Regular Consumption of Lipigo® Promotes the Reduction of Body Weight and Improves the Rebound Effect of Obese People Undergo a Comprehensive Weight Loss Program
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Study Design
2.3. Treatments
2.3.1. Dietary Program
2.3.2. Physical Activity Recommendations
2.3.3. Nutrition and Health Education Sessions
2.3.4. Lipigo® or Placebo
2.4. Endpoints
2.4.1. General Health Status Variables
2.4.2. Anthropometrics and Body Composition Variables
2.4.3. Dietary Variables
2.4.4. Physical Activity Variables
2.4.5. Compliance and Adverse Events
2.5. Statistical Analysis
3. Results
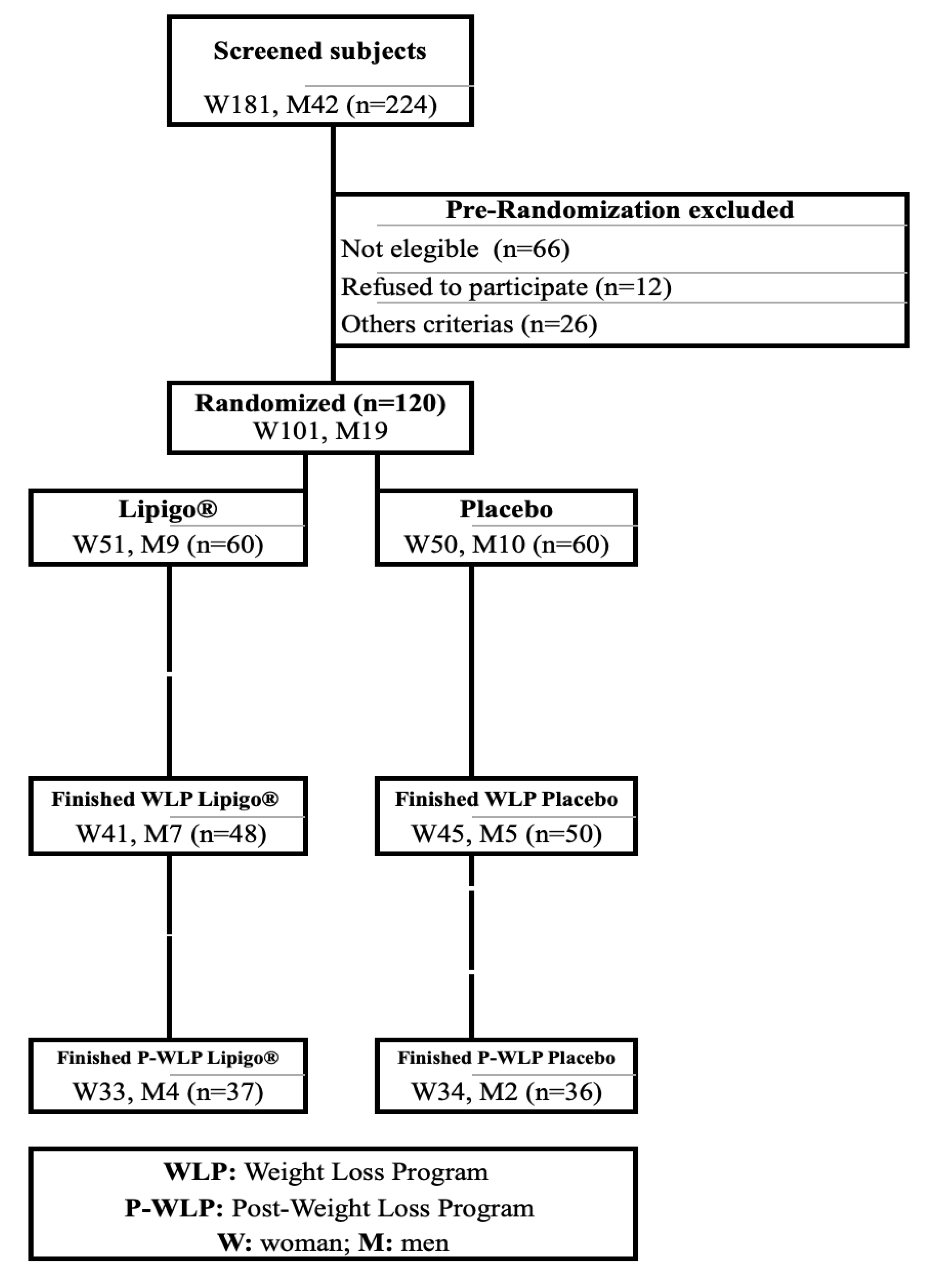
3.1. Recruitment and Study Population
3.2. Baseline Characteristics
3.3. Anthropometric and Body Composition Variables
3.4. Dietary Variables
3.5. Physical Activity Variables
3.6. Compliance and Adverse Events
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Inoue, Y.; Qin, B.; Poti, J.; Sokol, R.; Gordon-Larsen, P. Epidemiology of obesity in adults: Latest trends. Curr. Obes. Rep. 2018, 7, 276–288. [Google Scholar] [CrossRef] [PubMed]
- GBD 2015 Obesity Collaborators; Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; et al. Health effects of overweight and obesity in 195 countries over 25 years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Finucane, M.M.; Stevens, G.A.; Cowan, M.J.; Danaei, G.; Lin, J.K.; Paciorek, C.J.; Singh, G.M.; Gutierrez, H.R.; Lu, Y.; Bahalim, A.N.; et al. National, regional, and global trends in body-mass index since 1980: Systematic analysis of health examination surveys and epidemiological studies with 960 country-years and 9.1 million participants. Lancet 2011, 377, 557–567. [Google Scholar] [CrossRef]
- Misra, A.; Khurana, L. Obesity and the metabolic syndrome in developing countries. J. Clin. Endocrinol. Metab. 2008, 93, S9–S30. [Google Scholar] [CrossRef]
- Berger, N.A. Obesity and cancer pathogenesis. Ann. N. Y. Acad. Sci. 2014, 1311, 57–76. [Google Scholar] [CrossRef]
- WHO. World Health Organization (WHO), Obesity and Overweight. Fact sheet. 2014. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/ (accessed on 16 January 2017).
- Franks, P.W.; McCarthy, M.I. Exposing the exposures responsible for type 2 diabetes and obesity. Science 2016, 354, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Hooper, L.; Abdelhamid, A.; Moore, H.J.; Douthwaite, W.; Skeaff, C.M.; Summerbell, C.D. Effect of reducing total fat intake on body weight: Systematic review and meta-analysis of randomised controlled trials and cohort studies. BMJ 2012, 345, e7666. [Google Scholar] [CrossRef]
- Kroeger, C.M.; Hoddy, K.K.; Varady, K.A. Impact of weight regain on metabolic disease risk: A review of human trials. JObes 2014, 2014, 614519. [Google Scholar] [CrossRef]
- Loveman, E.; Frampton, G.K.; Shepherd, J.; Picot, J.; Cooper, K.; Bryant, J.; Welch, K.; Clegg, A. The clinical effectiveness and cost effectiveness of long-term weight management schemes for adults:systematic review. Health Technol. Assess (Rockv) 2011, 15, 1–6. [Google Scholar]
- Poddar, K.; Kolge, S.; Bezman, L.; Mullin, G.E.; Cheskin, L.J. Nutraceutical supplements for weight loss: A systematic review. Nutr. Clin. Pract. 2011, 26, 539–552. [Google Scholar] [CrossRef]
- Johansson, K.; Neovius, M.; Hemmingsson, E. Effects of anti-obesity drugs, diet and exercise on weight-loss maintenance after a very-low-calorie diet or low-calorie diet: A systematic review and meta-analysis of randomized controlled. Am. J. Clin. Nutr. 2014, 99, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Freimund, S.; Sauter, M.; Käppeli, O.; Dutler, H. A new non-degrading isolation process for 1,3-𝛽-d-glucan of high purity from baker’s yeast Saccharomyces cerevisiae. Carbohydr. Polym. 2003, 54, 159–171. [Google Scholar] [CrossRef]
- El Khoury, D.; Cuda, C.; Luhovyy, B.L.; Anderson, G.H. Beta glucan:Health benefits in obesity and metabolic syndrome. J. Nutr. Metab. 2012, 2012, 851362. [Google Scholar] [CrossRef]
- Stier, H.; Ebbeskotte, V.; Gruenwald, J. Immune-modulatory effects of dietary yeast beta-1,3/1,6-D-glucan. Nutr. J. 2014, 13, 38. [Google Scholar] [CrossRef]
- Jung, E.Y.; Cho, M.K.; Hong, Y.H.; Kim, J.H.; Park, Y.; Chang, U.J.; Suh, H.J. Yeast hydrolysate can reduce body weight and abdominal fat accumulation in obese adults. Nutrition 2014, 30, 25–32. [Google Scholar] [CrossRef]
- Nicolosi, R.; Bell, S.J.; Bistrian, B.R.; Greenberg, I.; Forse, R.A.; Blackburn, G.L. Plasma lipid changes after supplementation with beta-glucan fiber from yeast. Am. J. Clin. Nutr. 1999, 70, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Yanni, A.E.; Stamataki, N.S.; Konstantopoulos, P.; Stoupaki, M.; Abeliatis, A.; Nikolakea, I.; Perrea, D.; Karathanos, V.T.; Tentolouris, N. Controlling type-2 diabetes by inclusion of Cr-enriched yeast bread in the daily dietary pattern: A randomized clinical trial. Eur. J. Nutr. 2018, 57, 259–267. [Google Scholar] [CrossRef]
- Santas, J.; Lazaro, E.; Cuñé, J. Effect of a polysaccharide-rich hydrolysate from Saccharomyces cerevisiae (LipiGo®) in body weight loss: Randomised, double-blind, placebo-controlled clinical trial in overweight and obese adults. J. Sci. Food Agric. 2017, 97, 4250–4257. [Google Scholar] [CrossRef]
- Mancebo, R.; Castañé, X.; Cuñé, J.; Santas, J.; Rafecas, M.; Mirealles, M.; Mateos-Aparicio, I.; Heras, A. Fat-binder Obtained from Biomass Resulting from Beer Production. International application number: PCT/ES2013/070408. WO2014001589, 2014. [Google Scholar]
- Puri, K.S.; Suresh, K.R.; Gogtay, N.J.; Thatte, U.M. Declaration of Helsinki, 2008: Implications for stakeholders in research. J. Postgrad. Med. 2009, 55, 131–134. [Google Scholar]
- Spanish Society of Community Nutrition (SENC). Healthy nutrition Guide. Madrid: SENC. 2016. Available online: https://www.nutricioncomunitaria.org/es/noticia/guias-alimentarias-senc-2016 (accessed on 29 June 2020).
- Wycherley, T.P.; Moran, L.J.; Clifton, P.M.; Noakes, M.; Brinkworth, G.D. Effects of energy-restricted high-protein, low-fat compared with standard protein, low-fat diets: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2012, 96, 1281–1298. [Google Scholar] [CrossRef]
- WHO/FAO. Methodology of Nutritional Surveillance. Report of a Joint FAO/UNICEF/WHO Expert Committee; The World Health Organization Technical Report Series; WHO/FAO: Geneva, Switzerland, 1976. [Google Scholar]
- Sun, S.S.; Chumlea, W.C.; Heymsfield, S.B.; Lukaski, H.C.; Schoeller, D.; Friedl, K.; Kuczmarski, R.J.; Flegal, K.M.; Johnson, C.L.; Hubbard, V.S. Development of bioelectrical impedance analysis prediction equations for body composition with the use of a multicomponent model for use in epidemiologic surveys. Am. J. Clin. Nutr. 2003, 77, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Dehghan, M.; Merchant, A.T. Is bioelectrical impedance accurate for use in large epidemiological studies? Nutr. J. 2008, 7, 26. [Google Scholar] [CrossRef]
- Ortega, R.M.; Requejo, A.M.; López-Sobaler, A.M. Models of questionnaires for dietary studies, in the assessment of nutritional status. In Nutriguía Manual of Clinical Nu-trition in Primary Care; Ortega, R.M., Requejo, A.M., Eds.; Complutense: Madrid, India, 2006; pp. 456–467. [Google Scholar]
- Group USA Spanish Version Translated 3/2003-Short Last 7 Days Slef-Administered Version of the International Physical Activity Questionnaire (IPAQ) IPAQ–Revised August 2002. Available online: www.ipaq.ki.se (accessed on 29 June 2020).
- Dombrowski, S.U.; Knittle, K.; Avenell, A.; Araújo-Soares, V.; Sniehotta, F.F. Long term maintenance of weight loss with non-surgical interventions in obese adults: Systematic review and meta-analyses of randomised controlled trials. BMJ 2014, 348, g2646. [Google Scholar] [CrossRef]
- Pittler, M.; Abbot, N.; Harkness, E.; Ernst, E. Randomized, double-blind trial of chitosan for body weight reduction. Eur. J. Clin. Nutr. 1999, 53, 379–381. [Google Scholar] [CrossRef] [PubMed]
- Moraru, C.; Mincea, M.M.; Frandes, M.; Timar, B.; Ostafe, V. A Meta-Analysis on Randomised Controlled Clinical Trials Evaluating the Effect of the Dietary Supplement Chitosan on Weight Loss, Lipid Parameters and Blood Pressure. Medicina 2018, 54, 109. [Google Scholar] [CrossRef] [PubMed]
- Cornelli, U.; Belcaro, G.; Recchia, M.; D’Orazio, N. Long-Term Treatment of Overweight and Obesity with Polyglucosamine (PG L112): Randomized Study Compared with Placebo in Subjects after Caloric Restriction. Curr. Dev. Nutr. 2017, 1, e000919. [Google Scholar] [CrossRef]
- Pokhis, K.; Bitterlich, N.; Cornelli, U.; Cassano, G. Efficacy of polyglucosamine for weight loss-confirmed in a randomized double-blind, placebo-controlled clinical investigation. BMC Obes. 2015, 2, 25. [Google Scholar] [CrossRef]
- Cornelli, U.; Belcaro, G.; Cesarone, M.R.; Cornelli, M. Use of polyglucosamine and physical activity to reduce body weight and dyslipidemia in moderately overweight subjects. Minerva Cardioangiol. 2008, 56 (Suppl. 5), 71–78. [Google Scholar] [PubMed]
- Trivedi, V.R.; Satia, M.C.; Deschamps, A.; Maquet, V.; Shah, R.B.; Zinzuwadia, P.H.; Trivedi, J.V. Single-blind, placebo controlled randomised clinical study of chitosan for body weight reduction. Nutr. J. 2016, 15, 3. [Google Scholar] [CrossRef]
- Mhurchu, C.N.; Poppitt, S.D.; McGill, A.; Leahy, F.E.; Bennett, D.A.; Lin, R.B.; Ormrod, D.; Ward, L.; Strik, C.; Rodgers, A. The effect of the dietary supplement, Chitosan, on body weight: A randomised controlled trial in 250 overweight and obese adults. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 1149–1156. [Google Scholar] [CrossRef]
- Kaats, G.R.; Michalek, J.E.; Preuss, H.G. Evaluating Efficacy of a Chitosan Product Using a Double-Blinded, Placebo-Controlled Protocol. J. Am. Coll. Nutr. 2006, 25, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Woodgate, D.E.; Conquer, J.A. Effects of a stimulant-free dietary supplement on body weight and fat loss in obese adults: A six-week exploratory study. Curr. Ther. Res. Clin. Exp. 2003, 64, 248–262. [Google Scholar] [CrossRef]
- Duncan, D.; Colin, K.; Scharoun-Lee, M.; Ding, E.; Warner, E.; Bennett, G. Does perception equal reality? Weight misperception in relation to weight-related attitudes and behaviors among overweight and obese US adults. Int. J. Behav. Nutr. Phys. Act. 2011, 8, 20. [Google Scholar] [CrossRef] [PubMed]
- Coleman, L.; Loprinzi, P.D. The association between discrepant weight perceptions and objectively measured physical activity. Prev. Med. 2016, 87, 47–50. [Google Scholar] [CrossRef] [PubMed]
- Rashad, N.M.; Sayed, S.E.; Sherif, M.H.; Sitohy, M.Z. Effect of a 24-week weight management program on serum leptin level in correlation to anthropometric measures in obese female: A randomized controlled clinical trial. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 2230–2235. [Google Scholar] [CrossRef]
- Kyle, U.G.; Bosaeus, I.; De Lorenzo, A.D.; Deurenberg, P.; Elia, M.; Manuel Gómez, J.; Lilienthal Heitmann, B.; Kent-Smith, L.; Melchior, J.C.; Pirlich, M.; et al. Bioelectrical impedance analysis-part II: Utilization in clinical practice. Clin. Nutr. 2004, 23, 1430–1453. [Google Scholar] [CrossRef]
| Total | Placebo (n = 36) | Lipigo® (n = 37) | p-Value | ||
|---|---|---|---|---|---|
| Gender | (Female %, n) | 73 | 94.4 (34) | 89.2 (33) | 0.414 |
| Age | (years) | 50.9 ± 9.07 | 49.47 ± 10.4 | 52.3 ± 7.4 | 0.185 |
| Weight | (kg) | 82.85 ± 12.37 | 82.65 ± 12.5 | 83.04 ± 12.4 | 0.895 |
| BMI | (kg/m2) | 31.19 ± 3.44 | 31.2 ± 3.6 | 31.2 ± 3.3 | 0.976 |
| Waist circumference | (cm) | 97.76 ± 11.25 | 97.4 ± 11.6 | 98.2 ± 11.0 | 0.765 |
| FM | (kg) | 31.51 ± 7.65 | 32.3 ± 7.9 | 30.7 ± 7.5 | 0.400 |
| Lean mass | (kg) | 51.25 ± 10.15 | 50.3 ± 9.1 | 52.2 ± 11.1 | 0.439 |
| MM | (kg) | 35.21 ± 8.03 | 34.3 ± 6.8 | 36.2 ± 9.1 | 0.311 |
| SBP | (mmHg) | 111.47 ± 15.13 | 110.7 ± 15 | 112.3 ± 15.5 | 0.666 |
| DBP | (mmHg) | 77.19 ± 9.56 | 77.0 ± 9.7 | 77.4 ± 9.6 | 0.864 |
| Placebo (n = 36) | Lipigo® (n = 37) | p-Value | ||
|---|---|---|---|---|
| Weight (kg) | Baseline of WLP (V0) | 82.65 ± 12.48 | 82.65 ± 12.48 | 0.895 |
| End of WLP (V6) | 79.19 ± 12.3 | 78.61 ± 12.56 | 0.842 | |
| End of P-WLP (V9) | 80.18 ± 12.49 | 79.67 ± 13.01 | 0.864 | |
| BMI (kg/m2) | Baseline of WLP (V0) | 31.21 ± 3.65 | 31.18 ± 3.28 | 0.976 |
| End of WLP (V6) | 29.91 ± 3.54 | 29.39 ± 3.19 | 0.512 | |
| End of P-WLP(V9) | 30.27 ± 3.62 | 29.79 ± 3.32 | 0.558 | |
| Waist circumference (cm) | Baseline of WLP (V0) | 97.35 ± 11.6 | 98.18 ± 11.05 | 0.765 |
| End of WLP (V6) | 94.82 ± 11.82 | 93.41 ± 10.36 | 0.588 | |
| End of P-WLP (V9) | 91.4 ± 17.54 | 93.94 ± 10.66 | 0.456 | |
| Muscle mass (kg) | Baseline of WLP (V0) | 34.25 ± 6.84 | 36.19 ± 9.07 | 0.311 |
| End of WLP (V6) | 32.12 ± 6.55 | 33.23 ± 8.42 | 0.530 | |
| End of P-WLP (V9) | 32.51 ± 6.84 | 32.62 ± 6.87 | 0.944 | |
| Lean mass (kg) | Baseline of WLP (V0) | 50.32 ± 9.12 | 52.19 ± 11.14 | 0.439 |
| End of WLP (V6) | 48.75 ± 8.65 | 49.61 ± 9.01 | 0.680 | |
| End of P-WLP (V9) | 49.00 ± 9.11 | 48.92 ± 9.07 | 0.971 | |
| Fat mass (kg) | Baseline of WLP (V0) | 32.28 ± 7.86 | 30.74 ± 7.48 | 0.400 |
| End of WLP (V6) | 30.47 ± 7.21 | 29.01 ± 6.41 | 0.365 | |
| End of P-WLP (V9) | 31.32 ± 7.34 | 30.78 ± 7.17 | 0.755 |
| Placebo (n = 36) | Lipigo® (n = 37) | p-Value | ||
|---|---|---|---|---|
| Weight (kg) | Dif V0–V6 | −3.46 ± 2.00 | −4.43 ± 2.78 | 0.093 |
| Dif V6–V9 | 0.99 ± 2.35 | 1.06 ± 2.87 | 0.912 | |
| Rebound effect | Dif V0–V9 | −2.47 ± 3.41 | −3.36 ± 3.36 | 0.259 |
| BMI (kg/m2) | Dif V0–V6 | −1.31 ± 0.77 | −1.79 ± 1.35 | 0.063 |
| Dif V6–V9 | 0.36 ± 0.89 | 0.41 ± 1.09 | 0.831 | |
| Rebound effect | Dif V0–V9 | −0.94 ± 1.32 | −1.4 ± 1.58 | 0.178 |
| Waist circumference (cm) | Dif V0–V6 | −2.63 ± 3.1 | −4.34 ± 4.13 | 0.059 |
| Dif V6–V9 | −0.59 ± 3.3 | 0.53 ± 3.4 | 0.162 | |
| Rebound effect | Dif V0–V9 | −3.08 ± 3.44 | −3.48 ± 3.25 | 0.631 |
| Muscle mass (kg) | Dif V0–V6 | −2.14 ± 1.96 | −2.04 ± 1.85 | 0.837 |
| Dif V6–V9 | 0.39 ± 1.57 | −0.04 ± 1.36 | 0.218 | |
| Rebound effect | Dif V0–V9 | −1.74 ± 2.23 | −2.12 ± 1.74 | 0.444 |
| Lean mass (kg) | Dif V0–V6 | −1.57 ± 1.85 | −1.5 ± 1.76 | 0.872 |
| Dif V6–V9 | 0.25 ± 1.52 | −0.59 ± 1.57 | 0.024 | |
| Rebound effect | Dif V0–V9 | −1.32 ± 1.99 | −2.06 ± 1.84 | 0.108 |
| Fat mass (kg) | Dif V0–V6 | −1.81 ± 3.53 | −2.6 ± 2.56 | 0.282 |
| Dif V6–V9 | 0.85 ± 2.5 | 1.59 ± 2.56 | 0.214 | |
| Rebound effect | Dif V0–V9 | −0.96 ± 4.34 | −1.06 ± 3.32 | 0.908 |
| Placebo | Lipigo® | p-Value | ||
|---|---|---|---|---|
| Weight (kg) Overweight | Baseline of WLP (V0) | 74.25 ± 9.22 | 75.31 ± 6.30 | 0.706 |
| End of WLP (V6) | 71.18 ± 10.17 | 71.96 ± 7.77 | 0.810 | |
| End of P-WLP (V9) | 72.07 ± 10.41 | 72.65 ± 6.82 | 0.855 | |
| Dif V0–V6 | −2.96 ± 1.79 | −3.66 ± 2.60 | 0.732 | |
| Dif V6–V9 | 0.79 ± 1.83 | 0.92 ± 2.59 | 0.633 | |
| Rebound effect Dif V0–V9 | −2.17 ± 2.59 | −2.75 ± 3.27 | 0.806 | |
| Weight (kg) OB1 | Baseline of WLP (V0) | 84.77 ± 6.18 | 84.98 ± 8.77 | 0.942 |
| End of WLP (V6) | 81.69 ± 5.56 | 79.71 ± 9.53 | 0.503 | |
| End of P-WLP (V9) | 83.33 ± 6.66 | 80.79 ± 10.32 | 0.442 | |
| Dif V0–V6 | −3.08 ± 1.73 | −5.27 ± 2.76 | 0.017 | |
| Dif V6–V9 | 1.64 ± 1.65 | 1.09 ± 3.45 | 0.594 | |
| Rebound effect Dif V0–V9 | −1.44 ± 2.51 | −4.19 ± 3.61 | 0.026 | |
| BMI (kg/m2) Overweight | Baseline of WLP (V0) | 28.12 ± 1.14 | 28.34 ± 1.02 | 0.567 |
| End of WLP (V6) | 26.92 ± 1.27 | 27.05 ± 1.63 | 0.790 | |
| 9 months P-WLP (V9) | 27.26 ± 1.61 | 27.34 ± 1.48 | 0.881 | |
| Dif V0–V6 | 0.30 ± 0.72 | 0.37 ± 1.03 | 0.860 | |
| Dif V6–V9 | −1.16 ± 0.75 | −1.41 ± 1.0 | 0.807 | |
| Rebound effect Dif V0–V9 | −0.85 ± 1.05 | −1.01 ± 1.7 | 0.698 | |
| BMI (kg/m2) OB1 | Baseline of WLP (V0) | 31.98 ± 1.33 | 31.91 ± 1.22 | 0.883 |
| End of WLP (V6) | 30.89 ± 1.65 | 29.91 ± 1.78# | 0.136 | |
| End of P-WLP (V9) | 31.45 ± 1.99 | 30.30 ± 1.87 | 0.120 | |
| Dif V0–V6 | −1.09 ± 0.56 | −1.99 ± 1.09 | 0.010 | |
| Dif V6–V9 | 0.56 ± 0.63 | 0.4 ± 1.3 | 0.673 | |
| Rebound effect Dif V0–V9 | −0.52 ± 0.96 | −1.61 ± 1.43 | 0.025 | |
| Fat mass (kg) Overweight | Baseline of WLP (V0) | 26.55 ± 4.58 | 27.62 ± 3.69 | 0.481 |
| End of WLP (V6) | 24.96 ± 3.53 | 26.19 ± 4.42 | 0.391 | |
| End of P-WLP (V9) | 25.37 ± 4.55 | 26.94 ± 5.10 | 0.364 | |
| Dif V0–V6 | −1.58 ± 3.75 | −1.56 ± 2.76 | 0.982 | |
| Dif V6–V9 | 0.40 ± 1,94 | 0.75 ± 2.72 | 0.685 | |
| Rebound effect Dif V0–V9 | −1.18 ± 3.66 | −0.65 ± 3.63 | 0.804 | |
| Fat mass (kg) OB1 | Baseline of WLP (V0) | 32.91 ± 3.06 | 30.13 ± 7.78 | 0.222 |
| End of WLP (V6) | 31.48 ± 3.43 | 28.48 ± 5.56 | 0.145 | |
| End of P-WLP (V9) | 33.54 ± 3.69 | 31.31 ± 5.57 | 0.217 | |
| Dif V0–V6 | −1.44 ± 3.29 | −3.44±2.46 | 0.080 | |
| Dif V6–V9 | 2.06 ± 1.62 | 2.02 ± 2.53 | 0.958 | |
| Rebound effect Dif V0–V9 | 0.62 ± 3.80 | −1.62 ± 3.45 | 0.121 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valero-Pérez, M.; Bermejo, L.M.; López-Plaza, B.; García, M.A.; Palma-Milla, S.; Gómez-Candela, C. Regular Consumption of Lipigo® Promotes the Reduction of Body Weight and Improves the Rebound Effect of Obese People Undergo a Comprehensive Weight Loss Program. Nutrients 2020, 12, 1960. https://doi.org/10.3390/nu12071960
Valero-Pérez M, Bermejo LM, López-Plaza B, García MA, Palma-Milla S, Gómez-Candela C. Regular Consumption of Lipigo® Promotes the Reduction of Body Weight and Improves the Rebound Effect of Obese People Undergo a Comprehensive Weight Loss Program. Nutrients. 2020; 12(7):1960. https://doi.org/10.3390/nu12071960
Chicago/Turabian StyleValero-Pérez, Marlhyn, Laura M. Bermejo, Bricia López-Plaza, Meritxell Aguiló García, Samara Palma-Milla, and Carmen Gómez-Candela. 2020. "Regular Consumption of Lipigo® Promotes the Reduction of Body Weight and Improves the Rebound Effect of Obese People Undergo a Comprehensive Weight Loss Program" Nutrients 12, no. 7: 1960. https://doi.org/10.3390/nu12071960
APA StyleValero-Pérez, M., Bermejo, L. M., López-Plaza, B., García, M. A., Palma-Milla, S., & Gómez-Candela, C. (2020). Regular Consumption of Lipigo® Promotes the Reduction of Body Weight and Improves the Rebound Effect of Obese People Undergo a Comprehensive Weight Loss Program. Nutrients, 12(7), 1960. https://doi.org/10.3390/nu12071960






