Development of an Affordable, Sustainable and Efficacious Plant-Based Immunomodulatory Food Ingredient Based on Bell Pepper or Carrot RG-I Pectic Polysaccharides
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Extraction of Polysaccharide Rich Extracts
2.2.1. Lab-Scale Preparation of Bell Pepper and Carrot Polysaccharide Extracts for Activity-Guided Analyses
2.2.2. Industrial Scale Extraction of Bell Pepper RG-I and Carrot RG-I
2.3. Chemical Composition and Structure
2.3.1. Preparation of Subfractions Using Diethylaminoethyl (DEAE) Anion-Exchange Chromatography (Neutral, Moderately Acidic and Highly Acidic Subfractions)
2.3.2. Determination of Molecular Weight (MW) by Superdex 200 Size Exclusion Chromatography (HP-SEC)
2.3.3. Monosaccharide Composition Analysis
2.3.4. 1H-NMR
2.4. In Vitro Biological Function Assays
2.4.1. Immune Function
Phagocytosis Activity
Cytokine Secretion in Peripheral Blood Mononuclear Cells (PBMC) and Whole Blood from Healthy Donors
2.4.2. Microbiota Modulation
Colonic Batch Incubations
Microbial Metabolic Activity
Microbial Community Composition
2.5. Human Randomized, Double-Blind, Placebo-Controlled Proof of Concept Study
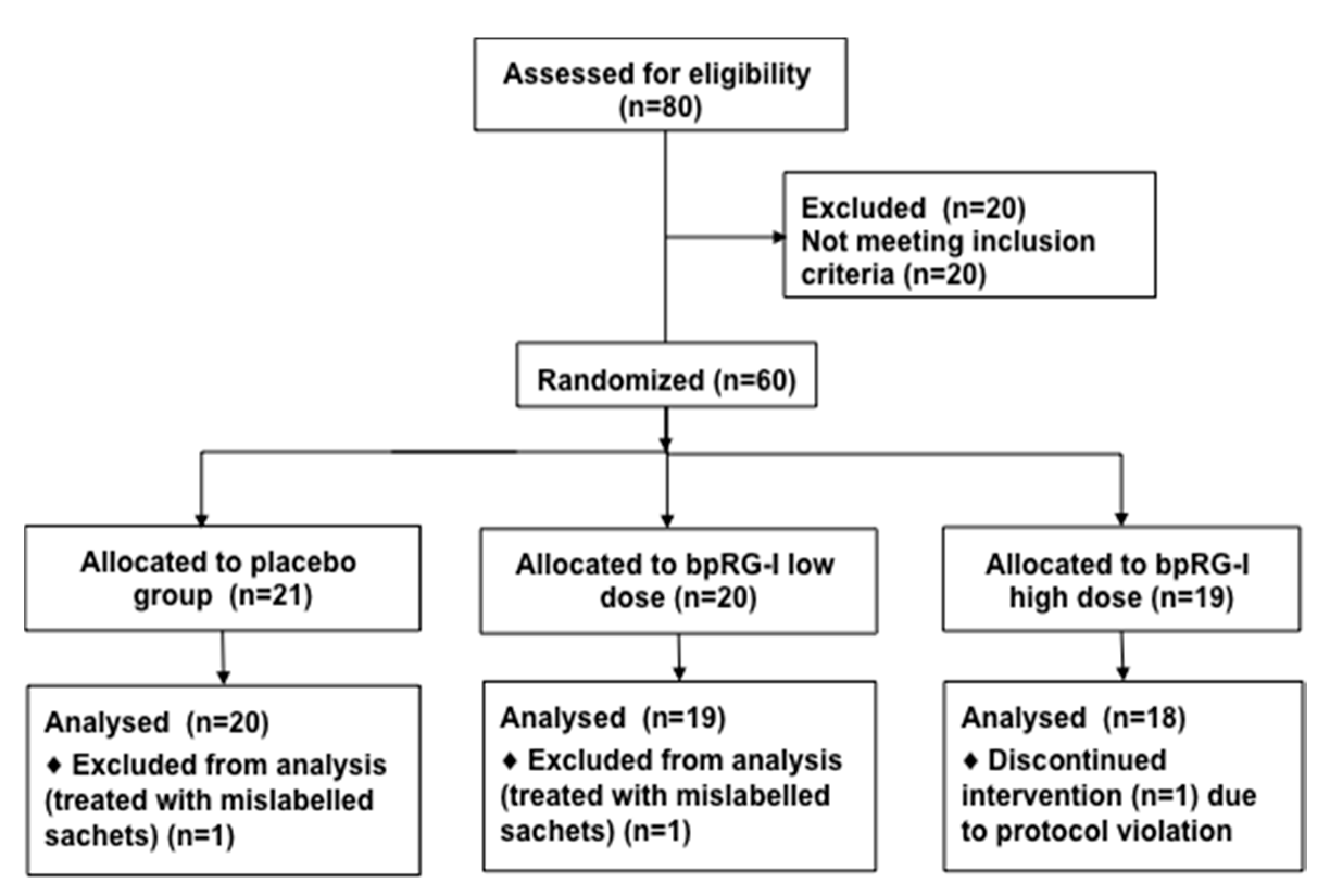
2.5.1. Participants and Study Design
2.5.2. Immune Function
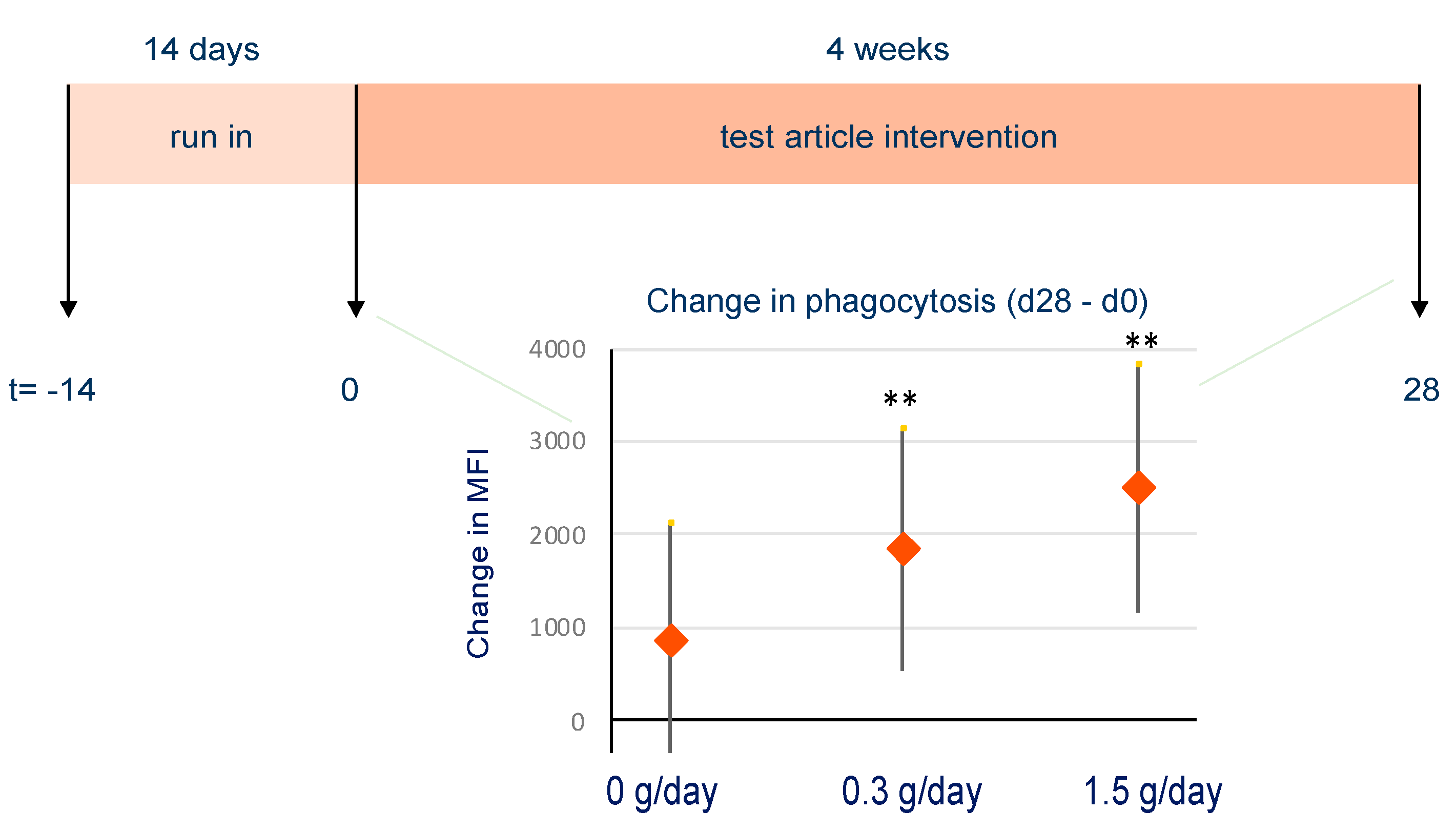
Phagocytosis Activity
Natural Killer (NK) Cell Activity
2.6. Statistics
3. Results
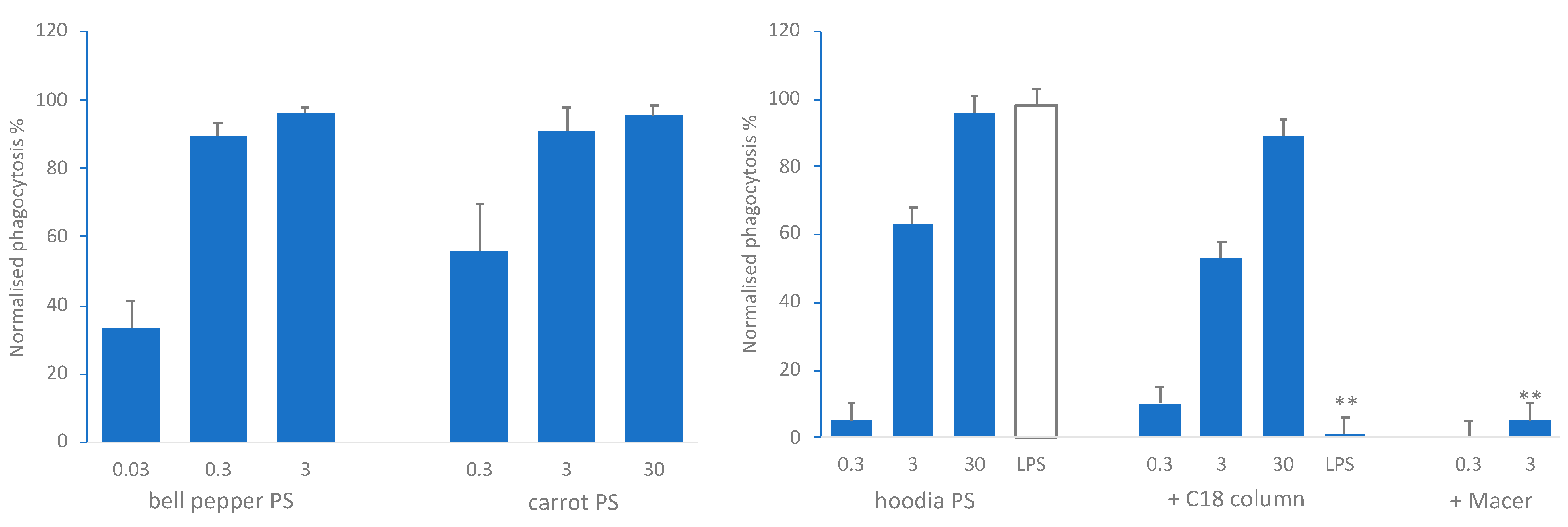
3.1. Polysaccharide Extracts Stimulate Immune Function In Vitro
3.2. Activity-Guided Identification of the Immunomodulatory Nutricophore from Hoodia, Bell Pepper and Carrot Polysaccharide-Enriched Hot Water Extracts
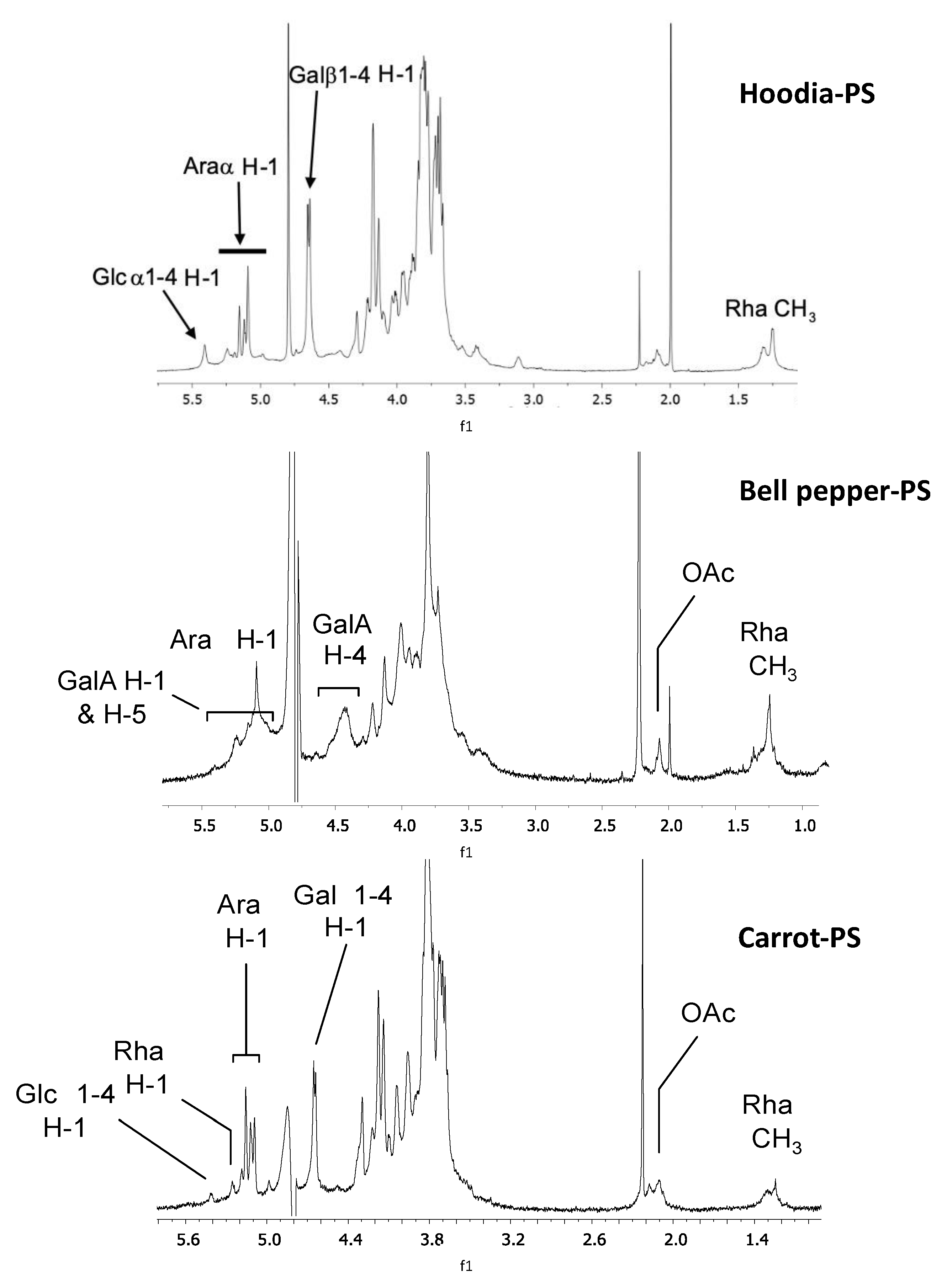
3.2.1. Characterization of Polysaccharides Extracts by 1H-NMR
3.2.2. Effect of Charge and Molecular Size on In Vitro Phagocytosis Activity
3.2.3. Effect of pH on Extraction of Bell Pepper and Carrot Polysaccharides
3.3. Monosaccharide Composition Analysis of Bell Pepper and Carrot
3.3.1. Lab-Scale Polysaccharide-Enriched Hot Water Extracts
3.3.2. Industrial Scale RG-I-Enriched Extracts
3.4. Functional/Biological Activity of bpRG-I and cRG-I In Vitro
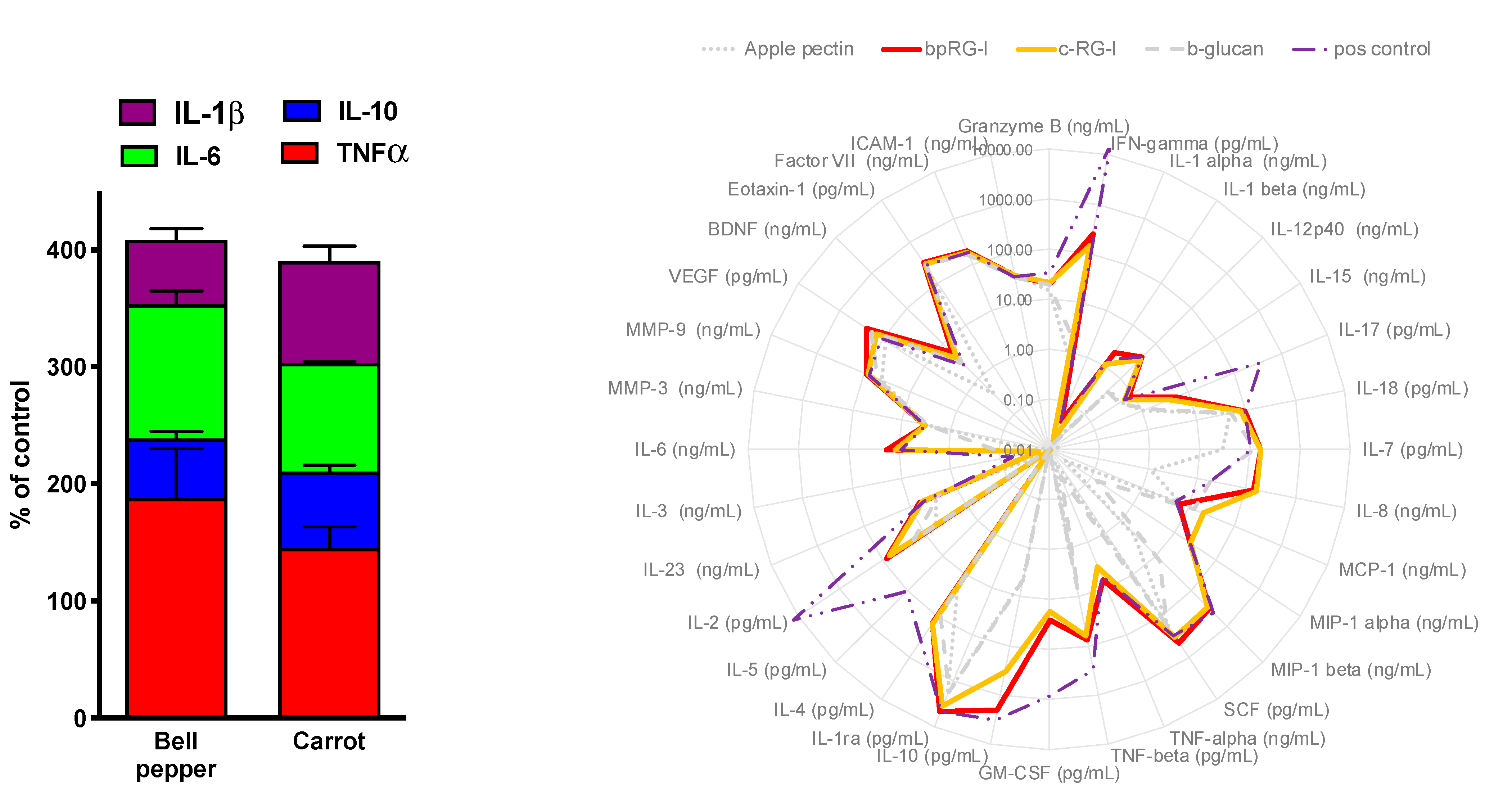
3.4.1. In Vitro Cytokine Secretion—PBMC and Whole Blood Assay
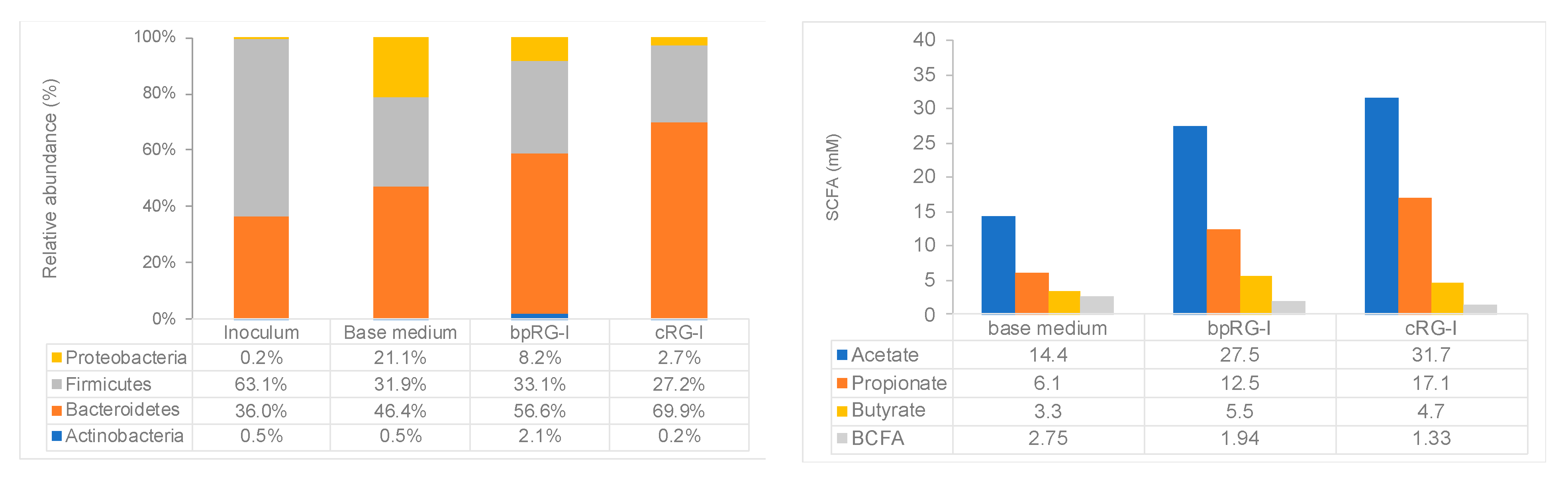
3.4.2. Effect of bpRG-I and cRG-I on Gut Microbiota In Vitro
3.5. Immunomodulatory Activity and Safety of bpRG-I in Human Proof of a Concept Study
3.5.1. Study Subjects
3.5.2. Safety and Tolerability
3.5.3. Effect of bpRG-I on Innate Immune Responsiveness and Fecal Microbiota Composition
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Forum of International Respiratory Societies. The Global Impact of Respiratory Disease, 2nd ed.; European Respiratory Society: Sheffield, UK, 2017. [Google Scholar]
- Jartti, T.; Gern, J.E. Role of Viral Infections in the Development and Exacerbation of Asthma in Children. J. Allergy Clin. Immun. 2017, 140, 895–906. [Google Scholar] [CrossRef]
- Rothberg, M.B.; Haessler, S.D.; Brown, R.B. Complications of Viral Influenza. Am. J. Med. 2008, 121, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Seida, J.K.; Durec, T.; Kuhle, S. North American (Panax Quinquefolius) and Asian Ginseng (Panax Ginseng) Preparations for Prevention of the Common Cold in Healthy Adults: A Systematic Review. Evid-Based Compl Alt. 2011, 2011, 282151. [Google Scholar] [CrossRef]
- Shah, S.A.; Sander, S.; White, C.M.; Rinaldi, M.; Coleman, C.I. Evaluation of Echinacea for the Prevention and Treatment of the Common Cold: A Meta-Analysis. Lancet Infect. Dis. 2007, 7, 473–480. [Google Scholar] [CrossRef]
- Poolsup, N.; Suthisisang, C.; Prathanturarug, S.; Asawamekin, A.; Chanchareon, U. Andrographis Paniculata in the Symptomatic Treatment of Uncomplicated Upper Respiratory Tract Infection: Systematic Review of Randomized Controlled Trials. J. Clin. Pharm. Ther. 2004, 29, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Philander, L.A. An Ethnobotany of Western Cape Rasta Bush Medicine. J. Ethnopharmacol. 2011, 138, 578–594. [Google Scholar] [CrossRef] [PubMed]
- Scaglione, F.; Ferrara, F.; Dugnani, S.; Falchi, M.; Santoro, G.; Fraschini, F. Immunomodulatory Effects of Two Extracts of Panax Ginseng C.A. Meyer. Drugs Exp. Clin. Res. 1990, XVI, 537–542. [Google Scholar]
- Scaglione, F.; Cogo, R.; Cocuzza, C.; Arcidiacono, M.; Beretta, A. Immunomodulatory Effects of Panax Ginseng C.A. Meyer (G115) on Alveolar Macrophages from Patients Suffering with Chronic Bronchitis. Int. J. Immunther. 1994, 10, 21–24. [Google Scholar]
- Scaglione, F.; Cattaneo, G.; Alessandria, M.; Cogo, R. Efficacy and Safety of the Standardized Ginseng Extract G115 for Potentiating Vaccination against Common Cold and/or Influenza Syndrome. Drugs Exptl. Clin. Res. 1996, XXII, 65–72. [Google Scholar]
- Predy, G.N.; Goel, V.; Lovlin, R.E.; Basu, T.K. Immune Modulating Effects of Daily Supplementation of COLD-FX (a Proprietary Extract of North American Ginseng) in Healthy Adults. J. Clin. Biochem. Nutr. 2006, 39, 162–167. [Google Scholar] [CrossRef][Green Version]
- Enaud, R.; Prevel, R.; Ciarlo, E.; Beaufils, F.; Wieërs, G.; Guery, B.; Delhaes, L. The Gut-Lung Axis in Health and Respiratory Diseases: A Place for Inter-Organ and Inter-Kingdom Crosstalks. Front. Cell Infect. Microbiol. 2020, 10, 9. [Google Scholar] [CrossRef]
- McElhaney, J.E.; Gravenstein, S.; Cole, S.K.; Davidson, E.; O’neill, D.; Petitjean, S.; Rumble, B.; Shan, J.J. A Placebo-Controlled Trial of a Proprietary Extract of North American Ginseng (CVT-E002) to Prevent Acute Respiratory Illness in Institutionalized Older Adults. J. Am. Geriatr. Soc. 2004, 52, 13–19. [Google Scholar] [CrossRef] [PubMed]
- McElhaney, J.E.; Simor, A.E.; McNeil, S.; Predy, G.N. Efficacy and Safety of CVT-E002, a Proprietary Extract of Panax Quinquefolius in the Prevention of Respiratory Infections in Influenza-Vaccinated Community-Dwelling Adults: A Multicenter, Randomized, Double-Blind, and Placebo-Controlled Trial. Influenza Res. Treat. 2011, 2011, 759051. [Google Scholar] [CrossRef] [PubMed]
- Predy, G.N.; Goel, V.; Lovlin, R.; Donner, A.; Stitt, L.; Basu, T.K. Efficacy of an Extract of North American Ginseng Containing Poly-Furanosyl-Pyranosyl-Saccharides for Preventing Upper Respiratory Tract Infections: A Randomized Controlled Trial. Can. Med. Assoc. J. 2005, 173, 1043–1048. [Google Scholar] [CrossRef]
- Weiss, R.F. Infuenza and Colds: Non-specific enhancement of resistance. In Weiss’s Herbal Medicine; Weiss, R.F., Ed.; Thieme: New York, NY, USA, 2001; pp. 225–233. [Google Scholar]
- Wu, D.; Zheng, J.; Mao, G.; Hu, W.; Ye, X.; Linhardt, R.J.; Chen, S. Rethinking the Impact of RG-I Mainly from Fruits and Vegetables on Dietary Health. Crit. Rev. Food Sci. 2019, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Mao, G.; Wu, D.; Wei, C.; Tao, W.; Ye, X.; Linhardt, R.J.; Orfila, C.; Chen, S. Reconsidering Conventional and Innovative Methods for Pectin Extraction from Fruit and Vegetable Waste: Targeting Rhamnogalacturonan I. Trends Food Sci. Tech. 2019, 94, 65–78. [Google Scholar] [CrossRef]
- Bermudez-Brito, M.; Sahasrabudhe, N.M.; Rösch, C.; Schols, H.A.; Faas, M.M.; de Vos, P. The Impact of Dietary Fibers on Dendritic Cell Responses in Vitro Is Dependent on the Differential Effects of the Fibers on Intestinal Epithelial Cells. Mol. Nutr. Food Res. 2015, 59, 698–710. [Google Scholar] [CrossRef] [PubMed]
- Riaz, M.; Rahman, N.U.; Zia-Ul-Haq, M.; Jaffar, H.Z.E.; Manea, R. Ginseng: A Dietary Supplement as Immune-Modulator in Various Diseases. Trends Food Sci. Tech. 2018, 83, 12–30. [Google Scholar] [CrossRef]
- Tang, C.; Ding, R.; Sun, J.; Liu, J.; Kan, J.; Jin, C. The Impacts of Natural Polysaccharides on Intestinal Microbiota and Immune Responses—A Review. Food Funct. 2019, 10, 2290–2312. [Google Scholar] [CrossRef]
- Singh, R.P.; Bashir, H.; Kumar, R. Emerging Role of Microbiota in Immunomodulation and Cancer Immunotherapy. Semin. Cancer Biol. 2020. [Google Scholar] [CrossRef]
- Beukema, M.; Faas, M.M.; Vos, P. de The Effects of Different Dietary Fiber Pectin Structures on the Gastrointestinal Immune Barrier: Impact via Gut Microbiota and Direct Effects on Immune Cells. Exp. Mol. Med. 2020, 52, 1364–1376. [Google Scholar] [CrossRef]
- Ferreira, S.S.; Passos, C.P.; Madureira, P.; Vilanova, M.; Coimbra, M.A. Structure–Function Relationships of Immunostimulatory Polysaccharides: A Review. Carbohydr. Polym. 2015, 132, 378–396. [Google Scholar] [CrossRef]
- Zhao, B.; Lv, C.; Lu, J. Natural Occurring Polysaccharides from Panax Ginseng C. A. Meyer: A Review of Isolation, Structures, and Bioactivities. Int. J. Biol. Macromol. 2019, 133, 324–336. [Google Scholar] [CrossRef]
- Maria-Ferreira, D.; Nascimento, A.M.; Cipriani, T.R.; Santana-Filho, A.P.; da Watanabe, P.S.; de Sant’Ana, D.M.G.; Luciano, F.B.; Bocate, K.C.P.; van den Wijngaard, R.M.; de Werner, M.F.P.; et al. Rhamnogalacturonan, a Chemically-Defined Polysaccharide, Improves Intestinal Barrier Function in DSS-Induced Colitis in Mice and Human Caco-2 Cells. Sci. Rep. UK 2018, 8, 12261. [Google Scholar] [CrossRef] [PubMed]
- Ruiter, G.A.D.; Schols, H.A.; Voragen, A.G.J.; Rombouts, F.M. Carbohydrate Analysis of Water-Soluble Uronic Acid-Containing Polysaccharides with High-Performance Anion-Exchange Chromatography Using Methanolysis Combined with TFA Hydrolysis Is Superior to Four Other Methods. Anal. Biochem. 1992, 207, 176–185. [Google Scholar] [CrossRef]
- Ahmed, A.E.R.; Labavitch, J.M. A Simplified Method for Accurate Determination of Cell Wall Uronide Content. J. Food Biochem. 1977, 1, 361–365. [Google Scholar] [CrossRef]
- Huisman, M.M.H.; Fransen, C.T.M.; Kamerling, J.P.; Vliegenthart, J.F.G.; Schols, H.A.; Voragen, A.G.J. The CDTA-soluble Pectic Substances from Soybean Meal Are Composed of Rhamnogalacturonan and Xylogalacturonan but Not Homogalacturonan. Biopolymers 2001, 58, 279–294. [Google Scholar] [CrossRef][Green Version]
- Perrone, P.; Hewage, C.M.; Thomson, A.R.; Bailey, K.; Sadler, I.H.; Fry, S.C. Patterns of Methyl and O-Acetyl Esterification in Spinach Pectins New Complexity. Phytochemistry 2002, 60, 67–77. [Google Scholar] [CrossRef]
- Den Abbeele, P.V.; Taminiau, B.; Pinheiro, I.; Duysburgh, C.; Jacobs, H.; Pijls, L.; Marzorati, M. Arabinoxylo-Oligosaccharides and Inulin Impact Inter-Individual Variation on Microbial Metabolism and Composition, Which Immunomodulates Human Cells. J. Agric. Food Chem. 2018, 66, 1121–1130. [Google Scholar] [CrossRef]
- Weirdt, R.D.; Possemiers, S.; Vermeulen, G.; Moerdijk-Poortvliet, T.C.W.; Boschker, H.T.S.; Verstraete, W.; de Wiele, T.V. Human Faecal Microbiota Display Variable Patterns of Glycerol Metabolism. FEMS Microbiol. Ecol. 2010, 74, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Props, R.; Kerckhof, F.-M.; Rubbens, P.; Vrieze, J.D.; Sanabria, E.H.; Waegeman, W.; Monsieurs, P.; Hammes, F.; Boon, N. Absolute Quantification of Microbial Taxon Abundances. ISME J. 2017, 11, 584–587. [Google Scholar] [CrossRef]
- Gandek, B.; Ware, J.E.; Aaronson, N.K.; Apolone, G.; Bjorner, J.B.; Brazier, J.E.; Bullinger, M.; Kaasa, S.; Leplege, A.; Prieto, L.; et al. Cross-Validation of Item Selection and Scoring for the SF-12 Health Survey in Nine Countries Results from the IQOLA Project. J. Clin. Epidemiol. 1998, 51, 1171–1178. [Google Scholar] [CrossRef]
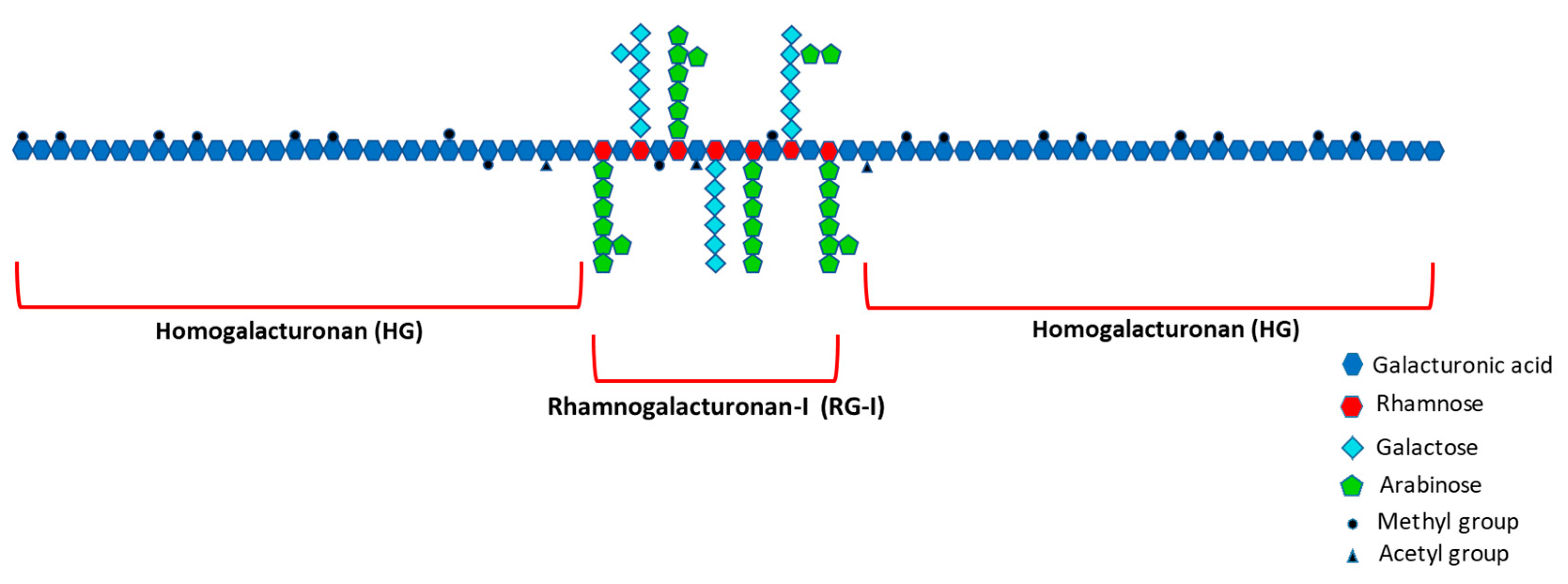
- Ridley, B.L.; O’Neill, M.A.; Mohnen, D. Pectins: Structure, Biosynthesis, and Oligogalacturonide-Related Signaling. Phytochemistry 2001, 57, 929–967. [Google Scholar] [CrossRef]
- Mohnen, D. Comprehensive Natural Products Chemistry; Barton, D., Meth-Cohn, O., Eds.; Elsevier Science Ltd.: Amsterdam, The Netherlands, 1999; ISBN 9780080912837. [Google Scholar]
- Mohnen, D. Pectin Structure and Biosynthesis. Curr. Opin. Plant Biol. 2008, 11, 266–277. [Google Scholar] [CrossRef]
- Harholt, J.; Suttangkakul, A.; Scheller, H.V. Biosynthesis of Pectin. Plant Physiol. 2010, 153, 384–395. [Google Scholar] [CrossRef]
- Coenen, G.J.; Bakx, E.J.; Verhoef, R.P.; Schols, H.A.; Voragen, A.G.J. Identification of the Connecting Linkage between Homo- or Xylogalacturonan and Rhamnogalacturonan Type I. Carbohydr. Polym. 2007, 70, 224–235. [Google Scholar] [CrossRef]
- Paulsen, B.S.; Barsett, H. Bioactive Pectic Polysaccharides. In Polysaccharides I; Heinze, T., Ed.; Springer: New York, NY, USA, 2005; ISBN 978-3-540-31583-4. [Google Scholar]
- Albersheim, P.; Neukom, H.; Deuel, H. Splitting of Pectin Chain Molecules in Neutral Solutions. Arch. Biochem. Biophys. 1960, 90, 46–51. [Google Scholar] [CrossRef]
- Helin, J.; Koek, J.H. Method for Isolation of Polysaccharides. Patent WO2012/148277, 1 November 2012. [Google Scholar]
- Jin, M.-Y.; Li, M.-Y.; Huang, R.-M.; Wu, X.-Y.; Sun, Y.-M.; Xu, Z.-L. Structural Features and Anti-Inflammatory Properties of Pectic Polysaccharides: A Review. Trends Food Sci. Technol. 2020, 107, 284–298. [Google Scholar] [CrossRef]
- Karboune, S.; Khodaei, N. Structures, Isolation and Health-Promoting Properties of Pectic Polysaccharides from Cell Wall-Rich Food by-Products: A Source of Functional Ingredients. Curr. Opin. Food Sci. 2016, 8, 50–55. [Google Scholar] [CrossRef]
- Den Abbeele, P.V.; Verstrepen, L.; Ghyselinck, J.; Albers, R.; Marzorati, M.; Mercenier, A. A Novel Non-Digestible, Carrot-Derived Polysaccharide (CRG-I) Selectively Modulates the Human Gut Microbiota While Promoting Gut Barrier Integrity: An Integrated In Vitro Approach. Nutrients 2020, 12, 1917. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yu, L.; Bi, H.; Li, X.; Ni, W.; Han, H.; Li, N.; Wang, B.; Zhou, Y.; Tai, G. Total Fractionation and Characterization of the Water-Soluble Polysaccharides Isolated from Panax Ginseng C. A. Meyer. Carbohydr. Polym. 2009, 77, 544–552. [Google Scholar] [CrossRef]
- Cho, Y.-J.; Son, H.-J.; Kim, K.-S. A 14-Week Randomized, Placebo-Controlled, Double-Blind Clinical Trial to Evaluate the Efficacy and Safety of Ginseng Polysaccharide (Y-75). J. Transl. Med. 2014, 12, 283. [Google Scholar] [CrossRef]
- McElhaney, J.; Goel, V.; Toane, B.; Hooten, J.; Shan, J. Efficacy of COLD-FX in the Prevention of Respiratory Symptoms in Community-Dwelling Adults: A Randomized, Double-Blinded, Placebo Controlled Trial. J. Altern. Complement. Med. 2006. [Google Scholar] [CrossRef]
- Zhang, X.; Li, S.; Sun, L.; Ji, L.; Zhu, J.; Fan, Y.; Tai, G.; Zhou, Y. Further Analysis of the Structure and Immunological Activity of an RG-I Type Pectin from Panax Ginseng. Carbohydr. Polym. 2012, 89, 519–525. [Google Scholar] [CrossRef]
- Beukema, M.; Jermendi, É.; van den Berg, M.A.; Faas, M.M.; Schols, H.A.; de Vos, P. The Impact of the Level and Distribution of Methyl-Esters of Pectins on TLR2-1 Dependent Anti-Inflammatory Responses. Carbohydr. Polym. 2021, 251, 117093. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Kim, Y.; Lee, H.; Kim, J.; Han, B.K.; Go, E.; Kwon, S.; Kang, J.-G.; You, S.; Kwon, J. Carrot Pomace Polysaccharide (CPP) Improves Influenza Vaccine Efficacy in Immunosuppressed Mice via Dendritic Cell Activation. Nutrients 2020, 12, 2740. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; In, G.; Han, S.-T.; Lee, M.-H.; Lee, J.-W.; Shin, K.-S. Structural Characteristics of a Red Ginseng Acidic Polysaccharide Rhamnogalacturonan I with Immunostimulating Activity from Red Ginseng. J. Ginseng. Res. 2020, 44, 570–579. [Google Scholar] [CrossRef] [PubMed]
- Dang, A.T.; Marsland, B.J. Microbes, Metabolites, and the Gut–Lung Axis. Mucosal Immunol. 2019, 12, 843–850. [Google Scholar] [CrossRef]
- Koh, A.; De Vadder, F.; Kovatcheva-Datchary, P.; Bäckhed, F. From Dietary Fiber to Host Physiology: Short-Chain Fatty Acids as Key Bacterial Metabolites. Cell 2016, 165, 1332–1345. [Google Scholar] [CrossRef]
- Netea, M.G.; Domínguez-Andrés, J.; Barreiro, L.B.; Chavakis, T.; Divangahi, M.; Fuchs, E.; Joosten, L.A.B.; van der Meer, J.W.M.; Mhlanga, M.M.; Mulder, W.J.M.; et al. Defining Trained Immunity and Its Role in Health and Disease. Nat. Rev. Immunol. 2020, 20, 375–388. [Google Scholar] [CrossRef]
- Gonçalves, P.; Araújo, J.R.; Santo, J.P.D. A Cross-Talk Between Microbiota-Derived Short-Chain Fatty Acids and the Host Mucosal Immune System Regulates Intestinal Homeostasis and Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2018, 24, 558–572. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.L.; Wesselingh, S.; Rogers, G.B. Host-microbiome Interactions in Acute and Chronic Respiratory Infections. Cell Microbiol. 2016, 18, 652–662. [Google Scholar] [CrossRef]
- Shinde, T.; Hansbro, P.M.; Sohal, S.S.; Dingle, P.; Eri, R.; Stanley, R. Microbiota Modulating Nutritional Approaches to Countering the Effects of Viral Respiratory Infections Including SARS-CoV-2 through Promoting Metabolic and Immune Fitness with Probiotics and Plant Bioactives. Microorganisms 2020, 8, 921. [Google Scholar] [CrossRef] [PubMed]
- Sencio, V.; Machado, M.G.; Trottein, F. The Lung–Gut Axis during Viral Respiratory Infections: The Impact of Gut Dysbiosis on Secondary Disease Outcomes. Mucosal Immunol. 2021, 1–9. [Google Scholar] [CrossRef]
| 1H-NMR Signal | Phagocytosis (Concentration) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Material MW Fraction | Rha 4-OH | GalA 1 | β1-4Gal 2 | Ara 2 | Rha 4-OH subst 3 | O-Ac 4 | GalA OCH3 5 | 3 μg/mL | 30 μg/mL |
| Hoodia-PS | + | ++ | |||||||
| >110 kD | 1 | 1 | 5 | 3 | 50% | 100% | n.d. | ++ | ++ |
| 70–110 kD | 1 | 6 | 6 | 1 | 30% | 10% | 46% | + | ++ |
| 40–70 kD | 1 | 8 | 5 | 2 | 25% | 7% | 40% | ± | + |
| Bell pepper-PS | ++ | ++ | |||||||
| >110 kD | 1 | 4 | n.d. | 3 | 20% | 8% | 40% | ++ | ++ |
| 70–110 kD | 1 | 9 | n.d. | 2 | 30% | 10% | 40% | ++ | ++ |
| 40–70 kD | 1 | 21 | n.d. | 1 | 25% | 4% | 40% | - | + |
| Carrot-PS | + | ++ | |||||||
| >110 kD | 1 | 1 | 12 | 11 | 55% | 150% | n.d. | + | ++ |
| 70–110 kD | 1 | 3 | 7 | 5 | 50% | 35% | 45% | - | + |
| 40–70 kD | 1 | 20 | 3 | 4 | 40% | 10% | 30% | - | - |
| Composition | Phagocytosis | |||||||
|---|---|---|---|---|---|---|---|---|
| Material | Extraction | GalA/Rha | Ara/Rha | Gal/Rha | GalA-meth | GalA-acet | 3 μg/mL | 30 μg/mL |
| bell pepper-PS | water | 6.9 | 4.4 | 0.0 | 50% | 20% | ++ | ++ |
| NaHCO3 | 5.3 | 0.0 | 0.0 | 0% | 0% | ++ | ++ | |
| carrot-PS | water | 10.9 | 11.8 | 5.3 | 60% | 20% | ++ | ++ |
| NaHCO3 | 7.6 | 0.0 | 5.3 | 0% | 0% | ++ | ++ | |
| Monosaccharides % (mol/mol) | Ratios and Regions | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rha | Ara | Gal | Fuc | Glc | Man | Xyl | GalA | GalA/Rha 1 | RG-I 2 | RG-I bb 3 | Ara/Rha 4 | Gal/Rha 5 | |
| Bell pepper-PS | 2.6 | 4.0 | 5.6 | 0.3 | 1.9 | 1.0 | 1.7 | 83 | 32 | 15 | 5.2 | 1.5 | 2.2 |
| Carrot-PS | 2.4 | 5.6 | 14.5 | 0.8 | 8.7 | 0.1 | 0.0 | 68 | 28 | 25 | 4.8 | 2.3 | 6.0 |
| Monosaccharides % (mol/mol) | Ratios and Regions | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rha | Ara | Gal | Fuc | Glc | Man | Xyl | GalA | GalA/Rha 1 | RG-I 2 | RG-I bb 3 | Ara/Rha 4 | Gal/Rha 5 | |
| bpRG-I | 2.8 | 6.2 | 6.9 | 0.2 | 28.2 | 1.2 | 1.2 | 53 | 19 | 19 | 5.6 | 2.2 | 2.5 |
| cRG-I | 9.6 | 33.9 | 21.0 | 0.7 | 6.1 | 0.8 | 0.8 | 27 | 3 | 74 | 19.2 | 3.5 | 2.2 |
| bpRG-I6 | 4.6 | 7.5 | 8.0 | 0.6 | 29.0 | 1.9 | 1.5 | 47 | 10 | 25 | 9.2 | 1.6 | 1.7 |
| cRG-I6 | 14.3 | 34.8 | 19.6 | 0.8 | 4.3 | 0.9 | 0.7 | 25 | 2 | 83 | 28.6 | 2.4 | 1.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McKay, S.; Oranje, P.; Helin, J.; Koek, J.H.; Kreijveld, E.; van den Abbeele, P.; Pohl, U.; Bothe, G.; Tzoumaki, M.; Aparicio-Vergara, M.; et al. Development of an Affordable, Sustainable and Efficacious Plant-Based Immunomodulatory Food Ingredient Based on Bell Pepper or Carrot RG-I Pectic Polysaccharides. Nutrients 2021, 13, 963. https://doi.org/10.3390/nu13030963
McKay S, Oranje P, Helin J, Koek JH, Kreijveld E, van den Abbeele P, Pohl U, Bothe G, Tzoumaki M, Aparicio-Vergara M, et al. Development of an Affordable, Sustainable and Efficacious Plant-Based Immunomodulatory Food Ingredient Based on Bell Pepper or Carrot RG-I Pectic Polysaccharides. Nutrients. 2021; 13(3):963. https://doi.org/10.3390/nu13030963
Chicago/Turabian StyleMcKay, Sue, Paul Oranje, Jari Helin, Jean H. Koek, Ellen Kreijveld, Pieter van den Abbeele, Ute Pohl, Gordana Bothe, Maria Tzoumaki, Marcela Aparicio-Vergara, and et al. 2021. "Development of an Affordable, Sustainable and Efficacious Plant-Based Immunomodulatory Food Ingredient Based on Bell Pepper or Carrot RG-I Pectic Polysaccharides" Nutrients 13, no. 3: 963. https://doi.org/10.3390/nu13030963
APA StyleMcKay, S., Oranje, P., Helin, J., Koek, J. H., Kreijveld, E., van den Abbeele, P., Pohl, U., Bothe, G., Tzoumaki, M., Aparicio-Vergara, M., Mercenier, A., Schols, H., & Albers, R. (2021). Development of an Affordable, Sustainable and Efficacious Plant-Based Immunomodulatory Food Ingredient Based on Bell Pepper or Carrot RG-I Pectic Polysaccharides. Nutrients, 13(3), 963. https://doi.org/10.3390/nu13030963






