Probiotic Strains and Intervention Total Doses for Modulating Obesity-Related Microbiota Dysbiosis: A Systematic Review and Meta-analysis
Abstract
1. Introduction
1.1. Probiotics for Dietary Supplementation Interventions
1.2. Microbiota Dysbiosis in Obesity-related Disorders: Obesogenic Substances
2. Materials and Methods
2.1. Eligibility Criteria
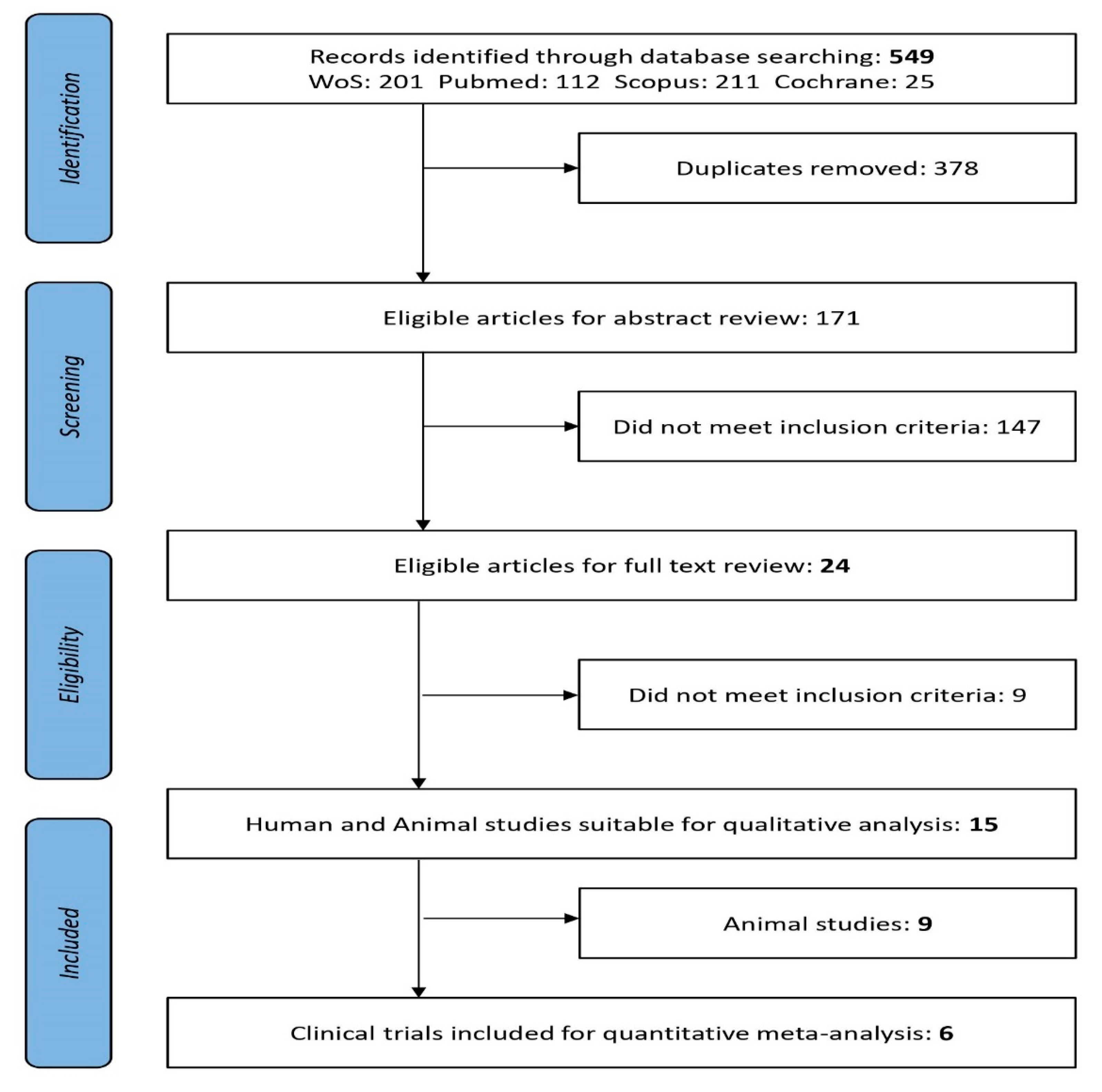
2.2. Search Strategy
2.3. Data Extraction and Analysis
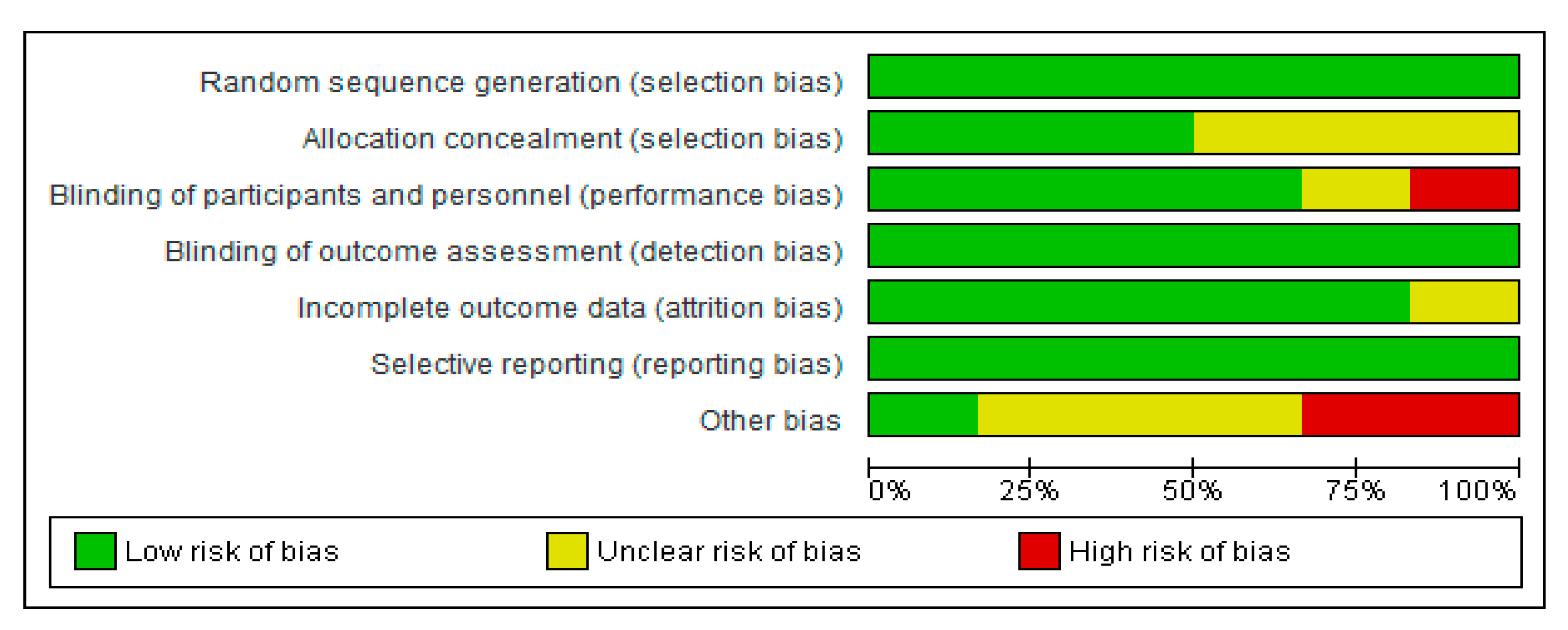
2.4. Risk of Bias (Quality) Assessment for the Selected Clinical Trials
2.5. Statistical Analysis
3. Results
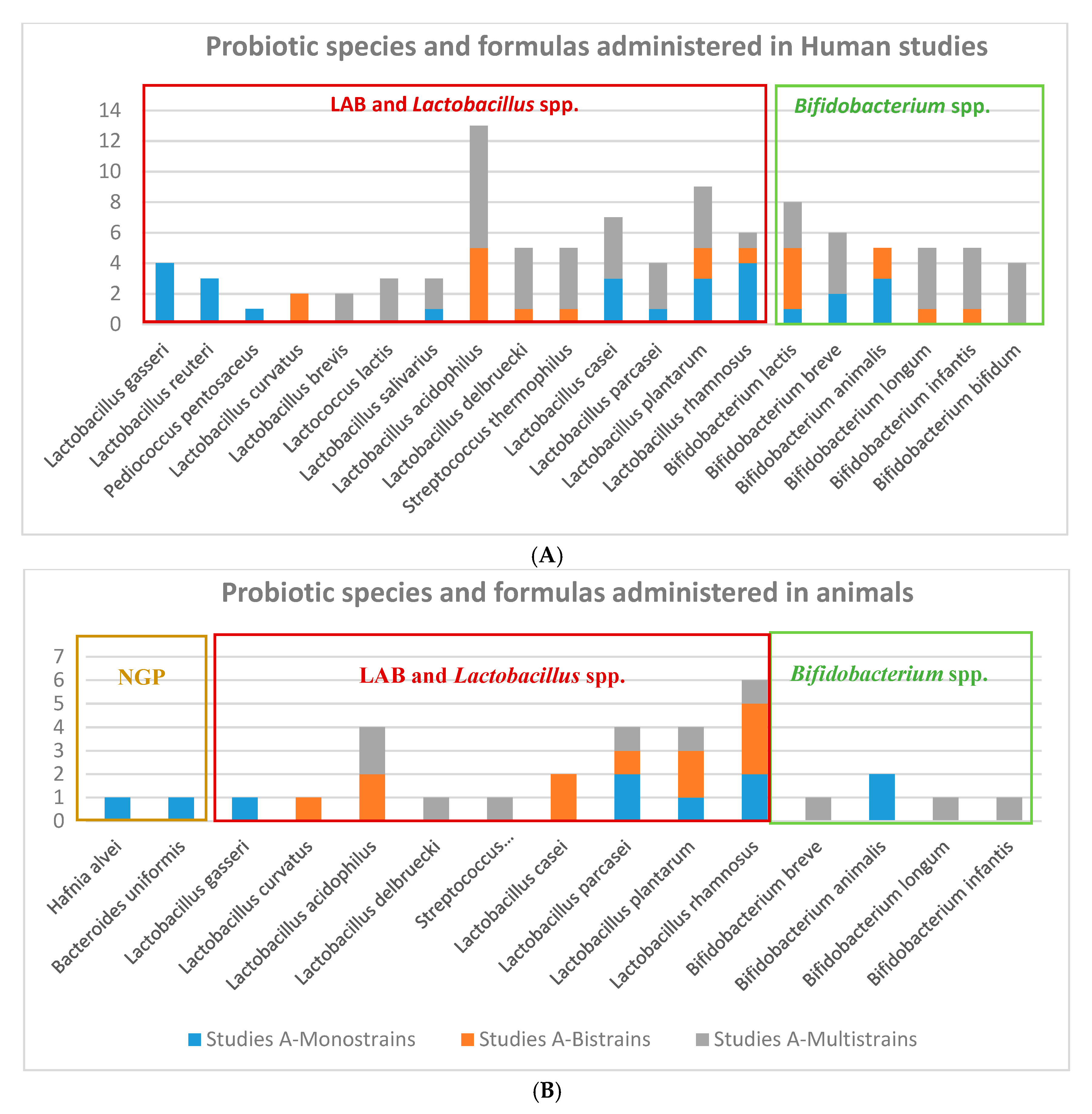
3.1. Research and Scientific Evidence on the Probiotic Strains and Doses Administered for Obesity-Related Disorders
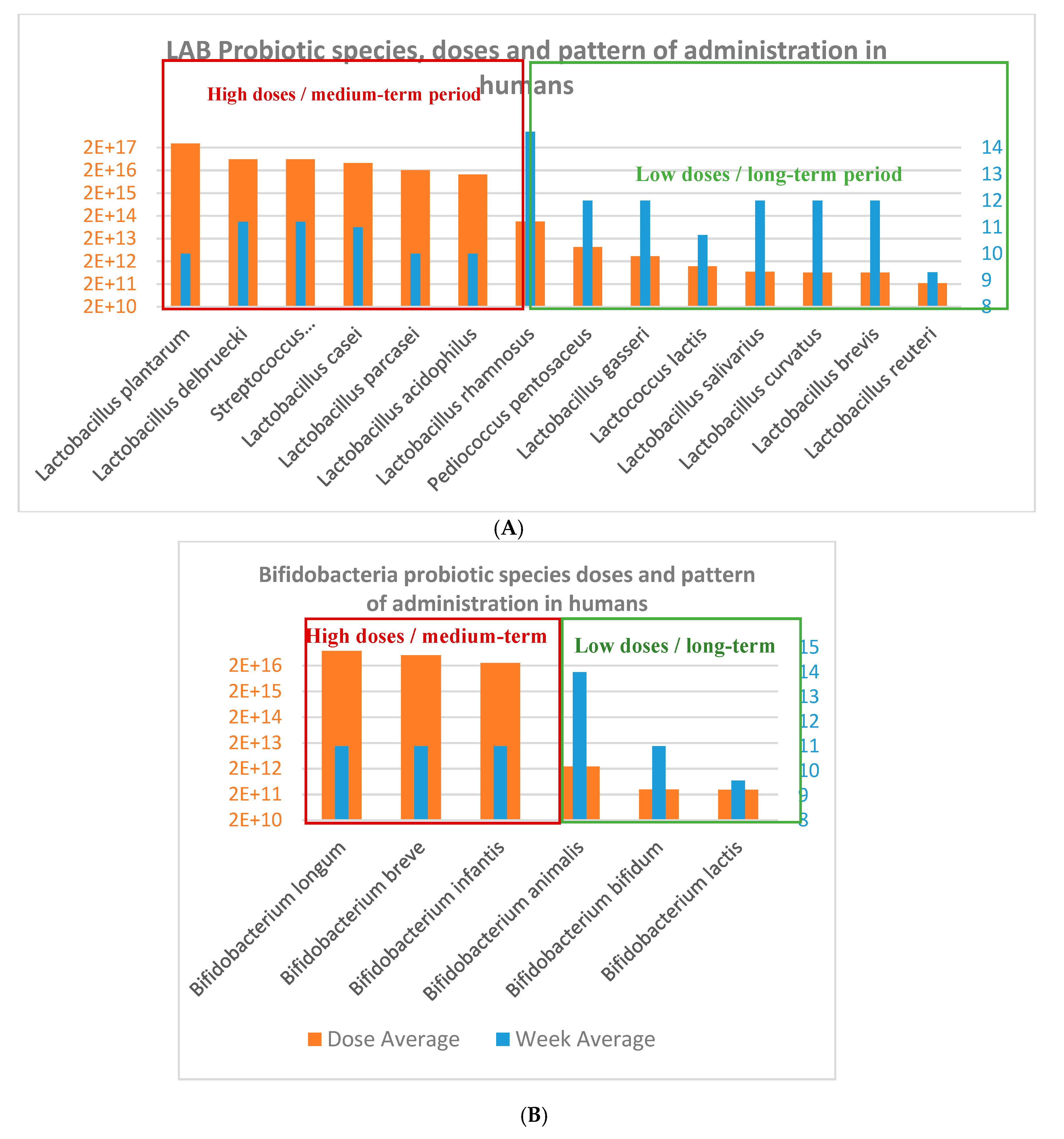
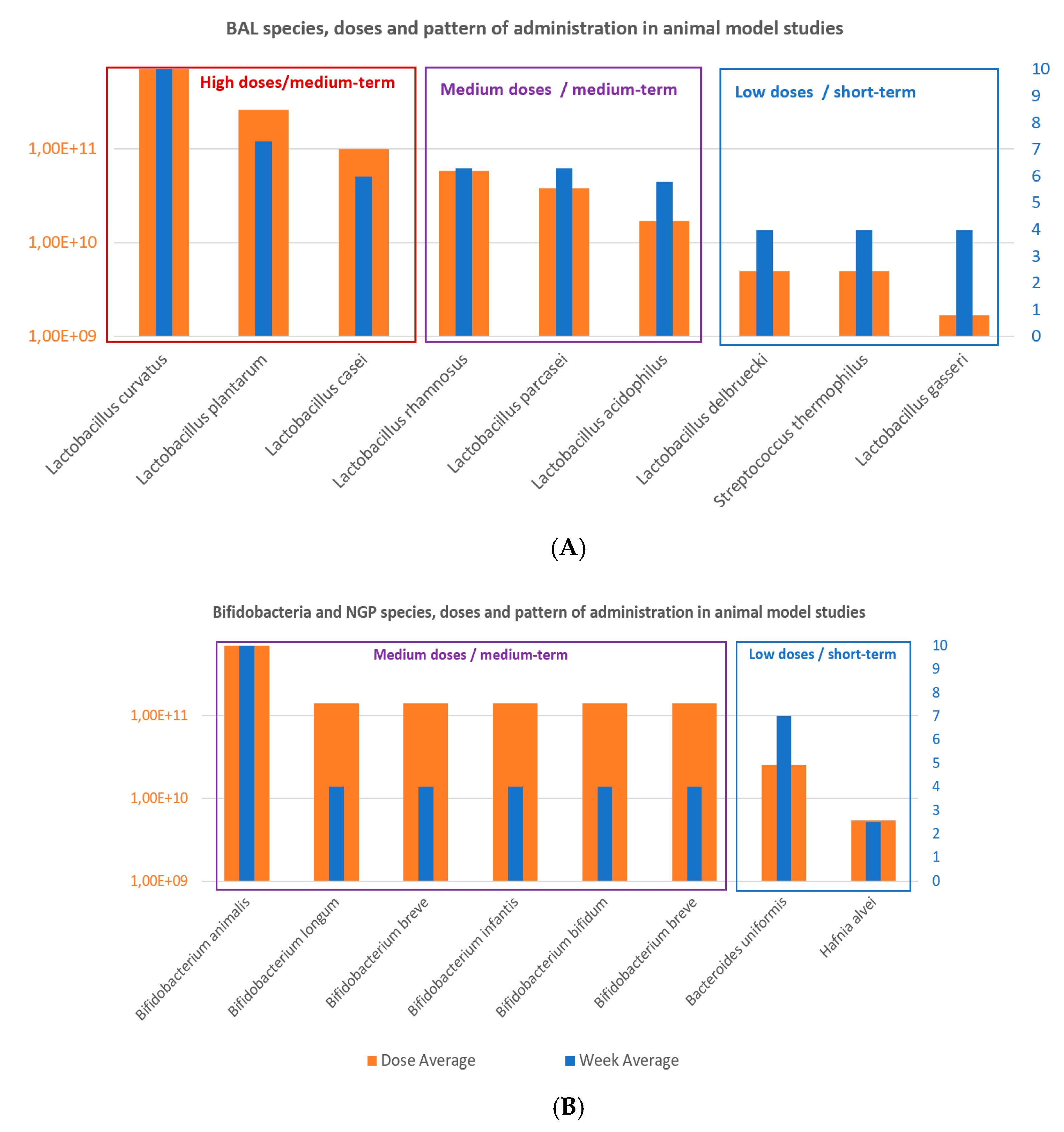
3.2. Probiotic Strains, Daily Doses and Intervention Total Doses in Obesity-related Clinical Studies
3.3. Probiotic Modulation Capacity on Individual Autochthony Microbiota and Clinical Parameters
3.3.1. Impact of Probiotics on Individual Autochthony Microbiota
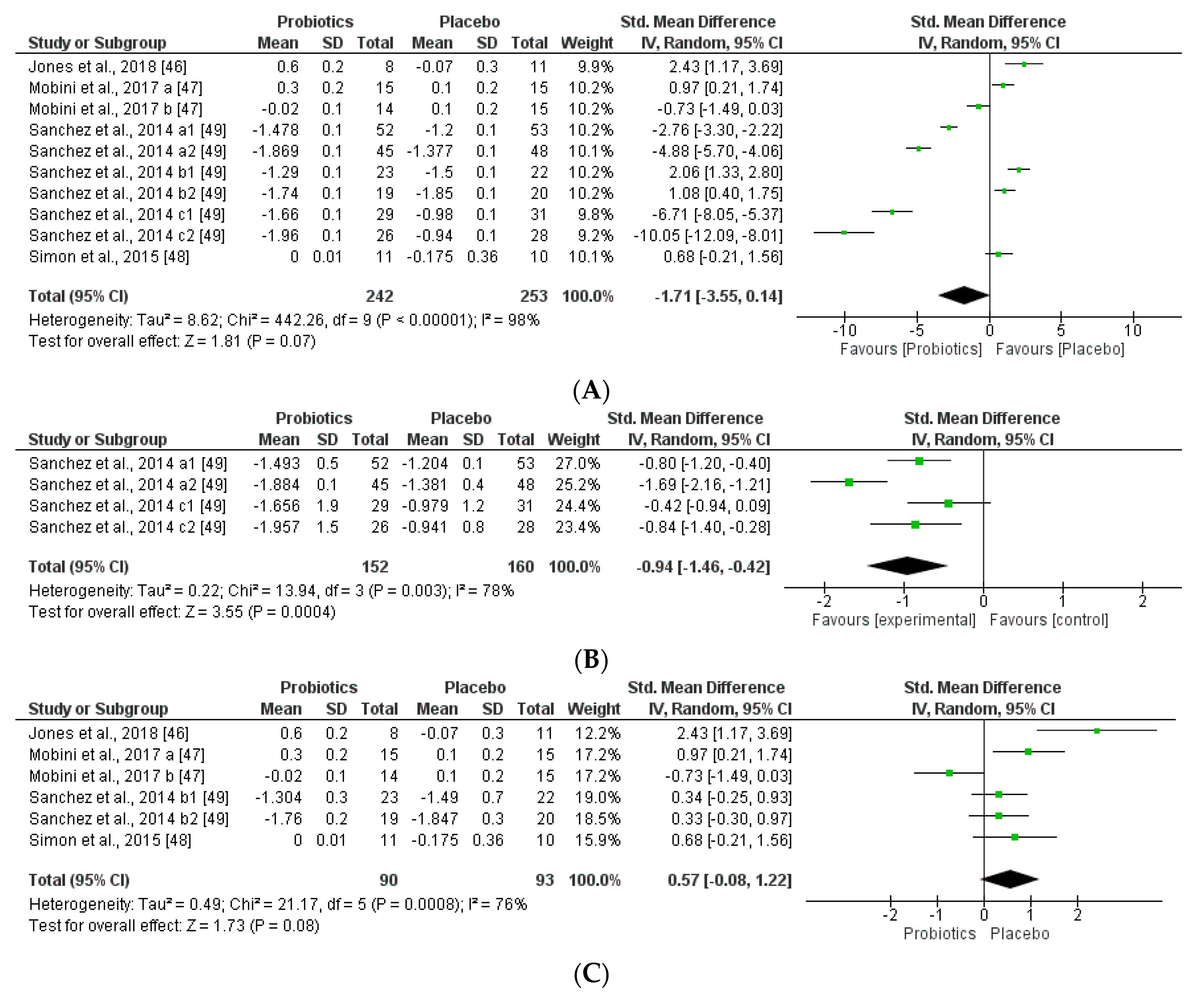
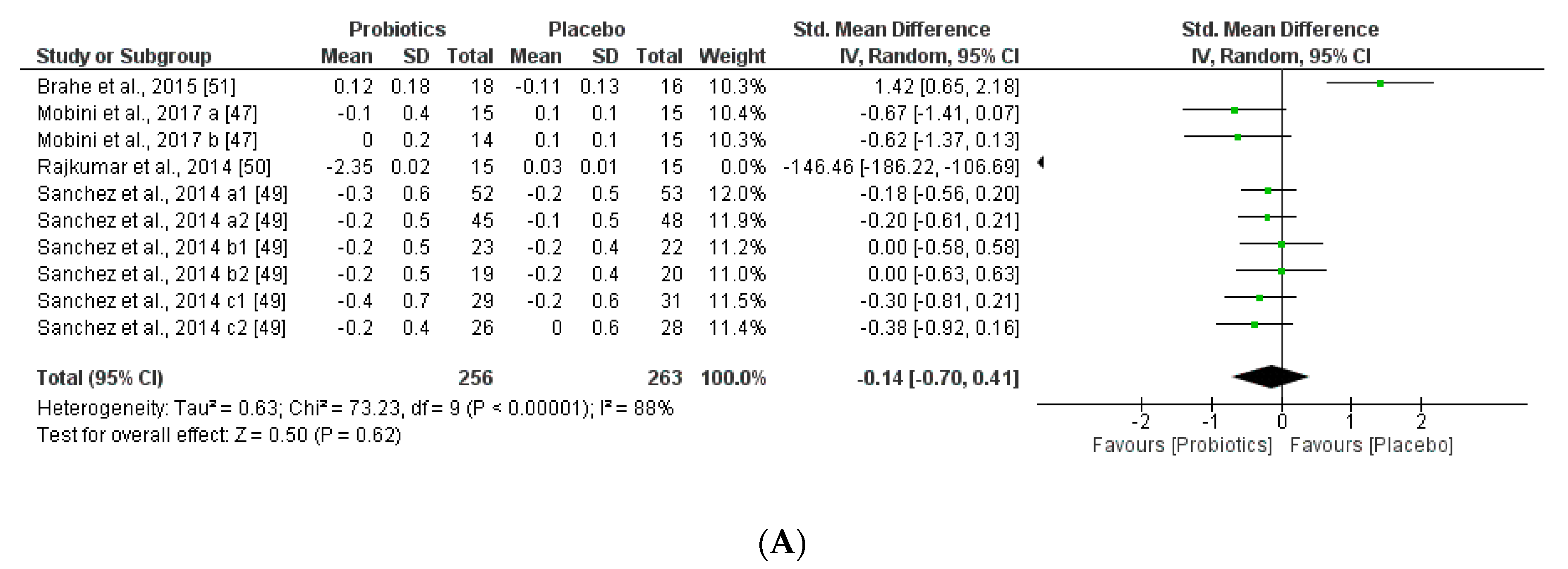
3.3.2. Impact on Obesity-related Clinical Parameters
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| aGLP-1 | Active Glucagon-like Peptide-1 |
| ALT | Liver toxicity biomarker alanine transaminase |
| BMI | Body Mass Index |
| CFU | Colony Forming Units |
| CRP | C-Reactive Protein |
| CT | Clinical Trials |
| FMT | Fecal Microbiota Transplant |
| FPG | Fasting Plasma Glucose |
| GLP-1 | Glucagon-like peptide-1 |
| GLP-2 | Glucagon-like peptide-2 |
| HDL | High density lipoprotein |
| hsCRP | high sensitivity C-reactive protein |
| ISI | Index Sensitivity Insulin |
| LDL | Low density lipoprotein |
| NAFL | Non-Alcoholic Fatty Liver |
| NAFLD | Non-Alcoholic Fatty Liver Disease |
| NASH | Non-Alcoholic Steatohepatitis |
| NGP | Next-generation probiotics |
| OA | Open Access |
| PCR | Polymerase Chain Reaction |
| qPCR | quantitative Polymerase Chain Reaction |
| TAG | Triglycerides |
| VLDL | Very low-density lipoprotein |
References
- Gardiner, G.; Heinemann, C.; Baroja, M.; Bruce, A.; Beuerman, D.; Madrenas, J.; Reid, G. Oral administration of the probiotic combination Lactobacillus rhamnosus GR-1 and L. fermentum RC-14 for human intestinal applications. Int. Dairy J. 2002, 12, 191–196. [Google Scholar] [CrossRef]
- Verna, E.; Lucak, S. Use of probiotics in gastrointestinal disorders: What to recommend? Ther. Adv. Gastroenterol. 2010, 3, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.; Hutkins, R.; Sanders, M.; Prescott, S.; Reimer, R.; Salminen, S.; Scott, K.; Stanton, C.; Swanson, K.; Cani, P.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Daliri, E.; Lee, B. New perspectives on probiotics in health and disease. Food Science and Human Wellness 2015, 4, 56–65. [Google Scholar] [CrossRef]
- Doron, S.; Snydman, D. Risk and Safety of Probiotics. Clin. Infect. Dis. 2015, 60, S129–S134. [Google Scholar] [CrossRef] [PubMed]
- Daliri, E.; Lee, B.; Oh, D. Safety of Probiotics in Health and Disease. In The Role of Functional Food Security in Global Health; Watson, R., Singh, R., Takahashi, T., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 603–622. [Google Scholar]
- Jiménez-Pranteda, M.L.; Pérez-Davó, A.; Monteoliva-Sánchez, M.; Ramos-Cormenzana, A.; Aguilera, M. Food Omics Validation: Towards Understanding Key Features for Gut Microbiota, Probiotics and Human Health. Food Anal. Methods 2015, 8, 272–289. [Google Scholar] [CrossRef]
- Ouwehand, A. A review of dose-responses of probiotics in human studies. Benef. Microbes 2017, 8, 143–151. [Google Scholar] [CrossRef]
- Forssten, S.; Ouwehand, A. Dose-Response Recovery of Probiotic Strains in Simulated Gastro-Intestinal Passage. Microorganisms 2020, 8, 112. [Google Scholar] [CrossRef]
- Pencina, M.; Louzao, D.; McCourt, B.; Adams, M.; Tayyabkhan, R.; Ronco, P.; Peterson, E. Supporting open access to clinical trial data for researchers: The Duke Clinical Research Institute–Bristol-Myers Squibb Supporting Open Access to Researchers Initiative. Am. Heart J. 2016, 172, 64–69. [Google Scholar] [CrossRef]
- Belizário, J.; Napolitano, M. Human microbiomes and their roles in dysbiosis, common diseases, and novel therapeutic approaches. Front. Microbiol. 2015, 6, 1050. [Google Scholar] [CrossRef]
- Lazar, V.; Ditu, L.; Pircalabioru, G.; Picu, A.; Petcu, L.; Cucu, N.; Chifiriuc, M. Gut Microbiota, Host Organism, and Diet Trialogue in Diabetes and Obesity. Front. Nutr. 2019, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Tenorio-Jiménez, C.; Martínez-Ramírez, M.; Gil, Á.; Gómez-Llorente, C. Effects of Probiotics on Metabolic Syndrome: A Systematic Review of Randomized Clinical Trials. Nutrients 2020, 12, 124. [Google Scholar] [CrossRef] [PubMed]
- Katsi, V.; Didagelos, M.; Skevofilax, S.; Armenis, I.; Kartalis, A.; Vlachopoulos, C.; Karvounis, H.; Tousoulis, D. GUT Microbiome-GUT Dysbiosis-Arterial Hypertension: New Horizons. Curr. Hypertens. Rev. 2019, 15, 40–46. [Google Scholar] [CrossRef]
- Pascale, A.; Marchesi, N.; Marelli, C.; Coppola, A.; Luzi, L.; Govoni, S.; Giustina, A.; Gazzaruso, C. Microbiota and metabolic diseases. Endocrine 2018, 61, 357–371. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Tripathi, P. Gut microbiome and type-2 diabetes: Where we are and where to go? J. Nutr. Biochem. 2019, 63, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Fontané, L.; Benaiges, D.; Goday, A.; Llauradó, G.; Pedro-Botet, J. Influence of the microbiota and probiotics in obesity. Clin. Investig. Arterioscler. 2018, 30, 271–279. [Google Scholar] [CrossRef]
- Dos Santos, C.; Ferreira, K.; Midori, P.; Fábio, L.; Darrieux, M.; Manzano, T. Childhood Obesity and Firmicutes/Bacteroidetes Ratio in the Gut Microbiota: A Systematic Review. Child. Obes. 2018, 14, 501–509. [Google Scholar] [CrossRef]
- Maruvada, P.; Leone, V.; Kaplan, L.; Chang, E. The Human Microbiome and Obesity: Moving beyond Associations. Cell Host Microbe 2017, 22, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Vallianou, N.; Stratigou, T.; Christodoulatos, G.; Dalamaga, M. Understanding the Role of the Gut Microbiome and Microbial Metabolites in Obesity and Obesity-Associated Metabolic Disorders: Current Evidence and Perspectives. Curr. Obes. Rep. 2019, 8, 317–332. [Google Scholar] [CrossRef] [PubMed]
- Barrea, L.; Muscogiuri, G.; Annunziata, G.; Laudisio, D.; Pugliese, G.; Salzano, C.; Colao, A.; Savastano, S. From gut microbiota dysfunction to obesity: Could short-chain fatty acids stop this dangerous course? Hormones 2019, 18, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Darbre, P.D. Endocrine Disruptors and Obesity. Curr. Obes. Rep. 2017, 6, 18–27. [Google Scholar] [CrossRef]
- Lai, K.; Ng, A.; Wan, H.; Wong, A.; Leung, C.; Li, R.; Wong, C. Dietary Exposure to the Environmental Chemical, PFOS on the Diversity of Gut Microbiota, Associated With the Development of Metabolic Syndrome. Front. Microbiol. 2018, 9, 2552. [Google Scholar] [CrossRef] [PubMed]
- Di Ciaula, A.; Portincasa, P. Diet and Contaminants: Driving the Rise to Obesity Epidemics? Curr. Med. Chem. 2019, 26, 3471–3482. [Google Scholar] [CrossRef] [PubMed]
- Gálvez-Ontiveros, Y.; Páez, S.; Monteagudo-Sanchez, C.; Rivas, A. Endocrine disruptors in food: Impact on gut microbiota and metabolic diseases: A systematic review. Nutrients 2020, 12, 1158. [Google Scholar] [CrossRef] [PubMed]
- Andújar, N.; Gálvez-Ontiveros, Y.; Zafra-Gómez, A.; Rodrigo, L.; Álvarez-Cubero, M.; Aguilera, M.; Monteagudo, C.; Rivas, A. Bisphenol A Analogues in Food and Their Hormonal and Obesogenic Effects: A Review. Nutrients 2019, 11, 2136. [Google Scholar] [CrossRef] [PubMed]
- Boto-Ordóñez, M.; Urpi-Sarda, M.; Queipo-Ortuño, M.; Tulipani, S.; Tinahones, F.; Andres-Lacueva, C. High levels of Bifidobacteria are associated with increased levels of anthocyanin microbial metabolites: A randomized clinical trial. Food Funct. 2014, 5, 1932–1938. [Google Scholar] [CrossRef]
- Santocchi, E.; Guiducci, L.; Fulceri, F.; Billeci, L.; Buzzigoli, E.; Apicella, F.; Calderoni, S.; Grossi, E.; Morales, M.; Muratori, F. Gut to brain interaction in Autism Spectrum Disorders: A randomized controlled trial on the role of probiotics on clinical, biochemical and neurophysiological parameters. BMC Psychiatry 2016, 16, 183. [Google Scholar] [CrossRef]
- Trinder, M.; Bisanz, J.; Burton, J.; Reid, G. Probiotic lactobacilli: A potential prophylactic treatment for reducing pesticide absorption in humans and wildlife. Benef. Microbes 2015, 6, 841–847. [Google Scholar] [CrossRef]
- O’Toole, P.; Marchesi, J.; Hill, C. Next-generation probiotics: The spectrum from probiotics to live biotherapeutics. Nat. Microbiol. 2017, 2, 17057. [Google Scholar] [CrossRef]
- Bansal, P.; Kumar, R.; Singh, J.; Dhanda, S. Next generation sequencing, biochemical characterization, metabolic pathway analysis of novel probiotic Pediococcus acidilactici NCDC 252 and it’s evolutionary relationship with other lactic acid bacteria. Mol. Biol. Rep. 2019, 46, 5883–5895. [Google Scholar] [CrossRef]
- Canese, K. An Updated PubMed Is on Its Way. NLM Tech. Bull. 2019, 427, e1. [Google Scholar]
- Higgins, J.P.; Altman, D.G.; Sterne, J. Chapter 8. In Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0; Higgins, J.P., Green, S., Eds.; The Cochrane Collaboration: London, UK, 2011; Available online: www.cochrane-handbook.org (accessed on 16 April 2020).
- Watson, P.; Petrie, A. Method agreement analysis: A review of correct methodology. Theriogenology 2010, 73, 1167–1179. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Alisi, A.; Bedogni, G.; Baviera, G.; Giorgio, V.; Porro, E.; Paris, C.; Giammaria, P.; Reali, L.; Anania, F.; Nobili, V. Randomised clinical trial: The beneficial effects of VSL#3 in obese children with non-alcoholic steatohepatitis. Aliment. Pharmacol. Ther. 2014, 39, 1276–1285. [Google Scholar] [CrossRef] [PubMed]
- Caimari, A.; del Bas, J.; Boqué, N.; Crescenti, A.; Puiggròs, F.; Chenoll, E.; Martorell, P.; Ramón, D.; Genovés, S.; Arolaad, L. Heat-killed Bifidobacterium animalis subsp. Lactis CECT 8145 increases lean mass and ameliorates metabolic syndrome in cafeteria-fed obese rats. J. Funct. Foods 2017, 38, 251–263. [Google Scholar] [CrossRef]
- Li, C.; Ding, Q.; Nie, S.; Zhang, Y.; Xiong, T.; Xie, M. Carrot Juice Fermented with Lactobacillus plantarum NCU116 Ameliorates Type 2 Diabetes in Rats. J. Agric. Food Chem. 2014, 62, 11884–11891. [Google Scholar] [CrossRef]
- Cano, P.; Santacruz, A.; Trejo, F.; Sanz, Y. Bifidobacterium CECT 7765 improves metabolic and immunological alterations associated with obesity in high-fat diet-fed mice. Obesity 2013, 21, 2310–2321. [Google Scholar] [CrossRef]
- Eaimworawuthikul, S.; Tunapong, W.; Chunchai, T.; Yasom, S.; Wanchai, K.; Suntornsaratoon, P.; Charoenphandhu, N.; Thiennimitr, P.; Chattipakorn, N.; Chattipakorn, S. Effects of probiotics, prebiotics or synbiotics on jawbone in obese-insulin resistant rats. Eur. J. Nutr. 2018, 58, 2801–2810. [Google Scholar] [CrossRef]
- Cerdó, T.; García-Santos, J.; Bermúdez, M.G.; Campoy, C. The Role of Probiotics and Prebiotics in the Prevention and Treatment of Obesity. Nutrients 2019, 11, 635. [Google Scholar] [CrossRef]
- Sanz, Y.; Santacruz, A.; Gauffin, P. Gut microbiota in obesity and metabolic disorders. Proc. Nutr. Soc. 2010, 69, 434–441. [Google Scholar] [CrossRef]
- Koutnikova, H.; Genser, B.; Monteiro-Sepulveda, M.; Faurie, J.M.; Rizkalla, S.; Schrezenmeir, J.; Clément, K. Impact of bacterial probiotics on obesity, diabetes and non-alcoholic fatty liver disease related variables: A systematic review and meta-analysis of randomised controlled trials. BMJ Open 2019, 9, e017995. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.; Pan, D.; Chang, B.; Jiang, M.; Sang, L. Probiotic mixture VSL#3: An overview of basic and clinical studies in chronic diseases. World J. Clin. Cases 2020, 8, 1361–1384. [Google Scholar] [CrossRef] [PubMed]
- Łukasik, J.; Szajewska, H. Effect of a multispecies probiotic on reducing the incidence of antibiotic-associated diarrhoea in children: A protocol for a randomised controlled trial. BMJ Open 2018, 8, e021214. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.; Alderete, T.; Martin, A.; Geary, B.; Hwang, D.; Palmer, S.; Goran, M. Probiotic supplementation increases obesity with no detectable effects on liver fat or gut microbiota in obese Hispanic adolescents: A 16-week, randomized, placebo-controlled trial. Pediatr. Obes. 2018, 13, 705–714. [Google Scholar] [CrossRef]
- Mobini, R.; Tremaroli, V.; Ståhlman, M.; Karlsson, F.; Levin, M.; Ljungberg, M.; Sohlin, M.; Bertéus, H.; Perkins, R.; Bäckhed, F. Metabolic effects of Lactobacillus reuteri DSM 17938 in people with type 2 diabetes: A randomized controlled trial. Diabetes Obes. Metab. 2017, 19, 579–589. [Google Scholar] [CrossRef]
- Simon, M.; Strassburger, K.; Nowotny, B.; Kolb, H.; Nowotny, P.; Burkart, V.; Zivehe, F.; Hwang, J.; Stehle, P.; Pacini, G.; et al. Intake of Lactobacillus reuteri. Improves Incretin and Insulin Secretion in Glucose-Tolerant Humans: A Proof of Concept. Diabetes Care 2015, 38, 1827–1834. [Google Scholar] [CrossRef]
- Sánchez, M.; Darimont, C.; Panahi, S.; Drapeau, V.; Marette, A.; Taylor, V.; Doré, J.; Tremblay, A. Effects of a Diet-Based Weight-Reducing Program with Probiotic Supplementation on Satiety Efficiency, Eating Behaviour Traits, and Psychosocial Behaviours in Obese Individuals. Nutrients 2017, 9, 284. [Google Scholar] [CrossRef]
- Rajkumar, H.; Mahmood, N.; Kumar, M.; Varikuti, S.; Challa, H.; Myakala, S. Effect of Probiotic (VSL#3) and Omega-3 on Lipid Profile, Insulin Sensitivity, Inflammatory Markers, and Gut Colonization in Overweight Adults: A Randomized, Controlled Trial. Mediat. Inflamm. 2014, 2014, 348959. [Google Scholar] [CrossRef]
- Brahe, L.; Le Chatelier, E.; Prifti, E.; Pons, N.; Kennedy, S.; Blædel, T.; Håkansson, J.; Kastrup, T.; Hansen, T.; Pedersen, O.; et al. Dietary modulation of the gut microbiota—A randomised controlled trial in obese postmenopausal women. Br. J. Nutr. 2015, 114, 406–417. [Google Scholar] [CrossRef]
- Bomhof, M.; Saha, D.; Reid, D.; Paul, H.; Reimer, R. Combined effects of oligofructose and Bifidobacterium animalis on gut microbiota and glycemia in obese rats. Obesity 2013, 22, 763–771. [Google Scholar] [CrossRef]
- Wang, G.; Li, X.; Zhao, J.; Zhang, H.; Chen, W. Lactobacillus casei CCFM419 attenuates type 2 diabetes via a gut microbiota dependent mechanism. Food Funct. 2017, 8, 3155–3164. [Google Scholar] [CrossRef] [PubMed]
- Natividad, J.; Lamas, B.; Pham, H.; Michel, M.; Rainteau, D.; Bridonneau, C.; da Costa, G.; van Hylckama, J.; Sovran, B.; Chamignon, C.; et al. Bilophila wadsworthia aggravates high fat diet induced metabolic dysfunctions in mice. Nat. Commun. 2018, 9, 2802. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kirkland, R.; Grunewald, Z.; Sun, Q.; Wicker, L.; de La Serre, C. Beneficial Effects of Non-Encapsulated or Encapsulated Probiotic Supplementation on Microbiota Composition, Intestinal Barrier Functions, Inflammatory Profiles, and Glucose Tolerance in High Fat Fed Rats. Nutrients 2019, 11, 1975. [Google Scholar] [CrossRef] [PubMed]
- Falcinelli, S.; Rodiles, A.; Hatef, A.; Picchietti, S.; Cossignani, L.; Merrifield, D.; Unniappan, S.; Carnevali, O. Dietary lipid content reorganizes gut microbiota and probiotic L. rhamnosus attenuates obesity and enhances catabolic hormonal milieu in zebrafish. Sci. Rep. 2017, 7, 5512. [Google Scholar] [CrossRef]
- Park, D.; Ahn, Y.; Park, S.; Huh, C.; Yoo, S.; Yu, R.; Sung, M.; McGregor, R.; Choi, M. Supplementation of Lactobacillus curvatus HY7601 and Lactobacillus plantarum KY1032 in Diet-Induced Obese Mice Is Associated with Gut Microbial Changes and Reduction in Obesity. PLoS ONE 2013, 8, e59470. [Google Scholar] [CrossRef]
- Wang, T.; Yan, H.; Lu, Y.; Li, X.; Wang, X.; Shan, Y.; Yi, Y.; Liu, B.; Zhou, Y.; Lü, X. Anti-obesity effect of Lactobacillus rhamnosus LS-8 and Lactobacillus crustorum MN047 on high-fat and high-fructose diet mice base on inflammatory response alleviation and gut microbiota regulation. Eur. J. Nutr. 2019. [Google Scholar] [CrossRef]
- Cano, P.; Santacruz, A.; Moya, Á.; Sanz, Y. Bacteroides uniformis CECT 7771 Ameliorates Metabolic and Immunological Dysfunction in Mice with High-Fat-Diet Induced Obesity. PLoS ONE 2012, 7, e41079. [Google Scholar] [CrossRef]
- Gómez Del Pulgar, E.M.; Benítez-Páez, A.; Sanz, Y. Safety Assessment of Bacteroides uniformis CECT 7771, a Symbiont of the Gut Microbiota in Infants. Nutrients 2020, 12, 551. [Google Scholar] [CrossRef]
- Legrand, R.; Lucas, N.; Dominique, M.; Azhar, S.; Deroissart, C.; Le Solliec, M.; Rondeaux, J.; Nobis, S.; Guérin, C.; Léon, F.; et al. Commensal Hafnia alvei strain reduces food intake and fat mass in obese mice—A new potential probiotic for appetite and body weight management. Int. J. Obes. 2020, 44, 1041–1051. [Google Scholar] [CrossRef]
- Iqbal, U.; Westfall, S.; Prakash, S. Novel microencapsulated probiotic blend for use in metabolic syndrome: Design and in-vivo analysis. Artif. Cells Nanomed. Biotechnol. 2018, 46, S116–S124. [Google Scholar] [CrossRef]
- Sudha, M.; Ahire, J.; Jayanthi, N.; Tripathi, A.; Nanal, S. Effect of multi-strain probiotic (UB0316) in weight management in overweight/obese adults: A 12-week double blind, randomised, placebo-controlled study. Benef. Microbes 2019, 10, 855–866. [Google Scholar] [CrossRef]
- Tao, Y.; Gu, Y.; Mao, X.; Zhang, L.; Pei, Y. Effects of probiotics on type II diabetes mellitus: A meta-analysis. J. Transl. Med. 2020, 18, 30. [Google Scholar] [CrossRef] [PubMed]
- Sáez-Lara, M.; Robles-Sanchez, C.; Ruiz-Ojeda, F.; Plaza-Diaz, J.; Gil, A. Effects of Probiotics and Synbiotics on Obesity, Insulin Resistance Syndrome, Type 2 Diabetes and Non-Alcoholic Fatty Liver Disease: A Review of Human Clinical Trials. Int. J. Mol. Sci. 2016, 17, 928. [Google Scholar] [CrossRef] [PubMed]
- Jeżewska-Frąckowiak, J.; Seroczyńska, K.; Banaszczyk, J.; Jedrzejczak, G.; Żylicz-Stachula, A.; Skowron, P.M. The promises and risks of probiotic Bacillus species. Acta Biochim. Pol. 2018, 65, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Onderdonk, A. Probiotics for Womenʼs Health. J. Clin. Gastroenterol. 2006, 40, 256–259. [Google Scholar] [CrossRef] [PubMed]
- Minami, J.; Iwabuchi, N.; Tanaka, M.; Yamauchi, K.; Xiao, J.Z.; Abe, F.; Sakane, N. Effects of Bifidobacterium breve B-3 on body fat reductions in pre-obese adults: A randomized, double-blind, placebo-controlled trial. Biosci. Microbiota Food Health 2018, 37, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Kothari, D.; Patel, S.; Kim, S. Probiotic supplements might not be universally-effective and safe: A review. Biomed. Pharmacother. 2019, 111, 537–547. [Google Scholar] [CrossRef]
- EFSA NDA Panel on Dietetic Products, Nutrition and Allergies. General scientific guidance for stakeholders on health claim applications. EFSA J. 2016, 14, 4367. [Google Scholar]
- Velayudham, A.; Dolganiuc, A.; Ellis, M.; Petrasek, J.; Kodys, K.; Mandrekar, P.; Szabo, G. VSL#3 probiotic treatment attenuates fibrosis without changes in steatohepatitis in a diet-induced nonalcoholic steatohepatitis model in mice. Hepatology 2009, 49, 989–997. [Google Scholar] [CrossRef]
- Mencarelli, A.; Distrutti, E.; Renga, B.; D’Amore, C.; Cipriani, S.; Palladino, G.; Donini, A.; Ricci, P.; Fiorucci, S. Probiotics modulate intestinal expression of nuclear receptor and provide counter-regulatory signals to inflammation-driven adipose tissue activation. PLoS ONE 2011, 6, e22978. [Google Scholar] [CrossRef]
- Ma, X.; Hua, J.; Li, Z. Probiotics improve high fat diet induced hepatic steatosis and insulin resistance by increasing hepatic NKT cells. J. Hepatol. 2008, 49, 821–830. [Google Scholar] [CrossRef] [PubMed]
- Memarrast, F.; Ghafouri-Fard, S.; Kolivand, S.; Jafary-Nodooshan, S.; Neyazi, N.; Sadroddiny, E.; Motevaseli, E. Comparative evaluation of probiotics effects on plasma glucose, lipid, and insulin levels in streptozotocin-induced diabetic rats. Diabetes Metab. Res. Rev. 2017, 33. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, M.; Tsai, W.; Jheng, Y.; Su, S.; Wang, S.; Lin, C.; Chen, Y.; Chang, W. The beneficial effects of Lactobacillus reuteri ADR-1 or ADR-3 consumption on type 2 diabetes mellitus: A randomized, double-blinded, placebo-controlled trial. Sci. Rep. 2018, 8, 16791. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.; Zhao, F.; Liu, W.; Lv, R.; Khine, W.; Han, J.; Sun, Z.; Lee, Y.; Zhang, H. Probiotic-directed modulation of gut microbiota is basal microbiome dependent. Gut Microbes 2020, 1–20. [Google Scholar] [CrossRef]
- Park, D.; Ahn, Y.; Huh, C.; Jeon, S.; Choi, M. The inhibitory effect of Lactobacillus plantarum KY1032 cell extract on the adipogenesis of 3T3-L1 Cells. J. Med. Food 2011, 14, 670–675. [Google Scholar] [CrossRef]
- Isolauri, E.; Sütas, Y.; Kankaanpää, P.; Arvilommi, H.; Salminen, S. Probiotics: Effects on immunity. Am. J. Clin. Nutr. 2001, 73, 444s–450s. [Google Scholar] [CrossRef]
- Kondo, S.; Xiao, J.; Satoh, T.; Odamaki, T.; Takahashi, S.; Sugahara, H.; Yaeshima, T.; Iwatsuki, K.; Kamei, A.; Abe, K. Antiobesity effects of bifidobacterium breve strain B-3 supplementation in a mouse model with high-fat diet-induced obesity. Biosci. Biotechnol. Biochem. 2010, 74, 1656–1661. [Google Scholar] [CrossRef]
- Delzenne, N.M.; Neyrinck, A.M.; Backhed, F.; Cani, P.D. Targeting gut microbiota in obesity: Effects of prebiotics and probiotics. Nat. Rev. Endocrinol. 2011, 7, 639–646. [Google Scholar] [CrossRef]
- Chen, J.; Wang, R.; Li, X.F.; Wang, R.L. Bifidobacterium adolescentis supplementation ameliorates visceral fat accumulation and insulin sensitivity in an experimental model of the metabolic syndrome. Br. J. Nutr. 2012, 107, 1429–1434. [Google Scholar] [CrossRef]
- Al-muzafar, H.; Amin, K. Probiotic mixture improves fatty liver disease by virtue of its action on lipid profiles, leptin, and inflammatory biomarkers. BMC Complement. Altern. Med. 2017, 17, 43. [Google Scholar] [CrossRef]
- Shin, N.R.; Lee, J.C.; Lee, H.; Kim, M.; Whon, T.; Lee, M.; Bae, J. An increase in the Akkermansia spp. population induced by metformin treatment improves glucose homeostasis in diet-induced obese mice. Gut 2014, 63, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Taverniti, V.; Koirala, R.; Dalla Via, A.; Gargari, G.; Leonardis, E.; Arioli, S.; Guglielmetti, S. Effect of Cell Concentration on the Persistence in the Human Intestine of Four Probiotic Strains Administered through a Multispecies Formulation. Nutrients 2019, 11, 285. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.; Xu, X.; Yang, B.; Lu, J.; Zhang, S.; Liu, L.; Nassar, K.; Zhang, C.; Zhang, M.; Pang, X.; et al. Stable Colonization of Orally Administered Lactobacillus casei SY13 Alters the Gut Microbiota. Biomed. Res. Int. 2020, 2020, 5281639. [Google Scholar] [CrossRef] [PubMed]
- Lagier, J.; Dubourg, G.; Million, M.; Cadoret, F.; Bilen, M.; Fenollar, F.; Levasseur, A.; Rolain, J.; Fournier, P.; Raoult, P. Culturing the human microbiota and culturomics. Nat. Rev. Microbiol. 2018, 16, 540–550. [Google Scholar] [CrossRef]
- Costa, M.; Weese, J. Methods and basic concepts for microbiota assessment. Vet. J. 2019, 249, 10–15. [Google Scholar] [CrossRef]
- Sergeev, I.N.; Aljutaily, T.; Walton, G.; Huarte, E. Effects of Synbiotic Supplement on Human Gut Microbiota, Body Composition and Weight Loss in Obesity. Nutrients 2020, 12, 222. [Google Scholar] [CrossRef]
- Million, M.; Maraninchi, M.; Henry, M.; Armougom, F.; Richet, H.; Carrieri, P.; Valero, R.; Raccah, D.; Vialettes, B.; Raoult, D. Obesity-associated gut microbiota is enriched in Lactobacillus reuteri and depleted in Bifidobacterium animalis and Methanobrevibacter smithii. Int. J. Obes. 2012, 36, 817–825. [Google Scholar] [CrossRef]
- Goyal, A.; Nimmakayala, K.; Zonszein, J. Is There a Paradox in Obesity? Cardiol. Rev. 2014, 22, 163–170. [Google Scholar] [CrossRef]
- Hassan, A.; Din, A.U.; Zhu, Y.; Zhang, K.; Li, T.; Wang, Y.; Luo, Y.; Wang, G. Updates in understanding the hypocholesterolemia effect of probiotics on atherosclerosis. Appl. Microbiol. Biotechnol. 2019, 103, 5993–6006. [Google Scholar] [CrossRef]
- Park, Y.; Zhang, J.; Steck, S.; Fung, T.; Hazlett, L.; Han, K.; Ko, S.; Merchant, A. Obesity mediates the association between Mediterranean diet consumption and insulin resistance and inflammation in US adults. J. Nutr. 2017, 147, 563–571. [Google Scholar] [CrossRef]
- Brown, B.I. Nutritional Management of Metabolic Endotoxemia: A Clinical Review. Altern. Ther. Health Med. 2017, 23, 42–54. [Google Scholar] [PubMed]
- Sudhakar, M.; Silambanan, S.; Chandran, A.; Prabhakaran, A.; Ramakrishnan, R. C-Reactive Protein (CRP) and Leptin Receptor in Obesity: Binding of Monomeric CRP to Leptin Receptor. Front. Immunol. 2018, 9, 1167. [Google Scholar] [CrossRef] [PubMed]
- Rouhani, M.; Hadi, A.; Ghaedi, E.; Salehi, M.; Mahdavi, A.; Mohammadi, H. Do probiotics, prebiotics and synbiotics affect adiponectin and leptin in adults? A systematic review and meta-analysis of clinical trials. Clin. Nutr. 2019, 38, 2031–2037. [Google Scholar] [CrossRef] [PubMed]
- Davis, P.; Lewenstein, B.; Simon, D.; Booth, J.; Connolly, M. Open access publishing, article downloads, and citations: Randomised controlled trial. BMJ 2008, 337, a568. [Google Scholar] [CrossRef] [PubMed]
- Rocca-Serra, P.; Salek, R.; Arita, M.; Correa, E.; Dayalan, S.; Gonzalez-Beltran, A.; Ebbels, T.; Goodacre, R.; Hastings, J.; Haug, K. Data standards can boost metabolomics research, and if there is a will, there is a way. Metabolomics 2016, 12, 14. [Google Scholar] [CrossRef]
- Rijkers, G.; Bengmark, S.; Enck, P.; Haller, D.; Herz, U.; Kalliomaki, M.; Kudo, S.; Lenoir-Wijnkoop, I.; Mercenier, A.; Myllyluoma, E.; et al. Guidance for Substantiating the Evidence for Beneficial Efects of Probiotics: Current Status and Recommendations for Future Research. J. Nutr. 2010, 140, 671S–676S. [Google Scholar] [CrossRef]
- Amabebe, E.; Robert, F.; Agbalalah, T.; Orubu, E. Microbial dysbiosis-induced obesity: Role of gut microbiota in homoeostasis of energy metabolism. Br. J. Nutr. 2020, 1–11. [Google Scholar] [CrossRef]
- Sadrzadeh-Yeganeh, H.; Elmadfa, I.; Djazayery, A.; Jalali, M.; Heshmat, R.; Chamary, M. The effects of probiotic and conventional yoghurt on lipid profile in women. Br. J. Nutr. 2010, 103, 1778–1783. [Google Scholar] [CrossRef]
- Baker, J.; Al-Nakkash, L.; Herbst-Kralovetz, M. Estrogen–gut microbiome axis: Physiological and clinical implications. Maturitas 2017, 103, 45–53. [Google Scholar] [CrossRef]
- Karrer, C.; de Boer, W.; Delmaar, C.; Cai, Y.; Crépet, A.; Hungerbühler, K.; von Goetz, N. Linking Probabilistic Exposure and Pharmacokinetic Modeling To Assess the Cumulative Risk from the Bisphenols BPA, BPS, BPF, and BPAF for Europeans. Environ. Sci. Technol. 2019, 53, 9181–9191. [Google Scholar] [CrossRef]
- Velmurugan, G.; Ramprasath, T.; Gilles, M.; Swaminathan, K.; Ramasamy, S. Gut Microbiota, Endocrine-Disrupting Chemicals, and the Diabetes Epidemic. Trends Endocrinol. Metab. 2017, 28, 612–625. [Google Scholar] [CrossRef] [PubMed]
- Aziz, R.; Hegazy, S.; Yasser, R.; Rizkallah, M.; ElRakaiby, M. Drug pharmacomicrobiomics and toxicomicrobiomics: From scattered reports to systematic studies of drug–microbiome interactions. Expert Opin. Drug Metabol. Toxicol. 2018, 14, 1043–1055. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Buschmann, M.; Gilbert, J. Pharmacomicrobiomics: The Holy Grail to Variability in Drug Response? Clin. Pharmacol. Ther. 2019, 106, 317–328. [Google Scholar] [CrossRef]
- Mikołajewska, K.; Stragierowicz, J.; Gromadzińska, J. Bisphenol A—Application, sources of exposure and potential risks in infants, children and pregnant women. Int. J. Occup. Med. Environ. Health 2015, 28, 209–241. [Google Scholar] [CrossRef]
- Zhang, W.; Yin, K.; Chen, L. Bacteria-mediated bisphenol A degradation. Appl. Microbiol. Biotechnol. 2013, 97, 5681–5689. [Google Scholar] [CrossRef] [PubMed]
- Dvořák, P.; Nikel, P.; Damborský, J.; de Lorenzo, V. Bioremediation 3.0: Engineering pollutant-removing bacteria in the times of systemic biology. Biotechnol. Adv. 2017, 35, 845–866. [Google Scholar] [CrossRef] [PubMed]
- Sang, H.; Hulvey, J.; Green, R.; Xu, H.; Im, J.; Chang, T.; Jung, G. A Xenobiotic Detoxification Pathway through Transcriptional Regulation in Filamentous Fungi. Mbio 2018, 9, e00457-18. [Google Scholar] [CrossRef]
- García-González, N.; Prete, R.; Perugini, M.; Merola, C.; Battista, N.; Corsetti, A. Probiotic antigenotoxic activity as a DNA bioprotective tool: A minireview with focus on endocrine disruptors. FEMS Microbiol. Lett. 2020, 367, fnaa041. [Google Scholar] [CrossRef]
- Bonk, F.; Popp, D.; Harms, H.; Centler, F. PCR-based quantification of taxa-specific abundances in microbial communities: Quantifying and avoiding common pitfalls. J. Microbiol. Methods 2018, 153, 139–147. [Google Scholar] [CrossRef]
- Martínez, N.; Hidalgo-Cantabrana, C.; Delgado, S.; Margolles, A.; Sánchez, B. Filling the gap between collection, transport and storage of the human gut microbiota. Sci. Rep. 2019, 9, 8327. [Google Scholar] [CrossRef]
- Smith, C.; Perfetti, T.; Hayes, A.; Berry, S.C. Obesity as a Source of Endogenous Compounds Associated with Chronic Disease: A review. Toxicol. Sci. 2020, 175, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Green, M.; Arora, K.; Prakash, S. Microbial Medicine: Prebiotic and Probiotic Functional Foods to Target Obesity and Metabolic Syndrome. Int. J. Mol. Sci. 2020, 21, 2890. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Pourang, A.; Burrall, B. Open access medical journals: Benefits and challenges. Clin. Dermatol. 2019, 37, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Lin, T.; Tsai, Y.; Wu, T.; Lai, W.; Lu, C.; Lai, H. Next generation probiotics in disease amelioration. J. Food Drug Anal. 2019, 27, 615–622. [Google Scholar] [CrossRef]
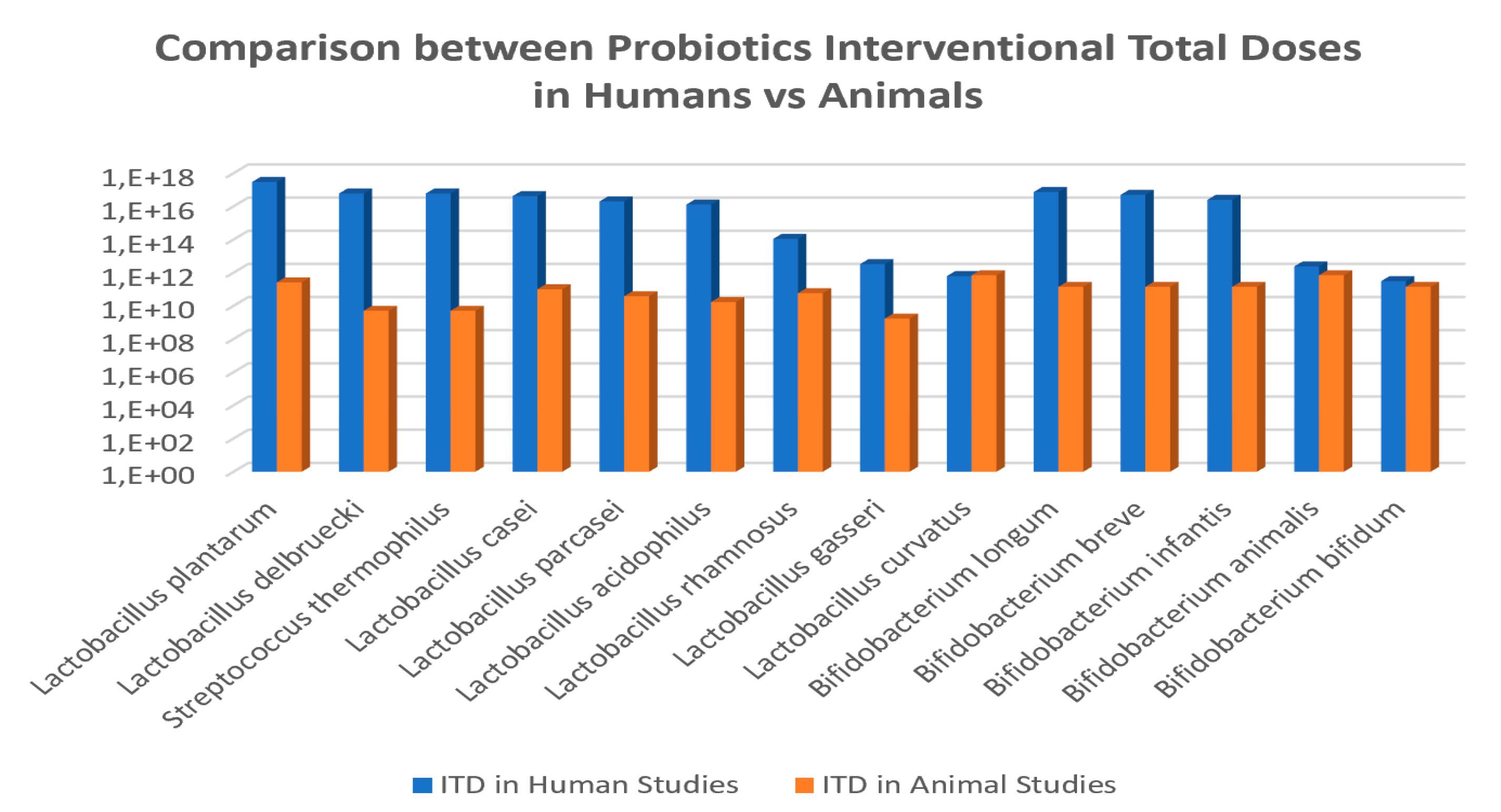

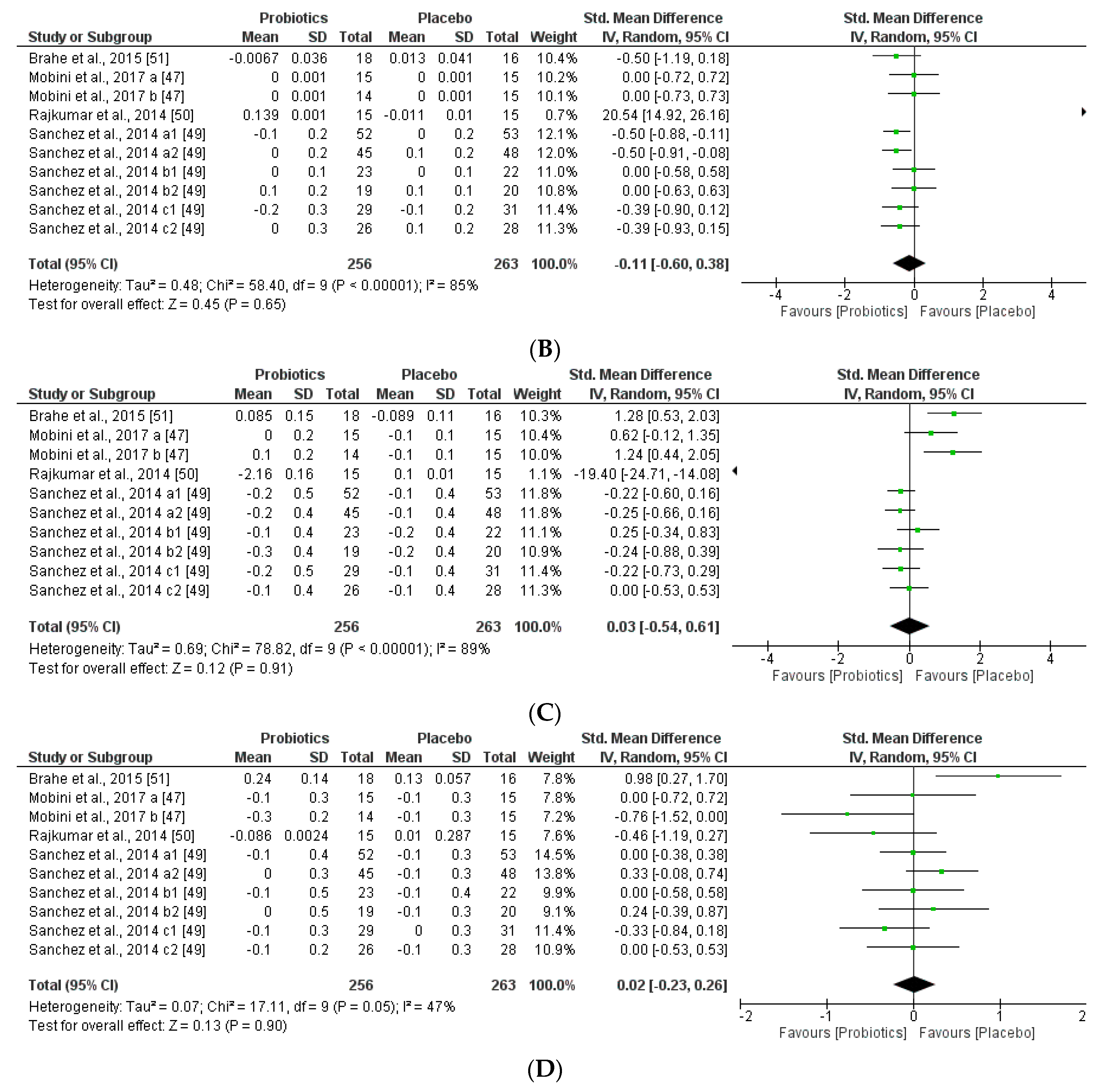
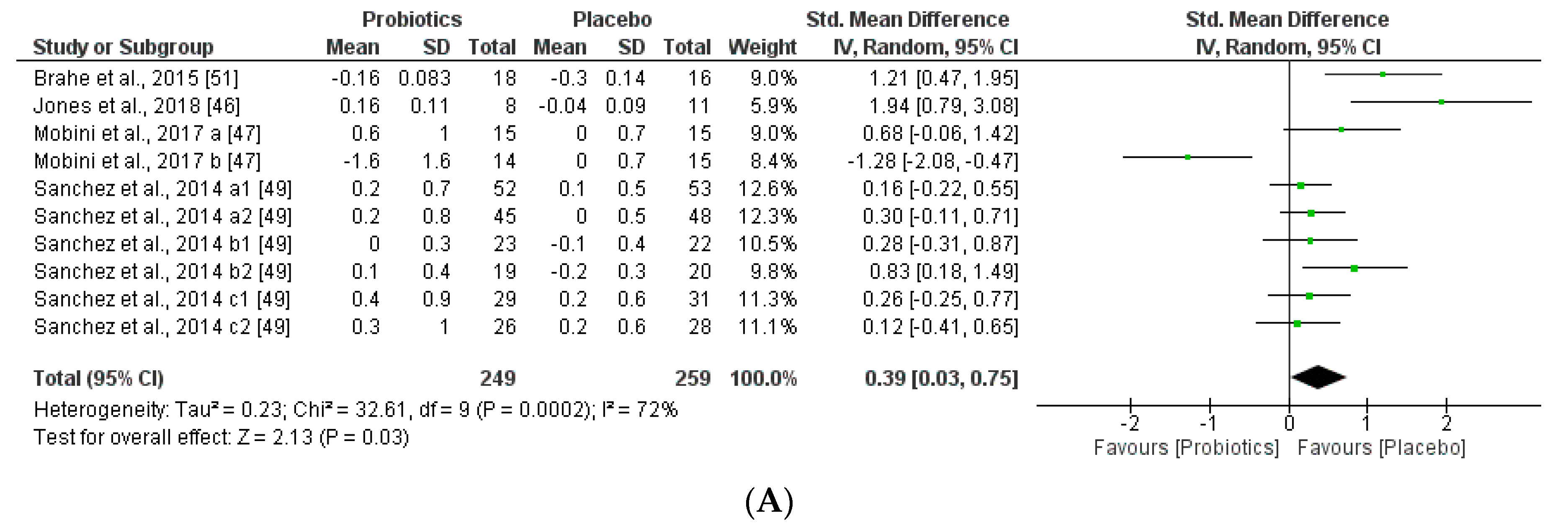
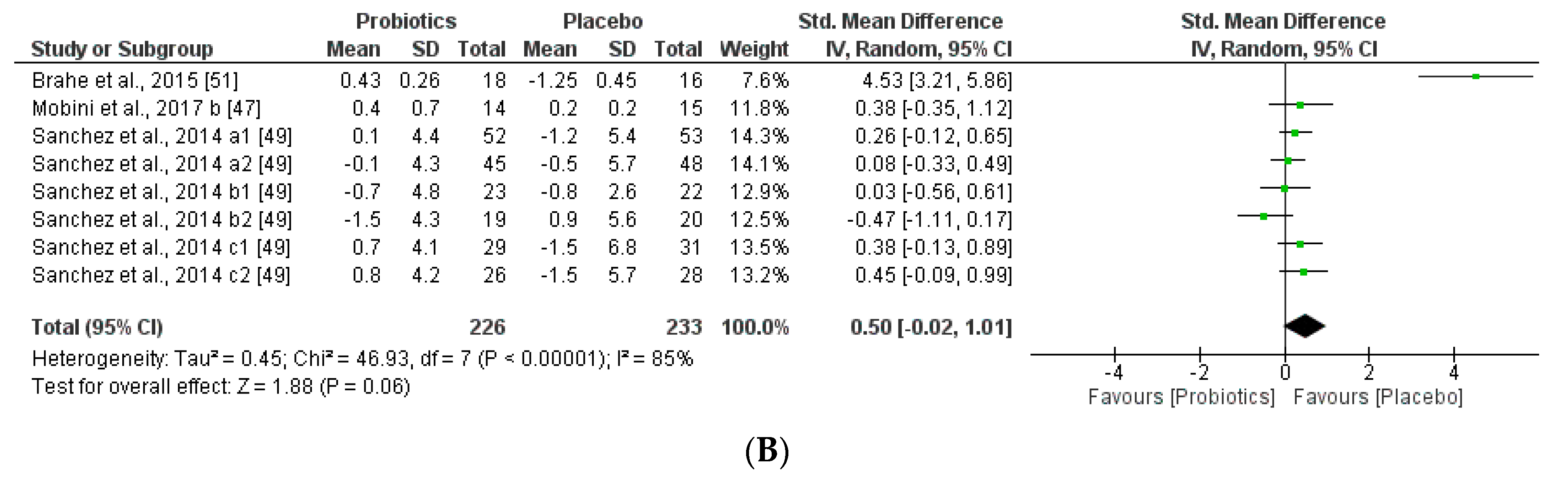
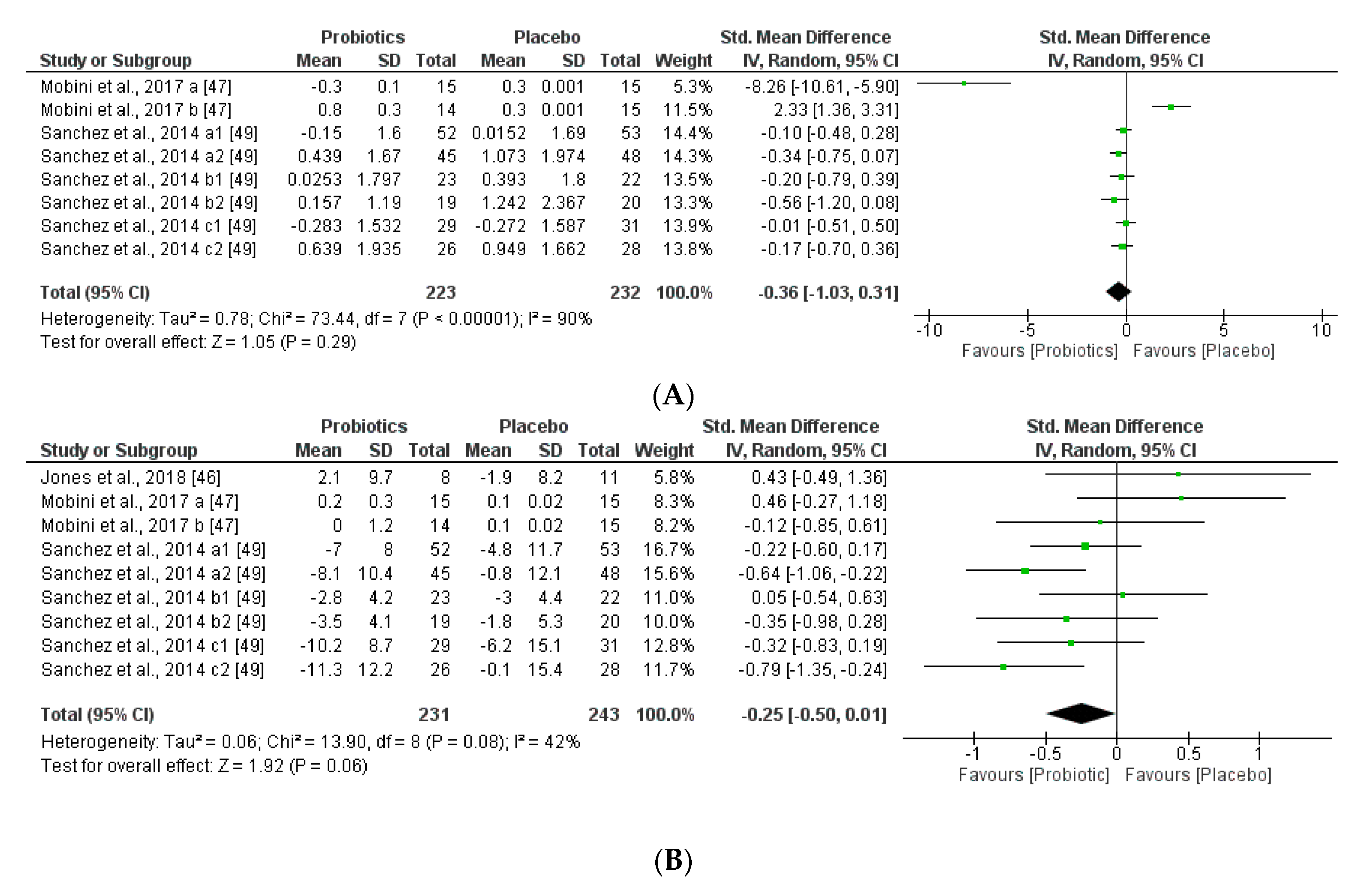
| Report Reference | Population Characteristics | Probiotic Strain | Probiotic Doses CFU/day | Pattern Administration ITD (CFU) | Microbiota Modulation Capacities | Clinical Impact and Parameter Modifications | ||
|---|---|---|---|---|---|---|---|---|
| Weight | Biomarkers | Gut Hormones | ||||||
| Jones et al. [46] | 19 Obese Latino adolescents | VSL#3* (Multispecies) | 1.35 × 1015 | 16–18 Weeks 1.5–1.7 × 1017 | No microbiota modification ↔ Firmicutes ↔ Bacteroides | ↑BMI ↑Adiposity ↑Fat mass | ↔ Glucose | ↔ Leptin, GLP-1 and GLP-2 ↔ Insulin levels |
| Rajkumar et al. [50] | 60 Overweight (30 women and 30 men) | VSL#3* (Multispecies) VSL#3*+ Omega-3 | 1.1 × 1011 | 6 weeks 4.6 × 1012 | ↑ Total bacteria ↑ Total anaerobes ↑ Lactabacillus, Bifidobacteria, Streptococcus and Bacteroides. ↓ Coliform, E.coli | ↓ BMI | ↑ HDL ↓ LDL, VLDL, TAG ↓ Glucose ↓hsCRP | ↓ Insulin levels |
| Brahe et al. [51] | 58 Obese postmenopausal women | L. paracasei subsp.paracaseiF19 | 9.4 × 1010 | 6 weeks 4 × 1012 | ↑Eubacterium rectaleand Ruminococcus torques | ↔ BMI | ↔ LDL, VLDL, TAG, Cholesterol, ↑hsCRP | ↔ ISI↔ Leptin |
| Simon et al. [48] | 10 Obese and 11 lean, all glucose-tolerant | Lactobacillus reuteri SD5865 | 2 × 1010 | 4 weeks 5.6 × 1011 | No microbiota modification ↔ Total bacterial ↔Enterobacteria ↔Lactobacilli, ↑Lactobacillus reuteri | ↑ BMI obese, ↔ Ectopic fat | ↔ Glucose | ↑ Insulin levels and C-peptide secretion ↑ GLP-1 and GLP-2 |
| Mobini et al. [47] | 46 (11 women and 36 men) Obese/diabetes | Lactobacillus reuteri DSM 17938 | Group 1: 1 × 108 | 12 weeks 8.4 × 109 | No microbiota modification Euryarcheota was initially elevated ↑Methanobacteria | ↑ BMI ↑ Weight | ↔ LDL, VLDL, TAG and Total Cholesterol | ↔ Leptin levels. |
| Group 2: 1 × 1010 | 8.4 × 1011 | No microbiota modification | ↔ BMI ↔ Weight | ↔Lipid metabolism | ↔ Leptin levels. ↑ ISI | |||
| Sánchez et al. [49] | 125 Healthy overweight men and women | Lactobacillus rhamnosus CGMCC1.3724 | 3.2 × 108 | 12 weeks 2.7 × 1010 24 weeks 5.4 × 1010 | ↓Subdoligranulum sp. in women No microbiota modification in men | ↓Weight in women ↔Weight in men | ↔Total Cholesterol | ↓Leptin levels in women. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Moreno, A.; Suárez, A.; Avanzi, C.; Monteoliva-Sánchez, M.; Aguilera, M. Probiotic Strains and Intervention Total Doses for Modulating Obesity-Related Microbiota Dysbiosis: A Systematic Review and Meta-analysis. Nutrients 2020, 12, 1921. https://doi.org/10.3390/nu12071921
López-Moreno A, Suárez A, Avanzi C, Monteoliva-Sánchez M, Aguilera M. Probiotic Strains and Intervention Total Doses for Modulating Obesity-Related Microbiota Dysbiosis: A Systematic Review and Meta-analysis. Nutrients. 2020; 12(7):1921. https://doi.org/10.3390/nu12071921
Chicago/Turabian StyleLópez-Moreno, Ana, Antonio Suárez, Camila Avanzi, Mercedes Monteoliva-Sánchez, and Margarita Aguilera. 2020. "Probiotic Strains and Intervention Total Doses for Modulating Obesity-Related Microbiota Dysbiosis: A Systematic Review and Meta-analysis" Nutrients 12, no. 7: 1921. https://doi.org/10.3390/nu12071921
APA StyleLópez-Moreno, A., Suárez, A., Avanzi, C., Monteoliva-Sánchez, M., & Aguilera, M. (2020). Probiotic Strains and Intervention Total Doses for Modulating Obesity-Related Microbiota Dysbiosis: A Systematic Review and Meta-analysis. Nutrients, 12(7), 1921. https://doi.org/10.3390/nu12071921







