Omega-3 Fatty Acid and Iron Supplementation Alone, but Not in Combination, Lower Inflammation and Anemia of Infection in Mycobacterium tuberculosis-Infected Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Ethics Statement
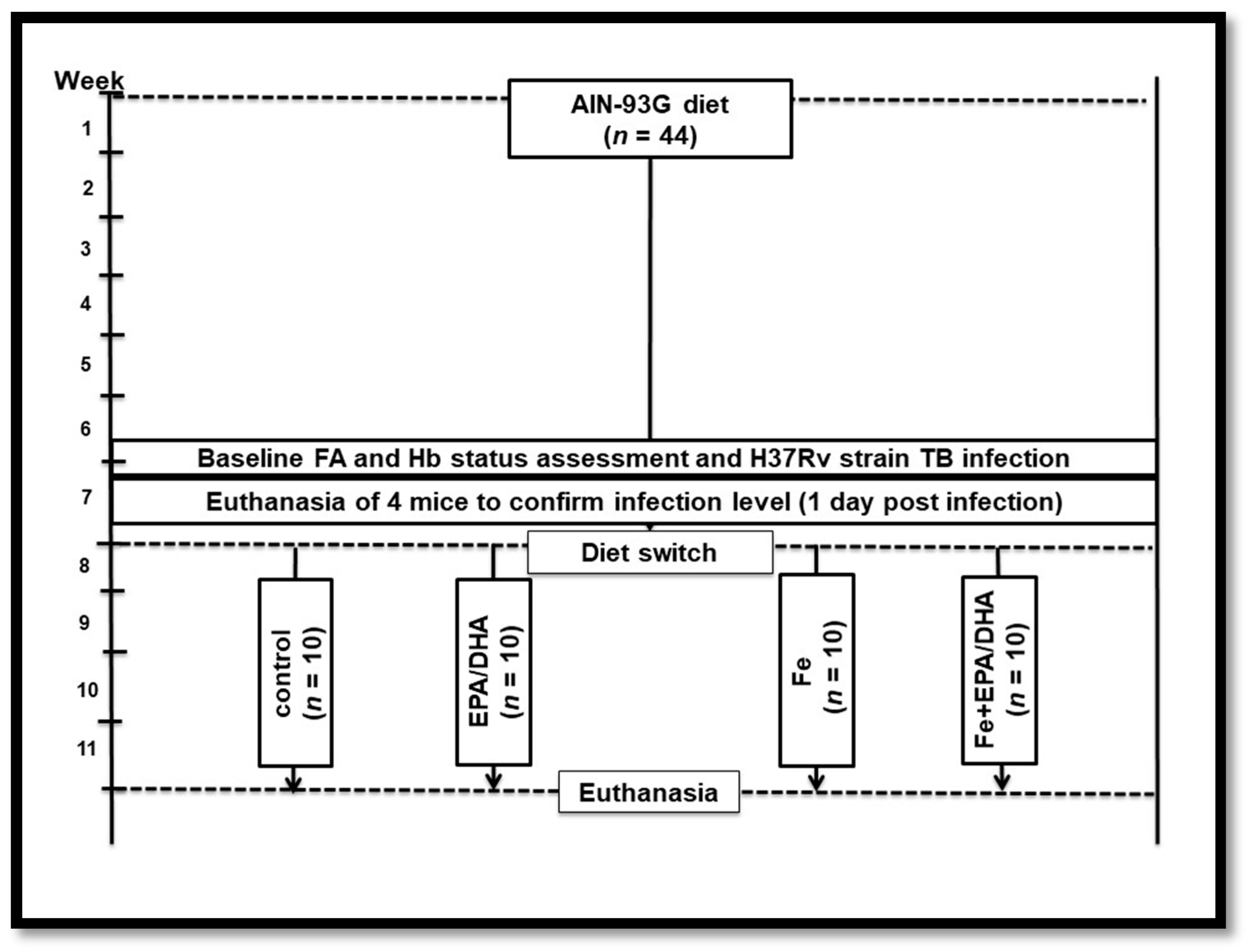
2.2. Experimental Design and Animal Diets
2.3. Aerosol Infection
2.4. Endpoint Blood and Tissue Collection
2.5. Total Phospholipid FA Composition Analysis
2.6. Lipid Mediator Analysis
2.7. Cytokine Analysis
2.8. Markers of Iron Status and Anemia of Infection
2.9. Lung Histopathology Analysis
2.10. Flow Cytometry
2.11. Bacterial Load Determination
2.12. Statistical Analyses
3. Results
3.1. Food Intake and Body Weight Gain
3.2. Total Phospholipid FA Composition of RBCs, PBMCs, and Crude Lung Homogenates
3.3. Biomarkers of Iron Status and Anemia of Infection
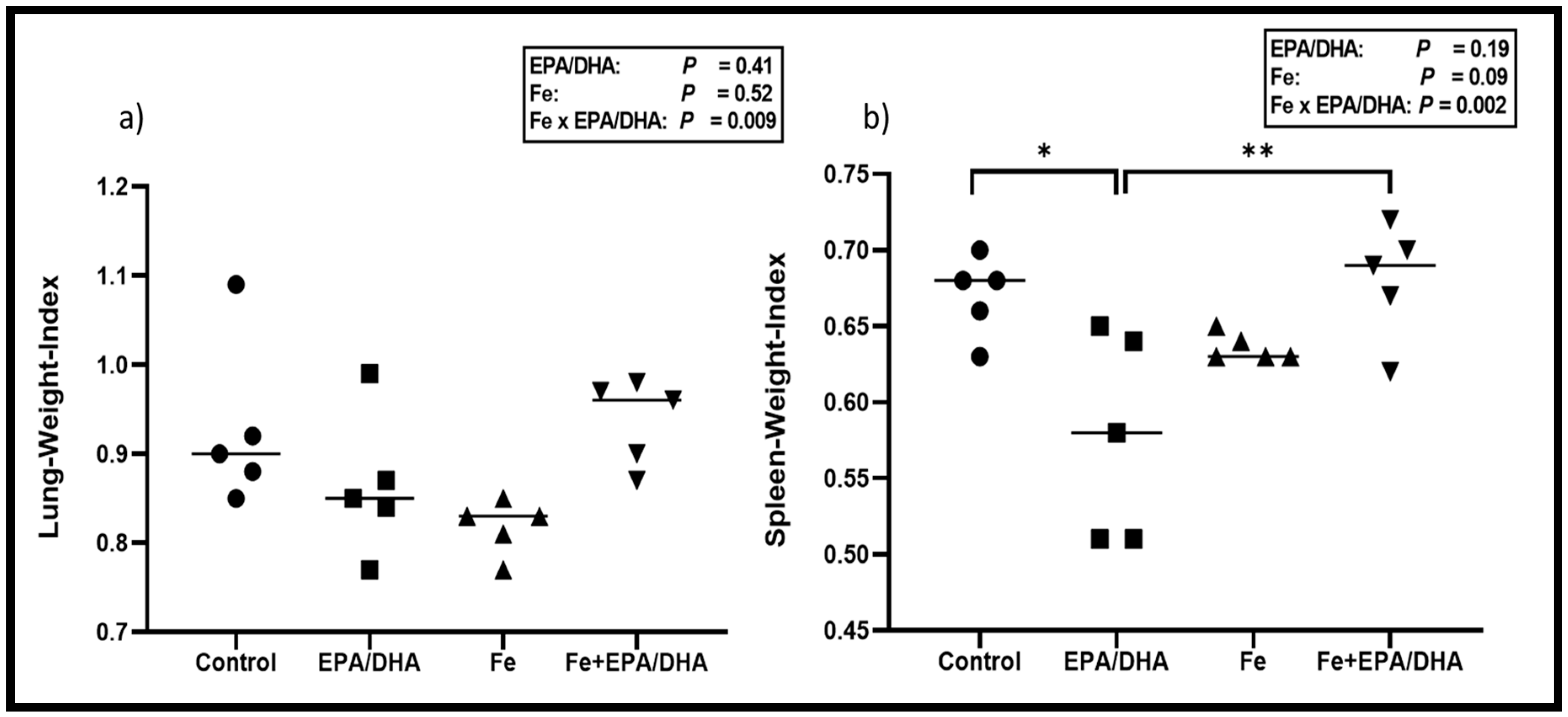
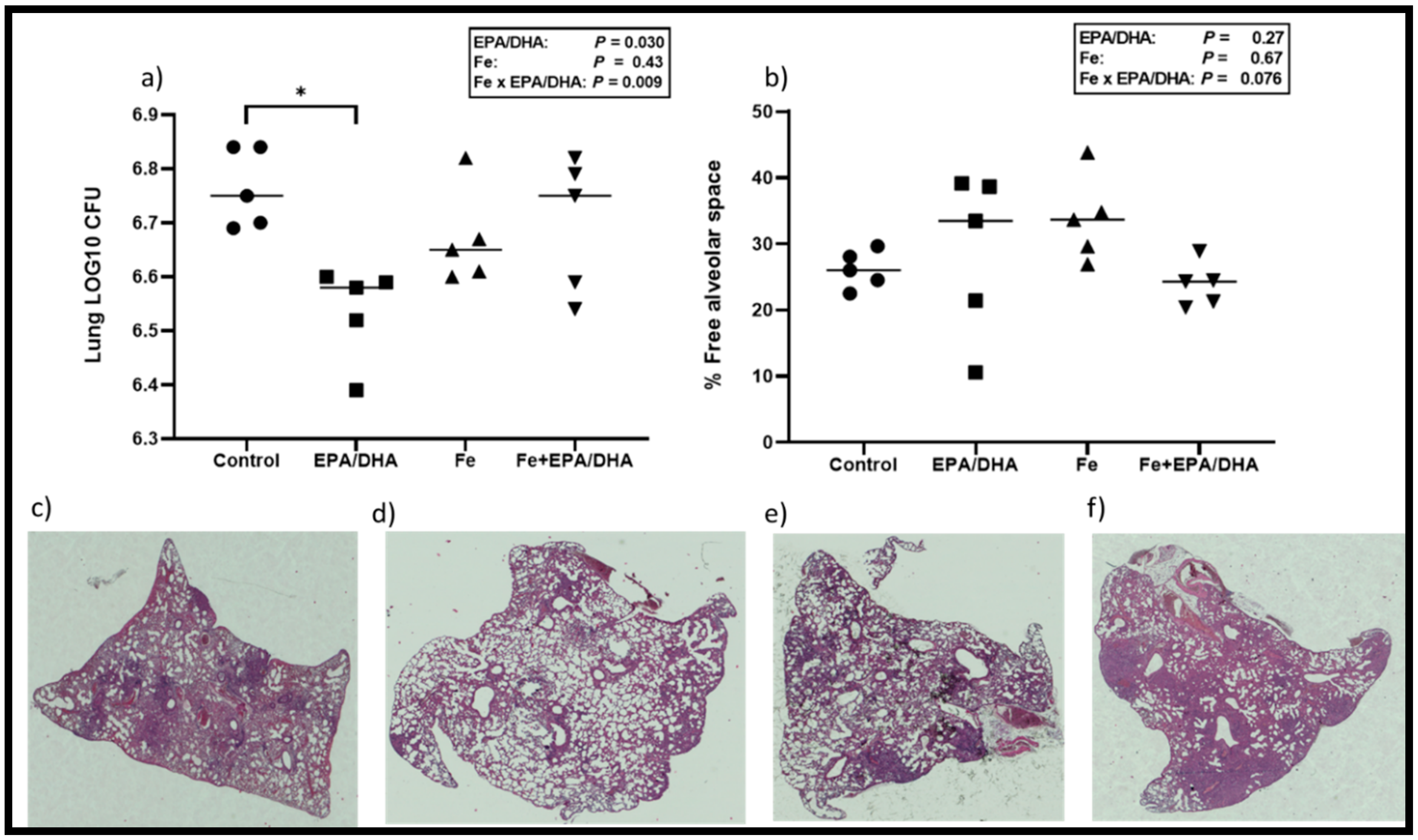
3.4. Tuberculosis Related Clinical Outcomes
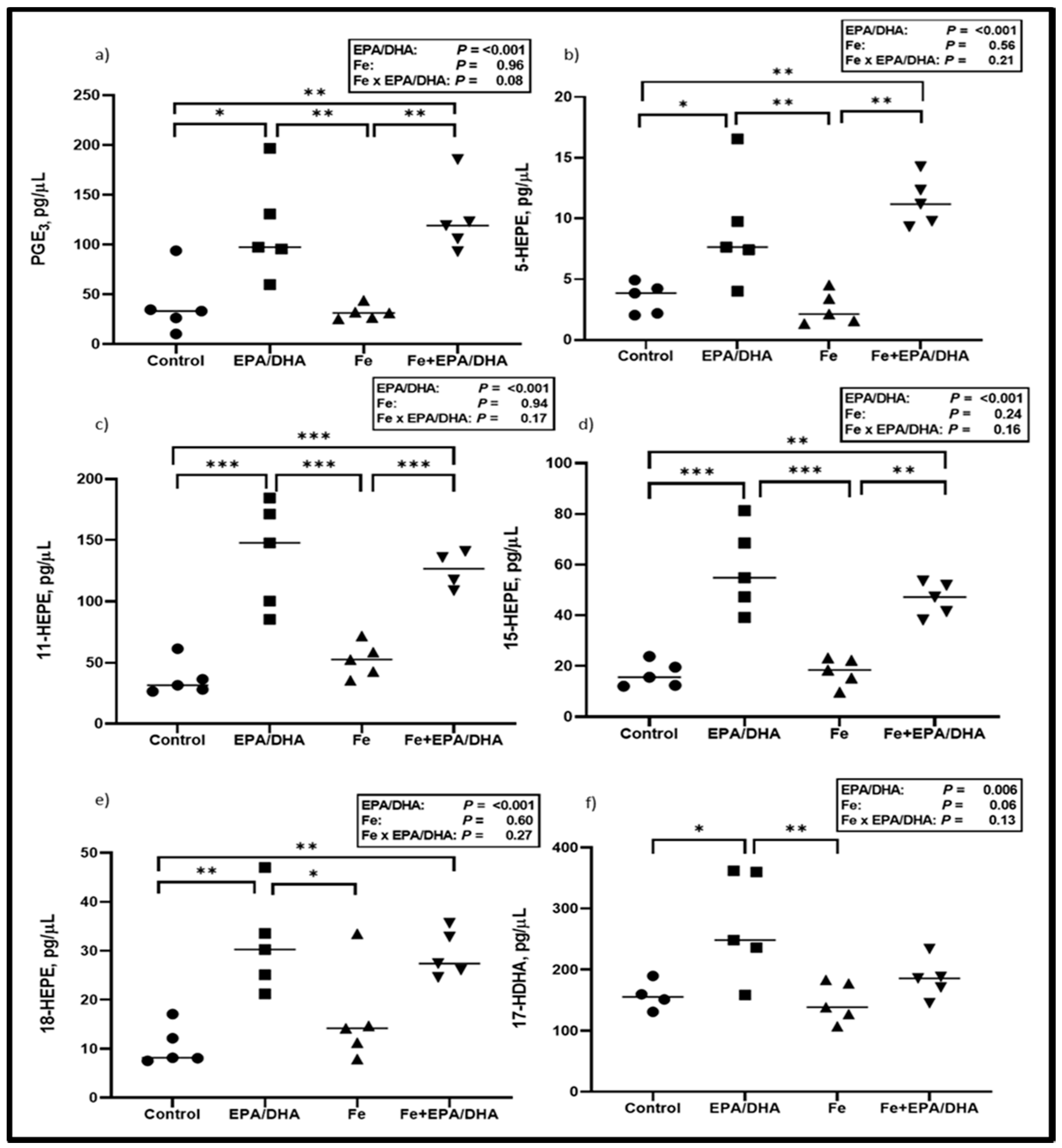
3.5. Lipid Mediators in Crude Lung Homogenates
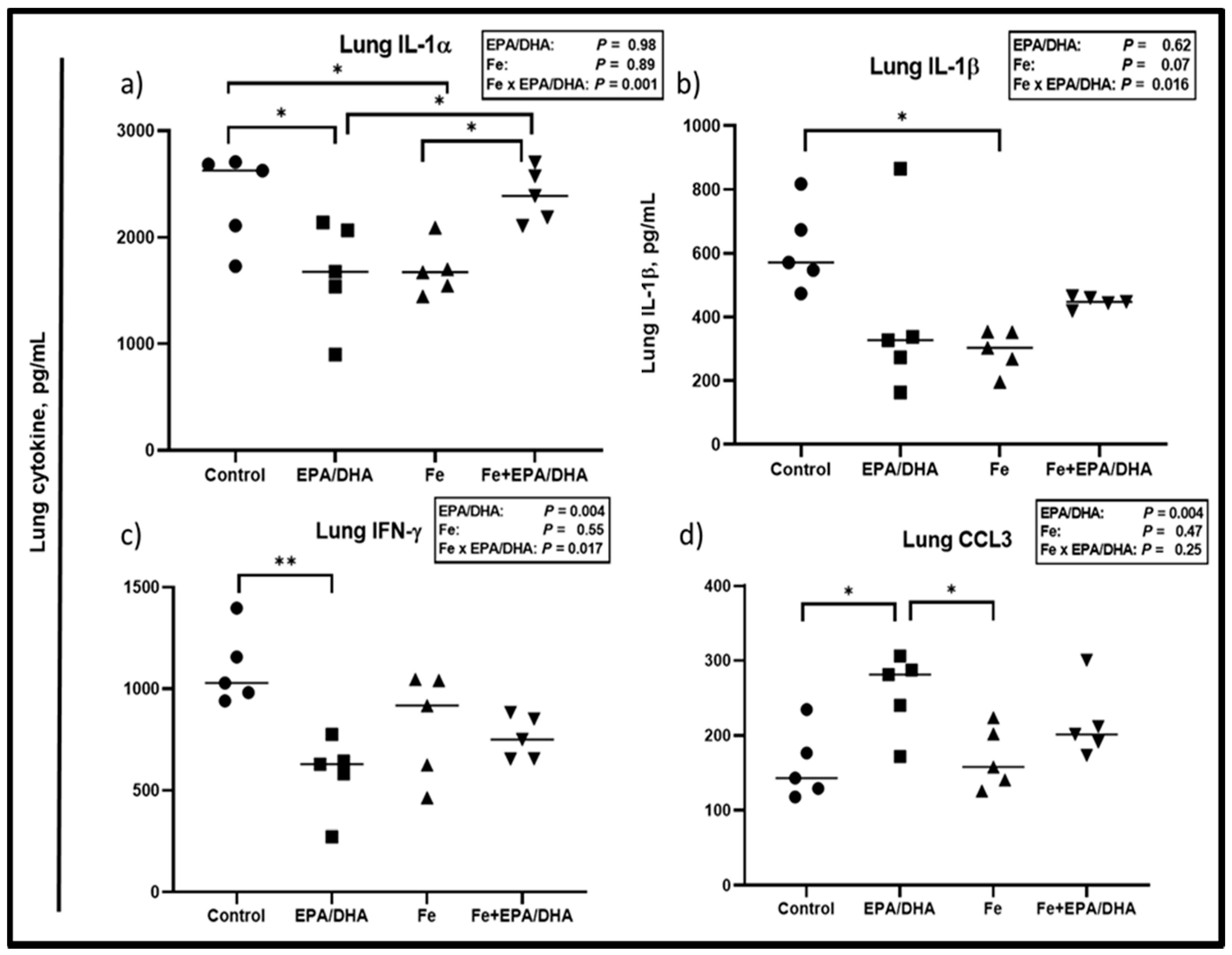
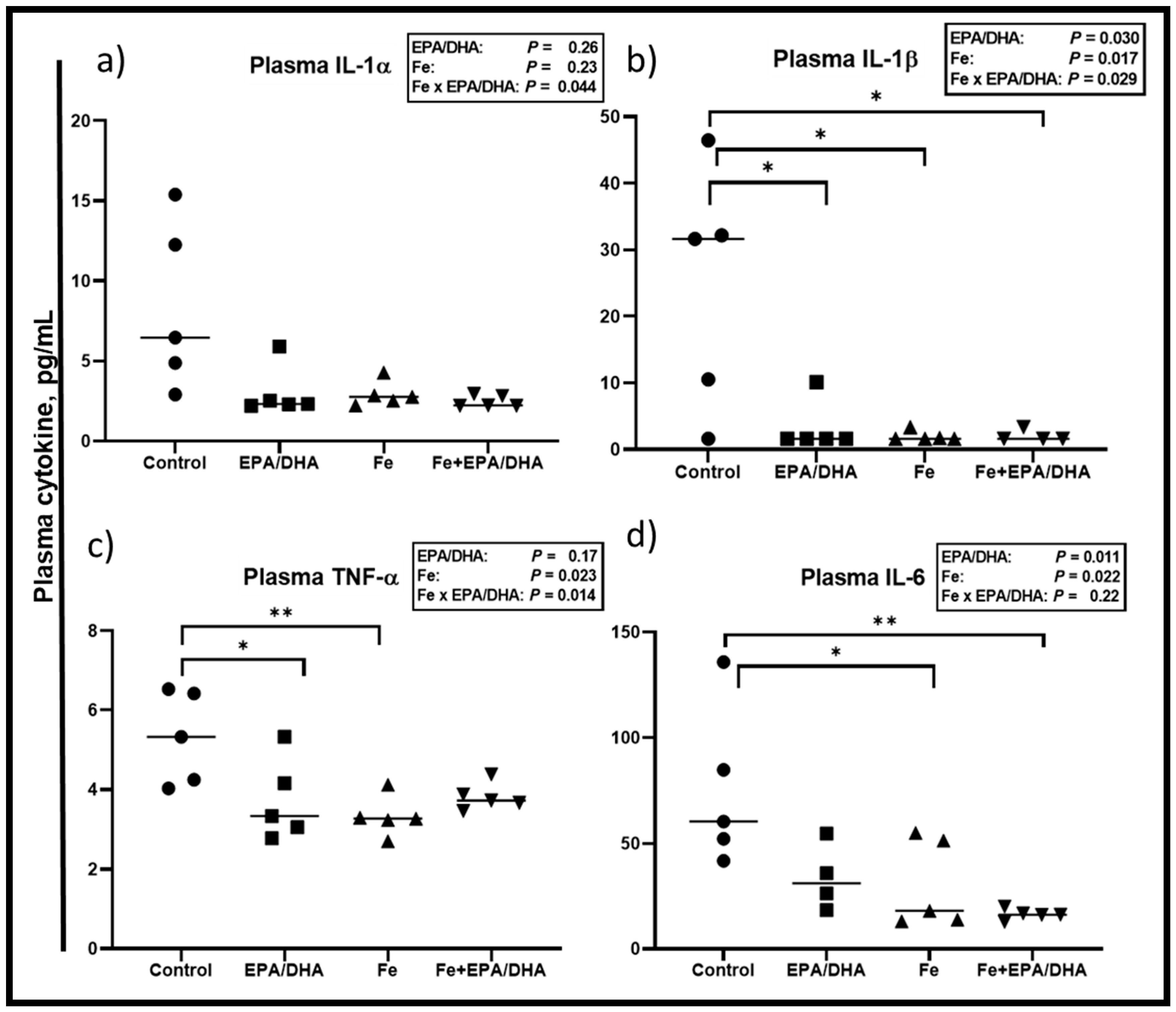
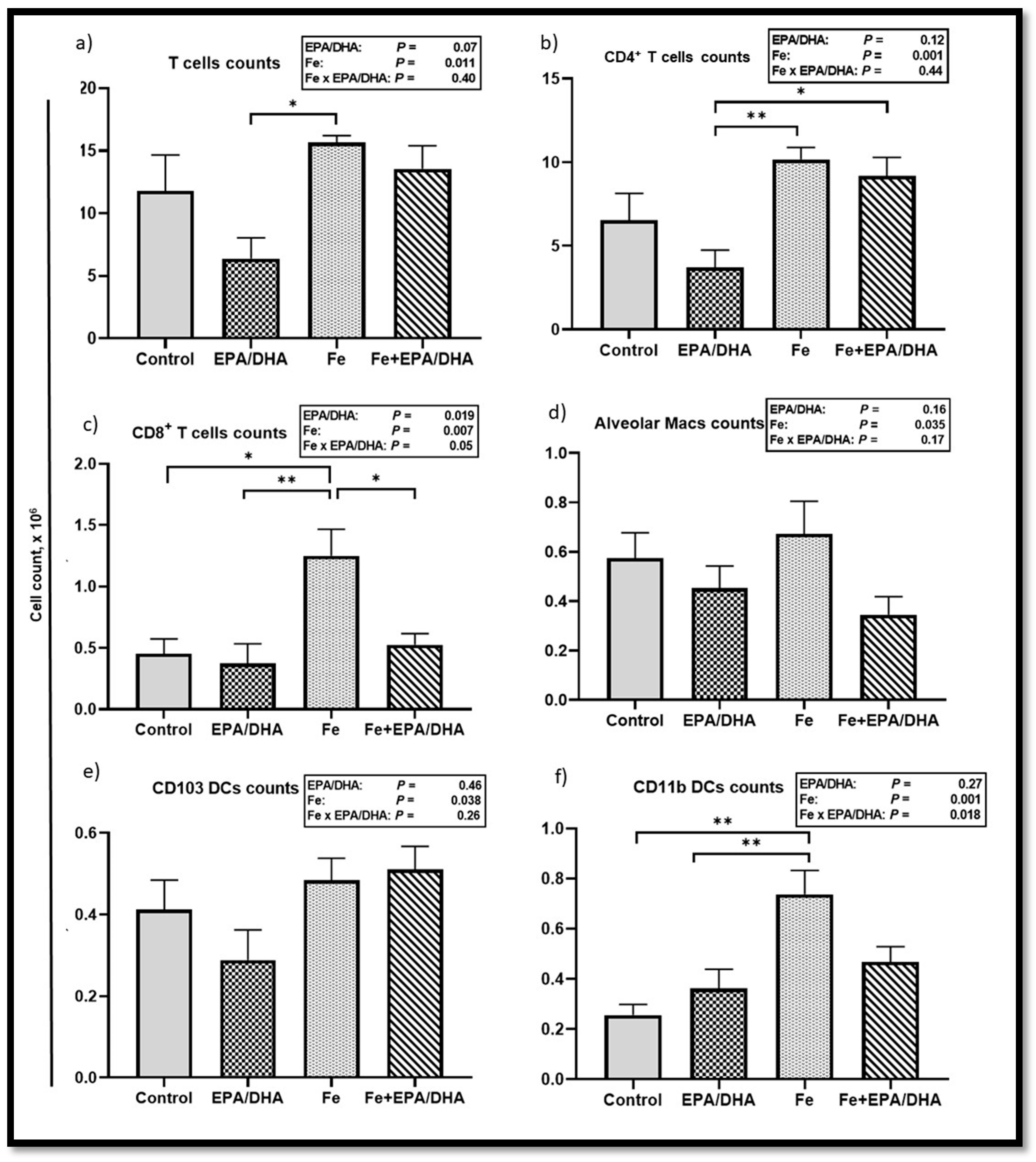
3.6. Other Markers of the Immune and Inflammatory Response
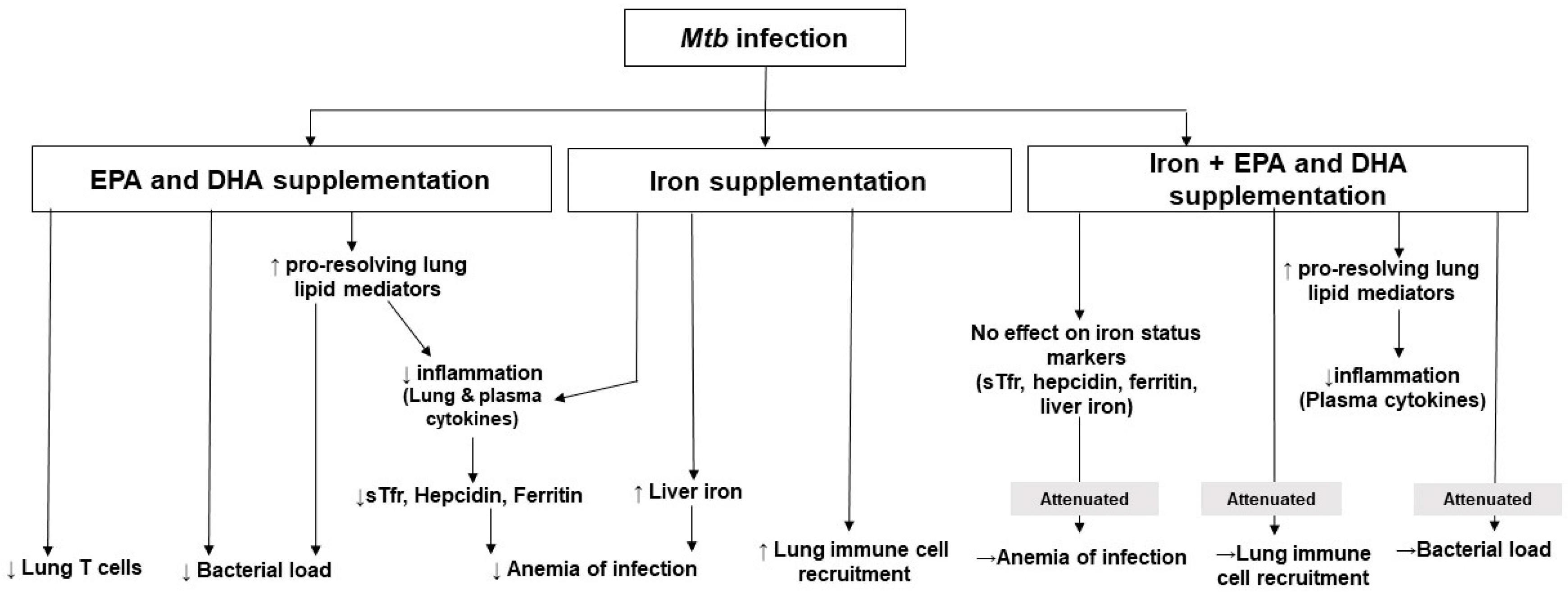
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Devi, U.; Rao, C.M.; Srivastava, V.K.; Rath, P.K.; Das, B.S. Effect of iron supplementation on mild to moderate anaemia in pulmonary tuberculosis. Br. J. Nutr. 2003, 90, 541–550. [Google Scholar] [CrossRef] [PubMed]
- Hella, J.; Cercamondi, C.I.; Mhimbira, F.; Sasamalo, M.; Stoffel, N.; Zwahlen, M.; Bodmer, T.; Gagneux, S.; Reither, K.; Zimmermann, M.B. Anemia in tuberculosis cases and household controls from Tanzania: Contribution of disease, coinfections, and the role of hepcidin. PLoS ONE 2018, 13, e0195985. [Google Scholar] [CrossRef] [PubMed]
- Isanaka, S.; Mugusi, F.; Urassa, W.; Willett, W.C.; Bosch, R.J.; Villamor, E.; Spiegelman, D.; Duggan, C.; Fawzi, W.W. Iron deficiency and anemia predict mortality in patients with tuberculosis. J. Nutr. 2011, 142, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Karyadi, E.; Schultink, W.; Nelwan, R.H.; Gross, R.; Amin, Z.; Dolmans, W.M.; van der Meer, J.W.; Hautvast, J.G.J.; West, C.E. Poor micronutrient status of active pulmonary tuberculosis patients in Indonesia. J. Nutr. 2000, 130, 2953–2958. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.W.; Kang, Y.; Yoon, Y.S.; Um, S.-W.; Lee, S.M.; Yoo, C.-G.; Kim, Y.W.; Han, S.K.; Shim, Y.-S.; Yim, J.-J. The prevalence and evolution of anemia associated with tuberculosis. J. Korean Med. Sci. 2006, 21, 1028–1032. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Taparia, M.P.; Yadav, D.; Koolwal, S. Study of Iron Metabolism in Pulmonary Tuberculosis Patients. Int. J. Health Sci. Res. 2018, 8, 70–77. [Google Scholar]
- Isanaka, S.; Aboud, S.; Mugusi, F.; Bosch, R.J.; Willett, W.C.; Spiegelman, D.; Duggan, C.; Fawzi, W.W. Iron status predicts treatment failure and mortality in tuberculosis patients: A prospective cohort study from Dar es Salaam, Tanzania. PLoS ONE 2012, 7, e37350. [Google Scholar] [CrossRef]
- Nagu, T.J.; Spiegelman, D.; Hertzmark, E.; Aboud, S.; Makani, J.; Matee, M.I.; Fawzi, W.; Mugusi, F. Anemia at the initiation of tuberculosis therapy is associated with delayed sputum conversion among pulmonary tuberculosis patients in Dar-es-Salaam, Tanzania. PLoS ONE 2014, 9, e91229. [Google Scholar] [CrossRef]
- Shimazaki, T.; Marte, S.; Saludar, N.; Dimaano, E.; Salva, E.; Ariyoshi, K.; Villarama, J.; Suzuki, M. Risk factors for death among hospitalised tuberculosis patients in poor urban areas in Manila, The Philippines. Int. J. TB Lung Dis. 2013, 17, 1420–1426. [Google Scholar] [CrossRef]
- Kerkhoff, A.D.; Meintjes, G.; Burton, R.; Vogt, M.; Wood, R.; Lawn, S.D. Relationship between blood concentrations of hepcidin and anemia severity, mycobacterial burden, and mortality among patients with HIV-associated tuberculosis. J. Infect. Dis. 2015, 213, 61–70. [Google Scholar] [CrossRef]
- Nemeth, E.; Tuttle, M.S.; Powelson, J.; Vaughn, M.B.; Donovan, A.; Ward, D.M.; Ganz, T.; Kaplan, J. Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science 2004, 306, 2090–2093. [Google Scholar] [CrossRef] [PubMed]
- Ganz, T.; Nemeth, E. Hepcidin and iron homeostasis. BBA Mol. Cell Res. 2012, 1823, 1434–1443. [Google Scholar] [CrossRef]
- Aschemeyer, S.; Qiao, B.; Stefanova, D.; Valore, E.V.; Sek, A.C.; Ruwe, T.A.; Vieth, K.R.; Jung, G.; Casu, C.; Rivella, S. Structure-function analysis of ferroportin defines the binding site and an alternative mechanism of action of hepcidin. Blood 2018, 131, 899–910. [Google Scholar] [CrossRef] [PubMed]
- Agoro, R.; Mura, C. Iron Supplementation Therapy, A Friend and Foe of Mycobacterial Infections? Pharmaceuticals 2019, 12, 75. [Google Scholar] [CrossRef] [PubMed]
- Kurthkoti, K.; Amin, H.; Marakalala, M.J.; Ghanny, S.; Subbian, S.; Sakatos, A.; Livny, J.; Fortune, S.M.; Berney, M.; Rodriguez, G.M. The capacity of Mycobacterium tuberculosis to survive iron starvation might enable it to persist in iron-deprived microenvironments of human granulomas. mBio 2017, 8. [Google Scholar] [CrossRef]
- Weiss, G.; Ganz, T.; Goodnough, L.T. Anemia of inflammation. Blood 2019, 133, 40–50. [Google Scholar] [CrossRef]
- Shaw, J.G.; Friedman, J.F. Iron deficiency anemia: Focus on infectious diseases in lesser developed countries. Anemia 2011. [Google Scholar] [CrossRef]
- Ganz, T. Anemia of inflammation. N. Engl. J. Med. 2019, 381, 1148–1157. [Google Scholar] [CrossRef]
- Kerkhoff, A.; Meintjes, G.; Opie, J.; Vogt, M.; Jhilmeet, N.; Wood, R.; Lawn, S. Anaemia in patients with HIV-associated TB: Relative contributions of anaemia of chronic disease and iron deficiency. Int. J. Tuberc. Lung Dis. 2016, 20, 193–201. [Google Scholar] [CrossRef]
- Minchella, P.A.; Donkor, S.; Owolabi, O.; Sutherland, J.S.; McDermid, J.M. Complex anemia in tuberculosis: The need to consider causes and timing when designing interventions. Clin. Infect. Dis. 2014, 60, 764–772. [Google Scholar] [CrossRef]
- Jordao, L.; Lengeling, A.; Bordat, Y.; Boudou, F.; Gicquel, B.; Neyrolles, O.; Becker, P.D.; Guzman, C.A.; Griffiths, G.; Anes, E. Effects of omega-3 and-6 fatty acids on Mycobacterium tuberculosis in macrophages and in mice. Microbes Infect. 2008, 10, 1379–1386. [Google Scholar] [CrossRef] [PubMed]
- Nienaber, A.; Ozturk, M.O.; Dolman, R.C.; Blaauw, R.; Zandberg, L.; King, S.; Britz, M.; Hayford, F.E.A.; Brombacher, F.; Loots, D.T.; et al. Omega-3 long-chain polyunsaturated fatty acids promote antibacterial and inflammation-resolving effects in Mycobacterium tuberculosis-infected C3HeB/FeJ mice, dependent on fatty acid status. Br. J. Nutr. 2020. under review. [Google Scholar]
- Serhan, C.N.; Chiang, N.; Dalli, J. New pro-resolving n-3 mediators bridge resolution of infectious inflammation to tissue regeneration. Mol. Asp. Med. 2017, 64, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Malan, L.; Baumgartner, J.; Calder, P.C.; Zimmermann, M.B.; Smuts, C.M. n–3 Long-chain PUFAs reduce respiratory morbidity caused by iron supplementation in iron-deficient South African schoolchildren: A randomized, double-blind, placebo-controlled intervention. Am. J. Clin. Nutr. 2014, 101, 668–679. [Google Scholar] [CrossRef] [PubMed]
- Malan, L.; Baumgartner, J.; Zandberg, L.; Calder, P.; Smuts, C. Iron and a mixture of DHA and EPA supplementation, alone and in combination, affect bioactive lipid signalling and morbidity of iron deficient South African school children in a two-by-two randomised controlled trial. Prostaglandins Leukot. Essent. Fatty Acids 2016, 105, 15–25. [Google Scholar] [CrossRef]
- Reeves, P.G.; Nielsen, F.H.; Fahey, G.C., Jr. AIN-93 purified diets for laboratory rodents: Final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J. Nutr. 1993, 11, 1939–1951. [Google Scholar] [CrossRef]
- Guler, R.; Parihar, S.P.; Spohn, G.; Johansen, P.; Brombacher, F.; Bachmann, M.F. Blocking IL-1α but not IL-1β increases susceptibility to chronic Mycobacterium tuberculosis infection in mice. Vaccine 2011, 29, 1339–1346. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Stanley, G.S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
- Parihar, S.; Ozturk, M.; Marakalala, M.; Loots, D.; Hurdayal, R.; Beukes, D.; Van Reenen, M.; Zak, D.; Mbandi, S.; Darboe, F. Protein kinase C-delta (PKC delta), a marker of inflammation and tuberculosis disease progression in humans, is important for optimal macrophage killing effector functions and survival in mice. Mucosal Immunol. 2018, 11, 578–579. [Google Scholar]
- Parihar, S.P.; Guler, R.; Khutlang, R.; Lang, D.M.; Hurdayal, R.; Mhlanga, M.M.; Suzuki, H.; Marais, A.D.; Brombacher, F. Statin Therapy Reduces the Mycobacterium tuberculosis Burden in Human Macrophages and in Mice by Enhancing Autophagy and Phagosome Maturation. J. Infect. Dis. 2014, 209, 754–763. [Google Scholar] [CrossRef]
- Rohner, F.; Namaste, S.M.; Larson, L.M.; Addo, O.Y.; Mei, Z.; Suchdev, P.S.; Williams, A.M.; Sakr Ashour, F.A.; Rawat, R.; Raiten, D.J. Adjusting soluble transferrin receptor concentrations for inflammation: Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) project. Am. J. Clin. Nutr. 2017, 106, 372S–382S. [Google Scholar] [PubMed]
- Schmidt, P.J. Regulation of iron metabolism by hepcidin under conditions of inflammation. J. Biol. Chem. 2015, 290, 18975–18983. [Google Scholar] [CrossRef] [PubMed]
- Ganz, T. Iron and infection. Int. J Hematol. 2018, 107, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Cole, S.; Brosch, R.; Parkhill, J.; Garnier, T.; Churcher, C.; Harris, D.; Gordon, S.; Eiglmeier, K.; Gas, S.; Barry, C., III. Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature 1998, 393, 537. [Google Scholar] [CrossRef]
- Lee, P.; Peng, H.; Gelbart, T.; Wang, L.; Beutler, E. Regulation of hepcidin transcription by interleukin-1 and interleukin-6. Proc. Natl. Acad. Sci. USA 2005, 102, 1906–1910. [Google Scholar] [CrossRef]
- Nemeth, E.; Rivera, S.; Gabayan, V.; Keller, C.; Taudorf, S.; Pedersen, B.K.; Ganz, T. IL-6 mediates hypoferremia of inflammation by inducing the synthesis of the iron regulatory hormone hepcidin. J. Clin. Investig. 2004, 113, 1271–1276. [Google Scholar] [CrossRef]
- Nicolas, G.; Chauvet, C.; Viatte, L.; Danan, J.L.; Bigard, X.; Devaux, I.; Beaumont, C.; Kahn, A.; Vaulont, S. The gene encoding the iron regulatory peptide hepcidin is regulated by anemia, hypoxia, and inflammation. J. Clin. Investig. 2002, 110, 1037–1044. [Google Scholar] [CrossRef]
- Millonig, G.; Ganzleben, I.; Peccerella, T.; Casanovas, G.; Brodziak-Jarosz, L.; Breitkopf-Heinlein, K.; Dick, T.P.; Seitz, H.-K.; Muckenthaler, M.U.; Mueller, S. Sustained submicromolar H2O2 levels induce hepcidin via signal transducer and activator of transcription 3 (STAT3). J. Biol. Chem. 2012, 287, 37472–37482. [Google Scholar] [CrossRef]
- Armitage, A.E.; Eddowes, L.A.; Gileadi, U.; Cole, S.; Spottiswoode, N.; Selvakumar, T.A.; Ho, L.-P.; Townsend, A.R.; Drakesmith, H. Hepcidin regulation by innate immune and infectious stimuli. Blood 2011, 118, 4129–4139. [Google Scholar] [CrossRef]
- Harms, K.; Kaiser, T. Beyond soluble transferrin receptor: Old challenges and new horizons. Best Pract. Res. Clin. Endocrinol. 2015, 29, 799–810. [Google Scholar] [CrossRef]
- Truman-Rosentsvit, M.; Berenbaum, D.; Spektor, L.; Cohen, L.A.; Belizowsky-Moshe, S.; Lifshitz, L.; Ma, J.; Li, W.; Kesselman, E.; Abutbul-Ionita, I. Ferritin is secreted via 2 distinct nonclassical vesicular pathways. Blood 2018, 131, 342–352. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.J.; Frazer, D.M. Current understanding of iron homeostasis. Am. J. Clin. Nutr. 2017, 106, 1559S–1566S. [Google Scholar] [CrossRef]
- Borthwick, L. The IL-1 cytokine family and its role in inflammation and fibrosis in the lung. Semin. Immunopathol. 2016, 38, 517–534. [Google Scholar] [CrossRef]
- Roca, F.J.; Ramakrishnan, L. TNF dually mediates resistance and susceptibility to mycobacteria via mitochondrial reactive oxygen species. Cell 2013, 153, 521–534. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.P.; Moideen, K.; Banurekha, V.V.; Nair, D.; Babu, S. Plasma pro-inflammatory cytokines are markers of disease severity and bacterial burden in pulmonary tuberculosis. Open For. Infect. Diss. 2019, 6, ofz257. [Google Scholar] [CrossRef]
- Del Bò, C.; Contino, D.; Riso, P.; Simonetti, P.; Ciappellano, S. Effect of horse meat consumption on iron status, lipid profile and fatty acid composition of red blood cell membrane: Preliminary study. In Riunione Nazionale SINU: Nutrizione, la Pietra D’Angolo; SINU: Milan, Italy, 2009; Volume 3, p. 1. [Google Scholar]
- Smuts, C.; Tichelaar, H.; Van Jaarsveld, P.; Badenhorst, C.; Kruger, M.; Laubscher, R.; Mansvelt, E.; Benade, A. The effect of iron fortification on the fatty acid composition of plasma and erythrocyte membranes in primary school children with and without iron deficiency. Prostaglandins Leukot. Essent. Fatty Acids 1995, 52, 59–67. [Google Scholar] [CrossRef]
- Agoro, R.; Benmerzoug, S.; Rose, S.; Bouyer, M.; Gozzelino, R.; Garcia, I.; Ryffel, B.; Quesniaux, V.F.; Mura, C. An iron-rich diet decreases the mycobacterial burden and correlates with hepcidin upregulation, lower levels of proinflammatory mediators, and increased T-cell recruitment in a model of mycobacterium bovis Bacille Calmette-Guerin infection. J. Infect. Dis. 2017, 216, 907–918. [Google Scholar] [CrossRef]
- Kolloli, A.; Singh, P.; Rodriguez, G.M.; Subbian, S. Effect of Iron Supplementation on the Outcome of Non-Progressive Pulmonary Mycobacterium tuberculosis Infection. J. Clin. Med. 2019, 8, 1155. [Google Scholar] [CrossRef]
- Serafín-López, J.; Chacón-Salinas, R.; Muñoz-Cruz, S.; Enciso-Moreno, J.; Estrada-Parra, S.A.; Estrada-Garcia, I. The effect of iron on the expression of cytokines in macrophages infected with Mycobacterium tuberculosis. Scand. J. Immunol. 2004, 60, 329–337. [Google Scholar] [CrossRef]
- McFarland, C.T.; Fan, Y.-Y.; Chapkin, R.S.; Weeks, B.R.; McMurray, D.N. Dietary polyunsaturated fatty acids modulate resistance to Mycobacterium tuberculosis in guinea pigs. J. Nutr. 2008, 138, 2123–2128. [Google Scholar] [CrossRef]
- Calder, P.C.; Newsholme, E.A. Polyunsaturated fatty acids suppress human peripheral blood lymphocyte proliferation and interleukin-2 production. Clin. Sci. 1992, 82, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. The relationship between the fatty acid composition of immune cells and their function. Prostaglandins Leukot. Essent. Fatty Acids 2008, 79, 101–108. [Google Scholar] [CrossRef]
- Cherayil, B.J. Iron and immunity: Immunological consequences of iron deficiency and overload. Arch. Immunol. Ther. Exp. 2010, 58, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Jabara, H.H.; Boyden, S.E.; Chou, J.; Ramesh, N.; Massaad, M.J.; Benson, H.; Bainter, W.; Fraulino, D.; Rahimov, F.; Sieff, C. A missense mutation in TFRC, encoding transferrin receptor 1, causes combined immunodeficiency. Nat. Genet. 2016, 48, 74. [Google Scholar] [CrossRef] [PubMed]
- Bresnahan, K.A.; Chileshe, J.; Arscott, S.; Nuss, E.; Surles, R.; Masi, C.; Kafwembe, E.; Tanumihardjo, S.A. The acute phase response affected traditional measures of micronutrient status in rural Zambian children during a randomized, controlled feeding trial. J. Nutr. 2014, 144, 972–978. [Google Scholar] [CrossRef]
- Ifeanyi, O.E. A Review on Iron Homeostasis and Anaemia in Pulmonary Tuberculosis. Int. J. Healthc. Med. Sci. 2018, 4, 84–89. [Google Scholar]
- Azcárate, I.G.; Sánchez-Jaut, S.; Marín-García, P.; Linares, M.; Pérez-Benavente, S.; García-Sánchez, M.; Uceda, J.; Kamali, A.N.; Morán-Jiménez, M.-J.; Puyet, A. Iron supplementation in mouse expands cellular innate defences in spleen and defers lethal malaria infection. BBA Mol. Basis Dis. 2017, 1863, 3049–3059. [Google Scholar] [CrossRef]
- Stefanova, D.; Raychev, A.; Arezes, J.; Ruchala, P.; Gabayan, V.; Skurnik, M.; Dillon, B.J.; Horwitz, M.A.; Ganz, T.; Bulut, Y. Endogenous hepcidin and its agonist mediate resistance to selected infections by clearing non–transferrin-bound iron. Blood 2017, 130, 245–257. [Google Scholar] [CrossRef]
- Harrington-Kandt, R.; Stylianou, E.; Eddowes, L.A.; Lim, P.J.; Stockdale, L.; Pinpathomrat, N.; Bull, N.; Pasricha, J.; Ulaszewska, M.; Beglov, Y. Hepcidin deficiency and iron deficiency do not alter tuberculosis susceptibility in a murine M.tb infection model. PLoS ONE 2018, 13, e0191038. [Google Scholar] [CrossRef]
- Cronjé, L.; Edmondson, N.; Eisenach, K.D.; Bornman, L. Iron and iron chelating agents modulate Mycobacterium tuberculosis growth and monocyte-macrophage viability and effector functions. FEMS Immunol. Med. Microbiol. 2005, 45, 103–112. [Google Scholar] [CrossRef]
- Kochan, I. The role of iron in bacterial infections, with special consideration of host-tubercle bacillus interaction. In Current Topics in Microbiology Immunology; Springer: Berlin/Heidelberg, Germany, 1973; Volume 60, pp. 1–30. [Google Scholar]
- Lounis, N.; Truffot-Pernot, C.; Grosset, J.; Gordeuk, V.R.; Boelaert, J.R. Iron and Mycobacterium tuberculosis infection. J. Clin. Virol. 2001, 20, 123–126. [Google Scholar] [CrossRef]
- Schaible, U.E.; Collins, H.L.; Priem, F.; Kaufmann, S.H. Correction of the iron overload defect in β-2-microglobulin knockout mice by lactoferrin abolishes their increased susceptibility to tuberculosis. J. Exp. Med. 2002, 196, 1507–1513. [Google Scholar] [CrossRef] [PubMed]
- Lenaerts, A.; Barry, C.E., III; Dartois, V. Heterogeneity in tuberculosis pathology, microenvironments and therapeutic responses. Immunol. Rev. 2015, 264, 288–307. [Google Scholar] [CrossRef] [PubMed]
| Group | Iron | Fat Source | LA | ALA | AA | DHA | EPA |
|---|---|---|---|---|---|---|---|
| Per 100 g diet | |||||||
| Control and uninfected | 40 ppm | 7 g soybean oil 3 g Coconut oil | 3.54 g | 0.44 g | <0.01 g | <0.01 g | <0.01 g |
| Fe | 130 ppm | 7 g Soybean oil 3 g Coconut oil | 3.48 g | 0.44 g | <0.01 g | <0.01 | <0.01 g |
| EPA/DHA | 40 ppm | 7 g Soybean oil 2.7 g Coconut oil 3 g Incromega TG4030 oil | 1.3 g | 0.43 g | <0.01 g | 0.06 g | 0.09 g |
| Fe+EPA/DHA | 123 ppm | 7 g Soybean oil 2.7 g Coconut oil 3 g Incromega TG4030 oil | 3.44 g | 0.43 g | <0.01 g | 0.06 g | 0.09 g |
| % Total FA | Control | EPA/DHA | Fe | Fe+EPA/DHA | p-Value 2 | ||
|---|---|---|---|---|---|---|---|
| EPA/DHA | Fe | Fe × EPA/DHA | |||||
| 20:5(n-3) (EPA) | |||||||
| RBC | 0.13 ± 0.00 b | 0.51 ± 0.04 a | 0.12 ± 0.00 b | 0.43 ± 0.02 a | <0.001 | 0.041 | 0.17 |
| PBMC | 0.20 ± 0.01 b | 0.89 ± 0.04 a | 0.16 ± 0.01 b | 0.55 ± 0.11 a | <0.001 | 0.038 | 0.19 |
| Lung | 0.17 ± 0.01 b | 0.40 ± 0.01 a | 0.17 ± 0.03 b | 0.38 ± 0.01 a | <0.001 | 0.74 | 0.63 |
| 22:6(n-3) (DHA) | |||||||
| RBC | 6.09 ± 0.21 b,c | 7.48 ± 0.41 a | 5.58 ± 0.22 c | 6.83 ± 0.25 a,b | <0.001 | 0.060 | 0.82 |
| PBMC | 9.04 ± 0.20 a | 9.82 ± 0.22 a | 7.57 ± 0.29 b | 9.52 ± 0.39 a | <0.001 | 0.008 | 0.060 |
| Lung | 8.08 ± 0.15 b | 9.68 ± 0.28 a | 7.99 ± 0.08 b | 9.86 ± 0.13 a | <0.001 | 0.75 | 0.38 |
| Total n-3 LCPUFAs | |||||||
| RBC | 6.63 ± 0.20 b,c | 8.59 ± 0.48 a | 6.06 ± 0.24 c | 7.80 ± 0.27 a,b | <0.001 | 0.046 | 0.74 |
| PBMC | 10.60 ± 0.21 b | 12.40 ± 0.33 a | 8.76 ± 0.42 c | 11.80 ± 0.64 a,b | <0.001 | 0.011 | 0.20 |
| Lung | 10.00 ± 0.14 b | 12.60 ± 0.26 a | 9.73 ± 0.10 b | 12.90 ± 0.12 a | <0.001 | 0.96 | 0.07 |
| 20:4(n-6) AA | |||||||
| RBC | 17.71 ± 0.27 a | 16.42 ± 0.30 a,b | 17.68 ± 0.40 a | 15.88 ± 0.28 b | <0.001 | 0.38 | 0.43 |
| PBMC | 16.36 ± 0.39 b | 14.76 ± 0.31 b | 20.94 ± 0.70 a | 17.34 ± 0.94 b | 0.001 | <0.001 | 0.14 |
| Lung | 14.40 ± 0.15 a,b | 13.32 ± 0.42 b | 14.76 ± 0.27 a | 14.31 ± 0.23 a,b | 0.023 | 0.013 | 0.25 |
| 22:5(n-6) (osbond) | |||||||
| RBC | 0.87 ± 0.10 a | 0.37 ± 0.01 c | 0.49 ± 0.01 b | 0.35 ± 0.00 c | <0.001 | 0.008 | 0.021 |
| PBMC | 1.42 ± 0.04 a | 0.74 ± 0.05 b | 1.01 ± 0.08 b | 0.80 ± 0.10 b | <0.001 | 0.025 | 0.005 |
| Lung | 1.13 ± 0.05 a | 0.54 ± 0.01 b | 1.01 ± 0.02 a | 0.54 ± 0.01 b | <0.001 | 0.27 | 0.050 |
| Total n-6 LCPUFAs | |||||||
| RBC | 20.20 ± 0.32 a | 18.7 ± 0.44 a | 20.10 ± 0.47 a,b | 18.00 ± 0.33 b | <0.001 | 0.33 | 0.45 |
| PBMC | 21.80 ± 0.41 b | 19.4 ± 0.54 b | 25.70 ± 0.78 a | 22.00 ± 1.16 b | 0.001 | <0.001 | 0.45 |
| Lung | 21.20 ± 0.19 a,b | 18.3 ± 0.50 c | 21.50 ± 0.37 a | 19.90 ± 0.35 b | <0.001 | 0.022 | 0.07 |
| Total n-6/n-3 LCPUFA ratio | |||||||
| RBC | 3.05 ± 0.05 b | 2.19 ± 0.07 c, | 3.32 ± 0.07 a | 2.31 ± 0.04 c | <0.001 | 0.004 | 0.19 |
| PBMC | 2.06 ± 0.02 b | 1.55 ±0.01 b | 2.94 ± 0.14 a | 1.91 ± 0.23 b | <0.001 | <0.001 | 0.07 |
| Lung | 2.12 ± 0.19 a | 1.45 ± 0.50 b | 2.21 ± 0.03 a | 1.54 ± 0.04 b | <0.001 | 0.039 | 0.97 |
| Iron Parameter | Non- Infected Reference (n = 3) 2 | Control (n = 5) | EPA/DHA (n = 5) | Fe (n = 5) | Fe+ EPA/DHA (n = 5) | p Value 3 | ||
|---|---|---|---|---|---|---|---|---|
| EPA/DHA | Fe | Fe × EPA/DHA | ||||||
| Hb (g/dL) | 14.4 ± 0.8 | 13.6 ± 0.3 a,b | 13.4 ± 0.3 a,b | 14.3 ± 0.4 a | 12.9 ± 0.4 b | 0.031 | 0.59 | 0.08 |
| Liver iron (µg/L) | 288.0 ± 14.0 | 210.8 ± 12.7 b,c * | 225.0 ± 16.7 b | 299.4 ± 15.9 a | 255.8 ± 18.4 a,b,c | 0.38 | 0.002 | 0.09 |
| Ferritin (ng/mL) | 26.1 ± 2.5 | 222.0 ± 43.9 a ** | 136.0 ± 32.5 a,b | 125.0 ± 23.4 b | 164.0 ± 13.8 a,b * | 0.51 | 0.63 | 0.045 |
| sTfR (ng/mL) | 2.3 ± 0.1 | 17.5 ± 2.8 a ** | 10.8 ± 2.4 b | 10.8 ± 2.2 b | 14.5 ± 0.9 a,b ** | 0.49 | 0.50 | 0.030 |
| Hepcidin (µg/mL) | 0.9 ± 1.1 | 14.3 ± 2.7 a ** | 8.1 ± 2.2 b | 8.7 ± 2.1 a,b | 10.1 ± 4.6 a,b ** | 0.71 | 0.36 | 0.044 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nienaber, A.; Baumgartner, J.; Dolman, R.C.; Ozturk, M.; Zandberg, L.; Hayford, F.E.A.; Brombacher, F.; Blaauw, R.; Parihar, S.P.; Smuts, C.M.; et al. Omega-3 Fatty Acid and Iron Supplementation Alone, but Not in Combination, Lower Inflammation and Anemia of Infection in Mycobacterium tuberculosis-Infected Mice. Nutrients 2020, 12, 2897. https://doi.org/10.3390/nu12092897
Nienaber A, Baumgartner J, Dolman RC, Ozturk M, Zandberg L, Hayford FEA, Brombacher F, Blaauw R, Parihar SP, Smuts CM, et al. Omega-3 Fatty Acid and Iron Supplementation Alone, but Not in Combination, Lower Inflammation and Anemia of Infection in Mycobacterium tuberculosis-Infected Mice. Nutrients. 2020; 12(9):2897. https://doi.org/10.3390/nu12092897
Chicago/Turabian StyleNienaber, Arista, Jeannine Baumgartner, Robin C. Dolman, Mumin Ozturk, Lizelle Zandberg, Frank E. A. Hayford, Frank Brombacher, Renee Blaauw, Suraj P. Parihar, Cornelius M. Smuts, and et al. 2020. "Omega-3 Fatty Acid and Iron Supplementation Alone, but Not in Combination, Lower Inflammation and Anemia of Infection in Mycobacterium tuberculosis-Infected Mice" Nutrients 12, no. 9: 2897. https://doi.org/10.3390/nu12092897
APA StyleNienaber, A., Baumgartner, J., Dolman, R. C., Ozturk, M., Zandberg, L., Hayford, F. E. A., Brombacher, F., Blaauw, R., Parihar, S. P., Smuts, C. M., & Malan, L. (2020). Omega-3 Fatty Acid and Iron Supplementation Alone, but Not in Combination, Lower Inflammation and Anemia of Infection in Mycobacterium tuberculosis-Infected Mice. Nutrients, 12(9), 2897. https://doi.org/10.3390/nu12092897






