Nutrition in Gynecological Diseases: Current Perspectives
Abstract
1. Introduction
2. Infertility
2.1. Mediterranean Diet
2.2. Fats
2.3. Vitamins
2.4. Probiotics
3. Polycystic Ovary Syndrome
4. Uterine Fibroids
4.1. Fats
4.2. Vegetables, Meat, and Phytochemicals
4.3. Dairy Foods and Vitamins
4.4. Pollutants and Metaloestrogens
5. Endometriosis
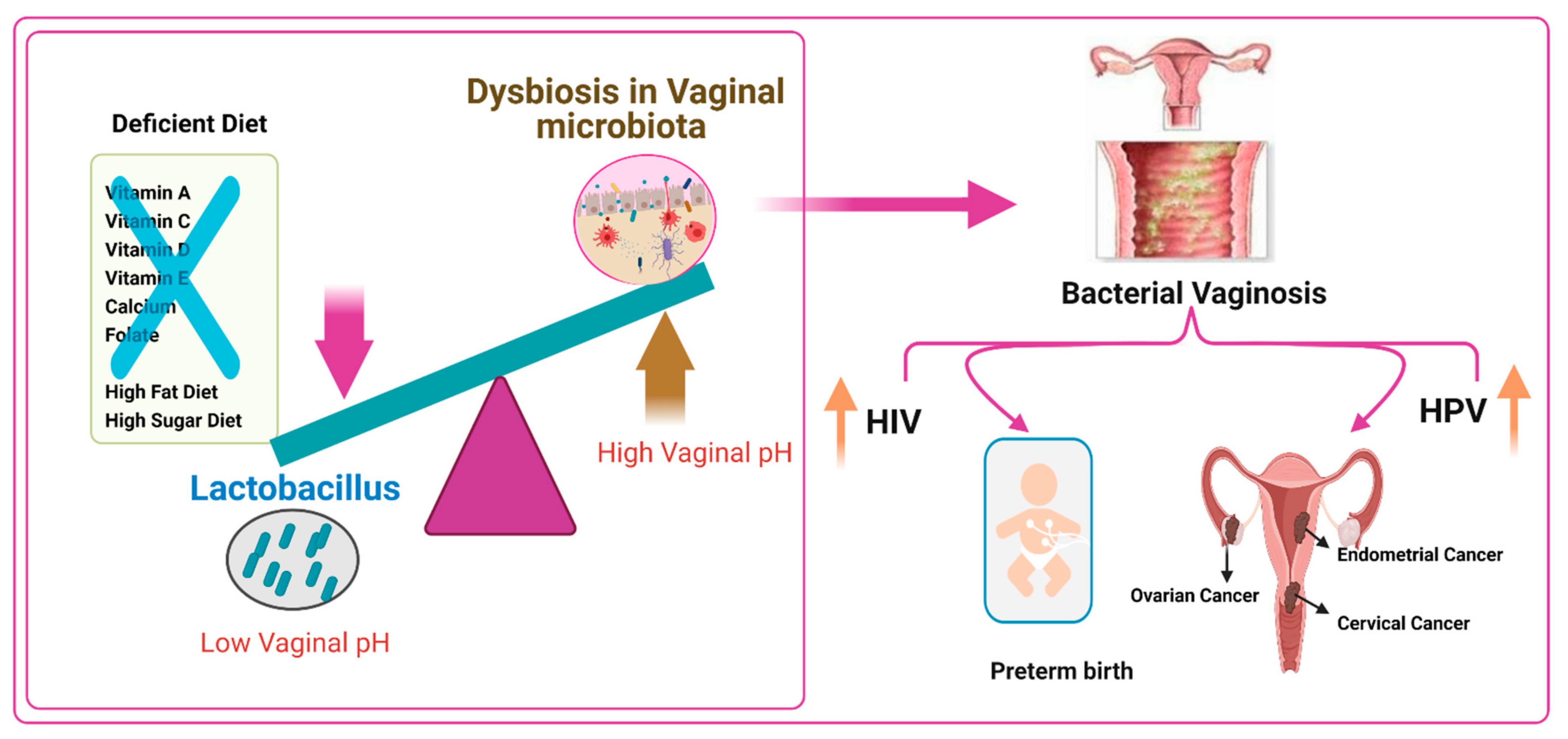
6. Vaginal Microbiome, Nutrients, and Female Reproductive Tract Infections
6.1. Bacterial Vaginosis and Vitamin D Deficiency
6.2. Role of a High-Fat Diet and a High-Sugar-Diet on Altering the Vaginal Microbiome
7. Gynecological Cancers
7.1. Cervical Cancer
7.2. Endometrial Cancer
7.3. Ovarian Cancer
8. Menstrual Disorders
8.1. Menorrhagia
8.1.1. Iron
8.1.2. Vitamin A
8.1.3. Vitamin B Complex
8.1.4. Vitamin C and Bioflavonoids
8.2. Dysmenorrhea
8.2.1. Calcium and Magnesium
8.2.2. Olive Oil
8.2.3. Fennel
8.2.4. Dietary Fiber
8.2.5. Omega-3 and Omega-6 Fatty Acids
8.2.6. Vitamin D
8.2.7. Vitamin E
8.2.8. Qixuehe
8.2.9. Zinc
8.2.10. Vitamin K
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Izetbegovic, S.; Alajbegovic, J.; Mutevelic, A.; Pasagic, A.; Masic, I. Prevention of diseases in gynecology. Int. J. Prev. Med. 2013, 4, 1347–1358. [Google Scholar]
- Kussmann, M.; Fay, L.B. Nutrigenomics and personalized nutrition: Science and concept. Per. Med. 2008, 5, 447–455. [Google Scholar] [CrossRef]
- Herceg, Z. Epigenetics and cancer: Towards an evaluation of the impact of environmental and dietary factors. Mutagenesis 2007, 22, 91–103. [Google Scholar] [CrossRef]
- Junien, C. Impact of diets and nutrients/drugs on early epigenetic programming. J. Inherit. Metab. Dis. 2006, 29, 359–365. [Google Scholar] [CrossRef]
- Dolinoy, C.D.; Weidman, J.R.; Jirtle, R.L. Epigenetic gene regulation: Linking early developmental environment to adult disease. Reprod. Toxicol. 2007, 23, 297–307. [Google Scholar] [CrossRef]
- Paluszczak, J.; Krajka-Kuzniak, V.; Baer-Dubowska, W. The effect of dietary polyphenols on the epigenetic regulation of gene expression in MCF7 breast cancer cells. Toxicol. Lett. 2010, 192, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Andreescu, N.; Puiu, M.; Niculescu, M. Effects of Dietary Nutrients on Epigenetic Changes in Cancer. Methods Mol. Biol. 2018, 1856, 121–139. [Google Scholar]
- Stephen, H.E.; Chandra, A. Declining estimates of infertility in the United States: 1982–2002. Fertil. Steril. 2006, 86, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Braga, D.P.; Halpern, G.; Setti, A.S.; Figueira, R.C.; Iaconelli, A., Jr.; Borges, E., Jr. The impact of food intake and social habits on embryo quality and the likelihood of blastocyst formation. Reprod. Biomed. Online 2015, 31, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Chavarro, J.E.; Rich-Edwards, J.W.; Rosner, B.A.; Willett, W.C. Protein intake and ovulatory infertility. Am. J. Obstet. Gynecol. 2008, 198, 210–e1. [Google Scholar] [CrossRef] [PubMed]
- Gaskins, J.A.; Chavarro, J.E. Diet and fertility: A review. Am. J. Obstet. Gynecol. 2018, 218, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Czeizel, E.A.; Bartfai, Z.; Banhidy, F. Primary prevention of neural-tube defects and some other congenital abnormalities by folic acid and multivitamins: History, missed opportunity and tasks. Ther. Adv. Drug Saf. 2011, 2, 173–188. [Google Scholar] [CrossRef] [PubMed]
- García-Fernández, E.; Rico-Cabanas, L.; Rosgaard, N.; Estruch, R.; Bach-Faig, A. Mediterranean diet and cardiodiabesity: A review. Nutrients 2014, 6, 3474–3500. [Google Scholar] [CrossRef] [PubMed]
- Karayiannis, D.; Kontogianni, M.D.; Mendorou, C.; Mastrominas, M.; Yiannakouris, N. Adherence to the Mediterranean diet and IVF success rate among non-obese women attempting fertility. Hum. Reprod. 2018, 33, 494–502. [Google Scholar] [CrossRef] [PubMed]
- Vujkovic, M.; de Vries, J.H.; Lindemans, J.; Macklon, N.S.; van der Spek, P.J.; Steegers, E.A.; Steegers-Theunissen, R.P. The preconception Mediterranean dietary pattern in couples undergoing in vitro fertilization/intracytoplasmic sperm injection treatment increases the chance of pregnancy. Fertil. Steril. 2010, 94, 2096–2101. [Google Scholar] [CrossRef]
- Twigt, J.M.; Bolhuis, M.E.; Steegers, E.A.; Hammiche, F.; Van Inzen, W.G.; Laven, J.S.; Steegers-Theunissen, R.P. The preconception diet is associated with the chance of ongoing pregnancy in women undergoing IVF/ICSI treatment. Hum. Reprod. 2012, 27, 2526–2531. [Google Scholar] [CrossRef]
- Gaskins, A.J.; Chiu, Y.H.; Williams, P.L.; Keller, M.G.; Toth, T.L.; Hauser, R.; Chavarro, J.E.; EARTH Study Team. Maternal whole grain intake and outcomes of in vitro fertilization. Fertil. Steril. 2016, 105, 1503–1510e4. [Google Scholar] [CrossRef]
- Mumford, S.L.; Browne, R.W.; Kim, K.; Nichols, C.; Wilcox, B.; Silver, R.M.; Connell, M.T.; Holland, T.L.; Kuhr, D.L.; Omosigho, U.R.; et al. Preconception plasma phospholipid fatty acids and fecundability. J. Clin. Endocrinol. Metab. 2018, 103, 4501–4510. [Google Scholar] [CrossRef]
- Saldeen, P.; Saldeen, T. Women and omega-3 Fatty acids. Obstet. Gynecol. Surv. 2004, 59, 722–730. [Google Scholar] [CrossRef]
- Abadia, L.; Gaskins, A.J.; Chiu, Y.H.; Williams, P.L.; Keller, M.; Wright, D.L.; Souter, I.; Hauser, R.; Chavarro, J.E.; Enviroment and Reproductive Health Study Team. Serum 25-hydroxyvitamin D concentrations and treatment outcomes of women undergoing assisted reproduction. Am. J. Clin. Nutr. 2016, 104, 729–735. [Google Scholar]
- Polyzos, N.P.; Anckaert, E.; Guzman, L.; Schiettecatte, J.; Van Landuyt, L.; Camus, M.; Smitz, J.; Tournaye, H. Vitamin D deficiency and pregnancy rates in women undergoing single embryo, blastocyst stage, transfer (SET) for IVF/ICSI. Hum. Reprod. 2014, 29, 2032–2040. [Google Scholar] [CrossRef] [PubMed]
- Chavarro, J.E.; Rich-Edwards, J.W.; Rosner, B.; Willett, W.C. A prospective study of dairy foods intake and anovulatory infertility. Hum. Reprod. 2007, 22, 1340–1347. [Google Scholar] [CrossRef]
- Arefi, S.; Khalili, G.; Iranmanesh, H.; Farifteh, F.; Hosseini, A.; Fatemi, H.M.; Lawrenz, B. Is the ovarian reserve influenced by vitamin D deficiency and the dress code in an infertile Iranian population? J. Ovarian Res. 2018, 11, 1–6. [Google Scholar] [CrossRef]
- Mg, S.; Brown, J.; Clarke, J.; Rj, H. Antioxidants for female subfertility. Cochrane Database Syst. Rev. 2013, 8, CD007807. [Google Scholar]
- Ruder, E.H.; Hartman, T.J.; Reindollar, R.H.; Goldman, M.B. Female dietary antioxidant intake and time to pregnancy among couples treated for unexplained infertility. Fertil. Steril. 2014, 101, 759–766. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef]
- Atassi, F.; Brassart, D.; Grob, P.; Graf, F.; Servin, A.L. Lactobacillus strains isolated from the vaginal microbiota of healthy women inhibit Prevotella bivia and Gardnerella vaginalis in coculture and cell culture. FEMS Immunol. Med. Microbiol. 2006, 48, 424–432. [Google Scholar] [CrossRef]
- Rose, W.A., II; McGowin, C.L.; Spagnuolo, R.A.; Eaves-Pyles, T.D.; Popov, V.L.; Pyles, R.B. Commensal bacteria modulate innate immune responses of vaginal epithelial cell multilayer cultures. PLoS ONE 2012, 7, e32728. [Google Scholar] [CrossRef] [PubMed]
- Younis, N.S.; Mahasneh, A. Probiotics and the envisaged role in treating human infertility. Middle E. Fertil. Soc. J. 2020, 25, 1–9. [Google Scholar] [CrossRef]
- Bhandari, P.; Prabha, V. Evaluation of profertility effect of probiotic Lactobacillus plantarum 2621 in a murine model. Indian J. Med. Res. 2015, 142, 79–84. [Google Scholar]
- Rocha, A.L.; Oliveira, F.R.; Azevedo, R.C.; Silva, V.A.; Peres, T.M.; Candido, A.L.; Gomes, K.B.; Reis, F.M. Recent advances in the understanding and management of polycystic ovary syndrome. F1000Research 2019, 8, 565. [Google Scholar] [CrossRef]
- Esfandyari, S.; Chugh, R.M.; Park, H.S.; Hobeika, E.; Ulin, M.; Al-Hendy, A. Mesenchymal Stem Cells as a Bio Organ. for Treatment of Female Infertility. Cells 2020, 9, 2253. [Google Scholar] [CrossRef] [PubMed]
- Esfandyari, S. miRNA-92a suppresses androgen-producing steroidogenic genes expression in h295r, a human pcos in-vitro theca-like cell model. Fertil. Steril. 2020, 114, e349–e350. [Google Scholar] [CrossRef]
- Azziz, R.; Woods, K.S.; Reyna, R.; Key, T.J.; Knochenhauer, E.S.; Yildiz, B.O. The prevalence and features of the polycystic ovary syndrome in an unselected population. J. Clin. Endocrinol. Metab. 2004, 89, 2745–2749. [Google Scholar] [CrossRef]
- Faghfoori, Z.; Fazelian, S.; Shadnoush, M.; Goodarzi, R. Nutritional management in women with polycystic ovary syndrome: A review study. Diabetes Metab. Syndr. Clin. Res. Rev. 2017, 11, S429–S432. [Google Scholar] [CrossRef]
- Douglas, C.C.; Gower, B.A.; Darnell, B.E.; Ovalle, F.; Oster, R.A.; Azziz, R. Role of diet in the treatment of polycystic ovary syndrome. Fertil. Steril. 2006, 85, 679–688. [Google Scholar] [CrossRef]
- Goss, A.M.; Gower, B.; Soleymani, T.; Stewart, M.; Pendergrass, M.; Lockhart, M.; Krantz, O.; Dowla, S.; Bush, N.; Barry, V.G.; et al. Effects of weight loss during a very low carbohydrate diet on specific adipose tissue depots and insulin sensitivity in older adults with obesity: A randomized clinical trial. Nutr. Metab. 2020, 17, 1–12. [Google Scholar] [CrossRef]
- Kasim-Karakas, S.E.; Almario, R.U.; Gregory, L.; Wong, R.; Todd, H.; Lasley, B.L. Metabolic and endocrine effects of a polyunsaturated fatty acid-rich diet in polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2004, 89, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Teegarden, D.; Donkin, S.S. Vitamin D: Emerging new roles in insulin sensitivity. Nutr. Res. Rev. 2009, 22, 82–92. [Google Scholar] [CrossRef]
- He, C.; Lin, Z.; Robb, S.W.; Ezeamama, A.E. Serum vitamin D levels and polycystic ovary syndrome: A systematic review and meta-analysis. Nutrients 2015, 7, 4555–4577. [Google Scholar] [CrossRef]
- Ott, J.; Wattar, L.; Kurz, C.; Seemann, R.; Huber, J.C.; Mayerhofer, K.; Vytiska-Binstorfer, E. Parameters for calcium metabolism in women with polycystic ovary syndrome who undergo clomiphene citrate stimulation: A prospective cohort study. Eur. J. Endocrinol. 2012, 166, 897. [Google Scholar] [CrossRef]
- Wojtusik, J.; Johnson, P.A. Vitamin D regulates anti-Mullerian hormone expression in granulosa cells of the hen. Biol. Reprod. 2012, 86, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Iliodromiti, S.; Kelsey, T.W.; Anderson, R.A.; Nelson, S.M. Can anti-Müllerian hormone predict the diagnosis of polycystic ovary syndrome? A systematic review and meta-analysis of extracted data. J. Clin. Endocrinol. Metab. 2013, 98, 3332–3340. [Google Scholar] [CrossRef]
- Elhusseini, H.; Lizneva, D.; Gavrilova-Jordan, L.; Eziba, N.; Abdelaziz, M.; Brakta, S.; Halder, S.; Al-Hebdy, A. Vitamin d and female reproduction. In A Critical Evaluation of Vitamin D: Basic Overview; Gower, S., Ed.; IntechOpen: London, UK, 2017; p. 297. [Google Scholar]
- Irani, M.; Minkoff, H.; Seifer, D.B.; Merhi, Z. Vitamin D increases serum levels of the soluble receptor for advanced glycation end products in women with PCOS. J. Clin. Endocrinol. Metab. 2014, 99, E886–E890. [Google Scholar] [CrossRef]
- Legro, R.S.; Arslanian, S.A.; Ehrmann, D.A.; Hoeger, K.M.; Murad, M.H.; Pasquali, R.; Welt, C.K. Diagnosis and treatment of polycystic ovary syndrome: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2013, 98, 4565–4592. [Google Scholar] [CrossRef]
- Rodriguez Paris, V.; Bertoldo, M.J. The mechanism of androgen actions in PCOS etiology. Med. Sci. 2019, 7, 89. [Google Scholar] [CrossRef]
- Zhang, X.; Zheng, Y.; Guo, Y.; Lai, Z. The effect of low carbohydrate diet on polycystic ovary syndrome: A meta-analysis of randomized controlled trials. Int. J. Endocrinol 2019. [Google Scholar] [CrossRef]
- Ebrahimi, R.; Bahiraee, A.; Niazpour, F.; Emamgholipour, S.; Meshkani, R. The role of microRNAs in the regulation of insulin signaling pathway with respect to metabolic and mitogenic cascades: A review. J. Cell. Biochem. 2019, 120, 19290–19309. [Google Scholar] [CrossRef] [PubMed]
- Emamgholipour, S.; Ebrahimi, R.; Bahiraee, A.; Niazpour, F.; Meshkani, R. Acetylation and insulin resistance: A focus on metabolic and mitogenic cascades of insulin signaling. Crit. Rev. Clin. Lab. Sci. 2020, 57, 196–214. [Google Scholar] [CrossRef]
- Moran, L.J.; Noakes, M.; Clifton, P.M.; Tomlinson, L.; Norman, R.J. Dietary composition in restoring reproductive and metabolic physiology in overweight women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2003, 88, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Jamilian, M.; Asemi, Z. The effects of soy isoflavones on metabolic status of patients with polycystic ovary syndrome. TJ Clin. Endocrinol. Metab. 2016, 101, 3386–3394. [Google Scholar] [CrossRef]
- Khani, B.; Mehrabian, F.; Khalesi, E.; Eshraghi, A. Effect of soy phytoestrogen on metabolic and hormonal disturbance of women with polycystic ovary syndrome. J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2011, 16, 297. [Google Scholar]
- Tehrani, H.G.; Allahdadian, M.; Zarre, F.; Ranjbar, H.; Allahdadian, F. Effect of green tea on metabolic and hormonal aspect of polycystic ovarian syndrome in overweight and obese women suffering from polycystic ovarian syndrome: A clinical trial. J. Educ. Health Promot. 2017, 6, 36. [Google Scholar] [CrossRef] [PubMed]
- Armanini, D.; Mattarello, M.J.; Fiore, C.; Bonanni, G.; Scaroni, C.; Sartorato, P.; Palermo, M. Licorice reduces serum testosterone in healthy women. Steroids 2004, 69, 763–766. [Google Scholar] [CrossRef]
- MCalderon-Montano, J.; Burgos-Morón, E.; Pérez-Guerrero, C.; López-Lázaro, M. A review on the dietary flavonoid kaempferol. Mini Rev. Med. Chem. 2011, 11, 298–344. [Google Scholar] [CrossRef]
- Tabrizi, F.P.; Hajizadeh-Sharafabad, F.; Vaezi, M.; Jafari-Vayghan, H.; Alizadeh, M.; Maleki, V. Quercetin and polycystic ovary syndrome, current evidence and future directions: A systematic review. J. Ovarian Res. 2020, 13, 11. [Google Scholar] [CrossRef]
- Shah, K.N.; Patel, S.S. Phosphatidylinositide 3-kinase inhibition: A new potential target for the treatment of polycystic ovarian syndrome. Pharm. Biol. 2016, 54, 975–983. [Google Scholar] [CrossRef]
- Wang, Z.; Zhai, D.; Zhang, D.; Bai, L.; Yao, R.; Yu, J.; Cheng, W.; Yu, C. Quercetin decreases insulin resistance in a polycystic ovary syndrome rat model by improving inflammatory microenvironment. Reprod. Sci. 2017, 24, 682–690. [Google Scholar] [CrossRef]
- Rezvan, N.; Moini, A.; Gorgani-Firuzjaee, S.; Hosseinzadeh-Attar, M.J. Oral quercetin supplementation enhances adiponectin receptor transcript expression in polycystic ovary syndrome patients: A randomized placebo-controlled double-blind clinical trial. Cell J. 2018, 19, 627. [Google Scholar]
- Khorshidi, M.; Moini, A.; Alipoor, E.; Rezvan, N.; Gorgani-Firuzjaee, S.; Yaseri, M.; Hosseinzadeh-Attar, M.J. The effects of quercetin supplementation on metabolic and hormonal parameters as well as plasma concentration and gene expression of resistin in overweight or obese women with polycystic ovary syndrome. Phytother. Res. 2018, 32, 2282–2289. [Google Scholar] [CrossRef]
- Oh, J.S.; Kim, H.; Vijayakumar, A.; Kwon, O.; Choi, Y.J.; Huh, K.B.; Chang, N. Association between dietary flavanones intake and lipid profiles according to the presence of metabolic syndrome in Korean women with type 2 diabetes mellitus. Nutr. Res. Pract. 2016, 10, 67–73. [Google Scholar] [CrossRef]
- Romualdi, D.; Costantini, B.; Campagna, G.; Lanzone, A.; Guido, M. Is there a role for soy isoflavones in the therapeutic approach to polycystic ovary syndrome? Results from a pilot study. Fertil. Steril. 2008, 90, 1826–1833. [Google Scholar] [CrossRef]
- Banaszewska, B.; Wrotyńska-Barczyńska, J.; Spaczynski, R.Z.; Pawelczyk, L.; Duleba, A.J. Effects of resveratrol on polycystic ovary syndrome: A double-blind, randomized, placebo-controlled trial. J. Clin. Endocrinol. Metab. 2016, 101, 4322–4328. [Google Scholar] [CrossRef]
- Günalan, E.; Yaba, A.; Yılmaz, B. The effect of nutrient supplementation in the management of polycystic ovary syndrome-associated metabolic dysfunctions: A critical review. J. Turk. Ger. Gynecol. Assoc. 2018, 19, 220. [Google Scholar] [CrossRef]
- Anderson, R.A. Chromium in the prevention and control of diabetes. Diab. Metab. 2000, 26, 22–28. [Google Scholar]
- Chakraborty, P.; Ghosh, S.; Goswami, S.K.; Kabir, S.N.; Chakravarty, B.; Jana, K. Altered trace mineral milieu might play an aetiological role in the pathogenesis of polycystic ovary syndrome. Biol. Trace Elem. Res. 2013, 152, 9–15. [Google Scholar] [CrossRef]
- Lucidi, R.S.; Thyer, A.C.; Easton, C.A.; Holden, A.E.; Schenken, R.S.; Brzyski, R.G. Effect of chromium supplementation on insulin resistance and ovarian and menstrual cyclicity in women with polycystic ovary syndrome. Fertil. Steril. 2005, 84, 1755–1757. [Google Scholar] [CrossRef] [PubMed]
- Jamilian, M.; Asemi, Z. Chromium supplementation and the effects on metabolic status in women with polycystic ovary syndrome: A randomized, double-blind, placebo-controlled trial. Ann. Nutr. Metab. 2015, 67, 42–48. [Google Scholar] [CrossRef]
- Jamilian, M.; Bahmani, F.; Siavashani, M.A.; Mazloomi, M.; Asemi, Z.; Esmaillzadeh, A. The effects of chromium supplementation on endocrine profiles, biomarkers of inflammation, and oxidative stress in women with polycystic ovary syndrome: A randomized, double-blind, placebo-controlled trial. Biol. Trace Elem. Res. 2016, 172, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Ullah, G.; Jung, P.; Machaca, K. Modeling Ca2+ signaling differentiation during oocyte maturation. Cell Calcium 2007, 42, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Mazloomi, S.; Sharifi, F.; Hajihosseini, R.; Kalantari, S.; Mazloomzadeh, S. Association between hypoadiponectinemia and low serum concentrations of calcium and vitamin D in women with polycystic ovary syndrome. ISRN Endocrinol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Dehghani Firouzabadi, R.; Aflatoonian, A.; Modarresi, S.; Sekhavat, L.; MohammadTaheri, S. Therapeutic effects of calcium & vitamin D supplementation in women with PCOS. Complement. Ther. Clin. Pract. 2012, 18, 85–88. [Google Scholar]
- Mirone, M.; Giannetta, E.; Isidori, A. Selenium and reproductive function. A systematic review. J. Endocrinol. Invest. 2013, 36, 28–36. [Google Scholar]
- Coskun, A.; Arikan, T.; Kilinc, M.; Arikan, D.C.; Ekerbiçer, H.Ç. Plasma selenium levels in Turkish women with polycystic ovary syndrome. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 168, 183–186. [Google Scholar] [CrossRef]
- Modarres, S.Z.; Heidar, Z.; Foroozanfard, F.; Rahmati, Z.; Aghadavod, E.; Asemi, Z. The effects of selenium supplementation on gene expression related to insulin and lipid in infertile polycystic ovary syndrome women candidate for in vitro fertilization: A randomized, double-blind, placebo-controlled trial. Biol. Trace Elem. Res. 2018, 183, 218–225. [Google Scholar] [CrossRef]
- Jamilian, M.; Razavi, M.; Fakhrie Kashan, Z.; Ghandi, Y.; Bagherian, T.; Asemi, Z. Metabolic response to selenium supplementation in women with polycystic ovary syndrome: A randomized, double-blind, placebo-controlled trial. Clin. Endocrinol. 2015, 82, 885–891. [Google Scholar] [CrossRef]
- Tubek, S. Zinc supplementation or regulation of its homeostasis: Advantages and threats. Biol. Trace Elem. Res. 2007, 119, 1–9. [Google Scholar] [CrossRef]
- Beletate, V.; el Dib, R.; Atallah, Á.N. Zinc supplementation for the prevention of type 2 diabetes mellitus. Cochrane Datab. Syst. Rev. 2007, 1, CD005525. [Google Scholar] [CrossRef]
- Guler, I.; Himmetoglu, O.; Turp, A.; Erdem, A.; Erdem, M.; Onan, M.A.; Taskiran, C.; Taslipinar, M.Y.; Guner, H. Zinc and homocysteine levels in polycystic ovarian syndrome patients with insulin resistance. Biol. Trace Elem. Res. 2014, 158, 297–304. [Google Scholar] [CrossRef]
- Saris, N.E.; Mervaala, E.; Karppanen, H.; Khawaja, J.A.; Lewenstam, A. Magnesium: An update on physiological, clinical and analytical aspects. Clin. Chim. Acta 2000, 294, 1–26. [Google Scholar] [CrossRef]
- Rumawas, M.E.; McKeown, N.M.; Rogers, G.; Meigs, J.B.; Wilson, P.W.; Jacques, P.F. Magnesium intake is related to improved insulin homeostasis in the framingham offspring cohort. J. Am. Coll. Nutr. 2006, 25, 486–492. [Google Scholar] [CrossRef]
- Ryan, L.G.; Syrop, C.H.; van Voorhis, B.J. Role, epidemiology, and natural history of benign uterine mass lesions. Clin. Obstet. Gynecol. 2005, 48, 312–324. [Google Scholar] [CrossRef]
- Ulin, M.; Ali, M.; Chaudhry, Z.T.; Al-Hendy, A.; Yang, Q. Uterine fibroids in menopause and perimenopause. Menopause 2020, 27, 238–242. [Google Scholar] [CrossRef]
- Wu, J.M.; Wechter, M.E.; Geller, E.J.; Nguyen, T.V.; Visco, A.G. Hysterectomy rates in the United States, 2003. Obstet. Gynecol. 2007, 110, 1091–1095. [Google Scholar] [CrossRef]
- Cardozo, E.R.; Clark, A.D.; Banks, N.K.; Henne, M.B.; Stegmann, B.J.; Segars, J.H. The estimated annual cost of uterine leiomyomata in the United States. Am. J. Obstet. Gynecol. 2012, 206, 211e1. [Google Scholar] [CrossRef] [PubMed]
- Ciebiera, M.; Ali, M.; Prince, L.; Jackson-Bey, T.; Atabiekov, I.; Zgliczyński, S.; Al-Hendy, A. The Evolving Role of Natural Compounds in the Medical Treatment of Uterine Fibroids. J. Clin. Med. 2020, 9, 1479. [Google Scholar] [CrossRef]
- Ali, M.; Esfandyari, S.; Al-Hendy, A. Evolving role of microRNAs in uterine fibroid pathogenesis: Filling the gap! Fertil. Steril. 2020, 113, 1167–1168. [Google Scholar] [CrossRef]
- Laughlin, K.S.; Schroeder, J.C.; Baird, D.D. New directions in the epidemiology of uterine fibroids. Semin. Reprod. Med. 2010, 28, 204–217. [Google Scholar] [CrossRef]
- Tinelli, A.; Vinciguerra, M.; Malvasi, A.; Andjić, M.; Babović, I.; Sparić, R. Uterine Fibroids and Diet. Int. J. Environ. Res. Publ. Health 2021, 18, 1066. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Pischon, T.; Hankinson, S.E.; Rifai, N.; Joshipura, K.; Willett, W.C.; Rimm, E.B. Dietary intake of trans fatty acids and systemic inflammation in women. Am. J. Clin. Nutr. 2004, 79, 606–612. [Google Scholar] [CrossRef]
- Wu, H.A.; Pike, M.C.; Stram, D.O. Meta-analysis: Dietary fat intake, serum estrogen levels, and the risk of breast cancer. J. Natl. Cancer Inst. 1999, 91, 529–534. [Google Scholar] [CrossRef] [PubMed]
- Baird, D.D.; Patchel, S.A.; Saldana, T.M.; Umbach, D.M.; Cooper, T.; Wegienka, G.; Harmon, Q.E. Uterine fibroid incidence and growth in an ultrasound-based, prospective study of young African Americans. Am. J. Obstet. Gynecol. 2020, 223, 402e1. [Google Scholar] [CrossRef] [PubMed]
- Kristal, R.A.; Shattuck, A.L.; Patterson, R.E. Differences in fat-related dietary patterns between black, Hispanic and White women: Results from the women’s health trial feasibility study in minority populations. Pub. Health Nutr. 1999, 2, 253–262. [Google Scholar] [CrossRef]
- Rosenberg, L.; Adams-Campbell, L.; Palmer, J.R. The black women’s health study: A follow-up study for causes and preventions of illness. J. Am. Med. Wom. Assoc. 1995, 50, 56–58. [Google Scholar]
- Wise, L.A.; Radin, R.G.; Kumanyika, S.K.; Ruiz-Narvaez, E.A.; Palmer, J.R.; Rosenberg, L. Prospective study of dietary fat and risk of uterine leiomyomata. Am. J. Clin. Nutr. 2014, 99, 1105–1116. [Google Scholar] [CrossRef]
- Brasky, T.M.; Bethea, T.N.; Wesselink, A.K.; Wegienka, G.R.; Baird, D.D.; Wise, L.A. Dietary fat intake and risk of uterine leiomyomata: A prospective ultrasound study. Am. J. Epidemiol. 2020, 189, 1538–1546. [Google Scholar] [CrossRef]
- Harris, H.R.; Eliassen, A.H.; Doody, D.R.; Terry, K.L.; Missmer, S.A. Dietary fat intake, erythrocyte fatty acids, and risk of uterine fibroids. Fertil. Steril. 2020, 114, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Wegienka, G. Are uterine leiomyoma a consequence of a chronically inflammatory immune system? Med. Hypotheses 2012, 79, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Chiaffarino, F.; Parazzini, F.; La Vecchia, C.; Chatenoud, L.; Di Cintio, E.; Marsico, S. Diet. and uterine myomas. Obstet. Gynecol. 1999, 94, 395–398. [Google Scholar] [PubMed]
- Nagata, C.; Nakamura, K.; Oba, S.; Hayashi, M.; Takeda, N.; Yasuda, K. Association of intakes of fat, dietary fibre, soya isoflavones and alcohol with uterine fibroids in Japanese women. Br. J. Nutr. 2009, 101, 1427–1431. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Castellucci, C.; Fiorini, R.; Greco, S.; Gagliardi, R.; Zannotti, A.; Giannubilo, S.R.; Ciavattini, A.; Frega, N.G.; Pacetti, D.; et al. Omega-3 fatty acids modulate the lipid profile, membrane architecture, and gene expression of leiomyoma cells. J. Cell. Physiol. 2018, 233, 7143–7156. [Google Scholar] [CrossRef] [PubMed]
- Kant, K.A.; Graubard, B.I. Ethnicity is an independent correlate of biomarkers of micronutrient intake and status in American adults. J. Nutr. 2007, 137, 2456–2463. [Google Scholar] [CrossRef] [PubMed]
- Timbo, B.B.; Ross, M.P.; McCarthy, P.V.; Lin, C.T. Dietary supplements in a national survey: Prevalence of use and reports of adverse events. J. Am. Diet. Assoc. 2006, 106, 1966–1974. [Google Scholar] [CrossRef] [PubMed]
- Wise, L.A.; Radin, R.G.; Palmer, J.R.; Kumanyika, S.K.; Boggs, D.A.; Rosenberg, L. Intake of fruit, vegetables, and carotenoids in relation to risk of uterine leiomyomata. Am. J. Clin. Nutr. 2011, 94, 1620–1631. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Zeng, Q.; Dong, S.; Qin, L.; Li, G.; Wang, P. Associations between uterine fibroids and lifestyles including diet, physical activity and stress: A case-control study in China. Asia Pac. J. Clin. Nutr. 2013, 22, 109–117. [Google Scholar] [PubMed]
- Sahin, K.; Ozercan, R.; Onderci, M.; Sahin, N.; Gursu, M.F.; Khachik, F.; Sarkar, F.H.; Munkarah, A.; Ali-Fehmi, R.; Kmak, D.; et al. Lycopene supplementation prevents the development of spontaneous smooth muscle tumors of the oviduct in Japanese quail. Nutr. Cancer 2004, 50, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Setchell, K.D.; Cassidy, A. Dietary isoflavones: Biological effects and relevance to human health. J. Nutr. 1999, 129, 758S–767S. [Google Scholar] [CrossRef] [PubMed]
- Greco, S.; Islam, M.S.; Zannotti, A.; Carpini, G.D.; Giannubilo, S.R.; Ciavattini, A.; Petraglia, F.; Ciarmela, P. Quercetin and indole-3-carbinol inhibit extracellular matrix expression in human primary uterine leiomyoma cells. Reprod. Biomed. Online 2020, 40, 593–602. [Google Scholar] [CrossRef]
- Lila, M.A.; Burton-Freeman, B.; Grace, M.; Kalt, W. Unraveling Anthocyanin Bioavailability for Human Health. Annu. Rev. Food Sci. Technol. 2016, 7, 375–393. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Feng, R.; Lu, Y.; Bowman, L.; Ding, M. Inhibitory effect on activator protein-1, nuclear factor-kappaB, and cell transformation by extracts of strawberries (Fragaria x ananassa Duch.). J. Agric. Food Chem. 2005, 53, 4187–4193. [Google Scholar] [CrossRef]
- Giampieri, F.; Islam, M.S.; Greco, S.; Gasparrini, M.; Forbes Hernandez, T.Y.; Delli Carpini, G.; Giannubilo, S.R.; Ciavattini, A.; Mezzetti, B.; Capocasa, F.; et al. Romina: A powerful strawberry with in vitro efficacy against uterine leiomyoma cells. J. Cell. Physiol. 2019, 234, 7622–7633. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Giampieri, F.; Janjusevic, M.; Gasparrini, M.; Forbes-Hernandez, T.Y.; Mazzoni, L.; Greco, S.; Giannubilo, S.R.; Ciavattini, A.; Mezzetti, B.; et al. Anthocyanin rich strawberry extract induces apoptosis and ROS while decreases glycolysis and fibrosis in human uterine leiomyoma cells. Oncotarget 2017, 8, 23575–23587. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.C.; Ramachandran, S.; Baek, S.H.; Kwon, S.H.; Kwon, K.Y.; Cha, S.D.; Bae, I.; Cho, C.H. Induction of growth inhibition and apoptosis in human uterine leiomyoma cells by isoliquiritigenin. Reprod. Sci. 2008, 15, 552–558. [Google Scholar] [CrossRef]
- Moore, A.B.; Castro, L.; Yu, L.; Zheng, X.; Di, X.; Sifre, M.I.; Kissling, G.E.; Newbold, R.R.; Bortner, C.D.; Dixon, D. Stimulatory and inhibitory effects of genistein on human uterine leiomyoma cell proliferation are influenced by the concentration. Hum. Reprod. 2007, 22, 2623–2631. [Google Scholar] [CrossRef] [PubMed]
- Di, X.; Yu, L.; Moore, A.B.; Castro, L.; Zheng, X.; Hermon, T.; Dixon, D. A low concentration of genistein induces estrogen receptor-alpha and insulin-like growth factor-I receptor interactions and proliferation in uterine leiomyoma cells. Hum. Reprod. 2008, 23, 1873–1883. [Google Scholar] [CrossRef]
- Beydoun, M.A.; Gary, T.L.; Caballero, B.H.; Lawrence, R.S.; Cheskin, L.J.; Wang, Y. Ethnic differences in dairy and related nutrient consumption among US adults and their association with obesity, central obesity, and the metabolic syndrome. Am. J. Clin. Nutr. 2008, 87, 1914–1925. [Google Scholar] [CrossRef]
- Lu, W.; Chen, H.; Niu, Y.; Wu, H.; Xia, D.; Wu, Y. Dairy products intake and cancer mortality risk: A meta-analysis of 11 population-based cohort studies. Nutr. J. 2016, 15, 91. [Google Scholar] [CrossRef]
- Shen, Y.; Xu, Q.; Xu, J.; Ren, M.L.; Cai, Y.L. Environmental exposure and risk of uterine leiomyoma: An epidemiologic survey. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 3249–3256. [Google Scholar]
- Wise, L.A.; Radin, R.G.; Palmer, J.R.; Kumanyika, S.K.; Rosenberg, L. A prospective study of dairy intake and risk of uterine leiomyomata. Am. J. Epidemiol. 2010, 171, 221–232. [Google Scholar] [CrossRef]
- Wise, L.A.; Ruiz-Narváez, E.A.; Haddad, S.A.; Rosenberg, L.; Palmer, J.R. Polymorphisms in vitamin D-related genes and risk of uterine leiomyomata. Fertil. Steril. 2014, 102, 503–510.e1. [Google Scholar] [CrossRef]
- Ciebiera, M.; Ali, M.; Prince, L.; Zgliczyński, S.; Jakiel, G.; Al-Hendy, A. The Significance of Measuring Vitamin D Serum Levels in Women with Uterine Fibroids. Reprod. Sci. 2020. [Google Scholar] [CrossRef]
- Al-Hendy, A.; Diamond, M.P.; El-Sohemy, A.; Halder, S.K. 1,25-dihydroxyvitamin D3 regulates expression of sex steroid receptors in human uterine fibroid cells. J. Clin. Endocrinol. Metab. 2015, 100, E572–E582. [Google Scholar] [CrossRef]
- Ciebiera, M.; Ali, M.; Zgliczyńska, M.; Skrzypczak, M.; Al-Hendy, A. Vitamins and uterine fibroids: Current data on pathophysiology and possible clinical relevance. Int. J. Mol. Sci. 2020, 21, 5528. [Google Scholar] [CrossRef]
- Ali, M.; Al-Hendy, A.; Yang, Q. Vitamin D, a promising natural compound with anti-uterine fibroid characteristics. Fertil. Steril. 2019, 111, 268–269. [Google Scholar] [CrossRef]
- Elkafas, H.; Ali, M.; Elmorsy, E.; Kamel, R.; Thompson, W.E.; Badary, O.; Al-Hendy, A.; Yang, Q. Vitamin d3 ameliorates dna damage caused by developmental exposure to endocrine disruptors in the uterine myometrial stem cells of Eker rats. Cells 2020, 9, 1459. [Google Scholar] [CrossRef]
- Ali, M.; Shahin, S.M.; Sabri, N.A.; Al-Hendy, A.; Yang, Q. Hypovitaminosis D exacerbates the DNA damage load in human uterine fibroids, which is ameliorated by vitamin D3 treatment. Acta Pharmacol. Sin. 2019, 40, 957–970. [Google Scholar] [CrossRef]
- ElHusseini, H.; Elkafas, H.; Abdelaziz, M.; Halder, S.; Atabiekov, I.; Eziba, N.; Ismail, N.; El Andaloussi, A.; Al-Hendy, A. Diet.-induced vitamin D deficiency triggers inflammation and DNA damage profile in murine myometrium. Int. J. Women’s Health 2018, 10, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Sheng, B.; Song, Y.; Liu, Y.; Jiang, C.; Zhu, X. Association between vitamin D and uterine fibroids: A study protocol of an open-label, randomised controlled trial. BMJ Open 2020, 10, e038709. [Google Scholar] [CrossRef] [PubMed]
- Prins, G.S.; Hu, W.Y.; Shi, G.B.; Hu, D.P.; Majumdar, S.; Li, G.; Huang, K.; Nelles, J.L.; Ho, S.M.; Walker, C.L.; et al. Bisphenol A promotes human prostate stem-progenitor cell self-renewal and increases in vivo carcinogenesis in human prostate epithelium. Endocrinology 2014, 155, 805–817. [Google Scholar] [CrossRef] [PubMed]
- Bariani, M.V.; Rangaswamy, R.; Siblini, H.; Yang, Q.; Al-Hendy, A.; Zota, A.R. The role of endocrine-disrupting chemicals in uterine fibroid pathogenesis. Curr. Opin. Endocrinol. Diabetes Obes. 2020, 27, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, E.B.; Louis, G.M.; Parsons, P.J.; Steuerwald, A.J.; Palmer, C.D.; Chen, Z.; Sun, L.; Hammoud, A.O.; Dorais, J.; Peterson, C.M. Increased urinary cobalt and whole blood concentrations of cadmium and lead in women with uterine leiomyomata: Findings from the ENDO Study. Reprod. Toxicol. 2014, 49, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Jackson, L.W.; Zullo, M.D.; Goldberg, J.M. The association between heavy metals, endometriosis and uterine myomas among premenopausal women: National Health and Nutrition Examination Survey 1999–2002. Hum. Reprod. 2008, 23, 679–687. [Google Scholar] [CrossRef] [PubMed]
- Maybin, J.A.; Critchley, H.O. Menstrual physiology: Implications for endometrial pathology and beyond. Hum. Reprod. Update 2015, 21, 748–761. [Google Scholar] [CrossRef]
- Karamian, A.; Paktinat, S.; Esfandyari, S.; Nazarian, H.; Ali Ziai, S.; Zarnani, A.H.; Salehpour, S.; Hosseinirad, H.; Karamian, A.; Novin, M.G. Pyrvinium pamoate induces in-vitro suppression of IL-6 and IL-8 produced by human endometriotic stromal cells. Hum. Exp. Toxicol. 2020, 4, 649–660. [Google Scholar]
- Nnoaham, K.E.; Hummelshoj, L.; Webster, P.; d’Hooghe, T.; de Cicco Nardone, F.; de Cicco Nardone, C.; Jenkinson, C.; Kennedy, S.H.; Zondervan, K.T.; Study, W.E. Impact of endometriosis on quality of life and work productivity: A multicenter study across ten countries. Fertil. Steril. 2011, 96, 366–373e8. [Google Scholar] [CrossRef]
- Taylor, R.N.; Lebovic, D.I.; Mueller, M.D. Angiogenic factors in endometriosis. Ann. N. Y. Acad. Sci. USA 2002, 955, 89–100. [Google Scholar] [CrossRef]
- Husby, G.K.; Haugen, R.S.; Moen, M.H. Diagnostic delay in women with pain and endometriosis. Acta Obstet. Gynecol. Scand. 2003, 82, 649–653. [Google Scholar] [CrossRef] [PubMed]
- Sourial, S.; Tempest, N.; Hapangama, D.K. Theories on the pathogenesis of endometriosis. Int. J. Reprod. Med. 2014, 2014, 179515. [Google Scholar] [CrossRef]
- Youseflu, S.; Jahanian Sadatmahalleh, S.; Mottaghi, A.; Kazemnejad, A. The association of food consumption and nutrient intake with endometriosis risk in Iranian women: A case-control study. Int. J. Reprod. BioMed. 2019, 17, 661. [Google Scholar]
- Aris, A.; Paris, K. Hypothetical link between endometriosis and xenobiotics-associated genetically modified food. Gynecol. Obstet. Fertil. 2010, 38, 747–753. [Google Scholar]
- Khanaki, K.; Nouri, M.; Ardekani, A.M.; Ghassemzadeh, A.; Shahnazi, V.; Sadeghi, M.R.; Darabi, M.; Mehdizadeh, A.; Dolatkhah, H.; Saremi, A.; et al. Evaluation of the relationship between endometriosis and omega-3 and omega-6 polyunsaturated fatty acids. Iran. Biomed. J. 2012, 16, 38. [Google Scholar]
- Halpern, G.; Schor, E.; Kopelman, A. Nutritional aspects related to endometriosis. Rev. Assoc. Méd. Brasil. 2015, 61, 519–523. [Google Scholar] [CrossRef]
- Lasco, A.; Catalano, A.; Benvenga, S. Improvement of primary dysmenorrhea caused by a single oral dose of vitamin D: Results of a randomized, double-blind, placebo-controlled study. Arch. Intern. Med. 2012, 172, 366–367. [Google Scholar] [CrossRef] [PubMed]
- Almassinokiani, F.; Khodaverdi, S.; Solaymani-Dodaran, M.; Akbari, P.; Pazouki, A. Effects of vitamin D on endometriosis-related pain: A double-blind clinical trial. Med. Sci. Monit. Int. Med. J. Exper. Clin. Res. 2016, 22, 4960. [Google Scholar] [CrossRef] [PubMed]
- Ngô, C.; Chéreau, C.; Nicco, C.; Weill, B.; Chapron, C.; Batteux, F. Reactive oxygen species controls endometriosis progression. Am. J. Pathol. 2009, 175, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Porpora, M.G.; Brunelli, R.; Costa, G.; Imperiale, L.; Krasnowska, E.K.; Lundeberg, T.; Nofroni, I.; Piccioni, M.G.; Pittaluga, E.; Ticino, A.; et al. A promise in the treatment of endometriosis: An observational cohort study on ovarian endometrioma reduction by N-acetylcysteine. Evid. Based Complement. Altern. Med. 2013, 2013, 240702. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Lim, W.; Bazer, F.W.; Whang, K.Y.; Song, G. Quercetin inhibits proliferation of endometriosis regulating cyclin D1 and its target microRNAs in vitro and in vivo. J. Nutr. Biochem. 2019, 63, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Zhuang, M.F.; Yang, Y.; Xie, S.W.; Cui, J.G.; Cao, L.; Zhang, T.T.; Zhu, Y. Preliminary study of quercetin affecting the hypothalamic-pituitary-gonadal axis on rat endometriosis model. Evid. Based Complement. Altern. Med. 2014, 2014, 781684. [Google Scholar] [CrossRef] [PubMed]
- Ergenoğlu, A.M.; Yeniel, A.Ö.; Erbaş, O.; Aktuğ, H.; Yildirim, N.; Ulukuş, M.; Taskiran, D. Regression of endometrial implants by resveratrol in an experimentally induced endometriosis model in rats. Reprod. Sci. 2013, 20, 1230–1236. [Google Scholar] [CrossRef]
- Khodarahmian, M.; Amidi, F.; Moini, A.; Kashani, L.; Salahi, E.; Danaii-Mehrabad, S.; Nashtaei, M.S.; Mojtahedi, M.F.; Esfandyari, S.; Sobhani, A. A randomized exploratory trial to assess the effects of resveratrol on VEGF and TNF-α 2 expression in endometriosis women. J. Reprod. Immunol. 2020, 143, 103248. [Google Scholar] [CrossRef]
- Zhou, A.; Hong, Y.; Lv, Y. Sulforaphane attenuates endometriosis in rat models through inhibiting pi3k/akt signaling pathway. Dose Res. 2019, 17, 1559325819855538. [Google Scholar] [CrossRef]
- Valipour, J.; Nashtaei, M.S.; Khosravizadeh, Z.; Mahdavinezhad, F.; Nekoonam, S.; Esfandyari, S.; Amidi, F. Effect of sulforaphane on apoptosis, reactive oxygen species and lipids peroxidation of human sperm during cryopreservation. Cryobiology 2021, 99, 122–130. [Google Scholar] [CrossRef]
- Stephens, F.B.; Constantin-Teodosiu, D.; Greenhaff, P.L. New insights concerning the role of carnitine in the regulation of fuel metabolism in skeletal muscle. J. Pshycol. 2007, 581, 431–444. [Google Scholar] [CrossRef]
- Tselekidou, E.D.; Vassiliadis, S.; Athanassakis, I. Establishment or Aggravation of Endometriosis by L-Carnitine: The Role of Pge1 and Pge2 in the Endometriosis-Induction Process. New Developments in Endometriosis. Available online: https://www.createspace.com (accessed on 15 January 2021).
- Gerbase, A.C.; Rowley, J.T.; Heymann, D.H.; Berkley, S.F.; Piot, P. Global prevalence and incidence estimates of selected curable STDs. Sex Transm. Infect. 1998, 74, S12–S16. [Google Scholar]
- Onisto, M.; Fasolato, S.; Veggian, R.; Caenazzo, C.; Garbisa, S. Hormonal and basement membrane markers for immunoidentification of cultured human trophoblast cells. Int. J. Gynaecol. Obstet. 1989, 30, 145–153. [Google Scholar] [CrossRef]
- Rabiu, K.A.; Adewunmi, A.A.; Akinlusi, F.M.; Akinola, O.I. Female reproductive tract infections: Understandings and care seeking behaviour among women of reproductive age in Lagos, Nigeria. BMC Women’s Health. 2010, 10, 8. [Google Scholar] [CrossRef]
- Moreno, I.; Simon, C. Relevance of assessing the uterine microbiota in infertility. Fertil. Steril. 2018, 110, 337–343. [Google Scholar] [CrossRef]
- Heil, B.A.; Paccamonti, D.L.; Sones, J.L. Role for the mammalian female reproductive tract microbiome in pregnancy outcomes. Physiol. Genomics 2019, 51, 390–399. [Google Scholar] [CrossRef]
- Krawinkel, M.B. Interaction of nutrition and infections globally: An overview. Ann. Nutr. Metab. 2012, 61, 39–45. [Google Scholar] [CrossRef]
- Cassotta, M.; Forbes-Hernández, T.Y.; Calderón Iglesias, R.; Ruiz, R.; Elexpuru Zabaleta, M.; Giampieri, F.; Battino, M. Links between nutrition, infectious diseases, and microbiota: Emerging technologies and opportunities for human-focused research. Nutrients 2020, 12, 1827. [Google Scholar] [CrossRef]
- Molenaar, M.C.; Singer, M.; Ouburg, S. The two-sided role of the vaginal microbiome in Chlamydia trachomatis and Mycoplasma genitalium pathogenesis. J. Reprod. Immunol. 2018, 130, 11–17. [Google Scholar] [CrossRef]
- Tuddenham, S.; Ghanem, K.G. A microbiome variable in the HIV-prevention equation. Science 2017, 356, 907–908. [Google Scholar] [CrossRef]
- Martin, D.H.; Marrazzo, J.M. The vaginal microbiome: Current understanding and future directions. J. Infect. Dis. 2016, 214, S36–S41. [Google Scholar] [CrossRef]
- Borgogna, J.L.; Shardell, M.D.; Yeoman, C.J.; Ghanem, K.G.; Kadriu, H.; Ulanov, A.V.; Gaydos, C.A.; Hardick, J.; Robinson, C.K.; Bavoil, P.M.; et al. The association of Chlamydia trachomatis and Mycoplasma genitalium infection with the vaginal metabolome. Sci. Rep. 2020, 10, 3420. [Google Scholar] [CrossRef]
- Ng, K.Y.; Mingels, R.; Morgan, H.; Macklon, N.; Cheong, Y. In vivo oxygen, temperature and pH dynamics in the female reproductive tract and their importance in human conception: A systematic review. Hum. Reprod. Update 2018, 24, 15–34. [Google Scholar] [CrossRef]
- Van Oostrum, N.; De Sutter, P.; Meys, J.; Verstraelen, H. Risks associated with bacterial vaginosis in infertility patients: A systematic review and meta-analysis. Hum. Reprod. 2013, 28, 1809–1815. [Google Scholar] [CrossRef]
- Koumans, E.H.; Sternberg, M.; Bruce, C.; McQuillan, G.; Kendrick, J.; Sutton, M.; Markowitz, L.E. The prevalence of bacterial vaginosis in the United States, 2001-2004; associations with symptoms, sexual behaviors, and reproductive health. Sex. Transm. Dis. 2007, 34, 864–869. [Google Scholar] [CrossRef] [PubMed]
- Thoma, M.E.; Klebanoff, M.A.; Rovner, A.J.; Nansel, T.R.; Neggers, Y.; Andrews, W.W.; Schwebke, J.R. Bacterial vaginosis is associated with variation in dietary indices. J. Nutr. 2011, 141, 1698–1704. [Google Scholar] [CrossRef] [PubMed]
- Smart, S.; Singal, A.; Mindel, A. Social and sexual risk factors for bacterial vaginosis. Sex. Transm. Infect. 2004, 80, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Neggers, Y.H.; Nansel, T.R.; Andrews, W.W.; Schwebke, J.R.; Yu, K.F.; Goldenberg, R.L.; Klebanoff, M.A. Dietary intake of selected nutrients affects bacterial vaginosis in women. J. Nutr. 2007, 137, 2128–2133. [Google Scholar] [CrossRef]
- Bodnar, L.M.; Krohn, M.A.; Simhan, H.N. Maternal vitamin D deficiency is associated with bacterial vaginosis in the first trimester of pregnancy. J. Nutr. 2009, 139, 1157–1161. [Google Scholar] [CrossRef]
- Mitchell, C.; Marrazzo, J. Bacterial vaginosis and the cervicovaginal immune response. Am. J. Reprod. Immunol. 2014, 71, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Dunlop, A.L.; Taylor, R.N.; Tangpricha, V.; Fortunato, S.; Menon, R. Maternal vitamin D, folate, and polyunsaturated fatty acid status and bacterial vaginosis during pregnancy. Infect. Dis. Obstet. Gynecol. 2011, 2011, 216217. [Google Scholar] [CrossRef]
- Hensel, K.J.; Randis, T.M.; Gelber, S.E.; Ratner, A.J. Pregnancy-specific association of vitamin D deficiency and bacterial vaginosis. Am. J. Obstet. Gynecol. 2011, 204, 41-e1. [Google Scholar] [CrossRef]
- Taheri, M.; Baheiraei, A.; Foroushani, A.R.; Nikmanesh, B.; Modarres, M. Treatment of vitamin D deficiency is an effective method in the elimination of asymptomatic bacterial vaginosis: A placebo-controlled randomized clinical trial. Indian J. Med. Res. 2015, 141, 799–806. [Google Scholar]
- Al-Ghazzewi, F.H.; Tester, R.F. Biotherapeutic agents and vaginal health. J. Appl. Microbiol. 2016, 121, 18–27. [Google Scholar] [CrossRef]
- Tuominen, H.; Rautava, S.; Syrjänen, S.; Collado, M.C.; Rautava, J. HPV infection and bacterial microbiota in the placenta, uterine cervix and oral mucosa. Sci. Rep. 2018, 8, 9787. [Google Scholar] [CrossRef]
- Atashili, J.; Poole, C.; Ndumbe, P.M.; Adimora, A.A.; Smith, J.S. Bacterial vaginosis and HIV acquisition: A meta-analysis of published studies. AIDS 2008, 22, 1493–1501. [Google Scholar] [CrossRef]
- Allsworth, J.E.; Lewis, V.A.; Peipert, J.F. Viral sexually transmitted infections and bacterial vaginosis: 2001–2004 national health and nutrition examination survey data. Sex. Transm. Dis. 2008, 35, 791–796. [Google Scholar] [CrossRef]
- White, M.C.; Hayes, N.S.; Richardson, L.C. Public health’s future role in cancer survivorship. Am. J. Prev. Med. 2015, 49, S550–S553. [Google Scholar] [CrossRef]
- Dunn, B.K.; Kramer, B.S. Cancer prevention: Lessons learned and future directions. Trends Cancer 2016, 2, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Sundstrom, K.; Elfstrom, K.M. Advances in cervical cancer prevention: Efficacy, effectiveness, elimination? PLoS MED 2020, 17, e1003035. [Google Scholar] [CrossRef]
- MacKintosh, M.L.; Crosbie, E.J. Prevention strategies in endometrial carcinoma. Curr. Oncol. Rep. 2018, 20, 101. [Google Scholar] [CrossRef]
- Temkin, S.M.; Bergstrom, J.; Samimi, G.; Minasian, L. Ovarian cancer prevention in high.-risk women. Clin. Obstet. Gynecol. 2017, 60, 738–757. [Google Scholar] [CrossRef]
- Szewczuk, M.; Gasiorowska, E.; Matysiak, K.; Nowak-Markwitz, E. The role of artificial nutrition in gynecological cancer therapy. Ginekol. Pol. 2019, 90, 167–172. [Google Scholar] [CrossRef]
- Koshiyama, M. The effects of the dietary and nutrient intake on gynecologic cancers. Healthcare 2019, 7, 88. [Google Scholar] [CrossRef]
- Key, T.J.; Bradbury, K.E.; Perez-Cornago, A.; Sinha, R.; Tsilidis, K.K.; Tsugane, S. Diet, nutrition, and cancer risk: What do we know and what is the way forward? BMJ 2020, 368, 368. [Google Scholar] [CrossRef]
- Ferenczy, A.; Franco, E. Persistent human papillomavirus infection and cervical neoplasia. Lancet Oncol. 2002, 3, 11–16. [Google Scholar] [CrossRef]
- Stebbing, J.; Hart, C.A. Antioxidants and cancer. Lancet Oncol. 2011, 12, 996. [Google Scholar] [CrossRef]
- Tomita, L.Y.; Roteli-Martins, C.M.; Villa, L.L.; Franco, E.L.; Cardoso, M.A. Associations of dietary dark-green and deep-yellow vegetables and fruits with cervical intraepithelial neoplasia: Modification by smoking. Br. J. Nutr. 2011, 105, 928–937. [Google Scholar] [CrossRef] [PubMed]
- Siegel, E.M.; Salemi, J.L.; Villa, L.L.; Ferenczy, A.; Franco, E.L.; Giuliano, A.R. Dietary consumption of antioxidant nutrients and risk of incident cervical intraepithelial neoplasia. Gynecol. Oncol. 2010, 118, 289–294. [Google Scholar] [CrossRef]
- Jia, Y.; Hu, T.; Hang, C.Y.; Yang, R.; Li, X.; Chen, Z.L.; Mei, Y.D.; Zhang, Q.H.; Huang, K.C.; Xiang, Q.Y.; et al. Case-control study of diet in patients with cervical cancer or precancerosis in Wufeng, a high incidence region in China. Asian Pac. J. Cancer Prev. 2012, 13, 5299–5302. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Zhu, H.; Lin, C.; Che, J.; Tian, X.; Han, S.; Zhao, H.; Zhu, Y.; Mao, D. Associations between antioxidant vitamins and the risk of invasive cervical cancer in Chinese women: A case-control study. Sci. Rep. 2015, 5, 13607. [Google Scholar] [CrossRef]
- Giuliano, A.R.; Siegel, E.M.; Roe, D.J.; Ferreira, S.; Luiza Baggio, M.; Galan, L.; Duarte-Franco, E.; Villa, L.L.; Rohan, T.E.; Marshall, J.R.; et al. Dietary intake and risk of persistent human papillomavirus (HPV) infection: The Ludwig-McGill HPV natural history study. J. Infect. Dis. 2003, 188, 1508–1516. [Google Scholar] [CrossRef]
- Ono, A.; Koshiyama, M.; Nakagawa, M.; Watanabe, Y.; Ikuta, E.; Seki, K.; Oowaki, M. The preventive effect of dietary antioxidants on cervical cancer development. Medicina 2020, 56, 604. [Google Scholar] [CrossRef]
- Yeo, A.S.; Schiff, M.A.; Montoya, G.; Masuk, M.; van Asselt-King, L.; Becker, T.M. Serum micronutrients and cervical dysplasia in Southwestern American Indian women. Nutr. Cancer 2000, 38, 141–150. [Google Scholar] [CrossRef]
- Huang, X.; Chen, C.; Zhu, F.; Zhang, Y.; Feng, Q.; Li, J.; Yu, Q.; Zhong, Y.; Luo, S.; Gao, J. Association between dietary vitamin A and HPV infection in American women: Data from NHANES 2003–2016. Biomed Res. Int. 2020, 2020, 4317610. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, S.; Raza, S.T.; Faizal Ahmed, A.A.; Abbas, S.; Mahdi, F. The role of vitamin e in human health and some diseases. Sultan Qaboos Univ. Med. J. 2014, 14, e157–e165. [Google Scholar] [PubMed]
- Hu, X.; Li, S.; Zhou, L.; Zhao, M.; Zhu, X. Effect of vitamin E supplementation on uterine cervical neoplasm: A meta-analysis of case-control studies. PLoS ONE 2017, 12, e0183395. [Google Scholar] [CrossRef]
- Vahedpoor, Z.; Jamilian, M.; Bahmani, F.; Aghadavod, E.; Karamali, M.; Kashanian, M.; Asemi, Z. Effects of long-term vitamin d supplementation on regression and metabolic status of cervical intraepithelial neoplasia: A randomized, double-blind, placebo-controlled trial. Horm. Cancer 2017, 8, 58–67. [Google Scholar] [CrossRef]
- Hernandez, B.Y.; McDuffie, K.; Wilkens, L.R.; Kamemoto, L.; Goodman, M.T. Diet. and premalignant lesions of the cervix: Evidence of a protective role for folate, riboflavin, thiamin, and vitamin B12. Cancer Causes Control 2003, 14, 859–870. [Google Scholar] [CrossRef] [PubMed]
- Zoberi, I.; Bradbury, C.M.; Curry, H.A.; Bisht, K.S.; Goswami, P.C.; Roti, J.L.; Gius, D. Radiosensitizing and anti-proliferative effects of resveratrol in two human cervical tumor cell lines. Cancer Lett. 2002, 175, 165–173. [Google Scholar] [CrossRef]
- Silva, G.Á.; Nunes, R.A.; Morale, M.G.; Boccardo, E.; Aguayo, F.; Termini, L. Oxidative stress: Therapeutic approaches for cervical cancer treatment. Clinics 2018, 73, e548s. [Google Scholar] [CrossRef] [PubMed]
- Venkatraman, M.; Anto, R.J.; Nair, A.; Varghese, M.; Karunagaran, D. Biological and chemical inhibitors of NF-kappaB sensitize SiHa cells to cisplatin-induced apoptosis. Mol. Carcinog. 2005, 44, 51–59. [Google Scholar] [CrossRef]
- Ciebiera, M.; Łukaszuk, K.; Męczekalski, B.; Ciebiera, M.; Wojtyła, C.; Słabuszewska-Jóźwiak, A.; Jakiel, G. Alternative oral agents in prophylaxis and therapy of uterine fibroids-an up-to-date review. Int. J. Mol. Sci. 2017, 18, 2586. [Google Scholar] [CrossRef] [PubMed]
- Min, K.J.; Kwon, T.K. Anticancer effects and molecular mechanisms of epigallocatechin-3-gallate. Integr. Med. Res. 2014, 3, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Pennant, M.E.; Mehta, R.; Moody, P.; Hackett, G.; Prentice, A.; Sharp, S.J.; Lakshman, R. Premenopausal abnormal uterine bleeding and risk of endometrial cancer. BJOG 2017, 124, 404–411. [Google Scholar] [CrossRef]
- Setiawan, V.W.; Yang, H.P.; Pike, M.C.; McCann, S.E.; Yu, H.; Xiang, Y.B.; Wolk, A.; Wentzensen, N.; Weiss, N.S.; Webb, P.M.; et al. Type I and II endometrial cancers: Have they different risk factors? J. Clin. Oncol. 2013, 31, 2607–2618. [Google Scholar] [CrossRef]
- Morice, P.; Leary, A.; Creutzberg, C.; Abu-Rustum, N.; Darai, E. Endometrial cancer. Lancet 2016, 387, 1094–1108. [Google Scholar] [CrossRef]
- Leslie, K.K.; Thiel, K.W.; Goodheart, M.J.; De Geest, K.; Jia, Y.; Yang, S. Endometrial cancer. Obstet. Gynecol. Clin. N. Am. 2012, 39, 255–268. [Google Scholar] [CrossRef]
- Bandera, E.V.; Gifkins, D.M.; Moore, D.F.; McCullough, M.L.; Kushi, L.H. Antioxidant vitamins and the risk of endometrial cancer: A dose-response meta-analysis. Cancer Causes Control 2009, 20, 699–711. [Google Scholar] [CrossRef]
- Cui, X.; Rosner, B.; Willett, W.C.; Hankinson, S.E. Antioxidant intake and risk of endometrial cancer: Results from the Nurses’ Health Study. Int. J. Cancer 2011, 128, 1169–1178. [Google Scholar] [CrossRef]
- Acmaz, G.; Aksoy, H.; Albayrak, E.; Baser, M.; Ozyurt, S.; Aksoy, U.; Unal, D. Evaluation of endometrial precancerous lesions in postmenopausal obese women—A high risk group? Asian Pac. J. Cancer Prev. 2014, 15, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Brasky, T.M.; Neuhouser, M.L.; Cohn, D.E.; White, E. Associations of long-chain omega-3 fatty acids and fish intake with endometrial cancer risk in the VITamins and Lifestyle cohort. Am. J. Clin. Nutr. 2014, 99, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, S.; Peeters, P.H.; Bezemer, I.D.; Dossus, L.; Biessy, C.; Sacerdote, C.; Berrino, F.; Panico, S.; Palli, D.; Tumino, R.; et al. Relationship of alcohol intake and sex steroid concentrations in blood in pre- and post-menopausal women: The European prospective investigation into cancer and nutrition. Cancer Causes Control 2006, 17, 1033–1043. [Google Scholar] [CrossRef] [PubMed]
- Fedirko, V.; Jenab, M.; Rinaldi, S.; Biessy, C.; Allen, N.E.; Dossus, L.; Onland-Moret, N.C.; Schütze, M.; Tjønneland, A.; Hansen, L.; et al. Alcohol drinking and endometrial cancer risk in the European Prospective Investigation into cancer and nutrition (EPIC) study. Ann. Epidemiol. 2013, 23, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.; Edefonti, V.; Parpinel, M.; Lagiou, P.; Franchi, M.; Ferraroni, M.; Decarli, A.; Zucchetto, A.; Serraino, D.; Dal Maso, L.; et al. Proanthocyanidins and other flavonoids in relation to endometrial cancer risk: A case-control study in Italy. Br. J. Cancer 2013, 109, 1914–1920. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Messina, M.J.; Persky, V.; Setchell, K.D.; Barnes, S. Soy intake and cancer risk: A review of the in vitro and in vivo data. Nutr. Cancer 1994, 21, 113–131. [Google Scholar] [CrossRef]
- Wang, L.; Lee, I.M.; Zhang, S.M.; Blumberg, J.B.; Buring, J.E.; Sesso, H.D. Dietary intake of selected flavonols, flavones, and flavonoid-rich foods and risk of cancer in middle-aged and older women. Am. J. Clin. Nutr. 2009, 89, 905–912. [Google Scholar] [CrossRef]
- Bandera, E.V.; Williams, M.G.; Sima, C.; Bayuga, S.; Pulick, K.; Wilcox, H.; Soslow, R.; Zauber, A.G.; Olson, S.H. Phytoestrogen consumption and endometrial cancer risk: A population-based case-control study in New Jersey. Cancer Causes Control 2009, 20, 1117–1127. [Google Scholar] [CrossRef]
- Ollberding, N.J.; Lim, U.; Wilkens, L.R.; Setiawan, V.W.; Shvetsov, Y.B.; Henderson, B.E.; Kolonel, L.N.; Goodman, M.T. Legume, soy, tofu, and isoflavone intake and endometrial cancer risk in postmenopausal women in the multiethnic cohort study. J. Natl. Cancer Inst. 2012, 104, 67–76. [Google Scholar] [CrossRef]
- Zhang, G.Q.; Chen, J.L.; Liu, Q.; Zhang, Y.; Zeng, H.; Zhao, Y. Soy intake is associated with lower endometrial cancer risk: A systematic review and meta-analysis of observational studies. Medicine 2015, 94, e2281. [Google Scholar] [CrossRef]
- Zhong, X.S.; Ge, J.; Chen, S.W.; Xiong, Y.Q.; Ma, S.J.; Chen, Q. Association between dietary isoflavones in soy and legumes and endometrial cancer: A systematic review and meta-analysis. J. Acad. Nutr. Diet. 2018, 118, 637–651. [Google Scholar] [CrossRef] [PubMed]
- Unfer, V.; Casini, M.L.; Costabile, L.; Mignosa, M.; Gerli, S.; Di Renzo, G.C. Endometrial effects of long-term treatment with phytoestrogens: A randomized, double-blind, placebo-controlled study. Fertil. Steril. 2004, 82, 145–148. [Google Scholar] [CrossRef] [PubMed]
- Mohr, S.B.; Garland, C.F.; Gorham, E.D.; Grant, W.B.; Garland, F.C. Is ultraviolet B irradiance inversely associated with incidence rates of endometrial cancer: An ecological study of 107 countries. Prev. Med. 2007, 45, 327–331. [Google Scholar] [CrossRef]
- McCullough, M.L.; Bandera, E.V.; Moore, D.F.; Kushi, L.H. Vitamin D and calcium intake in relation to risk of endometrial cancer: A systematic review of the literature. Prev. Med. 2008, 46, 298–302. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Rankin, G.O.; Li, Z.; DePriest, L.; Chen, Y.C. Kaempferol induces apoptosis in ovarian cancer cells through activating p53 in the intrinsic pathway. Food Chem. 2011, 128, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Chuwa, A.H.; Sone, K.; Oda, K.; Tanikawa, M.; Kukita, A.; Kojima, M.; Oki, S.; Fukuda, T.; Takeuchi, M.; Miyasaka, A.; et al. Kaempferol, a natural dietary flavonoid, suppresses 17beta-estradiol-induced survivin expression and causes apoptotic cell death in endometrial cancer. Oncol. Lett. 2018, 16, 6195–6201. [Google Scholar]
- Yang, T.O.; Crowe, F.; Cairns, B.J.; Reeves, G.K.; Beral, V. Tea and coffee and risk of endometrial cancer: Cohort study and meta-analysis. Am. J. Clin. Nutr. 2015, 101, 570–578. [Google Scholar] [CrossRef]
- Zhou, Q.; Li, H.; Zhou, J.G.; Ma, Y.; Wu, T.; Ma, H. Green tea, black tea consumption and risk of endometrial cancer: A systematic review and meta-analysis. Arch. Gynecol. Obstet. 2016, 293, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Coburn, S.B.; Bray, F.; Sherman, M.E.; Trabert, B. International patterns and trends in ovarian cancer incidence, overall and by histologic subtype. Int. J. Cancer 2017, 140, 2451–2460. [Google Scholar] [CrossRef]
- Jayson, G.C.; Kohn, E.C.; Kitchener, H.C.; Ledermann, J.A. Ovarian cancer. Lancet 2014, 384, 1376–1388. [Google Scholar] [CrossRef]
- Goff, B.A.; Mandel, L.; Muntz, H.G.; Melancon, C.H. Ovarian carcinoma diagnosis. Cancer 2000, 89, 2068–2075. [Google Scholar] [CrossRef]
- Kisielewski, R.; Mazurek, A.; Laudański, P.; Tołwińska, A. Inflammation and ovarian cancer—Current views. Ginekol. Pol. 2013, 84, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Shivappa, N.; Hébert, J.R.; Rosato, V.; Rossi, M.; Montella, M.; Serraino, D.; La Vecchia, C. Dietary inflammatory index and ovarian cancer risk in a large Italian case-control study. Cancer Causes Control 2016, 27, 897–906. [Google Scholar] [CrossRef]
- Dolecek, T.A.; McCarthy, B.J.; Joslin, C.E.; Peterson, C.E.; Kim, S.; Freels, S.A.; Davis, F.G. Prediagnosis food patterns are associated with length of survival from epithelial ovarian cancer. J. Am. Diet. Assoc. 2010, 110, 369–382. [Google Scholar] [CrossRef]
- Playdon, M.C.; Nagle, C.M.; Ibiebele, T.I.; Ferrucci, L.M.; Protani, M.M.; Carter, J.; Hyde, S.E.; Neesham, D.; Nicklin, J.L.; Mayne, S.T.; et al. Pre-diagnosis diet and survival after a diagnosis of ovarian cancer. Br. J. Cancer 2017, 116, 1627–1637. [Google Scholar] [CrossRef]
- Qiu, W.; Lu, H.; Qi, Y.; Wang, X. Dietary fat intake and ovarian cancer risk: A meta-analysis of epidemiological studies. Oncotarget 2016, 7, 37390–37406. [Google Scholar] [CrossRef]
- Bandera, E.V.; King, M.; Chandran, U.; Paddock, L.E.; Rodriguez-Rodriguez, L.; Olson, S.H. Phytoestrogen consumption from foods and supplements and epithelial ovarian cancer risk: A population-based case control study. BMC Wom. Health 2011, 11, 40. [Google Scholar] [CrossRef] [PubMed]
- Neill, A.S.; Ibiebele, T.I.; Lahmann, P.H.; Hughes, M.C.; Nagle, C.M.; Webb, P.M. Dietary phyto-oestrogens and the risk of ovarian and endometrial cancers: Findings from two Australian case-control studies. Br. J. Nutr. 2014, 111, 1430–1440. [Google Scholar] [CrossRef] [PubMed]
- Hedelin, M.; Löf, M.; Andersson, T.M.; Adlercreutz, H.; Weiderpass, E. Dietary phytoestrogens and the risk of ovarian cancer in the women’s lifestyle and health cohort study. Cancer Epidemiol. Biomarkers Prev. 2011, 20, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Hua, X.; Yu, L.; You, R.; Yang, Y.; Liao, J.; Chen, D.; Yu, L. Association among Dietary flavonoids, flavonoid subclasses and ovarian cancer risk: A meta-analysis. PLoS ONE 2016, 11, e0151134. [Google Scholar] [CrossRef] [PubMed]
- Shafabakhsh, R.; Asemi, Z. Quercetin: A natural compound for ovarian cancer treatment. J. Ovarian Res. 2019, 12, 55. [Google Scholar] [CrossRef]
- Luo, H.; Rankin, G.O.; Liu, L.; Daddysman, M.K.; Jiang, B.H.; Chen, Y.C. Kaempferol inhibits angiogenesis and VEGF expression through both HIF dependent and independent pathways in human ovarian cancer cells. Nutr. Cancer 2009, 61, 554–563. [Google Scholar] [CrossRef]
- Huang, H.; Chen, A.Y.; Ye, X.; Guan, R.; Rankin, G.O.; Chen, Y.C. Galangin, a flavonoid from lesser galangal, induced apoptosis via p53-dependent pathway in ovarian cancer cells. Molecules 2020, 25, 1579. [Google Scholar] [CrossRef]
- Lin, Y.G.; Kunnumakkara, A.B.; Nair, A.; Merritt, W.M.; Han, L.Y.; Armaiz-Pena, G.N.; Kamat, A.A.; Spannuth, W.A.; Gershenson, D.M.; Lutgendorf, S.K.; et al. Curcumin inhibits tumor growth and angiogenesis in ovarian carcinoma by targeting the nuclear factor-kappaB pathway. Clin. Cancer Res. 2007, 13, 3423–3430. [Google Scholar] [CrossRef] [PubMed]
- Pourhanifeh, M.H.; Darvish, M.; Tabatabaeian, J.; Fard, M.R.; Mottaghi, R.; Azadchehr, M.J.; Jahanshahi, M.; Sahebkar, A.; Mirzaei, H. Therapeutic role of curcumin and its novel formulations in gynecological cancers. J. Ovarian Res. 2020, 13, 30. [Google Scholar] [CrossRef]
- Wahl, H.; Tan, L.; Griffith, K.; Choi, M.; Liu, J.R. Curcumin enhances Apo2L/TRAIL-induced apoptosis in chemoresistant ovarian cancer cells. Gynecol. Oncol. 2007, 105, 104–112. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Wang, D.; Zou, D.; Wang, C.; Lopes-Bastos, B.; Jiang, W.G.; Chester, J.; Zhou, Q.; Cai, J. Re-purposing of curcumin as an anti-metastatic agent for the treatment of epithelial ovarian cancer: In vitro model using cancer stem cell enriched ovarian cancer spheroids. Oncotarget 2016, 7, 86374–86387. [Google Scholar] [CrossRef] [PubMed]
- Yallapu, M.M.; Maher, D.M.; Sundram, V.; Bell, M.C.; Jaggi, M.; Chauhan, S.C. Curcumin induces chemo/radio-sensitization in ovarian cancer cells and curcumin nanoparticles inhibit ovarian cancer cell growth. J. Ovarian Res. 2010, 3, 11. [Google Scholar] [CrossRef]
- Sun, Y.; Xun, K.; Wang, Y.; Chen, X. A systematic review of the anticancer properties of berberine, a natural product from Chinese herbs. Anticancer Drugs 2009, 20, 757–769. [Google Scholar] [CrossRef]
- Liu, L.; Fan, J.; Ai, G.; Liu, J.; Luo, N.; Li, C.; Cheng, Z. Berberine in combination with cisplatin induces necroptosis and apoptosis in ovarian cancer cells. Biol. Res. 2019, 52, 37. [Google Scholar] [CrossRef] [PubMed]
- Tse, A.K.; Wan, C.K.; Shen, X.L.; Yang, M.; Fong, W.F. Honokiol inhibits TNF-alpha-stimulated NF-kappaB activation and NF-kappaB-regulated gene expression through suppression of IKK activation. Biochem. Pharmacol. 2005, 70, 1443–1457. [Google Scholar] [CrossRef]
- Lee, J.S.; Sul, J.Y.; Park, J.B.; Lee, M.S.; Cha, E.Y.; Ko, Y.B. Honokiol induces apoptosis and suppresses migration and invasion of ovarian carcinoma cells via AMPK/mTOR signaling pathway. Int. J. Mol. Med. 2019, 43, 1969–1978. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.H.; Wu, T.Y.; Hsiao, Y.T.; Lin, J.H.; Hsu, S.C.; Hsia, T.C.; Yang, S.T.; Hsu, W.H.; Chung, J.G. Bufalin induces cell death in human lung cancer cells through disruption of DNA damage response pathways. Am. J. Chin. Med. 2014, 42, 729–742. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Dou, H.; Wang, Z.; Zhang, Q. Bufalin inhibits ovarian carcinoma via targeting mTOR/HIF-alpha pathway. Basic Clin. Pharmacol. Toxicol. 2020. [Google Scholar] [CrossRef]
- Kosuge, T.; Adachi, T.; Kamiya, H. Isolation of tetramethylpyrazine from culture of Bacillus natto, and biosynthetic pathways of tetramethylpyrazine. Nature 1962, 195, 1103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Ding, S.; Xia, L. Ligustrazine inhibits the proliferation and migration of ovarian cancer cells via regulating miR-211. Biosci. Rep. 2020, 41, BSR20200199. [Google Scholar] [CrossRef] [PubMed]
- Ciebiera, M.; Włodarczyk, M.; Ciebiera, M.; Zaręba, K.; Łukaszuk, K.; Jakiel, G. Vitamin D and Uterine Fibroids-Review of the Literature and Novel Concepts. Int. J. Mol. Sci. 2018, 19, 2051. [Google Scholar] [CrossRef]
- Dovnik, A.; Dovnik, N.F. Vitamin D and ovarian cancer: Systematic review of the literature with a focus on molecular mechanisms. Cells 2020, 9, 335. [Google Scholar] [CrossRef]
- Song, X.; Li, Z.; Ji, X.; Zhang, D. Calcium intake and the risk of ovarian cancer: A meta-analysis. Nutrients 2017, 9, 679. [Google Scholar] [CrossRef] [PubMed]
- Livdans-Forret, A.B.; Harvey, P.J.; Larkin-Thier, S.M. Menorrhagia: A synopsis of management focusing on herbal and nutritional supplements, and chiropractic. J. Can. Chiropr. Assoc. 2007, 51, 235–246. [Google Scholar]
- Geller, S.E.; Studee, L. Botanical and dietary supplements for menopausal symptoms: What works, what does not. J Wom. Health 2005, 14, 634–649. [Google Scholar] [CrossRef] [PubMed]
- Low, M.S.; Speedy, J.; Styles, C.E.; De-Regil, L.M.; Pasricha, S.R. Daily iron supplementation for improving anaemia, iron status and health in menstruating women. Cochrane Datab. Syst. Rev. 2016, 4, CD009747. [Google Scholar] [CrossRef] [PubMed]
- Lithgow, D.M.; Politzer, W.M. Vitamin A in the treatment of menorrhagia. S. Afr. Med. J. 1977, 51, 91–93. [Google Scholar]
- Ayre, J.E.; Bauld, W.A. Thiamine deficiency and high. estrogen findings in uterine cancer and in menorrhagia. Science 1946, 103, 441–445. [Google Scholar] [CrossRef]
- Cohen, J.D.; Rubin, H.W. Functional menorrhagia: Treatment with bioflavonoids and vitamin C. Curr. Ther. Res. Clin. Exp. 1960, 2, 539–542. [Google Scholar] [PubMed]
- Morrow, C.; Naumburg, E.H. Dysmenorrhea. Prim. Care 2009, 36, 19–32. [Google Scholar] [CrossRef]
- Dennehy, C.E. The use of herbs and dietary supplements in gynecology: An evidence-based review. J. Midwifery Women’s Health 2006, 51, 402–409. [Google Scholar] [CrossRef]
- Bajalan, Z.; Alimoradi, Z.; Moafi, F. Nutrition as a potential factor of primary dysmenorrhea: A systematic review of observational studies. Gynecol. Obstet. Invest. 2019, 84, 209–224. [Google Scholar] [CrossRef]
- Pattanittum, P.; Kunyanone, N.; Brown, J.; Sangkomkamhang, U.S.; Barnes, J.; Seyfoddin, V.; Marjoribanks, J. Dietary supplements for dysmenorrhoea. Cochrane Database Syst. Rev. 2016, 3, CD002124. [Google Scholar] [CrossRef]
- Naz, M.S.; Kiani, Z.; Fakari, F.R.; Ghasemi, V.; Abed, M.; Ozgoli, G. The effect of micronutrients on pain management of primary dysmenorrhea: A systematic review and meta-analysis. J. Caring Sci. 2020, 9, 47–56. [Google Scholar]
- Shin, H.J.; Na, H.S.; Do, S.H. Magnesium and Pain. Nutrients 2020, 9, 2184. [Google Scholar] [CrossRef] [PubMed]
- Chiang, Y.F.; Hung, H.C.; Chen, H.Y.; Huang, K.C.; Lin, P.H.; Chang, J.Y.; Huang, T.C.; Hsia, S.M. The inhibitory effect of extra virgin olive oil and its active compound oleocanthal on prostaglandin-induced uterine hypercontraction and pain-ex vivo and in vivo study. Nutrients 2020, 12, 3012. [Google Scholar] [CrossRef]
- Lee, H.W.; Ang, L.; Lee, M.S.; Alimoradi, Z.; Kim, E. Fennel for reducing pain in primary dysmenorrhea: A systematic review and meta-analysis of randomized controlled trials. Nutrients 2020, 12, 3438. [Google Scholar] [CrossRef] [PubMed]
- Nagata, C.; Hirokawa, K.; Shimizu, N.; Shimizu, H. Associations of menstrual pain with intakes of soy, fat and dietary fiber in Japanese women. Eur. J. Clin. Nutr. 2005, 59, 88–92. [Google Scholar] [CrossRef][Green Version]
- Mehrpooya, M.; Eshraghi, A.; Rabiee, S.; Larki-Harchegani, A.; Ataei, S. Comparison the effect of fish-oil and calcium supplementation on treatment of primary dysmenorrhea. Rev. Recent Clin. Trials 2017, 12, 148–153. [Google Scholar] [CrossRef]
- Sadeghi, N.; Paknezhad, F.; Rashidi Nooshabadi, M.; Kavianpour, M.; Jafari Rad, S.; Khadem Haghighian, H. Vitamin E and fish oil, separately or in combination, on treatment of primary dysmenorrhea: A double-blind, randomized clinical trial. Gynecol. Endocrinol. 2018, 34, 804–808. [Google Scholar] [CrossRef]
- Lerchbaum, E.; Rabe, T. Vitamin D and female fertility. Curr. Opin. Obstet. Gynecol. 2014, 26, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, A.; Avan, A.; Sadeghnia, H.R.; Esmaeili, H.; Tayefi, M.; Ghasemi, F.; Nejati Salehkhani, F.; Arabpour-Dahoue, M.; Rastgar-Moghadam, A.; Ferns, G.A.; et al. High. dose vitamin D supplementation can improve menstrual problems, dysmenorrhea, and premenstrual syndrome in adolescents. Gynecol. Endocrinol. 2018, 34, 659–663. [Google Scholar] [CrossRef]
- Abdi, F.; Amjadi, M.A.; Zaheri, F.; Rahnemaei, F.A. Role of vitamin D and calcium in the relief of primary dysmenorrhea: A systematic review. Obstet. Gynecol. Sci. 2021, 64, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Mao, X.; Su, J.; Geng, Y.; Guo, R.; Tang, S.; Li, J.; Xiao, X.; Xu, H.; Yang, H. A network pharmacology-based strategy deciphers the underlying molecular mechanisms of Qixuehe Capsule in the treatment of menstrual disorders. Chin. Med. 2017, 12, 23. [Google Scholar] [CrossRef] [PubMed]
- Zekavat, O.R.; Karimi, M.Y.; Amanat, A.; Alipour, F. A randomised controlled trial of oral zinc sulphate for primary dysmenorrhoea in adolescent females. Aust. N. Z. J. Obstet. Gynaecol. 2015, 55, 369–373. [Google Scholar] [CrossRef]
- Nasiadek, M.; Stragierowicz, J.; Klimczak, M.; Kilanowicz, A. The role of zinc in selected female reproductive system disorders. Nutrients 2020, 12, 2464. [Google Scholar] [CrossRef] [PubMed]
- Chao, M.T.; Wade, C.M.; Booth, S.L. Increase in plasma phylloquinone concentrations following acupoint injection for the treatment of primary dysmenorrhea. J. Acupunct. Meridian. Stud. 2014, 7, 151–154. [Google Scholar] [CrossRef]
- Wade, C.; Wang, L.; Zhao, W.J.; Cardini, F.; Kronenberg, F.; Gui, S.Q.; Ying, Z.; Zhao, N.Q.; Chao, M.T.; Yu, J. Acupuncture point injection treatment of primary dysmenorrhoea: A randomised, double blind, controlled study. BMJ Open 2016, 6, e008166. [Google Scholar] [CrossRef]
- Fujiwara, T.; Ono, M.; Mieda, M.; Yoshikawa, H.; Nakata, R.; Daikoku, T.; Sekizuka-Kagami, N.; Maida, Y.; Ando, H.; Fujiwara, H. Adolescent dietary habit-induced obstetric and gynecologic disease (ADHOGD) as a new hypothesis-possible involvement of clock system. Nutrients 2020, 12, 1294. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciebiera, M.; Esfandyari, S.; Siblini, H.; Prince, L.; Elkafas, H.; Wojtyła, C.; Al-Hendy, A.; Ali, M. Nutrition in Gynecological Diseases: Current Perspectives. Nutrients 2021, 13, 1178. https://doi.org/10.3390/nu13041178
Ciebiera M, Esfandyari S, Siblini H, Prince L, Elkafas H, Wojtyła C, Al-Hendy A, Ali M. Nutrition in Gynecological Diseases: Current Perspectives. Nutrients. 2021; 13(4):1178. https://doi.org/10.3390/nu13041178
Chicago/Turabian StyleCiebiera, Michał, Sahar Esfandyari, Hiba Siblini, Lillian Prince, Hoda Elkafas, Cezary Wojtyła, Ayman Al-Hendy, and Mohamed Ali. 2021. "Nutrition in Gynecological Diseases: Current Perspectives" Nutrients 13, no. 4: 1178. https://doi.org/10.3390/nu13041178
APA StyleCiebiera, M., Esfandyari, S., Siblini, H., Prince, L., Elkafas, H., Wojtyła, C., Al-Hendy, A., & Ali, M. (2021). Nutrition in Gynecological Diseases: Current Perspectives. Nutrients, 13(4), 1178. https://doi.org/10.3390/nu13041178






