Acute Effects of Beetroot Juice Supplements on Resistance Training: A Randomized Double-Blind Crossover
Abstract
1. Introduction
2. Material and Methods
2.1. Participants
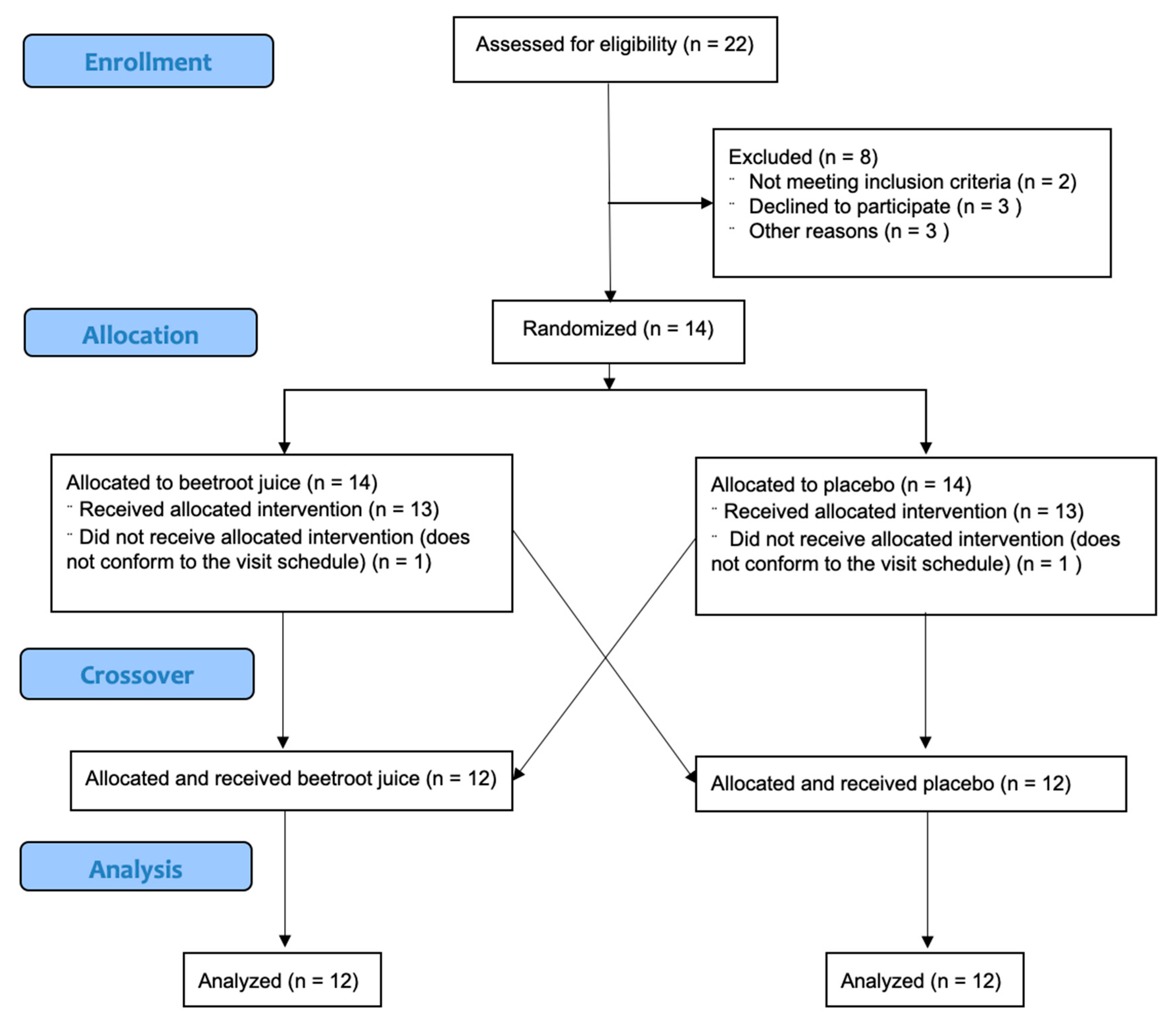
2.2. Design
2.3. Familiarization Protocol and One-Repetition Maximum Testing
2.4. Supplement Protocol
2.5. Exercise Protocol and Movement Velocity Measurement
2.6. Lactate Measurement and Ratings of Perceived Exertion
2.7. Statistical Analysis
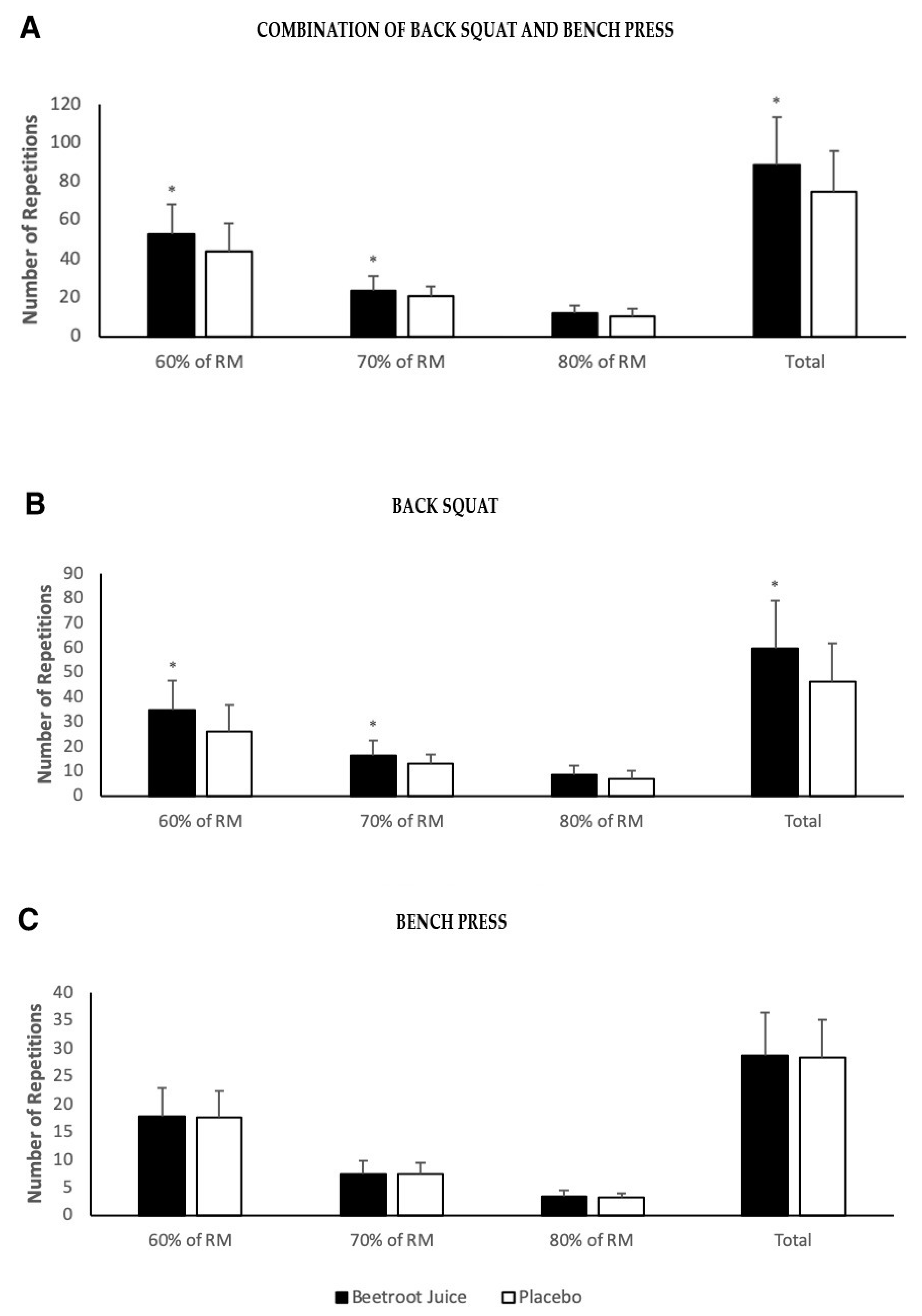
3. Results
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Domínguez, R.; Cuenca, E.; Maté-Muñoz, J.; García-Fernández, P.; Serra-Paya, N.; Estevan, M.; Herreros, P.; Garnacho-Castaño, M. Effects of beetroot juice supplementation on cardiorespiratory endurance in athletes: A systematic review. Nutrients 2017, 9, 43. [Google Scholar] [CrossRef]
- Vanhatalo, A.; Fulford, J.; Bailey, S.J.; Blackwell, J.R.; Winyard, P.G.; Jones, A.M. Dietary nitrate reduces muscle metabolic perturbation and improves exercise tolerance in hypoxia. J. Physiol. 2011, 589, 5517–5528. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O.; Weitzberg, E.; Cole, J.A.; Benjamin, N. Nitrate, bacteria and human health. Nat. Rev. Microbiol. 2004, 2, 593–602. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O.; Govoni, M. Inorganic nitrate is a possible source for systemic generation of nitric oxide. Redox. Biol. 2004, 37, 395–400. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Weitzberg, E.; Gladwin, M.T. The nitrate–nitrite–nitric oxide pathway in physiology and therapeutics. Nat. Rev. Drug Discov. 2008, 7, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Lansley, K.E.; Winyard, P.G.; Bailey, S.J.; Vanhatalo, A.; Wilkerson, D.P.; Blackwell, J.R.; Gilchrist, M.; Benjamin, N.; Jones, A.M. Acute dietary nitrate supplementation improves cycling time trial performance. Med. Sci. Sports Exerc. 2011, 43, 1125–1131. [Google Scholar] [CrossRef] [PubMed]
- Modin, A.; Björne, H.; Herulf, M.; Alving, K.; Weitzberg, E.; Lundberg, J.O. Nitrite-derived nitric oxide: A possible mediator of “acidic-metabolic vasodilation”. Acta. Physiol. 2001, 171, 9–16. [Google Scholar] [CrossRef]
- Ferguson, S.K.; Hirai, D.M.; Copp, S.W.; Holdsworth, C.T.; Allen, J.D.; Jones, A.M.; Musch, T.I.; Poole, D.C. Impact of dietary nitrate supplementation via beetroot juice on exercising muscle vascular control in rats. J. Physiol. 2012, 591, 547–557. [Google Scholar] [CrossRef]
- Wylie, L.J.; Bailey, S.J.; Kelly, J.; Blackwell, J.R.; Vanhatalo, A.; Jones, A.M. Influence of beetroot juice supplementation on intermittent exercise performance. Eur. J. Appl. Physiol. 2015, 116, 415–425. [Google Scholar] [CrossRef]
- Siervo, M.; Lara, J.; Ogbonmwan, I.; Mathers, J.C. Inorganic nitrate and beetroot juice supplementation reduces blood pressure in adults: A systematic review and meta-analysis. J. Nutr. 2013, 143, 818–826. [Google Scholar] [CrossRef]
- Muggeridge, D.J.; Howe, C.C.; Spendiff, O.; Pedlar, C.; James, P.E.; Easton, C. A single dose of beetroot juice enhances cycling performance in simulated altitude. Med. Sci. Sports Exerc. 2014, 46, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Cermak, N.M.; Gibala, M.J.; van Loon, L.J.C. Nitrate supplementation’s improvement of 10-km time-trial performance in trained cyclists. Int. J. Sport Nutr. Exerc. Metab. 2012, 22, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.J.; Fulford, J.; Vanhatalo, A.; Winyard, P.G.; Blackwell, J.R.; DiMenna, F.J.; Wilkerson, D.P.; Benjamin, N.; Jones, A.M. Dietary nitrate supplementation enhances muscle contractile efficiency during knee-extensor exercise in humans. J. Appl. Physiol. 2010, 109, 135–148. [Google Scholar] [CrossRef]
- Mosher, S.L.; Sparks, S.A.; Williams, E.L.; Bentley, D.J.; Mc Naughton, L.R. Ingestion of a nitric oxide enhancing supplement improves resistance exercise performance. J. Strength Cond. Res. 2016, 30, 3520–3524. [Google Scholar] [CrossRef] [PubMed]
- Williams, T.D.; Martin, M.P.; Mintz, J.A.; Rogers, R.R.; Ballmann, C.G. Effect of acute beetroot juice supplementation on bench press power, velocity, and repetition volume. J. Strength Cond. Res. 2020, 34, 924–928. [Google Scholar] [CrossRef]
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC consensus statement: Dietary supplements and the high-performance athlete. Br. J. Sports Med. 2018, 52, 439–455. [Google Scholar] [CrossRef]
- Hernández, A.; Schiffer, T.A.; Ivarsson, N.; Cheng, A.J.; Bruton, J.D.; Lundberg, J.O.; Weitzberg, E.; Westerblad, H. Dietary nitrate increases tetanic [Ca2+]i and contractile force in mouse fast-twitch muscle. J. Physiol. 2012, 590, 3575–3583. [Google Scholar] [CrossRef]
- Flanagan, S.D.; Looney, D.P.; Miller, M.J.S.; DuPont, W.H.; Pryor, L.; Creighton, B.C.; Sterczala, A.J.; Szivak, T.K.; Hooper, D.R.; Maresh, C.M.; et al. The effects of nitrate-rich supplementation on neuromuscular efficiency during heavy resistance exercise. J. Am. Coll. Nutr. 2016, 35, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Pareja-Blanco, F.; Rodríguez-Rosell, D.; Sánchez-Medina, L.; Sanchis-Moysi, J.; Dorado, C.; Mora-Custodio, R.; Yáñez-García, J.M.; Morales-Alamo, D.; Pérez-Suárez, I.; Calbet, J.A.L.; et al. Effects of velocity loss during resistance training on athletic performance, strength gains and muscle adaptations. Scand. J. Med. Sci. Sports 2016, 27, 724–735. [Google Scholar] [CrossRef] [PubMed]
- Haff, G.G. Current research. Strength Cond. J. 2012, 34, 76–78. [Google Scholar] [CrossRef]
- Pescatello, L.S.; Thompson, W.R.; Gordon, N.F. A preview of ACSM’S guidelines for exercise testing and prescription, eighth edition. ACSMs Health Fit. J. 2009, 13, 23–26. [Google Scholar] [CrossRef]
- Wylie, L.J.; Mohr, M.; Krustrup, P.; Jackman, S.R.; Ermιdis, G.; Kelly, J.; Black, M.I.; Bailey, S.J.; Vanhatalo, A.; Jones, A.M. Dietary nitrate supplementation improves team sport-specific intense intermittent exercise performance. Eur. J. Appl. Physiol. 2013, 113, 1673–1684. [Google Scholar] [CrossRef] [PubMed]
- Moran-Navarro, R.; Perez, C.E.; Mora-Rodriguez, R.; de la Cruz-Sanchez, E.; Gonzalez-Badillo, J.J.; Sanchez-Medina, L.; Pallares, J.G. Time course of recovery following resistance training leading or not to failure. Eur. J. Appl. Physiol. 2017, 117, 2387–2399. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.E.; Weir, J.P. Asep procedures recommendation i: Accurate assessment of muscular strength and power. J. Exerc. Physiol. Online 2001, 4, 1–21. [Google Scholar]
- González-Badillo, J.J.; Sánchez-Medina, L. Movement velocity as a measure of loading intensity in resistance training. Int. J. Sports Med. 2010, 31, 347–352. [Google Scholar] [CrossRef]
- Sánchez-Medina, L.; Pallarés, J.; Pérez, C.; Morán-Navarro, R.; González-Badillo, J. Estimation of relative load from bar velocity in the full back squat exercise. Sports Med. Int. Open. 2017, 1, E80–E88. [Google Scholar] [CrossRef]
- Govoni, M.; Jansson, E.A.; Weitzberg, E.; Lundberg, J.O. The increase in plasma nitrite after a dietary nitrate load is markedly attenuated by an antibacterial mouthwash. Nitric Oxide 2008, 19, 333–337. [Google Scholar] [CrossRef]
- Gallardo, E.J.; Coggan, A.R. What is in your beet juice? Nitrate and nitrite content of beet juice products marketed to athletes. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 345–349. [Google Scholar] [CrossRef]
- De Salles, B.F.; Simão, R.; Miranda, F.; Da Silva Novaes, J.; Lemos, A.; Willardson, J.M. Rest interval between sets in strength training. Sports Med. 2009, 39, 765–777. [Google Scholar] [CrossRef]
- Pérez-Castilla, A.; Piepoli, A.; Delgado-García, G.; Garrido-Blanca, G.; García-Ramos, A. Reliability and concurrent validity of seven commercially available devices for the assessment of movement velocity at different intensities during the bench press. J. Strength Cond. Res 2019, 33, 1258–1265. [Google Scholar] [CrossRef]
- Pallarés, J.G.; Sánchez-Medina, L.; Pérez, C.E.; De La Cruz-Sánchez, E.; Mora-Rodriguez, R. Imposing a pause between the eccentric and concentric phases increases the reliability of isoinertial strength assessments. J. Sports Sci. 2014, 32, 1165–1175. [Google Scholar] [CrossRef] [PubMed]
- Earle, R.; Baechle, T.R. The NSCA essentials of personal training text. Strength Cond. J. 2004, 26, 76–77. [Google Scholar] [CrossRef]
- Goodwin, M.L.; Harris, J.E.; Hernández, A.; Gladden, L.B. Blood lactate measurements and analysis during exercise: A guide for clinicians. J. Diabetes Sci. Technol. 2007, 1, 558–569. [Google Scholar] [CrossRef] [PubMed]
- Forsyth, J.J.; Farrally, M.R. A comparison of lactate concentration in plasma collected from the toe, ear, and fingertip after a simulated rowing exercise. Brit. J. Sport Med. 2000, 34, 35–38. [Google Scholar] [CrossRef]
- Robertson, R.J.; GOSS, F.L.; Rutkowski, J.; Lenz, B.; Dixon, C.; Timmer, J.; Frazee, K.; Dube, J.; Andreacci, J. Concurrent validation of the OMNI perceived exertion scale for resistance exercise. Med. Sci. Sports Exerc. 2003, 35, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Lakens, D. Calculating and reporting effect sizes to facilitate cumulative science: A practical primer for t-tests and ANOVAs. Front. Psychol. 2013, 4, 863. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988; p. 174. [Google Scholar]
- Coggan, A.R.; Leibowitz, J.L.; Spearie, C.A.; Kadkhodayan, A.; Thomas, D.P.; Ramamurthy, S.; Mahmood, K.; Park, S.; Waller, S.; Farmer, M.; et al. Acute dietary nitrate intake improves muscle contractile function in patients with heart failure. Circ. Heart Fail. 2015, 8, 914–920. [Google Scholar] [CrossRef]
- Coggan, A.R.; Hoffman, R.L.; Gray, D.A.; Moorthi, R.N.; Thomas, D.P.; Leibowitz, J.L.; Thies, D.; Peterson, L.R. A single dose of dietary nitrate increases maximal knee extensor angular velocity and power in healthy older men and women. J. Gerontol. A Biol. Sci. Med. Sci. 2019, 75, 1154–1160. [Google Scholar] [CrossRef]
- Bender, D.; Townsend, J.R.; Vantrease, W.C.; Marshall, A.C.; Henry, R.N.; Heffington, S.H.; Johnson, K.D. Acute beetroot juice administration improves peak isometric force production in adolescent males. Appl. Physiol. Nutr. Metab. 2018, 43, 816–821. [Google Scholar] [CrossRef]
- Whitfield, J.; Gamu, D.; Heigenhauser, G.J.F.; Van Loon, L.J.C.; Spriet, L.L.; Tupling, A.R.; Holloway, G.P. Beetroot juice increases human muscle force without changing Ca2+-handling proteins. Med. Sci. Sports Exerc. 2017, 49, 2016–2024. [Google Scholar] [CrossRef]
- Grgic, J.; Mikulic, P. Caffeine ingestion acutely enhances muscular strength and power but not muscular endurance in resistance-trained men. Eur. J. Sport Sci. 2017, 17, 1029–1036. [Google Scholar] [CrossRef]
- Ignarro, L.J.; Buga, G.M.; Wood, K.S.; Byrns, R.E.; Chaudhuri, G. Endothelium-derived relaxing factor produced and released from artery and vein is nitric oxide. Proc. Natl. Acad. Sci. USA 1987, 84, 9265–9269. [Google Scholar] [CrossRef]
- Escamilla, R.F. Knee biomechanics of the dynamic squat exercise. Med. Sci. Sports Exerc. 2001, 33, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Westerblad, H.; Allen, D.G.; Bruton, J.D.; Andrade, F.H.; Lannergren, J. Mechanisms underlying the reduction of isometric force in skeletal muscle fatigue. Acta Physiol. Scand. 1998, 162, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Bonilla Ocampo, D.A.; Paipilla, A.F.; Marín, E.; Vargas-Molina, S.; Petro, J.L.; Pérez-Idárraga, A. Dietary nitrate from beetroot juice for hypertension: A systematic review. Biomolecules 2018, 8, 134. [Google Scholar] [CrossRef] [PubMed]
- Raubenheimer, K.; Hickey, D.; Leveritt, M.; Fassett, R.; Ortiz de Zevallos Munoz, J.; Allen, J.D.; Briskey, D.; Parker, T.J.; Kerr, G.; Peake, J.M.; et al. Acute effects of nitrate-rich beetroot juice on blood pressure, hemostasis and vascular inflammation markers in healthy older adults: A randomized, placebo-controlled crossover study. Nutrients 2017, 9, 1270. [Google Scholar] [CrossRef]
- Hoon, M.W.; Jones, A.M.; Johnson, N.A.; Blackwell, J.R.; Broad, E.M.; Lundy, B.; Rice, A.J.; Burke, L.M. The effect of variable doses of inorganic nitrate-rich beetroot juice on simulated 2000-m rowing performance in trained athletes. Int. J. Sports Physiol. Perform. 2014, 9, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.J.; Qin, Z.; Wang, P.Y.; Sun, Y.; Liu, X. Muscle fatigue: General understanding and treatment. Exp. Mol. Med. 2017, 49, e384. [Google Scholar] [CrossRef] [PubMed]
- Martí, J.G.; Morales, A.M.; Bosch, M.P.; Cid, A.V.; Ferrari, M.R. El efecto del zumo de remolacha sobre la presión arterial y el ejercicio físico: Revisión sistemática. Rev. Esp. Nutr. Com. 2015, 21, 20–29. [Google Scholar]
- Jones, A.M. Dietary nitrate supplementation and exercise performance. Sports Med. 2014, 44, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Axe, D.D.; Bailey, J.E. Transport of lactate and acetate through the energized cytoplasmic membrane of Escherichia coli. Biotechnol. Bioeng. 1995, 47, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.M.; Ferguson, S.K.; Bailey, S.J.; Vanhatalo, A.; Poole, D.C. Fiber type- specific effects of dietary nitrate. Exerc. Sport Sci. 2016, 44, 53–60. [Google Scholar] [CrossRef] [PubMed]
- McMahon, S.; Jenkins, D. Factors affecting the rate of phosphocreatine resynthesis following intense exercise. Sports Med. 2002, 32, 761–784. [Google Scholar] [CrossRef] [PubMed]
- Shannon, O.M.; Barlow, M.J.; Duckworth, L.; Williams, E.; Wort, G.; Woods, D.; Siervo, M.; O’Hara, J.P. Dietary nitrate supplementation enhances short but no longer duration running time-trial performance. Eur. J. Appl. Physiol. 2017, 117, 775–785. [Google Scholar] [CrossRef]
- Murphy, M.C.; Eliot, K.; Heuertz, R.; Weiss, E.P. Whole beetroot consumption acutely improves running performance. J. Am. Diet. Assoc. 2011, 111, A16. [Google Scholar] [CrossRef]
- Jodra, P.; Domínguez, R.; Sánchez-Oliver, A.J.; Veiga-Herreros, P.; Bailey, S.J. Effect of beetroot juice supplementation on mood, perceived exertion, and performance during a 30-s wingate test. Int. J. Sports Physiol. Perform. 2020, 15, 243–248. [Google Scholar] [CrossRef]
- Husmann, F.; Bruhn, S.; Mittlmeier, T.; Zschorlich, V.; Behrens, M. Dietary nitrate supplementation improves exercise tolerance by reducing muscle fatigue and perceptual responses. Front. Physiol. 2019, 10, 404. [Google Scholar] [CrossRef]
- Presley, T.D.; Morgan, A.R.; Bechtold, E.; Clodfelter, W.; Dove, R.W.; Jennings, J.M.; Kraft, R.A.; Bruce King, S.; Laurienti, P.J.; Jack Rejeski, W.; et al. Acute effect of a high nitrate diet on brain perfusion in older adults. Nitric Oxide 2011, 24, 34–42. [Google Scholar] [CrossRef]
- Poulet, J.F.A.; Hedwig, B. New insights into corollary discharges mediated by identified neural pathways. Trends Neurosci. 2007, 30, 14–21. [Google Scholar] [CrossRef]
- DeMorree, H.M.; Klein, C.; Marcora, S.M. Perception of effort reflects central motor command during movement execution. Psychophysiology 2012, 49, 1242–1253. [Google Scholar]
- Saunders, B.; de Oliveira, L.F.; da Silva, R.P.; de Salles Painelli, V.; Goncalves, L.S.; Yamaguchi, G.; Mutti, T.; Maciel, E.; Roschel, H.; Artioli, G.G.; et al. Placebo in sports nutrition: A proof-of-principle study involving caffeine supplementation. Scand. J. Med. Sci. Sports 2017, 27, 1240–1247. [Google Scholar] [CrossRef] [PubMed]
- Szabo, A.; Szemerszky, R.; Dömötör, Z.; de la Vega, R.; Köteles, F. Creatine monohydrate ingestion-related placebo effects on brief anaerobic exercise performance. A laboratory investigation. Cuad. Psicol. Dep. 2017, 17, 81–86. [Google Scholar]
- Pallarés, J.G.; Fernández-Elías, V.E.; Ortega, J.F.; Muñoz, G.; Muñoz-Guerra, J.; Mora-Rodríguez, R. Neuromuscular responses to incremental caffeine doses: Performance and side effects. Med. Sci. Sports Excerc. 2013, 45, 2184–2192. [Google Scholar] [CrossRef] [PubMed]
| Variable | Value |
|---|---|
| AGE (YEARS) | 24 ± 3 |
| HEIGHT (M) | 1.75 ± 0.08 |
| WEIGHT (KG) | 73 ± 9.2 |
| BODY MASS INDEX (KG/M2) | 23.75 ± 2.6 |
| BODY FAT (%) | 15.8 ± 4 |
| MANUAL PRESSURE FORCE (KG) | 47.8 ± 7.1 |
| RT EXPERIENCE (YEARS) | 3.2 ± 0.9 |
| 1-RM BACK SQUAT (KG) | 93.2 ± 18.4 |
| 1-RM BENCH PRESS (KG) | 80.3 ± 19.2 |
| Variables | Back Squat | Bench Press | Total Session | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PLA | BJ | p-Value | ES | PLA | BJ | p-Value | ES | PLA | BJ | p-Value | ES | |
| Maximum Velocity (m·s−1) | ||||||||||||
| 60% 1-RM | 0.685 ± 0.094 | 0.697 ± 0.090 | 0.590 | 0.13 | 0.612 ± 0.082 | 0.609 ± 0.080 | 0.894 | 0.04 | 0.526 ± 0.051 | 0.522 ± 0.054 | 0.715 | 0.07 |
| 70% 1-RM | 0.611 ± 0.076 | 0.605 ± 0.083 | 0.727 | 0.07 | 0.431 ± 0.083 | 0.433 ± 0.058 | 0.896 | 0.03 | ||||
| 80% 1-RM | 0.512 ± 0.056 | 0.513 ± 0.085 | 0.971 | 0.01 | 0.306 ± 0.052 | 0.275 ± 0.053 | 0.135 | 0.59 | ||||
| Average | 0.603 ± 0.067 | 0.605 ± 0.075 | 0.877 | 0.02 | 0.449 ± 0.059 | 0.440 ± 0.051 | 0.414 | 0.16 | ||||
| Maximum Power (Watt) | ||||||||||||
| 60% 1-RM | 382 ± 111 | 389 ± 117 | 0.538 | 0.06 | 292 ± 94 | 289 ± 88 | 0.777 | 0.03 | 320 ± 90 | 318 ± 86 | 0.599 | 0.03 |
| 70% 1-RM | 395 ± 107 | 393 ± 116 | 0.816 | 0.02 | 238 ± 81 | 242 ± 81 | 0.690 | 0.05 | ||||
| 80% 1-RM | 378 ± 96 | 377 ± 108 | 0.961 | 0.01 | 191 ± 55 | 176 ± 66 | 0.257 | 0.25 | ||||
| Average | 392 ± 112 | 390 ± 104 | 0.857 | 0.02 | 249 ± 76 | 245 ± 77 | 0.520 | 0.05 | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ranchal-Sanchez, A.; Diaz-Bernier, V.M.; De La Florida-Villagran, C.A.; Llorente-Cantarero, F.J.; Campos-Perez, J.; Jurado-Castro, J.M. Acute Effects of Beetroot Juice Supplements on Resistance Training: A Randomized Double-Blind Crossover. Nutrients 2020, 12, 1912. https://doi.org/10.3390/nu12071912
Ranchal-Sanchez A, Diaz-Bernier VM, De La Florida-Villagran CA, Llorente-Cantarero FJ, Campos-Perez J, Jurado-Castro JM. Acute Effects of Beetroot Juice Supplements on Resistance Training: A Randomized Double-Blind Crossover. Nutrients. 2020; 12(7):1912. https://doi.org/10.3390/nu12071912
Chicago/Turabian StyleRanchal-Sanchez, Antonio, Victor Manuel Diaz-Bernier, Candelaria Alonso De La Florida-Villagran, Francisco Jesus Llorente-Cantarero, Julian Campos-Perez, and Jose Manuel Jurado-Castro. 2020. "Acute Effects of Beetroot Juice Supplements on Resistance Training: A Randomized Double-Blind Crossover" Nutrients 12, no. 7: 1912. https://doi.org/10.3390/nu12071912
APA StyleRanchal-Sanchez, A., Diaz-Bernier, V. M., De La Florida-Villagran, C. A., Llorente-Cantarero, F. J., Campos-Perez, J., & Jurado-Castro, J. M. (2020). Acute Effects of Beetroot Juice Supplements on Resistance Training: A Randomized Double-Blind Crossover. Nutrients, 12(7), 1912. https://doi.org/10.3390/nu12071912






