Effects of Monomeric and Oligomeric Flavanols on Kidney Function, Inflammation and Oxidative Stress in Runners: A Randomized Double-Blind Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Participants
2.3. Intervention
2.4. Study-Related Procedures
2.5. Urine Testing
2.6. Sample Calculation
2.7. Sample Randomization
2.8. Statictisal Analysis
3. Results
3.1. Baseline Characteritics of the Participants
3.2. Changes in Clinical and Performance Parameters after the Half-Marathon
3.3. Prevalence of Hematuria and Proteinuria after the Half-Marathon
3.4. Urinary Creatinine, Albumin and Electrolytes Concentrations
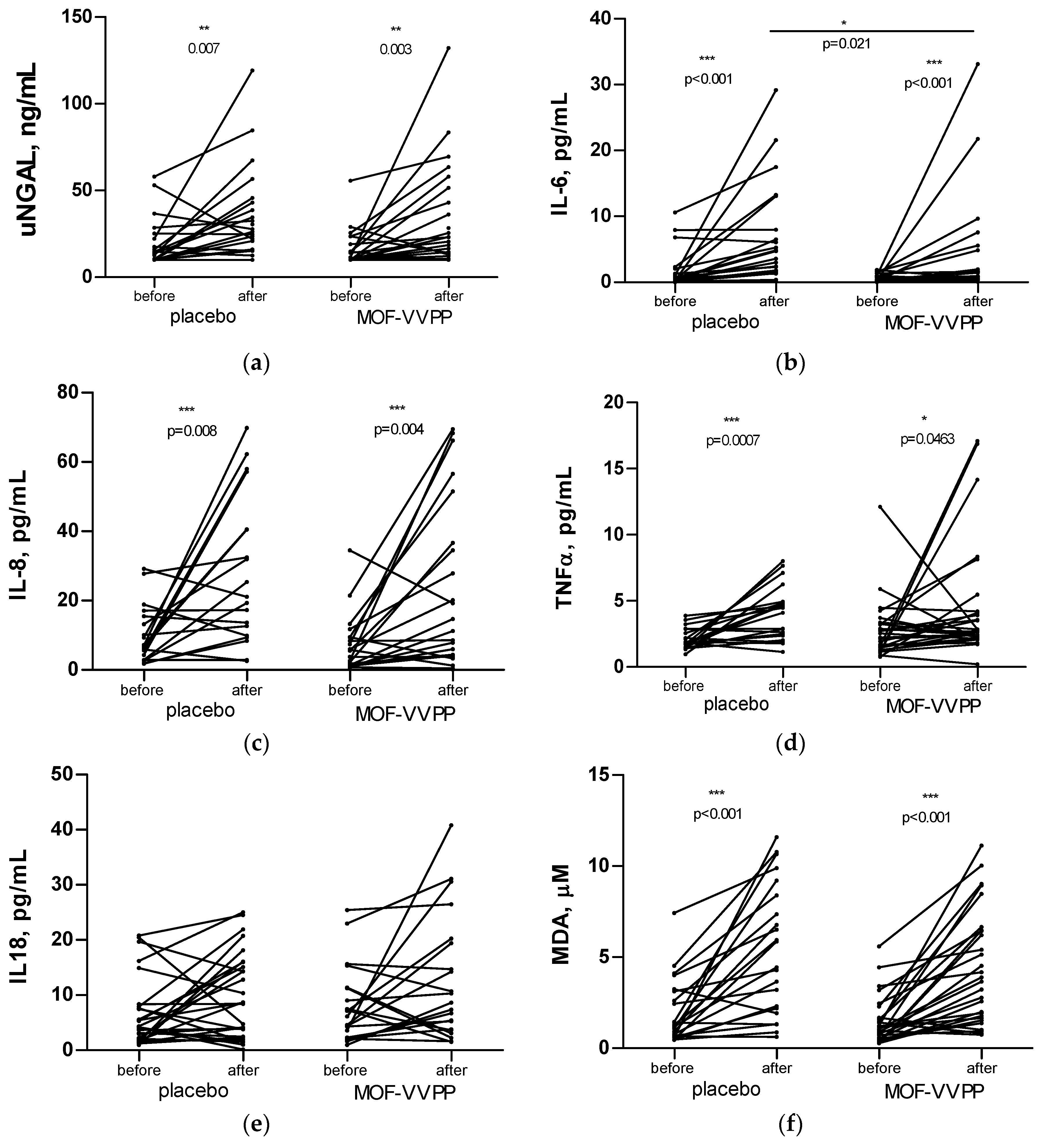
3.5. Biomarkers of Renal Inflammation and Oxidative Stress
3.6. Safety
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Venckunas, T.; Snieckus, A.; Trinkunas, E.; Baranauskiene, N.; Solianik, R.; Juodsnukis, A.; Streckis, V.; Kamandulis, S. Interval Running Training Improves Cognitive Flexibility and Aerobic Power of Young Healthy Adults. J. Strength Cond. Res. 2016, 30, 2114–2121. [Google Scholar] [CrossRef] [PubMed]
- Chakravarty, E.F.; Hubert, H.B.; Lingala, V.B.; Fries, J.F. Reduced Disability and Mortality Among Aging Runners. Arch. Intern. Med. 2008, 168, 1638–1646. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-C.; Brellenthin, A.G.; Thompson, P.D.; Sui, X.; Lee, I.-M.; Lavie, C.J. Running as a Key Lifestyle Medicine for Longevity. Prog. Cardiovasc. Dis. 2017, 60, 45–55. [Google Scholar] [CrossRef]
- Mansour, S.G.; Verma, G.; Pata, R.W.; Martin, T.G.; Perazella, M.A.; Parikh, C.R. Kidney Injury and Repair Biomarkers in Marathon Runners. Am. J. Kidney Dis. 2017, 70, 252–261. [Google Scholar] [CrossRef]
- Belli, T.; Macedo, D.V.; De Araújo, G.G.; Dos Reis, I.G.M.; Scariot, P.P.M.; Lazarim, F.L.; Nunes, L.A.S.; Brenzikofer, R.; Gobatto, C. Mountain Ultramarathon Induces Early Increases of Muscle Damage, Inflammation, and Risk for Acute Renal Injury. Front. Physiol. 2018, 9. [Google Scholar] [CrossRef]
- Wołyniec, W.; Kasprowicz, K.; Giebułtowicz, J.; Korytowska, N.; Zorena, K.; Bartoszewicz, M.; Tkachenko, R.; Renke, M.; Ratkowski, W.; Rita-Tkachenko, P. Changes in Water Soluble Uremic Toxins and Urinary Acute Kidney Injury Biomarkers After 10- and 100-km Runs. Int. J. Environ. Res. Public Health 2019, 16, 4153. [Google Scholar] [CrossRef]
- Banfi, G.; Schena, F.; Salvagno, G.L.; Tarperi, C.; Montagnana, M.; Gelati, M.; Banfi, G.; Guidi, G. Acute Variation of Estimated Glomerular Filtration Rate Following a Half-Marathon Run. Int. J. Sports Med. 2008, 29, 948–951. [Google Scholar] [CrossRef]
- Mohseni, M.; Silvers, S.; McNeil, R.; Diehl, N.; Vadeboncoeur, T.; Taylor, W.; Shapiro, S.; Roth, J.; Mahoney, S. Prevalence of Hyponatremia, Renal Dysfunction, and Other Electrolyte Abnormalities Among Runners Before and After Completing a Marathon or Half Marathon. Sports Health 2011, 3, 145–151. [Google Scholar] [CrossRef]
- Tian, Y.; Tong, T.K.; Lippi, G.; Huang, C.; Shi, Q.; Nie, J. Renal function parameters during early and late recovery periods following an all-out 21-km run in trained adolescent runners. Clin. Chem. Lab. Med. 2011, 49, 993–997. [Google Scholar] [CrossRef]
- Gross, M.; Baum, O. Supplemental Antioxidants and Adaptation to Physical Training. In Antioxidants in Sport Nutrition; Lamprecht, M., Ed.; CRC Press/Taylor & Francis 2015 by Taylor & Francis Group, LLC: Boca Raton, FL, USA, 2015. [Google Scholar]
- Junglee, N.A.; Di Felice, U.; Dolci, A.; Fortes, M.B.; Jibani, M.M.; Lemmey, A.B.; Walsh, N.P.; Macdonald, J.H. Exercising in a hot environment with muscle damage: Effects on acute kidney injury biomarkers and kidney function. Am. J. Physiol. Ren. Physiol. 2013, 305, F813–F820. [Google Scholar] [CrossRef]
- Rotunno, A.; Schwellnus, M.; Swanevelder, S.; Jordaan, E.; Van Rensburg, D.C.J.; Derman, W. Novel Factors Associated With Analgesic and Anti-inflammatory Medication Use in Distance Runners. Clin. J. Sport Med. 2018, 28, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Küster, M.; Renner, B.; Oppel, P.; Niederweis, U.; Brune, K. Consumption of analgesics before a marathon and the incidence of cardiovascular, gastrointestinal and renal problems: A cohort study. BMJ Open 2013, 3, 3. [Google Scholar] [CrossRef] [PubMed]
- Poortmans, J.R. Exercise and Renal Function. Sports Med. 1984, 1, 125–153. [Google Scholar] [CrossRef] [PubMed]
- Ageberg, E.; Bunke, S.; Andersson, K.; Nilsen, P.; Donaldson, A. The use of non-steroidal anti-inflammatories (NSAIDs) at the 2016 London marathon. Br. J. Sports Med. 2017, 51, 284–413. [Google Scholar] [CrossRef]
- Joslin, J.; Lloyd, J.; Kotlyar, T.; Wojcik, S. NSAID and other analgesic use by endurance runners during training, competition and recovery. S. Afr. J. Sports Med. 2013, 25, 101–104. [Google Scholar] [CrossRef]
- Martínez, S.; Aguilo, A.; Moreno, C.; Lozano, L.; Tauler, P. Use of Non-Steroidal Anti-Inflammatory Drugs among Participants in a Mountain Ultramarathon Event. Sports 2017, 5, 11. [Google Scholar] [CrossRef]
- Robert, A.-M. Substrate-protecting antiproteolytic agents for the prevention of pathological degradation of connective tissues. A review. Pathol. Boil. 2012, 60, 48–57. [Google Scholar] [CrossRef]
- Robert, A.M.; Robert, L.; Renard, G. Protection of cornea against proteolytic damage. Experimental study of procyanidolic oligomers (PCO) on bovine cornea. J. Français Ophtalmol. 2002, 25, 351–355. [Google Scholar]
- Changizi-Ashtiyani, S.; Najafi, H.; Firouzifar, M.R.; Shafaat, O. Grape seed extract for reduction of renal disturbances following reperfusion in rats. Iran. J. Kidney Dis. 2013, 7, 28–35. [Google Scholar]
- Nazima, B.; Manoharan, V.; Miltonprabu, S. Grape seed proanthocyanidins ameliorates cadmium-induced renal injury and oxidative stress in experimental rats through the up-regulation of nuclear related factor 2 and antioxidant responsive elements. Biochem. Cell Boil. 2015, 93, 210–226. [Google Scholar] [CrossRef]
- Pinna, C.; Morazzoni, P.; Sala, A. Proanthocyanidins from Vitis vinifera inhibit oxidative stress-induced vascular impairment in pulmonary arteries from diabetic rats. Phytomedicine 2017, 25, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Ruijters, E.J.B.; Haenen, G.R.; Willemsen, M.; Weseler, A.R.; Bast, A. Food-Derived Bioactives Can Protect the Anti-Inflammatory Activity of Cortisol with Antioxidant-Dependent and -Independent Mechanisms. Int. J. Mol. Sci. 2016, 17, 239. [Google Scholar] [CrossRef] [PubMed]
- Weseler, A.R.; Ruijters, E.J.B.; Drittij-Reijnders, M.-J.; Reesink, K.D.; Haenen, G.R.; Bast, A. Pleiotropic Benefit of Monomeric and Oligomeric Flavanols on Vascular Health - A Randomized Controlled Clinical Pilot Study. PLoS ONE 2011, 6, e28460. [Google Scholar] [CrossRef] [PubMed]
- Milenkovic, D.; Declerck, K.; Guttman, Y.; Kerem, Z.; Claude, S.; Weseler, A.R.; Bast, A.; Schroeter, H.; Morand, C.; Berghe, W.V. (−)-Epicatechin metabolites promote vascular health through epigenetic reprogramming of endothelial-immune cell signaling and reversing systemic low-grade inflammation. Biochem. Pharmacol. 2020, 173, 113699. [Google Scholar] [CrossRef]
- Bowtell, J.; Kelly, V.G. Fruit-Derived Polyphenol Supplementation for Athlete Recovery and Performance. Sports Med. 2019, 49, 3–23. [Google Scholar] [CrossRef]
- Somerville, V.; Bringans, C.; Braakhuis, A. Polyphenols and Performance: A Systematic Review and Meta-Analysis. Sports Med. 2017, 47, 1589–1599. [Google Scholar] [CrossRef]
- Lepage, G.; Munoz, G.; Champagne, J.; Roy, C.C. Preparative steps necessary for the accurate measurement of malondialdehyde by high-performance liquid chromatography. Anal. Biochem. 1991, 197, 277–283. [Google Scholar] [CrossRef]
- Fischer, M.A.J.G.; Gransier, T.J.M.; Beckers, L.M.G.; Bekers, O.; Bast, A.; Haenen, G.R. Determination of the antioxidant capacity in blood. Clin. Chem. Lab. Med. 2005, 43, 735–740. [Google Scholar] [CrossRef]
- Ruijters, E.J.; Weseler, A.R.; Kicken, C.; Haenen, G.R.; Bast, A. The flavanol (-)-epicatechin and its metabolites protect against oxidative stress in primary endothelial cells via a direct antioxidant effect. Eur. J. Pharmacol. 2013, 715, 147–153. [Google Scholar] [CrossRef]
- Wasung, M.E.; Chawla, L.S.; Madero, M. Biomarkers of renal function, which and when? Clin. Chim. Acta 2015, 438, 350–357. [Google Scholar] [CrossRef]
- Poortmans, J.R.; Blommaert, E.; Baptista, M.; De Broe, M.E.; Nouwen, E.J. Evidence of differential renal dysfunctions during exercise in men. Graefe’s Arch. Clin. Exp. Ophthalmol. 1997, 76, 88–91. [Google Scholar] [CrossRef] [PubMed]
- Lipman, G.S.; Krabak, B.J.; Rundell, S.D.; Shea, K.M.; Badowski, N.; Little, C. Incidence and Prevalence of Acute Kidney Injury During Multistage Ultramarathons. Clin. J. Sport Med. 2016, 26, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.A.; Sierra, A.P.R.; Benetti, M.; Ghorayeb, N.; Sierra, C.A.; Kiss, M.A.P.D.M.; Cury-Boaventura, M.F. Impact of Hot Environment on Fluid and Electrolyte Imbalance, Renal Damage, Hemolysis, and Immune Activation Postmarathon. Oxidative Med. Cell. Longev. 2017, 2017, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, L.; Walter, E.; Venn, R.; Galloway, R.; Pitsiladis, Y.; Sardat, F.; Forni, L.G. Acute kidney injury associated with endurance events—is it a cause for concern? A systematic review. BMJ Open Sport Exerc. Med. 2017, 3, e000093. [Google Scholar] [CrossRef]
- Nieman, D.C.; Henson, D.A.; Dumke, C.L.; Oley, K.; McAnulty, S.R.; Davis, J.M.; Murphy, E.A.; Utter, A.C.; Lind, R.H.; McAnulty, L.S. Ibuprofen use, endotoxemia, inflammation, and plasma cytokines during ultramarathon competition. Brain Behav. Immun. 2006, 20, 578–584. [Google Scholar] [CrossRef]
- Lipman, G.S.; Shea, K.; Christensen, M.; Phillips, C.; Burns, P.; Higbee, R.; Koskenoja, V.; Eifling, K.; Krabak, B.J. Ibuprofen vs. placebo effect on acute kidney injury in ultramarathons: A randomised controlled trial. Emerg. Med. J. 2017, 34, 637–642. [Google Scholar] [CrossRef]
- Lundberg, T.R.; Howatson, G. Analgesic and anti-inflammatory drugs in sports: Implications for exercise performance and training adaptations. Scand. J. Med. Sci. Sports 2018, 28, 2252–2262. [Google Scholar] [CrossRef]
- Da Silva, E.; Pinto, R.S.; Cadore, E.L.; Kruel, L.F. Nonsteroidal Anti-Inflammatory Drug Use and Endurance During Running in Male Long-Distance Runners. J. Athl. Train. 2015, 50, 295–302. [Google Scholar] [CrossRef]
- De Souza, R.F.; De Matos, D.G.; Ferreira, A.R.P.; Chilibeck, P.; Barros, N.D.A.; De Oliveira, A.S.; Cercato, L.M.; Da Silva, D.S.; Aidar, F.J. The effect of ibuprofen on muscle, hematological and renal function, hydric balance, pain, and performance during intense long-distance running. J. Strength Cond. Res. 2018, 1. [Google Scholar] [CrossRef]
- Hoffman, M.D.; Weiss, R.H. Does Acute Kidney Injury From an Ultramarathon Increase the Risk for Greater Subsequent Injury? Clin. J. Sport Med. 2016, 26, 417–422. [Google Scholar] [CrossRef]
- Mielgo-Ayuso, J.; Calleja-González, J.; Refoyo, I.; León-Guereño, P.; Martínez, A.C.; Del Coso, J. Exercise-Induced Muscle Damage and Cardiac Stress During a Marathon Could be Associated with Dietary Intake During the Week Before the Race. Nutrients 2020, 12, 316. [Google Scholar] [CrossRef] [PubMed]
- Weseler, A.R.; Bast, A. Masquelier’s grape seed extract: From basic flavonoid research to a well-characterized food supplement with health benefits. Nutr. J. 2017, 16, 5. [Google Scholar] [CrossRef] [PubMed]
- Kashi, D.; Shabir, A.; Da Boit, M.; Bailey, S.J.; Higgins, M.F. The Efficacy of Administering Fruit-Derived Polyphenols to Improve Health Biomarkers, Exercise Performance and Related Physiological Responses. Nutrients 2019, 11, 2389. [Google Scholar] [CrossRef] [PubMed]
- Vargas, F.; Romecín, P.; Guillen, A.I.G.; Wangesteen, R.; Vargas-Tendero, P.; Paredes, M.D.; Atucha, N.M.; Garcia-Estañ, J. Flavonoids in Kidney Health and Disease. Front. Physiol. 2018, 9, 9. [Google Scholar] [CrossRef]
- Wang, Q.-Z.; Gao, H.-Q.; Liang, Y.; Zhang, J.; Wang, J.; Qiu, J. Cofilin1 is involved in hypertension-induced renal damage via the regulation of NF-κB in renal tubular epithelial cells. J. Transl. Med. 2015, 13, 323. [Google Scholar] [CrossRef]
- Bolignano, D.; Donato, V.; Coppolino, G.; Campo, S.; Buemi, A.; Lacquaniti, A.; Buemi, M. Neutrophil Gelatinase–Associated Lipocalin (NGAL) as a Marker of Kidney Damage. Am. J. Kidney Dis. 2008, 52, 595–605. [Google Scholar] [CrossRef]
- De Geus, H.R.H.; Bakker, J.; Lesaffre, E.M.E.H.; Le Noble, J.L.M.L. Neutrophil Gelatinase-associated Lipocalin at ICU Admission Predicts for Acute Kidney Injury in Adult Patients. Am. J. Respir. Crit. Care Med. 2011, 183, 907–914. [Google Scholar] [CrossRef]
- De Geus, H.R.; Woo, J.G.; Wang, Y.; Devarajan, P.; Betjes, M.G.; Le Noble, J.L.; Bakker, J. Urinary Neutrophil Gelatinase-Associated Lipocalin Measured on Admission to the Intensive Care Unit Accurately Discriminates between Sustained and Transient Acute Kidney Injury in Adult Critically Ill Patients. Nephron Extra 2011, 1, 9–23. [Google Scholar] [CrossRef]
- Lippi, G.; Sanchis-Gomar, F. Exertional hematuria: Definition, epidemiology, diagnostic and clinical considerations. Clin. Chem. Lab. Med. 2019, 57, 1818–1828. [Google Scholar] [CrossRef]



| Compound | Vitis vinifera L. extract% (m/m) | Pinus pinaster L. extract% (m/m) |
|---|---|---|
| Total polyphenols | 90–95 | ca. 80 |
| Flavan-3-ols | 60–70 | n.a. |
| Other polyphenols | 20–35 | n.a. |
| Monomers | 25–30 | 7–20 |
| Oligomeric proanthocyanidins (2–5 units) | 35–40 | 19–38 |
| Dimeric proanthocyanidins | 30–35 | 11–22 |
| Polymers (>6 units) | 1–5 | 4–13 |
| Taxifolin | n.a. | <3 |
| Water | ca. 5 | ca. 4 |
| Characteristics | Placebo, n = 25 | MOF-VVPP, n = 29 | Total, n = 54 | p # |
|---|---|---|---|---|
| Male/female, n | 15/10 | 18/11 | 33/21 | 0.885 |
| Age, y | 47 ± 15 | 47 ± 11 | 47 13 | 0.823 |
| Blood pressure, mm Hg | ||||
| Systolic | 131 ± 14 | 132 ± 17 | 131 ± 15 | 0.755 |
| Diastolic | 82 ± 7 | 83 ± 11 | 83 ± 9 | 0.807 |
| HR, bmp | 59 ± 9 | 58 ± 8 | 59 ± 8 | 0.695 |
| BMI, kg/m2 | 24.8 ± 2.5 | 24.4 ± 2.1 | 24.6 ± 2.3 | 0.557 |
| Average training for 3 months before half-marathon, km/week | 30 (25–45) | 35 (25–48) | 30 (25–45) | 0,848 |
| Long-distance running experience, years | 10 (5–15) | 9 (5–14) | 9 (5–15) | 0.721 |
| Previous races, n | ||||
| 42.2 km | 0(0–3) | 1 (0–4) | 0 (0–4) | 0.722 |
| 21.1 km | 5.0 (1–12) * | 6 (3–15) | 5.5 (2.8–15) | 0.423 |
| 10 km | 10 (4–20) | 10 (4–17.5) | 10 (4–20) | 0.869 |
| 5 km | 4.5 (0.3–10) | 1 (0–10) | 3 (0–10) | 0.266 |
| Reported best 21.1-km finishing time, min | 110 (101–117) | 110 (96.5–127.5) | 110 (98–121) | 0.889 |
| Characteristics | Placebo, n = 25 | MOF-VVPP, n = 29 | p # | ||
|---|---|---|---|---|---|
| Before | After | Before | After | ||
| Body weight, kg | 74.7 ± 10.7 | 74.0 ± 10.7 * | 74.8 ± 11.2 | 74.2 ± 11.1 * | 0.935 |
| Change of body weight, % | 0.87 ± 0.69 | 0.77 ± 0.84 | 0.599 | ||
| Finishing time, min | |||||
| Male | 122.4 ± 11.2 | 117.4 ± 19.5 | 0.407 | ||
| Female | 122.9 ± 13.8 | 124.0 ± 19.2 | 0.882 | ||
| Tiredness, AU | 4.25 ± 2.52 | 4.89 ± 2.89 | 0.380 | ||
| Muscle pain, AU | 2.46 ± 3.11 | 2.97 ± 3.04 | 0.552 | ||
| Parameter | Placebo | MOF-VVPP | p * | ||
|---|---|---|---|---|---|
| Before | After | Before | After | ||
| Specific gravity, kg/L | 1.007 (1.005–1.018) | 1.015 b (1.006–1.024) | 1.006 (1.005–1.012) | 1.013 b (1.007–1.019) | 0.573 |
| Osmolality, mOsm/kg | 314 (207–704) | 407 (226–657) | 258 (164–485) | 374 (219–569) | 0.490 |
| Creatinine, mmol/L | 4.0 (2.4–7.8) | 10.7 c (6.3–18.0) | 3.6 (1.8–6.6) | 8.5 a (5.1–15.9) | 0.574 |
| Albumin **, mg/L | 5.0 (5.0–5.0) | 51.5 c (35.0–198.0) | 5.0 (5.0–5.0) | 39.0 c (13.8–105.3) | 0.335 |
| Albumin/Creatinine, mg/mmol | 0.47 (0.34–0.79) | 6.90 c (2.35–11.25) | 0.51 (0.35–1.02) | 4.2 c (2.2–10.7) | 0.534 |
| Urea, mmol/L | 131 (82–306) | 160 (98–225) | 121 (62–246) | 126 (88–281) | 0.842 |
| Sodium, mmol/L | 55.5 (28.0–121.8) | 50.0 (26.5–81.0) | 44.0 (25.0–72.1) | 34.5 (24.0–56.3) | 0.130 |
| Potassium, mmol/L | 36.1 (27.5–58.4) | 63.3 a (35.0–93.8) | 30.5 (16.1–54.0) | 53.1 a (29.8–86.2) | 0.475 |
| Chloride, mmol/L | 68.5 (39.3–138.3) | 40.0 b (30.0–90.0) | 55.0 (34.0–86.5) | 52.5 (35.3–72.8) | 0.762 |
| Uric acid, mmol/L | 0.91 (0.56–1.96) | 1.47 (0.64–2.20) | 0.95 (0.48–1.47) | 1.19 (0.56–2.00) | 0.486 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Semen, K.O.; Weseler, A.R.; Janssen, M.J.W.; Drittij-Reijnders, M.-J.; le Noble, J.L.M.L.; Bast, A. Effects of Monomeric and Oligomeric Flavanols on Kidney Function, Inflammation and Oxidative Stress in Runners: A Randomized Double-Blind Pilot Study. Nutrients 2020, 12, 1634. https://doi.org/10.3390/nu12061634
Semen KO, Weseler AR, Janssen MJW, Drittij-Reijnders M-J, le Noble JLML, Bast A. Effects of Monomeric and Oligomeric Flavanols on Kidney Function, Inflammation and Oxidative Stress in Runners: A Randomized Double-Blind Pilot Study. Nutrients. 2020; 12(6):1634. https://doi.org/10.3390/nu12061634
Chicago/Turabian StyleSemen, Khrystyna O., Antje R. Weseler, Marcel J. W. Janssen, Marie-José Drittij-Reijnders, Jos L. M. L. le Noble, and Aalt Bast. 2020. "Effects of Monomeric and Oligomeric Flavanols on Kidney Function, Inflammation and Oxidative Stress in Runners: A Randomized Double-Blind Pilot Study" Nutrients 12, no. 6: 1634. https://doi.org/10.3390/nu12061634
APA StyleSemen, K. O., Weseler, A. R., Janssen, M. J. W., Drittij-Reijnders, M.-J., le Noble, J. L. M. L., & Bast, A. (2020). Effects of Monomeric and Oligomeric Flavanols on Kidney Function, Inflammation and Oxidative Stress in Runners: A Randomized Double-Blind Pilot Study. Nutrients, 12(6), 1634. https://doi.org/10.3390/nu12061634





