Effect of Physical Exercise on Taste Perceptions: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Study Selection
2.3. Data Gathering and Analyses
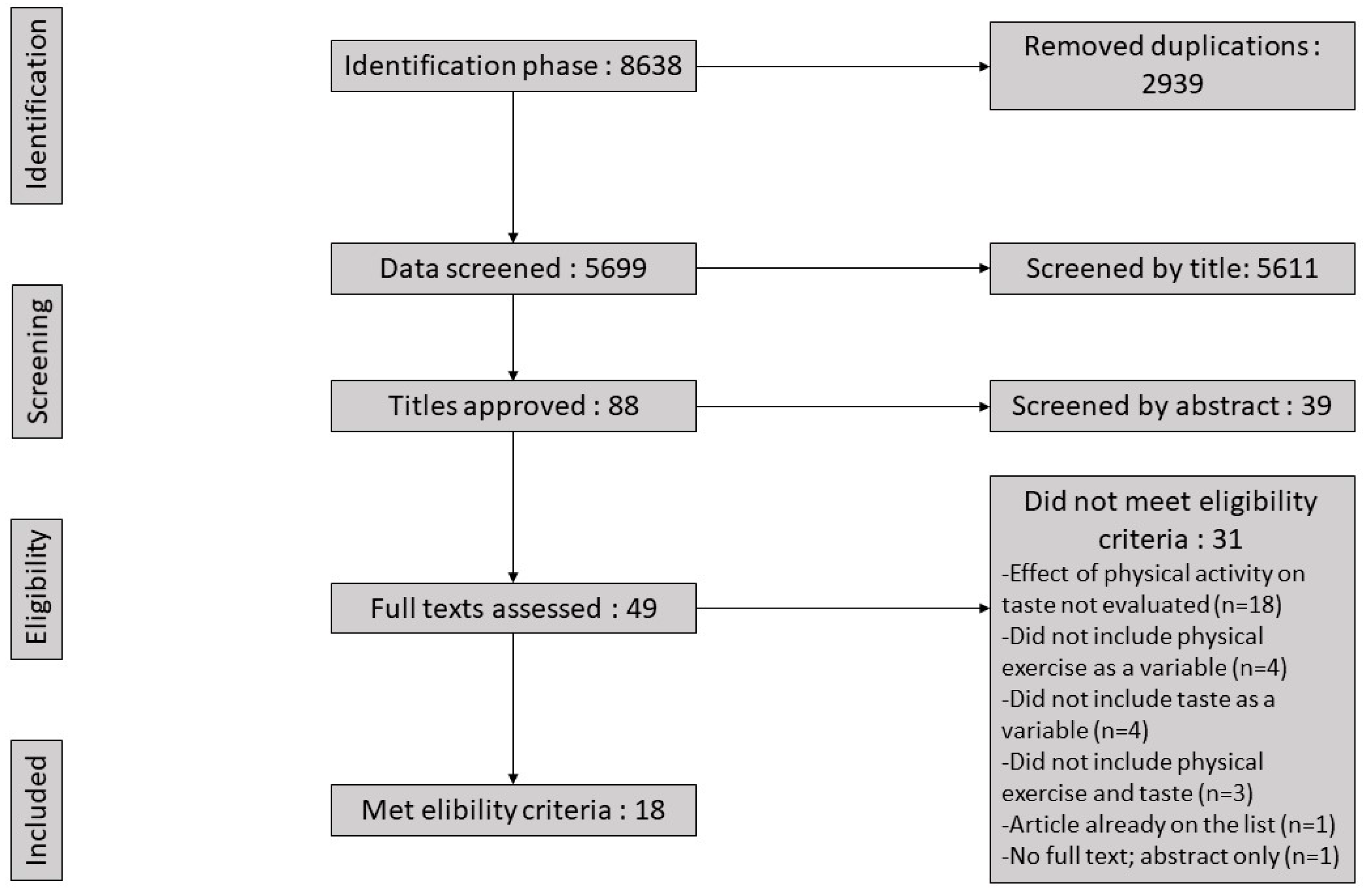
2.4. Risk of Bias and Quality Assessment
| Author, Year | Country | Design | N | Age (Years) | Sample Characteristics | Intervention Details | Type of Taste Measure | Main Results |
|---|---|---|---|---|---|---|---|---|
| Chrystal et al., 1995 [16] | United States of America | Experimental (nonrandomized trial) | 45 | 17–21 (experimental) 17–20 (control) | Division I varsity female swimmers (EX) and normal-weight females (CON) | Common conditions for all subjects: No caffeine, no MSG, no quinine, and no excessive sugar or excessive fat for 24 h before the test, with a low-fat, low-sucrose lunch 30 min before the test. No brushing teeth or chewing gum 30 min before the tests. Subjects were separated into 2 groups (exercise (EX) and control (CON)). Conditions for the CON group: 11 out of 17 of those who were initially chosen participated in all 4 tests; they completed different questionnaires on their eating habits, exercise habits, and other health-related traits in each session. Conditions for the EX group: Subjects were tested weekly for 4 weeks on the same day at the beginning of the athletic season. Subjects were then retested in the same manner at the end of the season for a total of 8 taste tests. | Visual analog scales. | The EX group and women who exercised more than 3 h/week (part of the CON group) all had decreased preferences for high sugar/fat stimuli when compared to CON (exercising less than 3 h/week). EX rated the sugar stimuli as significantly sweeter than CON but rated the fat stimuli as significantly less fatty. Significantly lower preference ratings were reported for the stimuli in the fall than controls in the winter (p < 0.01 and p < 0.05). |
| Kanarek et al., 1995 [17] | United States of America | Experimental (nonrandomized trial) | 55 | 18–21 | Healthy normal-weight female college students | Common conditions for all subjects: 34 out of 55 of those who were initially chosen were tested weekly for 4 weeks at the same time of day (11:30–12:00). Participants were asked to complete different questionnaires on their eating habits, exercise habits, sleep habits, and other health-related traits (TFEQ, POMS tests). Taste preference tests (tasting and rating samples of popcorn for palatability, saltiness, and fatness) were conducted in each session (4 total). | Taste preference with a hedonic rating scale. | Preference ratings for all samples of popcorn containing butter and salt were significantly higher for subjects who exercised more than 3 h/week compared to non-active and/or less-active groups (p < 0.01). |
| Nakagawa et al., 1996 [18] | Japan | Experimental | 55 | 30.0 (mental task; bitter/sour) 25.3 (mental task; sweet) 28.0 (physical task; bitter/sour) 23.7 (physical task; sweet) | Males and females | Common conditions for all subjects: Subjects were separated into 2 groups (10:30 and 13:30) and were tested daily for one session (approximately 1 h). The study included 2 conditions: mental task (MT) and physical task (PT). Conditions for the MT group: POMS questionnaire and taste tests. The test was repeated after 40 min of conditions. Conditions for the PT group: POMS questionnaire and taste tests. The test was repeated after 10 min of cycling on a 100-W ergometer rotating at 60 rpm at a considerable speed. | Time intensity scale test (taste intensity scale). | Significant decreases in perceived intensity and total amount of taste were observed for sourness in relation to the PT condition (p < 0.001 and p < 0.01). The buffering capacity of saliva was enhanced following hard exercise. |
| Horio and Kawamura, 1998 [19] | Japan | Experimental (nonrandomized trial) | 58 | 19–21 | Healthy non-active university students | Common conditions for all subjects: No food or drinks 1 h before the tests. Taste tests (sucrose (sweet), NaCI (salty), citric acid (sour), MSG (umami taste)) just before exercise, 3 min after beginning exercise, and right after a 20-min exercise session. The study included 2 conditions: exercise (EX) and control (CON). Conditions for the EX group: Taste tests, 30 min of cycling on an ergometer bicycle at a pedaling rate of 50 rpm with the intensity adjusted as a function of the calculated MHR; HR, BP, and skin temperature were monitored every 3 min during exercise. Conditions for the CON group: Taste tests, 30 min of rest, followed by taste tests. The CON condition was implemented prior to the EX condition for half the group, and vice versa. | Taste preference with a hedonic rating scale and triangle test; absolute taste detection threshold test. | Preference for sucrose and citric acid increased significantly post-exercise compared to pre-exercise (p < 0.01 and p < 0.05). |
| King et al., 1999 [20] | United Kingdom | Experimental (nonrandomized trial) | 16 | 21.3 | Healthy male students | Common conditions for all subjects in three situations (pre- and post-exercise drink): Participants completed questionnaires on physical activity levels, eating habits, and mental and physical health and performed a VO2max test. Then, they were assigned to one of 3 conditions in each session, drank a sample of a solution, rated their sensory characteristics, and ran on a treadmill (approximately 50 min at 70% VO2max). Afterwards, they drank another sample of the same solution, rated their sensory characteristics, and drank a larger quantity of the same drink before an ad libitum test meal was presented to them, with various questionnaires about hunger levels, appreciation of the meal, etc. | Visual analog scales. | A significant effect of time was observed for perceived pleasantness of the sweet drinks (p < 0.01). |
| Leshem et al., 1999 [21] | Israel | Experimental (nonrandomized trial) | 42 | 24.2 (control) 23.5 (exercisers) | Male students exercising (EX) 40–60 min 2–3 times a week and non-active male students (CON) | Common conditions for all subjects: No exercise the day before the experiment, a taste test in the morning (7:00–9:00) before eating or drinking anything other than water, a retest the following morning, and (12 h after exercise) an interview regarding their nutritional habits. Their preferences for salty or sweet snack items were considered based on added NaCl in soup and sugar in tea. Subjects were separated into 2 groups (exercise (EX) and control (CON)). Conditions for the EX group: Exercised in the evening (19:00–21:00) and had a second test within 30 min after the exercise session. Conditions for the CON group: Were asked to not consume anything 2 h before the evening taste test. | Preferred concentrations of salt and sugar were controlled by the participants; preferred snacks eaten were recorded. | The preference for salt increased significantly in each exercise group after exercise compared to the control (p < 0.05). |
| Wald and Leshem, 2003 [22] | Israel | Experimental (nonrandomized trial) | 80 | 23.5 (males) 23.1 (females) | Students exercising both in basketball and scheduled aerobics (males and females) | Common conditions for all subjects: No coffee or alcohol 12 h before testing. Participants drank 100 mL of a novel drink and swallowed a capsule, either empty (placebo) or containing 200, 400, or 600 mg NaCl. The subjects were then divided into 4 groups equally participating in basketball or aerobic exercises (MHR @ 96.1% after 60 min). Taste preference tests were conducted after each exercise session. Four exercise sessions (EX) were conducted, but the first one was only for familiarization (FAM). The 3 other sessions were the same but included test trials. | Taste/flavor preference and intensity with line rating. | Flavor/taste preference was significantly influenced by sodium concentration in a bell-shape manner (p < 0.005) and was highly dependent on sweat levels (p < 0.005). |
| Horio, 2004 [23] | Japan | Experimental (nonrandomized trial) | 44 | 19–21 | Healthy non-active university students (males and females) | Common conditions for all subjects: No food 1 h before the tests, breakfast at 8 am. Taste tests (containing glucose, sucrose, D-sorbitol, stevioside, erythritol, and saccharin) were conducted just before exercise (or control) and right after 20 min of exercise. The study included 2 conditions: exercise (EX) or control (CON). Conditions for the EX group: Taste tests, 30 min @ 50 rpm with the intensity adjusted as a function of the calculated MHR on a bicycle ergometer, followed by repeated taste tests. Conditions for the CON group: Taste tests, 30 min of rest, followed by repeated taste tests. The CON condition was implemented prior to the EX condition for half of the group, and vice versa. | Taste preference with a hedonic rating scale. | Preferences for sucrose, glucose, stevioside, D-sorbitol, and erythritol all increased significantly (p < 0.01, p < 0.05, p < 0.01, p < 0.01, and p < 0.01) following the EX condition (pre- vs. post-EX). |
| Passe et al., 2004 [24] | United States of America | Experimental (nonrandomized trial) | 50 | 36.4 | Triathletes (males and females) | Common conditions for all subjects in 5 situations: Drinks reviewed included diluted orange juice (DOJ), homemade 6% carbohydrate-electrolyte sports beverage (HCE), commercially available 6% carbohydrate-electrolyte sports beverage (CCE), and water (W). The first session was an orientation session, with water used for testing. In the 4 subsequent sessions, participants were assigned to a new solution/drink with varying concentrations. Subjects were assigned to a drink first and then exercised 75 min @ 80–85% of the age-predicted MDOJHR. During exercise, at the 30- and 60-min marks, participants completed a hedonic rating scale and a sensory rating scale for the assigned drink. | Descriptive line scale and taste preference with a hedonic rating scale. | DOJ and W sweetness intensity ratings were significantly lower than those for HCE and CCE (p < 0.05) 60 min after exercise. DOJ, HCE, and CCE had significantly lower saltiness ratings than W (p < 0.05) 60 min after exercise. In terms of tartness intensity ratings, after 60 min of exercise, DOJ was considered the tartest and W was considered the least tart, with a significant difference in ratings between all drinks (p < 0.05). |
| Havermans et al., 2009 [25] | Netherlands | Experimental (randomized trial) | 58 | 21.9 | Undergraduate students (males and females) | Common conditions for all subjects: No eating or drinking 2 h before the experiment. All participants tasted and rated all 3 solutions upon arrival. Subjects completed a 15-min cognitive task followed by sensory ratings of gastrointestinal feelings. One of the 3 drinks (containing the solutions) was given to each subject before and after exercise. Subjects were separated into 2 groups (TASTE and DRINK) with 3 visits. TASTE: Participants tasted and spit out the drink 5 min before exercise, engaged in 30 min of treadmill exercise @ 80% of the calculated MHR, performed a sensory rating of gastrointestinal feeling, tasted and spit out the same drink, and performed 15 min of a cognitive task followed by taste tests of all three drinks with ratings. DRINK: Participants consumed all of the drink 5 min before exercise, engaged in 30 min of treadmill exercise @ 80% of the calculated MHR, performed a sensory rating of gastrointestinal feeling, consumed all of the drink, and performed 15 min of a cognitive task followed by taste tests of all 3 solutions with ratings. | Line scales (visual analog scales). | No significant results. |
| Narukawa et al., 2009 [26] | Japan | Experimental (nonrandomized trial) | 35 | 24.7 | Normal-weight male and female runners with healthy habits | Common conditions for all subjects: No food or drink 1 h before the taste test (except water). Participants were all running in a half marathon. Subject sensory evaluations were performed 1 h before the half marathon and immediately after (2 tests in total). The participants were asked to assess their physical fatigue before and after the half marathon. For sensory evaluation, the triangle test was used, which included one glass of sucrose solution and 2 glasses of distilled water provided in descending order of sucrose concentration. | Triangle test and taste detection absolute threshold test. | The absolute taste detection threshold of sucrose decreased following the half marathon, dropping from 11.9 ± 1.0 mM (p = 0.14) to 7.7 ± 0.8 mM (p < 0.001). |
| Passe et al., 2009 [27] | United States of America | Experimental (nonrandomized trial) | 55 | 39.7 (males) 37.2 (females) | Normal-weight triathletes or runners (males and females) | Common conditions for all subjects: No physical activity and standardized nutrition the day before testing. Participants attend 6 visits, including one sedentary (SED) and 5 exercise (EX) testing conditions, with one sodium concentration for each session. SED: Blind-folded taste tests. EX: Blind-folded taste tests pre-exercise, aerobic circuit for 2 h @ 75% of the MHR, followed by taste tests at 60 and 120 min. Five test drinks, varying in sodium concentration (0,18, 30, 40, and 60 mmol/l), were evaluated in taste tests. | Visual analog scale and taste preference with a hedonic rating scale. | Exercise status and sodium level had a significant interaction with liking of the overall drink (p < 0.001). Exercise status had a significant main effect on the overall liking of the drink (p = 0.027). Exercise status and sodium level had a significant interaction with the liking of flavor and tartness (p = 0.046 and p = 0.034). The liking of sweetness, flavor, and tartness had a significant main effect for those in the exercise condition (p = 0.026, p = 0.035, and p = 0.004). Exercise status had a significant main effect on perceived intensity of tartness (p = 0.002). |
| Narukawa et al., 2010 [28] | Japan | Experimental (nonrandomized trial) | 13 | 29.8 | Normal-weight males and females in good physical health | Common conditions for all subjects: Arrival at 4 am in a fasted state (or at least no food 2 h prior). No drink or food 1 h before each taste test, but no restrictions during the session. Participants performed a 36-km mountain hike with 3 stops (16-, 25-, and 36-km marks) and 4 taste evaluations within 12 h. Participants tasted 2 solutions (100- and 300-mM sucrose solutions) 4 times (8) and rated their physical fatigue according to each distance. | Visual analog scale and a hedonic rating scale for palatability. | No significant results. |
| Wen and Song, 2010 [29] | China | Case control | 900 | 40–75 | Patients with confirmed cases of gastric cancer and controls without any cancer-related traits | Common conditions for all subjects: Subjects were selected from 2 groups (hospital patients and control group). All groups were asked to complete a questionnaire, and one-on-one interviews were conducted with each subject. The questionnaire included lifestyle habits, physical activity level, dietary habits, alcohol consumption, and other health-related sections. After interviews, a salt taste sensitivity threshold test (STST; 5 drops of NaCl solution were placed on the tongue) was performed for each participant. | Salt taste sensitivity threshold test (STST). | Significant correlation between physical activity and STST, with a Spearman rank correlation coefficient of 0.22 (p < 0.001). |
| Ali et al., 2011 [30] | New Zealand | Experimental (nonrandomized trial) | 14 | 24.4 | Healthy male recreational exercisers | Common conditions for all subjects: Controlled lifestyle and dietary factors 24 h before the test and a 3-h fast before each session. The group performed a sensory evaluation (including 3 sports drinks (high-carbohydrate/high-electrolytes, high-carbohydrate/low-electrolytes, low-carbohydrate/high-electrolytes) and water) 30 min before the exercise session. The other tests were conducted at 0, 30, 60, 90, and 120 min after the beginning of the exercise session. Participants performed a 60-min running session @ 70% of the MHR on a treadmill. All participants were tested weekly for 4 weeks (5 sessions in total, including the initial familiarization session). | Continuous analog scales. | Sweetness ratings were significantly higher during exercise compared to pre- and post-exercise (p < 0.001). Ratings for the high-carbohydrate/high-electrolyte drink decreased during exercise compared with post-exercise (p = 0.001). Ratings for the high-carbohydrate/low-electrolyte drink increased from pre-exercise to in-exercise (p < 0.001). Ratings for the low-carbohydrate/high-electrolyte drink increased from pre-exercise to in-exercise, and ratings were higher for pre- vs. post-exercise (p = 0.004 and p = 0.003). Sweetness ratings increased with exercise duration (p = 0.038). Saltiness ratings were lower in-exercise compared to pre-exercise (p = 0.003). Ratings decreased for pre- vs. in-exercise for the high-carbohydrate/high-electrolyte drink (p = 0.001). Saltiness ratings decreased with exercise duration (p < 0.01). |
| Martins et al., 2017 [31] | Norway | Experimental (randomized trial) | 46 | 34.4 | Sedentary healthy obese males and females | Common conditions for all subjects: 12-week exercise regimen (3 times/week) with monitoring of anthropometric measurements, body composition, maximal oxygen consumption, food preferences, and rewards before and after the end of the intervention (48 h after the last session). Fasting and postprandial subjective feelings of appetite and plasma concentrations of appetite hormones were measured before and after a standardized breakfast (every 30 min until 3 h) before and after the 12 weeks. Fat and sweet taste preferences and food rewards were measured using the Leeds Food Preference Questionnaire. Participants were separated into 3 groups: moderate-intensity continuous training (MICT), high-intensity interval training (HIIT), and 1/2 HIIT. | Leeds Food Preference Questionnaire. | Decreased explicit wanting, liking, and preference for savory relative to sweet foods (p < 0.001) was observed after 12 weeks. |
| Feeney et al., 2019 [32] | Ireland | Experimental (nonrandomized trial) | 30 | 19–51 | Healthy active (EX) and inactive (CON) males | Common conditions for all subjects: No strongly flavored food or drink 12 h before the test and no strenuous exercise 24 h before the test. Subjects were separated into 2 groups (inactive and active) according to their physical activity level. All participants were tested weekly for 2 weeks (2 sessions in total). Subjects were asked to taste 6 solutions with varying concentrations, to identify the substance, and to note the intensity and overall liking of the drinks (sucrose (sweet), citric acid (sour), sodium chloride (salt), quinine (bitter), and MSG (umami)). Participants were asked to complete different questionnaires (TFEQ and FFQ) on the last visit. | General labeled magnitude scale and general degree of liking scale. | The EX group was significantly better at identifying the umami taste compared to the CON group in general (p < 0.03). The EX group gave a higher rating for perceived intensity of high-concentration sucrose and high- and low-concentration umami than the CON group (p < 0.01, p < 0.01, and p < 0.05). The EX group also gave significantly lower ratings for the low-concentration carbohydrate taste and umami taste compared to the CON group (p < 0.01 and p < 0.01). |
| Josaphat et al., 2020 [33] | Canada | Experimental (nonrandomized trial) | 12 | 18–35 | Normal-weight non-athlete males | Common conditions for all subjects: No physical activity 24 h before the test and appetite sensation examination before sensory perception tests. In addition to the control group, subjects were separated into 2 groups with two visits: (1) EX9:40 and (2) EX10:30. EX9:40: 30 min of moderate to vigorous exercise on a treadmill at 70% of VO2max, followed by sensory perception tests and an ad libitum buffet-type meal. EX10:30: 90-min sedentary break before exercising, followed by sensory perception tests and an ad libitum buffet-type meal. Taste and smell tests were performed shortly after arrival and before lunch for both the CON and EX groups. For taste and smell tests, 10 liquid samples were considered. | Visual analog scales. | The timing of exercise in relation to a single ad libitum meal does not influence taste or smell perceptions or energy intake (p > 0.05 in all cases). |
| Author, Year | Variables Measured | Type of Exercise Description | Taste Experimental Protocol | Type of Taste Tests Description | Tests Duration |
|---|---|---|---|---|---|
| Chrystal et al., 1995 [16] | BMI, taste perceptions with hedonic and intensity scale, dietary restraint and disinhibition. | Exercise quantity was evaluated with a questionnaire and ranged from no exercise (control) to varsity college swimmers (exercise). | 16 samples of milk were presented to the subjects with varying concentrations of sucrose (0%, 5%, 10% and 20%) and fat (0%, 3.5%, 10.5% and 37.6%) which were served every session (4 or 8). | The participants evaluated sweetness, pleasantness, and fatness on 160-mm on visual analog scales. | 4 weeks, once/week or 8 weeks, once/week. |
| Kanarek et al., 1995 [17] | BMI, hedonic rating taste preferences, POMS results, menstrual cycle, dietary restraint and disinhibition and TFEQ. | Exercise quantity was evaluated with a questionnaire and ranged from no exercise to more than 3 h of exercise/week. | 9 samples of popcorn were presented to the subjects with varying concentrations of both butter (3.3 g, 10.0 g, 30.0 g) and salt (0.0 g, 1.5 g, 4.0 g) which were served every session (4). | The preference test offered a hedonic scale rating from 1 (least pleasant) to 9 (most pleasant) for palatability, saltiness, and fattiness. | 4 weeks, once/week. |
| Nakagawa et al., 1996 [18] | POMS results and time intensity test. | Exercise quantity was evaluated with POMS and self-examination. Participants also did a 10 min @100w for 60 rpm on an ergometer bicycle. | 3 different samples of mixed water with sweet (sucrose; 2.63 ± 10−1 M), bitter (quinine sulfate; 1.82 ± 10−5 M), and sour (citric acid; 1.37 ± 10−2 M) taste test which were tested 2 times/session. | Time intensity scale test (taste intensity scale) filled on an online computer with a 30cm slide-type input device. | One session. |
| Horio and Kawamura, 1998 [19] | VO2max, hedonic rating taste preferences, absolute taste detection threshold, and heart rate. | Exercise modality was 30 min @50rpm with adjusted intensity on bicycle ergometer in function of the calculated MHR: exercising heart rate in beats per min. = (Maximum age-adjusted heart rate - resting heart rate) × 50% + resting heart rate. | 5 different substances with different tastes (sucrose; sweet, NaCl; salty, citric acid; sour, caffeine; bitter, MSG; umami) with 5 varying concentrations, for a total of 25 drinks, were included in the taste preference test. The absolute taste detection threshold test had the same substances, but with 6 different concentrations. All tests were served before and after exercise and control condition for both tests and subjects had to detect the tastant-containing glass compared to a water-containing glass for every concentration. | The preference test offered a hedonic scale rating from 0 (extremely unpleasant) to 7 (extremely pleasant). The triangle test or the absolute taste detection threshold test consisted in the detection of the tastant-containing glass compared to a water-containing glass for every concentration. | 2–3 days/visits. |
| King et al., 1999 [20] | BMI, VO2max, sensory and hedonic taste ratings, hunger profile, and energy expenditure. | Running on a treadmill for 45 min @70% VO2max. | 3 conditions were present: exercise—bottled mineral water, exercise—low energy artificially sweetened fruit drink, exercise—high energy sucrose sweetened fruit drink. Each drink corresponds to a different session. | 100-mm visual analog scales with which participants evaluated pleasantness, sweetness, palatability, and their desire to drink it. | 4 sessions, 3 experimental. |
| Leshem et al., 1999 [21] | Body weight, hedonic rating taste preferences, and number of snacks eaten. | The intended sports program included fitness (10), basketball (6), and jogging (5) for 40–60 min in 3 groups. Exercise was evaluated in the morning before exercise, immediately after exercise, and on the following morning. | Participants would adjust the concentration of tomato soup by adding salt until they consider it “The most tasty”. Same applies for tea, except with sugar instead. Participants were also encouraged to eat freely from snacks that were both sugary or salty and the number of snacks eaten was recorded. | Adjusting concentration of salt or sugar until “The most tasty” concentration is attained (and then measured). Eating snacks freely and comparing which sugary or salty snacks were the most popular. | 24-h period testing. |
| Wald and Leshem, 2003 [22] | Body weight, body weight loss, taste/flavor preference, and estimated MHR. | The intended sports program included basketball or aerobic exercises with an MHR @96.1% after 60 min. | Participants were divided into 4 groups equally and a standardized drink (root beer) which they were unfamiliar with was served every session with a capsule containing different NaCl concentrations (0, 200, 400, 600 mg). | Evaluation of flavor/taste preference and intensity with a line rating ranging from 0.0 to 10.0cm (from disgusting to excellent). | 4 sessions. |
| Horio, 2004 [23] | Body weight, gustative perceptions and hedonic ratings, and fluid intake and predicted MHR. | Exercise modality was 75 min @80–85% of the age predicted MHR on bicycles ergometer, treadmills, and elliptical cross trainers. | 4 different drinks with varying concentrations of carbohydrates, electrolytes, and other components were the solutions presented to the participants; diluted orange juice, homemade sport drink, commercial sport drink, and water. Each visit, participants were paired with one of the drinks (4). | The participants evaluated sweetness, saltiness, tartness, and thirst quenching on 100-point descriptive line scales. Liking of the overall drink, liking of flavor, liking of sweetness was measured with a 9-point hedonic scale. | 5 sessions, 4 experimental. |
| Passe et al., 2004 [24] | Body weight, total body water, VO2max, hedonic rating taste preferences, and heart rate. | Exercise modality was 30 min @50 rpm with adjusted intensity in function of the calculated MHR: exercising heart rate in beats per min. = (Maximum age-adjusted heart rate - resting heart rate) × 50% + resting heart rate. | 5 different sweet substances (sucrose, glucose, stevioside, sorbitol, erythritol, saccharin) with 6 varying concentrations, for a total of 30 drinks, were served before and after EX and CON condition. | Preference hedonic rating scale ranging from +3 (extremely pleasant) to -3 (extremely unpleasant). | 2 days/visits. |
| Havermans et al., 2009 [25] | BMI, hedonic ratings for odor and taste, gastrointestinal sensory feelings, and MHR predicted by age. | Exercise quantity was evaluated with 30 min @80% of calculated MHR (207–0.7 * age; Gellish et al., 2007). | 3 different drinks (mali-flavored or sala-flavored lemonade, cream soda) were smelled, tasted, and evaluated for hedonic ratings by each participant at the beginning and the end of the session (2). Afterward, subjects were given one of the 3 solutions for the whole session, which they consumed before and after exercise and the cognitive task (2). | 100-mm and 200-mm line scales (visual analog scales) with hedonic ratings and sensory gastrointestinal feelings. | 3 sessions. |
| Narukawa et al., 2009 [26] | Absolute taste detection threshold and subjective physical fatigue. | Participants ran a half marathon as fast as possible at their own pace. | 6 different samples each containing 2 cups of distilled water and 1 containing sucrose were presented to the participants. Subjects had to detect the tastant-containing glass compared to the water-containing glasses for every concentration (40, 20, 10, 5, 2.5, and 1.25 mM). | The triangle test or the absolute taste threshold test consisted in the detection of the tastant-containing glass compared to water-containing glasses for every concentration. | One session. |
| Passe et al., 2009 [27] | Body mass, mean body mass loss, fluid intake, gustative perceptions and hedonic ratings, and MHR predicted by age. | Exercise modality was a 2-h circuit @70–75% of predicted MHR. | 5 different drinks with the same flavor (lemon-lime), carbohydrates concentration (6%), and potassium (2.9 mmol/L), but different concentrations of sodium (0, 18, 30, 40, and 60 mmol/L), were tested for the sedentary condition. Every other exercise testing day, only one sodium concentrated drink was tested (for a total of 5 sessions). | The participants evaluated sweetness, saltiness, tartness, bitterness, flavor strength, and ideal saltiness on 100-mm visual analog scales. Liking of the overall drink, sweetness, tartness, flavor, and saltiness was measured with a 9-point hedonic scale. | 6 visits, 2–3 sessions/weeks. |
| Narukawa et al., 2010 [28] | Body weight, taste sensitivity and palatability, subjective physical fatigue, number of steps, and energy consumption. | 12-h hike with 3 pauses at 16, 25 and 36 km marks. | 2 different drinks with varying concentrations of sucrose (100 and 300-Mm) were served to the participants. Participants evaluated each drink 4 times (including before the start of the hike) for a total of 8 measurements. Each test was followed by a 30-min break. | The participants evaluated intensity and the palatability on 100-mm visual analog scales. Subjects were also asked to rate the palatability on a hedonic scale ranging from -2 (unpleasant) to 2 (pleasant). | One session. |
| Wen and Song, 2010 [29] | BMI and STST. | Exercise quantity was evaluated with a questionnaire with 3 categories: never, 3 times or less/week, and 4 times or more/week. | 10 different samples were presented to each participant ranging from 0.22 to 58.4 g/L of NaCl. Between every sample, mouth would be rinsed and there would be a 30-s break before the other test. Participants evaluated the solutions once. | Taste sensitivity threshold test (STST); participants needed to associate each concentration with certain food. | One session. |
| Ali et al., 2011 [30] | Body mass, sensory evaluations, RPE, and MHR predicted by age. | Exercise modality was 60 min of moderate to vigorous exercise session on the treadmill at 70% calculated MHR. | 4 different drinks with key variances were served to the participants: (high-carbohydrate/high-electrolytes, high-carbohydrate/low-electrolytes, low-carbohydrate/high-electrolytes) or water. Participants were evaluating one substance each session (4). | The participants evaluated intensity of sweetness, saltiness, thirst-quenching ability, and overall liking on 100-mm continuous analog scales. | 5 weeks, once/week. |
| Martins et al., 2017 [31] | BMI, VO2max, fat and sweet taste preference, PA level, dietary habits, and MHR. | 12 weeks, 3 times/week with one of the following conditions: MICT (continuous exercise @75% of MHR for 250 kcal); HIIT (8s @85–90% of MHR and 12s of active recovery for 250 kcal); 1/2 HIIT (same protocol as HIIT, but for 125 kcal). Calories were measured according to VO2max. All sessions were performed on ergometers. | Participants were divided into 3 groups. Fat and sweet preferences were assessed with the Leeds Food Preference Questionnaire and a display of multiple images of different foods with varying energetic content and taste characteristics before and after a standardized breakfast and before and after the exercise intervention. | Leeds Food Preference Questionnaire protocol. | 12 weeks, 3 sessions/week. |
| Feeney et al., 2019 [32] | BMI, body fat, general taste perceptions, dietary restraint, and disinhibition. | Exercise quantity was evaluated with a questionnaire and ranged from inactive (CON), defined as less than 1 structured exercise session/week, or active (EX), defined as at least 4 structured exercise sessions/week. | 6 different substances of varying concentrations, ranging from low concentration to high concentration (total of 12 samples), of sucrose, acid citric, NaCl, MSG, quinine, and maltodextrin, were served every session (2). Participants were asked to identify the substance, to rate their intensity and their overall liking of the drinks. 30-sec break between substances and 2-min. break after 4 samples. | gLMS (general labeled magnitude scale), which is a validated scale for taste intensity, and gDOL (general degree of liking scale), which is a validated scale for liking of the stimuli. | 2 weeks, once/week. |
| Josaphat et al., 2020 [33] | Body mass, body fat, waist circumference, VO2max, gustative and olfactory sensory perception, dietary restraint and disinhibition. | Moderate to vigorous exercise session on the treadmill at 70% of VO2max. | 10 samples of milk with varying concentrations of fat (1%, 3.25%, 5%, 10%, and 15%) and sugary syrup (3%, 6%, 8%, 10%, and 12% carbohydrates) were served before and after the intervention. Ad libitum buffet-type meal composed of 14 liquid and solid items was served at lunch. The buffet was served in a private room with the same presentation and in controlled ambient conditions (odor, light, and temperature) on both visits. Appetite sensations were self-reported by the participants. | The participants evaluated sweetness, saltiness, fattiness, and liking on 100-mm visual analog scales. | 2 days/visit. |
3. Results
3.1. Study Selection
3.2. Study and Intervention Characteristics
3.3. Taste Protocols and Tests
3.4. Effectiveness of Intervention and Outcomes
3.5. Risk of Bias Assessment
4. Discussion
4.1. Overall Results and Takeaways
4.2. Sweet
4.3. Salty
4.4. Bitter/Sour/Umami
4.5. How Can We Measure Taste with a Novel Scope and How Can We Produce More Meaningful Results?
4.6. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lutter, M.; Nestler, E.J. Homeostatic and hedonic signals interact in the regulation of food intake. J. Nutr. 2009, 139, 629–632. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, C.A.; Cannon, G.; Levy, R.B.; Moubarac, J.C.; Louzada, M.L.; Rauber, F.; Khandpur, N.; Cediel, G.; Neri, D.; Martinez-Steele, E. Ultra-processed foods: What they are and how to identify them. Public Health Nutr. 2019, 22, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.D.; Ayuketah, A.; Brychta, R.; Cai, H.; Cassimatis, T.; Chen, K.Y.; Chung, S.T.; Costa, E.; Courville, A.; Darcey, V. Ultra-processed diets cause excess calorie intake and weight gain: An inpatient randomized controlled trial of ad libitum food intake. Cell Metab. 2019, 30, 67–77. [Google Scholar] [CrossRef]
- Lampuré, A.; Castetbon, K.; Deglaire, A.; Schlich, P.; Péneau, S.; Hercberg, S.; Méjean, C. Associations between liking for fat, sweet or salt and obesity risk in French adults: A prospective cohort study. Int. J. Behav. Nutr. Phys. Act. 2016, 13, 74. [Google Scholar] [CrossRef]
- Nasser, J. Taste, food intake and obesity. Obes. Rev. 2001, 2, 213–218. [Google Scholar] [CrossRef]
- Jayasinghe, S.N.; Kruger, R.; Walsh, D.C.; Cao, G.; Rivers, S.; Richter, M.; Breier, B.H. Is sweet taste perception associated with sweet food liking and intake? Nutrients 2017, 9, 750. [Google Scholar] [CrossRef] [PubMed]
- Després, J.P.; Lemieux, I. Abdominal obesity and metabolic syndrome. Nature 2006, 444, 881–887. [Google Scholar] [CrossRef] [PubMed]
- Golay, A.; Ybarra, J. Link between obesity and type 2 diabetes. Best Pract. Res. Clin. Endocrinol. Metab. 2005, 19, 649–663. [Google Scholar] [CrossRef]
- Church, T. Exercise in obesity, metabolic syndrome, and diabetes. Prog. Cardiovasc. Dis. 2011, 53, 412–418. [Google Scholar] [CrossRef]
- King, J.A.; Wasse, L.K.; Stensel, D.J. The acute effects of swimming on appetite, food intake, and plasma acylated ghrelin. J. Obes. 2011, D632-6. [Google Scholar] [CrossRef]
- Umabiki, M.; Tsuzaki, K.; Kotani, K.; Nagai, N.; Sano, Y.; Matsuoka, Y.; Kitaoka, K.; Okami, Y.; Sakane, N.; Higashi, A. The improvement of sweet taste sensitivity with decrease in serum leptin levels during weight loss in obese females. Tohoku J. Exp. Med. 2010, 220, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Sauer, H.; Ohla, K.; Dammann, D.; Teufel, M.; Zipfel, S.; Enck, P.; Mack, I. Changes in gustatory function and taste preference following weight loss. J. Pediatrics 2017, 182, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, M.E.; Reid, R.E.; King, N.A. Sensory profile of adults with reduced food intake and the potential roles of nutrition and physical activity interventions. Adv. Nutr. 2019, 10, 1120–1125. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef] [PubMed]
- Whiting, P.F.; Weswood, M.E.; Rutjes, A.W.; Reitsma, J.B.; Bossuyt, P.N.; Kleijnen, J. Evaluation of QUADAS, a tool for the quality assessment of diagnostic accuracy studies. BMC Med. Res. Methodol. 2006, 6, 9. [Google Scholar] [CrossRef]
- Crystal, S.; Frye, C.A.; Kanarek, R.B. Taste preferences and sensory perceptions in female varsity swimmers. Appetite 1995, 24, 25–36. [Google Scholar] [CrossRef]
- Kanarek, R.B.; Ryu, M.; Przypek, J. Preferences for foods with varying levels of salt and fat differ as a function of dietary restraint and exercise but not menstrual cycle. Physiol. Behav. 1995, 57, 821–826. [Google Scholar] [CrossRef]
- Nakagawa, M.; Mizuma, K.; Inui, T. Changes in taste perception following mental or physical stress. Chem. Senses 1996, 21, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Horio, T.; Kawamura, Y. Influence of physical exercise on human preferences for various taste solutions. Chem. Senses 1998, 23, 417–421. [Google Scholar] [CrossRef] [PubMed]
- King, N.A.; Appleton, K.; Rogers, P.J.; Blundell, J.E. Effects of sweetness and energy in drinks on food intake following exercise. Physiol. Behav. 1999, 66, 375–379. [Google Scholar] [CrossRef]
- Leshem, M.; Abutbul, A.; Eilon, R. Exercise increases the preference for salt in humans. Appetite 1999, 32, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Wald, N.; Leshem, M. Salt conditions a flavor preference or aversion after exercise depending on NaCl dose and sweat loss. Appetite 2003, 40, 277–284. [Google Scholar] [CrossRef]
- Horio, T. Effect of physical exercise on human preference for solutions of various sweet substances. Percept. Mot. Ski. 2004, 99, 1061–1070. [Google Scholar] [CrossRef]
- Passe, D.H.; Horn, M.; Stofan, J.; Murray, R. Palatability and voluntary intake of sports beverages, diluted orange juice, and water during exercise. Int. J. Sport Nutr. Exerc. Metab. 2004, 14, 272–284. [Google Scholar] [CrossRef]
- Havermans, R.C.; Salvy, S.J.; Jansen, A. Single-trial exercise-induced taste and odor aversion learning in humans. Appetite 2009, 53, 442–445. [Google Scholar] [CrossRef]
- Narukawa, M.; Ue, H.; Morita, K.; Kuga, S.; Isaka, T.; Hayashi, Y. Change in taste sensitivity to sucrose due to physical fatigue. Food Sci. Technol. Res. 2009, 15, 195–198. [Google Scholar] [CrossRef]
- Passe, D.H.; Stofan, J.R.; Rowe, C.L.; Horswill, C.A.; Murray, R. Exercise condition affects hedonic responses to sodium in a sport drink. Appetite 2009, 52, 561–567. [Google Scholar] [CrossRef]
- Narukawa, M.; Ue, H.; Uemura, M.; Morita, K.; Kuga, S.; Isaka, T.; Hayashi, Y. Influence of prolonged exercise on sweet taste perception. Food Sci. Technol. Res. 2010, 16, 513–516. [Google Scholar] [CrossRef]
- Wen, X.Y.; Song, F.M. Salt taste sensitivity, physical activity and gastric cancer. Asian Pac. J. Cancer Prev. 2010, 11, 1473–1477. [Google Scholar]
- Ali, A.; Duizer, L.; Foster, K.; Grigor, J.; Wei, W. Changes in sensory perception of sports drinks when consumed pre, during and post exercise. Physiol. Behav. 2011, 102, 437–443. [Google Scholar] [CrossRef]
- Martins, C.; Aschehoug, I.; Ludviksen, M.; Holst, J.; Finlayson, G.; Wisloff, U.; Morgan, L.; King, N.; Kulseng, B. High-intensity interval training, appetite, and reward value of food in the obese. Med. Sci. Sports Exerc. 2017, 49, 1851–1858. [Google Scholar] [CrossRef] [PubMed]
- Feeney, E.L.; Leacy, L.; O’Kelly, M.; Leacy, N.; Phelan, A.; Crowley, L.; Stynes, E.; de Casanove, A.; Horner, K. Sweet and umami taste perception differs with habitual exercise in males. Nutrients 2019, 11, 155. [Google Scholar] [CrossRef]
- Josaphat, K.J.; Drapeau, V.; Thivel, D.; Mathieu, M.E. Impact of Exercise Timing on Chemosensory Response, Appetite, and Energy Intake in Lean Males. Int. J. Sport Nutr. Exerc. Metab. 2020, 30, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, M.; Jeukendrup, A.; King, N.A.; Blundell, J.E. The relationship between substrate metabolism, exercise and appetite control. Sports Med. 2011, 41, 507–521. [Google Scholar] [CrossRef]
- Wan, H.Y.; Stickford, J.L.; Dawkins, E.J.; Lindeman, A.K.; Stager, J.M. Acute modulation in dietary behavior following glycogen depletion and postexercise supplementation in trained cyclists. Appl. Physiol. Nutr. Metab. 2018, 43, 1326–1333. [Google Scholar] [CrossRef] [PubMed]
- Pahnke, M.D.; Trinity, J.D.; Zachwieja, J.J.; Stofan, J.R.; Hiller, W.D.; Coyle, E.F. Serum sodium concentration changes are related to fluid balance and sweat sodium loss. Med. Sci. Sports Exerc. 2010, 42, 1669–1674. [Google Scholar] [CrossRef]
- Berridge, K.C.; Flynn, F.W.; Schulkin, J.; Grill, H.J. Sodium depletion enhances salt palatability in rats. Behav. Neurosci. 1984, 98, 652. [Google Scholar] [CrossRef]
- Church, T.; Martin, C.K. The obesity epidemic: A consequence of reduced energy expenditure and the uncoupling of energy intake? Obesity 2018, 26, 14–16. [Google Scholar] [CrossRef]
- Zheng, H.; Lenard, N.R.; Shin, A.C.; Berthoud, H.R. Appetite control and energy balance regulation in the modern world: Reward-driven brain overrides repletion signals. Int. J. Obes. 2009, 33, S8–S13. [Google Scholar] [CrossRef]
- Dorling, J.; Broom, D.R.; Burns, S.F.; Clayton, D.J.; Deighton, K.; James, L.J.; King, J.A.; Miyashita, M.; Thackray, A.E.; Batterham, R.L.; et al. Acute and chronic effects of exercise on appetite, energy intake, and appetite-related hormones: The modulating effect of adiposity, sex, and habitual physical activity. Nutrients 2018, 10, 1140. [Google Scholar] [CrossRef]
- Joo, J.; Williamson, S.A.; Vazquez, A.I.; Fernandez, J.R.; Bray, M.S. The influence of 15-week exercise training on dietary patterns among young adults. Int. J. Obes. 2019, 43, 1681–1690. [Google Scholar] [CrossRef] [PubMed]
- Schubert, M.M.; Sabapathy, S.; Leveritt, M.; Desbrow, B. Acute exercise and hormones related to appetite regulation: A meta-analysis. Sports Med. 2014, 44, 387–403. [Google Scholar] [CrossRef] [PubMed]
- Killgore, W.D.; Kipman, M.; Schwab, Z.J.; Tkachenko, O.; Preer, L.; Gogel, H.; Bark, J.S.; Mundy, E.A.; Olson, E.A.; Weber, M. Physical exercise and brain responses to images of high-calorie food. Neuroreport 2013, 24, 962–967. [Google Scholar] [CrossRef] [PubMed]
- Loper, H.B.; La Sala, M.; Dotson, C.; Steinle, N. Taste perception, associated hormonal modulation, and nutrient intake. Nutr. Rev. 2015, 73, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.E. Influence of Sensation and Liking on Eating and Drinking. In Handbook of Eating and Drinking: Interdisciplinary Perspectives; Springer: Cham, Switzlerland, 2020; pp. 131–155. [Google Scholar]
- De Graaf, K. Sensory Responses in Nutrition and Energy Balance: Role of Texture, Taste, and Smell in Eating Behavior. In Handbook of Eating and Drinking: Interdisciplinary Perspectives; Springer: Cham, Switzlerland, 2020; pp. 641–658. [Google Scholar]
- Johnson, F.; Wardle, J. Variety, palatability, and obesity. Adv. Nutr. 2014, 5, 851–859. [Google Scholar] [CrossRef]
- Gudziol, H.; Guntinas-Lichius, O. Electrophysiologic assessment of olfactory and gustatory function. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2019; Volume 164, pp. 247–262. [Google Scholar]
- Hardikar, S.; Wallroth, R.; Villringer, A.; Ohla, K. Shorter-lived neural taste representations in obese compared to lean individuals. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef]
- Yarnitsky, D.; Sprecher, E.; Zaslansky, R.; Hemli, J.A. Multiple session experimental pain measurement. Pain 1996, 67, 327–333. [Google Scholar] [CrossRef]
- Bijur, P.E.; Silver, W.; Gallagher, E.J. Reliability of the visual analog scale for measurement of acute pain. Acad. Emerg. Med. 2001, 8, 1153–1157. [Google Scholar] [CrossRef]
- Nakamura, Y.; Sanematsu, K.; Ohta, R.; Shirosaki, S.; Koyano, K.; Nonaka, K.; Shigemura, N.; Ninomiya, Y. Diurnal variation of human sweet taste recognition thresholds is correlated with plasma leptin levels. Diabetes 2008, 57, 2661–2665. [Google Scholar] [CrossRef]
- Noel, C.A.; Cassano, P.A.; Dando, R. College-aged males experience attenuated sweet and salty taste with modest weight gain. J. Nutr. 2017, 147, 1885–1891. [Google Scholar] [CrossRef]
- Overberg, J.; Hummel, T.; Krude, H.; Wiegand, S. Differences in taste sensitivity between obese and non-obese children and adolescents. Arch. Dis. Child. 2012, 97, 1048–1052. [Google Scholar] [CrossRef] [PubMed]
| Author, year | Risk of Bias | Applicability Concerns | |||||
|---|---|---|---|---|---|---|---|
| Patient Selection | Index Test | Reference Standard | Flow and Timing | Patient Selection | Index Test | Reference Standard | |
| Chrystal et al., 1995 [16] | (+) | (+) | (+) | (?) | (?) | (+) | (+) |
| Kanarek et al., 1995 [17] | (?) | (+) | (+) | (+) | (+) | (+) | (+) |
| Nakagawa et al., 1996 [18] | (+) | (+) | (+) | (?) | (+) | (+) | (+) |
| Horio and Kawamura, 1998 [19] | (?) | (+) | (+) | (+) | (+) | (+) | (+) |
| King et al., 1999 [20] | (+) | (+) | (+) | (+) | (+) | (+) | (+) |
| Leshem et al., 1999 [21] | (+) | (+) | (+) | (+) | (+) | (+) | (+) |
| Wald and Leshem, 2003 [22] | (+) | (+) | (+) | (+) | (+) | (+) | (+) |
| Horio, 2004 [23] | (?) | (+) | (+) | (+) | (?) | (+) | (+) |
| Passe et al., 2004 [24] | (?) | (+) | (+) | (?) | (+) | (+) | (+) |
| Havermans et al., 2009 [25] | (+) | (+) | (+) | (+) | (+) | (+) | (+) |
| Narukawa et al., 2009 [26] | (+) | (+) | (+) | (+) | (?) | (+) | (+) |
| Passe et al., 2009 [27] | (+) | (+) | (+) | (+) | (+) | (+) | (+) |
| Narukawa et al., 2010 [28] | (+) | (+) | (+) | (+) | (?) | (+) | (+) |
| Wen and Song, 2010 [29] | (+) | (+) | (+) | (+) | (?) | (+) | (+) |
| Ali et al., 2011 [30] | (+) | (+) | (+) | (+) | (+) | (+) | (+) |
| Martins et al., 2017 [31] | (+) | (+) | (+) | (+) | (+) | (+) | (+) |
| Feeney et al., 2019 [32] | (+) | (+) | (+) | (+) | (+) | (+) | (+) |
| Josaphat et al., 2020 [33] | (+) | (+) | (+) | (+) | (+) | (+) | (+) |
| Perceptions | ||||
|---|---|---|---|---|
| Intensity | Sensitivity | Preference | ||
| Taste | Sweet | ↑ (17, 18, 29) | ↑ (25) | ↑ (19, 20, 23, 24) ↓ (17, 29) |
| Salty | ↓ (28) | ↓ (33) | ↑ (18, 21, 22, 26) | |
| Sour | ↓ (16, 26) | - | ↑ (19, 26) | |
| Bitter | - | - | - | |
| Umami | ↑ (29) | ↑ (29) | ↓ (29) | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gauthier, A.-C.; Guimarães, R.d.F.; Namiranian, K.; Drapeau, V.; Mathieu, M.-E. Effect of Physical Exercise on Taste Perceptions: A Systematic Review. Nutrients 2020, 12, 2741. https://doi.org/10.3390/nu12092741
Gauthier A-C, Guimarães RdF, Namiranian K, Drapeau V, Mathieu M-E. Effect of Physical Exercise on Taste Perceptions: A Systematic Review. Nutrients. 2020; 12(9):2741. https://doi.org/10.3390/nu12092741
Chicago/Turabian StyleGauthier, Alexandre-Charles, Roseane de Fátima Guimarães, Khoosheh Namiranian, Vicky Drapeau, and Marie-Eve Mathieu. 2020. "Effect of Physical Exercise on Taste Perceptions: A Systematic Review" Nutrients 12, no. 9: 2741. https://doi.org/10.3390/nu12092741
APA StyleGauthier, A.-C., Guimarães, R. d. F., Namiranian, K., Drapeau, V., & Mathieu, M.-E. (2020). Effect of Physical Exercise on Taste Perceptions: A Systematic Review. Nutrients, 12(9), 2741. https://doi.org/10.3390/nu12092741





