The Role of Selenium Mineral Trace Element in Exercise: Antioxidant Defense System, Muscle Performance, Hormone Response, and Athletic Performance. A Systematic Review
Abstract
1. Introduction
2. Material and Methods
2.1. Search Strategy
2.2. Selection of Articles: Inclusion and Exclusion Criteria
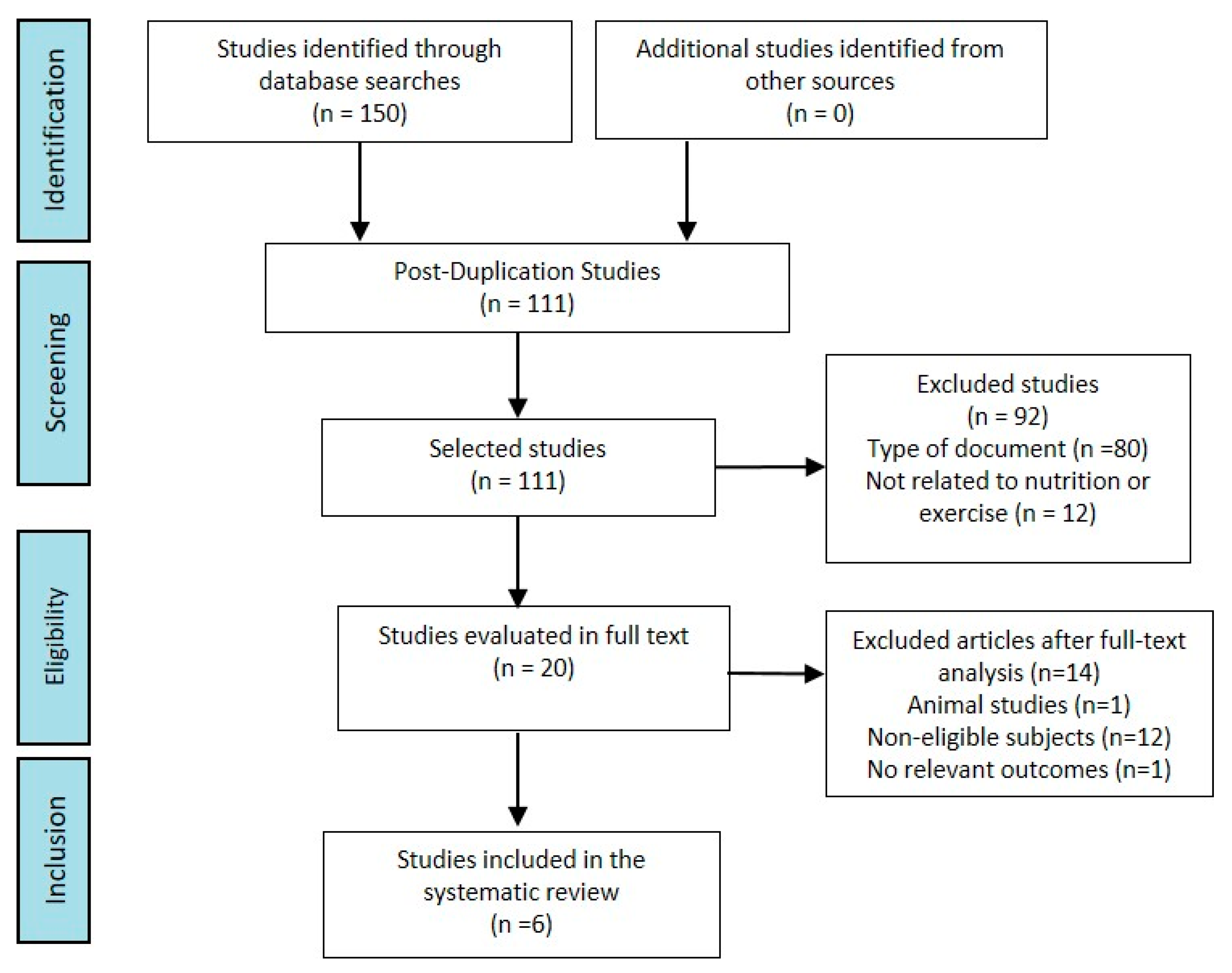
3. Results
3.1. Literature Search
3.2. Characteristics of the Studies
3.3. Assessment of the Methodological Quality
3.4. Findings of Included Studies
4. Discussion
4.1. Selenium Supplementation
4.2. Antioxidant Defense System
4.3. Muscle Performance
4.4. Hormone Response
4.5. Athletic Performance
4.6. Practical Applications
4.7. Limitations and Strengths
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vatansever, R.; Ozyigit, I.I.; Filiz, E. Essential and beneficial trace elements in plants, and their transport in roots: A review. Appl. Biochem. Biotechnol. 2017, 181, 464–482. [Google Scholar] [CrossRef]
- Mehdi, Y.; Hornick, J.-L.; Istasse, L.; Dufrasne, I. Selenium in the environment, metabolism and involvement in body functions. Molecules 2013, 18, 3292–3311. [Google Scholar] [CrossRef]
- Behne, D.; Weiler, H.; Kyriakopoulos, A. Effects of selenium deficiency on testicular morphology and function in rats. J. Reprod. Fertil. 1996, 106, 291–297. [Google Scholar] [CrossRef]
- Heffernan, S.M.; Horner, K.; De Vito, G.; Conway, G.E. The role of mineral and trace element supplementation in exercise and athletic performance: A systematic review. Nutrients 2019, 11, 696. [Google Scholar] [CrossRef]
- Speich, M.; Pineau, A.; Ballereau, F. Minerals, trace elements and related biological variables in athletes and during physical activity. Clin. Chim. Acta 2001, 312, 1–11. [Google Scholar] [CrossRef]
- Seo, D.-Y.; Heo, J.-W.; Ko, J.R.; Kwak, H.-B. Exercise and neuroinflammation in health and disease. Int. Neurourol. J. 2019, 23, S82. [Google Scholar] [CrossRef]
- Fernández-Lázaro, D.; Mielgo-Ayuso, J.; Seco Calvo, J.; Córdova Martínez, A.; Caballero García, A.; Fernandez-Lazaro, C.I. Modulation of Exercise-Induced Muscle Damage, Inflammation, and Oxidative Markers by Curcumin Supplementation in a Physically Active Population: A Systematic Review. Nutrients 2020, 12, 501. [Google Scholar] [CrossRef]
- Zoidis, E.; Seremelis, I.; Kontopoulos, N.; Danezis, G.P. Selenium-Dependent Antioxidant Enzymes: Actions and Properties of Selenoproteins. Antioxidants 2018, 7, 66. [Google Scholar] [CrossRef]
- Ranchordas, M.K.; Rogerson, D.; Soltani, H.; Costello, J.T. Antioxidants for preventing and reducing muscle soreness after exercise. Cochrane Database Syst. Rev. 2017. [Google Scholar] [CrossRef]
- Cisnero Prego, E.; Pupo Balboa, J.; Céspedes Miranda, E. Enzimas que participan como barreras fisiológicas para eliminar los radicales libres: III. Glutatión peroxidasa. Rev. Cuba. Investig. Biomed. 1997, 1, 10–15. [Google Scholar]
- Cisneros Prego, E. La glutatión reductasa y su importancia biomédica. Rev. Cuba. Investig. Biomed. 1995, 14, 10–12. [Google Scholar]
- García Triana, B.; García Morales, O.; Clapes Hernández, S.; Rodes Fernández, L.; García Piñeiro, J.C. Enzimas que participan como barreras fisiológicas para eliminar los radicales libres: I. Superóxido dismutasas. Rev. Cuba. Investig. Biomed. 1995, 14, 15–21. [Google Scholar]
- Margaritis, I.; Tessier, F.; Prou, E.; Marconnet, P.; Marini, J. Effects of endurance training on skeletal muscle oxidative capacities with and without selenium supplementation. J. Trace Elem. Med. Biol. 1997, 11, 37–43. [Google Scholar] [CrossRef]
- Córdova, A.; Mielgo-Ayuso, J.; Fernandez-Lazaro, C.I.; Caballero-García, A.; Roche, E.; Fernández-Lázaro, D. Effect of iron supplementation on the modulation of iron metabolism, muscle damage biomarkers and cortisol in professional cyclists. Nutrients 2019, 11, 500. [Google Scholar] [CrossRef]
- Alfredo, C.; Diego, F.; Juan, M.; Calvo, S.; Jesús, C.G.A. Effect of magnesium supplementation on muscular damage markers in basketball players during a full season. J. Magnes. Res. 2017, 30, 61–70. [Google Scholar]
- Rotruck, J.T.; Pope, A.L.; Ganther, H.E.; Swanson, A.; Hafeman, D.G.; Hoekstra, W. Selenium: Biochemical role as a component of glutathione peroxidase. Science 1973, 179, 588–590. [Google Scholar] [CrossRef]
- Neve, J. Human selenium supplementation as assessed by changes in blood selenium concentration and glutathione peroxidase activity. J. Trace Elem. Med. Biol. 1995, 9, 65–73. [Google Scholar] [CrossRef]
- Neek, L.S.; Gaeini, A.A.; Choobineh, S. Effect of zinc and selenium supplementation on serum testosterone and plasma lactate in cyclist after an exhaustive exercise bout. Biol. Trace Elem. Res. 2011, 144, 454–462. [Google Scholar] [CrossRef]
- Savory, L.A.; Kerr, C.J.; Whiting, P.; Finer, N.; McEneny, J.; Ashton, T. Selenium supplementation and exercise: Effect on oxidant stress in overweight adults. Obesity 2012, 20, 794–801. [Google Scholar] [CrossRef]
- Burk, R.F. Protection against free radical injury by selenoenzymes. Pharmacol. Ther. 1990, 45, 383–385. [Google Scholar] [CrossRef]
- Bloomer, R.J.; Falvo, M.J.; Schilling, B.K.; Smith, W.A. Prior exercise and antioxidant supplementation: Effect on oxidative stress and muscle injury. J. Int. Soc. Sports Nutr. 2007, 4, 9. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Cabrera, M.C.; Salvador-Pascual, A.; Cabo, H.; Ferrando, B.; Viña, J. Redox modulation of mitochondriogenesis in exercise. Does antioxidant supplementation blunt the benefits of exercise training? Free Radic. Biol. Med. 2015, 86, 37–46. [Google Scholar] [CrossRef]
- Kanter, M.M. Free radicals, exercise, and antioxidant supplementation. Int. J. Sport Nutr. Exerc. Metab. 1994, 4, 205–220. [Google Scholar] [CrossRef]
- Urso, M.L.; Clarkson, P.M. Oxidative stress, exercise, and antioxidant supplementation. Toxicology 2003, 189, 41–54. [Google Scholar] [CrossRef]
- Manzanares Castro, W. Selenium in critical patients with systemic inflammatory response. Nutr. Hosp. 2007, 22, 295–306. [Google Scholar]
- Tessier, F.; Margaritis, I.; Richard, M.-J.; Moynot, C.; Marconnet, P. Selenium and training effects on the glutathione system and aerobic performance. Med. Sci. Sports Exerc. 1995, 27, 390–396. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 151, W-65–W-94. [Google Scholar]
- Law, M.; Stewart, D.; Letts, L.; Pollock, N.; Bosch, J.; Westmorland, M. Guidelines for Critical Review Form—Quantitative Studies 1998; McMaster University: Hamilton, ON, Canada, 2008. [Google Scholar]
- Zamora, A.; Tessier, F.; Marconnet, P.; Margaritis, I.; Marini, J.-F. Mitochondria changes in human muscle after prolonged exercise, endurance training and selenium supplementation. Eur. J. Appl. Physiol. Occup. Physiol. 1995, 71, 505–511. [Google Scholar] [CrossRef]
- Tessier, F.; Hida, H.; Favier, A.; Marconnet, P. Muscle GSH-Px activity after prolonged exercise, training, and selenium supplementation. Biol. Trace Elem. Res. 1995, 47, 279–285. [Google Scholar] [CrossRef]
- Hays, S.M.; Macey, K.; Nong, A.; Aylward, L.L. Biomonitoring Equivalents for selenium. Regul. Toxicol. Pharmacol. 2014, 70, 333–339. [Google Scholar] [CrossRef]
- Yang, G.; Yin, S.; Zhou, R.; Gu, L.; Yan, B.; Liu, Y. Studies of safe maximal daily dietary Se-intake in a seleniferous area in China. Part II: Relation between Se-intake and the manifestation of clinical signs and certain biochemical alterations in blood and urine. J. Trace Elem. Electrolytes Health Dis. 1989, 3, 123–130. [Google Scholar] [PubMed]
- Schrauzer, G.N. Nutritional selenium supplements: Product types, quality, and safety. J. Am. Coll. Nutr. 2001, 20, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Brenneisen, P.; Steinbrenner, H.; Sies, H. Selenium, oxidative stress, and health aspects. Mol. Aspects Med. 2005, 26, 256–267. [Google Scholar] [CrossRef]
- Klotz, L.-O.; Kröncke, K.-D.; Buchczyk, D.P.; Sies, H. Role of copper, zinc, selenium and tellurium in the cellular defense against oxidative and nitrosative stress. J. Nutr. 2003, 133, 1448S–1451S. [Google Scholar] [CrossRef] [PubMed]
- Akil, M.; Gurbuz, U.; Bicer, M.; Sivrikaya, A.; Mogulkoc, R.; Baltaci, A.K. Effect of selenium supplementation on lipid peroxidation, antioxidant enzymes, and lactate levels in rats immediately after acute swimming exercise. Biol. Trace Elem. Res. 2011, 142, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Brady, P.S.; Brady, L.J.; Ullrey, D.E. Selenium, vitamin E and the response to swimming stress in the rat. J. Nutr. 1979, 109, 1103–1109. [Google Scholar] [CrossRef]
- Morabito, R.; Remigante, A.; Marino, A. Melatonin Protects Band 3 Protein in Human Erythrocytes against H2O2-Induced Oxidative Stress. Molecules 2019, 24, 2741. [Google Scholar] [CrossRef]
- Egan, B.; Zierath, J.R. Exercise metabolism and the molecular regulation of skeletal muscle adaptation. Cell Metab. 2013, 17, 162–184. [Google Scholar] [CrossRef]
- Fernández-Landa, J.; Fernández-Lázaro, D.; Calleja-González, J.; Caballero-García, A.; Córdova, A.; León-Guereño, P.; Mielgo-Ayuso, J. Long-Term Effect of Combination of Creatine Monohydrate Plus β-Hydroxy β-Methylbutyrate (HMB) on Exercise-Induced Muscle Damage and Anabolic/Catabolic Hormones in Elite Male Endurance Athletes. Biomolecules 2020, 10, 140. [Google Scholar] [CrossRef]
- Mujika, I. Quantification of training and competition loads in endurance sports: Methods and applications. Int. J. Sports Physiol. Perform. 2017, 12, S29–S217. [Google Scholar] [CrossRef]
- Pingitore, A.; Lima, G.P.P.; Mastorci, F.; Quinones, A.; Iervasi, G.; Vassalle, C. Exercise and oxidative stress: Potential effects of antioxidant dietary strategies in sports. Nutrition 2015, 31, 916–922. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Wang, T.; Li, Q.; Li, D. Prevention of Keshan disease by selenium supplementation: A systematic review and meta-analysis. Biol. Trace Elem. Res. 2018, 186, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Simpson, R.J.; Kunz, H.; Agha, N.; Graff, R. Exercise and the Regulation of Immune Functions. Prog. Mol. Biol. Transl. Sci. 2015, 135, 355–380. [Google Scholar] [PubMed]
- Kieliszek, M.; Lipinski, B. Selenium supplementation in the prevention of coronavirus infections (COVID-19). Med. Hypotheses 2020, 143, 109878. [Google Scholar] [CrossRef]
| Level of Participants | Active | 1 Study [18] |
| Regularly trained athletes | 4 Studies [13,26,29,30] | |
| No Regular Training before the Study | 1 Study [19] | |
| Age Range (years) | 20–35 years | 5 Studies [13,19,26,29,30] |
| Not Specified | 1 Study [18] | |
| Se Plasma Level (µg/l) | Assayed | 4 Studies [13,19,26,30] |
| Not Assayed | 2 Studies [18,29] | |
| Type of Administration of Selenium | Organic selenium in form of selenomethionine | 4 Studies [13,26,29,30] |
| Salts of sodium selenite (Na2Se03) | 2 Studies [18,19] | |
| Dosage Used | 180 µg single dose | 3 Studies [13,26,29] |
| 200 µg single dose | 2 Studies [18,19] | |
| 240 µg single dose | 1 Study [30] | |
| Moment of Supplementation | Daily | 6 Studies [13,18,19,26,29,30] |
| Duration of Treatment | 4 weeks | 1 Study [18] |
| 10 weeks | 4 Studies [13,26,29,30] | |
| 14 weeks | 1 Study [19] |
| References | Margaritis et al. 1997 [13] | Zamora et al. 1995 [29] | Savory et al. 2012 [19] | Tessier et al. 1994 [26] | Neek et al. 2011 [18] | Tessier et al. 1995 [30] | TI | |
|---|---|---|---|---|---|---|---|---|
| ITEMS | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 6 |
| 2 | 1 | 1 | 1 | 1 | 1 | 1 | 6 | |
| 3 | 1 | 1 | 1 | 1 | 1 | 1 | 6 | |
| 4 | 1 | 1 | 1 | 1 | 1 | 1 | 6 | |
| 5 | 1 | 1 | 1 | 1 | 1 | 1 | 6 | |
| 6 | 0 | 0 | 1 | 0 | 0 | 0 | 2 | |
| 7 | 1 | 1 | 1 | 1 | 1 | 1 | 6 | |
| 8 | 1 | 1 | 1 | 1 | 1 | 1 | 6 | |
| 9 | 0 | 1 | 1 | 1 | 1 | 1 | 5 | |
| 10 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | |
| 11 | 1 | 1 | 1 | 1 | 1 | 1 | 6 | |
| 12 | 1 | 1 | 1 | 1 | 1 | 1 | 6 | |
| 13 | 1 | 1 | 1 | 1 | 1 | 1 | 6 | |
| 14 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 15 | 1 | 1 | 1 | 1 | 1 | 1 | 6 | |
| 16 | 1 | 1 | 1 | 1 | 1 | 1 | 6 | |
| TS | 12 | 13 | 14 | 13 | 15 | 13 | ||
| % | 75 | 81.3 | 87.5 | 81.3 | 93.8 | 81.3 | ||
| MQ | G | VG | VG | VG | E | VG | ||
| Authors & Year | Study Design | Population | Intervention | Analyzed Results | Main Conclusions | |
|---|---|---|---|---|---|---|
| Savory et al., 2012 [19] | Placebo-controlled, double-blind, crossover | 20 healthy subjects 9♂ & 1♀ NW: 4 ♂ & 6 ♀ 27.9 ± 2.2 y BMI 22.8 ± 0.4 kg/m2 OW: 5 ♂ & 5 ♀ 31.4 ± 1.9 y BMI 28.0 ± 0.8 kg/m2 | Supplementation: 200 μg Se (sodium selenite) once per day * 3 weeks’ placebo (not containing glucose) during another 3-week period. Washout period 2 moth. Order treatment: NW: Se/Placebo OW: Placebo/Se 14-week total period PhA: test 30 min treadmill session at 70% VO2peak | [Se] | Post Se treatment period ↑ *[Se] NW & OW compared to week 0 | |
| Post Se treatment period ↑* [Se] NW & OW compared to placebo treatment | ||||||
| TAS, GSH, SOD | Placebo period & Se treatment period
| |||||
| LH | Placebo period
| |||||
Se treatment period
| ||||||
| Placebo vs. Se treatment post PhA: OW ↓*LH; NW↔LH | ||||||
| Tessier et al., 1994 [26] | Placebo-controlled, double-blind, randomized | 24 ♂ healthy students 22.9 ± 2.1 y; 8.0 ± 8.7 Kg; 178.0 ± 6.6 cm; Body fat 11.2 ± 4.4 % PbG n = 12 ♂ 22.5 ± 2.0 y; 67.3 ±7.0 Kg; 177.4 ± 7.0 cm; Body fat 10.4 ± 3.9 % SeG n = 12 ♂ 23.2 ± 2.3 y; 68.7 ± 10.4 Kg; 178.7 ± 6.3 cm; Body fat 12.3 ± 4.8 % | Supplementation: 180 μg Se (Seleniomethionine) once per day * 10-week period PhA: 10-week endurance training program 4-week nontraining | Pre-PhA vs. Post-PhA | SeG vs. PbG | |
| [Se] | ↑*[Se] SeG ↓[Se] PbG | # [Se] | ||||
| GTtotal | ↓* SeGr ↓* PbGr | † GTtotal | ||||
| GSSG | ↓ GSSG SeGr ↓ GSSG PbGr | † GSSG | ||||
| GPx plasma | ↑* SeG ↑PbG | # GPx | ||||
| EGPx | ↑* SeG ↑PbG | # EGPx | ||||
| EGR | ↑* SeG ↑*PbG | † EGR | ||||
| Vitamin E | ↓SeG ↑PbG | † Vitamin E | ||||
| VO2max | ↑* SeG ↑*PbG | † VO2max | ||||
| SeG: ↑ VO2max positive correlated ↑GPX (r:0.66 p < 0.05 n = 12) | ||||||
| Author/s—Year | Study Design | Population | Intervention | Analyzed Results | Results and Main Conclusions | |
|---|---|---|---|---|---|---|
| Neek et al., 2011 [18] | Placebo-controlled, double-blind, randomized | 16 ♂ road cyclists Pb n = 8 ♂ cyclists 66.1 ± 4.2 Kg; 176.8 ± 8.0 cm; BMI 21.1 ± 4.4 Kg/m2 SeG n = 8 ♂ cyclists 61.6 ± 4.7 Kg; 177.7 ± 4.2 cm; BMI 20.5 ± 1.2 Kg/m2 | Supplementation: 200 μg de Selenium (sodium selenite) once per day * 4-week period PhA: cycling exhaustive exercise 4-week | Pre-PhA vs. Post-PhA | SeG vs. PbG | |
| Tt | ↑* SeG ↑* PbG | ↔ Tt | ||||
| Tf | ↑* SeG ↑* PbG | ↔ Tf | ||||
| [La] | ↑* SeG ↑* PbG | ↔ [La] | ||||
| Margaritis et al., 1997 [13] | Placebo-controlled, double-blind, randomized | 24 ♂ healthy subjects 22.9 ± 2.2 y; 68.0 ± 8.7 Kg; 178.1 ± 6.6 cm; Body fat 11.2 ± 4.9 % PbG n = 12 ♂ 22.5 ± 2.0 y; 67.3 ± 7.0 Kg; 177.4 ± 7.0 cm; Body fat 10.1 ± 4.0% SeG n = 12 ♂ 23.3 ± 2.4 y; 68.8 ± 10.4 kg; 178.8 ± 6.4 cm; Body fat 12.6 ± 4.9 % | Supplementation: 180 μg Se (Seleniomethionine) once per day * 10-week period PhA: 10-wk endurance training program, 3 sessions per week | Pre-PhA vs. Post-PhA | SeG vs. PbG | |
| [Se] | ↑*[Se] SeG ↓[Se] PbG | # SeG vs. PbG | ||||
| GPx plasma | ↑*SeG ↑PbG | # SeG vs. PbG | ||||
| GPx muscle | ↓ SeG ↓ PbG | † SeG vs. PbG | ||||
| Vitamin E | ↓ SeG ↑PbG | † SeG vs. PbG | ||||
| CK | ↓ SeG ↓PbG | † SeG vs. PbG | ||||
| Cyt Ox | ↑ SeG ↑PbG | # SeG vs. PbG | ||||
| SDH | ↑ SeG ↑ PbG | † SeG vs. PbG | ||||
| MHC I | ↑SeG ↑PbG | # SeG vs. PbG | ||||
| MHC II | ↓SeG ↓PbG | † SeG vs. PbG | ||||
| MHC I - MHC II co-expressed | ↑SeG ↑PbG | # SeG vs. PbG | ||||
| VO2max | ↑*SeG ↑*PbG | # SeG vs. PbG | ||||
| VO2total | ↑*SeG ↑*PbG | # SeG vs. PbG | ||||
| Author/s—Year | Study Design | Population | Intervention | Analyzed Results | Main Conclusions | |||
|---|---|---|---|---|---|---|---|---|
| Zamora et al., 1995 [29] | Placebo-controlled, double-blind, randomized | 24 ♂ healthy students; 22.9 ± 2.1 y; 68.0 ± 8.7 Kg; 178.1 ± 6.6 cm; Body fat 11.2 ± 4.4 % PbG n = 12 ♂ 22.5 ± 2.0 y; 67.3 ± 7.0 Kg; 177.4 ± 7.0 cm; Body fat 10.1 ± 3.9 % SeG n = 12 ♂ 23.28 ± 2.36 y; 68.78 ± 10.44 Kg; 178.7 ± 6.3 cm; Body fat 12.3 ± 4.8 % | Supplementation: 180 μg Se (Seleniomethionine) once per day * 10-week period PhA: 10-week endurance training programme (3 sessions per week) after a 4-week period of restricted | At rest | Post-PhA | |||
| Pre-PhA vs. Post-PhA | SeG vs. PbG | Pre-PhA vs. Post-PhA | SeG vs. PbG | |||||
| Muscle mitochondria morphometric parameters QA Aa â | ↑*SelG ↔ PbG ↑*SelGr ↔ PbG ↔ SelGr ↔ PbG | #SelGr vs PbG #SelGr vs PbG #SelGr vs PbG | ↑*SelG ↔ PbG ↑*SelG ↔ PbG ↔ SelGr ↔ PbG | #SelGr vs PbG ↔ SelGr vs PbG #SelGr vs PbG | ||||
| VO2max | ↔ SelGr ↔ PbG | † SelGr vs PbG | ↔ SelGr ↔ PbG | † SelGr vs PbG | ||||
| Body fat % | ↔ SelGr ↔ PbG | † SelGr vs PbG | ↔ SelGr ↔ PbG | † SelGr vs PbG | ||||
| BMI Kg*m2 | ↔ SelGr ↔ PbG | † SelGr vs PbG | ↔ SelGr ↔ PbG | † SelGr vs PbG | ||||
| Tessier et al., 1995 [30] | Placebo-controlled, double-blind, randomized | 24 ♂ healthy; 22.9 ± 2.1 y | Supplementation: 240 μg organic selenium (70% selenomethionine) Selenion® once per day *10-week period PhA: 4-week deconditioning period with no training, followed by running endurance training lasting 10 week (3 sessions/week). | Pre-PhA vs. Post-PhA | SeG vs. PbG | |||
| [Se] | ↑*SelG ↓PbG | † SelGr vs PbG | ||||||
| Vitamin E | ↓SelG ↑PbG | † SelGr vs PbG | ||||||
| GPx muscle | ||||||||
| PhA cronic | ↓SelG ↓PbG | † SelGr vs PbG | ||||||
| PhA acute | ↑*SelG ↓*PbG | # SelGr vs PbG | ||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Lázaro, D.; Fernandez-Lazaro, C.I.; Mielgo-Ayuso, J.; Navascués, L.J.; Córdova Martínez, A.; Seco-Calvo, J. The Role of Selenium Mineral Trace Element in Exercise: Antioxidant Defense System, Muscle Performance, Hormone Response, and Athletic Performance. A Systematic Review. Nutrients 2020, 12, 1790. https://doi.org/10.3390/nu12061790
Fernández-Lázaro D, Fernandez-Lazaro CI, Mielgo-Ayuso J, Navascués LJ, Córdova Martínez A, Seco-Calvo J. The Role of Selenium Mineral Trace Element in Exercise: Antioxidant Defense System, Muscle Performance, Hormone Response, and Athletic Performance. A Systematic Review. Nutrients. 2020; 12(6):1790. https://doi.org/10.3390/nu12061790
Chicago/Turabian StyleFernández-Lázaro, Diego, Cesar I. Fernandez-Lazaro, Juan Mielgo-Ayuso, Lourdes Jiménez Navascués, Alfredo Córdova Martínez, and Jesús Seco-Calvo. 2020. "The Role of Selenium Mineral Trace Element in Exercise: Antioxidant Defense System, Muscle Performance, Hormone Response, and Athletic Performance. A Systematic Review" Nutrients 12, no. 6: 1790. https://doi.org/10.3390/nu12061790
APA StyleFernández-Lázaro, D., Fernandez-Lazaro, C. I., Mielgo-Ayuso, J., Navascués, L. J., Córdova Martínez, A., & Seco-Calvo, J. (2020). The Role of Selenium Mineral Trace Element in Exercise: Antioxidant Defense System, Muscle Performance, Hormone Response, and Athletic Performance. A Systematic Review. Nutrients, 12(6), 1790. https://doi.org/10.3390/nu12061790









