The Effect of Ingesting Carbohydrate and Proteins on Athletic Performance: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Abstract
1. Introduction
2. Materials and Methods
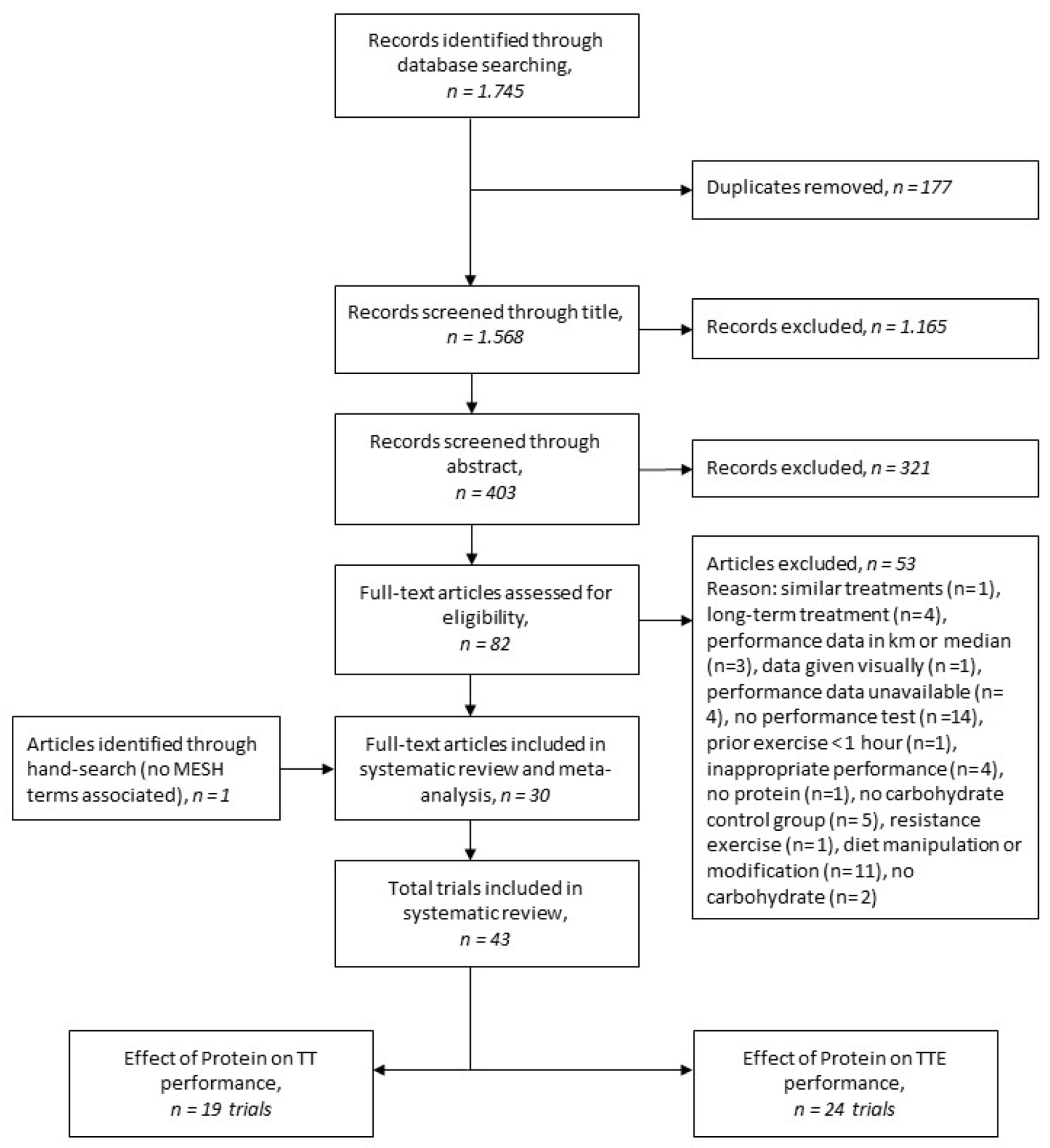
2.1. Literature Search
2.2. Inclusion and Exclusion Criteria
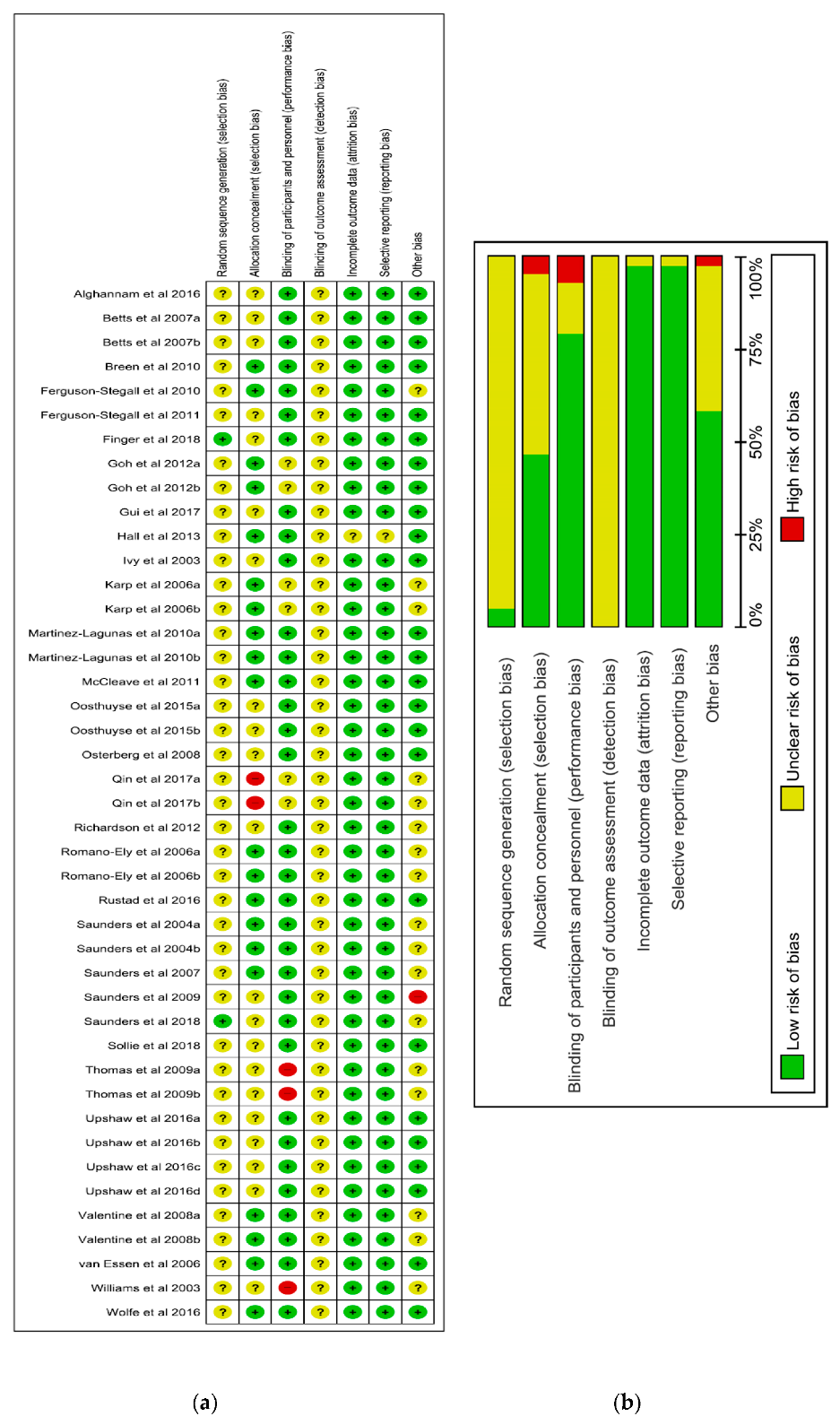
2.3. Methodological Quality Assessment
2.4. Data Extraction
2.5. Statistical Analysis
3. Results
3.1. Overview of Included Studies
3.1.1. Subjects
3.1.2. Study Protocol
3.1.3. Intervention and Control Products
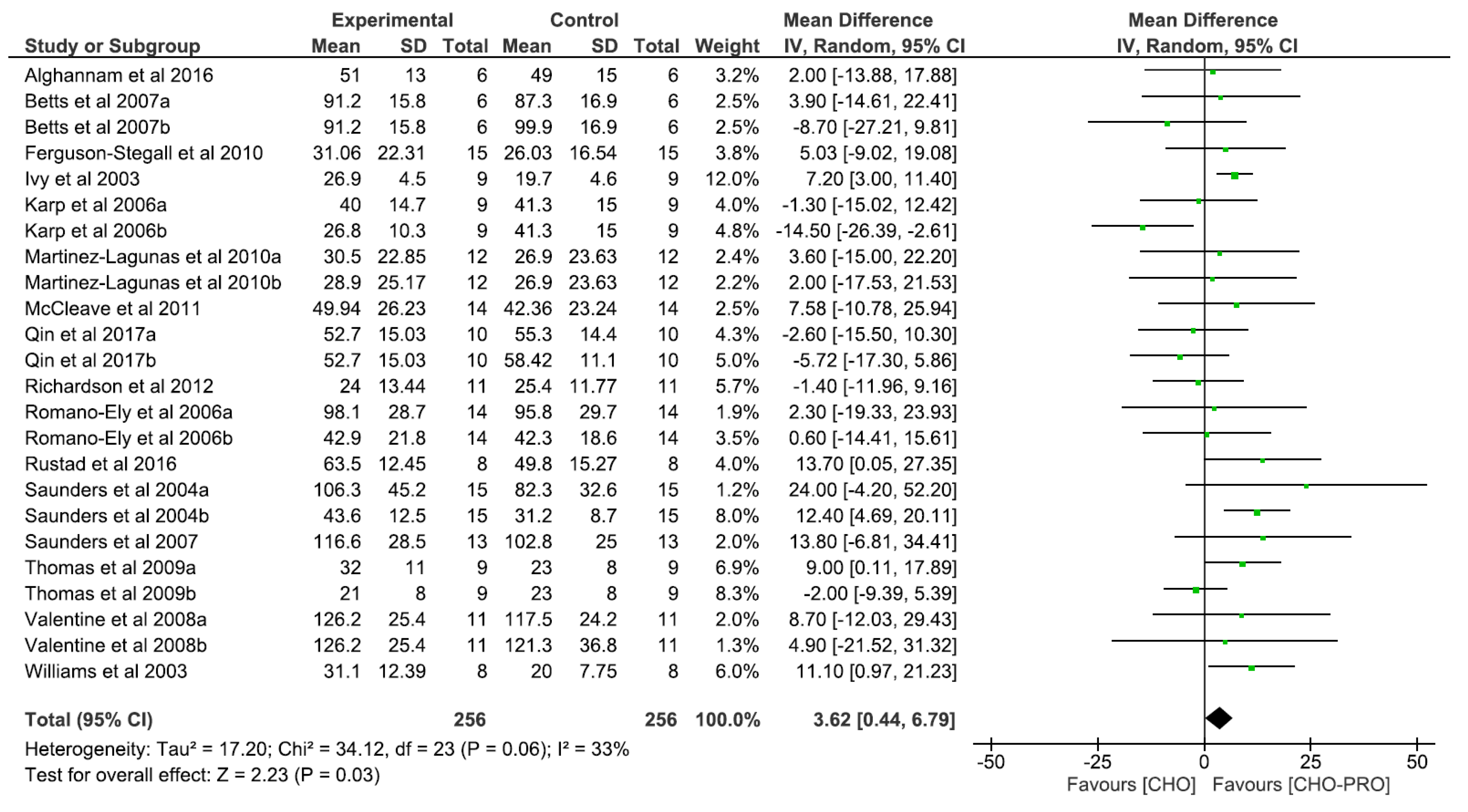
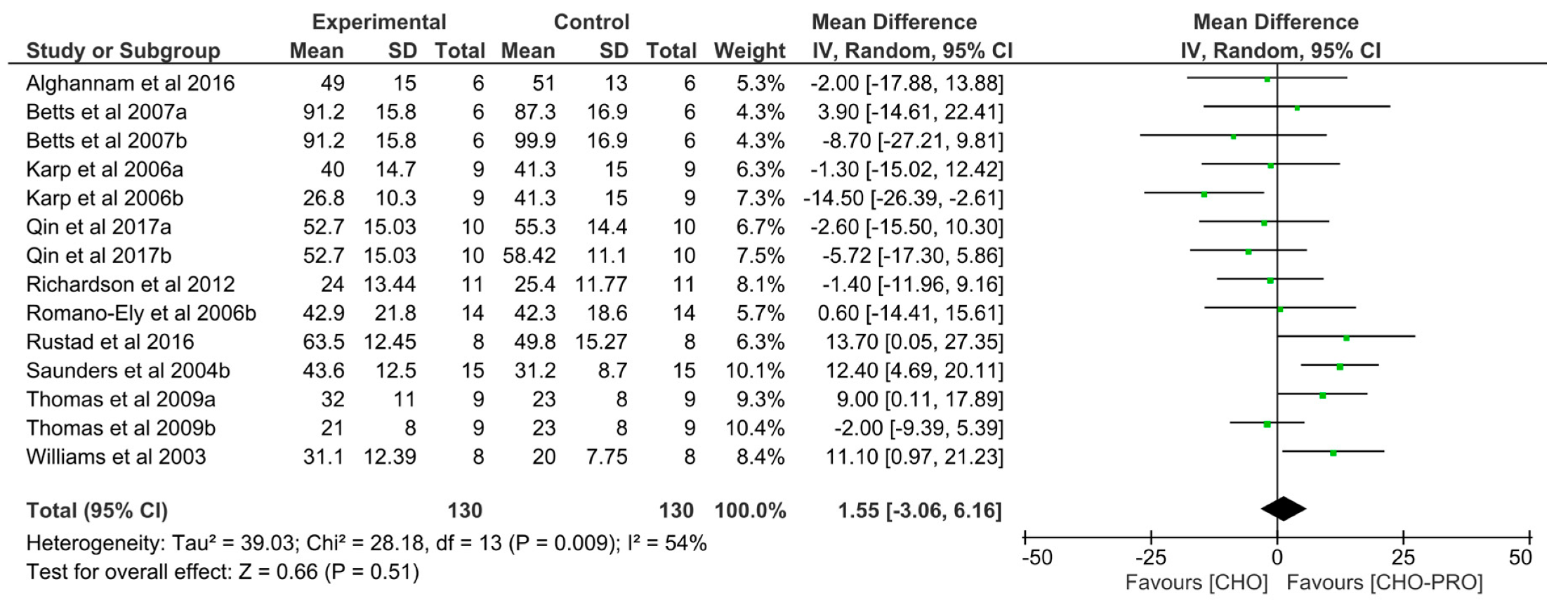
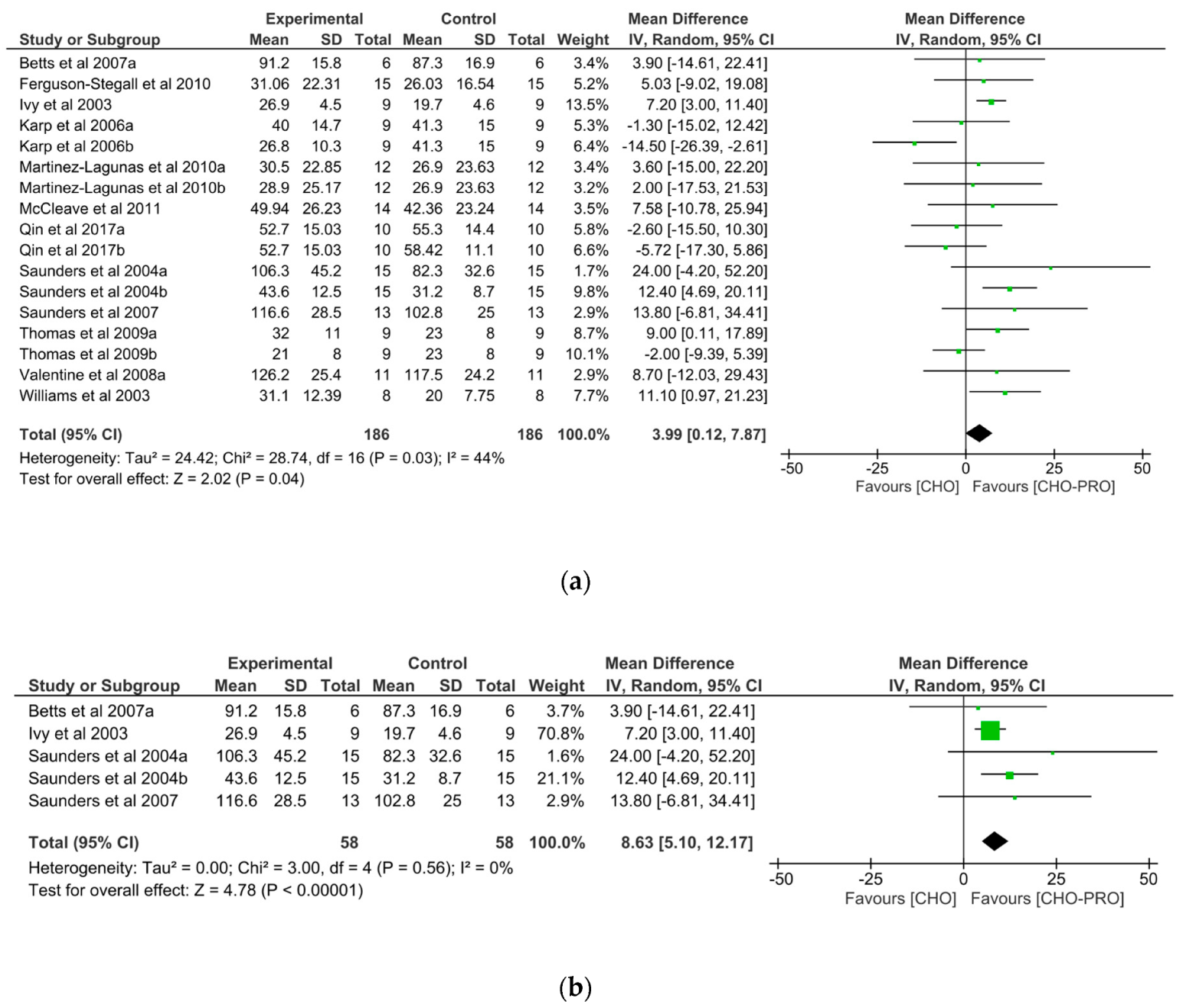
3.2. Effect of Carbohydrate and Protein (CHO-PRO) vs. Carbohydrate (CHO) Supplementation on Time-To-Exhaustion (TTE)
3.3. Effect of CHO-PRO vs. CHO Intake during TTE on Performance
3.4. Effect of CHO-PRO vs. CHO Intake during Recovery on Subsequent TTE
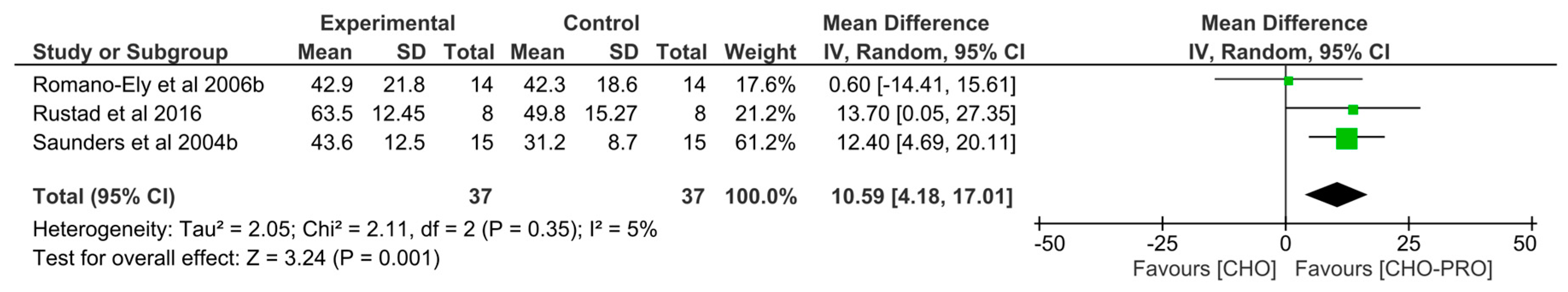
3.4.1. Effect of CHO-PRO vs. CHO Intake during Short-Term Recovery on Subsequent TTE
3.4.2. Effect of CHO-PRO vs. CHO Intake during Long-Term Recovery on Subsequent TTE
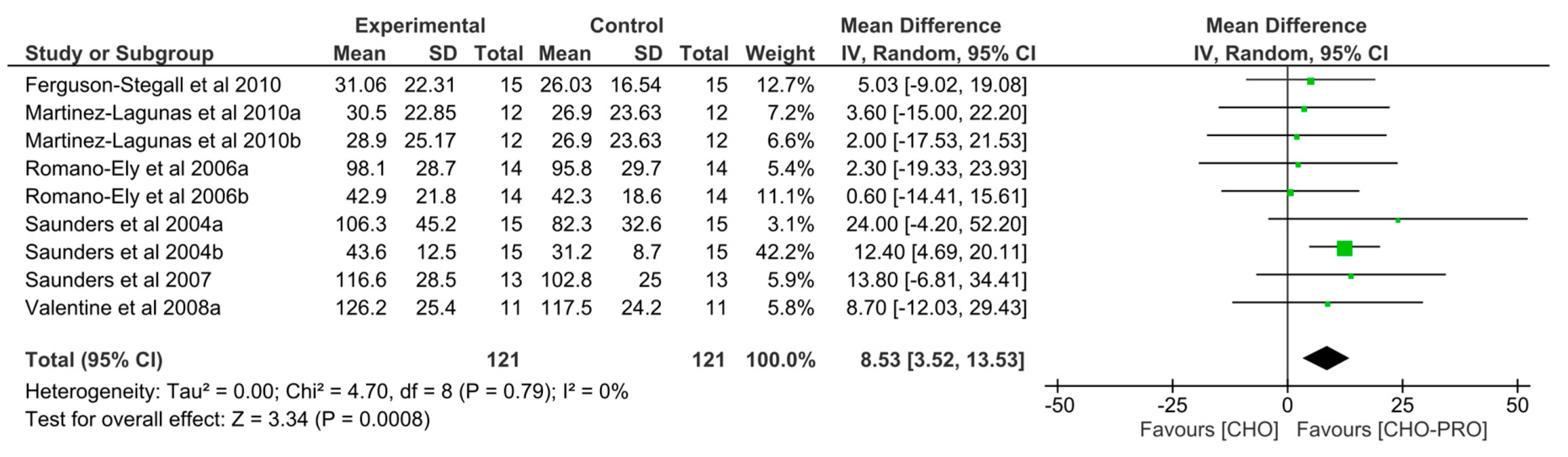
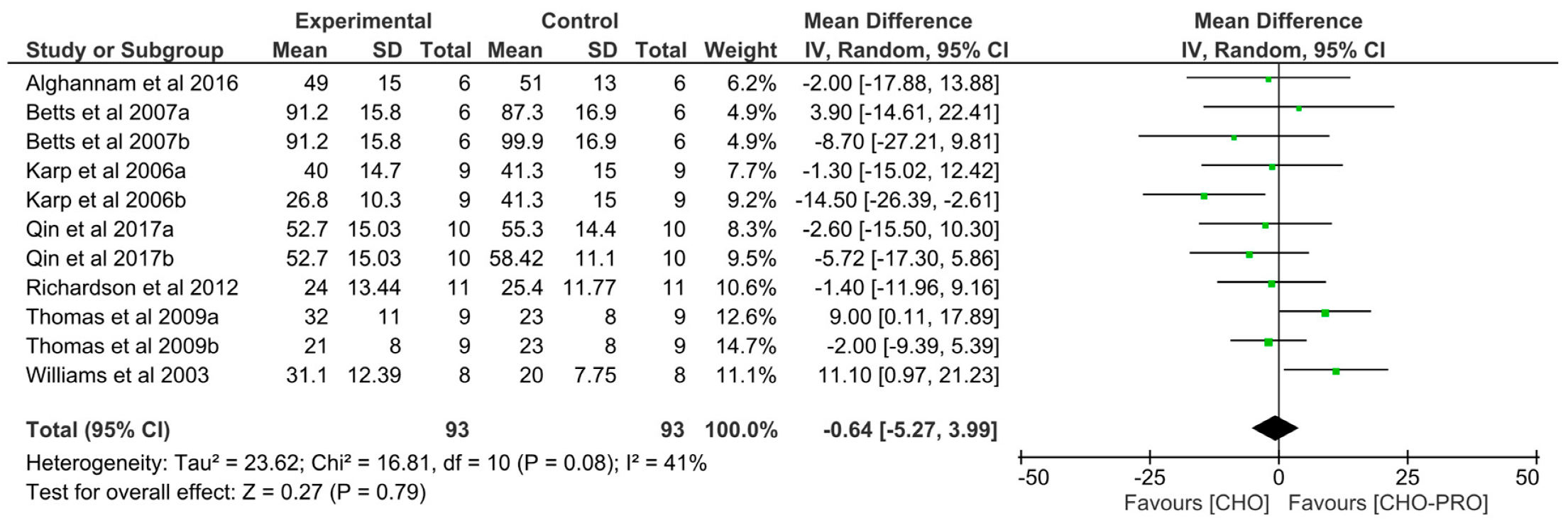
3.5. Effect of Isocaloric Supplementation (i.e., CHO-PRO vs. CHO) on TTE Performance
3.6. Effect of Non-Isocaloric CHO-PRO vs. CHO Supplementation on TTE Performance
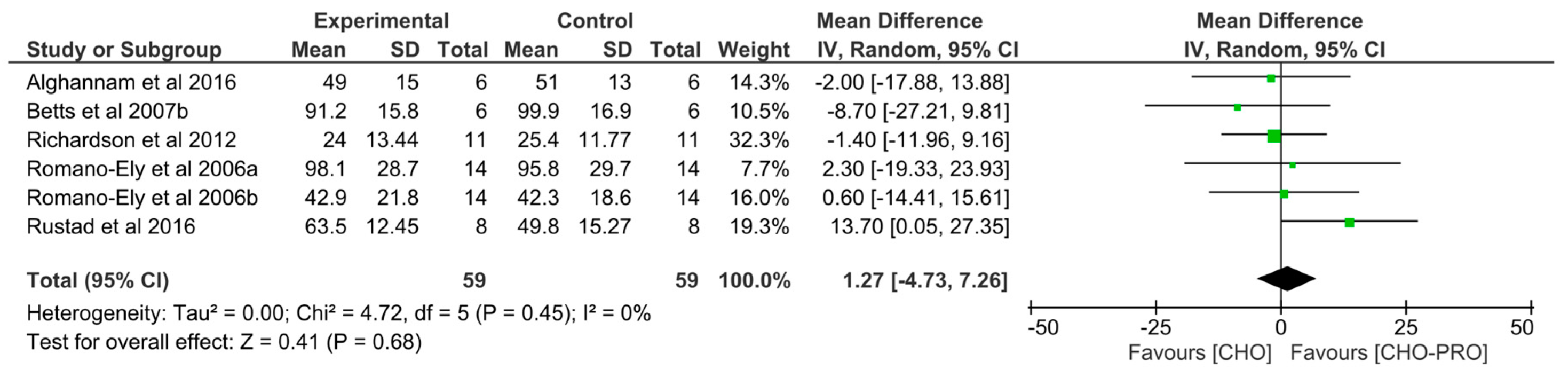
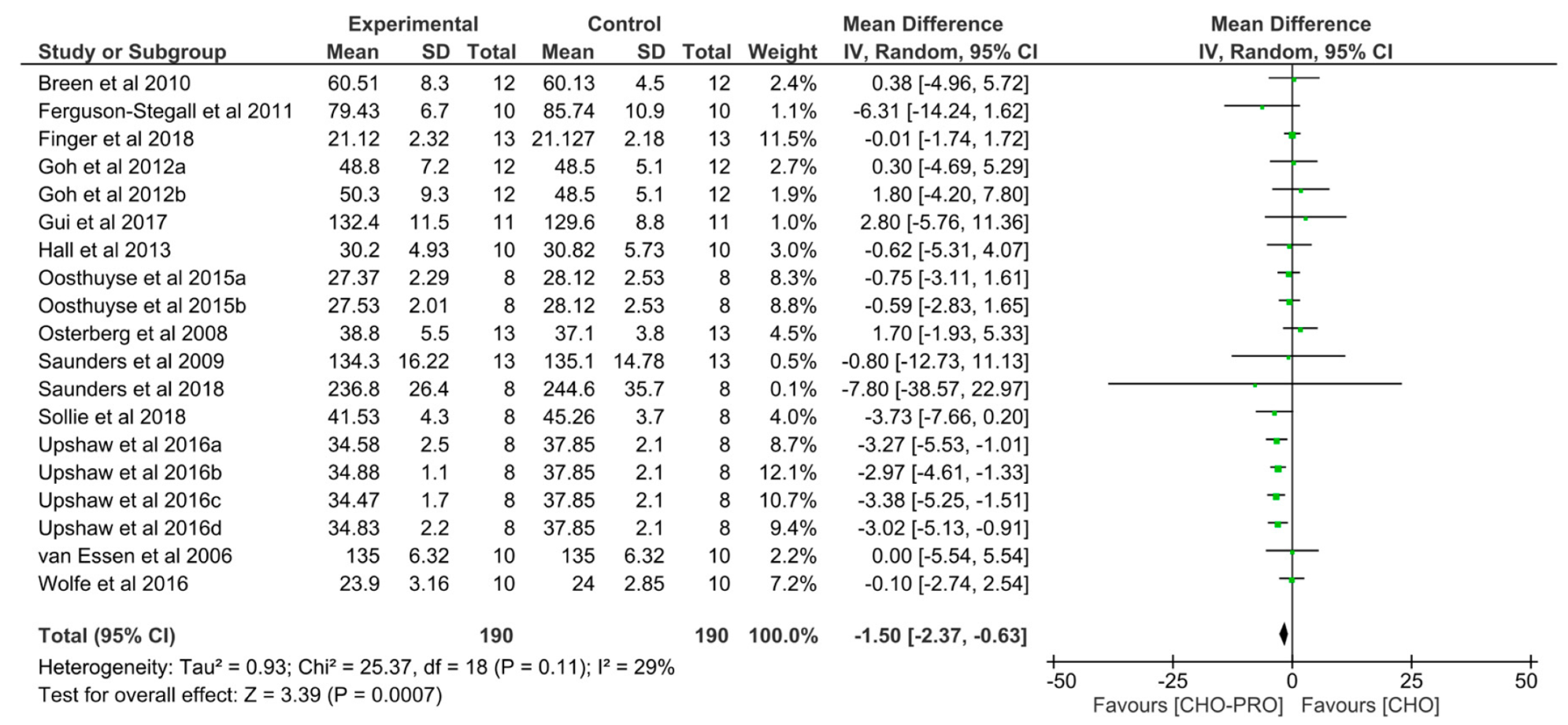
3.7. Effect of CHO-PRO vs. CHO Supplementation on Time-Trial (TT) Performance
4. Discussion
4.1. Future Research
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Beelen, M.; Burke, L.M.; Gibaia, M.J.; Loon, L.J.C. Van nutritional strategies to promote postexercise recovery synthesis. Int. J. Sport Nutr. Exerc. Metab. 2010, 20, 515–533. [Google Scholar] [CrossRef] [PubMed]
- Jentjens, R.; Jeukendrup, A.E. Determinants of post-exercise glycogen synthesis during short-term recovery. Sports Med. 2003, 33, 117–144. [Google Scholar] [CrossRef]
- Thomas, D.T.; Erdman, K.A.; Burke, L.M. American college of sports medicine joint position statement nutrition and athletic performance. Med. Sci. Sport. Exerc. 2016, 48, 543–568. [Google Scholar]
- Romijn, J.A.; Coyle, E.F.; Sidossis, L.S.; Gastaldelli, A.; Horowitz, J.F.; Endert, E.; Wolfe, R.R. Regulation of endogenous fat and carbohydrate metabolism in relation to exercise intensity and duration. Am. J. Physiol. 1993, 265, E380–E391. [Google Scholar] [CrossRef] [PubMed]
- van Loon, L.J.; Greenhaff, P.L.; Constantin-Teodosiu, D.; Saris, W.H.; Wagenmakers, A.J. The effects of increasing exercise intensity on muscle fuel utilisation in humans. J. Physiol. 2001, 536, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Ørtenblad, N.; Nielsen, J.; Saltin, B.; Holmberg, H.C. Role of glycogen availability in sarcoplasmic reticulum Ca 2+ kinetics in human skeletal muscle. J. Physiol. 2011, 589, 711–725. [Google Scholar] [CrossRef]
- Gejl, K.D.; Hvid, L.G.; Frandsen, U.; Jensen, K.; Sahlin, K.; Ørtenblad, N. Muscle glycogen content modifies SR Ca2+ release rate in elite endurance athletes. Med. Sci. Sports Exerc. 2014, 46, 496–505. [Google Scholar] [CrossRef]
- van Loon, L.J.; Saris, W.H.; Kruijshoop, M.; Wagenmakers, A.J. Maximizing postexercise muscle glycogen synthesis: Carbohydrate supplementation and the application of amino acid or protein. Am. J. Clin. Nutr. 2000, 72, 106–111. [Google Scholar] [CrossRef]
- Ivy, J.L.; Katz, A.L.; Cutler, C.L.; Sherman, W.M.; Coyle, E.F. Muscle glycogen synthesis after exercise: Effect of time of carbohydrate ingestion. J. Appl. Physiol. 1988, 64, 1480–1485. [Google Scholar] [CrossRef]
- Rustad, P.I.; Sailer, M.; Cumming, K.T.; Jeppesen, P.B.; Kolnes, K.J.; Sollie, O.; Franch, J.; Ivy, J.L.; Daniel, H.; Jensen, J. Intake of protein plus carbohydrate during the first two hours after exhaustive cycling improves performance the following day. PLoS ONE 2016, 11, 1–25. [Google Scholar] [CrossRef]
- Richardson, K.L.; Coburn, J.W.; Beam, W.C.; Brown, L.E. Effects of isocaloric carbohydrate vs. carbohydrate-protein supplements on cycling time to exhaustion. J. Strength Cond. Res. 2012, 26, 1361–1365. [Google Scholar] [CrossRef]
- Osterberg, K.L.; Zachwieja, J.J.; Smith, J.W. Carbohydrate and carbohydrate + protein for cycling time-trial performance. J. Sports Sci. 2008, 26, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Betts, J.; Williams, C.; Duffy, K.; Gunner, F. The influence of carbohydrate and protein ingestion during recovery from prolonged exercise on subsequent endurance performance. J. Sports Sci. 2007, 25, 1449–1460. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.B.; Raven, P.B.; Fogt, D.L.; Ivy, J.L. Effects of recovery beverages on glycogen restoration and endurance exercise performance. J. Strength Cond. Res. 2003, 17, 12–19. [Google Scholar] [PubMed]
- Saunders, M.J.; Luden, N.D.; Herrick, J.E. Consumption of an oral carbohydrate-protein gel improves cycling endurance and prevents postexercise muscle damage. J. Strength Cond. Res. 2007, 21, 678–684. [Google Scholar]
- Saunders, M.J.; Kane, M.D.; Kent Todd, M. Effects of a carbohydrate-protein beverage on cycling endurance and muscle damage. Med. Sci. Sports Exerc. 2004, 36, 1233–1238. [Google Scholar] [CrossRef]
- Romano-Ely, B.C.; Todd, M.K.; Saunders, M.J.; St. Laurent, T. Effect of an isocaloric carbohydrate-protein-antioxidant drink on cycling performance. Med. Sci. Sports Exerc. 2006, 38, 1608–1616. [Google Scholar] [CrossRef]
- Valentine, R.J.; Saunders, M.J.; Todd, M.K.; Laurent, T.G.S. Influence of carbohydrate-protein beverage on cycling endurance and indices of muscle disruption. Int. J. Sport Nutr. Exerc. Metab. 2008, 18, 363–378. [Google Scholar] [CrossRef]
- Thomas, K.; Morris, P.; Stevenson, E. Improved endurance capacity following chocolate milk consumption compared with 2 commercially available sport drinks. Appl. Physiol. Nutr. Metab. 2009, 34, 78–82. [Google Scholar] [CrossRef]
- Qin, L.; Wang, Q.R.; Fang, Z.L.; Wang, T.; Yu, A.Q.; Zhou, Y.J.; Zheng, Y.; Yi, M.Q. Effects of three commercially available sports drinks on substrate metabolism and subsequent endurance performance in a postprandial state. Nutrients 2017, 9, 377. [Google Scholar] [CrossRef]
- Martinez-Lagunas, V.; Ding, Z.; Bernard, J.R.; Wang, B.; Ivy, J.L. Added protein maintains efficacy of a low-carbohydrate sports drink. J. Strength Cond. Res. 2010, 24, 48–59. [Google Scholar] [CrossRef] [PubMed]
- Ferguson-Stegall, L.; McCleave, E.L.; Ding, Z.; Kammer, L.M.; Wang, B.; Doerner, P.G.; Liu, Y.; Ivy, J.L. The effect of a low carbohydrate beverage with added protein on cycling endurance performance in trained athletes. J. Strength Cond. Res. 2010, 24, 2577–2586. [Google Scholar] [CrossRef] [PubMed]
- Ivy, J.L.; Res, P.T.; Sprague, R.C.; Widzer, M.O. Effect of a carbohydrate-protein supplement on endurance performance during exercise of varying intensity. Int. J. Sport Nutr. Exerc. Metab. 2003, 13, 382–395. [Google Scholar] [CrossRef] [PubMed]
- McCleave, E.L.; Ferguson-Stegall, L.; Ding, Z.; Doerner, P.G.; Wang, B.; Kammer, L.M.; Ivy, J.L. A low carbohydrate-protein supplement improves endurance performance in female athletes. J. Strength Cond. Res. 2011, 25, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Karp, J.R.; Johnston, J.D.; Tecklenburg, S.; Mickleborough, T.D.; Fly, A.D.; Stager, J.M. Chocolate milk as a post-exercise recovery aid. Int. J. Sport Nutr. Exerc. Metab. 2006, 16, 78–91. [Google Scholar] [CrossRef] [PubMed]
- Alghannam, A.F.; Jedrzejewski, D.; Bilzon, J.; Thompson, D.; Tsintzas, K.; Betts, J.A. Influence of post-exercise carbohydrate-protein ingestion on muscle glycogen metabolism in recovery and subsequent running exercise. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 572–580. [Google Scholar] [CrossRef]
- Saunders, M.J.; Moore, R.W.; Luden, N.D.; Pratt, C.A.; Kies, A.K. Carbohydrate and protein hydrolysate coingestion’s improvement of late- exercise time-trial performance. Int. J. Sport Nutr. Exerc. Metab. 2009, 19, 136–149. [Google Scholar] [CrossRef]
- Hall, A.H.; Leveritt, M.D.; Ahuja, K.D.K.; Shing, C.M. Coingestion of carbohydrate and protein during training reduces training stress and enhances subsequent exercise performance. Appl. Physiol. Nutr. Metab. 2013, 38, 597–604. [Google Scholar] [CrossRef]
- Upshaw, A.U.; Wong, T.S.; Bandegan, A.; Lemon, P.W.R. Cycling time trial performance 4 hours after glycogen-lowering exercise is similarly enhanced by recovery nondairy chocolate beverages versus chocolate milk. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 65–70. [Google Scholar] [CrossRef]
- Gui, Z.; Sun, F.; Si, G.; Chen, Y. Effect of protein and carbohydrate solutions on running performance and cognitive function in female recreational runners. PLoS ONE 2017, 12, 1–13. [Google Scholar] [CrossRef]
- Sollie, O.; Jeppesen, P.B.; Tangen, D.S.; Jernerén, F.; Nellemann, B.; Valsdottir, D.; Madsen, K.; Turner, C.; Refsum, H.; Skålhegg, B.S.; et al. Protein intake in the early recovery period after exhaustive exercise improves performance the following day. J. Appl. Physiol. 2018, 125, 1731–1742. [Google Scholar] [CrossRef] [PubMed]
- Van Essen, M.; Gibala, M.J. Failure of protein to improve time trial performance when added to a sports drink. Med. Sci. Sports Exerc. 2006, 38, 1476–1483. [Google Scholar] [CrossRef] [PubMed]
- Breen, L.; Tipton, K.D.; Jeukendrup, A.E. No effect of carbohydrate-protein on cycling performance and indices of recovery. Med. Sci. Sports Exerc. 2010, 42, 1140–1148. [Google Scholar] [CrossRef] [PubMed]
- Ferguson-Stegall, L.; McCleave, E.L.; Ding, Z.; Doerner, P.G., III; Wang, B.; Liao, Y.H.; Kammer, L.; Liu, Y.; Hwang, J.; Dessard, B.M.; et al. Postexercise Carbohydrate-Protein supplementation improves subsequent exercise performance and intracellular signaling for protein synthesis. J. Strength Cond. Res. 2011, 25, 1210–1224. [Google Scholar] [CrossRef] [PubMed]
- Goh, Q.; Boop, C.A.; Luden, N.D.; Smith, A.G.; Womack, C.J.; Saunders, M.J. Recovery from cycling exercise: Effects of carbohydrate and protein beverages. Nutrients 2012, 4, 568–584. [Google Scholar] [CrossRef]
- Wolfe, A.S.; Brandt, S.A.; Krause, I.A.; Mavison, R.W.; Aponte, J.A.; Ferguson-Stegall, L.M. Shorter duration time trial performance and recovery is not improved by inclusion of protein in a multiple carbohydrate supplement. J. Strength Cond. Res. 2017, 31, 2509–2518. [Google Scholar] [CrossRef]
- Oosthuyse, T.; Carstens, M.; Millen, A.M.E. Whey or casein hydrolysate with carbohydrate for metabolism and performance in cycling. Int. J. Sports Med. 2015, 36, 636–646. [Google Scholar] [CrossRef]
- Finger, D.; Farinha, J.B.; Brusco, C.M.; Boeno, F.P.; Cadore, E.L.; Pinto, R.S.; Lanferdini, F.J.; Helal, L. Ingestion of carbohydrate or carbohydrate plus protein does not enhance performance during endurance exercise: A randomized crossover placebo-controlled clinical trial. Appl. Physiol. Nutr. Metab. 2018, 43, 937–944. [Google Scholar] [CrossRef]
- Saunders, M.J.; Luden, N.D.; DeWitt, C.R.; Gross, M.C.; Rios, A.D. Protein supplementation during or following a marathon run influences post-exercise recovery. Nutrients 2018, 10, 1–14. [Google Scholar]
- Zawadzki, K.M.; Yaspelkis, B.B.; Ivy, J.L. Carbohydrate-protein complex increases the rate of muscle glycogen storage after exercise. J. Appl. Physiol. 1992, 72, 1854–1859. [Google Scholar] [CrossRef]
- Levenhagen, D.K.; Carr, C.; Carlson, M.G.; Maron, D.J.; Borel, M.J.; Flakoll, P.J. Postexercise protein intake enhances whole-body and leg protein accretion in humans. Med. Sci. Sports Exerc. 2002, 34, 828–837. [Google Scholar] [CrossRef] [PubMed]
- Stepto, N.K.; Martin, D.T.; Fallon, K.E.; Hawley, J.A. Metabolic demands of intense aerobic interval training in competitive cyclists. Med. Sci. Sports Exerc. 2001, 33, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, R.A.; Rasmussen, P.; Siebenmann, C.; Díaz, V.; Gassmann, M.; Pesta, D.; Gnaiger, E.; Nordsborg, N.B.; Robach, P.; Lundby, C. Determinants of time trial performance and maximal incremental exercise in highly trained endurance athletes. J. Appl. Physiol. 2011, 111, 1422–1430. [Google Scholar] [CrossRef]
- Karsten, B.; Baker, J.; Naclerio, F.; Klose, A.; Bianco, A.; Nimmerichter, A. Time trials versus time-to-exhaustion tests: Effects on critical power, W0, and oxygen-uptake kinetics. Int. J. Sports Physiol. Perform. 2018, 13, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Floyd, J.C.; Fajans, S.S.; Pek, S.; Thiffault, C.A.; Knopf, R.F.; Conn, J.W.; Arbor, A. Synergistic Effect of Certain Amino Acid Pairs upon Insulin Secretion in Man. Diabetes 1970, 19, 102–108. [Google Scholar] [CrossRef]
- Liu, Z.; Jeppesen, P.B.; Gregersen, S.; Chen, X.; Hermansen, K. Dose- and glucose-dependent effects of amino acids on insulin secretion from isolated mouse islets and clonal INS-1E beta-cells. Rev. Diabetes Stud. 2009, 5, 232–244. [Google Scholar] [CrossRef]
- Van Loon, L.J.C.; Saris, W.H.M.; Verhagen, H.; Wagenmakers, A.J.M. Plasma insulin responses after ingestion of different amino acid or. Am. J. Clin. Nutr. 2000, 72, 96–105. [Google Scholar] [CrossRef]
- Yue, Y.; Madsen, S.; Hedemann, M.S.; Knudsen, K.E.B.; Sparsø, F.V.; Laursen, A.; Jensen, H.M.; Knudsen, T.A.; Purup, S. Effect of food ingredients on glucagon-like peptide-1 secretion in STC-1 and HuTu-80 cells. Int. J. Food Sci. Technol. 2019, 54, 3149–3155. [Google Scholar] [CrossRef]
- Sucher, S.; Markova, M.; Hornemann, S.; Pivovarova, O.; Rudovich, N.; Thomann, R.; Schneeweiss, R.; Rohn, S.; Pfeiffer, A.F.H. Comparison of the effects of diets high in animal or plant protein on metabolic and cardiovascular markers in type 2 diabetes: A randomized clinical trial. Diabetes Obes. Metab. 2017, 19, 944–952. [Google Scholar] [CrossRef]
- Lopez-Sanchez, P.; Fredriksson, N.; Larsson, A.; Altskär, A.; Ström, A. High sugar content impacts microstructure, mechanics and release of calcium-alginate gels. Food Hydrocoll. 2018, 84, 26–33. [Google Scholar] [CrossRef]
- Needleman, I.; Ashley, P.; Meehan, L.; Petrie, A.; Weiler, R.; McNally, S.; Ayer, C.; Hanna, R.; Hunt, I.; Kell, S.; et al. Poor oral health including active caries in 187 UK professional male football players: Clinical dental examination performed by dentists. Br. J. Sports Med. 2016, 50, 41–44. [Google Scholar] [CrossRef] [PubMed]
| Citation, Year | Participants | Study Design | Preceding Exercise | Recovery Duration (hr) | Performance Exercise | Performance Duration (min) | Supplement Administration |
|---|---|---|---|---|---|---|---|
| Martinez-Lagunas et al. 2010 | 12 (5F, 7M) VO2max: 57.3 ± 2.7 Road cyclists/triathletes | Crossover DB | Cycling: 150 min, 55–75%VO2max | 0 | Cycling TTE 80% VO2max |
| 255.4 ± 9.1 mL during EX every 20 min |
| Betts et al. 2007 | 6 M VO2max: 61.4 ± 7.3 Recreationally active | Crossover DB | Running: 70 min, 70% VO2max | 4 | Running TTE 70% VO2max |
| 581 mL/hr during REC every 30 min |
| Saunders et al. 2004 | 15 M VO2peak: 52.6 ± 10.3 Cyclists | Crossover DB |
| 0 12–15 |
|
| 1.8 mL/kg every 15 min during EX, and 10 mL/kg PRE-EX |
| Romano-Ely et al. 2006 | 14 M VO2max: 59.8 ± 11.9 Physical active | Crossover DB |
| 0 22–24 |
|
| 2 mL/kg every 15 min during EX, 10 mL/kg BW and 10 mL/kg PRE-EX |
| Saunders et al. 2007 | 13 (5F, 8M) VO2peak: 57.6 ± 6.7 Recreationally active | Crossover DB | None | 0 | Cycling TTE 70% VO2max | CHO-PRO: 116.6 ± 28.5 CHO: 102.8 ± 25 | 0.146g CHO/kg BW/serv and 0.0365g PRO/kgBW/serv during EX and PRE-EX |
| Williams et al. 2003 | 8 M VO2max: 62.4 ± 1.1 Trained cyclists | Crossover N.D. | Cycling: 120 min, 65–75%VO2max | 4 | Cycling TTE 85% VO2max | CHO-PRO: 31.1 ± 12.39 CHO: 20 ± 7.75 | 355 mL during REC at t = 0 and 2 hr PRE-EX |
| Ivy et al. 2003 | 9 M VO2max: 61.3 ± 2.4 Cyclists | Crossover DB | Cycling: 3 hr; 45–75% VO2max | 0 | Cycling TTE 85% Pmax | CHO-PRO: 26.9 ± 4.5 CHO: 19.7 ± 4.6 | 200 mL every 20 min during EX |
| Ferguson-Stegall et al. 2010 | 15 (7F, 8M) VO2max: 56.15 ± 1.02 Cyclists/triathletes | Crossover DB | Cycling: 180 min, 45–70% VO2max | 0 | Cycling TTE 74–85% VO2max | CHO-PRO: 31.06 ± 22.31 CHO: 26.03 ± 16.54 | 275 mL every 20 min during EX |
| Rustad et al. 2016 | 8 M VO2max: 69.6 ± 1.3 Endurance trained | Crossover DB | Cycling to fatigue, 72–90% VO2max | ~18 | Cycling TTE 72% VO2max | CHO-PRO: 63.5 ± 12.45 CHO: 49.8 ± 15.27 | 1.2 g/kg BW/hr during REC every 30 min during the first 2 hr of REC |
| Thomas et al. 2009 | 9 M VO2max: 59.1 ± 1.5 N.D. | Crossover DB | Cycling to fatigue, 60–90% Pmax | 4 | Cycling TTE 70% Pmax |
| 1 g/kg BW during REC at t = 0 and 2 hr |
| Valentine et al. 2008 | 12 M VO2max: 53.4 ± 7.2 Active (incl. cycling) | Crossover DB | None | 0 | Cycling TTE 75% VO2max | CHO-PRO: 126.2 ± 25.4
| 250 mL every 15 min during EX |
| Richardson et al. 2012 | 11 (5F, 7M) M: VO2max: 50 ± 7.3 F: VO2max: 45.2 ± 3.9 Physically active (incl. cycling) | Crossover DB | Cycling TTE 75% VO2max | 3 | Cycling TTE 75% VO2max | CHO-PRO: 24 ± 13.44 CHO: 25.4 ± 11.77 | 1.5 g/kg BW/hr during REC every 30 min |
| Qin et al. 2017 | 10 M VO2max: 48.1 ± 8.4 Endurance runners/cyclists | Crossover DB | Cycling: 60 min, 70%VO2max | 6 | Cycling TTE 70% VO2max | CHO-PRO: 52.7 ± 15.03
| 1.8 mL/kg BW ever 15 min during EX, and 10 mL/kg PRE-EX |
| McCleave et al. 2011 | 14 F VO2max: 46.74 ± 1.6 Triathletes | Crossover DB | Cycling: 3 hr; 45–70% VO2max | 0 | Cycling TTE ~75% VO2max | CHO-PRO: 49.94 ± 26.23 CHO: 42.36 ± 23.24 | 275 mL every 20 min during EX |
| Karp et al. 2006 | 9 M VO2max: 65 ± 9 Cyclists | Crossover SB | Interval cycling workout | 4 | Cycling TTE 70% VO2max |
| 509.1±36 mL during REC |
| Alghannam et al. 2016 | 6 (5M, 1F) VO2max: 64 ± 4 Recreational runners | Crossover DB | Cycling TTE 70% VO2max | 4 | Cycling TTE 70% VO2max | CHO-PRO: 49 ± 15 CHO: 51 ± 13 | 10 mL/kg/hr during REC |
| Osterberg et al. 2008 | 13 M VO2max: 56 ± 0.12 Cyclists | Crossover DB | Cycling ~120 min SS | 0 | Cycling TT 7kJ/kg BW | CHO-PRO: 38.8 ± 5.5 CHO: 37.1 ± 3.8 | 250 mL every 15 min during PRE-EX |
| Saunders et al. 2009 | 13 M VO2max: 60.8 ± 1.6 Recreational cyclists | Crossover DB | None | 0 | Cycling TT 60 km | CHO-PRO: 134.3 ± 16.2 CHO: 135.1 ± 14.8 | 200 mL every 5 km and 500 mL PRE-EX |
| Hall et al. 2013 | 10 M VO2max: 66.2 ± 6 Cyclists | Crossover DB | Interval cycling workout ~2.5 hr | 4 | Cycling TT 7kJ/kg BW | CHO-PRO: 30.2 ± 4.93 CHO: 30.82 ± 5.73 | 250 mL every 15 min during PRE-EX, 30 mL every 5 min during TT, and recovery supplement |
| Upshaw et al. 2016 | 8 M VO2max: 61.2 ± 1.4 Cyclists | Crossover DB | Cycling intervals | 4 | Cycling TT 20 km | CHO: 37.85 ± 2.1
| CHO 247 kJ and CHO-PRO 2.107 kJ during REC |
| Gui et al. 2017 | 11 F VO2max: 49 ± 6.6 Recreational runners | Crossover DB | None | 0 | Running, TT 21 km | CHO-PRO: 132.4 ± 11.5 CHO: 129.6 ± 8.8 | 150 mL every 2.5 km during EX |
| Sollie et al. 2018 | 8 M VO2max: 74 ± 1.6 Cyclists | Crossover SB | Cycling to fatigue, interval + sprint, 50–90% VO2max | ~18 | Cycling; Preloaded TT 30 min at 73% W and TT | CHO-PRO: 41.53 ± 1.51 CHO: 45.26 ± 1.32 | 7.06 mL/kg BW/hr during the first 2 hr of REC |
| Finger et al. 2018 | 13 M VO2max: 62.2 ± 5.4 Amateur athletes | Crossover DB | SDT: 10 km running 40 km cycling | 0 | Duathlon, TT 5 km running | CHO-PRO: 21.12 ± 2.315 CHO: 21.17 ± 2.175 | 150 mL at 5, 20 and 35 km during 40 km cycling (PRE-EX) |
| van Essen et al. 2006 | 10 M VO2max: 63 ± 2 Cyclists/triathletes | Crossover DB | None | 0 | Cycling TT 80 km | CHO-PRO: 135 ± 6.3 CHO: 135 ± 6.3 | 250 mL every 15 min during EX |
| Breen et al. 2010 | 12 M VO2max: 62.7 ± 6.3 Cyclists | Crossover DB | Cycling; 120 min, 55% VO2max | 0 | Cycling TT (880±27 kJ) | CHO-PRO: 60.51 ± 8.3 CHO: 60.13 ± 4.5 | 270 mL 15 min of PRE-EX |
| Ferguson-Stegall et al. 2011 | 10 (5F, 5M) F: VO2max: 47.6 ± 1.5 M: VO2max: 57.7 ± 2.8 Cyclists | Crossover DB | Cycling; 1.5 hr at 70% VO2max + 10 min intervals | 4 | Cycling TT 40 km | CHO-PRO: 79.43 ± 2.11 CHO: 85.74 ± 3.44 | 1000–1400 mL during REC |
| Saunders et al. 2018 | 16 (11F, 5M) N.D. Amateur athletes | Parallel study DB | None | 0 | Marathon run, TT | CHO-PRO: 236.8 ± 26.4 CHO: 244.6 ± 35.7 | 4.5 ± 1.4 gels CHO and 5.9±1.5 gels CHO-PRO during EX |
| Goh et al. 2012 | 12 M VO2max:65 ± 7 Cyclists | Crossover DB | Interval cycling workout ~1 hr | 4 | Cycling: 20 min at 70% VO2max + 20 km TT |
| 750 mL during REC |
| Wolfe et al. 2016 | 10 (2F, 8M) VO2max: 54.6 ± 6.5 Cyclists/Triathletes | Crossover DB | Cycling, TT 40 km | 0.5 | Cycling, TT 10 km | CHO-PRO: 23.9 ± 1.0 CHO: 24 ± 0.9 | 1925 mL during the protocol |
| Oosthuyse et al. 2015 | 8 M VO2max: 60.9 ± 5.1 Cyclists | Crossover DB | Cycling; 2 hr at 60% Wattmax | 0 | Cycling, TT 16 km |
| 400 mL prior EX and 200 mL every 15 min during EX |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kloby Nielsen, L.L.; Tandrup Lambert, M.N.; Jeppesen, P.B. The Effect of Ingesting Carbohydrate and Proteins on Athletic Performance: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2020, 12, 1483. https://doi.org/10.3390/nu12051483
Kloby Nielsen LL, Tandrup Lambert MN, Jeppesen PB. The Effect of Ingesting Carbohydrate and Proteins on Athletic Performance: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients. 2020; 12(5):1483. https://doi.org/10.3390/nu12051483
Chicago/Turabian StyleKloby Nielsen, Lotte Lina, Max Norman Tandrup Lambert, and Per Bendix Jeppesen. 2020. "The Effect of Ingesting Carbohydrate and Proteins on Athletic Performance: A Systematic Review and Meta-Analysis of Randomized Controlled Trials" Nutrients 12, no. 5: 1483. https://doi.org/10.3390/nu12051483
APA StyleKloby Nielsen, L. L., Tandrup Lambert, M. N., & Jeppesen, P. B. (2020). The Effect of Ingesting Carbohydrate and Proteins on Athletic Performance: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients, 12(5), 1483. https://doi.org/10.3390/nu12051483





