Association of Sodium, Potassium and Sodium-to-Potassium Ratio with Urine Albumin Excretion among the General Chinese Population
Abstract
1. Introduction
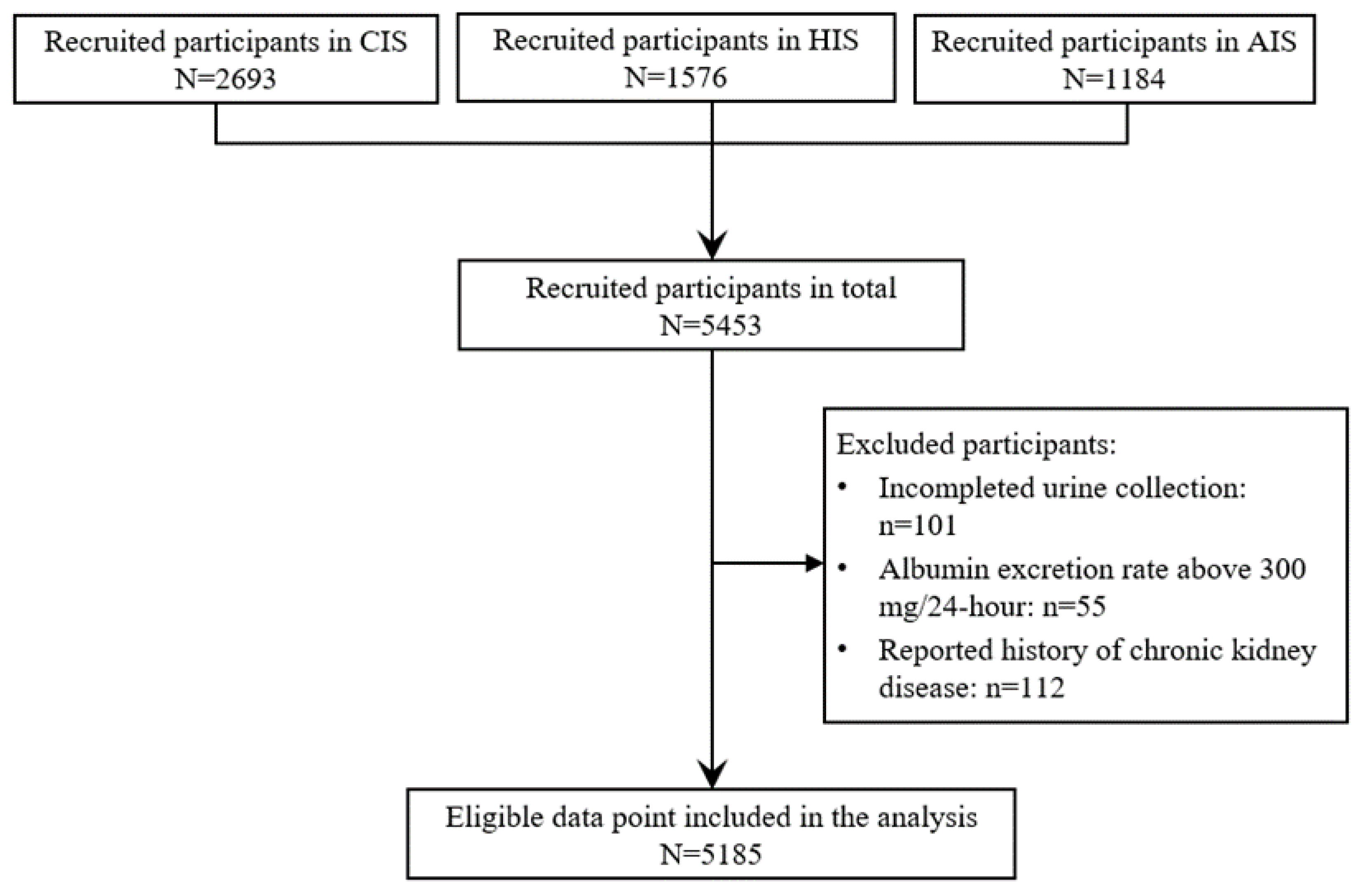
2. Materials and Methods
2.1. Participants
2.2. Data Collection
2.3. Statistical Analysis
3. Results
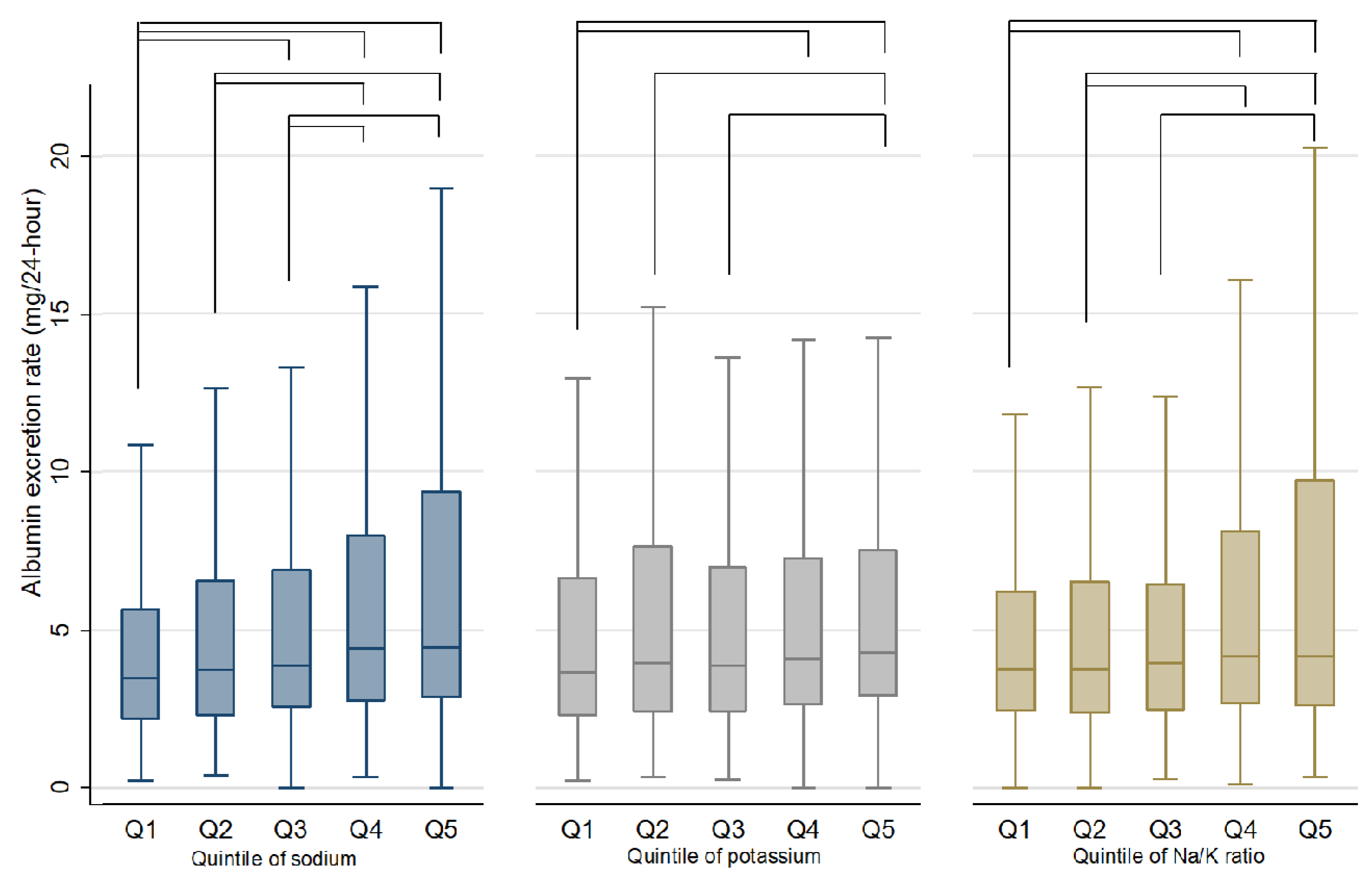
3.1. AER Levels across the Quintiles of Sodium, Potassium and Na/K Ratio
3.2. Adjusted Association between the Levels of Sodium, Potassium, Na/K Ratio and AER Level
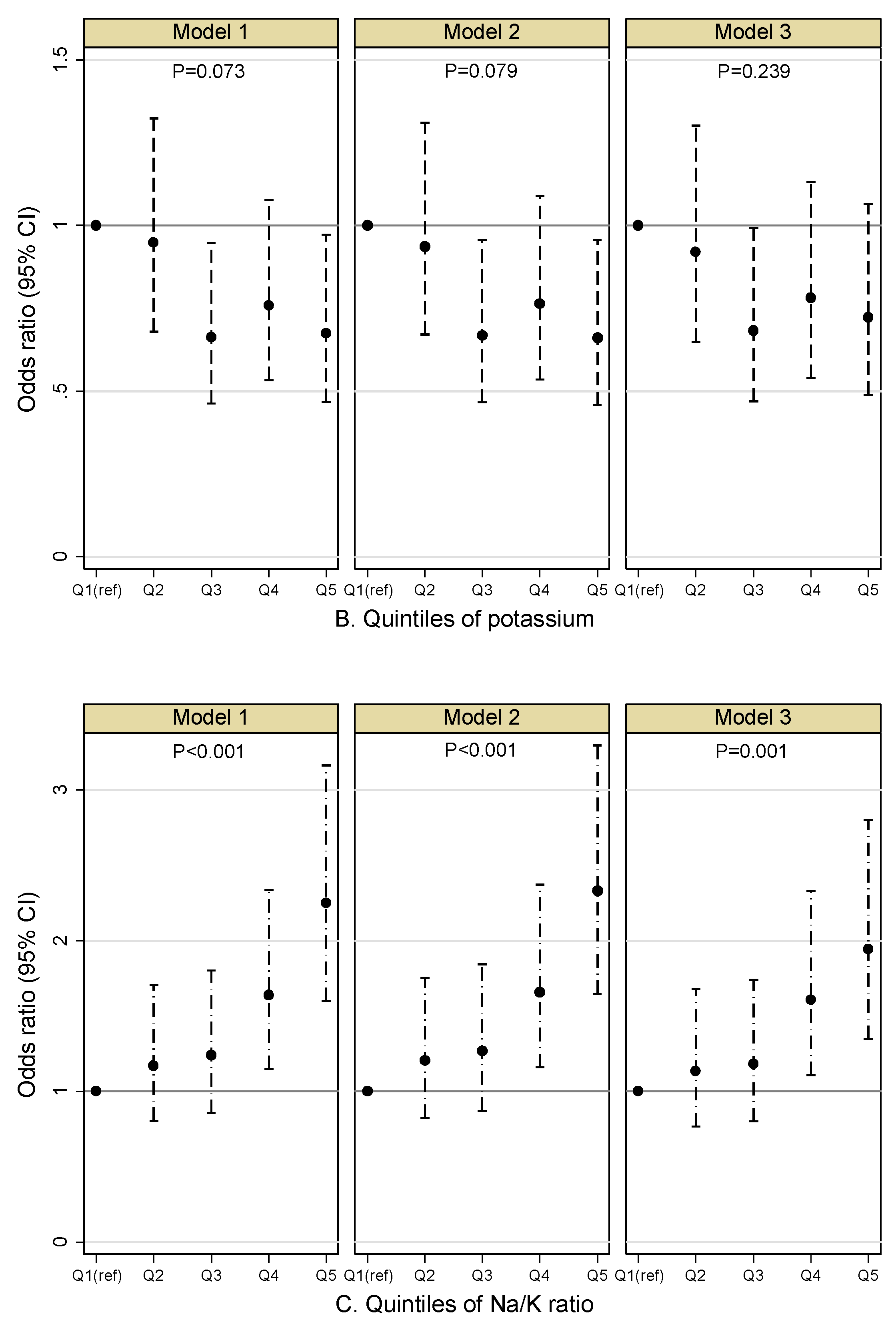
3.3. Adjusted Association between the Quintiles of Sodium, Potassium, Na/K Ratio and Proportions of Albuminuria
3.4. Sensitivity Analysis
4. Discussion
Strengths and Weaknesses of This Study
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, L.; Wang, F.; Wang, L.; Wang, W.; Liu, B.; Liu, J.; Chen, M.; He, Q.; Liao, Y.; Yu, X.; et al. Prevalence of chronic kidney disease in China: A cross-sectional survey. Lancet 2012, 379, 815–822. [Google Scholar] [CrossRef]
- Lv, J.C.; Zhang, L.X. Prevalence and Disease Burden of Chronic Kidney Disease. Adv. Exp. Med. Biol. 2019, 1165, 3–15. [Google Scholar] [PubMed]
- Yang, C.; Wang, H.; Zhao, X.; Matsushita, K.; Coresh, J.; Zhang, L.; Zhao, M.H. CKD in China: Evolving Spectrum and Public Health Implications. Am. J. Kidney Dis. 2020, 76, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Levin, A.; Stevens, P.E. Summary of KDIGO 2012 CKD Guideline: Behind the scenes, need for guidance, and a framework for moving forward. Kidney Int. 2014, 85, 49–61. [Google Scholar] [CrossRef]
- Matsushita, K.; van der Velde, M.; Astor, B.C.; Woodward, M.; Levey, A.S.; de Jong, P.E.; Coresh, J.; Gansevoort, R.T. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: A collaborative meta-analysis. Lancet 2010, 375, 2073–2081. [Google Scholar]
- Sung, K.C.; Ryu, S.; Lee, J.Y.; Lee, S.H.; Cheong, E.; Hyun, Y.Y.; Lee, K.B.; Kim, H.; Byrne, C.D. Urine Albumin/Creatinine Ratio Below 30 mg/g is a Predictor of Incident Hypertension and Cardiovascular Mortality. J. Am. Heart Assoc. 2016, 5, e003245. [Google Scholar] [CrossRef]
- Xu, J.W.; Wu, J.; Chen, X.R.; Yan, L.X.; Cai, X.N.; Ma, J.X. Association between 24 h urinary sodium excretion and microalbuminuria among Chinese people aged from 18 to 69 years old. Zhonghua Yu Fang Yi Xue Za Zhi 2019, 53, 459–463. [Google Scholar]
- Jardine, M.J.; Li, N.; Ninomiya, T.; Feng, X.; Zhang, J.; Shi, J.; Zhang, Y.; Zhang, R.; Zhang, J.; Hao, J.; et al. Dietary Sodium Reduction Reduces Albuminuria: A Cluster Randomized Trial. J. Ren. Nutr. 2019, 29, 276–284. [Google Scholar] [CrossRef]
- Deriaz, D.; Guessous, I.; Vollenweider, P.; Devuyst, O.; Burnier, M.; Bochud, M.; Ponte, B. Estimated 24-h urinary sodium and sodium-to-potassium ratio are predictors of kidney function decline in a population-based study. J. Hypertens. 2019, 37, 1853–1860. [Google Scholar] [CrossRef]
- Jones-Burton, C.; Mishra, S.I.; Fink, J.C.; Brown, J.; Gossa, W.; Bakris, G.L.; Weir, M.R. An In-Depth Review of the Evidence Linking Dietary Salt Intake and Progression of Chronic Kidney Disease. Am. J. Nephrol. 2006, 26, 268–275. [Google Scholar] [CrossRef]
- Kieneker, L.M.; Bakker, S.J.; de Boer, R.A.; Navis, G.J.; Gansevoort, R.T.; Joosten, M.M. Low potassium excretion but not high sodium excretion is associated with increased risk of developing chronic kidney disease. Kidney Int. 2016, 90, 888–896. [Google Scholar] [CrossRef] [PubMed]
- Smyth, A.; Dunkler, D.; Gao, P.; Teo, K.K.; Yusuf, S.; O’Donnell, M.J.; Mann, J.F.; Clase, C.M. The relationship between estimated sodium and potassium excretion and subsequent renal outcomes. Kidney Int. 2014, 86, 1205–1212. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; McFann, K.; Chonchol, M.; de Boer, I.H.; Kendrick, J. Association between Dietary Sodium and Potassium Intake with Chronic Kidney Disease in US Adults: A Cross-Sectional Study. Am. J. Nephrol. 2013, 37, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Hendriksen, M.A.H.; Over, E.A.B.; Navis, G.; Joles, J.A.; Hoorn, E.J.; Gansevoort, R.T.; Boshuizen, H.C. Limited Salt Consumption Reduces the Incidence of Chronic Kidney Disease: A Modeling Study. J. Public Health 2018, 40, e351–e358. [Google Scholar] [CrossRef] [PubMed]
- Wei, K.Y.; Gritter, M.; Vogt, L.; de Borst, M.H.; Rotmans, J.I.; Hoorn, E.J. Dietary potassium and the kidney: Lifesaving physiology. Clin. Kidney J. 2020, 13, 952–968. [Google Scholar] [CrossRef]
- Elfassy, T.; Zhang, L.; Raij, L.; Bibbins-Domingo, K.; Lewis, C.E.; Allen, N.B.; Liu, K.J.; Peralta, C.A.; Odden, M.C.; Zeki Al Hazzouri, A. Results of the CARDIA study suggest that higher dietary potassium may be kidney protective. Kidney Int. 2020, 98, 187–194. [Google Scholar] [CrossRef]
- McLean, R.M. Measuring Population Sodium Intake: A Review of Methods. Nutrients 2014, 6, 4651–4662. [Google Scholar] [CrossRef]
- McLean, R.M.; Farmer, V.L.; Nettleton, A.; Cameron, C.M.; Cook, N.R.; Campbell, N.R.C.; Consortium, T. Assessment of dietary sodium intake using a food frequency questionnaire and 24-hour urinary sodium excretion: A systematic literature review. J. Clin. Hypertens. 2017, 19, 1214–1230. [Google Scholar] [CrossRef]
- Oh, S.W.; Koo, H.S.; Han, K.H.; Han, S.Y.; Chin, H.J. Associations of sodium intake with obesity, metabolic disorder, and albuminuria according to age. PLoS ONE 2017, 12, e0188770. [Google Scholar] [CrossRef]
- Lin, J.; Hu, F.B.; Curhan, G.C. Associations of diet with albuminuria and kidney function decline. Clin. J. Am. Soc. Nephrol. 2010, 5, 836–843. [Google Scholar] [CrossRef]
- Shim, J.S.; Oh, K.; Kim, H.C. Dietary Assessment Methods in Epidemiologic Studies. Epidemiol. Health 2014, 36, e2014009. [Google Scholar] [CrossRef]
- Inker, L.A. Albuminuria: Time to Focus on Accuracy. Am. J. Kidney Dis. 2014, 63, 378–381. [Google Scholar] [CrossRef] [PubMed]
- Younes, N.; Cleary, P.A.; Steffes, M.W.; de Boer, I.H.; Molitch, M.E.; Rutledge, B.N.; Lachin, J.M.; Dahms, W. Comparison of Urinary Albumin-Creatinine Ratio and Albumin Excretion Rate in the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Study. Clin. J. Am. Soc. Nephrol. 2010, 5, 1235. [Google Scholar] [CrossRef]
- He, F.J.; Zhang, P.; Luo, R.; Li, Y.; Chen, F.; Zhao, Y.; Zhao, W.; Li, D.; Chen, H.; Wu, T. An Application-based programme to reinforce and maintain lower salt intake (AppSalt) in schoolchildren and their families in China. BMJ Open 2019, 9, e027793. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; He, F.J.; Li, Y.; Li, C.; Wu, J.; Ma, J.; Zhang, B.; Wang, H.; Li, Y.; Han, J.; et al. Reducing Salt Intake in China with “Action on Salt China” (ASC): Protocol for Campaigns and Randomized Controlled Trials. JMIR Res. Protoc. 2020, 9, e15933. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Tang, B.; Liu, M.; Bai, Y.; Yan, W.; Zhou, X.; Xu, Z.; He, J.; Jin, D.; Sun, J.; et al. A town level comprehensive intervention study to reduce salt intake in China: Protocol for a cluster randomised controlled trial. BMJ Open 2020, 10, e032976. [Google Scholar] [CrossRef]
- Zhang, X.; Hu, X.; Ma, J.; Zhang, P.; Li, Y.; Luo, R.; He, F.J.; MacGregor, G.A.; Wang, J.; Yin, Z. Cluster randomised controlled trial of home cook intervention to reduce salt intake in China: A protocol study. BMJ Open 2020, 10, e033842. [Google Scholar] [CrossRef]
- Han, S.Y.; Hong, J.W.; Noh, J.H.; Kim, D.J. Association of the estimated 24-h urinary sodium excretion with albuminuria in adult koreans: The 2011 Korea National Health and Nutrition Examination Survey. PLoS ONE 2014, 9, e109073. [Google Scholar] [CrossRef]
- Aaron, K.J.; Campbell, R.C.; Judd, S.E.; Sanders, P.W.; Muntner, P. Association of dietary sodium and potassium intakes with albuminuria in normal-weight, overweight, and obese participants in the Reasons for Geographic and Racial Differences in Stroke (REGARDS) Study. Am. J. Clin. Nutr. 2011, 94, 1071–1078. [Google Scholar] [CrossRef]
- Forman, J.P.; Scheven, L.; de Jong, P.E.; Bakker, S.J.; Curhan, G.C.; Gansevoort, R.T. Association between sodium intake and change in uric acid, urine albumin excretion, and the risk of developing hypertension. Circulation 2012, 125, 3108–3116. [Google Scholar] [CrossRef]
- Hattori, H.; Hirata, A.; Kubo, S.; Nishida, Y.; Nozawa, M.; Kawamura, K.; Hirata, T.; Kubota, Y.; Sata, M.; Kuwabara, K.; et al. Estimated 24 h Urinary Sodium-to-Potassium Ratio Is Related to Renal Function Decline: A 6-Year Cohort Study of Japanese Urban Residents. Int. J. Environ. Res. Public. Health 2020, 17, 5811. [Google Scholar] [CrossRef] [PubMed]
- Koo, H.; Hwang, S.; Kim, T.H.; Kang, S.W.; Oh, K.-H.; Ahn, C.; Kim, Y.H. The Ratio of Urinary Sodium and Potassium and Chronic Kidney Disease Progression: Results from the KoreaN Cohort Study for Outcomes in Patients with Chronic Kidney Disease (KNOW-CKD). Medicine 2018, 97, e12820. [Google Scholar] [CrossRef] [PubMed]
- He, F.J.; Marciniak, M.; Visagie, E.; Markandu, N.D.; Anand, V.; Dalton, R.N.; MacGregor, G.A. Effect of modest salt reduction on blood pressure, urinary albumin, and pulse wave velocity in white, black, and Asian mild hypertensives. Hypertension 2009, 54, 482–488. [Google Scholar] [CrossRef] [PubMed]
- He, F.J.; Marciniak, M.; Carney, C.; Markandu, N.D.; Anand, V.; Fraser, W.D.; Dalton, R.N.; Kaski, J.C.; MacGregor, G.A. Effects of potassium chloride and potassium bicarbonate on endothelial function, cardiovascular risk factors, and bone turnover in mild hypertensives. Hypertension 2010, 55, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; He, F.J.; Wang, C.; MacGregor, G.A. Twenty-Four-Hour Urinary Sodium and Potassium Excretion in China: A Systematic Review and Meta-Analysis. J. Am. Heart Assoc. 2019, 8, e012923. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, P.; Wu, J.; Ma, J.; Xu, J.; Zhang, X.; Luo, R.; Liu, M.; Sun, Y.; Li, X.; et al. Twenty-Four-Hour Urinary Sodium and Potassium Excretion and Their Associations With Blood Pressure Among Adults in China: Baseline Survey of Action on Salt China. Hypertension 2020, 76, 1580–1588. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guideline: Potassium Intake for Adults and Children; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- Nomura, K.; Asayama, K.; Jacobs, L.; Thijs, L.; Staessen, J.A. Renal function in relation to sodium intake: A quantitative review of the literature. Kidney Int. 2017, 92, 67–78. [Google Scholar] [CrossRef]
- Tonelli, M.; Sacks, F.; Pfeffer, M.; Jhangri, G.S.; Curhan, G. Biomarkers of inflammation and progression of chronickidney disease. Kidney Int. 2005, 68, 237–245. [Google Scholar] [CrossRef]
- Ajjarapu, A.S.; Hinkle, S.N.; Li, M.; Francis, E.C.; Zhang, C. Dietary Patterns and Renal Health Outcomes in the General Population: A Review Focusing on Prospective Studies. Nutrients 2019, 11, 1877. [Google Scholar] [CrossRef]
- Patik, J.C.; Lennon, S.L.; Farquhar, W.B.; Edwards, D.G. Mechanisms of Dietary Sodium-Induced Impairments in Endothelial Function and Potential Countermeasures. Nutrients 2021, 13, 270. [Google Scholar] [CrossRef] [PubMed]
- Verhave, J.C.; Hillege, H.L.; Burgerhof, J.G.; Janssen, W.M.; Gansevoort, R.T.; Navis, G.J.; de Zeeuw, D.; de Jong, P.E. Sodium intake affects urinary albumin excretion especially in overweight subjects. J. Intern. Med. 2004, 256, 324–330. [Google Scholar] [CrossRef] [PubMed]

| Variables | Overall (n = 5185) | Albuminuria | p | |
|---|---|---|---|---|
| No (n = 4798) | Yes (n = 387) | |||
| Age (years), mean ± SD | 49.5 ± 12.8 | 49.2 ± 12.8 | 52.9 ± 12.1 | <0.001 |
| Gender: male, n (%) | 2500 (48.2) | 2279 (47.5) | 221 (57.1) | <0.001 |
| Region, n (%) | ||||
| North | 2419 (46.7) | 2263 (47.2) | 156 (40.3) | 0.009 |
| South | 2766 (53.4) | 2535 (52.8) | 231 (59.7) | |
| AER (mg/24 h), median (IQR) | 3.9 (2.5~7.3) | 3.7 (2.4~5.9) | 57.6 (39.4~99.4) | <0.001 |
| Sodium (g/24 h), mean ± SD | 4.3 ± 1.8 | 4.3 ± 1.8 | 4.8 ± 2.0 | <0.001 |
| Potassium (g/24 h) | 1.6 ± 0.6 | 1.6 ± 0.6 | 1.6 ± 0.6 | 0.993 |
| Na/K ratio, mean ± SD | 5.0 ± 2.1 | 5.0 ± 2.1 | 5.6 ± 2.3 | <0.001 |
| SBP (mmHg), mean ± SD | 125.2 ± 19.0 | 124.2 ± 18.4 | 137.9 ± 21.4 | <0.001 |
| DBP (mmHg), mean ± SD | 79.0 ± 11.1 | 78.5 ± 10.8 | 85.5 ± 12.7 | <0.001 |
| Waist circumference (cm), mean ± SD | 84.7 ± 10.6 | 84.3 ± 10.5 | 89.6 ± 10.5 | <0.001 |
| BMI, n (%) | ||||
| <24 | 1982 (38.2) | 1900 (39.6) | 82 (21.2) | <0.001 |
| 24–28 | 2086 (40.2) | 1923 (40.1) | 163 (42.1) | |
| ≥28 | 1117 (21.5) | 975 (20.3) | 142 (36.7) | |
| Education years, n (%) | ||||
| 0–6 | 2047 (39.5) | 1882 (39.2) | 165 (42.7) | 0.156 |
| 7–9 | 1749 (33.7) | 1615 (33.7) | 134 (34.6) | |
| ≥10 | 1389 (26.8) | 1301 (27.1) | 88 (22.7) | |
| Smoking: yes, n (%) | 1395 (26.9) | 1273 (26.5) | 122 (31.5) | 0.033 |
| Alcohol intake, n (%) | ||||
| No | 3061 (59.0) | 2836 (59.1) | 225 (58.1) | 0.606 |
| Occasionally | 1622 (31.3) | 1503 (31.3) | 119 (30.7) | |
| Frequently | 501 (9.7) | 458 (9.6) | 43 (11.1) | |
| Physical activity: active, n (%) | 1618 (31.2) | 1480 (30.8) | 138 (35.7) | 0.050 |
| High blood pressure, n (%) | 1662 (32.1) | 1424 (29.7) | 238 (61.5) | <0.001 |
| Antihypertensive medication: yes, n (%) | 801 (15.4) | 673 (14.0) | 128 (33.1) | <0.001 |
| Diabetes, n (%) | 282 (5.4) | 221 (4.6) | 61 (15.8) | <0.001 |
| Models | β | 95%CI | p |
|---|---|---|---|
| Sodium | |||
| Model 1 | 0.090 | 0.071~0.108 | <0.001 |
| Model 2 | 0.095 | 0.077~0.114 | <0.001 |
| Model 3 | 0.069 | 0.050~0.087 | <0.001 |
| Potassium | |||
| Model 1 | 0.011 | −0.041~0.063 | 0.679 |
| Model 2 | 0.001 | −0.051~0.053 | 0.977 |
| Model 3 | 0.039 | −0.012~0.089 | 0.135 |
| Na/K ratio | |||
| Model 1 | 0.041 | 0.027~0.055 | <0.001 |
| Model 2 | 0.045 | 0.031~0.059 | <0.001 |
| Model 3 | 0.026 | 0.012~0.040 | <0.001 |
| Models | β | 95%CI | p |
|---|---|---|---|
| Sodium | |||
| Model 1 | 0.092 | 0.074~0.110 | <0.001 |
| Model 2 | 0.098 | 0.080~0.117 | <0.001 |
| Model 3 | 0.071 | 0.053~0.089 | <0.001 |
| Potassium | |||
| Model 1 | 0.023 | −0.029~0.074 | 0.386 |
| Model 2 | 0.012 | −0.040~0.063 | 0.661 |
| Model 3 | 0.045 | −0.006~0.095 | 0.081 |
| Na/K ratio | |||
| Model 1 | 0.035 | 0.022~0.048 | <0.001 |
| Model 2 | 0.039 | 0.025~0.052 | <0.001 |
| Model 3 | 0.022 | 0.009~0.035 | 0.001 |
| Factors | Model 1 | Model 2 | Model 3 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| OR | 95%CI | p | OR | 95%CI | p | OR | 95%CI | p | |
| Sodium (g/24 h) | |||||||||
| Q1 (≤2.78) | Ref | <0.001 | Ref | <0.001 | Ref | 0.001 | |||
| Q2 (2.79~3.62) | 1.288 | 0.882~1.880 | 1.339 | 0.915~1.959 | 1.306 | 0.882~1.934 | |||
| Q3 (3.63~4.48) | 1.549 | 1.062~2.260 | 1.593 | 1.089~2.330 | 1.440 | 0.972~2.132 | |||
| Q4 (4.49~5.63) | 2.089 | 1.439~3.031 | 2.167 | 1.487~3.158 | 2.058 | 1.391~3.043 | |||
| Q5 (>5.63) | 2.460 | 1.680~3.602 | 2.564 | 1.739~3.780 | 2.080 | 1.382~3.131 | |||
| Potassium (g/24 h) | |||||||||
| Q1 (≤1.07) | Ref | 0.094 | Ref | 0.106 | Ref | 0.215 | |||
| Q2 (1.08~1.34) | 1.011 | 0.725~1.410 | 1.000 | 0.715~1.397 | 0.998 | 0.705~1.413 | |||
| Q3 (1.35~1.60) | 0.678 | 0.473~0.972 | 0.681 | 0.474~0.980 | 0.691 | 0.474~1.007 | |||
| Q4 (1.61~1.98) | 0.795 | 0.558~1.132 | 0.800 | 0.560~1.143 | 0.812 | 0.560~1.178 | |||
| Q5 (>1.98) | 0.735 | 0.510~1.058 | 0.721 | 0.499~1.042 | 0.769 | 0.521~1.133 | |||
| Na/K ratio | |||||||||
| Q1 (≤3.33) | Ref | <0.001 | Ref | <0.001 | Ref | 0.002 | |||
| Q2 (3.34~4.22) | 1.185 | 0.818~1.717 | 1.213 | 0.835~1.762 | 1.144 | 0.777~1.683 | |||
| Q3 (4.23~5.15) | 1.254 | 0.868~1.811 | 1.280 | 0.884~1.853 | 1.204 | 0.820~1.768 | |||
| Q4 (5.16~6.46) | 1.619 | 1.140~2.300 | 1.639 | 1.150~2.337 | 1.595 | 1.103~2.305 | |||
| Q5 (>6.46) | 2.191 | 1.563~3.072 | 2.271 | 1.611~3.204 | 1.915 | 1.332~2.752 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Y.; Zhang, P.; Li, Y.; He, F.J.; Wu, J.; Xu, J.; Zhang, X.; Li, X.; Song, J. Association of Sodium, Potassium and Sodium-to-Potassium Ratio with Urine Albumin Excretion among the General Chinese Population. Nutrients 2021, 13, 3456. https://doi.org/10.3390/nu13103456
Sun Y, Zhang P, Li Y, He FJ, Wu J, Xu J, Zhang X, Li X, Song J. Association of Sodium, Potassium and Sodium-to-Potassium Ratio with Urine Albumin Excretion among the General Chinese Population. Nutrients. 2021; 13(10):3456. https://doi.org/10.3390/nu13103456
Chicago/Turabian StyleSun, Yuewen, Puhong Zhang, Yuan Li, Feng J. He, Jing Wu, Jianwei Xu, Xiaochang Zhang, Xian Li, and Jing Song. 2021. "Association of Sodium, Potassium and Sodium-to-Potassium Ratio with Urine Albumin Excretion among the General Chinese Population" Nutrients 13, no. 10: 3456. https://doi.org/10.3390/nu13103456
APA StyleSun, Y., Zhang, P., Li, Y., He, F. J., Wu, J., Xu, J., Zhang, X., Li, X., & Song, J. (2021). Association of Sodium, Potassium and Sodium-to-Potassium Ratio with Urine Albumin Excretion among the General Chinese Population. Nutrients, 13(10), 3456. https://doi.org/10.3390/nu13103456






