Resting Energy Expenditure during Breastfeeding: Body Composition Analysis vs. Predictive Equations Based on Anthropometric Parameters
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects and Data Collection
2.2. Bioelectrical Impedance Analysis
2.3. Resting Energy Expenditure (REE) Predictive Equations
2.4. Statistical Analysis
3. Results
3.1. Subjects’ Characteristics and Body Composition Parameters
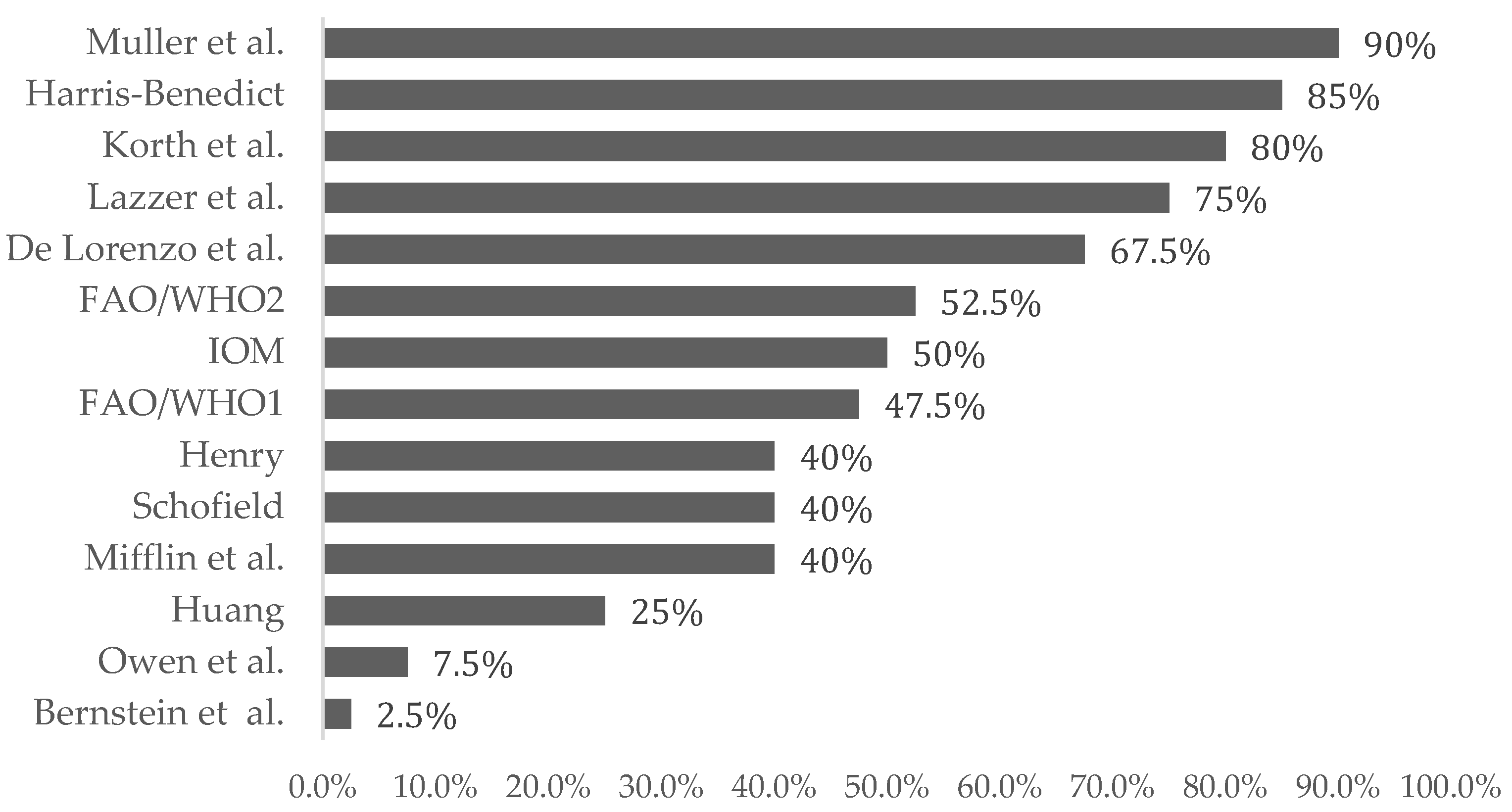
3.2. Estimated and Predicted Resting Energy Expenditure (REE)
4. Discussion
Author Contributions
Founding
Acknowledgments
Conflicts of Interest
References
- Hill, J.O.; Wyatt, H.R.; Peters, J.C. The Importance of Energy Balance. Eur. Endocrinol. 2013, 9, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Sparling, T.M.; Nesbitt, R.C.; Henschke, N.; Gabrysch, S. Nutrients and perinatal depression: A systematic review. J. Nutr. Sci. 2017, 6, 1–13. [Google Scholar] [CrossRef]
- Poston, L.; Caleyachetty, R.; Cnattingius, S.; Corvalán, C.; Uauy, R.; Herring, S.; Gillman, M. Preconceptional and maternal obesity: Epidemiology and health consequences. Lancet Diabetes Endocrinol. 2016, 4, 1025–1036. [Google Scholar] [CrossRef]
- Lovelady, C. Balancing exercise and food intake with lactation to promote post-partum weight loss. Proc. Nutr. Soc. 2011, 70, 181–184. [Google Scholar] [CrossRef]
- Innis, S.M. Impact of maternal diet on human milk composition and neurological development of infants. Am. J. Clin. Nutr. 2014, 99, 734–741. [Google Scholar] [CrossRef]
- Delsoglio, M.; Achamrah, N.; Berger, M.M.; Pichard, C. Indirect Calorimetry in Clinical Practice. J. Clin. Med. 2019, 8, 1387. [Google Scholar] [CrossRef]
- Singer, P.; Singer, J. Clinical Guide for the Use of Metabolic Carts: Indirect Calorimetry-No Longer the Orphan of Energy Estimation. Nutr. Clin. Pract. 2016, 31, 30–38. [Google Scholar] [CrossRef]
- Blundell, J.E.; Caudwell, P.; Gibbons, C.; Hopkins, M.; Naslund, E.; King, N.; Finlayson, G. Role of resting metabolic rate and energy expenditure in hunger and appetite control: A new formulation. Dis. Model. Mech. 2012, 5, 608–613. [Google Scholar] [CrossRef]
- Gunderson, E.P. Childbearing and obesity in women: Weight before, during, and after pregnancy. Obstet. Gynecol. Clin. N. Am. 2009, 36, 317–332. [Google Scholar] [CrossRef]
- Butte, N.F.; King, J.C. Energy requirements during pregnancy and lactation. Public Health Nutr. 2005, 8, 1010–1027. [Google Scholar] [CrossRef]
- Harris, J.A.; Benedict, F.G. A Biometric Study of Human Basal Metabolism. Proc. Natl. Acad. Sci. USA 1918, 4, 370–373. [Google Scholar] [CrossRef] [PubMed]
- Frankenfield, D.C.; Muth, E.R.; Rowe, W.A. The Harris-Benedict Studies of Human Basal Metabolism: History and Limitations. J. Am. Diet. Assoc. 1998, 98, 439–445. [Google Scholar] [CrossRef]
- Amaro-Gahete, F.J.; Sanchez-Delgado, G.; Alcantara, J.M.A.; Martinez-Tellez, B.; Muñoz-Hernandez, V.; Merchan-Ramirez, E.; Löf, M.; Labayen, I.; Ruiz, J.R. Congruent Validity of Resting Energy Expenditure Predictive Equations in Young Adults. Nutrients 2019, 11, 223. [Google Scholar] [CrossRef] [PubMed]
- Barak, N.; Wall-Alonso, E.; Cheng, A.; Sitrin, M.D. Use of bioelectrical impedance analysis to predict energy expenditure of hospitalized patients receiving nutrition support. JPEN J. Parenter. Enter. Nutr. 2003, 27, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Weyer, C.; Snitker, S.; Rising, R.; Bogardus, C.; Ravussin, E. Determinants of energy expenditure and fuel utilization in man: Effects of body composition, age, sex, ethnicity and glucose tolerance in 916 subjects. Int. J. Obes. Relat. Metab. Disord. 1999, 23, 715–722. [Google Scholar]
- Ferraro, R.; Ravussin, E. Fat mass in predicting resting metabolic rate. Am. J. Clin. Nutr. 1992, 56, 460. [Google Scholar] [CrossRef]
- Ravussin, E.; Lillioja, S.; Anderson, T.E.; Christin, L.; Bogardus, C. Determinants of 24-hour energy expenditure in man. Methods and results using a respiratory chamber. J. Clin. Investig. 1986, 78, 1568–1578. [Google Scholar] [CrossRef]
- Redondo, B. Resting energy expenditure; assessment methods and applications. Nutr. Hosp. 2015, 26, 245–254. [Google Scholar]
- Bera, T.K. Bioelectrical Impedance Methods for Noninvasive Health Monitoring: A Review. J. Med. Eng. 2014, 2014, 381251. [Google Scholar]
- Wasyluk, W.; Wasyluk, M.; Zwolak, A.; Łuczyk, R.J. Limits of body composition assessment by bioelectrical impedance analysis (BIA). J. Educ. Health Sport 2019, 9, 35–44. [Google Scholar]
- Mialich, M.S.; Jmf, S.; Jordao, A. Analysis of body composition: A critical review of the use of bioelectrical impedance analysis. Int. J. Clin. Nutr. 2014, 2, 1–10. [Google Scholar]
- Pereira, L.C.R.; Purcell, S.A.; Elliott, S.A.; McCargar, L.J.; Bell, R.C.; Robson, P.J.; Prado, C.M. The use of whole body calorimetry to compare measured versus predicted energy expenditure in postpartum women. Am. J. Clin. Nutr. 2019, 109, 554–565. [Google Scholar] [CrossRef] [PubMed]
- Korth, O.; Bosy-Westphal, A.; Zschoche, P.; Gluer, C.C.; Heller, M.; Muller, M.J. Influence of methods used in body composition analysis on the prediction of resting energy expenditure. Eur. J. Clin. Nutr. 2007, 61, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Euro WHO. Available online: http://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi (accessed on 21 March 2020).
- Heyward, V.H.; Stolarczyk, L.M. Applied Body Composition Assessment; Human Kinetics: Champaign, IL, USA, 1996; pp. 1–215. [Google Scholar]
- Bernstein, R.S.; Thornton, J.C.; Yang, M.U.; Wang, J.; Redmond, A.M.; Pierson, R.N., Jr.; Pi-Sunyer, F.X.; van Itallie, T.B. Prediction of the resting metabolic rate in obese patients. Am. J. Clin. Nutr. 1983, 37, 595–602. [Google Scholar]
- Owen, O.E.; Kavle, E.; Owen, R.S.; Polansky, M.; Caprio, S.; Mozzoli, M.A.; Kendrick, Z.V.; Bushman, M.C.; Boden, G. Areappraisal of caloric requirements in healthy women. Am. J. Clin. Nutr. 1986, 44, 1–19. [Google Scholar] [CrossRef]
- Mifflin, M.D.; St Jeor, S.T.; Hill, L.A.; Scott, B.J.; Daugherty, S.A.; Koh, Y.O. A new predictive equation for resting energy expenditure in healthy individuals. Am. J. Clin. Nutr. 1990, 51, 241–247. [Google Scholar] [CrossRef]
- Schofield, W.N. Predicting basal metabolic rate, new standards and rewiew of previous work. Hum. Nutr. Clin. Nutr. 1985, 39, 5–41. [Google Scholar]
- Food and Agriculture Organization; World Health Organization; United Nations. Energy and Protein Requirements; WHO Technical Report Series 724; World Health Organization: Geneva, Switzerland, 1985. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein and Amino Acids (Macronutrients); The National Academic Press: Washington, DC, USA, 2005; p. 1357. [Google Scholar]
- Müller, M.J.; Bosy-Westphal, A.; Klaus, S.; Kreymann, G.; Luhrmann, P.M.; Neuhauser-Berthold, M.; Noack, R.; Pirke, K.M.; Platte, P.; Selberg, O.; et al. World health organization equations have shortcomings for predicting resting energy expenditure in persons from a modern, affluent population: Generation of a new reference standard from a retrospective analysis of a German database of resting energy expenditure. Am. J. Clin. Nutr. 2004, 80, 1379–1390. [Google Scholar]
- De Lorenzo, A.; Tagliabue, A.; Andreoli, A.; Testolin, G.; Comelli, M.; Deurenberg, P. Measured and predicted resting metabolic rate in Italian males and females, aged 18–59 y. Eur. J. Clin. Nutr. 2001, 55, 208–214. [Google Scholar]
- Lazzer, S.; Agosti, F.; Silverstri, P.; Derumeaux-Burel, H.; Sartorio, A. Prediction of resting energy expenditure in severely obese Italian women. J. Edndocrinol. Invest. 2007, 30, 20–27. [Google Scholar] [CrossRef]
- Henry, C.J. Basal metabolic rate studies in humans: Measurement and development of new equations. Public Health Nutr. 2005, 8, 1133–1352. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.C.; Kormas, N.; Steinbeck, K.; Loughnan, G.; Caterson, I.D. Resting metabolic rate in severely obese diabetic and nondiabetic subjects. Obes. Res. 2004, 12, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Siervo, M.; Bertoli, S.; Battezzati, A.; Wells, J.C.; Lara, J.; Ferraris, C.; Tagliabue, A. Accuracy of predictive equations for the measurement of resting energy expenditure in older subjects. Clin. Nutr. 2014, 33, 613–619. [Google Scholar] [PubMed]
- Weijs, P.J.; Vansant, G.A. Validity of predictive equations for resting energy expenditure in Belgian normal weight to morbid obese women. Clin. Nutr. 2010, 29, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Callegari-Jacques, S.M. Biostatistcs: Priciples and Applications, 2nd ed.; Artemed: Porto Alegre, Brazil, 2003. [Google Scholar]
- Bzikowska-Jura, A.; Czerwonogrodzka-Senczyna, A.; Olędzka, G.; Szostak-Węgierek, D.; Weker, H.; Wesołowska, A. Maternal Nutrition and Body Composition During Breastfeeding: Association with Human Milk Composition. Nutrients 2018, 10, 1379. [Google Scholar] [CrossRef]
- Hu, M.; Qin, R.; Lin, X.; Ding, Y.; Li, F.; Wang, Z. Dietary status of lactating women from five cities of China in 2015–2016. J. Hyg. Res. 2019, 48, 220–225. [Google Scholar]
- Aubuchon-Endsley, N.L.; Kennedy, T.S.; Gilchrist, M.; Thomas, D.G.; Grant, S. Relationships among Socioeconomic Status, Dietary Intake, and Stress in Breastfeeding Women. J. Acad. Nutr. Diet. 2015, 115, 939–946. [Google Scholar] [CrossRef]
- Marangoni, F.; Cetin, I.; Verduci, E.; Canzone, G.; Giovannini, M.; Scollo, P.; Corsello, G.; Poli, A. Maternal Diet and Nutrient Requirements in Pregnancy and Breastfeeding. An Italian Consensus Document. Nutrients 2016, 8, 629. [Google Scholar]
- De Sousa, T.M.; Maioli, T.U.; Dos Santos, A.L.S.; Dos Santos, L.C. Energy expenditure in the immediate postpartum period: Indirect calorimetry versus predictive equations. Nutrition 2017, 39–40, 36–42. [Google Scholar] [CrossRef]
- Jesus, P.; Achamrah, N.; Grigioni, S.; Charles, J.; Rimbert, A.; Folope, V.; Petit, A.; Déchelotte, P.; Coëer, M. Validity of predictive equations for resting energy expenditure according to the body mass index in a population of 1726 patients followed in a Nutrition Unit. Clin. Nutr. 2015, 34, 529–535. [Google Scholar]
- Hashizume, N.; Tanaka, Y.; Yoshida, M.; Fukahori, S.; Ishii, S.; Saikusa, N.; Masui, D.; Higashidate, N.; Sakamoto, S.; Tsuruhisa, S.; et al. Resting energy expenditure prediction using bioelectrical impedance analysis in patients with severe motor and intellectual disabilities. Brain Dev. 2019, 41, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Jesus, P.; Marin, B.; Fayemendy, P.; Nicol, M.; Lautrette, G.; Sourisseau, H.; Preux, P.M.; Couratier, P.; Desport, J.C. Resting energy expenditure equations in amyotrophic lateral sclerosis, creation of an ALS-specific equation. Clin. Nutr. 2019, 38, 1657–1665. [Google Scholar] [CrossRef] [PubMed]
- Zusman, O.; Theilla, M.; Cohen, J.; Kagan, I.; Bendavid, I.; Singer, P. Resting energy expenditure, calorie and protein consumption in critically ill patients: A retrospective cohort study. Crit. Care 2016, 20, 367. [Google Scholar] [CrossRef] [PubMed]
- Frankenfield, D.; Roth-Yousey, L.; Compher, C. Comparison of predictive equations for resting metabolic rate in healthy nonobese and obese adults: A systematic review. J. Am. Diet. Assoc. 2005, 105, 775–789. [Google Scholar] [CrossRef] [PubMed]
- Kamimura, M.A.; Avesani, C.M.; Bazanelli, A.P.; Baria, F.; Draibe, S.A.; Cuppari, L. Are prediction equations reliable for estimating resting energy expenditure in chronic kidney disease patients? Nephrol. Dial. Transplant. 2011, 26, 544–550. [Google Scholar] [CrossRef]
- Khor, S.M.; Mohd, B.B. Assessing the resting energy expenditure of cancer patients in the Penang General Hospital. Malays. J. Nutr. 2011, 17, 43–53. [Google Scholar]
- Ramos, F.M.; Rossato, L.T.; Ramires, B.R.; Pimentel, G.D.; Venancio, L.S.; Orsatti, F.L.; de Oliveira, E.P. Comparison of predictive equations of resting energy expenditure in older adults with chronic obstructive pulmonary disease. Rev. Port. Pneumol. (2006) 2017, 23, 40–42. [Google Scholar] [CrossRef]
- Behera, K.K.; Joseph, M.; Shetty, S.K.; Chacko, A.; Sahoo, M.K.; Mahendri, N.V.; Nair, V.; Nadig, S.; Thomas, N. Resting energy expenditure in subjects with fibro-calculous pancreatic diabetes. J. Diabetes 2014, 6, 158–163. [Google Scholar]
- Marra, M.; Cioffi, I.; Sammarco, R.; Santarpia, L.; Contaldo, F.; Scalfi, L.; Pasanisi, F. Are Raw BIA Variables Useful for Predicting Resting Energy Expenditure in Adults with Obesity? Nutrients 2019, 11, 216. [Google Scholar] [CrossRef]
- Amaro-Gahete, F.J.; Jurado-Fasoli, L.; DelaO, A.; Gutierrez, Á.; Castillo, M.J.; Ruiz, J.R. Accuracy and Validity of Resting Energy Expenditure Predictive Equations in Middle-Aged Adults. Nutrients 2018, 10, 1635. [Google Scholar] [CrossRef]
- Wilms, B.; Schmid, S.M.; Ernst, B.; Thurnheer, M.; Mueller, M.J.; Schultes, B. Poor prediction of resting energy expenditure in obese women by established equations. Metabolism 2010, 59, 1181–1189. [Google Scholar] [CrossRef] [PubMed]
- Lam, Y.Y.; Ravussin, E. Analysis of energy metabolism in humans: A review of methodologies. Mol. Metab. 2016, 5, 1057–1071. [Google Scholar] [CrossRef] [PubMed]
- Elia, M. Organ and tissue contribution to metabolic rate. In Energy Metabolism: Tissue Determinants and Cellular Corollaries; Kinney, J.M., Tucker, H.N., Eds.; Raven Press: New York, NY, USA, 1992; pp. 61–80. [Google Scholar]
- Fiaccadori, E.; Morabito, S.; Cabassi, A.; Regolisti, G. Body cell mass evaluation in critically ill patients: Killing two birds with one stone. Crit. Care 2014, 18, 139. [Google Scholar] [CrossRef] [PubMed]
| Equations | Factors Used for Calculation | REE Predictive Equations (kcal/d) |
|---|---|---|
| Harris Benedict | Sex, W (kg), H (cm), age (y) | W × 9.5634 + H × 1.8496 − age × 4.6756 + 655.0955 |
| Bernstein et al. | Sex, W (kg), H (cm), age (y) | 7.48 × W − 0.42 × H − 3 × age + 844 |
| Owen et al. | Sex, W (kg) | W × 7.18 + 795 |
| Mifflin et al. | Sex, W (kg), H (cm), age (y) | 9.99 × W + 6.2 × H − 4.92 × age − 161 |
| Schofield | Sex, W (kg), H (m), age (y) | Age 18–30 y: (0.057 × W + 1.148 × H + 0.411) × 239 Age 31–60 y: (0.034 × W + 0.006 × H + 3.53) × 239 |
| FAO 1/WHO 2 | W (kg) | Age 18–30 y: 16.7 × W + 496 Age 31–60 y: 8.7 × W + 829 |
| FAO/WHO | W (kg), H (m) | Age 18–30 y: 13.3 × W + 334 × H + 35 Age 31–60 y: 8.7 × W − 25 × H + 865 |
| IOM 3 | W (kg), H (m), age (y) | 247 − 2.637 × age + 401 × H (m) + 8.6 × W |
| Müller et al. | Sex, W (kg), age (y) | (0.047 × W + 0.01452 × age + 3.21) × 239 |
| Korth et al. | Sex, W (kg), H (cm), age (y) | (41.4 × W + 35 × H − 19.1 × age − 1731.2)/4.186 |
| De Lorenzo et al. | Sex, W (kg), H (cm), age (y) | (46.322 × W + 15.744 × H − 16.66 × age + 944)/4.186 |
| Lazzer et al. | Sex, W (kg), H (m), age (y) | (0.042 × W + 3.619 × H − 2.678) × 239 |
| Henry | Sex, W (kg), age (y) | Age 18–30 y: (0.0546 × W + 2.33) × 239 Age 31–60 y: (0.0407 × W + 2.9) × 239 |
| Huang et al. | Sex, W (kg), H (cm), age (y) | 10.158 × W + 3993 × H − 1.44 × age + 60.655 |
| Mean ± SD | Median (Interquartile Range) | |
|---|---|---|
| Age (years) | 32.1 (6.2) | 31.0 (30.0–35.0) |
| Height (cm) | 166.6 (6.6) | 166.5 (162.0–172.5) |
| Pre-pregnancy weight | 61.4 (10.8) | 58.0 (53.8–69.0) |
| Pre-pregnancy body mass index (kg/m2) | 22.1 (3.3) | 21.1 (19.5–23.7) |
| Weight gain during pregnancy | 14.5 (4.6) | 14.0 (11.5–16.5) |
| Weight at first month postpartum (kg) | 64.5 (12.2) | 62.3 (54.8–70.9) |
| Body mass index at first month postpartum (kg/m2) | 23.0 (3.6) | 22.7 (20.4–24.8) |
| Fat mass–FM (kg) | 19.8 (10.3) | 17.9 (11.3–23.0) |
| Fat mass–FM (%) | 28.2 (8.4) | 28.5 (20.6–33.0) |
| Fat free mass–FFM (kg) | 45.4 (3.9) | 45.7 (43.0–48.4) |
| Fat free mass–FFM (%) | 71.8 (8.4) | 71.5 (67.0–79.4) |
| Total body water–TBW (L) | 32.4 (3.8) | 31.2 (29.4–35.2) |
| Total body water–TBW (%) | 51.2 (5.1) | 50.3 (47.0–55.3) |
| Extracellular water–ECW (L) | 15.0 (1.9) | 14.7 (13.8–16.3) |
| Extracellular water–ECW (%) | 46.3 (2.7) | 46.4 (45.4–48.0) |
| Intracellular water–ICW (L) | 17.4 (2.3) | 16.8 (16.0–18.9) |
| Intracellular water–ICW (%) | 53.7 (2.7) | 53.6 (52.0–54.7) |
| ECW/ICW | 0.87 (0.09) | 0.87 (0.83–0.92) |
| Body cell mass BCM (kg) | 23.9 (2.9) | 23.6 (22.3–25.9) |
| Extracellular mass–ECM (kg) | 21.5 (1.9) | 21.4 (20.1–23.3) |
| Protein mass–PM (kg) | 9.0 (1.4) | 9.0 (8.5–9.9) |
| Muscles (kg) | 19.9 (1.9) | 19.8 (18.8–21.4) |
| Minerals (kg) | 3.8 (0.6) | 3.7 (3.5–4.1) |
| Total body potassium–TBK (g) | 106.4 (12.0) | 104.6 (98.7–114.8) |
| Total body calcium–TBCa (g) | 892.3 (87.2) | 879 (836.5–953) |
| Glycogen (g) | 415.7 (38.1) | 418.5 (391.0–444.0) |
| Dry weight (kg) | 63.6 (12.2) | 61.2 (53.5–69.9) |
| Body volume (L) | 62.3 (13.1) | 59.8 (51.5–69.5) |
| Method | Energy Expenditure (kcal/day) | ||
|---|---|---|---|
| Median | 95% Cl | Spearman Correlation Coefficient | |
| BIA 1 | 1515.0 ± 68.4 | 1477.0–1582.0 | - |
| Harris–Benedict | 1441.0 ± 131.2 | 1361.5–1551.1 | 0.854 * |
| Bernstein et al. | 1149.7 ± 92.0 | 1088.7–1215.0 | 0.818 * |
| Owen et al. | 1236.6 ± 90.2 | 1179.1–1303.7 | 0.797 * |
| Mifflin et al. | 1344.9 ± 150.3 | 1230.1–1476.9 | 0.872 * |
| Schofield | 1366.2 ±159.7 | 1276.8–1495.3 | 0.820 * |
| FAO/WHO 2 | 1381.5 ± 162.8 | 1290.0–1516.1 | 0.791 * |
| FAO/WHO 3 | 1383.8 ±159.7 | 1294.2–1518.1 | 0.825 * |
| IOM | 1375.0 ± 123.1 | 1280.7–1480.3 | 0.866 * |
| Müller et al. | 1576.8 ± 143.8 | 1479.9–1683.4 | 0.748 * |
| Korth et al. | 1461.8 ± 159.7 | 1339.5–1601.1 | 0.870 * |
| De Lorenzo et al. | 1423.2 ± 152.5 | 1316.1–1552.6 | 0.858 * |
| Lazzer et al. | 1423.8 ± 163.1 | 1298.3–1576.5 | 0.850 * |
| Henry | 1328.0 ± 149.7 | 1248.5–1461.7 | 0.793 * |
| Huang et al. | 1309.9 ± 142.2 | 1208.1–1427.4 | 0.841 * |
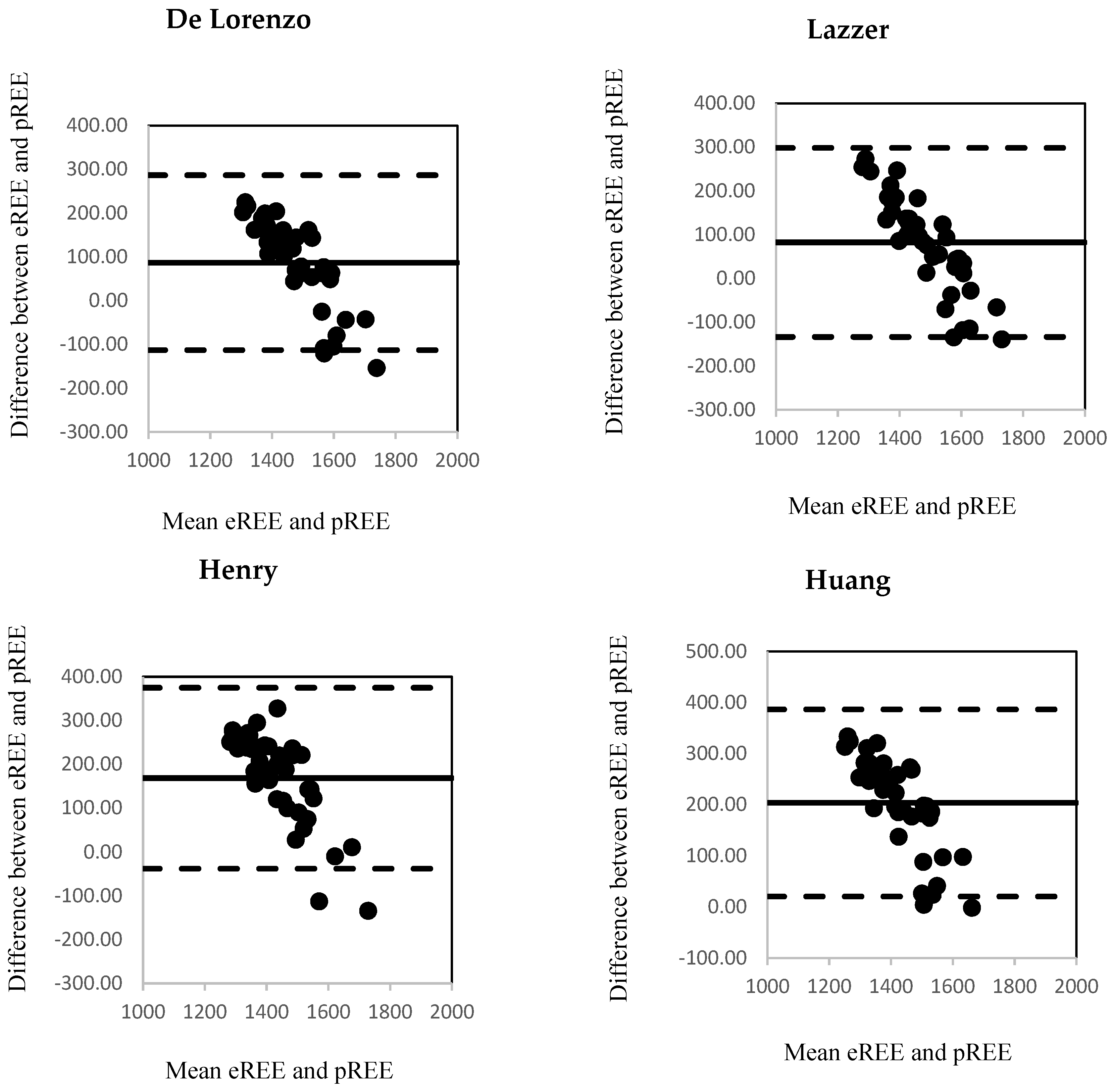
| ΔREE 3 kcal/day | SD | ΔREE 1 + 1.96 SD | ΔREE 1−1.96 SD | Range of LoA 4 | |
|---|---|---|---|---|---|
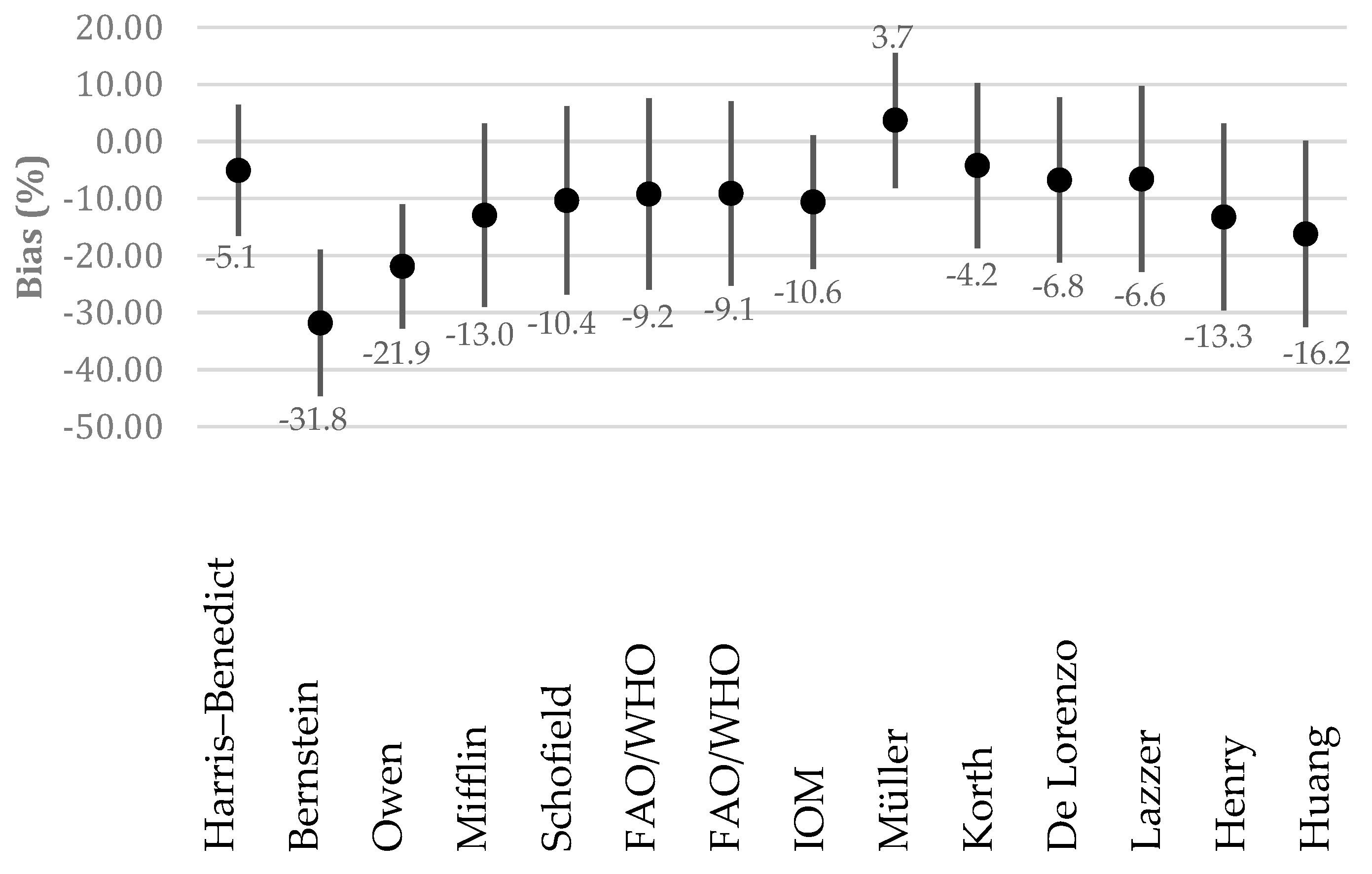
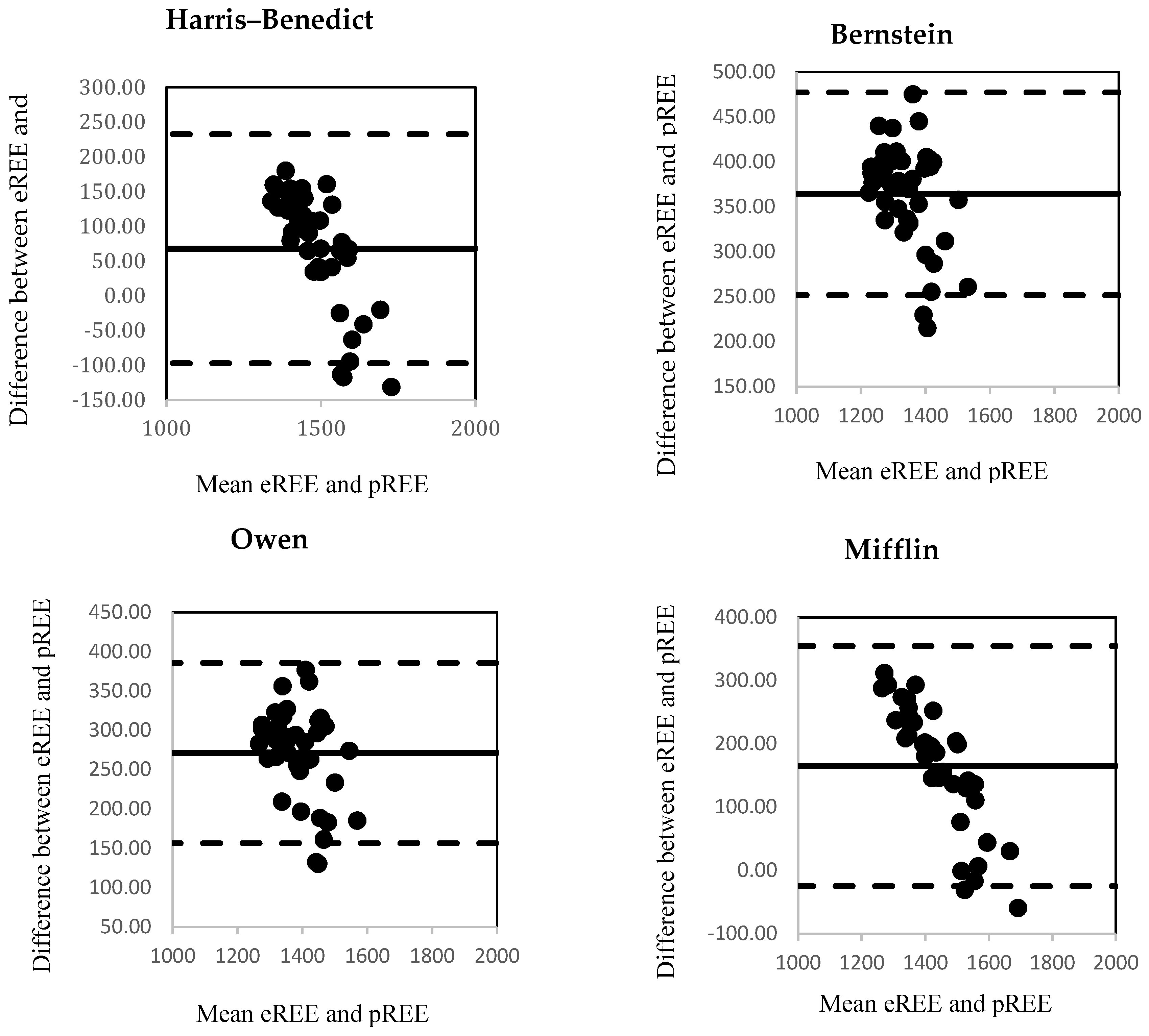
| Harris–Benedict | 67.54 *** | 84.10 | −97.30 | 232.37 | 329.67 |
| Bernstein et. al. | 364.54 *** | 57.56 | 251.73 | 477.35 | 729.08 |
| Owen et al. | 271.13 *** | 58.41 | 156.64 | 385.62 | 542.25 |
| Mifflin et al. | 164.86 *** | 96.70 | −24.68 | 354.40 | 379.08 |
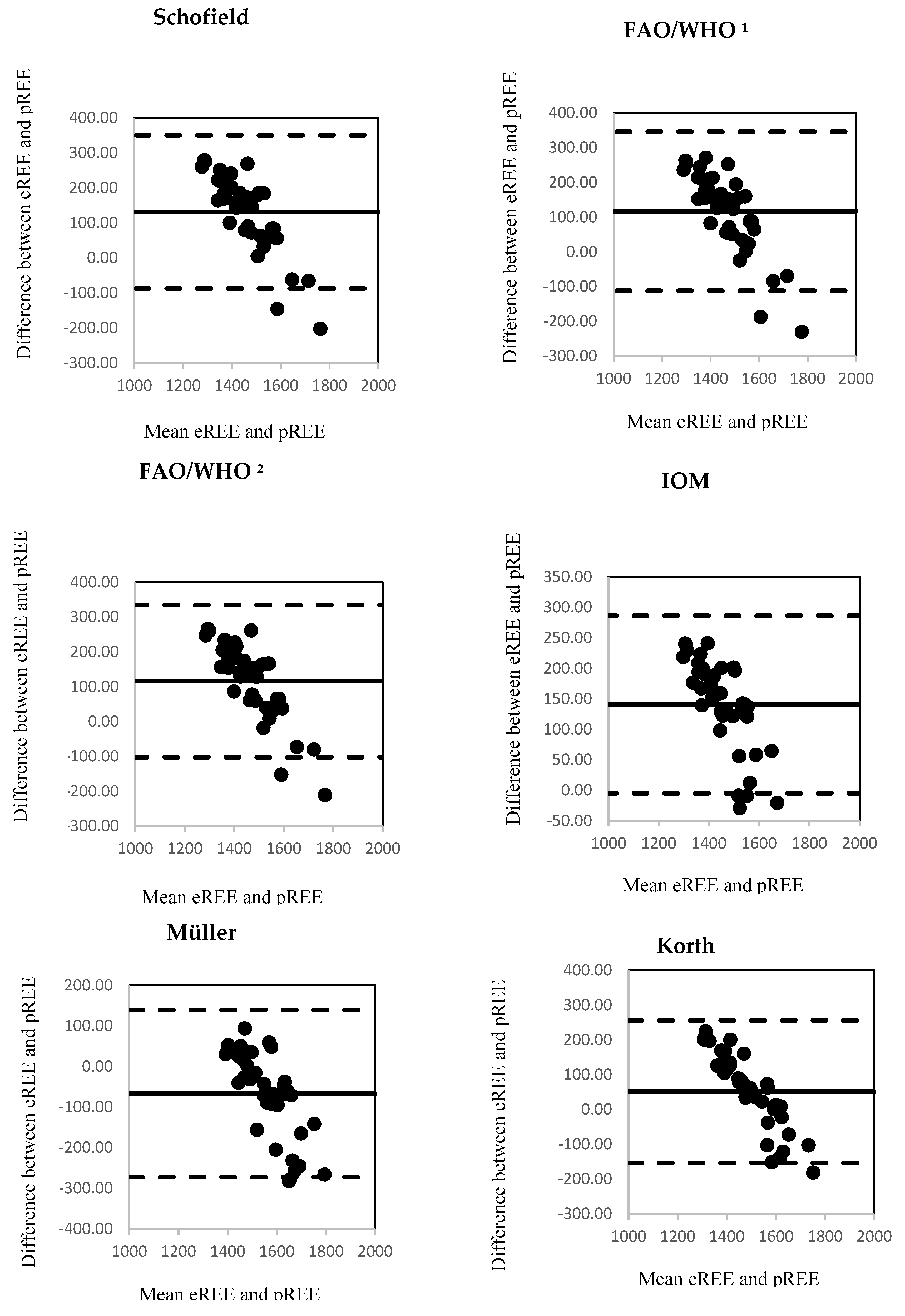
| Schofield | 131.91 *** | 111.66 | −86.93 | 350.76 | 437.70 |
| FAO/WHO 1 | 117.23 *** | 116.84 | −111.77 | 346.23 | 458.00 |
| FAO/WHO 2 | 116.40 *** | 111.38 | −101.90 | 334.69 | 436.59 |
| IOM | 140.86 *** | 74.27 | −4.71 | 286.43 | 291.14 |
| Müller et al. | −66.43 ** | 104.99 | −272.22 | 139.36 | 411.58 |
| Korth et al. | 51.37 * | 104.49 | −153.42 | 256.17 | 409.58 |
| De Lorenzo et al. | 86.88 *** | 101.95 | −112.94 | 286.70 | 399.65 |
| Lazzer et al. | 82.51 *** | 110.23 | −133.55 | 298.57 | 432.11 |
| Henry | 168.60 *** | 105.29 | −37.76 | 374.97 | 412.73 |
| Huang et al. | 203.76 *** | 93.32 | 20.85 | 386.67 | 407.52 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bzikowska-Jura, A.; Szulińska, A.; Szostak-Węgierek, D. Resting Energy Expenditure during Breastfeeding: Body Composition Analysis vs. Predictive Equations Based on Anthropometric Parameters. Nutrients 2020, 12, 1274. https://doi.org/10.3390/nu12051274
Bzikowska-Jura A, Szulińska A, Szostak-Węgierek D. Resting Energy Expenditure during Breastfeeding: Body Composition Analysis vs. Predictive Equations Based on Anthropometric Parameters. Nutrients. 2020; 12(5):1274. https://doi.org/10.3390/nu12051274
Chicago/Turabian StyleBzikowska-Jura, Agnieszka, Adriana Szulińska, and Dorota Szostak-Węgierek. 2020. "Resting Energy Expenditure during Breastfeeding: Body Composition Analysis vs. Predictive Equations Based on Anthropometric Parameters" Nutrients 12, no. 5: 1274. https://doi.org/10.3390/nu12051274
APA StyleBzikowska-Jura, A., Szulińska, A., & Szostak-Węgierek, D. (2020). Resting Energy Expenditure during Breastfeeding: Body Composition Analysis vs. Predictive Equations Based on Anthropometric Parameters. Nutrients, 12(5), 1274. https://doi.org/10.3390/nu12051274






