Dietary Management of Heart Failure: DASH Diet and Precision Nutrition Perspectives
Abstract
:1. Introduction
2. Methods
3. Heart Failure Overview
3.1. Heart Failure
3.2. Prevalence and Importance of Interventions
4. Current Cardiovascular Disease Risk Reduction Recommendations
5. Dietary Approaches for Heart Failure Management
5.1. Sodium and Fluid Restriction
5.2. Body Weight Management
5.2.1. Overweight, Obesity, and Underweight
5.2.2. Inadequate Intake and Malnutrition
5.3. Dietary Patterns
The DASH Diet
5.4. Nutritional Supplements and Treatments
6. Physiological Mechanisms of the DASH Diet in Heart Failure
6.1. Potential Mechanisms
6.2. Specificity and Efficacy Depending on Type of Heart Failure Diagnosis
6.3. Metabolic and Metabolomic Biomarkers
7. DASH Interventions in Heart Failure: Food/Nutrients and Education Interventions
7.1. Controlled Feeding Trials
7.2. Behavioral and Lifestyle Interventions
7.3. Behavior Change Strategies
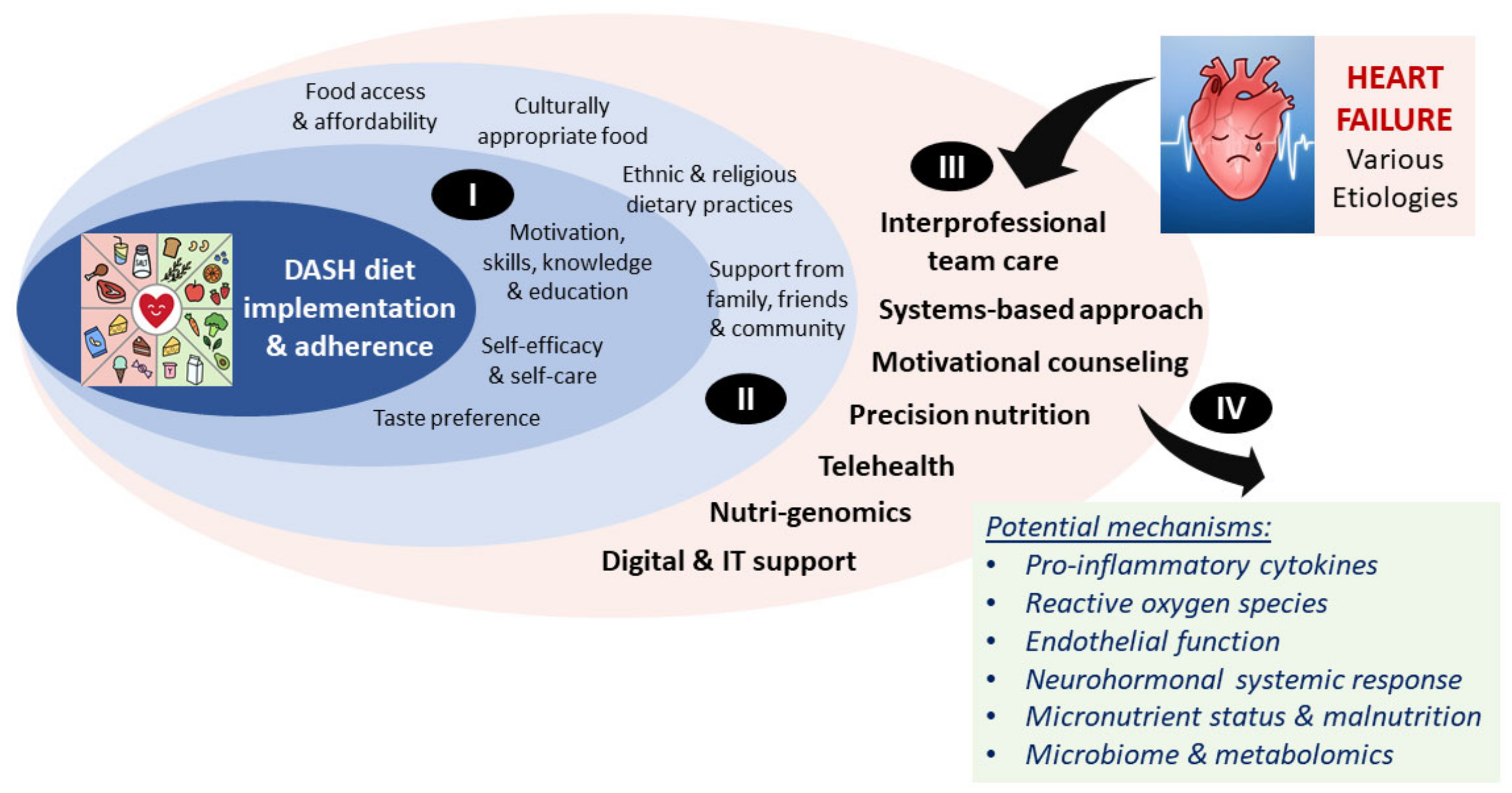
8. Implementation Analyses
9. Opportunities for Precision Nutrition and Health in Heart Failure
10. Summary and Recommendations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Study | Appel et al., NEJM 1997 [53] | Sacks et al., NEJM 2001 (Design: Svetkey et al., J Am Diet Assoc 1999) [21,22] | Lin et al., J Am Diet Assoc 2007 [97] | Hummel et al., Hypertension 2012 [94] |
|---|---|---|---|---|
| DASH diet name used in study | Combination diet | DASH diet at low, intermediate, and high sodium levels | Established recommendations + DASH diet (EST+DASH) | Sodium-restricted DASH diet (DASH/SRD) |
| Diet comparators | Control; Fruits-and-vegetables | Control at low, intermediate, and high sodium levels | Control; Established recommendations | None (DASH/SRD compared to baseline diet) |
| Energy level associated with provided nutrient analyses (kcal/d) | 2100 | 2100 | 1705 | 2040 |
| Macronutrients | ||||
| Fat (% of total kcal) | 26 | 26 | 26 | 29 |
| Saturated fat (% of total kcal) | 7 | 5 | 8 | Not available |
| Monounsaturated fat (% of total kcal) | 10 | 13 | 10 | Not available |
| Polyunsaturated fat (% of total kcal) | 7 | 8 | 6 | Not available |
| Carbohydrates (% of total kcal) | 57 | 56 | 57 | 58 |
| Protein (% of total kcal) | 18 | 18 | 18 | 19 |
| Cholesterol (mg/d) | 151 | 150 | 205 | 211 |
| Fiber (g/d) | Not available | 32 | 22 | 34 |
| Micronutrients | ||||
| Sodium (mg/d) | 2859 | 1150 or 2300 or 3450 | 2412 | 1450 |
| Potassium (mg/d) | 4415 | 4700 | 3256 | 4274 |
| Magnesium (mg/d) | 480 | 500 | 311 | Not available |
| Calcium (mg/d) | 1265 | 1250 | 907 | 1350 |
| Nutrient values based on: | Menu nutrient analyses | Menu nutrient targets described in study design publication | Mean intakes calculated from 24-h recall data at 18-month timepoint in intervention. Energy based on absolute intake (unadjusted) | Mean menu nutrient analyses from days 16-21 of the study |
| Intervention duration | 8 weeks | 30 days | 18 months | 3 weeks |
| Participants | N = 459, adults with systolic BP < 160 mmHg and diastolic BP ranging 80–95 mmHg, 225 females and 234 males | N = 412, adults with BP > 120/80 mmHg, ~57% females and ~43% males | N = 810, prehypertensive adults not taking antihypertensives, ~60% females and ~40% males | N = 14, treated hypertension, compensated HFpEF, 13 females and 1 male |
| Mean BP changes in DASH diet group compared to control (mmHg) | Ambulatory 24-h BP compared to baseline | |||
| Systolic (mmHg) | −5.5 | −5.9 high; −5.0 intermediate; −2.2 low | −4.6 at 6 months; −2.1 at 18 months | −7 |
| Diastolic (mmHg) | −3.0 | −2.9 high; −2.5 intermediate; −1.0 low | −2.1 at 6 months; −1.0 at 18 months | −5 |
Appendix B
| Study | Omic Platform | Bio-Sample | Metabolites | Associated Foods/Food Components |
|---|---|---|---|---|
| [83] | Targeted University of Michigan Metabolomics Center GC/MS + LC/MS | Serum (DASH style diet) | Short-chain acyl carnitine metabolites increased significantly, L-carnitine and propionyl carnitine correlated with improved left ventricular cardiac function | Results associated with dietary changes and energy utilization; no specific foods were discussed other than increased fiber intake |
| [77] | Untargeted Metabolon, Inc (Durham, NC) UHPLC/MS + GC/MS | Plasma (DASH-Sodium trial specimens) | Sodium intake significantly associated with changes in 6 metabolite pathways. Adjustment for change in diet pattern or blood pressure did not alter associations. Significant pathways (most increased with sodium restriction, while γ-glutamyl decreased with sodium restriction): Fatty acid Food component or plant group Benzoate metabolism Methionine metabolism Tryptophan metabolism γ-glutamyl amino acid | Greatest responder to reduced sodium intake was increase in 4-ethylephenylsulfate (Benzoate pathway) gut metabolite associated with soy intake; 4-allylpheol sulfate & homostachydrine (food component or plant group metabolites) may be related to herbs and seasonings added to low sodium diet to increase palatability; decrease in isovalerate (fatty acid pathway) may be gut microbiota changes related to salt intake |
| [76] | Untargeted Metabolon, Inc (Durham, NC) UHPLC/MS + GC/MS | Serum (original DASH trial specimens) | Multiple metabolites differed significantly DASH vs. Control (97) and DASH vs. fruit/veg (67); metabolites represented classes of lipids, amino acids, xenobiotics and food components, cofactors and vitamins, carbohydrates 10 candidate biomarkers for assessing adherence to DASH diet: N-methylproline Stachydrine (proline betaine) Tryptophan betaine Methyl glucopyranoside β-cryptoxanthin Theobromine 7-methylurate 3-methylxanthine 7-methlyxanthine Chiro-inositol | Citrus fruits (N-methylproline and stachydrine) Legumes (Tryptophan betaine) Cereals and cereal products (glucopyranoside) Total fruits (Methyl-β-glucopyranoside) Carotenoid-rich fruits and vegetables (β-cryptoxanthin) Caffeine and chocolate (theobromine, 7-methylurate, 3- and 7-methylxanthine) Fruit, beans, grains, nuts, seeds (chiro-inositol, phospholipids) |
| [81] | Untargeted University of Colorado RP HPLC/TOF MS, ESI negative and positive | Urine & food (DASH style trial) | Food specific compounds from DASH diet containing 12 specified foods were detected in urine along with other metabolites (13-190 metabolites per food); 16 urinary metabolites were significantly associated with BP and 6 with change in BP: 3-indolebutyric acid 1-beta-D-Ribofuranosyl)-1,4-dihydronicotinamide Kynuramine Physoperuvine 2 unidentified compounds (mass of 265.0971 and 157.0373) | 12 specific foods from a DASH diet and their metabolites were characterized in this study: Apples and apple juice, beef, blueberries, broccoli, chicken, coffee, cucumber, grapefruit, peanut butter, pork, tilapia |
| [79] | Untargeted West Coast Metabolomics Center, University of California Davis CHS LC/QTOF MS, ESI positive, ESI negative | Plasma (DASH-Sodium trial specimens, control diet only) | Baseline comparisons exhibited no differences, but 3 metabolites (2 lipids and one organic acid derivative) differed significantly in salt sensitive subjects with change in sodium intake: Tocolpherol alpha 2-ketoisocaprioic acid Citramalic acid | Salt sensitive individuals exhibit an altered metabolome on high salt intake, no specific foods or food components were investigated |
| [78] | Untargeted Metabolon, Inc (Durham, NC) RP UHPLC/MS ESI positive early and late, ESI negative, Hydrophilic UHPLC/MS ESI negative | Urine (DASH-Sodium trial specimens) | Identified additional novel urinary metabolites associated with DASH diet: N-methylglutamate 3,5-dihydroxybenzoic acid Phloroglucinol sulfate Galactonate Replicated 8 significant urine metabolites identified in serum of original DASH trial distinguishing DASH vs. Control; and 9 significant urine metabolites identical in DASH-high and low sodium diets. Most Influential DASH metabolites: N-methylproline Stachydrine (proline betaine) Chiro-inositol Theobromine Tryptophan betaine 3-methylxanthine | Plant foods (N-methylglutamate) Whole grains and cereals (3,5-dihydroxybenzoic acid) Beans and lentils (Phloroglucinol sulfate) Dairy, pulses and seeds (Galactonate) |
| [82] | Untargeted GC/MS Duke University | Plasma & urine (DASH style trial) | Urine—19 metabolites (phenolic acids and microbial metabolites) differed significantly DASH vs. Control; Plasma—8 metabolites differed significantly DASH vs. Control; Major classes included phenolics, amino acid, organooxygen compounds, and cofactors | Whole grains, fruits, vegetables, herbs (polyphenol and phenolic acid rich foods) Protein sources, legumes (amino acids, organooxygen, keto acid derivatives, quinolones) |
References
- Virani, S.S.; Alonso, A.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Delling, F.N.; et al. Heart Disease and Stroke Statistics—2020 Update: A Report from the American Heart Association. Circulation 2020, 141, e139–e596. [Google Scholar] [CrossRef] [PubMed]
- Dassanayaka, S.; Jones, S.P. Recent Developments in Heart Failure. Circ. Res. 2015, 117, e58–e63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colin-Ramirez, E.; McAlister, F.; Zheng, Y.; Sharma, S.; Ezekowitz, J.A. Changes in dietary intake and nutritional status associated with a significant reduction in sodium intake in patients with heart failure. A sub-analysis of the SODIUM-HF pilot study. Clin. Nutr. ESPEN 2016, 11, e26–e32. [Google Scholar] [CrossRef]
- Inamdar, A.A.; Inamdar, A.C. Heart Failure: Diagnosis, Management and Utilization. J. Clin. Med. 2016, 5, 62. [Google Scholar] [CrossRef]
- Bozkurt, B.; Coats, A.J.; Tsutsui, H.; Abdelhamid, C.M.; Adamopoulos, S.; Albert, N.; Anker, S.D.; Atherton, J.; Böhm, M.; Butler, J.; et al. Universal definition and classification of heart failure: A report of the Heart Failure Society of America, Heart Failure Association of the European Society of Cardiology, Japanese Heart Failure Society and Writing Committee of the Universal Definition of Heart Failure. Eur. J. Heart Fail. 2021, 23, 352–380. [Google Scholar] [CrossRef] [PubMed]
- Triposkiadis, F.; Butler, J.; Abboud, F.M.; Armstrong, P.W.; Adamopoulos, S.; Atherton, J.J.; Backs, J.; Bauersachs, J.; Burkhoff, D.; Bonow, R.O.; et al. The continuous heart failure spectrum: Moving beyond an ejection fraction classification. Eur. Heart J. 2019, 40, 2155–2163. [Google Scholar] [CrossRef]
- Weldy, C.S.; Ashley, E.A. Towards precision medicine in heart failure. Nat. Rev. Cardiol. 2021, 18, 745–762. [Google Scholar] [CrossRef]
- Hartupee, J.; Mann, D.L. Neurohormonal activation in heart failure with reduced ejection fraction. Nat. Rev. Cardiol. 2017, 14, 30–38. [Google Scholar] [CrossRef] [Green Version]
- Schwinger, R.H.G. Pathophysiology of heart failure. Cardiovasc. Diagn. Ther. 2021, 11, 263–276. [Google Scholar] [CrossRef]
- Mcmurray, J.; Petrie, M.C.; Murdoch, D.R.; Davie, A.P. Clinical epidemiology of heart failure: Public and private health burden. Eur. Heart J. 1998, 19, 9–16. [Google Scholar]
- Heidenreich, P.A.; Albert, N.M.; Allen, L.A.; Bluemke, D.A.; Butler, J.; Fonarow, G.C.; Ikonomidis, J.S.; Khavjou, O.; Konstam, M.A.; Maddox, T.M.; et al. Forecasting the Impact of Heart Failure in the United States. Circ. Heart Fail. 2013, 6, 606–619. [Google Scholar] [CrossRef] [Green Version]
- Urbich, M.; Globe, G.; Pantiri, K.; Heisen, M.; Bennison, C.; Wirtz, H.S.; Di Tanna, G.L. A Systematic Review of Medical Costs Associated with Heart Failure in the USA (2014–2020). PharmacoEconomics 2020, 38, 1219–1236. [Google Scholar] [CrossRef]
- Blecker, S.; Paul, M.; Taksler, G.; Ogedegbe, G.; Katz, S. Heart Failure–Associated Hospitalizations in the United States. J. Am. Coll. Cardiol. 2013, 61, 1259–1267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- U.S. Department of Agriculture; U.S. Department of Health and Human Services. Dietary Guidelines for Americans, 2020–2025, 9th ed.; USDA: Washington, DC, USA, 2020. Available online: https://www.dietaryguidelines.gov/sites/default/files/2020-12/Dietary_Guidelines_for_Americans_2020-2025.pdf (accessed on 18 January 2021).
- Salehi-Abargouei, A.; Maghsoudi, Z.; Shirani, F.; Azadbakht, L. Effects of Dietary Approaches to Stop Hypertension (DASH)-style diet on fatal or nonfatal cardiovascular diseases—Incidence: A systematic review and meta-analysis on observational prospective studies. Nutrition 2013, 29, 611–618. [Google Scholar] [CrossRef]
- Van Horn, L.; Carson, J.A.S.; Appel, L.J.; Burke, L.E.; Economos, C.; Karmally, W.; Lancaster, K.; Lichtenstein, A.H.; Johnson, R.K.; Thomas, R.J.; et al. Recommended Dietary Pattern to Achieve Adherence to the American Heart Association/American College of Cardiology (AHA/ACC) Guidelines: A Scientific Statement from the American Heart Association. Circulation 2016, 134, e505–e529. [Google Scholar] [CrossRef] [PubMed]
- Force, U.P.S.T.; Krist, A.H.; Davidson, K.W.; Mangione, C.M.; Barry, M.J.; Cabana, M.; Caughey, A.B.; Donahue, K.; Doubeni, C.A.; Epling, J.W.; et al. Behavioral Counseling Interventions to Promote a Healthy Diet and Physical Activity for Cardiovascular Disease Prevention in Adults with Cardiovascular Risk Factors. JAMA 2020, 324, 2069–2075. [Google Scholar] [CrossRef]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Bohm, M.; Burri, H.; Butler, J.; Celutkiene, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef]
- Butler, T. Dietary management of heart failure: Room for improvement? Br. J. Nutr. 2016, 115, 1202–1217. [Google Scholar] [CrossRef] [Green Version]
- Vest, A.R.; Chan, M.; Deswal, A.; Givertz, M.M.; Lekavich, C.; Lennie, T.; Litwin, S.E.; Parsly, L.; Rodgers, J.E.; Rich, M.W.; et al. Nutrition, Obesity, and Cachexia in Patients with Heart Failure: A Consensus Statement from the Heart Failure Society of America Scientific Statements Committee. J. Card. Fail. 2019, 25, 380–400. [Google Scholar] [CrossRef]
- Svetkey, L.P.; Sacks, F.M.; Obarzanek, E.; Vollmer, W.M.; Appel, L.J.; Lin, P.-H.; Karanja, N.M.; Harsha, D.W.; Bray, G.A.; Aickin, M.; et al. The DASH Diet, Sodium Intake and Blood Pressure Trial (DASH-Sodium): Rationale and Design. J. Am. Diet. Assoc. 1999, 99, S96–S104. [Google Scholar] [CrossRef]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R.; Simons-Morton, D.G.; et al. Effects on Blood Pressure of Reduced Dietary Sodium and the Dietary Approaches to Stop Hypertension (DASH) Diet. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef]
- Weinberger, M.H. Salt Sensitivity of Blood Pressure in Humans. Hypertension 1996, 27, 481–490. [Google Scholar] [CrossRef]
- Aliti, G.B.; Rabelo, E.R.; Clausell, N.; Rohde, L.E.; Biolo, A.; Beck-da-Silva, L. Aggressive fluid and sodium restriction in acute decompensated heart failure: A randomized clinical trial. JAMA Intern. Med. 2013, 173, 1058–1064. [Google Scholar] [CrossRef]
- Mahtani, K.R.; Heneghan, C.; Onakpoya, I.; Tierney, S.; Aronson, J.K.; Roberts, N.; Hobbs, F.D.R.; Nunan, D. Reduced Salt Intake for Heart Failure: A Systematic Review. JAMA Intern. Med. 2018, 178, 1693–1700. [Google Scholar] [CrossRef]
- Damgaard, M.; Norsk, P.; Gustafsson, F.; Kanters, J.K.; Christensen, N.J.; Bie, P.; Friberg, L.; Gadsbøll, N. Hemodynamic and neuroendocrine responses to changes in sodium intake in compensated heart failure. Am. J. Physiol. Integr. Comp. Physiol. 2006, 290, R1294–R1301. [Google Scholar] [CrossRef] [Green Version]
- Yancy, C.W.; Jessup, M.; Bozkurt, B.; Butler, J.; Casey, D.E.; Drazner, M.H.; Fonarow, G.C.; Geraci, S.A.; Horwich, T.; Januzzi, J.L.; et al. 2013 ACCF/AHA guideline for the management of heart failure: A report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation 2013, 128, e240–e327. [Google Scholar] [CrossRef] [PubMed]
- Johansson, P.; van der Wal, M.H.; Stromberg, A.; Waldreus, N.; Jaarsma, T. Fluid restriction in patients with heart failure: How should we think? Eur. J. Cardiovasc. Nurs. 2016, 15, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Dunlap, M.E.; Hauptman, P.J.; Amin, A.N.; Chase, S.L.; Chiodo, J.A.; Chiong, J.R.; Dasta, J.F. Current Management of Hyponatremia in Acute Heart Failure: A Report from the Hyponatremia Registry for Patients with Euvolemic and Hypervolemic Hyponatremia (HN Registry). J. Am. Heart Assoc. 2017, 6, e005261. [Google Scholar] [CrossRef]
- Alhafez, B.A.; Lavie, C.J.; Milani, R.V.; Ventura, H.O. Interactions of hypertension, obesity, left ventricular hypertrophy, and heart failure. Curr. Opin. Cardiol. 2021, 36, 453–460. [Google Scholar]
- Aggarwal, M.; Bozkurt, B.; Panjrath, G.; Aggarwal, B.; Ostfeld, R.J.; Barnard, N.D.; Gaggin, H.; Freeman, A.M.; Allen, K.; Madan, S.; et al. Lifestyle Modifications for Preventing and Treating Heart Failure. J. Am. Coll. Cardiol. 2018, 72, 2391–2405. [Google Scholar] [CrossRef]
- Ayinapudi, K.; Samson, R.; Le Jemtel, T.H.; Marrouche, N.F.; Oparil, S. Weight Reduction for Obesity-Induced Heart Failure with Preserved Ejection Fraction. Curr. Hypertens. Rep. 2020, 22, 47. [Google Scholar] [CrossRef] [PubMed]
- Shimada, Y.J.; Tsugawa, Y.; Brown, D.F.; Hasegawa, K. Bariatric Surgery and Emergency Department Visits and Hospitalizations for Heart Failure Exacerbation: Population-Based, Self-Controlled Series. J. Am. Coll. Cardiol. 2016, 67, 895–903. [Google Scholar] [CrossRef]
- Mikhalkova, D.; Holman, S.R.; Jiang, H.; Saghir, M.; Novak, E.; Coggan, A.R.; O’Connor, R.; Bashir, A.; Jamal, A.; Ory, D.S.; et al. Bariatric Surgery-Induced Cardiac and Lipidomic Changes in Obesity-Related Heart Failure with Preserved Ejection Fraction. Obesity 2017, 26, 284–290. [Google Scholar] [CrossRef] [Green Version]
- Evangelista, L.S.; Heber, D.; Li, Z.; Bowerman, S.; Hamilton, M.A.; Fonarow, G. Reduced Body Weight and Adiposity with a High-Protein Diet Improves Functional Status, Lipid Profiles, Glycemic Control, and Quality of Life in Patients with Heart Failure. J. Cardiovasc. Nurs. 2009, 24, 207–215. [Google Scholar] [CrossRef] [Green Version]
- Kitzman, D.W.; Brubaker, P.H.; Morgan, T.M.; Haykowsky, M.J.; Hundley, G.; Kraus, W.E.; Eggebeen, J.; Nicklas, B.J. Effect of Caloric Restriction or Aerobic Exercise Training on Peak Oxygen Consumption and Quality of Life in Obese Older Patients with Heart Failure with Preserved Ejection Fraction. JAMA 2016, 315, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, J.R.; Heidenreich, P. Obesity and survival in patients with heart failure and preserved systolic function: A U-shaped relationship. Am. Heart J. 2010, 159, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Padwal, R.; McAlister, F.; Mcmurray, J.; Cowie, M.; Rich, M.W.; Pocock, S.; Swedberg, K.; Maggioni, A.P.; Gamble, G.; Ariti, C.; et al. The obesity paradox in heart failure patients with preserved versus reduced ejection fraction: A meta-analysis of individual patient data. Int. J. Obes. 2013, 38, 1110–1114. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, R.; Stokes, M.; Elliott, A.; Munawar, D.A.; Khokhar, K.B.; Thiyagarajah, A.; Hendriks, J.; Linz, D.; Gallagher, C.; Kaye, D.; et al. Complex interaction of obesity, intentional weight loss and heart failure: A systematic review and meta-analysis. Heart 2019, 106, 58–68. [Google Scholar] [CrossRef]
- Vest, A.R.; Wu, Y.; Hachamovitch, R.; Young, J.B.; Cho, L. The Heart Failure Overweight/Obesity Survival Paradox. JACC Heart Fail. 2015, 3, 917–926. [Google Scholar] [CrossRef]
- Obata, Y.; Kakutani, N.; Kinugawa, S.; Fukushima, A.; Yokota, T.; Takada, S.; Ono, T.; Sota, T.; Kinugasa, Y.; Takahashi, M.; et al. Impact of Inadequate Calorie Intake on Mortality and Hospitalization in Stable Patients with Chronic Heart Failure. Nutrients 2021, 13, 874. [Google Scholar] [CrossRef]
- Wawrzeńczyk, A.; Anaszewicz, M.; Wawrzeńczyk, A.; Budzyński, J. Clinical significance of nutritional status in patients with chronic heart failure—A systematic review. Heart Fail. Rev. 2019, 24, 671–700. [Google Scholar] [CrossRef] [PubMed]
- Shan, Z.; Li, Y.; Baden, M.Y.; Bhupathiraju, S.N.; Wang, D.D.; Sun, Q.; Rexrode, K.M.; Rimm, E.B.; Qi, L.; Willett, W.C.; et al. Association Between Healthy Eating Patterns and Risk of Cardiovascular Disease. JAMA Intern. Med. 2020, 180, 1090–1100. [Google Scholar] [CrossRef]
- Kerley, C.P. Dietary patterns and components to prevent and treat heart failure: A comprehensive review of human studies. Nutr. Res. Rev. 2018, 32, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Sanches Machado d’Almeida, K.; Ronchi Spillere, S.; Zuchinali, P.; Corrêa Souza, G. Mediterranean Diet and Other Dietary Patterns in Primary Prevention of Heart Failure and Changes in Cardiac Function Markers: A Systematic Review. Nutrients 2018, 10, 58. [Google Scholar] [CrossRef] [Green Version]
- Lara, K.M.; Levitan, E.; Gutierrez, O.M.; Shikany, J.M.; Safford, M.M.; Judd, S.E.; Rosenson, R.S. Dietary Patterns and Incident Heart Failure in U.S. Adults Without Known Coronary Disease. J. Am. Coll. Cardiol. 2019, 73, 2036–2045. [Google Scholar] [CrossRef] [PubMed]
- Padilha, G.D.R.; D’Almeida, K.S.M.; Spillere, S.R.; Souza, G.C. Dietary Patterns in Secondary Prevention of Heart Failure: A Systematic Review. Nutrients 2018, 10, 828. [Google Scholar] [CrossRef] [Green Version]
- Levitan, E.B.; Lewis, C.E.; Tinker, L.F.; Eaton, C.B.; Ahmed, A.; Manson, J.E.; Snetselaar, L.G.; Martin, L.W.; Trevisan, M.; Howard, B.V.; et al. Mediterranean and DASH diet scores and mortality in women with heart failure: The Women’s Health Initiative. Circ. Heart Fail. 2013, 6, 1116–1123. [Google Scholar] [CrossRef] [Green Version]
- Papadaki, A.; Martinez-Gonzalez, M.A.; Alonso-Gómez, A.; Rekondo, J.; Salas-Salvadó, J.; Corella, D.; Ros, E.; Fitó, M.; Estruch, R.; Lapetra, J.; et al. Mediterranean diet and risk of heart failure: Results from the PREDIMED randomized controlled trial. Eur. J. Heart Fail. 2017, 19, 1179–1185, Erratum in 2019, 21, 389–391, doi:10.1002/ejhf.1346. [Google Scholar] [CrossRef]
- Kerley, C.P. A Review of Plant-based Diets to Prevent and Treat Heart Failure. Card. Fail. Rev. 2018, 4, 54–61. [Google Scholar] [CrossRef]
- Abu-Sawwa, R.; Dunbar, S.B.; Quyyumi, A.A.; Sattler, E.L. Nutrition intervention in heart failure: Should consumption of the DASH eating pattern be recommended to improve outcomes? Heart Fail. Rev. 2019, 24, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Burgermaster, M.; Rudel, R.; Seres, D. Dietary Sodium Restriction for Heart Failure: A Systematic Review of Intervention Outcomes and Behavioral Determinants. Am. J. Med. 2020, 133, 1391–1402. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A clinical trial of the effects of dietary patterns on blood pressure. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef] [Green Version]
- Campos, C.L.; Wood, L.; Burke, G.L.; Bahrami, H.; Bertoni, A.G. Dietary Approaches to Stop Hypertension Diet Concordance and Incident Heart Failure: The Multi-Ethnic Study of Atherosclerosis. Am. J. Prev. Med. 2019, 56, 819–826. [Google Scholar] [CrossRef]
- Goyal, P.; Balkan, L.; Ringel, J.B.; Hummel, S.L.; Sterling, M.R.; Kim, S.; Arora, P.; Jackson, E.A.; Brown, T.M.; Shikany, J.M.; et al. The Dietary Approaches to Stop Hypertension (DASH) Diet Pattern and Incident Heart Failure. J. Card. Fail. 2021, 27, 512–521. [Google Scholar] [CrossRef]
- Okonko, D.O.; Shah, A. Mitochondrial dysfunction and oxidative stress in CHF. Nat. Rev. Cardiol. 2014, 12, 6–8. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Khan, F.; Fonarow, G.C.; Sreenivasan, J.; Greene, S.J.; Khan, S.U.; Usman, M.S.; Vaduganathan, M.; Fudim, M.; Anker, S.D.; et al. Dietary interventions and nutritional supplements for heart failure: A systematic appraisal and evidence map. Eur. J. Heart Fail. 2021, 23, 1468–1476. [Google Scholar] [CrossRef]
- Mortensen, S.A.; Rosenfeldt, F.; Kumar, A.; Dolliner, P.; Filipiak, K.J.; Pella, D.; Alehagen, U.; Steurer, G.; Littarru, G.P.; Q-SYMBIO Study Investigators. The Effect of Coenzyme Q10 on Morbidity and Mortality in Chronic Heart Failure: Results From Q-SYMBIO: A Randomized Double-Blind Trial. JACC Heart Fail. 2014, 2, 641–649. [Google Scholar] [CrossRef]
- Di Lorenzo, A.; Iannuzzo, G.; Parlato, A.; Cuomo, G.; Testa, C.; Coppola, M.; D’Ambrosio, G.; Oliviero, D.A.; Sarullo, S.; Vitale, G.; et al. Clinical Evidence for Q10 Coenzyme Supplementation in Heart Failure: From Energetics to Functional Improvement. J. Clin. Med. 2020, 9, 1266. [Google Scholar] [CrossRef] [PubMed]
- Barbarawi, M.; Lakshman, H.; Barbarawi, O.; Alabdouh, A.; Al Kasasbeh, M.; Djousse, L.; Manson, J.E. Omega-3 supplementation and heart failure: A meta-analysis of 12 trials including 81,364 participants. Contemp. Clin. Trials 2021, 107, 106458. [Google Scholar] [CrossRef]
- Ponikowski, P.; Kirwan, B.-A.; Anker, S.D.; McDonagh, T.; Dorobantu, M.; Drozdz, J.; Fabien, V.; Filippatos, G.; Göhring, U.M.; Keren, A.; et al. Ferric carboxymaltose for iron deficiency at discharge after acute heart failure: A multicentre, double-blind, randomised, controlled trial. Lancet 2020, 396, 1895–1904. [Google Scholar] [CrossRef]
- Levitan, E.B.; Wolk, A.; Mittleman, M. Relation of Consistency with the Dietary Approaches to Stop Hypertension Diet and Incidence of Heart Failure in Men Aged 45 to 79 Years. Am. J. Cardiol. 2009, 104, 1416–1420. [Google Scholar] [CrossRef] [Green Version]
- Hummel, S.L.; Seymour, E.M.; Brook, R.D.; Sheth, S.S.; Ghosh, E.; Zhu, S.; Weder, A.B.; Kovács, S.J.; Kolias, T.J. Low-Sodium DASH Diet Improves Diastolic Function and Ventricular–Arterial Coupling in Hypertensive Heart Failure with Preserved Ejection Fraction. Circ. Heart Fail. 2013, 6, 1165–1171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiavaroli, L.; Viguiliouk, E.; Nishi, S.K.; Mejia, S.B.; Rahelić, D.; Kahleova, H.; Salas-Salvadó, J.; Kendall, C.W.C.; Sievenpiper, J.L. DASH Dietary Pattern and Cardiometabolic Outcomes: An Umbrella Review of Systematic Reviews and Meta-Analyses. Nutrients 2019, 11, 338. [Google Scholar] [CrossRef] [Green Version]
- Rifai, L.; Pisano, C.; Hayden, J.; Sulo, S.; Silver, M.A. Impact of the DASH diet on endothelial function, exercise capacity, and quality of life in patients with heart failure. Bayl. Univ. Med. Cent. Proc. 2015, 28, 151–156. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, H.T.; Bertoni, A.G.; Nettleton, J.A.; Bluemke, D.A.; Levitan, E.B.; Burke, G.L. DASH Eating Pattern Is Associated with Favorable Left Ventricular Function in the Multi-Ethnic Study of Atherosclerosis. J. Am. Coll. Nutr. 2012, 31, 401–407. [Google Scholar] [CrossRef] [Green Version]
- He, F.J.; MacGregor, G.A. Potassium: More beneficial effects. Climacteric 2003, 6, 36–48. [Google Scholar] [PubMed]
- Rifai, L.; Silver, M.A. A Review of the DASH Diet as an Optimal Dietary Plan for Symptomatic Heart Failure. Prog. Cardiovasc. Dis. 2015, 58, 548–554. [Google Scholar] [CrossRef] [PubMed]
- Sciatti, E.; Lombardi, C.; Ravera, A.; Vizzardi, E.; Bonadei, I.; Carubelli, V.; Gorga, E.; Metra, M. Nutritional Deficiency in Patients with Heart Failure. Nutrients 2016, 8, 442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haring, B.; Von Ballmoos, M.C.W.; Appel, L.J.; Sacks, F.M. Healthy Dietary Interventions and Lipoprotein (a) Plasma Levels: Results from the Omni Heart Trial. PLoS ONE 2014, 9, e114859. [Google Scholar] [CrossRef] [Green Version]
- Mamic, P.; Chaikijurajai, T.; Tang, W.W. Gut microbiome—A potential mediator of pathogenesis in heart failure and its comorbidities: State-of-the-art review. J. Mol. Cell. Cardiol. 2020, 152, 105–117. [Google Scholar] [CrossRef]
- Shah, S.J.; Katz, D.H.; Deo, R.C. Phenotypic Spectrum of Heart Failure with Preserved Ejection Fraction. Heart Fail. Clin. 2014, 10, 407–418. [Google Scholar] [CrossRef] [Green Version]
- Shah, S.J. Precision Medicine for Heart Failure with Preserved Ejection Fraction: An Overview. J. Cardiovasc. Transl. Res. 2017, 10, 233–244. [Google Scholar] [CrossRef]
- Juraschek, S.P.; Kovell, L.C.; Appel, L.J.; Miller, E.R.; Sacks, F.M.; Chang, A.R.; Christenson, R.H.; Rebuck, H.; Mukamal, K.J. Effects of Diet and Sodium Reduction on Cardiac Injury, Strain, and Inflammation. J. Am. Coll. Cardiol. 2021, 77, 2625–2634. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hodge, R.A.; Stevens, V.L.; Hartman, T.J.; McCullough, M.L. Identification and Reproducibility of Plasma Metabolomic Biomarkers of Habitual Food Intake in a US Diet Validation Study. Metabolites 2020, 10, 382. [Google Scholar] [CrossRef]
- Maruvada, P.; Lampe, J.W.; Wishart, D.S.; Barupal, D.; Chester, D.N.; Dodd, D.; Feunang, Y.D.; Dorrestein, P.C.; Dragsted, L.O.; Draper, J.; et al. Perspective: Dietary Biomarkers of Intake and Exposure—Exploration with Omics Approaches. Adv. Nutr. 2019, 11, 200–215. [Google Scholar] [CrossRef]
- Tang, H.-Y.; Wang, C.-H.; Ho, H.-Y.; Lin, J.-F.; Lo, C.-J.; Huang, C.-Y.; Cheng, M.-L. Characteristic of Metabolic Status in Heart Failure and Its Impact in Outcome Perspective. Metabolites 2020, 10, 437. [Google Scholar] [CrossRef]
- Chang, P.; Lei, S.; Zhang, X.; Zhang, J.; Wang, X.; Wu, J.; Wang, J.; Geng, J.; Chen, B.; Yu, J. Metabonomics Analysis of Myocardial Metabolic Dysfunction in Patients with Cardiac Natriuretic Peptide Resistance. Cardiol. Res. Pract. 2020, 2020, 1416945. [Google Scholar] [CrossRef] [PubMed]
- Israr, M.Z.; Bernieh, D.; Salzano, A.; Cassambai, S.; Yazaki, Y.; Heaney, L.M.; Jones, D.J.; Ng, L.L.; Suzuki, T. Association of gut-related metabolites with outcome in acute heart failure. Am. Heart J. 2021, 234, 71–80. [Google Scholar] [CrossRef]
- González-Domínguez, R.; Jáuregui, O.; Mena, P.; Hanhineva, K.; Tinahones, F.J.; Angelino, D.; Andrés-Lacueva, C. Quantifying the human diet in the crosstalk between nutrition and health by multi-targeted metabolomics of food and microbiota-derived metabolites. Int. J. Obes. 2020, 44, 2372–2381. [Google Scholar] [CrossRef]
- Playdon, M.C.; Moore, S.C.; Derkach, A.; Reedy, J.; Subar, A.F.; Sampson, J.N.; Albanes, D.; Gu, F.; Kontto, J.; Lassale, C.; et al. Identifying biomarkers of dietary patterns by using metabolomics. Am. J. Clin. Nutr. 2016, 105, 450–465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCullough, M.L.; Maliniak, M.L.; Stevens, V.L.; Carter, B.D.; Hodge, R.A.; Wang, Y. Metabolomic markers of healthy dietary patterns in US postmenopausal women. Am. J. Clin. Nutr. 2019, 109, 1439–1451. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.E.; Song, R.J.; Xu, X.; Gerszten, R.E.; Ngo, D.; Clish, C.B.; Corlin, L.; Ma, J.; Xanthakis, V.; Jacques, P.F.; et al. Proteomic and Metabolomic Correlates of Healthy Dietary Patterns: The Framingham Heart Study. Nutrients 2020, 12, 1476. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Hu, E.A.; Wong, K.E.; Yu, B.; Steffen, L.M.; Seidelmann, S.B.; Boerwinkle, E.; Coresh, J.; Rebholz, C.M. Serum Metabolites Associated with Healthy Diets in African Americans and European Americans. J. Nutr. 2020, 151, 40–49. [Google Scholar] [CrossRef]
- Rebholz, C.M.; Lichtenstein, A.H.; Zheng, Z.; Appel, L.J.; Coresh, J. Serum untargeted metabolomic profile of the Dietary Approaches to Stop Hypertension (DASH) dietary pattern. Am. J. Clin. Nutr. 2018, 108, 243–255. [Google Scholar] [CrossRef]
- Derkach, A.; Sampson, J.; Joseph, J.; Playdon, M.; Stolzenberg-Solomon, R.Z. Effects of dietary sodium on metabolites: The Dietary Approaches to Stop Hypertension (DASH)–Sodium Feeding Study. Am. J. Clin. Nutr. 2017, 106, 1131–1141. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.; Lichtenstein, A.H.; Wong, K.E.; Appel, L.J.; Coresh, J.; Rebholz, C.M. Urine Metabolites Associated with the Dietary Approaches to Stop Hypertension (DASH) Diet: Results from the DASH-Sodium Trial. Mol. Nutr. Food Res. 2020, 65, e2000695. [Google Scholar] [CrossRef]
- Chaudhary, P.; Wainford, R.D. Association of urinary sodium and potassium excretion with systolic blood pressure in the Dietary Approaches to Stop Hypertension Sodium Trial. J. Hum. Hypertens. 2021, 35, 577–587. [Google Scholar] [CrossRef]
- Gordon-Dseagu, V.L.Z.; Derkach, A.; Xiao, Q.; Williams, I.; Sampson, J.; Stolzenberg-Solomon, R.Z. The association of sleep with metabolic pathways and metabolites: Evidence from the Dietary Approaches to Stop Hypertension (DASH)—sodium feeding study. Metabolomics 2019, 15, 48. [Google Scholar] [CrossRef]
- Mathew, A.V.; Seymour, E.M.; Byun, J.; Pennathur, S.; Hummel, S.L. Altered Metabolic Profile with Sodium-Restricted Dietary Approaches to Stop Hypertension Diet in Hypertensive Heart Failure with Preserved Ejection Fraction. J. Card. Fail. 2015, 21, 963–967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reisdorph, N.A.; Hendricks, A.E.; Tang, M.; Doenges, K.A.; Reisdorph, R.M.; Tooker, B.C.; Quinn, K.; Borengasser, S.J.; Nkrumah-Elie, Y.; Frank, D.N.; et al. Nutrimetabolomics reveals food-specific compounds in urine of adults consuming a DASH-style diet. Sci. Rep. 2020, 10, 1157. [Google Scholar] [CrossRef]
- Pourafshar, S.; Nicchitta, M.; Tyson, C.; Svetkey, L.; Corcoran, D.; Bain, J.; Muehlbauer, M.; Ilkayeva, O.; O’Connell, T.; Lin, P.-H.; et al. Urine and Plasma Metabolome of Healthy Adults Consuming the DASH (Dietary Approaches to Stop Hypertension) Diet: A Randomized Pilot Feeding Study. Nutrients 2021, 13, 1768. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Anderson, C.A.; Hu, E.A.; Zheng, Z.; Appel, L.J.; He, J.; Feldman, H.I.; Anderson, A.H.; Ricardo, A.C.; Bhat, Z.; et al. Plasma Metabolomic Signatures of Healthy Dietary Patterns in the Chronic Renal Insufficiency Cohort (CRIC) Study. J. Nutr. 2021, 151, 2894–2907. [Google Scholar] [CrossRef]
- Hummel, S.L.; Seymour, E.M.; Brook, R.D.; Kolias, T.J.; Sheth, S.S.; Rosenblum, H.R.; Wells, J.M.; Weder, A.B. Low-Sodium Dietary Approaches to Stop Hypertension Diet Reduces Blood Pressure, Arterial Stiffness, and Oxidative Stress in Hypertensive Heart Failure with Preserved Ejection Fraction. Hypertension 2012, 60, 1200–1206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hummel, S.L.; Karmally, W.; Gillespie, B.W.; Helmke, S.; Teruya, S.; Wells, J.; Trumble, E.; Jimenez, O.; Marolt, C.; Wessler, J.D.; et al. Home-Delivered Meals Postdischarge from Heart Failure Hospitalization. Circ. Heart Fail. 2018, 11, e004886. [Google Scholar] [CrossRef]
- Epstein, D.E.; Sherwood, A.; Smith, P.J.; Craighead, L.; Caccia, C.; Lin, P.-H.; Babyak, M.A.; Johnson, J.J.; Hinderliter, A.; Blumenthal, J.A. Determinants and Consequences of Adherence to the Dietary Approaches to Stop Hypertension Diet in African-American and White Adults with High Blood Pressure: Results from the ENCORE Trial. J. Acad. Nutr. Diet. 2012, 112, 1763–1773. [Google Scholar] [CrossRef] [Green Version]
- Lin, P.-H.; Appel, L.J.; Funk, K.; Craddick, S.; Chen, C.; Elmer, P.; McBurnie, M.A.; Champagne, C. The PREMIER Intervention Helps Participants Follow the Dietary Approaches to Stop Hypertension Dietary Pattern and the Current Dietary Reference Intakes Recommendations. J. Am. Diet. Assoc. 2007, 107, 1541–1551. [Google Scholar] [CrossRef]
- Abshire, M.; Xu, J.; Baptiste, D.-L.; Almansa, J.R.; Xu, J.; Cummings, A.; Andrews, M.J.; Himmelfarb, C.D. Nutritional Interventions in Heart Failure: A Systematic Review of the Literature. J. Card. Fail. 2015, 21, 989–999. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basuray, A.; Dolansky, M.; Josephson, R.; Sattar, A.; Grady, E.M.; Vehovec, A.; Gunstad, J.; Redle, J.; Fang, J.; Hughes, J.W. Dietary Sodium Adherence Is Poor in Chronic Heart Failure Patients. J. Card. Fail. 2015, 21, 323–329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seid, M.A.; Abdela, O.A.; Zeleke, E.G. Adherence to self-care recommendations and associated factors among adult heart failure patients. From the patients’ point of view. PLoS ONE 2019, 14, e0211768. [Google Scholar] [CrossRef] [Green Version]
- Desai, A.S.; Stevenson, L.W. Rehospitalization for heart failure: Predict or prevent? Circulation 2012, 126, 501–506. [Google Scholar] [CrossRef] [Green Version]
- Jencks, S.F.; Williams, M.V.; Coleman, E.A. Rehospitalizations among Patients in the Medicare Fee-for-Service Program. N. Engl. J. Med. 2009, 360, 1418–1428. [Google Scholar] [CrossRef]
- Freedland, K.E.; Carney, R.M.; Rich, M.W.; Steinmeyer, B.C.; Rubin, E.H. Cognitive Behavior Therapy for Depression and Self-Care in Heart Failure Patients: A Randomized Clinical Trial. JAMA Intern. Med. 2015, 175, 1773–1782. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodriguez, M.A.; Friedberg, J.P.; DiGiovanni, A.; Wang, B.; Wylie-Rosett, J.; Hyoung, S.; Natarajan, S. A Tailored Behavioral Intervention to Promote Adherence to the DASH Diet. Am. J. Health Behav. 2019, 43, 659–670. [Google Scholar] [CrossRef] [PubMed]
- Ohannessian, R. Telemedicine: Potential applications in epidemic situations. Eur. Res. Telemed./Recherche Européenne Télémédecine 2015, 4, 95–98. [Google Scholar] [CrossRef]
- Monaghesh, E.; Hajizadeh, A. The role of telehealth during COVID-19 outbreak: A systematic review based on current evidence. BMC Public Health 2020, 20, 1193. [Google Scholar] [CrossRef]
- Rosen, D.; McCall, J.D.; Primack, B.A. Telehealth Protocol to Prevent Readmission Among High-Risk Patients with Congestive Heart Failure. Am. J. Med. 2017, 130, 1326–1330. [Google Scholar] [CrossRef]
- Tiede, M.; Dwinger, S.; Herbarth, L.; Härter, M.; Dirmaier, J. Long-term effectiveness of telephone-based health coaching for heart failure patients: A post-only randomised controlled trial. J. Telemed. Telecare 2016, 23, 716–724. [Google Scholar] [CrossRef]
- Kelly, J.T.; Warner, M.; Conley, M.; Reidlinger, D.P.; Hoffmann, T.; Craig, J.; Tong, A.; Reeves, M.; Johnson, D.W.; Palmer, S.; et al. Feasibility and acceptability of telehealth coaching to promote healthy eating in chronic kidney disease: A mixed-methods process evaluation. BMJ Open 2019, 9, e024551. [Google Scholar] [CrossRef]
- DeVore, A.D.; Granger, B.B.; Fonarow, G.C.; Al-Khalidi, H.R.; Albert, N.M.; Lewis, E.F.; Butler, J.; Piña, I.L.; Heidenreich, P.A.; Allen, L.A.; et al. Care Optimization Through Patient and Hospital Engagement Clinical Trial for Heart Failure: Rationale and design of CONNECT-HF. Am. Heart J. 2019, 220, 41–50. [Google Scholar] [CrossRef]
- Balas, E.A.; Boren, S.A. Managing Clinical Knowledge for Health Care Improvement. Yearb. Med. Inform. 2000, 9, 65–70. [Google Scholar] [CrossRef] [Green Version]
- Mazumdar, M.; Poeran, J.V.; Ferket, B.S.; Zubizarreta, N.; Agarwal, P.; Gorbenko, K.; Craven, C.K.; Zhong, X.T.; Moskowitz, A.J.; Gelijns, A.C.; et al. Developing an Institute for Health Care Delivery Science: Successes, challenges, and solutions in the first five years. Health Care Manag. Sci. 2020, 24, 234–243. [Google Scholar] [CrossRef]
- Eccles, M.P.; Mittman, B.S. Welcome to Implementation Science. Implement. Sci. 2006, 1, 1. [Google Scholar] [CrossRef] [Green Version]
- Stetler, C.B.; Legro, M.W.; Wallace, C.M.; Bowman, C.; Guihan, M.; Hagedorn, H.; Kimmel, B.; Sharp, N.D.; Smith, J.L. The role of formative evaluation in implementation research and the QUERI experience. J. Gen. Intern. Med. 2006, 21 (Suppl. S2), S1–S8. [Google Scholar] [CrossRef]
- Bauer, M.S.; Damschroder, L.J.; Hagedorn, H.; Smith, J.; Kilbourne, A.M. An introduction to implementation science for the non-specialist. BMC Psychol. 2015, 3, 32. [Google Scholar] [CrossRef] [Green Version]
- Murofushi, K.; Badaracco, C.; County, C.; Gonzales-Pacheco, D.; Silzle, C.; Watowicz, R.; Moloney, L. Implementation Science in Evidence-based Nutrition Practice: Considerations for the Registered Dietitian Nutritionist. J. Acad. Nutr. Diet. 2020, 121, 1392–1400. [Google Scholar] [CrossRef]
- Garner, J.A.; Proaño, G.V.; Kelley, K.; Banna, J.C.; Emenaker, N.J.; Sauer, K. Revising the Academy’s Research Priorities: Methods of the Research Priorities and Strategies Development Task Force, 2017–2019. J. Acad. Nutr. Diet. 2020, 121, 2275–2286. [Google Scholar] [CrossRef] [PubMed]
- Pearson, N.; Naylor, P.-J.; Ashe, M.C.; Fernandez, M.; Yoong, S.L.; Wolfenden, L. Guidance for conducting feasibility and pilot studies for implementation trials. Pilot Feasibility Stud. 2020, 6, 167. [Google Scholar] [CrossRef]
- Sampson, U.K.; McGlynn, E.A.; Perlin, J.B.; Frisse, M.E.; Arnold, S.B.; Benz, E.J., Jr.; Brennan, T.; Briss, P.; Buntin, M.J.B.; Khosla, S.; et al. Advancing the Science of Healthcare Service Delivery: The NHLBI Corporate Healthcare Leaders’ Panel. Glob. Heart 2018, 13, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Sampson, U.K.; Kaplan, R.M.; Cooper, R.S.; Diez Roux, A.V.; Marks, J.S.; Engelgau, M.M.; Peprah, E.; Mishoe, H.; Boulware, L.E.; Felix, K.L.; et al. Reducing Health Inequities in the U.S. J. Am. Coll. Cardiol. 2016, 68, 517–524. [Google Scholar] [CrossRef] [PubMed]
- De Batlle, J.; Matejcic, M.; Chajes, V.; Moreno-Macias, H.; Amadou, A.; Slimani, N.; Cox, D.G.; Clavel-Chapelon, F.; Fagherazzi, G.; Romieu, I. Determinants of folate and vitamin B12 plasma levels in the French E3N-EPIC cohort. Eur. J. Nutr. 2018, 57, 751–760. [Google Scholar] [CrossRef]
- Gomez-Delgado, F.; Alcala-Diaz, J.F.; Leon-Acuña, A.; Lopez-Moreno, J.; Lista, F.J.D.; Gomez-Marin, B.; Roncero-Ramos, I.; Yubero-Serrano, E.M.; Rangel-Zuñiga, O.A.; Vals-Delgado, C.; et al. Apolipoprotein E genetic variants interact with Mediterranean diet to modulate postprandial hypertriglyceridemia in coronary heart disease patients: CORDIOPREV study. Eur. J. Clin. Investig. 2019, 49, e13146. [Google Scholar] [CrossRef]
- Zeevi, D.; Korem, T.; Zmora, N.; Israeli, D.; Rothschild, D.; Weinberger, A.; Ben-Yacov, O.; Lador, D.; Avnit-Sagi, T.; Lotan-Pompan, M.; et al. Personalized Nutrition by Prediction of Glycemic Responses. Cell 2015, 163, 1079–1094. [Google Scholar] [CrossRef] [Green Version]
- Mendes-Soares, H.; Raveh-Sadka, T.; Azulay, S.; Ben-Shlomo, Y.; Cohen, Y.; Ofek, T.; Stevens, J.; Bachrach, D.; Kashyap, P.; Segal, L.; et al. Model of personalized postprandial glycemic response to food developed for an Israeli cohort predicts responses in Midwestern American individuals. Am. J. Clin. Nutr. 2019, 110, 63–75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berry, S.E.; Valdes, A.M.; Drew, D.A.; Asnicar, F.; Mazidi, M.; Wolf, J.; Capdevila, J.; Hadjigeorgiou, G.; Davies, R.; Al Khatib, H.; et al. Human postprandial responses to food and potential for precision nutrition. Nat. Med. 2020, 26, 964–973. [Google Scholar] [CrossRef]
- National Institutes of Health. NIH-Wide Strategic Plan Fiscal Years 2021–2025. 2021. Available online: https://www.nih.gov/sites/default/files/about-nih/strategic-plan-fy2021-2025-508.pdf (accessed on 3 October 2021).
- Cresci, S.; Pereira, N.L.; Ahmad, F.; Byku, M.; Fuentes, L.D.L.; Lanfear, D.E.; Reilly, C.M.; Owens, A.T.; Wolf, M.J. Heart Failure in the Era of Precision Medicine: A Scientific Statement from the American Heart Association. Circ. Genom. Precis. Med. 2019, 12, 458–485. [Google Scholar] [CrossRef] [PubMed]
- Konstantinidou, V.; Ruiz, L.A.D.; Ordovás, J.M. Personalized Nutrition and Cardiovascular Disease Prevention: From Framingham to PREDIMED. Adv. Nutr. 2014, 5, 368S–371S. [Google Scholar] [CrossRef] [Green Version]
- Corella, D.; Carrasco, P.; Sorlí, J.V.; Estruch, R.; Rico-Sanz, J.; Martínez-González, M.; Salas-Salvadó, J.; Covas, M.I.; Coltell, O.; Arós, F.; et al. Mediterranean Diet Reduces the Adverse Effect of the TCF7L2-rs7903146 Polymorphism on Cardiovascular Risk Factors and Stroke Incidence: A randomized controlled trial in a high-cardiovascular-risk population. Diabetes Care 2013, 36, 3803–3811. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaikijurajai, T.; Tang, W.H.W. Gut Microbiome and Precision Nutrition in Heart Failure: Hype or Hope? Curr. Heart Fail. Rep. 2021, 18, 23–32. [Google Scholar] [CrossRef]

| Study | Aim | Design & Diet | Subject Group | Omic Method | Bio-Sample | Main Finding |
|---|---|---|---|---|---|---|
| Mathew A, et al., 2015 J Card Fail [90] | Characterize metabolomic changes following DASH Sodium-restricted diet intervention | Single arm, Controlled feeding study, 3 weeks DASH sodium reduced diet Ş | Adults with HTN and HFpEF (n = 13), predominately female | GC-MS and LC-MS, targeted | Serum | Short-chain acyl carnitine metabolites increased significantly, correlated with improved cardiac function |
| Derkach A, et al., 2017 AJCN [86] | Evaluate the effect of sodium intake on metabolomic profiles following control and DASH diet intervention | RCT, Crossover Controlled feeing study, 2-week run-in, 12 weeks DASH vs. Control arms each with 3 sodium levels for 4 weeks each intervention ¥ | Adults with HTN, M + F, mixed race, n = 119 from DASH-Sodium trial; Samples compared high-versus low or medium sodium, independent of diet arm | LC-MS, untargeted | Plasma | Sodium intake significantly associated with changes in 6 metabolite pathways. Adjustment for change in diet pattern or blood pressure did not alter associations |
| Rebholz CM, et al., 2018 AJCN [85] | Identify metabolites associated with DASH diet pattern | RCT, Parallel, Controlled feeding study, 3-week Control run-in, 8 weeks feeding DASH diet or Control or Fruit & Vegetable-rich diet ≠ | Adults with pre-and stage 1 HTN, M + F, mixed race, n = 329 (control 108, fruit/veg 111, DASH 110) | GC-MS and LC-MS, untargeted | Serum | Multiple metabolites differed significantly DASH vs. Control (97) and DASH vs. fruit/veg (67); Identified 10 most influential metabolites as candidate biomarkers for assessing adherence to DASH diet; metabolites represented classes of lipids, amino acids, xenobiotics and food components, cofactors and vitamins, carbohydrates |
| Reisdorph N, et al., 2020 Sci Report [91] | Characterize changes in metabolome following DASH diet intervention, and characterize food specific compounds (FSC) | RCT, Crossover, Controlled feeding study, 2-week habitual diet run-in, 6-week feeding each of two DASH style diets Ş | Adults, M + F, n = 19 | LC-MS, untargeted | Urine and foods | FSC from DASH diet were detected in urine along with other metabolites; 16 metabolites were significantly associated with BP and 6 with change in BP |
| Chaudhary P, et al., 2021 Hypertension [88] | Evaluate the effect of sodium intake on metabolomic and lipidomic profiles between salt-sensitive and salt-resistant individuals | RCT, Crossover Controlled feeing study, 2-week run-in, 12 weeks DASH vs. Control arms each with 3 sodium levels for 4 weeks each intervention ¥ | Adults with HTN, M + F, mixed race, n = 191 (106 salt-sensitive, 85 salt-resistant) from DASH-Sodium trial; Samples compared high-versus low sodium on Control diet arm | LC-MS, untargeted | Plasma | Baseline comparisons exhibited no differences, but 3 metabolites differed significantly in salt sensitive subjects with change in sodium intake |
| Kim H, et al., 2021 MNFR [87] | Determine if candidate serum biomarkers from original DASH trial replicate in urine in DASH-Sodium trial | RCT, Crossover Controlled feeing study, 2-week run-in, 12 weeks DASH vs. Control arms each with 3 sodium levels for 4 weeks each intervention ¥ | Adults with HTN, M + F, mixed race, n = 193 (control), n = 202 (DASH) from DASH-Sodium trial | LC-MS, untargeted | Urine | Identified several novel metabolite markers of the DASH diet; Replicated 8 significant urine metabolites identified in serum of original DASH trial that distinguish DASH vs. Control; Identified 9 significant urine metabolites identical in DASH-high sodium and DASH-low sodium diets |
| Pourafshar S, et al., 2021 Nutrients [92] | Characterize changes in metabolome following DASH diet intervention | RCT, Parallel, Controlled feeding study, 1 week Control run-in, 2 week feeding DASH diet or Control Ş | Adults with HTN, M + F, mixed race, n = 20 | GC-MS, untargeted | Plasma and urine | Urine—19 metabolites differed significantly DASH vs. Control; Plasma—8 metabolites differed significantly DASH vs. Control; Major classes included phenolics, amino acid, organooxygen compounds, and cofactors |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wickman, B.E.; Enkhmaa, B.; Ridberg, R.; Romero, E.; Cadeiras, M.; Meyers, F.; Steinberg, F. Dietary Management of Heart Failure: DASH Diet and Precision Nutrition Perspectives. Nutrients 2021, 13, 4424. https://doi.org/10.3390/nu13124424
Wickman BE, Enkhmaa B, Ridberg R, Romero E, Cadeiras M, Meyers F, Steinberg F. Dietary Management of Heart Failure: DASH Diet and Precision Nutrition Perspectives. Nutrients. 2021; 13(12):4424. https://doi.org/10.3390/nu13124424
Chicago/Turabian StyleWickman, Brooke E., Byambaa Enkhmaa, Ronit Ridberg, Erick Romero, Martin Cadeiras, Frederick Meyers, and Francene Steinberg. 2021. "Dietary Management of Heart Failure: DASH Diet and Precision Nutrition Perspectives" Nutrients 13, no. 12: 4424. https://doi.org/10.3390/nu13124424
APA StyleWickman, B. E., Enkhmaa, B., Ridberg, R., Romero, E., Cadeiras, M., Meyers, F., & Steinberg, F. (2021). Dietary Management of Heart Failure: DASH Diet and Precision Nutrition Perspectives. Nutrients, 13(12), 4424. https://doi.org/10.3390/nu13124424






