Role of Vitamin C in Osteoporosis Development and Treatment—A Literature Review
Abstract
1. Introduction
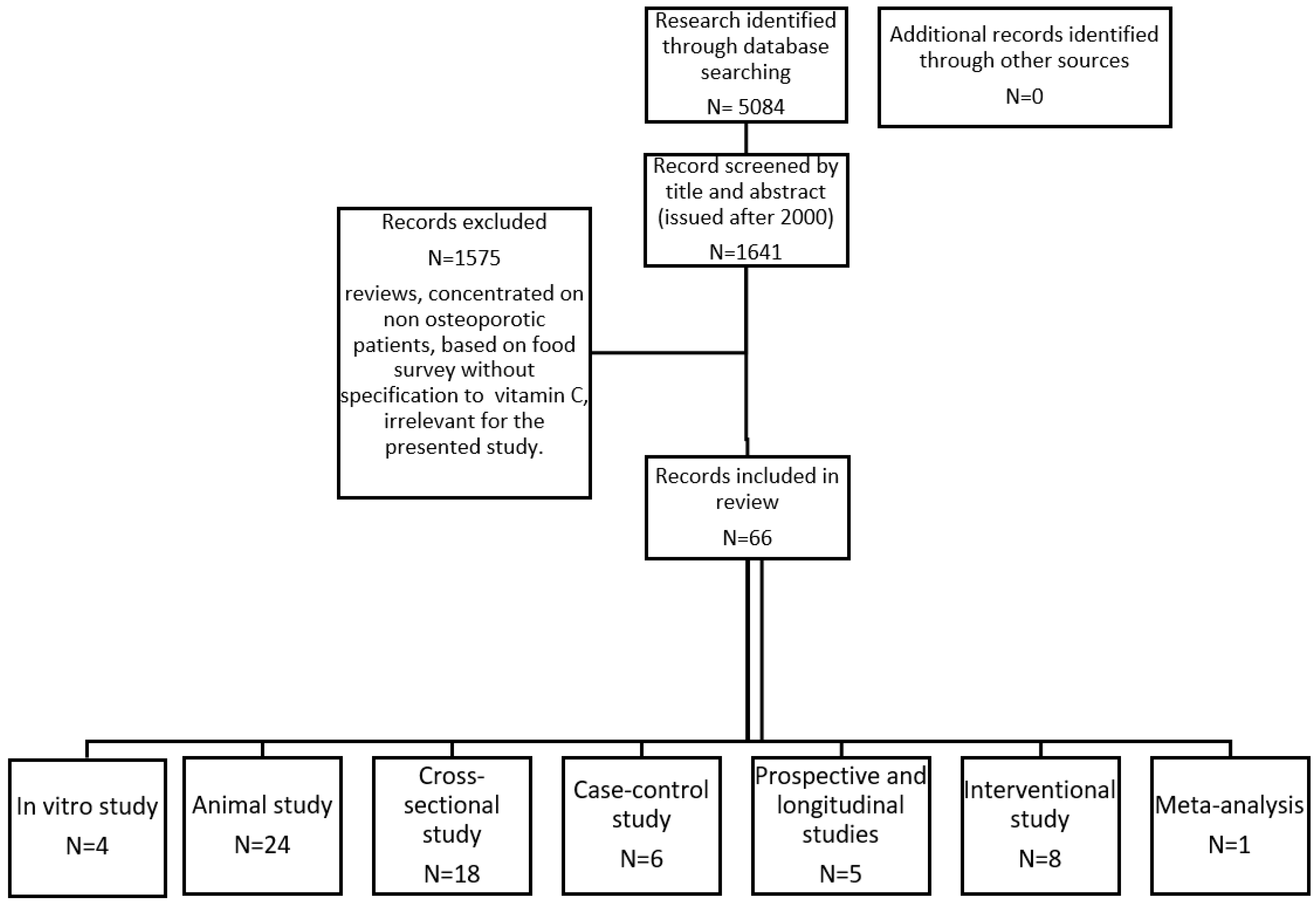
2. Materials and Methods
3. Results
3.1. Molecular Function of Vitamin C in Bone Tissue
3.1.1. Studies on Animal Models
3.1.2. In Vitro Models
3.2. Cross-Sectional Studies
3.3. Case-Control Study
3.4. Longitudinal and Prospective Studies
3.5. Interventional Studies
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bijlsma, A.Y.; Meskers, C.G.; Westendorp, R.G.; Maier, A.B. Chronology of age-related disease definitions: Osteoporosis and sarcopenia. Ageing Res. Rev. 2012, 11, 320–324. [Google Scholar] [CrossRef] [PubMed]
- The World Health Report 2004: Changing History; World Health Organization: Geneva, Switzerland, 2004.
- Kanis, J.A.; Compston, J.; Cooper, C.; Hernlund, E.; Ivergard, M.; Johansson, H. The burden of fractures in the European Union in 2010. Osteoporos. Int. 2012, 23 (Suppl. 2), S57. [Google Scholar]
- Pouresmaeili, F.; Kamalidehghan, B.; Kamarehei, M.; Goh, Y.M. A comprehensive overview on osteoporosis and its risk factors. Ther. Clin. Risk Manag. 2018, 14, 2029–2049. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.A.; Cooper, C.; Rizzoli, R.; Reginster, J.Y. Scientific Advisory Board of the European Society for Clinical and Economic Aspects of Osteoporosis (ESCEO) and the Committees of Scientific Advisors and National Societies of the International Osteoporosis Foundation (IOF). European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos. Int. 2019, 31, 209. [Google Scholar] [CrossRef]
- Muñoz-Garach, A.; García-Fontana, B.; Muñoz-Torres, M. Nutrients and Dietary Patterns Related to Osteoporosis. Nutrients 2020, 12, E1986. [Google Scholar] [CrossRef]
- Xu, Z.; Xie, Z.; Sun, J.; Huang, S.; Chen, Y.; Li, C.; Sun, X.; Xia, B.; Tian, L.; Guo, C.; et al. Gut Microbiome Reveals Specific Dysbiosis in Primary Osteoporosis. Front. Cell. Infect. Microbiol. 2020, 10, 160. [Google Scholar] [CrossRef]
- Snoddy, A.M.E.; Buckley, H.R.; Elliott, G.E.; Standen, V.G.; Arriaza, B.T.; Halcrow, S.E. Macroscopic features of scurvy in human skeletal remains: A literature synthesis and diagnostic guide. Am. J. Phys. Anthropol. 2018, 167, 876–895. [Google Scholar] [CrossRef]
- Tucker, K.L. Osteoporosis prevention and nutrition. Curr. Osteoporos. Rep. 2009, 7, 111–117. [Google Scholar] [CrossRef]
- Palacios, C. The role of nutrients in bone health, from A to Z. Crit. Rev. Food Sci. Nutr. 2006, 46, 621–628. [Google Scholar] [CrossRef]
- Malmir, H.; Shab-Bidar, S.; Djafarian, K. Vitamin C intake in relation to bone mineral density and risk of hip fracture and osteoporosis: A systematic review and meta-analysis of observational studies. Br. J. Nutr. 2018, 119, 847–858. [Google Scholar] [CrossRef]
- Zeng, L.F.; Luo, M.H.; Liang, G.H.; Yang, W.Y.; Xiao, X.; Wei, X.; Yu, J.; Guo, D.; Chen, H.Y.; Pan, J.K.; et al. Can Dietary Intake of Vitamin C-Oriented Foods Reduce the Risk of Osteoporosis, Fracture, and BMD Loss? Systematic Review with Meta-Analyses of Recent Studies. Front. Endocrinol. 2020, 10, 844. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Liu, C.; Bo, Y.; You, J.; Zhu, Y.; Duan, D.; Cui, H.; Lu, Q. Dietary vitamin C intake and the risk of hip fracture: A dose-response meta-analysis. Osteoporos. Int. 2018, 29, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Padayatty, S.J.; Levine, M. Vitamin C: The known and the unknown and Goldilocks. Oral. Dis. 2016, 22, 463–493. [Google Scholar] [CrossRef] [PubMed]
- Smirnoff, N.; Conklin, P.L.; Loewus, F.A. Biosynthesis of ascorbic acid in plants: A Renaissance. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2001, 52, 437–467. [Google Scholar] [CrossRef] [PubMed]
- Fain, O. Musculoskeletal manifestations of scurvy. Joint Bone Spine 2005, 72, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Finck, H.; Hart, A.R.; Jennings, A.; Welch, A.A. Is there a role for vitamin C in preventing osteoporosis and fractures? A review of the potential underlying mechanisms and current epidemiological evidence. Nutr. Res. Rev. 2014, 27, 268–283. [Google Scholar] [CrossRef]
- Drouin, G.; Godin, J.R.; Pagé, B. The genetics of vitamin C loss in vertebrates. Curr. Genom. 2011, 12, 371–378. [Google Scholar] [CrossRef]
- Doba, T.; Burton, G.W.; Ingold, K.U. Antioxidant and co-antioxidant activity of vitamin C. The effect of vitamin C, either alone or in the presence of vitamin E or a water-soluble vitamin E analogue, upon the peroxidation of aqueous multilamellar phospholipid liposomes. Biochim. Biophys. Acta. 1985, 835, 298–303. [Google Scholar] [CrossRef]
- Padayatty, S.J.; Katz, A.; Wang, Y.; Eck, P.; Kwon, O.; Lee, J.H.; Chen, S.; Corpe, C.; Dutta, A.; Dutta, S.K.; et al. Vitamin C as an antioxidant: Evaluation of its role in disease prevention. J. Am. Coll. Nutr. 2003, 22, 18–35. [Google Scholar] [CrossRef]
- Bates, C.J. Vitamin C deficiency in guinea pigs: Variable sensitivity of collagen at different sites. Int. J. Vitam. Nutr. Res. 1979, 49, 77–86. [Google Scholar]
- Tsuchiya, H.; Bates, C.J. Ascorbic acid deficiency in guinea pigs: Contrasting effects of tissue ascorbic acid depletion and of associated inanition on status indices related to collagen and vitamin D. Br. J. Nutr. 1994, 72, 745–752. [Google Scholar] [CrossRef] [PubMed]
- Bates, C.J.; Tsuchiya, H. Comparison of vitamin C deficiency with food restriction on collagen cross-link ratios in bone, urine and skin of weanling guinea-pigs. Br. J. Nutr. 2003, 89, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Kipp, D.E.; McElvain, M.; Kimmel, D.B.; Akhter, M.P.; Robinson, R.G.; Lukert, B.P. Scurvy results in decreased collagen synthesis and bone density in the guinea pig animal. Bone 1996, 18, 281–288. [Google Scholar] [CrossRef]
- Aghajanian, P.; Hall, S.; Wongworawat, M.D.; Mohan, S. The Roles and Mechanisms of Actions of Vitamin C in Bone: New Developments. J. Bone Miner. Res. 2015, 30, 1945–1955. [Google Scholar] [CrossRef]
- Kim, W.; Bae, S.; Kim, H.; Kim, Y.; Choi, J.; Lim, S.Y.; Lee, H.J.; Lee, J.; Choi, J.; Jang, M.; et al. Ascorbic acid insufficiency induces the severe defect on bone formation via the down-regulation of osteocalcin production. Anat. Cell Biol. 2013, 46, 254–261. [Google Scholar] [CrossRef]
- Mohan, S.; Kapoor, A.; Singgih, A.; Zhang, Z.; Taylor, T.; Yu, H.; Chadwick, R.B.; Chung, Y.S.; Donahue, L.R.; Rosen, C.; et al. Spontaneous fractures in the mouse mutant sfx are caused by deletion of the gulonolactone oxidase gene, causing vitamin C deficiency. J. Bone Miner. Res. 2005, 20, 1597–1610. [Google Scholar] [CrossRef]
- Gabbay, K.H.; Bohren, K.M.; Morello, R.; Bertin, T.; Liu, J.; Vogel, P. Ascorbate synthesis pathway: Dual role of ascorbate in bone homeostasis. J. Biol. Chem. 2010, 285, 19510–19520. [Google Scholar] [CrossRef]
- Park, J.K.; Lee, E.M.; Kim, A.Y.; Lee, E.J.; Min, C.W.; Kang, K.K.; Lee, M.M.; Jeong, K.S. Vitamin C deficiency accelerates bone loss inducing an increase in PPAR-γ expression in SMP30 knockout mice. Int. J. Exp. Pathol. 2012, 93, 332–340. [Google Scholar] [CrossRef]
- Nishijima, K.; Ohno, T.; Amano, A.; Kishimoto, Y.; Kondo, Y.; Ishigami, A.; Tanaka, S. Bone Degeneration and Its Recovery in SMP30/GNL-Knockout Mice. J. Nutr. Health Aging 2017, 21, 573–578. [Google Scholar] [CrossRef]
- Li, Z.; Kuhn, G.; Schirmer, M.; Müller, R.; Ruffoni, D. Impaired bone formation in ovariectomized mice reduces implant integration as indicated by longitudinal in vivo micro-computed tomography. PLoS ONE 2017, 12, e0184835. [Google Scholar] [CrossRef]
- Zhu, L.L.; Cao, J.; Sun, M.; Yuen, T.; Zhou, R.; Li, J.; Peng, Y.; Moonga, S.S.; Guo, L.; Mechanick, J.I.; et al. Vitamin C prevents hypogonadal bone loss. PLoS ONE 2012, 7, e47058. [Google Scholar] [CrossRef] [PubMed]
- Deyhim, F.; Strong, K.; Deyhim, N.; Vandyousefi, S.; Stamatikos, A.; Faraji, B. Vitamin C reverses bone loss in an osteopenic rat model of osteoporosis. Int. J. Vitam. Nutr. Res. 2018, 88, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, J.; Sun, L.; Kumar, T.R.; Blair, H.C.; Zaidi, M. Follicle-stimulating hormone stimulates TNF production from immune cells to enhance osteoblast and osteoclast formation. Proc. Natl. Acad. Sci. USA. 2006, 103, 14925–14930. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.K.; Kim, G.J.; Yoo, H.S.; Song, D.H.; Chung, K.H.; Lee, K.J.; Koo, Y.T.; An, J.H. Vitamin C Activates Osteoblastogenesis and Inhibits Osteoclastogenesis via Wnt/β-Catenin/ATF4 Signaling Pathways. Nutrients 2019, 11, 506. [Google Scholar] [CrossRef]
- Urban, K.; Höhling, H.J.; Lüttenberg, B.; Szuwart, T.; Plate, U. Biomineralisation Research Unit. An in vitro study of osteoblast vitality influenced by the vitamins C and E. Head Face Med. 2012, 8, 25. [Google Scholar] [CrossRef]
- Takamizawa, S.; Maehata, Y.; Imai, K.; Senoo, H.; Sato, S.; Hata, R. Effects of ascorbic acid and ascorbic acid 2-phosphate, a long-acting vitamin C derivative, on the proliferation and differentiation of human osteoblast-like cells. Cell Biol. Int. 2004, 28, 255–265. [Google Scholar] [CrossRef]
- Hadzir, S.N.; Ibrahim, S.N.; Abdul Wahab, R.M.; Zainol Abidin, I.Z.; Senafi, S.; Ariffin, Z.Z.; Abdul Razak, M.; Zainal Ariffin, S.H. Ascorbic acid induces osteoblast differentiation of human suspension mononuclear cells. Cytotherapy 2014, 16, 674–682. [Google Scholar] [CrossRef]
- Otsuka, E.; Yamaguchi, A.; Hirose, S.; Hagiwara, H. Characterization of osteoblastic differentiation of stromal cell line ST2 that is induced by ascorbic acid. Am. J. Physiol. 1999, 277, C132–C138. [Google Scholar] [CrossRef]
- Lindsey, R.C.; Cheng, S.; Mohan, S. Vitamin C effects on 5-hydroxymethylcytosine and gene expression in osteoblasts and chondrocytes: Potential involvement of PHD2. PLoS ONE 2019, 14, e0220653. [Google Scholar] [CrossRef]
- Xing, W.; Singgih, A.; Kapoor, A.; Alarcon, C.M.; Baylink, D.J.; Mohan, S. Nuclear factor-E2-related factor-1 mediates ascorbic acid induction of osterix expression via interaction with antioxidant-responsive element in bone cells. J. Biol. Chem. 2007, 282, 22052–22061. [Google Scholar] [CrossRef]
- Malicev, E.; Woyniak, G.; Knezevic, M.; Radosavljević, D.; Jeras, M. Vitamin C induced apoptosis in human articular chondrocytes. Pflugers Arch. 2000, 440 (Suppl. 5), R46–R48. [Google Scholar] [CrossRef] [PubMed]
- Chang, Z.; Huo, L.; Li, P.; Wu, Y.; Zhang, P. Ascorbic acid provides protection for human chondrocytes against oxidative stress. Mol. Med. Rep. 2015, 12, 7086–7092. [Google Scholar] [CrossRef] [PubMed]
- Temu, T.M.; Wu, K.Y.; Gruppuso, P.A.; Phornphutkul, C. The mechanism of ascorbic acid-induced differentiation of ATDC5 chondrogenic cells. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E325–E334. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.I.; Park, K.Y.; Lee, Y.; Park, M.; Kim, J. Vitamin C-linker-conjugated tripeptide AHK stimulates BMP-2-induced osteogenic differentiation of mouse myoblast C2C12 cells. Differentiation 2018, 101, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Xing, W.; Pourteymoor, S.; Mohan, S. Ascorbic acid regulates osterix expression in osteoblasts by activation of prolyl hydroxylase and ubiquitination-mediated proteosomal degradation pathway. Physiol. Genom. 2011, 43, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Aghajanian, P.; Pourteymoor, S.; Alarcon, C.; Mohan, S. Prolyl Hydroxylase Domain-Containing Protein 2 (Phd2) Regulates Chondrocyte Differentiation and Secondary Ossification in Mice. Sci. Rep. 2016, 6, 35748. [Google Scholar] [CrossRef]
- Tsuneto, M.; Yamazaki, H.; Yoshino, M.; Yamada, T.; Hayashi, S. Ascorbic acid promotes osteoclastogenesis from embryonic stem cells. Biochem. Biophys. Res. Commun. 2005, 335, 1239–1246. [Google Scholar] [CrossRef]
- Le Nihouannen, D.; Barralet, J.E.; Fong, J.E.; Komarova, S.V. Ascorbic acid accelerates osteoclast formation and death. Bone 2010, 46, 1336–1343. [Google Scholar] [CrossRef]
- Kim, M.H.; Lee, H.J. Osteoporosis, vitamin C intake, and physical activity in Korean adults aged 50 years and over. J. Phys. Ther. Sci. 2016, 28, 725–730. [Google Scholar] [CrossRef]
- Kim, Y.A.; Kim, K.M.; Lim, S.; Choi, S.H.; Moon, J.H.; Kim, J.H.; Kim, S.W.; Jang, H.C.; Shin, C.S. Favorable effect of dietary vitamin C on bone mineral density in postmenopausal women (KNHANES IV, 2009): Discrepancies regarding skeletal sites, age, and vitamin D status. Osteoporos. Int. 2015, 26, 2329–2337. [Google Scholar] [CrossRef]
- Finck, H.; Hart, A.R.; Lentjes, M.A.; Jennings, A.; Luben, R.N.; Khaw, K.T.; Welch, A.A. Cross-sectional and prospective associations between dietary and plasma vitamin C, heel bone ultrasound, and fracture risk in men and women in the European Prospective Investigation into Cancer in Norfolk cohort. Am. J. Clin. Nutr. 2015, 102, 1416–1424. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.E.; Cho, S.H.; Park, H.M.; Chang, Y.K. Relationship between bone mineral density and dietary intake of β-carotene, vitamin C, zinc and vegetables in postmenopausal Korean women: A cross-sectional study. J. Int. Med. Res. 2016, 44, 1103–1114. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, M.; Nakamura, M.; Ogawa, K.; Ikoma, Y.; Ando, F.; Shimokata, H.; Yano, M. Dietary patterns of antioxidant vitamin and carotenoid intake associated with bone mineral density: Findings from post-menopausal Japanese female subjects. Osteoporos. Int. 2011, 22, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Lee, J.S.; Johnson, M.A. Poor Socioeconomic and Nutritional Status Are Associated with Osteoporosis in Korean Postmenopausal Women: Data from the Fourth Korea National Health and Nutrition Examination Survey (KNHANES) 2009. J. Am. Coll. Nutr. 2015, 34, 400–407. [Google Scholar] [CrossRef]
- Pasco, J.A.; Henry, M.J.; Wilkinson, L.K.; Nicholson, G.C.; Schneider, H.G.; Kotowicz, M.A. Antioxidant vitamin supplements and markers of bone turnover in a community sample of nonsmoking women. J. Womens Health 2006, 15, 295–300. [Google Scholar] [CrossRef]
- Karamati, M.; Yousefian-Sanni, M.; Shariati-Bafghi, S.E.; Rashidkhani, B. Major nutrient patterns and bone mineral density among postmenopausal Iranian women. Calcif. Tissue Int. 2014, 94, 648–658. [Google Scholar] [CrossRef]
- Ilich, J.Z.; Brownbill, R.A.; Tamborini, L. Bone and nutrition in elderly women: Protein, energy, and calcium as main determinants of bone mineral density. Eur. J. Clin. Nutr. 2003, 57, 554–565. [Google Scholar] [CrossRef]
- Rivas, A.; Romero, A.; Mariscal-Arcas, M.; Monteagudo, C.; López, G.; Lorenzo, M.L.; Ocaña-Peinado, F.M.; Olea-Serrano, F. Association between dietary antioxidant quality score (DAQs) and bone mineral density in Spanish women. Nutr. Hosp. 2012, 27, 1886–1893. [Google Scholar] [CrossRef]
- De França, N.A.; Camargo, M.B.; Lazaretti-Castro, M.; Martini, L.A. Antioxidant intake and bone status in a cross-sectional study of Brazilian women with osteoporosis. Nutr. Health 2013, 22, 133–142. [Google Scholar] [CrossRef]
- Liu, Z.M.; Leung, J.; Wong, S.Y.; Wong, C.K.; Chan, R.; Woo, J. Greater fruit intake was associated with better bone mineral status among Chinese elderly men and women: Results of Hong Kong Mr. Os and Ms. Os studies. J. Am. Med. Dir. Assoc. 2015, 16, 309–315. [Google Scholar] [CrossRef]
- Morton, D.J.; Barrett-Connor, E.L.; Schneider, D.L. Vitamin C supplement use and bone mineral density in postmenopausal women. J. Bone Miner. Res. 2001, 16, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Wolf, R.L.; Cauley, J.A.; Pettinger, M.; Jackson, R.; Lacroix, A.; Leboff, M.S.; Lewis, C.E.; Nevitt, M.C.; Simon, J.A.; Stone, K.L.; et al. Lack of a relation between vitamin and mineral antioxidants and bone mineral density: Results from the Women’s Health Initiative. Am. J. Clin. Nutr. 2005, 82, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Melaku, Y.A.; Gill, T.K.; Taylor, A.W.; Adams, R.; Shi, Z. Association between nutrient patterns and bone mineral density among ageing adults. Clin. Nutr. ESPEN 2017, 22, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.A.; Hudes, E.S. Relation of ascorbic acid to bone mineral density and self-reported fractures among US adults. Am. J. Epidemiol. 2001, 154, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Casale, M.; von Hurst, P.R.; Beck, K.L.; Shultz, S.; Kruger, M.C.; O’Brien, W.; Conlon, C.A.; Kruger, R. Lean Mass and Body Fat Percentage Are Contradictory Predictors of Bone Mineral Density in Pre-Menopausal Pacific Island Women. Nutrients 2016, 8, 470. [Google Scholar] [CrossRef]
- Prynne, C.J.; Mishra, G.D.; O’Connell, M.A.; Muniz, G.; Laskey, M.A.; Yan, L.; Prentice, A.; Ginty, F. Fruit and vegetable intakes and bone mineral status: A cross sectional study in 5 age and sex cohorts. Am. J. Clin. Nutr. 2006, 83, 1420–1428. [Google Scholar] [CrossRef]
- Maïmoun, L.; Simar, D.; Caillaud, C.; Peruchon, E.; Sultan, C.; Rossi, M.; Mariano-Goulart, D. Effect of antioxidants and exercise on bone metabolism. J. Sports Sci. 2008, 26, 251–258. [Google Scholar] [CrossRef]
- Houston, Z.L. Development and Validation of a Semi-Quantitative Food Frequency Questionnaire to Assess. Dietary Intake of Adult Women Living in New Zealand; Massey University: Albany, NY, USA, 2014. [Google Scholar]
- Sugiura, M.; Nakamura, M.; Ogawa, K.; Ikoma, Y.; Yano, M. High Vitamin C Intake with High Serum β-Cryptoxanthin Associated with Lower Risk for Osteoporosis in Post-Menopausal Japanese Female Subjects: Mikkabi Cohort Study. J. Nutr. Sci. Vitaminol. 2016, 62, 185–191. [Google Scholar] [CrossRef]
- Martínez-Ramírez, M.J.; Palma Pérez, S.; Delgado-Martínez, A.D.; Martínez-González, M.A.; De la Fuente Arrillaga, C.; Delgado-Rodríguez, M. Vitamin C, vitamin B12, folate and the risk of osteoporotic fractures. A case-control study. Int. J. Vitam. Nutr. Res. 2007, 77, 359–368. [Google Scholar] [CrossRef]
- Sahni, S.; Hannan, M.T.; Gagnon, D.; Blumberg, J.; Cupples, L.A.; Kiel, D.P.; Tucker, K.L. High vitamin C intake is associated with lower 4-year bone loss in elderly men. J. Nutr. 2008, 138, 1931–1938. [Google Scholar] [CrossRef]
- Masse, P.G.; Jougleux, J.L.; CTranchant, C.; Dosy, J.; Caissie, M.; PCoburn, S. Enhancement of calcium/vitamin d supplement efficacy by administering concomitantly three key nutrients essential to bone collagen matrix for the treatment of osteopenia in middle-aged women: A one-year follow-up. J. Clin. Biochem. Nutr. 2010, 46, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Sahni, S.; Hannan, M.T.; Gagnon, D.; Blumberg, J.; Cupples, L.A.; Kiel, D.P.; Tucker, K.L. Protective effect of total and supplemental vitamin C intake on the risk of hip fracture—A 17-year follow-up from the Framingham Osteoporosis Study. Osteoporos. Int. 2009, 20, 1853–1861. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, H.M.; New, S.A.; Golden, M.H.; Campbell, M.K.; Reid, D.M. Nutritional associations with bone loss during the menopausal transition: Evidence of a beneficial effect of calcium, alcohol, and fruit and vegetable nutrients and of a detrimental effect of fatty acids. Am. J. Clin. Nutr. 2004, 79, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Park, H.M.; Heo, J.; Park, Y. Calcium from plant sources is beneficial to lowering the risk of osteoporosis in postmenopausal Korean women. Nutr. Res. 2011, 31, 27–32. [Google Scholar] [CrossRef]
- Lumbers, M.; New, S.A.; Gibson, S.; Murphy, M.C. Nutritional status in elderly female hip fracture patients: Comparison with an age-matched home living group attending day centres. Br. J. Nutr. 2001, 85, 733–740. [Google Scholar] [CrossRef]
- New, S.A.; Robins, S.P.; Campbell, M.K.; Martin, J.C.; Garton, M.J.; Bolton-Smith, C.; Grubb, D.A.; Lee, S.J.; Reid, D.M. Dietary influences on bone mass and bone metabolism: Further evidence of a positive link between fruit and vegetable consumption and bone health? Am. J. Clin. Nutr. 2000, 71, 142–151. [Google Scholar] [CrossRef]
- Murata, A. Smoking and vitamin C. World Rev. Nutr. Diet. 1991, 64, 31–57. [Google Scholar] [CrossRef]
- Kuo, S.M.; Stout, A.; Wactawski-Wende, J.; Leppert, P.C. Ascorbic acid status in postmenopausal women with hormone replacement therapy. Maturitas 2002, 41, 45–50. [Google Scholar] [CrossRef]
- Sun, L.L.; Li, B.L.; Xie, H.L.; Fan, F.; Yu, W.Z.; Wu, B.H.; Xue, W.Q.; Chen, Y.M. Associations between the dietary intake of antioxidant nutrients and the risk of hip fracture in elderly Chinese: A case-control study. Br. J. Nutr. 2014, 112, 1706–1714. [Google Scholar] [CrossRef]
- Maggio, D.; Barabani, M.; Pierandrei, M.; Polidori, M.C.; Catani, M.; Mecocci, P.; Senin, U.; Pacifici, R.; Cherubini, A. Marked decrease in plasma antioxidants in aged osteoporotic women: Results of a cross-sectional study. J. Clin. Endocrinol. Metab. 2003, 88, 1523–1527. [Google Scholar] [CrossRef]
- Bjarnason, N.H.; Riis, B.J.; Christiansen, C. The effect of fluvastatin on parameters of bone remodeling. Osteoporos. Int. 2001, 12, 380–384. [Google Scholar] [CrossRef] [PubMed]
- Kaptoge, S.; Welch, A.; McTaggart, A.; Mulligan, A.; Dalzell, N.; Day, N.E.; Bingham, S.; Khaw, K.T.; Reeve, J. Effects of dietary nutrients and food groups on bone loss from the proximal femur in men and women in the 7th and 8th decades of age. Osteoporos. Int. 2003, 14, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Ramos, M.; Vargas, L.A.; Fortoul Van der Goes, T.I.; Cervantes-Sandoval, A.; Mendoza-Nunez, V.M. Supplementation of ascorbic acid and alpha-tocopherol is useful to preventing bone loss linked to oxidative stress in elderly. J. Nutr. Health Aging 2010, 14, 467–472. [Google Scholar] [CrossRef]
- Stunes, A.K.; Syversen, U.; Berntsen, S.; Paulsen, G.; Stea, T.H.; Hetlelid, K.J.; Lohne-Seiler, H.; Mosti, M.P.; Bjørnsen, T.; Raastad, T.; et al. High doses of vitamin C plus E reduce strength training-induced improvements in areal bone mineral density in elderly men. Eur. J. Appl. Physiol. 2017, 117, 1073–1084. [Google Scholar] [CrossRef] [PubMed]
- Mainini, G.; Rotondi, M.; Di Nola, K.; Pezzella, M.T.; Iervolino, S.A.; Seguino, E.; D’Eufemia, D.; Iannicelli, I.; Torella, M. Oral supplementation with antioxidant agents containing alpha lipoic acid: Effects on postmenopausal bone mass. Clin. Exp. Obstet. Gynecol. 2012, 39, 489–493. [Google Scholar] [PubMed]
- Chuin, A.; Labonté, M.; Tessier, D.; Khalil, A.; Bobeuf, F.; Doyon, C.Y.; Rieth, N.; Dionne, I.J. Effect of antioxidants combined to resistance training on BMD in elderly women: A pilot study. Osteoporos. Int. 2009, 20, 1253–1258. [Google Scholar] [CrossRef]
- Kiel, D.P.; Mercier, C.A.; Dawson-Hughes, B.; Cali, C.; Hannan, M.T.; Anderson, J.J. The effects of analytic software and scan analysis technique on the comparison of dual X-ray absorptiometry with dual photon absorptiometry of the hip in the elderly. J. Bone Miner. Res. 1995, 10, 1130–1136. [Google Scholar] [CrossRef]
- Chavan, S.N.; More, U.; Mulgund, S.; Saxena, V.; Sontakke, A.N. Effect of supplementation of vitamin C and E on oxidative stress in osteoporosis. Indian J. Clin. Biochem. 2007, 22, 101–105. [Google Scholar] [CrossRef]
| Study | Study Population | Dietary Assessment | Outcome Measure(s) and Lab Analyses | Results |
|---|---|---|---|---|
| New SA et al., 2000 [78] UK | 62 healthy women pre- and postmenopausal (range 45–54 years old) | FFQ | LS and FN BMD Quantitative CT at the ultradistal radial total, trabecular, and cortical sites Urine concentration of pyridinoline and deoxypyridinoline Serum level of osteocalcin | Daily VC intake: 103.4 ± 65.6 mg/d (23.8–453.2). Mean deoxypyridinoline excretion was significantly lower with higher intakes of VC (p < 0.02). No correlation with BMD parameters in any localisation. |
| Simon JA et al., 2001 [65] USA | 3778 premenopausal women (34 ± 8 years old) 3165 postmenopausal women (62 ± 13 years old) 6137 men (44 ± 16 years old) 20–90 years old | 24-h dietary recall | FN BMD Serum level of VC and VE Self-reported fractures | In a premenopausal women group, increasing levels of dietary VC were independently associated with BMD and did not correlate with prevalence of self-reported fractures. In a postmenopausal women group, neither VC intake nor VC serum concentration was associated with BMD and or prevalence of self-reported fractures. Among postmenopausal women who had no history of smoking or oestrogen use, serum ascorbic acid levels were inversely associated with BMD. In smoking postmenopausal women with history of oestrogen use, higher VC supplementation was associated with a lower prevalence of fractures. |
| Ilich JZ et al., 2003 [58] USA | 136 postmenopausal (at least 5 years) women, generally health, 68.7 ± 7.1 years old | 3-day dietary record (2 weeks and 1 weekend day); Ca-FFQ | TB, LS, FN, forearm and hand BMD Serum levels of PTH and vitamin D | A weak but statistically significant correlation between VC dietary intake and bone mass was observed. VC intake was a significant element of stepwise regression models explaining loss of Ward’s triangle, trochanter, shaft and total femur BMD. |
| Wolf RL et al., 2005 [63] USA | 11,068 postmenopausal women aged 50–79 years old | FFQ 122 food items supplements were estimated with an interviewer-administered questionnaire | TB, LS, TH BMD Serum concentrations of retinol, carotenoids, and tocopherols measured (N = 379) | The beneficial effect of current hormone therapy use on FN TB, LS and TH BMD was greater in women with higher VC level. An independent positive influence of VC intake on BMD was not observed. |
| Prynne CJ et al., 2006 [67] UK | Three groups: 101 girls and 111 boys 16–18 years old 90 women 23–37 years old and 67 women and 67 men 60–83 years old | 7-day food diaries | TB, LS, TH, FN and greater trochanter BMD; bone area and bone mineral content | In the group of boys, significant positive associations were found between dietary VC and BMD. A negative association between hip BMD and VC intake was found in the group of older women. |
| Pasco JA et al., 2006 [56] Australia | 533 postmenopausal, non-smoking women 48–89 year old (mean 68.3) 26 supplemented vitamin C and 22 vitamin C and/or E | Ca-FFQ Self-reported supplement and medication use, diet, and lifestyle questionnaire | TB BMD Serum bone resorption markers: C-telopeptide and bone-specific AP | Lower concentration of C-telopeptide was observed in group with increased exposure to VC and/or VE. |
| Sugiura M et al., 2011 [54] Japan | 293 postmenopausal women | FFQ 97 food items | BMD of nondominant forearm | High intake of VC (170–625 mg/day) with β-cryptoxanthin was inversely associated with low radial BMD and may be beneficial to bone health. |
| Rivas A et al., 2012 [59] Spain | 280 women divided into 3 groups: ≤35, 35–45 and ≥45 years old | 24-h diet recall | BMD of calcaneus | BMD was higher in the participants defined as high antioxidant consumers in all age groups. |
| De França NA et al., 2013 [60] Brazil | 150 postmenopausal women, 68.7 ± 9.1 year old (range 48–87 year old) | 3-day food diary, non-consecutive days | TB, LS, FN and TF BMD | No relationship between the BMD and daily antioxidant intake was found. |
| Karamati M et al., 2014 [57] Iran | 151 postmenopausal women aged 60.3 years old | FFQ 168 food item | LS and FN BMD | Mean BMD of the lumbar spine of women in the highest tertile of the first pattern contain folate, total fiber, potassium, vitamin A, C, K, B6, b-carotene, magnesium, copper, and manganese scores was significantly higher than those in the lowest tertile (mean difference 0.08; 95% confidence interval 0.02–0.15; p = 0.01). No correlation between separate nutrients or nutrient patterns and BMD was found. |
| Kim YA et al., 2015 [51] South Korea | 1196 postmenopausal female | 24-h dietary recall | LS, FN and TH BMD | Dietary vitamin C intake tertile was significantly positively associated with BMD at all sites (R = 0.513 for LS and R = 0.657 for FN; p < 0.05 for each). The multiple-adjusted odds ratio for osteoporosis for dietary VC <100 mg/day was 1.790 (95% CI 1.333–2.405; p < 0.001). However, a significant association between VC intake and BMD was only observed in subjects with vitamin D deficiency and aged 50–59 years or >70 years. |
| Finck H et al., 2015 [52] UK | 4510 participants (women: 2616, men 1898), the mean age 60 ± 10 years old | 7-day diet diary | History of bone fractures VC plasma concentration | A linear inverse relationship between quintiles of plasma VC and the prevalence of hip fractures (HR: 0.82; P-trend = 0.016) in men. |
| Liu ZM et al., 2015 [61] China | 2000 men and 2000 women aged 65 years and older 72.5 ± 5.2 years old | FFQ 266 food items | BMD, bone mineral content, and bone area at TB, TH, LS and FN | TB and FN BMD were significantly positively associated with fruit intake in both men and women. Adjustment for VC intake, but not dietary acid load, attenuated the association between fruit intake and bone mass. |
| Kim MH et al., 2015 [55] South Korea | 1467 postmenopausal women Age 65.2 ± 0.3 years old | 24-h recall | TF, FN and LS BMD | Participants consuming less VC than the estimated average requirement showed higher odds (OR = 1.49; 95% Cl 1.10–2.03; p < 0.05) of having osteoporosis than their counterparts. |
| Kim MH et al., 2016 [50] South Korea | Osteoporosis (N = 244 men and 968 female) Healthy control (N = 1382 men and 453 female) 50 years old and over | 24-h recall | BMD (no specific location) | Higher VC intake levels were associated with a lower risk of osteoporosis; the result was statistically significant (OR = 0.67; 95% CI: 0.47–0.97; p = 0.0371) only in the highest intake quartile group. No association was seen in the group with high physical activity. Significantly lower VC intake (81 ± 2.1 vs. 113.1 ± 2.0 mg/d; p < 0.05) was noted in the osteoporosis patients group. |
| Kim DE et al., 2016 [53] South Korea | 189 postmenopausal women aged 60.63 ± 6.39 years old | FFQ 103 food items | LS, FN and TH BMD | T-score of the LS, FN and TH was positively correlated with intake of VC (r2 = 0.157, p = 0.048; r2 = 0.324, p < 0.001; r2 = 0.182, p = 0.003, respectively). |
| Casale M et al., 2016 [66] New Zealand | 175 post-menarcheal and pre-menopausal women 16–45 years old | FFQ 220 food items | TB BMD | There was no relationship between VC intake and total body BMD. |
| Melaku YA et al., 2017 [64] Australia | 1135 participants (N = 520 men, 615 female) 50 year old and over | FFQ 167 food items | BMD (no specific location) | Three nutritional patterns: mixed (potassium, calcium, fibre, retinol and vitamin B12); animal-sourced (cholesterol, protein, vitamin B12 and fat) and plant-sourced (fibre, carotene, VC and lutein). Whereas animal- and plant-sourced nutrient patterns were not associated with BMD, mixed source pattern may have had a beneficial effect on BMD reduction prevention. |
| Study | Study Population | Dietary Assessment | Outcome Measure(s) and Lab Analyses | Results |
|---|---|---|---|---|
| Lumbers M. et al., 2001 [77] UK | 75 women with femur neck fracture 80.5 ± 11.9 years old (range 61–103) 50 age-matched independent-living group of females attending one of three local day centres 79.8 ± 7.5 years old (range 63–95) | 24-h dietary recall | Serum VC level | Plasma VC in fracture patients was significantly higher (7.52 ± 3.77 mg/L) than in control group (3.66 ± 2.50 mg/L p < 0.001). Daily intake of VC in diet was higher in group with hip fracture (60.7 ± 33.2 mg/d) than in control group (55.2 ± 38.8 mg/d) but it was not statistically significant. |
| Maggio D et al., 2003 [82] Italy | 75 osteoporotic (T-score ≤ −3.5) 75 controls (T-Score ≥ −1) postmenopausal women Age over 60 years old | Mini Nutritional Assessment questionnaire | FN BMD Plasma VC level | FN BMD showed a positively statistically significant correlation with plasma VC (r = 0.26, p = 0.05). Plasma VC was significantly lower in osteoporotic group than in control (30 ± 3.7 vs. 55.5 ± 13.1 µmol/L; p < 0.001). |
| Martínez-Ramírez MJ et al., 2007 [71] Spain | Research group: N = 167, aged ≥ 65, osteoporotic fracture in 6–24 months prior to inclusion in the study Control group: N = 167 | FFQ 136 food items | Plasma VC level | Daily dietary VC intake was higher in group with hip fracture (283 ± 12.8 mg/d) than in control group (263 ± 9.9 mg/d), but it was not statistically significant. Plasma VC in bone fracture patients was significantly lower (3.1 ± 0.3 mg/L) than in the control group (4.1 ± 0.3 mg/L; p = 0.012). For both dietary intake and serum levels, the lowest quartile of VC was associated with the highest risk of fracture. VC serum levels presented a linear trend (p = 0.03) with a significantly reduced fracture risk for the upper quartile (OR = 0.31; 95% CI 0.11–0.87) compared with the lowest quartile. |
| Park HM et al., 2011 [76] South Korea | 72 osteoporotic women 72 controls (range 50–70 years old) | FFQ 117 food items | LS, FN and TF BMD | A significant reduction in the risk of osteoporosis for third quartile (136.9–176.3 mg/d) versus the lowest daily dietary VC intake (≤91.5 mg/d) was found. The correlation between VC intake and femoral neck BMD was presented (r = 0.190; p < 0.05) |
| Sun et al., 2014 [81] USA | 726 elderly with hip fracture and 726 control subjects | FFQ 79 food items | None | The OR of hip fracture for the highest (>167 mg/d for men and >171 mg/d for women) vs. the lowest (<55 mg/d for men and <49 mg/d for women) quartile of VC intake was 0.39 (95% CI 0.28, 0.56). Daily dietary VC intake was significantly lower in both men and women with osteoporotic fracture (M: 77 ± 40 mg/d; F: 82 ± 47 mg/d)) compared to healthy control subjects (M: 102 ± 60 mg/d p < 0.001; F: 106 ± 57 mg/d p < 0.001). |
| Study | Study Group | Follow-Up in Years | Dietary Assessment | Outcome Measure(s) and Lab Analyses | Results |
|---|---|---|---|---|---|
| Kaptoge S et al., 2003 [84] UK | 470 women and 474 men mean age 72 (range 67–79 years old) | 2–5 | 7-day food diaries | TH BMD performed twice an average of 3 years apart | Women in the lowest tertile of dietary VC intake (7–57 mg/d) lost BMD at a faster rate compared to those in the middle (58–98 mg/d) (p = 0.015) and upper (p = 0.010) tertiles (99–363 mg/d). No relationship between VC intake and BMD loss was observed in men. |
| Macdonald HM et al., 2004 [75] UK | 891 women aged 45–55 years old at baseline | 5–7 | FFQ 98 food items | LS and FN BMD at baseline and the end of observation | In premenopausal women, calcium intake and dietary fruit and vegetable intake were associated with FN BMD. |
| Sahni S et al., 2008 [72] USA | 334 men and 540 women mean age 75 years old | 4 | FFQ 126 food items | BMD of right femoral neck and trochanter, lumbar spine, radial shaft at baseline used dual-photon absorptiometer; in follow-up used dual X-ray absorptiometry | No significant effects of VC intake on BMD in women was observed. LS and T BMD loss was significantly lower in smoking men with higher dietary VC intake. FN and T BMD loss was significantly lower in men with higher total VC intake, lower calcium and total VE intake. |
| Sahni S et al., 2009 [74] USA | 366 men and 592 women, mean age 75 ± 5 years old In the 17 years of follow-up, 100 hip fractures and in the 15 years of follow up 180 non-vertebral osteoporotic fracture were reported among 976 participants | 15–17 | FFQ 126 food items | Clinical report of non-vertebral osteoporotic fractures | The group characterized by the highest tertile of total VC intake had a significantly lower risk of hip and non-vertebral osteoporotic fractures compared to subjects in the lowest tertile. Subjects in the highest category of supplemental VC intake had significantly lower risk of hip and non-vertebral osteoporotic fracture compared to non-supplementing study participants. A protective trend was observed for dietary VC and risk of hip fracture as well as non-vertebral osteoporotic fractures. |
| Sugiura M et al., 2016 [70] Japan | 187 post-menopausal female Mean age round 60 * | 4 | FFQ 97 food items | Non-dominant forearm BMD measured at baseline and follow up. Concentration of 6 serum carotenoids | High VC intake with high serum concentration of β-cryptoxanthin was inversely associated with osteoporosis development risk. |
| Study | Study Group | Follow-Up | Intervention | Outcome Measure(s) and Lab Analyses | Results |
|---|---|---|---|---|---|
| Bjarnason NH et al., 2001 [83] Denmark | 68 postmenopausal women 65 years old and above; hip and/or spine BMD < −2.0 SD; serum cholesterol < 5.2 mmol/L | 12 weeks | 2 groups: 1. VC 500 mg/d; N = 23 2. 40 mg Fluvastatin + VC 500 mg/d; N = 45 | Biochemical bone marker measurements (AP, OC, U-CTX, S-CTX) at visits –2, 0, 4 and 12 weeks. | No effect of the treatments on the markers of bone formation. |
| Chavan SN et al., 2007 [90] India | 75 osteoporosis patients age 45–70 years and 50 healthy controls age 20–50 years (did not undergo the intervention) * | 13 weeks | 3 groups: 1. VE 400 mg/d; N = 25 2. VC 500 mg/d; N = 25 3. VE 400 mg/d + VC 500 mg/d; N = 25 | Serum concentrations of malondialdehyde, glutathione peroxidase, glutathione reductase, superoxide dismutase, Ca+, Pi measured at baseline, 45th day and at the end of the study. | A significant fall in the concentration of serum malondialdehyde (p < 0.001), TrACP (p < 0.01) in all supplemented groups. Antioxidant status was reflected by significant rise in concentration of serum superoxide dismutase (p < 0.001) and erythrocyte GSH (p < 0.001) after 90 days of antioxidant supplementation in osteoporosic patients. |
| Maïmoun L et al., 2008 [68] France | 9 postmenopausal women and 4 men 69–79 years old | 8 weeks | VC 500 mg/d + vitamin E 100 mg/d + aerobic training programme | Serum concentration of ionized calcium, PTH, 25-hydroxyvitamin D, 1,25-dihydroxyvitamin D, osteocalcin, bone AP, urinary type I collagen, C-telopeptide, IGF-1 and IGFBP-3 | After 8 weeks vitamin d level and bone alkaline phosphatase was statistically significantly higher, rest serum markers stayed constant. Intervention combining VC and VE supplementation combined with aerobic training might improve the calciotropic hormone profile which is altered in the elderly and associated with bone loss. |
| Chuin A et al., 2009 [88] Canada | 34 postmenopausal women 61–73 years old | 6 months | 3-day dietary record Four groups: 1. Placebo; N = 7 2. Antioxidant (VE 600 mg/d + VC 1000 mg/d); N = 8 3. Exercise (60-min exercise sessions 3 x/week) + placebo; N = 11 4. Exercise + antioxidants; N = 8 | BMD of FN and LS at baseline and at the end of the study. | The placebo group displayed a significant loss in LS BMD (p < 0.05) over 6 months; no changes observed in the other groups. |
| Ruiz-Ramos M et al., 2010 [85] México | 90 elderly participants Group 1: 67.6 ± 7.3 years old; 66% female Group 2: 68.2 ± 7.3 years old; 87% female Group 3: 68.8 ± 8.5 years old; 68% female | 1 year (follow-up 0, 3, 6, 9, 12 months) | Divided into 3 groups: 1. Placebo; N = 30 2. VC 500 mg/d + alpha-tocopherol 400 IU/d; N = 30 3. VC 1000 mg/d + alpha-tocopherol 400 IU/d N = 30 | BMD of hip and spine at baseline and at the end of the study; Serum measurements of thiobarbituric acid reactive substances, total antioxidant status, superoxide dismutase, and glutation peroxidase | Differences observed in the hip BMD between the treatment groups and placebo, indicating a possible beneficial effect of antioxidants as a coadjuvant in the prevention and treatment of osteoporosis. |
| Masse PG et al., 2010 [73] Canada | 60 women with osteopenia 35–55 years old, non-oestrogen users | 1 year | 3 non-consecutive days food recall; 3 groups: 1. Placebo, normal BMD; N = 20 2. Osteopenic, calcium 1000 mg/d + vitamin D3 250 IU/d; N = 20 3. Osteopenic, calcium 1000 mg/d + vitamin D3 250 IU/d + VC 500 mg/d + vitamin B6 75 mg/d + prolin 500 mg/d; N = 20 | BMD of LS and femoral sites at baseline and at the end of the study. Blood sample: intact PTH, estradiol, calcium and inorganic phosphorus, OC, bone AP, vitamin D3 and B6. Urinary free deoxypyridinoline, type I collagen helical peptide, creatinine, vitamin B6 metabolites. | Osteopenic patients treated with the conventional calcium/vitamin D supplement continued to lose bone minerals to a much greater extent than the normal BMD control participants. The combination of calcium/vitamin D with collagen-related nutrients deterred further bone loss at all bone sites. Markers of bone turnover decreased significantly in both osteopenic groups. Although biomarkers of resorption did not change, PTH and 1,25(OH)2D3-induced osteoclastic activity were significantly reduced. |
| Mainini G et al., 2012 [87] Italy | 44 postmenopausal women with osteopenia (49–75 years old) | 1 year | 2 groups: 1. alpha lipoic acid 300 mg + VC 30 mg + VE 5 mg + selenium 2.75 mg + calcium 500 mg + vitamin D3 400 IU twice a day; N = 23 2. calcium 500 mg + vitamin D3 400 IU twice a day; N = 21 | BMD of non-dominant foot at baseline and after 1-year follow-up; heel quantitative ultrasonometry | The treatment with alpha lipoic acid led to a significantly higher estimated BMD compared to the control group (p = 0.048). |
| Stunes AK et al., 2017 [86] Norway | 35 men age 68 ± 6 years old | 12 weeks | 4-weekday dietary registration 2 groups: 1. Placebo + strength training program n = 18 2. VC 1000 mg/d, VE 235 mg/d + strength training program | BMD of the LS, TH, FN, TB at baseline and after 12 weeks. Blood concentration of vitamins C, E, D; CTX-1. P1NP, osteoprotegerin, intact OC, PTH, insulin, sclerostin, DKK1, TNF-α, leptin, adiponectin, resistin, RANKL | High doses of VC + VE supplementation blunted some of the positive skeletal effects from strength training. Twelve weeks of resistance training intervention increased the LS and TH BMD. In the control group, physical activity increased TH and LS BMD, insulin-like growth factor and leptin concentrations. This effect was not present in the antioxidant supplementation group. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brzezińska, O.; Łukasik, Z.; Makowska, J.; Walczak, K. Role of Vitamin C in Osteoporosis Development and Treatment—A Literature Review. Nutrients 2020, 12, 2394. https://doi.org/10.3390/nu12082394
Brzezińska O, Łukasik Z, Makowska J, Walczak K. Role of Vitamin C in Osteoporosis Development and Treatment—A Literature Review. Nutrients. 2020; 12(8):2394. https://doi.org/10.3390/nu12082394
Chicago/Turabian StyleBrzezińska, Olga, Zuzanna Łukasik, Joanna Makowska, and Konrad Walczak. 2020. "Role of Vitamin C in Osteoporosis Development and Treatment—A Literature Review" Nutrients 12, no. 8: 2394. https://doi.org/10.3390/nu12082394
APA StyleBrzezińska, O., Łukasik, Z., Makowska, J., & Walczak, K. (2020). Role of Vitamin C in Osteoporosis Development and Treatment—A Literature Review. Nutrients, 12(8), 2394. https://doi.org/10.3390/nu12082394





