Effects of Ashwagandha (Withania somnifera) on VO2max: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Literature Search
2.2. Study Selection
2.3. Quality of the Evidence and Risk of Bias
2.4. Data Collection
2.5. Statistical Analysis
3. Results
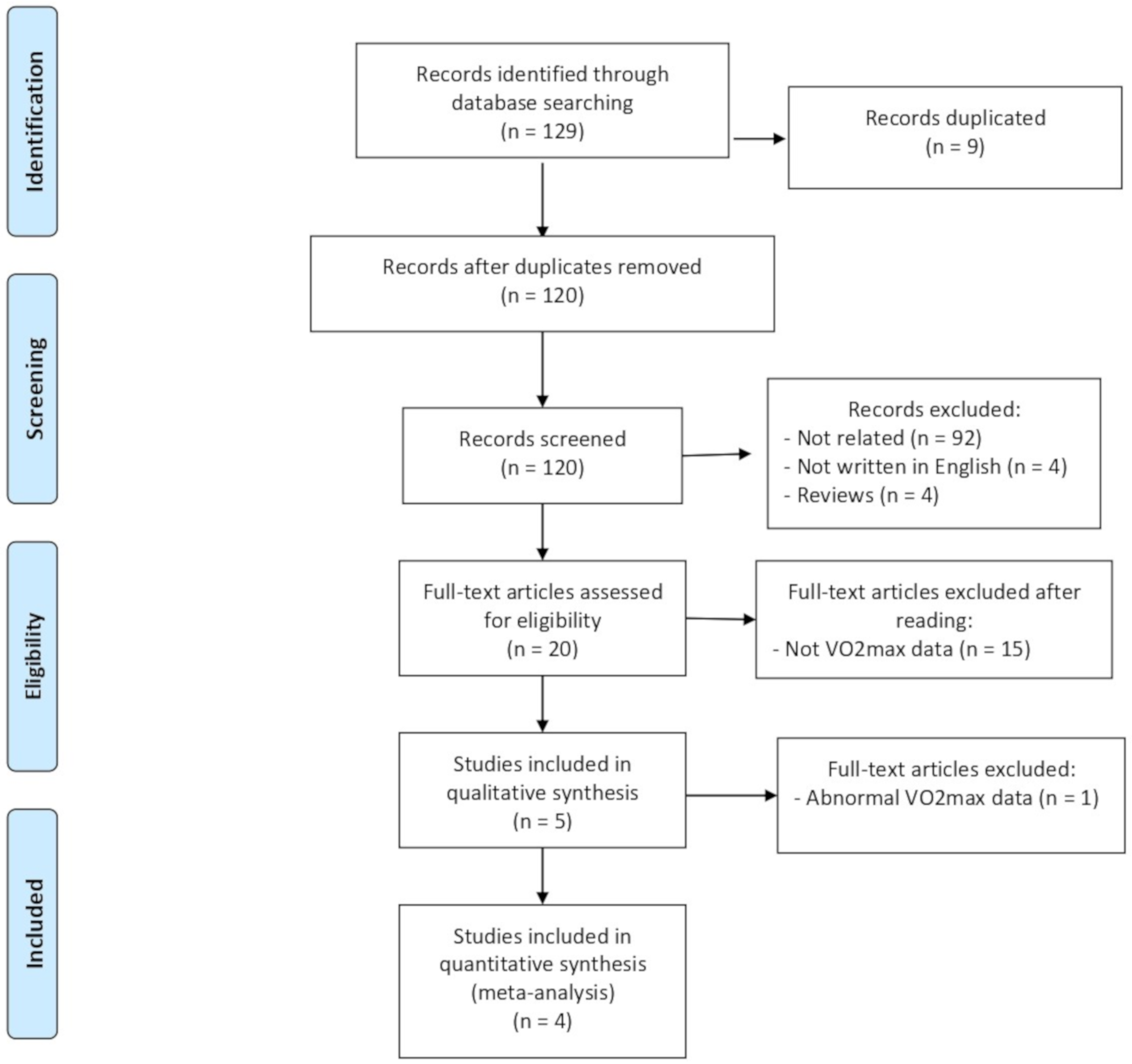
3.1. Study Selection
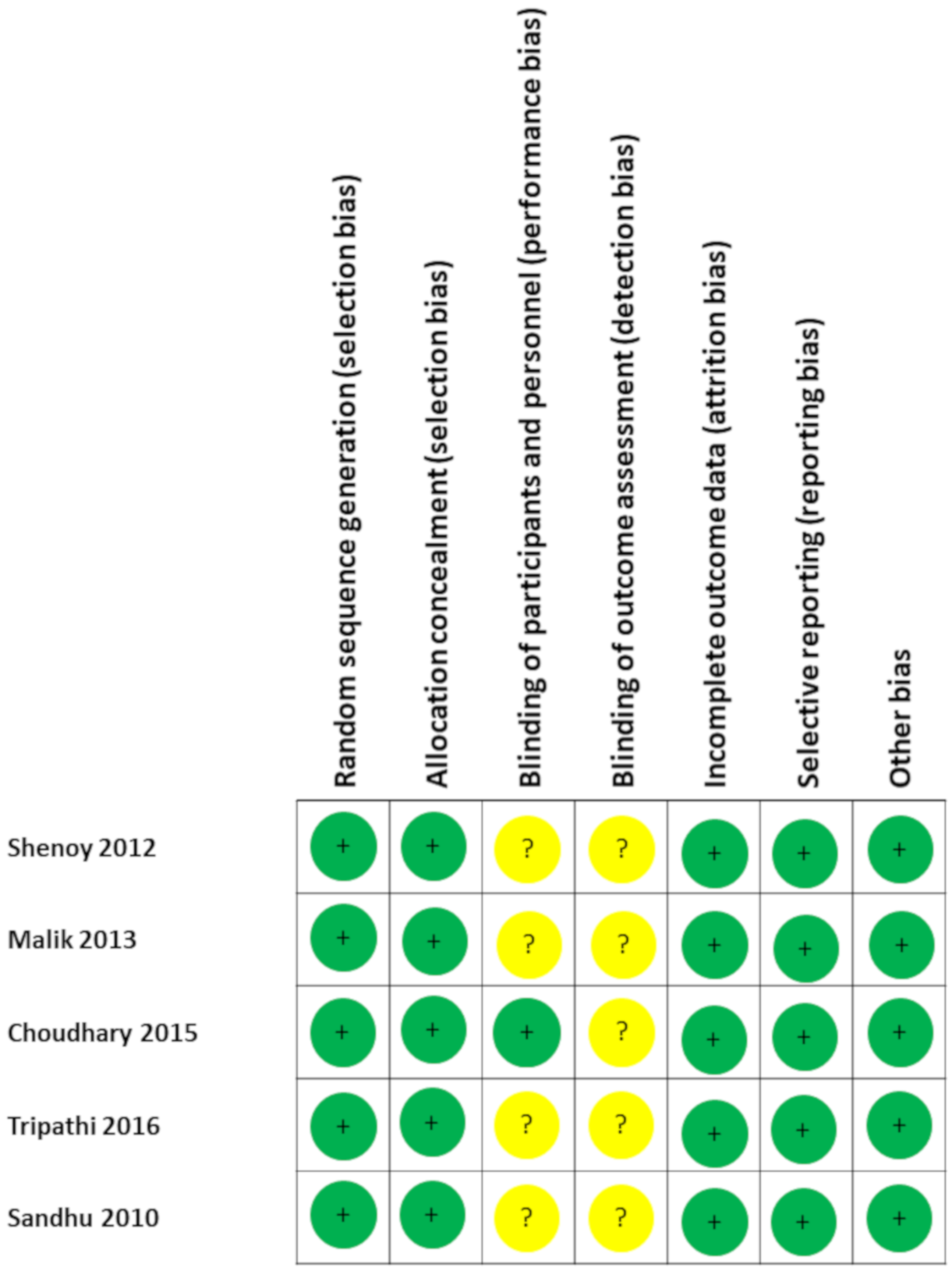
3.2. Quality of Evidence and Risk of Bias
3.3. Study Characteristics
3.4. Interventions
3.5. Outcome Measures
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Dela, F.; Finkenzeller, T.; Ingersen, A.; Potzelsberger, B.; Muller, E. Trajectories of cardio-metabolic health in successful aging. Scand. J. Med. Sci. Sports 2019, 29 (Suppl. 1), 44–51. [Google Scholar] [CrossRef]
- Mooses, M.; Hackney, A.C. Anthropometrics and Body Composition in East African Runners: Potential Impact on Performance. Int. J. Sports Physiol. Perform. 2017, 12, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Brandon, L.J. Physiological factors associated with middle distance running performance. Sports Med. 1995, 19, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Helgerud, J.; Engen, L.C.; Wisloff, U.; Hoff, J. Aerobic endurance training improves soccer performance. Med. Sci. Sports Exerc. 2001, 33, 1925–1931. [Google Scholar] [CrossRef] [PubMed]
- Midgley, A.W.; McNaughton, L.R.; Wilkinson, M. Is there an optimal training intensity for enhancing the maximal oxygen uptake of distance runners? Empirical research findings, current opinions, physiological rationale and practical recommendations. Sports Med. 2006, 36, 117–132. [Google Scholar] [CrossRef] [PubMed]
- Steele, J.; Butler, A.; Comerford, Z.; Dyer, J.; Lloyd, N.; Ward, J.; Fisher, J.; Gentil, P.; Scott, C.; Ozaki, H. Similar acute physiological responses from effort and duration matched leg press and recumbent cycling tasks. PeerJ 2018, 6, e4403. [Google Scholar] [CrossRef]
- Myers, J.; Prakash, M.; Froelicher, V.; Do, D.; Partington, S.; Atwood, J.E. Exercise capacity and mortality among men referred for exercise testing. N. Engl. J. Med. 2002, 346, 793–801. [Google Scholar] [CrossRef]
- Blair, S.N.; Kampert, J.B.; Kohl, H.W., 3rd; Barlow, C.E.; Macera, C.A.; Paffenbarger, R.S., Jr.; Gibbons, L.W. Influences of cardiorespiratory fitness and other precursors on cardiovascular disease and all-cause mortality in men and women. JAMA 1996, 276, 205–210. [Google Scholar] [CrossRef]
- Rebollo-Ramos, M.; Velazquez-Diaz, D.; Corral-Perez, J.; Barany-Ruiz, A.; Perez-Bey, A.; Fernandez-Ponce, C.; Garcia-Cozar, F.J.; Ponce-Gonzalez, J.G.; Cuenca-Garcia, M. Aerobic fitness, Mediterranean diet and cardiometabolic risk factors in adults. Endocrinol. Diabetes Nutr. 2019. [Google Scholar] [CrossRef]
- Galvez Casas, A.; Rodriguez Garcia, P.L.; Garcia-Canto, E.; Rosa Guillamon, A.; Perez-Soto, J.J.; Tarraga Marcos, L.; Tarraga Lopez, P. Aerobic capacity and quality of life in school children from 8 to 12. Clin. Investig. Arterioscler. 2015, 27, 239–245. [Google Scholar] [CrossRef]
- Dar, N.J.; Hamid, A.; Ahmad, M. Pharmacologic overview of Withania somnifera, the Indian Ginseng. Cell. Mol. Life Sci. 2015, 72, 4445–4460. [Google Scholar] [CrossRef] [PubMed]
- Chukwuma, C.I.; Matsabisa, M.G.; Ibrahim, M.A.; Erukainure, O.L.; Chabalala, M.H.; Islam, M.S. Medicinal plants with concomitant anti-diabetic and anti-hypertensive effects as potential sources of dual acting therapies against diabetes and hypertension: A review. J. Ethnopharmacol. 2019, 235, 329–360. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.Y.; Li, R.; Cui, J.; Hannink, M.; Gu, Z.; Fritsche, K.L.; Lubahn, D.B.; Simonyi, A. Withania somnifera and Its Withanolides Attenuate Oxidative and Inflammatory Responses and Up-Regulate Antioxidant Responses in BV-2 Microglial Cells. Neuromol. Med. 2016, 18, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, N.; Shrivastava, D.; Ahmad Mir, B.; Kumar, S.; Govil, S.; Vahedi, M.; Bisen, P.S. Metabolomic and biotechnological approaches to determine therapeutic potential of Withania somnifera (L.) Dunal: A review. Phytomedicine 2018, 50, 127–136. [Google Scholar] [CrossRef]
- Hassannia, B.; Logie, E.; Vandenabeele, P.; Vanden Berghe, T.; Vanden Berghe, W. Withaferin A: From ayurvedic folk medicine to preclinical anti-cancer drug. Biochem. Pharm. 2019. [Google Scholar] [CrossRef]
- Kaur, P.; Mathur, S.; Sharma, M.; Tiwari, M.; Srivastava, K.K.; Chandra, R. A biologically active constituent of withania somnifera (ashwagandha) with antistress activity. Indian J. Clin. Biochem. 2001, 16, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Singh, N.; Samuel, S.S.; Bora, H.K.; Sharma, S.; Pachauri, S.D.; Dwivedi, A.K.; Siddiqui, H.H.; Hanif, K. Withania somnifera shows a protective effect in monocrotaline-induced pulmonary hypertension. Pharm. Biol. 2015, 53, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Yenisetti, S.C.; Manjunath, M.J.; Muralidhara, C. Neuropharmacological Properties of Withania somnifera - Indian Ginseng: An Overview on Experimental Evidence with Emphasis on Clinical Trials and Patents. Recent Pat. CNS Drug Discov. 2016, 10, 204–215. [Google Scholar] [CrossRef]
- Ahmed, W.; Mofed, D.; Zekri, A.R.; El-Sayed, N.; Rahouma, M.; Sabet, S. Antioxidant activity and apoptotic induction as mechanisms of action of Withania somnifera (Ashwagandha) against a hepatocellular carcinoma cell line. J. Int. Med. Res. 2018, 46, 1358–1369. [Google Scholar] [CrossRef]
- Pradhan, R.; Kumar, R.; Shekhar, S.; Rai, N.; Ambashtha, A.; Banerjee, J.; Pathak, M.; Dwivedi, S.N.; Dey, S.; Dey, A.B. Longevity and healthy ageing genes FOXO3A and SIRT3: Serum protein marker and new road map to burst oxidative stress by Withania somnifera. Exp. Gerontol. 2017, 95, 9–15. [Google Scholar] [CrossRef]
- Chandrasekhar, K.; Kapoor, J.; Anishetty, S. A prospective, randomized double-blind, placebo-controlled study of safety and efficacy of a high-concentration full-spectrum extract of ashwagandha root in reducing stress and anxiety in adults. Indian J. Psychol. Med. 2012, 34, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Ahmed, R.S.; Chandra, N.; Arora, V.K.; Ali, A. In vivo, Extract from Withania somnifera Root Ameliorates Arthritis via Regulation of Key Immune Mediators of Inflammation in Experimental Model of Arthritis. Antiinflamm. Antiallergy Agents Med. Chem. 2019, 18, 55–70. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, D.; Bhattacharyya, S.; Bose, S. Efficacy and Safety of Ashwagandha (Withania somnifera (L.) Dunal) Root Extract in Improving Memory and Cognitive Functions. J. Diet. Suppl. 2017, 14, 599–612. [Google Scholar] [CrossRef] [PubMed]
- Anju, T.R.; Smijin, S.; Jobin, M.; Paulose, C.S. Altered muscarinic receptor expression in the cerebral cortex of epileptic rats: Restorative role of Withania somnifera. Biochem. Cell Biol. 2018, 96, 433–440. [Google Scholar] [CrossRef]
- Singh, A.; Naidu, P.S.; Gupta, S.; Kulkarni, S.K. Effect of natural and synthetic antioxidants in a mouse model of chronic fatigue syndrome. J. Med. Food 2002, 5, 211–220. [Google Scholar] [CrossRef]
- Kuboyama, T.; Tohda, C.; Komatsu, K. Effects of Ashwagandha (roots of Withania somnifera) on neurodegenerative diseases. Biol. Pharm. Bull. 2014, 37, 892–897. [Google Scholar] [CrossRef]
- Ramakanth, G.S.; Uday Kumar, C.; Kishan, P.V.; Usharani, P. A randomized, double blind placebo controlled study of efficacy and tolerability of Withaina somnifera extracts in knee joint pain. J. Ayurveda Integr. Med. 2016, 7, 151–157. [Google Scholar] [CrossRef]
- Sharma, A.K.; Basu, I.; Singh, S. Efficacy and Safety of Ashwagandha Root Extract in Subclinical Hypothyroid Patients: A Double-Blind, Randomized Placebo-Controlled Trial. J. Altern. Complement. Med. 2018, 24, 243–248. [Google Scholar] [CrossRef]
- Li, W.; Zhang, C.; Du, H.; Huang, V.; Sun, B.; Harris, J.P.; Richardson, Q.; Shen, X.; Jin, R.; Li, G.; et al. Withaferin A suppresses the up-regulation of acetyl-coA carboxylase 1 and skin tumor formation in a skin carcinogenesis mouse model. Mol. Carcinog. 2016, 55, 1739–1746. [Google Scholar] [CrossRef]
- Cakici, N.; van Beveren, N.J.M.; Judge-Hundal, G.; Koola, M.M.; Sommer, I.E.C. An update on the efficacy of anti-inflammatory agents for patients with schizophrenia: A meta-analysis. Psychol. Med. 2019, 49, 2307–2319. [Google Scholar] [CrossRef]
- Durg, S.; Shivaram, S.B.; Bavage, S. Withania somnifera (Indian ginseng) in male infertility: An evidence-based systematic review and meta-analysis. Phytomedicine 2018, 50, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Durg, S.; Dhadde, S.B.; Vandal, R.; Shivakumar, B.S.; Charan, C.S. Withania somnifera (Ashwagandha) in neurobehavioural disorders induced by brain oxidative stress in rodents: A systematic review and meta-analysis. J. Pharm. Pharmacol. 2015, 67, 879–899. [Google Scholar] [CrossRef] [PubMed]
- Pratte, M.A.; Nanavati, K.B.; Young, V.; Morley, C.P. An alternative treatment for anxiety: A systematic review of human trial results reported for the Ayurvedic herb ashwagandha (Withania somnifera). J. Altern. Complement. Med. 2014, 20, 901–908. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, J.S.; Shah, B.; Shenoy, S.; Chauhan, S.; Lavekar, G.S.; Padhi, M.M. Effects of Withania somnifera (Ashwagandha) and Terminalia arjuna (Arjuna) on physical performance and cardiorespiratory endurance in healthy young adults. Int. J. Ayurveda Res. 2010, 1, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, B.; Shetty, A.; Langade, D.G. Efficacy of Ashwagandha (Withania somnifera [L.] Dunal) in improving cardiorespiratory endurance in healthy athletic adults. Ayu 2015, 36, 63–68. [Google Scholar] [CrossRef]
- Tripathi, R.; Salve, B.; Petare, A.; Raut, A.; Rege, N. Effect of Withania somnifera on physical and cardiovascular performance induced by physical stress in healthy human volunteers. Int. J. Basic Clin. Pharmacol. 2016, 5, 2510–2516. [Google Scholar]
- Malik, A.; Mehta, V.; Dahiya, V. Effect of ashwagandha (withania somnifera) root powder supplementation on the vo2 max. and hemoglobin in hockey players. Int. J. Behav. Soc. Mov. Sci. 2013, 2, 91–99. [Google Scholar]
- Shenoy, S.; Chaskar, U.; Sandhu, J.S.; Paadhi, M.M. Effects of eight-week supplementation of Ashwagandha on cardiorespiratory endurance in elite Indian cyclists. J. Ayurveda Integr. Med. 2012, 3, 209–214. [Google Scholar] [CrossRef]
- Raut, A.A.; Rege, N.N.; Tadvi, F.M.; Solanki, P.V.; Kene, K.R.; Shirolkar, S.G.; Pandey, S.N.; Vaidya, R.A.; Vaidya, A.B. Exploratory study to evaluate tolerability, safety, and activity of Ashwagandha (Withania somnifera) in healthy volunteers. J. Ayurveda Integr. Med. 2012, 3, 111–114. [Google Scholar] [CrossRef]
- Williams, C.J.; Williams, M.G.; Eynon, N.; Ashton, K.J.; Little, J.P.; Wisloff, U.; Coombes, J.S. Genes to predict VO2max trainability: A systematic review. BMC Genom. 2017, 18, 831. [Google Scholar] [CrossRef]
- Honig, C.R.; Connett, R.J.; Gayeski, T.E. O2 transport and its interaction with metabolism; a systems view of aerobic capacity. Med. Sci. Sports Exerc. 1992, 24, 47–53. [Google Scholar] [CrossRef]
- Pette, D.; Staron, R.S. Cellular and molecular diversities of mammalian skeletal muscle fibers. Rev. Physiol. Biochem. Pharmacol. 1990, 116, 1–76. [Google Scholar]
- MacInnis, M.J.; Gibala, M.J. Physiological adaptations to interval training and the role of exercise intensity. J. Physiol. 2017, 595, 2915–2930. [Google Scholar] [CrossRef]
- Dominguez, R.; Cuenca, E.; Mate-Munoz, J.L.; Garcia-Fernandez, P.; Serra-Paya, N.; Estevan, M.C.; Herreros, P.V.; Garnacho-Castano, M.V. Effects of Beetroot Juice Supplementation on Cardiorespiratory Endurance in Athletes. A Systematic Review. Nutrients 2017, 9, 43. [Google Scholar] [CrossRef]
- Dhuley, J.N. Adaptogenic and cardioprotective action of ashwagandha in rats and frogs. J. Ethnopharmacol. 2000, 70, 57–63. [Google Scholar] [CrossRef]
- Begum, V.H.; Sadique, J. Effect of Withania somnifera on glycosaminoglycan synthesis in carrageenin-induced air pouch granuloma. Biochem. Med. Metab. Biol 1987, 38, 272–277. [Google Scholar] [CrossRef]
- Ziauddin, M.; Phansalkar, N.; Patki, P.; Diwanay, S.; Patwardhan, B. Studies on the immunomodulatory effects of Ashwagandha. J. Ethnopharmacol. 1996, 50, 69–76. [Google Scholar] [CrossRef]
- Mishra, L.C. Scientific Basis for Ayurvedic Therapies; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Biswal, B.M.; Sulaiman, S.A.; Ismail, H.C.; Zakaria, H.; Musa, K.I. Effect of Withania somnifera (Ashwagandha) on the development of chemotherapy-induced fatigue and quality of life in breast cancer patients. Integr. Cancer Ther. 2013, 12, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Lopresti, A.L.; Smith, S.J.; Malvi, H.; Kodgule, R. An investigation into the stress-relieving and pharmacological actions of an ashwagandha (Withania somnifera) extract: A randomized, double-blind, placebo-controlled study. Medicine 2019, 98, e17186. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Dey, A.; Hadimani, M.B.; Marcović, T.; Emerald, M. Chemistry and pharmacology of Withania somnifera: An update. TANG 2015, 5, 1–13. [Google Scholar] [CrossRef]
- Mishra, L.C.; Singh, B.B.; Dagenais, S. Scientific basis for the therapeutic use of Withania somnifera (ashwagandha): A review. Altern. Med. Rev. 2000, 5, 334–346. [Google Scholar] [PubMed]

| RCT | Weeks | Groups, Sample Size and Sex | Age (Years) | Country | Population |
|---|---|---|---|---|---|
| Shenoy 2012 | 8 | AS: 20 (M and F) CG: 20 (M and F) | 18–27 | India | Elite cyclists |
| Malik 2013 | 8 | AS: 16 (M) CG: 16 (M) | 16–19 | India | Hockey players |
| Choudhary 2015 | 12 | AS: 25 (M and F) CG: 25 (M and F) | 20–45 | India | Athletes |
| Tripathi 2016 | 2 | AS: 10 (M) CG: 10 (M) | 18–45 | India | Healthy adults |
| Sandhu 2010 | 8 | AS: 10 (M and F) CG: 10 (M and F) | 18–25 | India | Healthy adults |
| RCT | Ashwagandha Group | Control Group | Dose (mg) | Duration of the Study | Daily Frequency | Total Dose (g) |
|---|---|---|---|---|---|---|
| Type of Supplementation | Type of Supplementation | |||||
| Shenoy 2012 | Ashwagandha in gelatin capsules | Capsules containing starch powder | 500 | 8 weeks | twice | 56 |
| Malik 2013 | Roots of WS | Sugar power was filled in gelatin capsules | 500 | 8 weeks | once | 28 |
| Choudhary 2015 | One capsule of KSM-66 Ashwagandha | Identical capsules containing sucrose | 300 | 12 weeks | twice | 50.4 |
| Tripathi 2016 | WS aqueous extract in the capsule form | Maize starch capsule | 330 | 2 weeks | once | 4.62 |
| Sandhu 2010 | WS filled in gelatin capsules | Capsules filled with flour | 500 | 8 weeks | once | 28 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Gómez, J.; Villafaina, S.; Adsuar, J.C.; Merellano-Navarro, E.; Collado-Mateo, D. Effects of Ashwagandha (Withania somnifera) on VO2max: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 1119. https://doi.org/10.3390/nu12041119
Pérez-Gómez J, Villafaina S, Adsuar JC, Merellano-Navarro E, Collado-Mateo D. Effects of Ashwagandha (Withania somnifera) on VO2max: A Systematic Review and Meta-Analysis. Nutrients. 2020; 12(4):1119. https://doi.org/10.3390/nu12041119
Chicago/Turabian StylePérez-Gómez, Jorge, Santos Villafaina, José Carmelo Adsuar, Eugenio Merellano-Navarro, and Daniel Collado-Mateo. 2020. "Effects of Ashwagandha (Withania somnifera) on VO2max: A Systematic Review and Meta-Analysis" Nutrients 12, no. 4: 1119. https://doi.org/10.3390/nu12041119
APA StylePérez-Gómez, J., Villafaina, S., Adsuar, J. C., Merellano-Navarro, E., & Collado-Mateo, D. (2020). Effects of Ashwagandha (Withania somnifera) on VO2max: A Systematic Review and Meta-Analysis. Nutrients, 12(4), 1119. https://doi.org/10.3390/nu12041119









