The Use of Curcumin as a Complementary Therapy in Ulcerative Colitis: A Systematic Review of Randomized Controlled Clinical Trials
Abstract
1. Introduction
2. Methods
2.1. Protocol and Registration
2.2. Information Sources and Search Strategies
- Population: individuals with IBD (UC or CD) of either sex and from any age group;
- Intervention: curcumin supplementation in the form of spice, capsule, or enema;
- Comparison: placebo or conventional drug therapy;
- Outcomes: disease activity, clinical, or endoscopic inflammatory activity;
- Study design: randomized clinical trials (RCTs).
2.3. Eligibility Criteria
2.4. Study Selection and Data Collection Process
2.5. Risk of Bias Assessment
3. Results
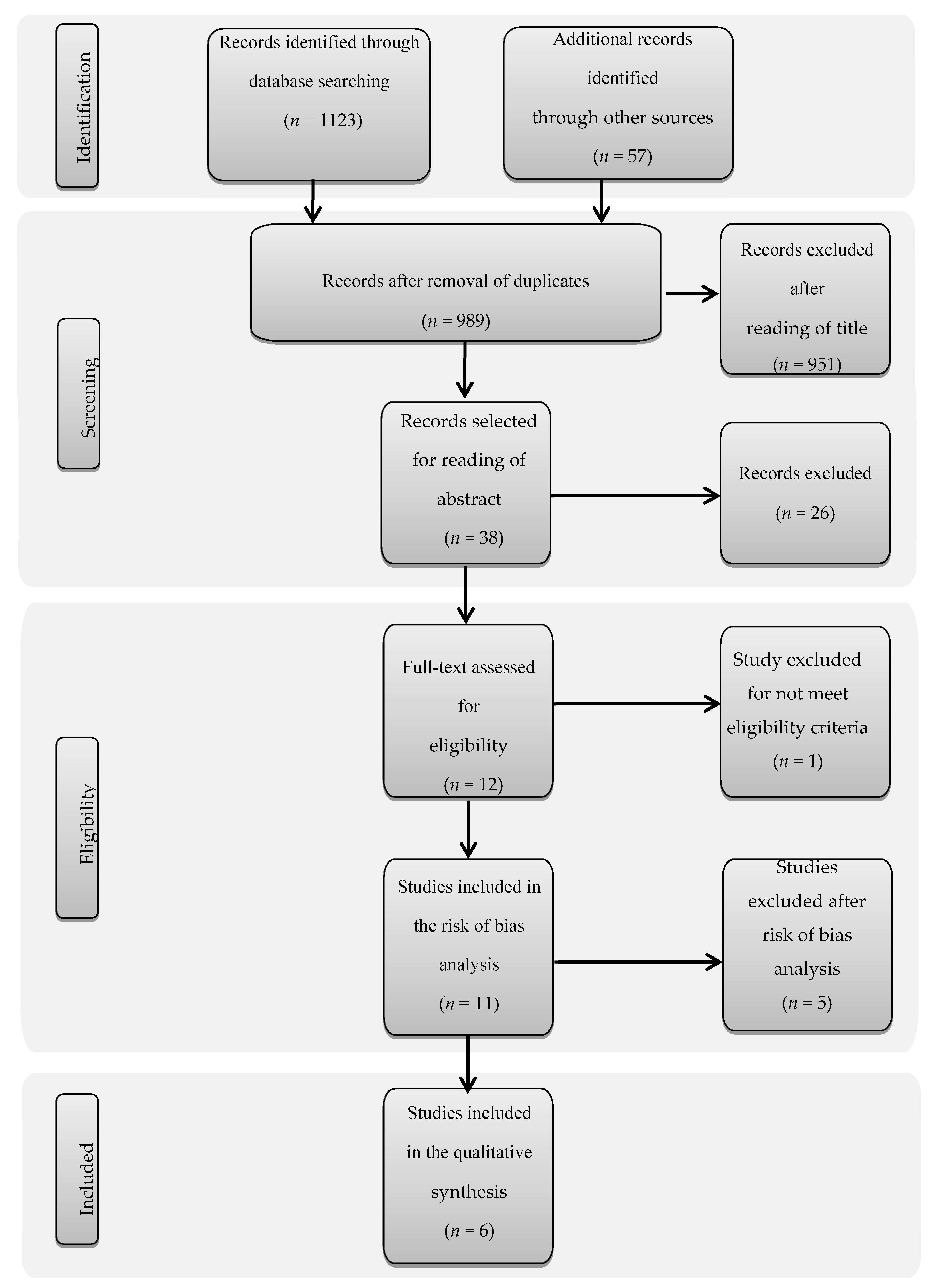
3.1. Search Results
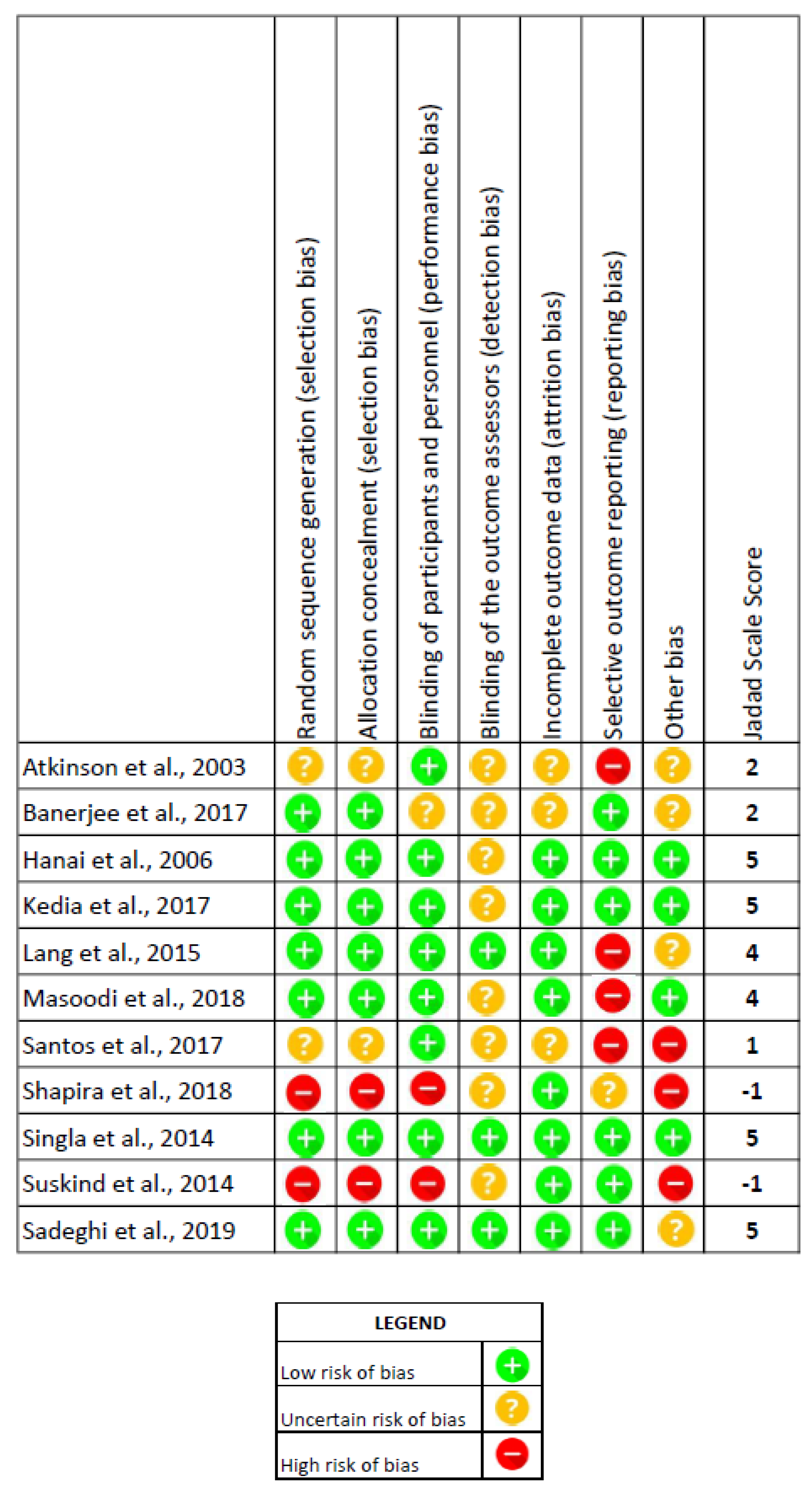
3.2. Assessment of the Risk of Bias and Excluded RCTs
3.3. Characteristics of Selected Articles
3.4. Outcomes after Intervention
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Magro, F.; Gionchetti, P.; Eliakim, R.; Ardizzone, S.; Armuzzi, A.; Barreiro-de Acosta, M.; Burisch, J.; Gecse, K.B.; Hart, A.L.; Hindryckx, P.; et al. Third European evidence-based consensus on diagnosis and management of ulcerative colitis. Part 1: Definitions, diagnosis, extra-intestinal manifestations, pregnancy, cancer surveillance, surgery, and ileo-anal pouch disorders. J. Crohn’s Colitis 2017, 11, 649–670. [Google Scholar] [CrossRef] [PubMed]
- Stapley, S.A.; Rubin, G.P.; Alsina, D.; Shephard, E.A.; Rutter, M.D.; Hamilton, W.T. Clinical features of bowel disease in patients aged <50 years in primary care: A large case-control study. Br. J. Gen. Pract. 2017, 67, e336–e344. [Google Scholar] [CrossRef] [PubMed]
- Ilhara, S.; Hirata, Y.; Koike, K. TGF-β in inflammatory bowel disease: A key regulator of immune cells, epithelium and the intestinal microbiota. J. Gastroenterol. 2017, 52, 777–787. [Google Scholar] [CrossRef]
- D’Haens, G.R.; Sartor, R.B.; Silverberg, M.S.; Petersson, J.; Rutgeerts, P. Future directions in inflammatory bowel disease management. J. Crohn’s Colitis 2014, 8, 726–734. [Google Scholar] [CrossRef] [PubMed]
- Kawalec, P.; Malinowski, K.P. Indirect health costs in ulcerative colitis and Crohn’s disease: A systematic review and meta-analysis. Expert Rev. Pharm. Outcomes Res. 2015, 15, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Harbord, M.; Eliakim, R.; Bettenworth, D.; Karmiris, K.; Katsanos, K.; Kopylov, U.; Kucharzik, T.; Molnár, T.; Raine, T.; Sebastian, S.; et al. Third European evidence-based consensus on diagnosis and management of ulcerative colitis. Part 2: Current management. J. Crohn’s Colitis 2017, 11, 769–784. [Google Scholar] [CrossRef] [PubMed]
- Ben-Horin, S.; Mao, R.; Chen, M. Optimizing biologic treatment in IBD: Objective measures, but when, how and how often? BMC Gastroenterol. 2015, 15, 178. [Google Scholar] [CrossRef] [PubMed]
- Munkholm, P.; Langholz, E.; Davidsen, M.; Binder, V. Frequency of glucocorticoid resistance and dependency in Crohn’s disease. Gut 1994, 35, 360–362. [Google Scholar] [CrossRef]
- Faubion, W.A., Jr.; Loftus, E.V., Jr.; Harmsen, W.S.; Zinsmeister, A.R.; Sandborn, W.J. The natural history of corticosteroid therapy for inflammatory bowel disease: A population-based study. Gastroenterology 2001, 121, 255–260. [Google Scholar] [CrossRef]
- Ho, G.T.; Chiam, P.; Drummond, H.; Loane, J.; Arnott, I.D.; Satsangi, J. The efficacy of corticosteroid therapy in inflammatory bowel disease: Analysis of a 5-year UK inception cohort. Aliment. Pharmacol. Ther. 2006, 24, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, F.; Zhang, H.J.; Sheng, J.Q.; Yan, W.F.; Ma, M.X.; Fan, R.Y.; Gu, F.; Li, C.F.; Chen, D.F.; et al. Corticosteroid therapy in ulcerative colitis: Clinical response and predictors. World J. Gastroenterol. 2015, 21, 3005–3015. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Beevers, S.C.; Huang, S. Targets of curcumin. Curr. Cancer Drug Targets 2012, 12, 332–347. [Google Scholar] [CrossRef] [PubMed]
- Kocaadam, B.; Sanlier, N. Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Crit. Rev. Food Sci. Nutr. 2017, 57, 2889–2895. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Patchva, S.; Koh, W.; Aggarwal, B.B. Discovery of curcumin, a component of the golden spice, and its miraculous biological activities. Clin. Exp. Pharmacol. Physiol. 2012, 39, 283–299. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Prasad, S.; Kim, J.H.; Patchva, S.; Webb, L.J.; Priyadarsini, I.K.; Aggarwal, B.B. Multitargeting by curcumin as revealed by molecular interaction studies. Nat. Prod. Rep. 2011, 2011, 1937–1955. [Google Scholar] [CrossRef]
- Baliga, M.S.; Joseph, N.; Venkataranganna, M.V.; Saxena, A.; Ponemone, V.; Fayad, R. Curcumin, an active component of turmeric in the prevention and treatment of ulcerative colitis: Preclinical and clinical observations. Food Funct. 2012, 3, 1109–1117. [Google Scholar] [CrossRef]
- Taylor, R.A.; Leonard, M.C. Curcumin for inflammatory bowel disease: A review of human studies. Altern. Med. Rev. 2011, 16, 152–156. [Google Scholar]
- Khalaf, H.; Jass, J.; Olsson, E.P. Differential cytokine regulation by NF-κB and AP-1 in Jurkat T-cells. BMC Immunol. 2010, 11, 26. [Google Scholar] [CrossRef]
- Hutton, B.; Salanti, G.; Caldwell, D.M.; Chaimani, A.; Schmid, C.H.; Cameron, C.; Ioannidis, J.P.; Straus, S.; Thorlund, K.; Jansen, J.P.; et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: Checklist and explanations. Ann. Intern. Med. 2015, 162, 777–784. [Google Scholar] [CrossRef]
- O’Connor, D.; Green, S.; Higgins, P.T.J. Chapter 5: Defining the review question and developing criteria for including studies. In Cochrane Handbook for Systematic Review A of Interventions Version 5.0; Higgins, J.P.T., Greeen, S., Eds.; The Cochrane Collaboration: London, UK, 2008. [Google Scholar]
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0. Updated; The Cochrane Collaboration: London, UK, 2011. [Google Scholar]
- Jadad, A.R.; Moore, R.A.; Carroll, D.; Jenkinson, C.; Reynolds, D.J.; Gavaghan, D.J.; McQuay, H.J. Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control. Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef]
- Shapira, S.; Leshno, A.; Katz, D.; Maharshak, N.; Hevroni, G.; Jean-David, M.; Kraus, S.; Galazan, L.; Aroch, I.; Kazanov, D.; et al. Of mice and men: A novel dietary supplement for the treatment of ulcerative colitis. Ther. Adv. Gastroenterol. 2018, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Suskind, D.L.; Wahbeh, G.; Burpee, T.; Cohen, M.; Christie, D.; Weber, W. Tolerability of curcumin in pediatric inflammatory bowel disease: A forced dose titration study. J. Pediatric Gastroenterol. Nutr. 2013, 56, 277. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, R.J.; Hunter, J.O. Double blind, placebo controlled randomized trial of Curcuma extract in the treatment of steroid dependent inflammatory bowel disease. Gastroenterology 2003, 124, A205. [Google Scholar] [CrossRef]
- Banerjee, R.; Medaboina, K.; Boramma, G.G.; Amsrala, S.; Reddy, D.N. Novel bio-enhanced curcumin with mesalamine forinduction of remission in mild to moderate ulcerative colitis. Gastroenterology 2017, 152, S587. [Google Scholar] [CrossRef]
- Santos, L.R. Perfil Inflamatório de Pacientes com Colite Ulcerativa em uso de Cúrcuma Longa. Master’s Thesis, UFG-Faculdade de Nutrição, Goiânia, Brazil, 2017; p. 34. [Google Scholar]
- Hanai, H.; Iida, T.; Takeuchi, K.; Watanabe, F.; Maruyama, Y.; Andoh, A.; Tsujikawa, T.; Fujiyama, Y.; Mitsuyama, K.; Sata, M.; et al. Curcumin maintenance therapy for ulcerative colitis: Randomized, multicenter, double-blind, placebo-controlled trial. Clin. Gastroenterol. Hepatol. 2006, 4, 1502–1506. [Google Scholar] [CrossRef]
- Singla, V.; Pratap Mouli, V.; Garg, S.K.; Rai, T.; Choudhury, B.N.; Verma, P.; Deb, R.; Tiwari, V.; Rohatgi, S.; Dhingra, R.; et al. Induction with NCB-02 (curcumin) enema for mild-to-moderate distal ulcerative colitis—A randomized, placebo-controlled, pilot study. J. Crohn’s Colitis 2014, 8, 208–214. [Google Scholar] [CrossRef]
- Lang, A.; Salomon, N.; Wu, J.C.; Kopylov, U.; Lahat, A.; Har-Noy, O.; Ching, J.Y.; Cheong, P.K.; Avidan, B.; Gamus, D.; et al. Curcumin in combination with mesalamine induces remission in patients with mild-to-moderate ulcerative colitis in a randomized controlled trial. Clin. Gastroenterol. Hepatol. 2015, 13, 1444–1449. [Google Scholar] [CrossRef]
- Masoodi, M.; Mahdiabadi, M.A.; Mokhtare, M.; Agah, S.; Kashani, A.H.; Rezadoost, A.M.; Sabzikarian, M.; Talebi, A.; Sahebkar, A. The efficacy of curcuminoids in improvement of ulcerative colitis symptoms and patients’self-reported well-being: A randomized double-blind controlled trial. J. Cell. Biochem. 2018, 119, 9552–9559. [Google Scholar] [CrossRef] [PubMed]
- Kedia, S.; Bhatia, V.; Thareja, S.; Garg, S.; Mouli, V.P.; Bopanna, S.; Tiwari, V.; Makharia, G.; Ahuja, V. Low dose oral curcumin is not effective in induction of remission in mild to moderate ulcerative colitis: Results from a randomized double blind placebo controlled trial. World J. Gastrointest. Pharmacol. Ther. 2017, 8, 147–154. [Google Scholar] [CrossRef]
- Sadeghi, N.; Mansoori, A.; Shayesteh, A.; Hashemi, S.J. The effect of curcumin supplementation on clinical outcomes and inflammatory markers in patients with ulcerative colitis. Phytother. Res. 2019, 34, 1123–1133. [Google Scholar] [CrossRef]
- Yallapu, M.M.; Jaggi, M.; Chauhan, S.C. Curcumin nanoformulations: A future nanomedicine for cancer. Drug Discov. Today 2012, 17, 71–80. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Refined exposure assessment for curcumin (E 100). EFSA J. 2014, 12, 3876. [Google Scholar] [CrossRef]
- JECFA. Curcumin. (Prepared by Ivan Stankovic). Chemical and Technical Assessment Compendıum Addendum 11/Fnp 52 Add.11/29. Monographs 2014, 1, 417. [Google Scholar]
- Lao, C.D.; Ruffin, M.T.; Normolle, D.; Heath, D.D.; Murray, S.I.; Bailey, J.M.; Boggs, M.E.; Crowell, J.; Rock, C.L.; Brenner, D.E. Dose escalation of a curcuminoid formulation. BMC Complement. Altern. Med. 2006, 6, 10. [Google Scholar] [CrossRef] [PubMed]
- Schiborr, C.; Kocher, A.; Behnam, D.; Jandasek, J.; Toelstede, S.; Frank, J. The oral bioavailability of curcumin from micronized powder and liquid micelles is significantly increased in healthy humans and differs between sexes. Mol. Nutr. Food Res. 2014, 58, 516–527. [Google Scholar] [CrossRef] [PubMed]
- Ohno, M.; Nishida, A.; Sugitani, Y.; Nishino, K.; Inatomi, O.; Sugimoto, M.; Kawahara, M.; Andoh, A. Nanoparticle curcumin ameliorates experimental colitis via modulation of gut microbiota and induction of regulatory T cells. PLoS ONE 2017, 12, e0185999. [Google Scholar] [CrossRef] [PubMed]
- Jamwal, R. Bioavailable curcumin formulations: A review of pharmacokinetic studies in healthy volunteers. J. Integr. Med. 2018, 16, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Shoba, G.; Joy, D.; Joseph, T.; Majeed, M.; Rajendran, R.; Srinivas, P.S. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998, 64, 353–356. [Google Scholar] [CrossRef]
- Li, Q.; Zhai, W.; Jiang, Q.; Huang, R.; Liu, L.; Dai, J.; Gong, W.; Du, S.; Wu, Q. Curcumin-piperine mixtures in self-microemulsifying drug delivery system for ulcerative colitis therapy. Int. J. Pharm. 2015, 490, 22–31. [Google Scholar] [CrossRef]
- Kumar, G.; Mittal, S.; Sak, K.; Tuli, H.S. Molecular mechanisms underlying chemopreventive potencial of curcumin: Current challenges and future perspectives. Life Sci. J. 2016, 148, 313–328. [Google Scholar] [CrossRef]
- Platel, K.; Srinivasan, K. Studies on the influence of dietary spices on food transit time in experimental rats. Nutr. Res. 2001, 21, 1309–1314. [Google Scholar] [CrossRef]
- Tanko, H.; Carrier, D.J.; Duan, L.; Clausen, E. Pre-and post-harvest processing of medicinal plants. Plant Genet. Resour. 2005, 3, 304–313. [Google Scholar] [CrossRef]
- Figueiredo, A.C.; Barroso, J.G.; Pedro, L.G.; Scheffer, J.J. Factors affecting secondary metabolite production in plants: Volatile components and essential oils. Flavour Fragr. J. 2008, 23, 213–226. [Google Scholar] [CrossRef]
- Choudhury, B.U.; Nath, A.; Hazarika, S.; Ansari, M.A.; Buragohain, J.; Mishra, D. Effects of pre-harvest soil management practices and post-harvest processing on phytochemical qualities of turmeric (Curcuma longa). Indian J. Agric. Sci. 2017, 87, 1002–1007. [Google Scholar]
| PubMed | |
| #1 (Inflammatory Bowel Disease [Mesh] or Inflammatory Bowel Disease [Tiab] or Crohn Disease [Mesh] or Crohn Disease [Tiab] or Proctocolitis [Mesh] or Proctocolitis [Tiab]) | #2 (Curcuma [Mesh] or Curcuma [Tiab] or Curcumin [Mesh] or Curcumin * [Tiab]) |
| #1 AND #2 | |
| Scopus | |
| #1 (TITLE-ABS-KEY ((“Inflammatory Bowel Disease” or “Crohn Disease” or proctocolitis))) | #2 (TITLE-ABS-KEY ((curcuma or curcumin *))) |
| #1 AND #2 | |
| Web of Science | |
| #1 (“Inflammatory Bowel Disease” or “Crohn Disease” or Proctocolitis) | #2 (Curcuma or Curcumin *) |
| #1 AND #2 | |
| Lilacs | |
| #1 tw: (tw: ((mh: “inflamatory bowel diseases” or “doenças inflamatórias intestinais” or mh: “crohn disease” or “doença de crohn” or mh: proctocolitis or “retocolite ulcerativa”))) | #2 (tw: (tw: ((mh: curcumin or curcumina or curcuma)))) |
| #1 AND #2 | |
| Food Science and Technology Abstracts | |
| #1 (“Inflammatory Bowel Disease” or “Crohn Disease” or Proctocolitis) | #2 (Curcuma or Curcumin *) |
| #1 AND #2 | |
| ScienceDirect | |
| #1 (“Inflammatory Bowel Disease” or “Crohn Disease” or Proctocolitis) | # (curcumin or curcumina or curcuma) |
| #1 AND #2 | |
| Cochrane Library | |
| #1 MeSH descriptor: [Inflammatory Bowel Diseases] explode all trees | #8 Proctocolitis |
| #2 “Inflammatory Bowel Disease” | #9 #7 or #8 |
| #3 #1 or #2 | #10 #3 or #6 or #9 |
| #4 MeSH descriptor: [Crohn Disease] explode all trees | #11 MeSH descriptor: [Curcuma] explode all trees |
| #5 “Crohn Disease” | #12 (Curcuma or Curcumin or Curcumin *) |
| #6 #4 or #5 | #13 #11 or #12 |
| #7 MeSH descriptor: [Proctocolitis] explode all trees | #14 #10 and #13 |
| Author, Year and Country | Study Design | Characterization of UC Population | Intervention | Variables of Interest Analyzed | Results |
|---|---|---|---|---|---|
| Hanai et al. 2006 [28] | Multicenter, randomized, double-blind, placebo-controlled | In remission | Curcumin (capsule) 2 g + 1.5–3 g 5ASA or 1–3 g sulfasalazine/day (n = 45) or placebo + 5ASA/sulfasalazine (n = 44) | Clinical Activity Index and Endoscopic Index assessed at baseline and every 2 months up to 12 months |
|
| n = 89 (49♂/30♀) | |||||
| Japão | 25–61 years | 6 months | |||
| Singla et al. 2014 [29] | Pilot study, double-blind, randomized, placebo-controlled | Mild/moderate proctitis and proctosigmoiditis | 140 mg NCB-02 (standardized extract curcumin) enema + oral 1.6 g 5ASA/day (n = 28) or placebo enema + oral 1.6 g 5ASA/day (n = 22) | UCDAI and endoscopic activity by mucosal appearance score at baseline and after 8 weeks |
|
| n = 45 (22♂/23♀) | |||||
| India | 23–49 years | 8 weeks | |||
| Lang et al. 2015 [30] | Multicenter, randomized, double-blind, placebo-controlled | Mild/moderate proctitis/left colitis/pancolitis | 95% pure curcumin (capsule)—3 g + 4 g 5ASA/day (n = 26) or placebo + 4 g 5ASA/day (n = 24) | SCCAI and Mayo endoscopic score assessed at baseline and after 4 weeks |
|
| n = 50 (17♂/33♀) | |||||
| Israel, Hong Kong and Cyprus. | 27–55 years | 4 weeks | |||
| Masoodi et al. 2018 [31] | Single-center, Double-blind, randomized, placebo-controlled | Mild/moderate left colitis/pancolitis | Nanomicellar curcumin (capsule) 80 mg 3x/day = 240 mg + 3 g 5ASA/day (n = 28) or placebo + 3 g 5ASA/day (n = 28) | SCCAI assessed at baseline, and at 2 and 4 weeks |
|
| n = 56 (28♂/28♀) | 4 weeks | ||||
| Iran | 25–54 years | ||||
| Kedia et al. 2017 [32] | Single-center, Double-blind, randomized, placebo-controlled | Mild/moderate proctitis/left colitis/pancolitis | Curcumin (capsule) 450 mg/day + 2.4 g 5-ASA/day (n = 29) or placebo + 2.4 g 5-ASA/day (n = 33) | UCDAI and endoscopic Baron score evaluation assessed at baseline, and at 4 and 8 weeks |
|
| n = 62 (41♂/21♀) | |||||
| India | 24–48 years | 8 weeks | |||
| Sadeghi et al. 2019 [33] | Double-blind, randomized, placebo-controlled | Mild/moderate proctitis/left colitis/pancolitis) | Curcumin (capsule-turmeric extract) 1.500 mg/day + routine drugs (n = 35) or placebo + routine drugs (n = 35) | SCCAI, IBDQ-9, ESR, hs-CRP, anthropometric indices and dietary intakes were assessed at baseline and after 8 weeks |
|
| n = 70 (21♂/49♀) | |||||
| Iran | 27–53 years | 8 weeks |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coelho, M.R.; Romi, M.D.; Ferreira, D.M.T.P.; Zaltman, C.; Soares-Mota, M. The Use of Curcumin as a Complementary Therapy in Ulcerative Colitis: A Systematic Review of Randomized Controlled Clinical Trials. Nutrients 2020, 12, 2296. https://doi.org/10.3390/nu12082296
Coelho MR, Romi MD, Ferreira DMTP, Zaltman C, Soares-Mota M. The Use of Curcumin as a Complementary Therapy in Ulcerative Colitis: A Systematic Review of Randomized Controlled Clinical Trials. Nutrients. 2020; 12(8):2296. https://doi.org/10.3390/nu12082296
Chicago/Turabian StyleCoelho, Mariana Roque, Marcela Diogo Romi, Daniele Masterson Tavares Pereira Ferreira, Cyrla Zaltman, and Marcia Soares-Mota. 2020. "The Use of Curcumin as a Complementary Therapy in Ulcerative Colitis: A Systematic Review of Randomized Controlled Clinical Trials" Nutrients 12, no. 8: 2296. https://doi.org/10.3390/nu12082296
APA StyleCoelho, M. R., Romi, M. D., Ferreira, D. M. T. P., Zaltman, C., & Soares-Mota, M. (2020). The Use of Curcumin as a Complementary Therapy in Ulcerative Colitis: A Systematic Review of Randomized Controlled Clinical Trials. Nutrients, 12(8), 2296. https://doi.org/10.3390/nu12082296






