Nutritional Intervention to Prevent the Functional Decline in Community-Dwelling Older Adults: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Eligibility Criteria
2.2. Study Identification
2.3. Study Selection
2.4. Data Collection and Study Quality
3. Results
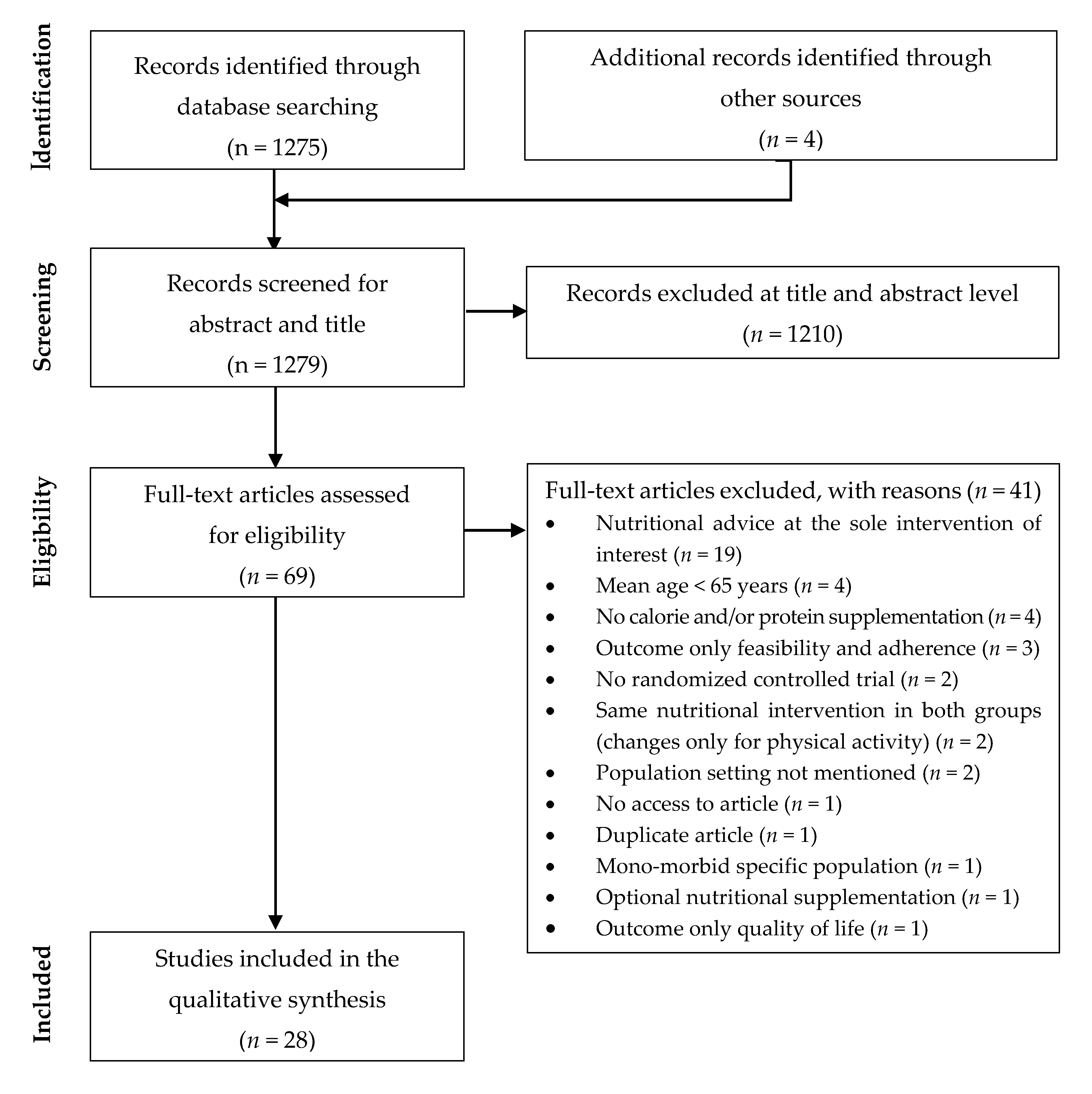
3.1. Study Selection
3.2. Study Characteristics
3.3. Risk of Bias within Studies
3.4. Main Findings
3.4.1. Unimodal Nutritional Interventions
3.4.2. Combined Nutritional and Physical Activity/Exercise Interventions
Healthy Older Adults
Older Adults with Low-Protein Intake or Limited Mobility
Frail Older Adults
Sarcopenic Older Adults
Summary
3.4.3. Combined Nutritional and Testosterone Therapy Interventions
3.4.4. Multimodal Interventions (>2 Interventions)
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Decade of Healthy Ageing. Available online: https://www.who.int/ageing/decade-of-healthy-ageing (accessed on 7 August 2020).
- Jaul, E.; Barron, J. Age-Related Diseases and Clinical and Public Health Implications for the 85 Years Old and Over Population. Front. Public Health 2017, 5, 335. [Google Scholar] [CrossRef] [PubMed]
- Keller, K.; Engelhardt, M. Strength and muscle mass loss with aging process. Age and strength loss. Muscles Ligaments Tendons J. 2013, 3, 346–350. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.S.; Cohen, H.J.; Pieper, C.F.; Fillenbaum, G.G.; Kraus, W.E.; Huffman, K.M.; Cornish, M.A.; Shiloh, A.; Flynn, C.; Sloane, R.; et al. Physical Performance Across the Adult Life Span: Correlates With Age and Physical Activity. J. Gerontol. Biol. Sci. Med. Sci. 2017, 72, 572–578. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Sobestiansky, S.; Michaelsson, K.; Cederholm, T. Sarcopenia prevalence and associations with mortality and hospitalisation by various sarcopenia definitions in 85–89 year old community-dwelling men: A report from the ULSAM study. BMC Geriatr. 2019, 19, 318. [Google Scholar] [CrossRef]
- Kim, M.; Won, C.W. Prevalence of sarcopenia in community-dwelling older adults using the definition of the European Working Group on Sarcopenia in Older People 2: Findings from the Korean Frailty and Aging Cohort Study. Age Ageing 2019, 48, 910–916. [Google Scholar] [CrossRef]
- Purcell, S.A.; MacKenzie, M.; Barbosa-Silva, T.G.; Dionne, I.J.; Ghosh, S.; Olobatuyi, O.V.; Siervo, M.; Ye, M.; Prado, C.M. Sarcopenia Prevalence Using Different Definitions in Older Community-Dwelling Canadians. J. Nutr. Health Aging 2020, 24, 783–790. [Google Scholar] [CrossRef]
- Clark, B.C.; Manini, T.M. Functional consequences of sarcopenia and dynapenia in the elderly. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 271–276. [Google Scholar] [CrossRef]
- Malmstrom, T.K.; Miller, D.K.; Simonsick, E.M.; Ferrucci, L.; Morley, J.E. SARC-F: A symptom score to predict persons with sarcopenia at risk for poor functional outcomes. J. Cachexia Sarcopenia Muscle 2016, 7, 28–36. [Google Scholar] [CrossRef]
- Schaap, L.A.; van Schoor, N.M.; Lips, P.; Visser, M. Associations of Sarcopenia Definitions, and Their Components, With the Incidence of Recurrent Falling and Fractures: The Longitudinal Aging Study Amsterdam. J. Gerontol. Biol. Sci. Med. Sci. 2018, 73, 1199–1204. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, W.; Wang, C.; Tao, W.; Dou, Q.; Yang, Y. Sarcopenia as a predictor of hospitalization among older people: A systematic review and meta-analysis. BMC Geriatr. 2018, 18, 188. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, L.; Cyrino, E.S.; Antunes, M.; Santos, D.A.; Sardinha, L.B. Sarcopenia and physical independence in older adults: The independent and synergic role of muscle mass and muscle function. J. Cachexia Sarcopenia Muscle 2017, 8, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Akune, T.; Muraki, S.; Oka, H.; Tanaka, S.; Kawaguchi, H.; Tokimura, F.; Yoshida, H.; Suzuki, T.; Nakamura, K.; Yoshimura, N. Incidence of certified need of care in the long-term care insurance system and its risk factors in the elderly of Japanese population-based cohorts: The ROAD study. Geriatr. Gerontol. Int. 2014, 14, 695–701. [Google Scholar] [CrossRef]
- Graf, C.E.; Karsegard, V.L.; Spoerri, A.; Makhlouf, A.M.; Ho, S.; Herrmann, F.R.; Genton, L. Body composition and all-cause mortality in subjects older than 65 y. Am. J. Clin. Nutr. 2015, 101, 760–767. [Google Scholar] [CrossRef] [PubMed]
- Graf, C.E.; Herrmann, F.R.; Spoerri, A.; Makhlouf, A.M.; Sorensen, T.I.A.; Ho, S.; Karsegard, V.L.; Genton, L. Impact of body composition changes on risk of all-cause mortality in older adults. Clin. Nutr. 2016, 35, 1499–1505. [Google Scholar] [CrossRef]
- Janssen, I.; Shepard, D.S.; Katzmarzyk, P.T.; Roubenoff, R. The healthcare costs of sarcopenia in the United States. J. Am. Geriatr. Soc. 2004, 52, 80–85. [Google Scholar] [CrossRef]
- Mijnarends, D.M.; Luiking, Y.C.; Halfens, R.J.G.; Evers, S.; Lenaerts, E.L.A.; Verlaan, S.; Wallace, M.; Schols, J.M.; Meijers, J.M. Muscle, Health and Costs: A Glance at their Relationship. J. Nutr. Health Aging 2018, 22, 766–773. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef]
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cruz-Jentoft, A.; Goisser, S.; Hooper, L.; Kiesswetter, E.; Maggio, M.; Raynaud-Simon, A.; Sieber, C.C.; et al. ESPEN guideline on clinical nutrition and hydration in geriatrics. Clin. Nutr. 2019, 38, 10–47. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Downs, S.H.; Black, N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J. Epidemiol. Community Health 1998, 52, 377–384. [Google Scholar] [CrossRef]
- O’Connor, S.R.; Tully, M.A.; Ryan, B.; Bradley, J.M.; Baxter, G.D.; McDonough, S.M. Failure of a numerical quality assessment scale to identify potential risk of bias in a systematic review: A comparison study. BMC Res Notes. 2015, 8, 224. [Google Scholar] [CrossRef] [PubMed]
- Ellis, A.C.; Hunter, G.R.; Goss, A.M.; Gower, B.A. Oral Supplementation with Beta-Hydroxy-Beta-Methylbutyrate, Arginine, and Glutamine Improves Lean Body Mass in Healthy Older Adults. J. Diet Suppl. 2019, 16, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Kerr, D.A.; Meng, X.; Devine, A.; Solah, V.; Binns, C.W.; Prince, R.L. Two-Year Whey Protein Supplementation Did Not Enhance Muscle Mass and Physical Function in Well-Nourished Healthy Older Postmenopausal Women. J. Nutr. 2015, 145, 2520–2526. [Google Scholar] [CrossRef]
- Ottestad, I.; Lovstad, A.T.; Gjevestad, G.O.; Hamarsland, H.; Saltyte Benth, J.; Andersen, L.F.; Bye, A.; Biong, A.S.; Retterstøl, K.; Iversen, P.O.; et al. Intake of a Protein-Enriched Milk and Effects on Muscle Mass and Strength. A 12-Week Randomized Placebo Controlled Trial among Community-Dwelling Older Adults. J. Nutr. Health Aging 2017, 21, 1160–1169. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.O.; Lee, K.R. Preventive effect of protein-energy supplementation on the functional decline of frail older adults with low socioeconomic status: A community-based randomized controlled study. J. Gerontol. Biol. Sci. Med. Sci. 2013, 68, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.M.; Verlaan, S.; Bautmans, I.; Brandt, K.; Donini, L.M.; Maggio, M.; McMurdo, M.E.; Mets, T.; Seal, C.; Wijers, S.L.; et al. Effects of a vitamin D and leucine-enriched whey protein nutritional supplement on measures of sarcopenia in older adults, the PROVIDE study: A randomized, double-blind, placebo-controlled trial. J. Am. Med. Dir. Assoc. 2015, 16, 740–747. [Google Scholar] [CrossRef]
- Mori, H.; Tokuda, Y. Effect of whey protein supplementation after resistance exercise on the muscle mass and physical function of healthy older women: A randomized controlled trial. Geriatr. Gerontol. Int. 2018, 18, 1398–1404. [Google Scholar] [CrossRef]
- Berton, L.; Bano, G.; Carraro, S.; Veronese, N.; Pizzato, S.; Bolzetta, F.; De Rui, M.; Valmorbida, E.; De Ronch, I.; Perissinotto, E.; et al. Effect of Oral Beta-Hydroxy-Beta-Methylbutyrate (HMB) Supplementation on Physical Performance in Healthy Old Women Over 65 Years: An Open Label Randomized Controlled Trial. PLoS ONE 2015, 10, e0141757. [Google Scholar] [CrossRef]
- Kirk, B.; Mooney, K.; Cousins, R.; Angell, P.; Jackson, M.; Pugh, J.N.; Coyles, G.; Amirabdollahian, F.; Khaiyat, O. Effects of exercise and whey protein on muscle mass, fat mass, myoelectrical muscle fatigue and health-related quality of life in older adults: A secondary analysis of the Liverpool Hope University-Sarcopenia Ageing Trial (LHU-SAT). Eur. J. Appl. Physiol. 2020, 120, 493–503. [Google Scholar] [CrossRef]
- Kirk, B.; Mooney, K.; Amirabdollahian, F.; Khaiyat, O. Exercise and Dietary-Protein as a Countermeasure to Skeletal Muscle Weakness: Liverpool Hope University-Sarcopenia Aging Trial (LHU-SAT). Front. Physiol. 2019, 10, 445. [Google Scholar] [CrossRef]
- Markofski, M.M.; Jennings, K.; Timmerman, K.L.; Dickinson, J.M.; Fry, C.S.; Borack, M.S.; Reidy, P.T.; Deer, R.R.; Randolph, A.; Rasmussen, B.B.; et al. Effect of Aerobic Exercise Training and Essential Amino Acid Supplementation for 24 Weeks on Physical Function, Body Composition, and Muscle Metabolism in Healthy, Independent Older Adults: A Randomized Clinical Trial. J. Gerontol. Biol. Sci. Med. Sci. 2019, 74, 1598–1604. [Google Scholar] [CrossRef] [PubMed]
- Seino, S.; Sumi, K.; Narita, M.; Yokoyama, Y.; Ashida, K.; Kitamura, A.; Shinkai, S. Effects of Low-Dose Dairy Protein Plus Micronutrient Supplementation during Resistance Exercise on Muscle Mass and Physical Performance in Older Adults: A Randomized, Controlled Trial. J. Nutr. Health Aging 2018, 22, 59–67. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho Bastone, A.; Nobre, L.N.; de Souza Moreira, B.; Rosa, I.F.; Ferreira, G.B.; Santos, D.D.; Monteiro, N.K.; Alves, M.D.; Gandra, R.A.; de Lira, E.M. Independent and combined effect of home-based progressive resistance training and nutritional supplementation on muscle strength, muscle mass and physical function in dynapenic older adults with low protein intake: A randomized controlled trial. Arch. Gerontol. Geriatr. 2020, 89, 104098. [Google Scholar] [CrossRef] [PubMed]
- Bonnefoy, M.; Boutitie, F.; Mercier, C.; Gueyffier, F.; Carre, C.; Guetemme, G.; Ravis, B.; Laville, M.; Cornu, C. Efficacy of a home-based intervention programme on the physical activity level and functional ability of older people using domestic services: A randomised study. J. Nutr. Health Aging 2012, 16, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Englund, D.A.; Kirn, D.R.; Koochek, A.; Zhu, H.; Travison, T.G.; Reid, K.F.; von Berens, Å.; Melin, M.; Cederholm, T.; Gustafsson, T.; et al. Nutritional Supplementation With Physical Activity Improves Muscle Composition in Mobility-Limited Older Adults, The VIVE2 Study: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Gerontol. Biol. Sci. Med. Sci. 2017, 73, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Fielding, R.A.; Travison, T.G.; Kirn, D.R.; Koochek, A.; Reid, K.F.; von Berens, A.; Zhu, H.; Folta, S.C.; Sacheck, J.M.; Nelson, M.E.; et al. Effect of Structured Physical Activity and Nutritional Supplementation on Physical Function in Mobility-Limited Older Adults: Results from the VIVE2 Randomized Trial. J. Nutr. Health Aging 2017, 21, 936–942. [Google Scholar] [CrossRef]
- Ikeda, T.; Aizawa, J.; Nagasawa, H.; Gomi, I.; Kugota, H.; Nanjo, K.; Jinno, T.; Masuda, T.; Morita, S. Effects and feasibility of exercise therapy combined with branched-chain amino acid supplementation on muscle strengthening in frail and pre-frail elderly people requiring long-term care: A crossover trial. Appl. Physiol. Nutr. Metab. 2016, 41, 438–445. [Google Scholar] [CrossRef]
- Yamada, M.; Nishiguchi, S.; Fukutani, N.; Aoyama, T.; Arai, H. Mail-Based Intervention for Sarcopenia Prevention Increased Anabolic Hormone and Skeletal Muscle Mass in Community-Dwelling Japanese Older Adults: The INE (Intervention by Nutrition and Exercise) Study. J. Am. Med. Dir. Assoc. 2015, 16, 654–660. [Google Scholar] [CrossRef]
- Bjorkman, M.P.; Suominen, M.H.; Kautiainen, H.; Jyvakorpi, S.K.; Finne-Soveri, H.U.; Strandberg, T.E.; Pitkälä, K.H.; Tilvis, R. Effect of Protein Supplementation on Physical Performance in Older People with Sarcopenia-A Randomized Controlled Trial. J. Am. Med. Dir. Assoc. 2020, 21, 226–232. [Google Scholar] [CrossRef]
- Zhu, L.Y.; Chan, R.; Kwok, T.; Cheng, K.C.; Ha, A.; Woo, J. Effects of exercise and nutrition supplementation in community-dwelling older Chinese people with sarcopenia: A randomized controlled trial. Age Ageing 2019, 48, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Suzuki, T.; Saito, K.; Yoshida, H.; Kobayashi, H.; Kato, H.; Katayama, M. Effects of exercise and amino acid supplementation on body composition and physical function in community-dwelling elderly Japanese sarcopenic women: A randomized controlled trial. J. Am. Geriatr. Soc. 2012, 60, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Kemmler, W.; Grimm, A.; Bebenek, M.; Kohl, M.; von Stengel, S. Effects of Combined Whole-Body Electromyostimulation and Protein Supplementation on Local and Overall Muscle/Fat Distribution in Older Men with Sarcopenic Obesity: The Randomized Controlled Franconia Sarcopenic Obesity (FranSO) Study. Calcif. Tissue Int. 2018, 103, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Kemmler, W.; Weissenfels, A.; Teschler, M.; Willert, S.; Bebenek, M.; Shojaa, M.; Kohl, M.; Freiberger, E.; Sieber, C.; von Stengel, S. Whole-body electromyostimulation and protein supplementation favorably affect sarcopenic obesity in community-dwelling older men at risk: The randomized controlled FranSO study. Clin. Interv. Aging 2017, 12, 1503–1513. [Google Scholar] [CrossRef]
- Kim, H.; Kim, M.; Kojima, N.; Fujino, K.; Hosoi, E.; Kobayashi, H.; Somekawa, S.; Niki, Y.; Yamashiro, Y.; Yoshida, H. Exercise and Nutritional Supplementation on Community-Dwelling Elderly Japanese Women with Sarcopenic Obesity: A Randomized Controlled Trial. J. Am. Med. Dir. Assoc. 2016, 17, 1011–1019. [Google Scholar] [CrossRef]
- Bhasin, S.; Apovian, C.M.; Travison, T.G.; Pencina, K.; Moore, L.L.; Huang, G.; Campbell, W.W.; Li, Z.; Howland, A.S.; Chen, R.; et al. Effect of Protein Intake on Lean Body Mass in Functionally Limited Older Men: A Randomized Clinical Trial. JAMA Intern. Med. 2018, 178, 530–541. [Google Scholar] [CrossRef]
- Visvanathan, R.; Piantadosi, C.; Lange, K.; Naganathan, V.; Hunter, P.; Cameron, I.D.; Chapman, I. The Randomized Control Trial of the Effects of Testosterone and a Nutritional Supplement on Hospital Admissions in Undernourished, Community Dwelling, Older People. J. Nutr. Health Aging 2016, 20, 769–779. [Google Scholar] [CrossRef]
- Deer, R.R.; Dickinson, J.M.; Baillargeon, J.; Fisher, S.R.; Raji, M.; Volpi, E. A Phase I Randomized Clinical Trial of Evidence-Based, Pragmatic Interventions to Improve Functional Recovery after Hospitalization in Geriatric Patients. J. Gerontol. Biol. Sci. Med. Sci. 2019, 74, 1628–1636. [Google Scholar] [CrossRef]
- Romera-Liebana, L.; Orfila, F.; Segura, J.M.; Real, J.; Fabra, M.L.; Moller, M.; Lancho, S.; Ramirez, A.; Marti, N.; Cullell, M.; et al. Effects of a Primary Care-Based Multifactorial Intervention on Physical and Cognitive Function in Frail, Elderly Individuals: A Randomized Controlled Trial. J. Gerontol. Biol. Sci. Med. Sci. 2018, 73, 1668–1674. [Google Scholar] [CrossRef]
- Ng, T.P.; Feng, L.; Nyunt, M.S.; Feng, L.; Niti, M.; Tan, B.Y.; Chan, G.; Khoo, S.A.; Chan, S.M.; Yap, P.; et al. Nutritional, Physical, Cognitive, and Combination Interventions and Frailty Reversal Among Older Adults: A Randomized Controlled Trial. Am. J. Med. 2015, 128, 1225–1236. [Google Scholar] [CrossRef]
- Cawood, A.L.; Elia, M.; Stratton, R.J. Systematic review and meta-analysis of the effects of high protein oral nutritional supplements. Ageing Res. Rev. 2012, 11, 278–296. [Google Scholar] [CrossRef]
- World Health Organization. Physical Activity and Older Adults. Available online: https://www.who.int/dietphysicalactivity/factsheet_olderadults/en/ (accessed on 18 August 2020).
- Taylor, D. Physical activity is medicine for older adults. Postgrad. Med. J. 2014, 90, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Negm, A.M.; Kennedy, C.C.; Thabane, L.; Veroniki, A.A.; Adachi, J.D.; Richardson, J.; Cameron, I.D.; Giangregorio, A.; Petropoulou, M.; Alsaad, S.M.; et al. Management of Frailty: A Systematic Review and Network Meta-analysis of Randomized Controlled Trials. J. Am. Med. Dir. Assoc. 2019, 20, 1190–1198. [Google Scholar] [CrossRef] [PubMed]
- Ten Haaf, D.S.M.; Nuijten, M.A.H.; Maessen, M.F.H.; Horstman, A.M.H.; Eijsvogels, T.M.H.; Hopman, M.T.E. Effects of protein supplementation on lean body mass, muscle strength, and physical performance in nonfrail community-dwelling older adults: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2018, 108, 1043–1059. [Google Scholar] [CrossRef]
- De Spiegeleer, A.; Beckwee, D.; Bautmans, I.; Petrovic, M. Sarcopenia Guidelines Development group of the Belgian Society of G., Geriatrics. Pharmacological Interventions to Improve Muscle Mass, Muscle Strength and Physical Performance in Older People: An Umbrella Review of Systematic Reviews and Meta-analyses. Drugs Aging 2018, 35, 719–734. [Google Scholar] [CrossRef] [PubMed]
- Borst, S.E.; Mulligan, T. Testosterone replacement therapy for older men. Clin. Interv. Aging 2007, 2, 561–566. [Google Scholar] [CrossRef]
- Beck, A.M.; Damkjaer, K.; Beyer, N. Multifaceted nutritional intervention among nursing-home residents has a positive influence on nutrition and function. Nutrition 2008, 24, 1073–1080. [Google Scholar] [CrossRef]
- Beck, A.M.; Christensen, A.G.; Hansen, B.S.; Damsbo-Svendsen, S.; Moller, T.K. Multidisciplinary nutritional support for undernutrition in nursing home and home-care: A cluster randomized controlled trial. Nutrition 2016, 32, 199–205. [Google Scholar] [CrossRef]
- Vellas, B.; Guigoz, Y.; Garry, P.J.; Nourhashemi, F.; Bennahum, D.; Lauque, S.; Albarede, J.L. The Mini Nutritional Assessment (MNA) and its use in grading the nutritional state of elderly patients. Nutrition 1999, 15, 116–122. [Google Scholar] [CrossRef]
- Cederholm, T.; Jensen, G.L.; Correia, M.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.J.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. Clin. Nutr. 2019, 38, 207–217. [Google Scholar] [CrossRef]

| Studies | Population | Intervention | Outcomes | Results | Limitations | QS |
|---|---|---|---|---|---|---|
| Ellis et al. 2019 [24] | Healthy men and women 65–89 yrs n = 34 | 6 months INT: amino acid supplement; 3 g HMB, 14g L-arginine, 14 g L-glutamine/day (n = 17) CO: isocaloric placebo (n = 17) | Lean body mass (DXA) and quadriceps muscle volume (MRI) Physical performance: eight-foot up-and-go test, 25-foot walk test, 12-step timed stair climb | Significant improvement in lean body mass and timed stair climb in INT No change in quadriceps volume and other physical performance parameters | Small sample size, no monitoring of nutrient intake, no assessment of muscle strength | 19/28 |
| Zhu et al. 2015 [25] | Healthy women Mean age 74.3 ± 2.7 yrs n = 219 | 2 years INT: protein supplement; 30 g whey, 1×/day (n = 109) CO: placebo (n = 110) | ASMM (DXA) Muscle strength: handgrip, lower limb muscle strength Physical performance: TUG | No change in ASMM, lower limb muscle strength and TUG between groups Significant decrease in handgrip in INT | High protein intake at inclusion | 23/28 |
| Ottestad et al. 2017 [26] | Healthy men and womenHandgrip strength < 20 kg in women, < 30 kg in men Gait speed < 1m/s 5-time sit to stand test ≥ 8.4s ≥ 70 yrs n = 50 | 12 weeks INT: protein-enriched milk; 20 g protein, 2×/day (n = 24) CO: isocaloric placebo (n = 26) | ASMM (DXA) Muscle strength: handgrip, 1-repetition maximum leg and chest press, 5-time sit-to-stand test Physical performance: stair climbing test (20 steps) | Significant improvement of chest press in INT and CO, but no difference between groups No change in other parameters between groups | High dropout rate in both groups, tests not performed on all subjects | 22/28 |
| Kim et al. 2013 [27] | Frail men and women Gait speed < 0.6 m/s MNA < 24 ≥ 65 yrs n = 87 | 12 weeks INT: nutritional supplement; 200 kcal, 12.5 g protein, 2×/day (n = 43) CO: no treatment (n = 42) | Muscle strength: handgrip Physical performance: one-leg stance SPPB, TUG | No difference between groups in handgrip and one-leg stance SPPB score stable in INT, but decreased in CO Significant improvement of TUG in INT | High dropout rate in INT, no placebo in CO, multiple testing | 20/28 |
| Bauer et al. 2015 [28] | Sarcopenic men and women BMI 20–30 kg/m2 SPPB score: 4-9 Skeletal muscle mass / body weight × 100: < 37% in men and < 28% in women Mean age 77.7 yrs n = 380 | 13 weeks INT: nutritional supplement; 20 g whey protein, 3 g total leucine, 800 IU vitamin D, 2×/day (n = 184) CO: isocaloric placebo (n = 196) | ASMM (DXA) Muscle strength: handgrip, 5-time sit-to-stand test Physical performance: SPPB, gait speed (4-m walk), balance test | Significant improvement in ASMM and 5-time sit-to-stand test in INT compared with CO No difference in handgrip, SPPB, gait speed and balance scores between groups | High dropout rate, no monitoring of nutrient intake, definition of sarcopenia not clear | 24/28 |
| Studies | Population | Intervention | Outcomes | Results | Limitations | QS |
|---|---|---|---|---|---|---|
| Mori et al. 2018 [29] | Healthy women Aged 65–80 yrs n = 81 | 24 weeks Group A: supervised and home-based resistance exercise; 2×/week + protein supplement; 22.3 g whey, 2×/week 5 min after exercise (n = 27) Group B: supervised and home-based resistance exercise; 2×/week (n = 27) Group C: protein supplement; 22.3 g whey, 2×/week (n = 27) | Upper and lower limb muscle mass (BIA) Muscle strength: handgrip, knee extension strength Physical performance: gait speed | Significant improvement in upper and lower limb muscle mass, handgrip strength and gait speed in exercise + protein supplement compared to protein supplement only Significant improvement in lower limb muscle mass and knee extension strength in exercise + protein supplement compared to exercise only and protein supplement only | Compliance to the intervention not reported, characteristics of lost to follow-up not described | 19/28 |
| Berton et al. 2015 [30] | Healthy women Mean age 69.5 ± 5.3 yrs n = 80 | 8 weeks Supervised endurance and resistance exercise; 2×/week + INT: nutritional supplement; 330 kcal, 20 g proteins, 1.5 g HMB, 1×/day (n = 40) CO: no treatment (n = 40) | ASMM (DXA) Muscle strength: isometric knee extension torque, isokinetic strength, handgrip Physical performance: SPPB, 6MWT | No difference in ASMMI, handgrip and SPPB between groups Significant improvement in isometric knee extension torque, isokinetic strength and in 6MWT in INT | No monitoring of nutrient intake, description of exercise training not clear | 22/28 |
| Kirk et al. 2020 [31] | Non-frail and untrained men and women Mean age 69 ± 6 yrs n = 123 | 16 weeks Group A: no treatment (n = 34) Group B: supervised resistance and functional exercise; 50 min, 3×/week (n = 29) Group C: supervised resistance and functional exercise; 50 min, 3×/week + leucine-enriched whey protein supplement; based on individual body-weight 1.5 g/kg/day (n = 22) Group D: leucine-enriched whey protein supplement; based on individual body weight 1.5 g/kg/day (n = 38) | Muscle mass and skeletal muscle index (muscle mass/height2) (BIA) | No significant change in muscle mass and skeletal muscle index | Lack of compliance to protein supplement, high dropout rate in protein supplementation group, lack of external validity | 17/28 |
| Kirk et al. 2019 [32] | Non-frail and untrained men and women Mean age 68 ± 5 yrs n = 51 | 16 weeks Group A: supervised resistance and functional exercise; 50 min, 3×/week (n = 29) Group B: supervised resistance and functional exercise; 50 min, 3×/week + leucine-enriched whey protein supplement; based on individual body-weight 1.5 g/kg/day (n = 22) | Muscle strength: 5-repetition maximum in leg press, chest press, and bicep curl Physical performance: SPPB, 25-m obstacle course, 6MWT | Significant improvement in all parameters in the both groups, with no difference between groups | Lack of compliance to protein supplement, no muscle mass assessment, lack of external validity | 16/28 |
| Markofski et al. 2018 [33] | Non-frail independent men and women Low active <7500 steps/day Not engaged in an exercise program Mean age 72 ± 1 yrs n = 50 | 24 weeks Group A: nutritional supplement; 15 g essential amino acids, 1×/day + supervised endurance exercise; 50 min, 3×/week (n = 14) Group B: placebo supplement + supervised endurance exercise; 50 min, 3×/week (n = 11) Group C: nutritional supplement; 15 g essential amino acids, 1×/day (n = 13) Group D: placebo (n = 12) | Lean body mass (DXA) Muscle strength: isokinetic leg strength Physical performance: 20-m walk, 20-m walk with carry, 400-m walk test, VO2 | Compared to baseline: No significant change in lean mass Significant improvement of isokinetic leg strength only in the nutritional supplement + exercise group Significant improvement of walking speed and VO2 peak in both exercise groups, irrespective of supplementation type | No monitoring of nutrient intake, statistical power limited, non-frail not defined, no sample size calculation | 19/28 |
| Seino et al. 2018 [34] | Non-disabled men and women Not engaged in an exercise program Mean age 73.5 yrs n = 82 | 12 weeks Group A: supervised resistance exercise; 60 min, 2×/week + nutritional supplement; 114 kcal, 10.5 g protein, 1×/day + micronutrient beverage, 1×/day (n = 41) Group B: supervised resistance exercise; 60 min, 2×/week (n = 41) | Lean body mass, ASMM (DXA) Muscle strength: handgrip, knee-extension strength, 5-time sit-to-stand test Physical performance: assessed by single leg stand, gait speed, TUG | Significant improvement in lean body mass and ASMM in exercise and nutritional supplement group compared to exercise only No significant differences between groups in handgrip strength, knee-extension strength, single leg stand, gait speed, TUG, 5-time sit-to-stand test | Few male subjects, compliance to the intervention not mentioned | 22/28 |
| De Carvalho Bastone et al. 2020 [35] | Men and women Low protein intake < 1 g/kg/day Able to walk independently Handgrip strength <20 kg in women, <30 kg in men Mean age 75.9 ± 6.7 yrs n = 80 | 3 months Group A: home-based supervised progressive resistance exercise program; 60 min, 3×/week (n = 20) Group B: protein supplement; 40 g protein/day (n = 20) Group C: combined resistance exercise program and protein supplementation (n = 20) Group D: daily routine (n = 20) | Skeletal muscle index: absolute skeletal muscle mass / height squared (BIA) Muscle strength: handgrip, 30 s and 5-time sit-to-stand test Physical performance: gait speed (8-m course), TUG | Significant improvement in handgrip, gait speed, and 5-time sit-to-stand test in resistance training only and combined resistance plus protein supplement groups compared to control group No significant difference in all parameters between resistance training only and combined resistance plus protein supplement groups | No monitoring of nutrient intake, lack of statistical power | 21/28 |
| Bonnefoy et al. 2012 [36] | Men and women Able to walk independently; risk of becoming frail: gait speed < 0.8 m/s ± PASE < 64 for men, < 52 for women Median age 84 yrs n = 102 | 4 months INT: self-administered mobility, strength, balance and endurance exercise program; 20 min, 1×/day + protein supplement; 10 g/day during 1.5 months (n = 53) CO: no treatment (n = 49) | Fat-free mass (device?) Physical performance: TUG, walking speed, maximum walking time, 1-min sit-to-stand test, six-stair climbing time | Only significant reduction in maximum walking time in control group | Low compliance, protein supplementation only during 1.5 month, evaluators and participants not blinded | 17/28 |
| Englund et al. 2017 [37] | Men and Women Mobility limitations: SPPB ≤ 9 Low serum vitamin D level Mean age 77.5 ± 5.4 yrs n = 149 | 6 months Group A: supervised endurance, resistance, balance and flexibility exercise; 60 min, 3×/week + nutritional supplement; 150 kcal, 20 g whey protein, 800 UI vitamin D, 1×/day (n = 74) Group B: supervised endurance, resistance, balance and flexibility exercise; 60 min, 3×/week + placebo, 30 kcal, 1×/day (n = 75) | ASMM (DXA) Muscle strength: isokinetic strength | No improvement in ASMM in both groups Improvement in muscle strength in both groups No significant differences for all parameters between groups | No monitoring of nutrient intake | 21/28 |
| Fielding et al. 2017 [38] | Men and Women Mobility limitations: SPPB ≤ 9 Low serum vitamin D level Mean age 77.5 ± 5.4 yrs n = 149 | 6 months Group A: supervised endurance, resistance, balance and flexibility exercise; 60 min, 3×/week + nutritional supplement; 150 kcal, 20 g whey protein, 800 UI vitamin D, 1×/day (n = 74) Group B: supervised endurance, resistance, balance and flexibility exercise; 60 min, 3×/week + placebo, 30 kcal, 1×/day (n = 75) | Physical performance: gait speed (400-m walk capacity), SPPB | Significant improvement in gait speed and SPPB in both groups but no significant difference between groups | No monitoring of nutrient intake | 21/28 |
| Ikeda et al. 2016 [39] | Pre-frail and frail men and women according to Fried et al. Mean age 78.4 ± 7.8 yrs and 80.4 ± 8.9 yrs in the 2 groups n = 52 | Cross-over design: two time 3 months of supplementation combined with exercise, washout of 1 month with exercise only Supplementation: 6g of BCAA or 6 g of maltodextrin, 2×/week 10 min before exercise Exercise: supervised resistance, endurance, balance exercise; 2×/week | Muscle strength: handgrip, upper and lower limb isometric strength Physical performance: TUG, dynamic balance ability | Significant improvement in lower limb isometric strength and dynamic balance ability in BCAA group compared to the control group after crossover | High dropout rate, no monitoring of nutrient intake, population including both pre-frail and frail subjects | 19/28 |
| Yamada et al. 2015 [40] | Non-frail and frail (Cardiovascular Health Study criteria) men and women Able to walk independently Age ≥ 65 yrs n = 227 | 6 months Group A: pedometer-based walking program; increasing of daily steps by 10% each month with ankle weight 0.5 kg + nutritional supplement; 200 kcal, 10 g protein, 12.5 ug vitamin D, 1×/day (n = 79) Group B: pedometer-based walking program; increasing of daily steps by 10% each month with ankle weight 0.5 kg (n = 71) Group C: daily routine (n = 77) | Skeletal muscle mass index: muscle mass/height2 (BIA) | Significant improvement of skeletal muscle index in exercise + nutrition and exercise alone compared to control group Effects more pronounced in the subgroup of frail subjects | No monitoring of nutrient intake, compliance and number of steps in both groups before and after intervention not reported | 20/28 |
| Bjorkman et al. 2020 [41] | Sarcopenic men and women Able to walk independently Handgrip <20 kg in women, < 30 kg in men or gait speed <0.80 m/s 75–96 yrs n = 218 | 12 months Instructions on low-intensity home-based exercise, importance of dietary protein and vitamin D supplementation 20 µg/day + Group A: no treatment (n = 72) Group B: isocaloric placebo (n = 73) Group C: protein supplement; 20g whey protein, 2×/day (n = 73) | Skeletal muscle index: skeletal muscle mass / height2 (BIA) Muscle strength: handgrip physical performance: SPPB | No significant differences in skeletal muscle index and physical performance between groups Significant reduction in muscle strength in all groups | Dropout higher in control group compared to other groups, low compliance and adherence in intervention groups | 21/28 |
| Zhu et al. 2019 [42] | Sarcopenic men and women Sarcopenia: Asian Working Group criteria ≥ 65 yrs n = 113 | 12 weeks Group A: supervised endurance and resistance exercise; 45 min, 1×/week + home session, resistance exercise, min 1×/week (n = 40) Group B: supervised endurance and resistance exercise; 45 min, 1×/week + home session, resistance exercise, min 1×/week nutritional supplement + oral nutritional supplement; 231 kcal, 8.61 g protein, 1×/day (n = 36) Group C: daily routine (n = 37) | ASSM (DXA) Muscle strength: handgrip strength, leg extensors strength, 5-time sit-to-stand test Physical performance: gait speed (6-m walk test) | Compared to control group: Significant improvement of ASMMI in exercise + nutritional supplement group only Significant improvement of leg extensors strength and 5-time sit-to-stand test in both intervention groups No significant improvement of handgrip and gait speed | No monitoring of nutrient intake, high dropout rate | 21/28 |
| Kim et al. 2012 [43] | Sarcopenic women Sarcopenia: ASMMI < 6.42 kg/m2, knee extension strength < 1.01 Nm/kg, gait speed < 1.22 m/s, BMI < 22.0 kg/m2 ≥ 75 yrs n = 155 | 3 months Group A: supervised resistance and balance exercise; 60 min, 2×/week + protein supplement; 3.0 g of amino acid, 2×/day (n = 38) Group B: supervised resistance and balance exercise; 60 min, 2×/week (n = 39) Group C: protein supplement; 3.0 g of amino acid, 2×/day (n = 39) Group D: health education (n = 39) | Leg muscle mass (BIA) Muscle strength: knee extension strength Physical performance: walking speed | Compared to health education group: Significant increase in leg muscle mass and muscle strength only in the exercise + nutrition group Signification increase of walking speed in exercise + nutrition group and exercise only groups | Multiple testing, no monitoring of nutrient intake | 21/28 |
| Kemmler et al. 2018 [44] | Sarcopenic and obese men Sarcopenia: ASSM/BMI <0.789 Obesity: fat mass > 27% ≥ 70 yrs n = 67 | 16 weeks INT: whole-body electro-myostimulation; 14 to 20 min, 1.5×/week + whey protein (aim: protein intake 1.7–1.8 g/kg/day) and 800 UI vitamin D/day (n = 33) CO: no treatment (n = 34) | Muscle distribution of intra-fascial fat-free muscle volume of the mid-thigh (MRI) and ASMM (BIA) Muscle strength: leg-extensor strength Physical performance: 10-m gait velocity | Significant improvement of all parameters in intervention group compared to baseline and compared to control group | Sarcopenia and obesity not defined according usual definitions, lower protein intake than prescribed in intervention group, high MRI assessment refusal rate | 19/28 |
| Kemmler et al. 2017 [45] | Sarcopenic and obese men Sarcopenia: ASSM/BMI < 0.789 Obesity: fat mass > 27% ≥ 70 yrs n = 100 | 16 weeks Group A: whole-body electro-myostimulation; 14 to 20 min, 1.5×/week + whey protein (aim: protein intake 1.7–1.8 g/kg/day) and 800 UI vitamin D/day (n = 33) Group B: whey protein (aim: protein intake 1.7–1.8 g/kg/day) and 800 UI vitamin D/day (n = 33) Group C: no treatment (n = 34) | Skeletal muscle index: ASSM/BMI (BIA) Muscle strength: handgrip | Significant improvement of skeletal muscle index in the 2 intervention groups Significant increase in handgrip strength with electromyostimulation only | Sarcopenia and obesity not defined according usual definitions, pro tein intake lower than prescribed in both protein-supplemented group | 19/28 |
| Kim et al. 2016 [46] | Sarcopenic and obese women Sarcopenia: skeletal muscle index < 5.67 kg/m2, handgrip < 17 kg, gait speed < 1.0 m/s Obesity: fat mass > 32% > 70 yrs n = 139 | 3 months Group A: supervised resistance and endurance exercise; 60 min, 2×/week + nutritional supplement 3.0 g leucine, 20 mg vitamin D and 350 mL of tea catechin fortified, 1×/day (n = 36) Group B: supervised resistance and endurance exercise; 60 min, 2×/week (n = 36) Group C: nutritional supplement 3.0 g leucine, 20 mg vitamin D and 350 mL of tea catechin fortified, 1×/day (n = 34) Group D: health education (n = 34) | ASMM (BIA) Muscle strength: handgrip Physical performance: 11-m gait velocity | No significant changes in ASMM and handgrip strength between the groups Significant improvement of gait velocity in exercise + nutrition group | Multiple testing, no monitoring of nutrient intake | 22/28 |
| Studies | Population | Intervention | Outcomes | Results | Limitations | QS |
|---|---|---|---|---|---|---|
| Bhasin et al. 2018 [47] | Men Mobility limitations: SPPB 3–10 Daily protein intake < 0.83 g/kg/day Mean age 73 ± 5.8 yrs n = 92 | 6 months Group A: placebo injections weekly + 0.8 g/kg/day protein (n = 24) Group B: placebo injections weekly + 1.3 g/kg/day protein (n = 24) Group C: testosterone enanthate 100 mg intramuscularly weekly + 0.8 g/kg/day protein (n = 22) Group D: testosterone enanthate 100 mg intramuscularly weekly + 1.3 g/kg/day protein (n = 22) | Lean body mass, ASSM (DXA) Muscle strength: maximal leg press and chest press strength Physical performance: 6MWT, stair climbing, 50-m walk carrying a load equaling 20% body mass | Regardless of whether patients received testosterone or placebo: no change in muscle mass, muscle strength, and physical function between men assigned to 0.8 vs 1.3 g/kg/d of protein Regardless of whether patients received 0.8 vs 1.3 g/kg/d of protein: change in lean mass, ASSM and muscle strength but not in physical function in men randomized to testosterone compared to placebo | No combined physical activity, only men, pre-packaged controlled meals (not representative of reality), statistical power limited | 24/28 |
| Visvanathan et al. [48] 2016 | Undernourished men and women MNA: 17 and 23.5 BMI < 22 kg/m2 or self-reported weight loss ≥7.5% in the last 3 months Aged ≥ 65 yrs n = 53 | 12 months INT: nutritional supplement; 500–800 kcal, 1×/day + oral testosterone; undecanoate 40 mg/day women, 160 mg/day men (n = 25) CO: nutritional supplement; 35–45 kcal, 1×/day + placebo medication (n = 26) | Muscle mass (BIA) Muscle strength: handgrip Hospital admissions | No significant difference in all parameters in each arm and between the treatment arms | Expected sample size not reached, high dropout rate, no monitoring of nutrient intake | 22/28 |
| Studies | Population | Intervention | Outcomes | Results | Limitations | QS |
|---|---|---|---|---|---|---|
| Deer et al. 2019 [49] | Men and women admitted to hospital for an acute medical illness Residing at home before and after hospitalization Able to walk across a small room 2 weeks before hospitalization and to stand independently Mean age 78.1 ± 8.0 yrs n = 100 | During 4 weeks after discharge Group A: protein supplement; 20 g whey protein, 2×/day (n = 20) Group B: in-home resistance training program, 3×/week + placebo supplement (n = 21) Group C: combined protein supplementation + resistance training (n = 20) Group D: single testosterone injection; enanthate 100 mg for women, 200 mg for men (n = 19) Group E: placebo (n = 20) | Lean body mass and ASMM (DXA) Physical performance: SPPB 30-day readmission | Significant improvement in SPPB score in all active intervention groups compared to placebo No significant differences in body composition between groups Readmission rates highest in placebo (28%), followed by exercise + placebo (15%), protein supplementation (12%), exercise + protein supplementation (11%) and testosterone (5%) | Pilot study with many interventions studied in the same trial, statistical power limited, no monitoring of nutrient intake | 20/28 |
| Romera-Liebana et al. 2017 [50] | Prefrail and frail men and women TUG: 10–20s No severe cognitive impairment Mean age 77.3 yrs n = 352 | Follow-up at 3 and 18 months INT: multimodal therapy; supervised exercise, endurance, resistance, flexibility, balance training; 60min, 2×/week, for 6 weeks + nutritional supplement; 156 kcal, 11.8g protein, 1×/day, for 6 weeks + memory workshop; 90 min, 2×/week for 6 weeks + medication review (n = 176) CO: standard care (n = 176) | Muscle strength: handgrip strength Physical performance: SPPB, standing balance, stretching and unipodal station test | Significant improvement in all parameters in the intervention group at 3 and 18 months compared to control group | No monitoring of nutrient intake, no assessment of muscle mass, high dropout rate at 18 months, compliance to the intervention not mentioned | 22/28 |
| Ng et al. 2015 [51] | Pre-frail and frail men and women according to Fried et al. Able to walk independently Mean age 70.0 ± 4.7 yrs n = 246 | 24 weeks Group A: resistance and balance exercise; supervised 12 weeks, 90 min, 2×/week and home-based, 12 weeks, 90 min, 2×/week (n = 48) Group B: nutritional supplement; 300 kcal, 12 g proteins, 1×/day (n = 49) Group C: cognitive training; 2h, 1×/week (n = 50) Group D: combined intervention (exercise, nutritional supplement and cognitive training) (n = 49) Group E: standard care (n = 50) | Frailty score status: unintentional weight, slowness (6m fast gait speed), weakness (knee extension strength), exhaustion (SF-12 scale) and low-activity (longitudinal ageing physical activity questionnaire) Hospitalization rate: self-reported | Significant reduction of frailty score in exercise and combined intervention groups only compared to baseline No difference in hospitalization rate | No monitoring of nutrient intake, population including both pre-frail and frail subjects | 23/28 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mareschal, J.; Genton, L.; Collet, T.-H.; Graf, C. Nutritional Intervention to Prevent the Functional Decline in Community-Dwelling Older Adults: A Systematic Review. Nutrients 2020, 12, 2820. https://doi.org/10.3390/nu12092820
Mareschal J, Genton L, Collet T-H, Graf C. Nutritional Intervention to Prevent the Functional Decline in Community-Dwelling Older Adults: A Systematic Review. Nutrients. 2020; 12(9):2820. https://doi.org/10.3390/nu12092820
Chicago/Turabian StyleMareschal, Julie, Laurence Genton, Tinh-Hai Collet, and Christophe Graf. 2020. "Nutritional Intervention to Prevent the Functional Decline in Community-Dwelling Older Adults: A Systematic Review" Nutrients 12, no. 9: 2820. https://doi.org/10.3390/nu12092820
APA StyleMareschal, J., Genton, L., Collet, T.-H., & Graf, C. (2020). Nutritional Intervention to Prevent the Functional Decline in Community-Dwelling Older Adults: A Systematic Review. Nutrients, 12(9), 2820. https://doi.org/10.3390/nu12092820






