mallorquí-Bagué, N.; Mestre-Bach, G.; Neto-Antao, M.I.; et al. Validation of the Spanish version of the Yale Food Addiction Scale 2.0 (YFAS 2.0) and clinical correlates in a sample of eating disorder, gambling disorder, and healthy control participants. Front. Psychiatry 2018, 9, 208. [Google Scholar] [CrossRef]
- Meule, A.; von Rezori, V.; Blechert, J. Food Addiction and Bulimia Nervosa. Eur. Eat. Disord. Rev. 2014, 22, 331–337. [Google Scholar] [CrossRef]
- Smith, D.G.; Robbins, T.W. The neurobiological underpinnings of obesity and binge eating: A rationale for adopting the food addiction model. Biol. Psychiatry 2013, 73, 804–810. [Google Scholar] [CrossRef]
- Penzenstadler, L.; Soares, C.; Karila, L.; Khazaal, Y. Systematic Review of Food Addiction as Measured with the Yale Food Addiction Scale: Implications for the Food Addiction Construct. Curr. Neuropharmacol. 2018, 17, 526–538. [Google Scholar] [CrossRef] [PubMed]
- Burrows, T.; Kay-Lambkin, F.; Pursey, K.; Skinner, J.; Dayas, C. Food addiction and associations with mental health symptoms: A systematic review with meta-analysis. J. Hum. Nutr. Diet. 2018, 31, 544–572. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Murcia, S.; Granero, R.; Wolz, I.; Baño, M.; Mestre-Bach, G.; Steward, T.; Agüera, Z.; Hinney, A.; Diéguez, C.; Casanueva, F.F.; et al. Food addiction in gambling disorder: Frequency and clinical outcomes. Front. Psychol. 2017, 8, 473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piccinni, A.; Marazziti, D.; Cargioli, C.; Mauri, M.; Stallone, T. Food addiction: Is it a nosological category or a psychopathological dimension? Preliminary results of an Italian study. Horm. Mol. Biol. Clin. Investig. 2018, 36. [Google Scholar] [CrossRef]
- Fernandez-Aranda, F.; Karwautz, A.; Treasure, J. Food addiction: A transdiagnostic construct of increasing interest. Eur. Eat. Disord. Rev. 2018, 26, 536–540. [Google Scholar] [CrossRef]
- Burrows, T.; Hides, L.; Brown, R.; Dayas, C.; Kay-Lambkin, F. Differences in Dietary Preferences, Personality and Mental Health in Australian Adults with and without Food Addiction. Nutrients 2017, 9, 285. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, E.; Kim, H.S.; Lacroix, E.; de Fátima Vasques, M.; Durante, C.R.; Pereira, D.; Cabral, J.R.; Bernstein, P.S.; Garcia, X.; Ritchie, E.V.; et al. The Clinical Utility of Food Addiction: Characteristics and Psychosocial Impairments in a Treatment-Seeking Sample. Nutrients 2020, 12, 3388. [Google Scholar] [CrossRef]
- Nunes-Neto, P.R.; Köhler, C.A.; Schuch, F.B.; Solmi, M.; Quevedo, J.; Maes, M.; Murru, A.; Vieta, E.; McIntyre, R.S.; McElroy, S.L.; et al. Food addiction: Prevalence, psychopathological correlates and associations with quality of life in a large sample. J. Psychiatr. Res. 2018, 96, 145–152. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Z.; Ma, Y.; Han, Y.; Liu, Y.; Yang, K.; Zhen, S.; Wen, D. Psychosocial Correlates of Food Addiction and Its Association with Quality of Life in a Non-Clinical Adolescent Sample. Nutrients 2018, 10, 837. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steward, T.; Mestre-Bach, G.; Vintró-Alcaraz, C.; Lozano-Madrid, M.; Agüera, Z.; Fernández-Formoso, J.A.; Granero, R.; Jiménez-Murcia, S.; Vilarrasa, N.; García-Ruiz-de-Gordejuela, A.; et al. Food addiction and impaired executive functions in women with obesity. Eur. Eat. Disord. Rev. 2018, 26, 574–584. [Google Scholar] [CrossRef]
- Lacroix, E.; von Ranson, K.M. Prevalence of social, cognitive, and emotional impairment among individuals with food addiction. Eat. Weight Disord. 2020, 26, 1253–1258. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.A.; Buil-Cosiales, P.; Corella, D.; Bulló, M.; Fitó, M.; Vioque, J.; Romaguera, D.; Alfredo Martínez, J.; Wärnberg, J.; López-Miranda, J.; et al. Cohort profile: Design and methods of the PREDIMED-Plus randomized trial. Int. J. Epidemiol. 2019, 48, 387–388o. [Google Scholar] [CrossRef] [Green Version]
- Mataix, J. Tablas de Composición de Alimentos, 4th ed.; Universidad de Granada: Granada, Spain, 2003. [Google Scholar]
- Moreiras, O.; Carvajal, A.; Cabrera, L. Tablas de Composición de Alimentos [Food Composition Tables], 9th ed.; Ediciones Pirámide: Madrid, Spain, 2005. [Google Scholar]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR®); American Psychiatric Association Publishing: Arlington, VA, USA, 2010; ISBN 0890426651, 9780890426654. [Google Scholar]
- Beck, A.T.; Steer, R.A.; Brown, G.K. Manual for the Beck Depression Inventory-II; Psychological Corporation: San Antonio, TX, USA, 1996; Volume 1, p. 82. [Google Scholar]
- Sanz, J.; Perdigón, A.L.; Vázquez, C. Adaptación española del Inventario para la Depresión de Beck-II (BDI-II): 2. Propiedades psicométricas en población general|Revista de psicología. Clínica Salud 2003, 14, 249–280. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Dsm-5TM, 5th ed.; American Psychiatric Association: Arlington, VA, USA, 2013; ISBN 978-0-89042-554-1. [Google Scholar]
- Ware, J.J.; Sherbourne, C. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection—PubMed. Med. Care 1992, 30, 473–483. [Google Scholar] [CrossRef]
- Alonso, J.; Prieto, L.; Anto, J. La versión española del SF-36 Health Survey (Cuestionario de Salud SF-36): Un instrumento para la medida de los resultados clínicos. Med. Clin. 1995, 104, 771–776. [Google Scholar]
- Nasreddine, Z.S.; Phillips, N.A.; Bédirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef]
- Ojeda del Pozo, N.; del Pino Sáez, R.; Ibarretxe Bilbao, N.; Schretlen, D.J.; Peña Lasa, J. Test de evaluación cognitiva de Montreal: Normalización y estandarización de la prueba en población española. Rev. Neurol. 2016, 63, 488. [Google Scholar] [CrossRef]
- Stata-Corp Stata Statistical Software: Release 17 2021. Available online: www.stata.com (accessed on 13 August 2021).
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Kelley, K.; Preacher, K.J. On effect size. Psychol. Methods 2012, 17, 137–152. [Google Scholar] [CrossRef]
- Finner, H.; Roters, M. On the false discovery rate and expected type I errors. Biometr. J. 2001, 43, 985–1005. [Google Scholar] [CrossRef]
- Rubin, D.B. Multiple Imputation for Nonresponse in Surveys; John Wiley & Sons, Inc.: New York, NY, USA, 1987. [Google Scholar]
- Rubin, D.B. Multiple Imputation after 18+ Years. J. Am. Stat. Assoc. 1996, 91, 473–489. [Google Scholar] [CrossRef]
- Hauck, C.; Weiß, A.; Schulte, E.M.; Meule, A.; Ellrott, T. Prevalence of “Food Addiction” as Measured with the Yale Food Addiction Scale 2.0 in a Representative German Sample and Its Association with Sex, Age and Weight Categories. Obes. Facts 2017, 10, 12–24. [Google Scholar] [CrossRef]
- Flint, A.J.; Gearhardt, A.N.; Corbin, W.R.; Brownell, K.D.; Field, A.E.; Rimm, E.B. Food-addiction scale measurement in 2 cohorts of middle-aged and older women. Am. J. Clin. Nutr. 2014, 99, 578–586. [Google Scholar] [CrossRef]
- Shlisky, J.; Bloom, D.E.; Beaudreault, A.R.; Tucker, K.L.; Keller, H.H.; Freund-Levi, Y.; Fielding, R.A.; Cheng, F.W.; Jensen, G.L.; Wu, D.; et al. Nutritional considerations for healthy aging and reduction in age-related chronic disease. Adv. Nutr. 2017, 8, 17–26. [Google Scholar] [CrossRef] [Green Version]
- Van den Beld, A.W.; Kaufman, J.M.; Zillikens, M.C.; Lamberts, S.W.J.; Egan, J.M.; van der Lely, A.J. The physiology of endocrine systems with ageing. Lancet Diabetes Endocrinol. 2018, 6, 647–658. [Google Scholar] [CrossRef] [Green Version]
- Gearhardt, A.N.; Boswell, R.G.; White, M.A. The association of “food addiction” with disordered eating and body mass index. Eat. Behav. 2014, 15, 427–433. [Google Scholar] [CrossRef] [Green Version]
- Pedram, P.; Wadden, D.; Amini, P.; Gulliver, W.; Randell, E.; Cahill, F.; Vasdev, S.; Goodridge, A.; Carter, J.C.; Zhai, G.; et al. Food Addiction: Its Prevalence and Significant Association with Obesity in the General Population. PLoS ONE 2013, 8, e74832. [Google Scholar] [CrossRef] [Green Version]
- Siren, R.; Eriksson, J.G.; Vanhanen, H. Waist circumference a good indicator of future risk for type 2 diabetes and cardiovascular disease. BMC Public Health 2012, 12, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hruby, A.; Hu, F.B. The Epidemiology of Obesity: A Big Picture. Pharmacoeconomics 2015, 33, 673–689. [Google Scholar] [CrossRef] [PubMed]
- Pedram, P.; Sun, G. Hormonal and dietary characteristics in obese human subjects with and without food addiction. Nutrients 2015, 7, 223–238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarkar, S.; Kochhar, K.P.; Khan, N.A. Fat addiction: Psychological and physiological trajectory. Nutrients 2019, 11, 2785. [Google Scholar] [CrossRef] [Green Version]
- Heiskanen, T.H.; Niskanen, L.K.; Hintikka, J.J.; Koivumaa-Honkanen, H.T.; Honkalampi, K.M.; Haatainen, K.M.; Viinamäki, H.T. Metabolic Syndrome and Depression: A Cross-Sectional Analysis. J. Clin. Psychiatry 2006, 67, 1422–1427. [Google Scholar] [CrossRef]
- Skilton, M.R.; Moulin, P.; Terra, J.L.; Bonnet, F. Associations between Anxiety, Depression, and the Metabolic Syndrome. Biol. Psychiatry 2007, 62, 1251–1257. [Google Scholar] [CrossRef] [PubMed]
- Dunbar, J.A.; Reddy, P.; Davis-Lameloise, N.; Philpot, B.; Laatikainen, T.; Kilkkinen, A.; Bunker, S.J.; Best, J.D.; Vartiainen, E.; Lo, S.K.; et al. Depression: An important comorbidity with metabolic syndrome in a general population. Diabetes Care 2008, 31, 2368–2373. [Google Scholar] [CrossRef] [Green Version]
- Koponen, H.; Jokelainen, J.; Keinänen-Kiukaanniemi, S.; Kumpusalo, E.; Vanhala, M. Metabolic Syndrome Predisposes to Depressive Symptoms: A Population-Based 7-Year Follow-Up Study. J. Clin. Psychiatry 2008, 69, 178. [Google Scholar] [CrossRef]
- Simon, G.E.; Ludman, E.J.; Linde, J.A.; Operskalski, B.H.; Ichikawa, L.; Rohde, P.; Finch, E.A.; Jeffery, R.W. Association between obesity and depression in middle-aged women. Gen. Hosp. Psychiatry 2008, 30, 32–39. [Google Scholar] [CrossRef] [Green Version]
- Hryhorczuk, C.; Sharma, S.; Fulton, S.E. Metabolic disturbances connecting obesity and depression. Front. Neurosci. 2013, 7, 177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Munguía, L.; Jiménez-Murcia, S.; Granero, R.; Baenas, I.; Agüera, Z.; Sánchez, I.; Codina, E.; del Pino-Gutiérrez, A.; Testa, G.; Treasure, J.; et al. Emotional regulation in eating disorders and gambling disorder: A transdiagnostic approach. J. Behav. Addict. 2021. [Google Scholar] [CrossRef] [PubMed]
- Beranuy, M.; Machimbarrena, J.M.; Vega-Osés, M.A.; Carbonell, X.; Griffiths, M.D.; Pontes, H.M.; González-Cabrera, J. Spanish Validation of the Internet Gaming Disorder Scale–Short Form (IGDS9-SF): Prevalence and Relationship with Online Gambling and Quality of Life. Int. J. Environ. Res. Public Health 2020, 17, 1562. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, T.; Feng, Z.; Jiang, L.; Yu, Q.; Liu, K. Associations of health behaviors, food preferences, and obesity patterns with the incidence of mild cognitive impairment in the middle-aged and elderly population: An 18-year cohort study. J. Affect. Disord. 2020, 275, 180–186. [Google Scholar] [CrossRef]
- Torres, S.J.; Lautenschlager, N.T.; Wattanapenpaiboon, N.; Greenop, K.R.; Beer, C.; Flicker, L.; Alfonso, H.; Nowson, C.A. Dietary patterns are associated with cognition among older people with mild cognitive impairment. Nutrients 2012, 4, 1542–1551. [Google Scholar] [CrossRef] [Green Version]
- Scarmeas, N.; Stern, Y.; Mayeux, R.; Manly, J.J.; Schupf, N.; Luchsinger, J.A. Mediterranean Diet and Mild Cognitive Impairment. Arch. Neurol. 2009, 66, 216–225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burmeister, J.M.; Hinman, N.; Koball, A.; Hoffmann, D.A.; Carels, R.A. Food addiction in adults seeking weight loss treatment. Implications for psychosocial health and weight loss. Appetite 2013, 60, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Sawamoto, R.; Nozaki, T.; Nishihara, T.; Furukawa, T.; Hata, T.; Komaki, G.; Sudo, N. Predictors of successful long-term weight loss maintenance: A two-year follow-up. Biopsychosoc. Med. 2017, 11, 1–10. [Google Scholar] [CrossRef]
- Gordon, E.L.; Merlo, L.J.; Durning, P.E.; Perri, M.G. Longitudinal Changes in Food Addiction Symptoms and Body Weight among Adults in a Behavioral Weight-Loss Program. Nutrients 2020, 12, 3687. [Google Scholar] [CrossRef] [PubMed]
- Murray, S.M.; Tweardy, S.; Geliebter, A.; Avena, N.M. A Longitudinal Preliminary Study of Addiction-Like Responses to Food and Alcohol Consumption Among Individuals Undergoing Weight Loss Surgery. Obes. Surg. 2019, 29, 2700–2703. [Google Scholar] [CrossRef]
- Sevinçer, G.M.; Konuk, N.; Bozkurt, S.; Coşkun, H. Food addiction and the outcome of bariatric surgery at 1-year: Prospective observational study. Psychiatry Res. 2016, 244, 159–164. [Google Scholar] [CrossRef] [PubMed]
| Total Sample (n = 448) | FA Negative (n = 422) | FA Positive (n = 26) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | χ2 | df | p | ||
| Sex | Male | 217 | 48.4% | 208 | 49.3% | 9 | 34.6% | 2.11 | 1 | 0.146 |
| Female | 231 | 51.6% | 214 | 50.7% | 17 | 65.4% | ||||
| Origin | Europe | 440 | 98.2% | 415 | 98.3% | 25 | 96.2% | 0.67 | 1 | 0.414 |
| South America | 8 | 1.8% | 7 | 1.7% | 1 | 3.8% | ||||
| Civil status | Single | 17 | 3.8% | 16 | 3.8% | 1 | 3.8% | 4.14 | 3 | 0.247 |
| Married | 351 | 78.3% | 333 | 78.9% | 18 | 69.2% | ||||
| Divorced-separated | 30 | 6.7% | 29 | 6.9% | 1 | 3.8% | ||||
| Widowed | 50 | 11.2% | 44 | 10.4% | 6 | 23.1% | ||||
| School | University (high) | 42 | 9.4% | 38 | 9.0% | 4 | 15.4% | 1.39 | 3 | 0.708 |
| University (grade) | 37 | 8.3% | 35 | 8.3% | 2 | 7.7% | ||||
| Secondary | 129 | 28.8% | 123 | 29.1% | 6 | 23.1% | ||||
| Primary | 240 | 53.6% | 226 | 53.6% | 14 | 53.8% | ||||
| Employment | Unemployed | 80 | 17.9% | 76 | 18.0% | 4 | 15.4% | 1.48 | 4 | 0.831 |
| Work at home | 46 | 10.3% | 43 | 10.2% | 3 | 11.5% | ||||
| Retired | 288 | 64.3% | 271 | 64.2% | 17 | 65.4% | ||||
| Unemployed (incomes) | 21 | 4.7% | 19 | 4.5% | 2 | 7.7% | ||||
| Unemployed (no-incomes) | 13 | 2.9% | 13 | 3.1% | 0 | 0.0% | ||||
| Group weight | Over-weight | 123 | 27.5% | 122 | 28.9% | 1 | 3.8% | 14.64 | 3 | 0.002 * |
| Obesity I (BMI 30-35) | 218 | 48.7% | 206 | 48.8% | 12 | 46.2% | ||||
| Obesity II (BMI 35–40) | 103 | 23.0% | 90 | 21.3% | 13 | 50.0% | ||||
| Obesity III (BMI >40) | 4 | 0.9% | 4 | 0.9% | 0 | 0.0% | ||||
| Mean | SD | Mean | SD | Mean | SD | F | df | p | ||
| Age, years-old | 65.25 | 4.63 | 65.22 | 4.63 | 65.73 | 4.64 | 0.30 | 1.446 | 0.582 | |
| FA Negative (n = 422) | FA Positive (n = 26) | |||||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | p | |d| | |
| Total cholesterol, mg/dL | 207.70 | 40.10 | 215.00 | 36.63 | 0.366 | 0.19 |
| Triglycerides, mg/dL | 160.81 | 77.69 | 155.08 | 81.02 | 0.716 | 0.07 |
| LDL cholesterol, mg/dL | 125.65 | 33.69 | 128.58 | 32.47 | 0.666 | 0.09 |
| HDL cholesterol, mg/dL | 51.11 | 12.95 | 55.46 | 10.02 | 0.093 | 0.38 |
| Albumin, g/dL | 4.43 | 0.50 | 4.52 | 0.24 | 0.401 | 0.21 |
| Glucose, mg/dL | 116.81 | 30.18 | 110.81 | 18.53 | 0.317 | 0.24 |
| Insulin, mIU/ml | 18.68 | 8.64 | 18.36 | 8.37 | 0.855 | 0.04 |
| HOMA-IR | 5.45 | 3.08 | 5.13 | 2.66 | 0.607 | 0.11 |
| HbA1c, % | 6.13 | 0.76 | 6.10 | 0.81 | 0.849 | 0.04 |
| Alanine aminotransferase, U/L | 26.95 | 12.60 | 23.20 | 11.10 | 0.139 | 0.32 |
| Aspartate aminotransferase, U/L | 24.12 | 8.45 | 22.85 | 6.81 | 0.454 | 0.17 |
| Systolic blood pressure, mm Hg | 140.75 | 15.03 | 139.69 | 13.21 | 0.725 | 0.07 |
| Diastolic blood pressure, mmHg | 79.93 | 9.50 | 79.73 | 9.76 | 0.918 | 0.02 |
| Physical activity total energy expenditure, MET·min/week | 849.05 | 801.20 | 769.23 | 832.43 | 0.623 | 0.10 |
| Weight, kg | 85.42 | 13.39 | 91.29 | 14.50 | 0.031 * | 0.42 |
| BMI, kg/m2 | 32.38 | 3.39 | 35.06 | 3.07 | <0.001 * | 0.83 † |
| Waist circumference, cm | 107.53 | 10.15 | 111.88 | 11.02 | 0.035 * | 0.41 |
| Total energy intake, kcal/day | 2398.66 | 573.70 | 2527.12 | 815.43 | 0.282 | 0.18 |
| Carbohydrate, g/d | 241.40 | 78.35 | 245.12 | 92.33 | 0.816 | 0.04 |
| Carbohydrate, % | 39.93 | 6.53 | 38.48 | 7.04 | 0.276 | 0.21 |
| Protein, g/d | 100.57 | 20.83 | 104.97 | 26.46 | 0.304 | 0.19 |
| Protein, % | 17.10 | 2.90 | 17.20 | 2.87 | 0.869 | 0.03 |
| Total fat, g/d | 107.66 | 28.71 | 119.47 | 46.04 | 0.052 | 0.31 |
| Total fat, % | 40.52 | 6.12 | 42.55 | 5.66 | 0.099 | 0.35 |
| SFAs, g | 27.69 | 9.21 | 31.65 | 12.89 | 0.039 * | 0.35 |
| SFAs, % | 10.33 | 1.92 | 11.15 | 1.99 | 0.034 * | 0.42 |
| MUFAs, g/d | 55.28 | 14.98 | 63.53 | 25.87 | 0.010 * | 0.39 |
| MUFAs, % | 20.91 | 4.20 | 22.70 | 3.75 | 0.035 * | 0.45 |
| PUFAs, g/d | 18.12 | 6.58 | 19.58 | 8.19 | 0.280 | 0.20 |
| PUFAs, % | 6.80 | 1.80 | 6.93 | 1.41 | 0.710 | 0.08 |
| Trans fatty acids, g/d | 0.65 | 0.41 | 0.87 | 0.50 | 0.009 * | 0.48 |
| Prevalence | n | % | n | % | p | |h| |
| Hypertension | 317 | 75.1% | 23 | 88.5% | 0.123 | 0.35 |
| Diabetes | 121 | 28.7% | 6 | 23.1% | 0.539 | 0.13 |
| Hypercholesterolemia | 216 | 51.2% | 13 | 50.0% | 0.907 | 0.02 |
| FA Negative (n = 422) | FA Positive (n = 26) | |||||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | p | |d| | |
| BDI total score | 7.97 | 6.42 | 14.92 | 9.38 | <0.001 * | 0.87 † |
| SF-36 total score | 78.34 | 16.35 | 63.46 | 18.33 | <0.001 * | 0.86 † |
| SF-36 physical function | 75.84 | 18.98 | 63.27 | 18.05 | 0.001 * | 0.68 † |
| SF-36 physical role | 77.86 | 33.33 | 48.08 | 41.79 | <0.001 * | 0.79 † |
| SF-36 physical pain | 70.43 | 22.54 | 62.31 | 21.41 | 0.075 | 0.37 |
| SF-36 general health | 76.21 | 16.78 | 66.92 | 17.27 | 0.007 * | 0.55 † |
| SF-36 vitality | 62.73 | 20.28 | 46.15 | 22.06 | <0.001 * | 0.78 † |
| SF-36 social function | 92.48 | 14.29 | 84.73 | 19.11 | 0.009 * | 0.46 |
| SF-36 emotional role | 87.92 | 28.19 | 67.96 | 42.69 | 0.001 * | 0.55 † |
| SF-36 mental health | 75.10 | 18.83 | 64.31 | 17.89 | 0.005 * | 0.59 † |
| MoCA total | 23.58 | 4.31 | 21.42 | 6.54 | 0.017 * | 0.39 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Camacho-Barcia, L.; Munguía, L.; Lucas, I.; de la Torre, R.; Salas-Salvadó, J.; Pintó, X.; Corella, D.; Granero, R.; Jiménez-Murcia, S.; González-Monje, I.; et al. Metabolic, Affective and Neurocognitive Characterization of Metabolic Syndrome Patients with and without Food Addiction. Implications for Weight Progression. Nutrients 2021, 13, 2779. https://doi.org/10.3390/nu13082779
Camacho-Barcia L, Munguía L, Lucas I, de la Torre R, Salas-Salvadó J, Pintó X, Corella D, Granero R, Jiménez-Murcia S, González-Monje I, et al. Metabolic, Affective and Neurocognitive Characterization of Metabolic Syndrome Patients with and without Food Addiction. Implications for Weight Progression. Nutrients. 2021; 13(8):2779. https://doi.org/10.3390/nu13082779
Chicago/Turabian StyleCamacho-Barcia, Lucía, Lucero Munguía, Ignacio Lucas, Rafael de la Torre, Jordi Salas-Salvadó, Xavier Pintó, Dolores Corella, Roser Granero, Susana Jiménez-Murcia, Inmaculada González-Monje, and et al. 2021. "Metabolic, Affective and Neurocognitive Characterization of Metabolic Syndrome Patients with and without Food Addiction. Implications for Weight Progression" Nutrients 13, no. 8: 2779. https://doi.org/10.3390/nu13082779
APA StyleCamacho-Barcia, L., Munguía, L., Lucas, I., de la Torre, R., Salas-Salvadó, J., Pintó, X., Corella, D., Granero, R., Jiménez-Murcia, S., González-Monje, I., Esteve-Luque, V., Cuenca-Royo, A., Gómez-Martínez, C., Paz-Graniel, I., Forcano, L., & Fernández-Aranda, F. (2021). Metabolic, Affective and Neurocognitive Characterization of Metabolic Syndrome Patients with and without Food Addiction. Implications for Weight Progression. Nutrients, 13(8), 2779. https://doi.org/10.3390/nu13082779














