Harnessing Microbes for Sustainable Development: Food Fermentation as a Tool for Improving the Nutritional Quality of Alternative Protein Sources
Abstract
1. Introduction
2. Fermentation Process
3. The Benefits of Food Fermentations from the Sustainability Point of View
3.1. The Multiple Levels of Food Production Sustainability
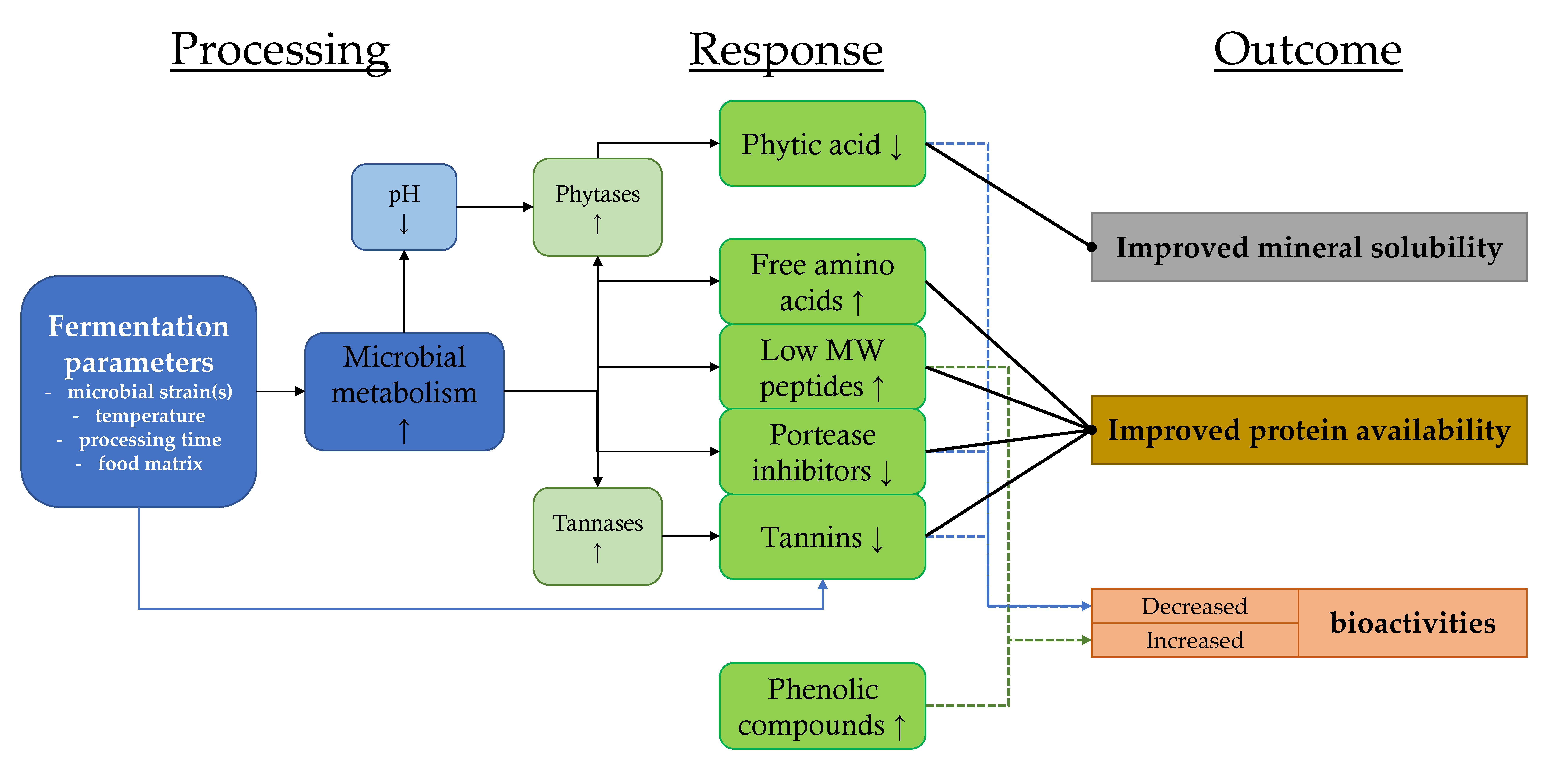
3.2. Improving the Nutritional Quality of Sustainable Protein Sources by Reducing Antinutritional Factors
In Vitro Protein Digestibility as a Measure of Nutritional Quality
4. The Effects of Fermentation on Nutritional Quality: Special Focus on Protein and Micronutrient Availability in Legume and Cereal Products
4.1. Legumes
Fermentation Increases Nutrient Availability in Commonly Consumed Legumes
4.2. Cereals
4.2.1. Applying Fermentation to Boost the Nutritional Quality of Cereal and Quinoa Products
4.2.2. Fortification of Fermented Cereal Food Products with Pseudocereal and Legume-Based Ingredients
5. From In Vitro to In Vivo
6. The Prospects of Research and Process Development in the Field of Food Fermentation
6.1. Process Optimization for Nutritionally Desirable Outcomes
6.2. Critical Points
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Springmann, M.; Wiebe, K.; Mason-D’Croz, D.; Sulser, T.B.; Rayner, M.; Scarborough, P. Health and Nutritional Aspects of Sustainable Diet Strategies and their Association with Environmental Impacts: A Global Modelling Analysis with Country-Level Detail. Lancet Planet. Health 2018, 2, e451–e461. [Google Scholar] [CrossRef]
- Willett, W.; Rockström, J.; Loken, B.; Springmann, M.; Lang, T.; Vermeulen, S.; Garnett, T.; Tilman, D.; DeClerck, F.; Wood, A. Food in the Anthropocene: The EAT–Lancet Commission on Healthy Diets from Sustainable Food Systems. Lancet 2019, 393, 447–492. [Google Scholar] [CrossRef]
- Tomé, D. Digestibility Issues of Vegetable Versus Animal Proteins: Protein and Amino Acid requirements—Functional Aspects. Food Nutr. Bull. 2013, 34, 272–274. [Google Scholar] [CrossRef] [PubMed]
- Sá, A.G.A.; Moreno, Y.M.F.; Carciofi, B.A.M. Food Processing for the Improvement of Plant Proteins Digestibility. Crit. Rev. Food Sci. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.O.; Bracarense, A.P.F. Phytic Acid: From Antinutritional to Multiple Protection Factor of Organic Systems. J. Food Sci. 2016, 81, R1357–R1362. [Google Scholar] [CrossRef] [PubMed]
- Barrett, A.H.; Farhadi, N.F.; Smith, T.J. Slowing Starch Digestion and Inhibiting Digestive Enzyme Activity using Plant Flavanols/Tannins—A Review of Efficacy and Mechanisms. LWT 2018, 87, 394–399. [Google Scholar] [CrossRef]
- Clemente, A.; Olias, R. Beneficial Effects of Legumes in Gut Health. Curr. Opin. Food Sci. 2017, 14, 32–36. [Google Scholar] [CrossRef]
- Hutkins, R.W. Microbiology and Technology of Fermented Foods; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- Mota de Carvalho, N.; Costa, E.M.; Silva, S.; Pimentel, L.; Fernandes, T.H.; Pintado, M.E. Fermented Foods and Beverages in Human Diet and their Influence on Gut Microbiota and Health. Fermentation 2018, 4, 90. [Google Scholar] [CrossRef]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A. Health Benefits of Fermented Foods: Microbiota and Beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef]
- Cantatore, V.; Filannino, P.; Giuseppe, G.; De Pasquale, I.; Pan, S.; Gobbetti, M.; Di Cagno, R. Lactic Acid Fermentation to Re-Cycle Apple By-Products for Wheat Bread Fortification. Front. Microbiol. 2019, 10, 2574. [Google Scholar] [CrossRef]
- Lemos Júnior, W.J.F.; Guerra, A.F.; da Silva Duarte, V.; Treu, L.; Tarrah, A.; Campanaro, S.; Luchese, R.H.; Giacomini, A.; Corich, V. Draft Genome Sequence Data of Lactobacillus Paracasei Strain DTA83 Isolated from Infant Stools. Data Brief 2019, 22, 1064–1067. [Google Scholar] [CrossRef] [PubMed]
- Guerra, A.F.; Lemos Júnior, W.J.F.; dos Santos, G.O.; Andrighetto, C.; Gianomini, A.; Corich, V.; Luchese, R.H. Lactobacillus Paracasei Probiotic Properties and Survivability Under Stress-Induced by Processing and Storage of Ice Cream Bar or Ice-Lolly. Ciência Rural 2018, 48, e20170601. [Google Scholar] [CrossRef]
- Filannino, P.; Tlais, A.Z.; Morozova, K.; Cavoski, I.; Scampicchio, M.; Gobbetti, M.; Di Cagno, R. Lactic Acid Fermentation Enriches the Profile of Biogenic Fatty Acid Derivatives of Avocado Fruit (Persea Americana Mill.). Food Chem. 2020, 317, 126384. [Google Scholar] [CrossRef] [PubMed]
- Shiferaw Terefe, N.; Augustin, M.A. Fermentation for Tailoring the Technological and Health Related Functionality of Food Products. Crit. Rev. Food Sci. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Michaelsen, K.F.; Hoppe, C.; Roos, N.; Kaestel, P.; Stougaard, M.; Lauritzen, L.; Mølgaard, C.; Girma, T.; Friis, H. Choice of Foods and Ingredients for Moderately Malnourished Children 6 Months to 5 Years of Age. Food Nutr. Bull. 2009, 30, S343–S404. [Google Scholar] [CrossRef] [PubMed]
- Lang, J.M.; Eisen, J.A.; Zivkovic, A.M. The Microbes we Eat: Abundance and Taxonomy of Microbes Consumed in a Day’s Worth of Meals for Three Diet Types. PeerJ 2014, 2, e659. [Google Scholar] [CrossRef] [PubMed]
- Lemos Júnior, W.J.F.; Guerra, A.F.; Tarrah, A.; da Silva Duarte, V.; Giacomini, A.; Luchese, R.H.; Corich, V. Safety and Stability of Two Potentially Probiotic Lactobacillus Strains After in Vitro Gastrointestinal Transit. Probiotics Antimicrob. Proteins 2019, 1–10. [Google Scholar] [CrossRef]
- Derrien, M.; van Hylckama Vlieg, J.E. Fate, Activity, and Impact of Ingested Bacteria within the Human Gut Microbiota. Trends Microbiol. 2015, 23, 354–366. [Google Scholar] [CrossRef]
- Kabak, B.; Dobson, A.D. An Introduction to the Traditional Fermented Foods and Beverages of Turkey. Crit. Rev. Food Sci. Nutr. 2011, 51, 248–260. [Google Scholar] [CrossRef]
- Bell, V.; Ferrão, J.; Fernandes, T. Nutritional Guidelines and Fermented Food Frameworks. Foods 2017, 6, 65. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, D.; Dong, Q.; Zhao, Z. Identifying a Set of Influential Spreaders in Complex Networks. Sci. Rep. 2016, 6, 27823. [Google Scholar] [CrossRef] [PubMed]
- Doeun, D.; Davaatseren, M.; Chung, M. Biogenic Amines in Foods. Food Sci. Biotechnol. 2017, 26, 1463–1474. [Google Scholar] [CrossRef] [PubMed]
- Mataragas, M.; Bellio, A.; Rovetto, F.; Astegiano, S.; Decastelli, L.; Cocolin, L. Risk-Based Control of Food-Borne Pathogens Listeria Monocytogenes and Salmonella Enterica in the Italian Fermented Sausages Cacciatore and Felino. Meat Sci. 2015, 103, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Soro-Yao, A.A.; Kouakou Brou, G.A.; Thonart, P.; Djè, K.M. The use of Lactic Acid Bacteria Starter Cultures during the Processing of Fermented Cereal-Based Foods in West Africa: A Review. Trop. Life Sci. Res. 2014, 25, 81–100. [Google Scholar] [PubMed]
- Montemurro, M.; Pontonio, E.; Gobbetti, M.; Rizzello, C.G. Investigation of the Nutritional, Functional and Technological Effects of the Sourdough Fermentation of Sprouted Flours. Int. J. Food Microbiol. 2019, 302, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Adam, G.O.A.; Hua, Y.; Chamba, M.V.M.; Gasmalla, M.A. Functional Properties and in Vitro Protein Digestibility of Fermented Sorghum and Broad Bean (Visia Faba L. Major) Blended Flour. Pak. J. Food Sci. 2013, 23, 10–16. [Google Scholar]
- Tamang, J.P.; Shin, D.; Jung, S.; Chae, S. Functional Properties of Microorganisms in Fermented Foods. Front. Microbiol. 2016, 7, 578. [Google Scholar] [CrossRef]
- Day, C.N.; Morawicki, R.O. Effects of Fermentation by Yeast and Amylolytic Lactic Acid Bacteria on Grain Sorghum Protein Content and Digestibility. J. Food Qual. 2018, 2018, 3964392. [Google Scholar] [CrossRef]
- Lorusso, A.; Coda, R.; Montemurro, M.; Rizzello, C.G. Use of Selected Lactic Acid Bacteria and Quinoa Flour for Manufacturing Novel Yogurt-Like Beverages. Foods 2018, 7, 51. [Google Scholar] [CrossRef]
- Çabuk, B.; Nosworthy, M.G.; Stone, A.K.; Korber, D.R.; Tanaka, T.; House, J.D.; Nickerson, M.T. Effect of Fermentation on the Protein Digestibility and Levels of Non-Nutritive Compounds of Pea Protein Concentrate. Food Technol. Biotechnol. 2018, 56, 257–264. [Google Scholar] [CrossRef]
- Ghosh, D.; Chattopadhyay, P. Preparation of Idli Batter, its Properties and Nutritional Improvement during Fermentation. J. Food Sci. Technol. 2011, 48, 610–615. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Lee, S.H.; Jin, H.M.; Hahn, Y.; Madsen, E.L.; Jeon, C.O. Metatranscriptomic Analysis of Lactic Acid Bacterial Gene Expression during Kimchi Fermentation. Int. J. Food Microbiol. 2013, 163, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Syal, P.; Vohra, A. Probiotic Potential of Yeasts Isolated from Traditional Indian Fermented Foods. Int. J. Microbiol. Res. 2013, 5, 390–398. [Google Scholar] [CrossRef]
- Sharma, R. Market Trends and Opportunities for Functional Dairy Beverages. Aust. J. Dairy Technol. 2005, 60, 195–198. [Google Scholar]
- Buech, J. The Rising Popularity of Fermented Drinks. Available online: https://web.archive.org/web/20200225200011/https%3A%2F%2Fwww.mintel.com%2Fblog%2Fdrink-market-news%2Fthe-rising-popularity-of-fermented-drinks (accessed on 25 February 2020).
- Caddy, T. Interview with an Innovator: Chef Niklas Ekstedt. Available online: https://web.archive.org/web/20200225200348/https://www.mintel.com/blog/foodservice-market-news/interview-with-an-innovator-chef-niklas-ekstedt (accessed on 25 February 2020).
- Pollock Communications. What’s Trending in Nutrition: Top 10 Superfoods for 2020. Available online: https://www.lpollockpr.com/in-the-news/nutrition-experts-forecast-2020-will-usher-in-the-ultimate-food-revolution/ (accessed on 25 February 2020).
- Askew, K. There is a Mega-Trend around Fermentation: The Rising Star of Fermented Foods. Available online: https://web.archive.org/web/20200225200959/https%3A%2F%2Fwww.foodnavigator.com%2FArticle%2F2018%2F05%2F04%2FThere-is-a-mega-trend-around-fermentation-The-rising-star-of-fermented-foods (accessed on 25 February 2020).
- Xiang, H.; Sun-Waterhouse, D.; Waterhouse, G.I.; Cui, C.; Ruan, Z. Fermentation-Enabled Wellness Foods: A Fresh Perspective. Food Sci. Hum. Wellness 2019, 8, 203–243. [Google Scholar] [CrossRef]
- Dey, G. Non-dairy Probiotic Foods: Innovations and market trends. In Innovations in Technologies for Fermented Food and Beverage Industries; Springer: Berlin/Heidelberg, Germany, 2018; pp. 159–173. [Google Scholar]
- Statista. Global Market Size of Fermentation Products 2018 & 2023; Statista: Hamburg, Germany, 2019. [Google Scholar]
- Research and Markets. Global Fermented Food and Ingredients Market 2019–2023: Focus on Food Type, Ingredient Type and Distribution Channel. Available online: https://web.archive.org/web/20200225200621/https://www.globenewswire.com/news-release/2019/04/17/1805321/0/en/Global-Fermented-Food-and-Ingredients-Market-2019-2023-Focus-on-Food-Type-Ingredient-Type-and-Distribution-Channel.html (accessed on 25 February 2020).
- Kumar, B.V.; Vijayendra, S.V.N.; Reddy, O.V.S. Trends in Dairy and Non-Dairy Probiotic Products-a Review. J. Food Sci. Technol. 2015, 52, 6112–6124. [Google Scholar] [CrossRef]
- Market Data Forecast. Europe Kombucha Market by Types, by Flavours and by Region: Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2019–2024. Available online: https://web.archive.org/web/20200225202112/https://www.marketdataforecast.com/market-reports/europe-kombucha-market (accessed on 25 February 2020).
- FMI. U.S. Grocery Shopper Trends. 2017. Available online: https://www.fmi.org/docs/default-source/webinars/trends-2017-webinar-7-18-2017.pdf (accessed on 25 February 2020).
- Symrise. Food for Future. White Paper. 2019. Available online: https://www.symrise.com/fileadmin/symrise/marketing/flavours_and_wellbeing/culinary/191209_SYM_Factsheet_Alternative_Proteines.pdf (accessed on 25 February 2020).
- Ebner, S.; Smug, L.N.; Kneifel, W.; Salminen, S.J.; Sanders, M.E. Probiotics in Dietary Guidelines and Clinical Recommendations Outside the European Union. World J. Gastroenterol. WJG 2014, 20, 16095. [Google Scholar] [CrossRef]
- Salva, S.; Villena, J.; Alvarez, S. Immunomodulatory Activity of Lactobacillus Rhamnosus Strains Isolated from Goat Milk: Impact on Intestinal and Respiratory Infections. Int. J. Food Microbiol. 2010, 141, 82–89. [Google Scholar] [CrossRef]
- Reid, G.; Kort, R.; Alvarez, S.; Bourdet-Sicard, R.; Benoit, V.; Cunningham, M.; Saulnier, D.M.; van Hylckama Vlieg, J.E.; Verstraelen, H.; Sybesma, W. Expanding the Reach of Probiotics through Social Enterprises. Benef. Microbes 2018, 9, 707–715. [Google Scholar] [CrossRef]
- Westerik, N.; Wacoo, A.P.; Anyimo, E.; Matovu, W.; Reid, G.; Kort, R.; Sybesma, W. Improving Health and Wealth by Introduction of an Affordable Bacterial Starter Culture for Probiotic Yoghurt Production in Uganda. Challenges 2019, 10, 2. [Google Scholar] [CrossRef]
- Wacoo, A.P.; Mukisa, I.M.; Meeme, R.; Byakika, S.; Wendiro, D.; Sybesma, W.; Kort, R. Probiotic Enrichment and Reduction of Aflatoxins in a Traditional African Maize-Based Fermented Food. Nutrients 2019, 11, 265. [Google Scholar] [CrossRef] [PubMed]
- Kort, R.; Westerik, N.; Serrano, L.M.; Douillard, F.P.; Gottstein, W.; Mukisa, I.M.; Tuijn, C.J.; Basten, L.; Hafkamp, B.; Meijer, W.C. A Novel Consortium of Lactobacillus Rhamnosus and Streptococcus Thermophilus for Increased Access to Functional Fermented Foods. Microb. Cell Factories 2015, 14, 195. [Google Scholar] [CrossRef] [PubMed]
- Reale, A.; Konietzny, U.; Coppola, R.; Sorrentino, E.; Greiner, R. The Importance of Lactic Acid Bacteria for Phytate Degradation during Cereal Dough Fermentation. J. Agric. Food Chem. 2007, 55, 2993–2997. [Google Scholar] [CrossRef] [PubMed]
- García-Mantrana, I.; Monedero, V.; Haros, M. Reduction of Phytate in Soy Drink by Fermentation with Lactobacillus Casei Expressing Phytases from Bifidobacteria. Plant Foods Hum. Nutr. 2015, 70, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Amritha, G.K.; Venkateswaran, G. Use of Lactobacilli in Cereal-Legume Fermentation and as Potential Probiotics towards Phytate Hydrolysis. Probiotics Antimicrob. Proteins 2018, 10, 647–653. [Google Scholar] [CrossRef]
- García-Mantrana, I.; Yebra, M.J.; Haros, M.; Monedero, V. Expression of Bifidobacterial Phytases in Lactobacillus Casei and their Application in a Food Model of Whole-Grain Sourdough Bread. Int. J. Food Microbiol. 2016, 216, 18–24. [Google Scholar] [CrossRef]
- Yildirim-Elikoglu, S.; Erdem, Y.K. Interactions between Milk Proteins and Polyphenols: Binding Mechanisms, Related Changes, and the Future Trends in the Dairy Industry. Food Rev. Int. 2018, 34, 665–697. [Google Scholar] [CrossRef]
- Riedl, K.M.; Hagerman, A.E. Tannin−Protein Complexes as Radical Scavengers and Radical Sinks. J. Agric. Food Chem. 2001, 49, 4917–4923. [Google Scholar] [CrossRef]
- de las Rivas, B.; Rodríguez, H.; Anguita, J.; Muñoz, R. Bacterial Tannases: Classification and Biochemical Properties. Appl. Microbiol. Biotechnol. 2019, 103, 603–623. [Google Scholar] [CrossRef]
- Kosińska, A.; Karamać, M.; Penkacik, K.; Urbalewicz, A.; Amarowicz, R. Interactions between Tannins and Proteins Isolated from Broad Bean Seeds (Vicia Faba Major) Yield Soluble and Non-Soluble Complexes. Eur. Food Res. Technol. 2011, 233, 213–222. [Google Scholar] [CrossRef]
- Jiménez, N.; Esteban-Torres, M.; Mancheño, J.M.; de las Rivas, B.; Muñoz, R. Tannin Degradation by a Novel Tannase Enzyme Present in some Lactobacillus Plantarum Strains. Appl. Environ. Microbiol. 2014, 80, 2991–2997. [Google Scholar] [CrossRef] [PubMed]
- Rahbé, Y.; Ferrasson, E.; Rabesona, H.; Quillien, L. Toxicity to the Pea Aphid Acyrthosiphon Pisum of Anti-Chymotrypsin Isoforms and Fragments of Bowman–Birk Protease Inhibitors from Pea Seeds. Insect Biochem. Mol. Biol. 2003, 33, 299–306. [Google Scholar] [CrossRef]
- Chen, Y.; Xu, Z.; Zhang, C.; Kong, X.; Hua, Y. Heat-Induced Inactivation Mechanisms of Kunitz Trypsin Inhibitor and Bowman-Birk Inhibitor in Soymilk Processing. Food Chem. 2014, 154, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Clemente, A.; Jimenez, E.; Marin-Manzano, M.C.; Rubio, L.A. Active Bowman–Birk Inhibitors Survive Gastrointestinal Digestion at the Terminal Ileum of Pigs Fed Chickpea-based Diets. J. Sci. Food Agric. 2008, 88, 513–521. [Google Scholar] [CrossRef]
- Kumar, V.; Rani, A.; Mittal, P.; Shuaib, M. Kunitz Trypsin Inhibitor in Soybean: Contribution to Total Trypsin Inhibitor Activity as a Function of Genotype and Fate during Processing. J. Food Meas. Charact. 2019, 13, 1583–1590. [Google Scholar] [CrossRef]
- Phengnuam, T.; Suntornsuk, W. Detoxification and Anti-Nutrients Reduction of Jatropha Curcas Seed Cake by Bacillus Fermentation. J. Biosci. Bioeng. 2013, 115, 168–172. [Google Scholar] [CrossRef]
- Yousif, N.E.; El Tinay, A.H. Effect of Fermentation on Sorghum Protein Fractions and in Vitro Protein Digestibility. Plant Foods Hum. Nutr. 2001, 56, 175–182. [Google Scholar] [CrossRef]
- Arte, E.; Rizzello, C.G.; Verni, M.; Nordlund, E.; Katina, K.; Coda, R. Impact of Enzymatic and Microbial Bioprocessing on Protein Modification and Nutritional Properties of Wheat Bran. J. Agric. Food Chem. 2015, 63, 8685–8693. [Google Scholar] [CrossRef]
- Osman, M.A. Changes in Sorghum Enzyme Inhibitors, Phytic Acid, Tannins and in Vitro Protein Digestibility Occurring during Khamir (Local Bread) Fermentation. Food Chem. 2004, 88, 129–134. [Google Scholar] [CrossRef]
- Vilcacundo, R.; Barrio, D.; Carpio, C.; García-Ruiz, A.; Rúales, J.; Hernández-Ledesma, B.; Carrillo, W. Digestibility of Quinoa (Chenopodium Quinoa Willd.) Protein Concentrate and its Potential to Inhibit Lipid Peroxidation in the Zebrafish Larvae Model. Plant Foods Hum. Nutr. 2017, 72, 294–300. [Google Scholar] [CrossRef]
- Picariello, G.; Mamone, G.; Nitride, C.; Addeo, F.; Ferranti, P. Protein Digestomics: Integrated Platforms to Study Food-Protein Digestion and Derived Functional and Active Peptides. Trends Anal. Chem. 2013, 52, 120–134. [Google Scholar] [CrossRef]
- Dupont, D.; Alric, M.; Blanquet-Diot, S.; Bornhorst, G.; Cueva, C.; Deglaire, A.; Denis, S.; Ferrua, M.; Havenaar, R.; Lelieveld, J. Can Dynamic in Vitro Digestion Systems Mimic the Physiological Reality? Crit. Rev. Food Sci. Nutr. 2018, 59, 1546–1562. [Google Scholar] [CrossRef] [PubMed]
- Shani-Levi, C.; Alvito, P.; Andrés, A.; Assunҫão, R.; Barberá, R.; Blanquet-Diot, S.; Bourlieu, C.; Brodkorb, A.; Cilla, A.; Deglaire, A. Extending in Vitro Digestion Models to Specific Human Populations: Perspectives, Practical Tools and Bio-Relevant Information. Trends Food Sci. Technol. 2017, 60, 52–63. [Google Scholar] [CrossRef]
- De Pasquale, I.; Pontonio, E.; Gobbetti, M.; Rizzello, C.G. Nutritional and Functional Effects of the Lactic Acid Bacteria Fermentation on Gelatinized Legume Flours. Int. J. Food Microbiol. 2019, 316, 108426. [Google Scholar] [CrossRef] [PubMed]
- Celmeli, T.; Sari, H.; Canci, H.; Sari, D.; Adak, A.; Eker, T.; Toker, C. The Nutritional Content of Common Bean (Phaseolus Vulgaris L.) Landraces in Comparison to Modern Varieties. Agronomy 2018, 8, 166. [Google Scholar] [CrossRef]
- Coda, R.; Melama, L.; Rizzello, C.G.; Curiel, J.A.; Sibakov, J.; Holopainen, U.; Pulkkinen, M.; Sozer, N. Effect of Air Classification and Fermentation by Lactobacillus Plantarum VTT E-133328 on Faba Bean (Vicia Faba L.) Flour Nutritional Properties. Int. J. Food Microbiol. 2015, 193, 34–42. [Google Scholar] [CrossRef]
- Bartkiene, E.; Krungleviciute, V.; Juodeikiene, G.; Vidmantiene, D.; Maknickiene, Z. Solid State Fermentation with Lactic Acid Bacteria to Improve the Nutritional Quality of Lupin and Soya Bean. J. Sci. Food Agric. 2015, 95, 1336–1342. [Google Scholar] [CrossRef]
- Starzyńska-Janiszewska, A.; Stodolak, B.; Mickowska, B. Effect of Controlled Lactic Acid Fermentation on Selected Bioactive and Nutritional Parameters of Tempeh obtained from Unhulled Common Bean (Phaseolus Vulgaris) Seeds. J. Sci. Food Agric. 2014, 94, 359–366. [Google Scholar] [CrossRef]
- Jakubczyk, A.; Karaś, M.; Baraniak, B.; Pietrzak, M. The Impact of Fermentation and in Vitro Digestion on Formation Angiotensin Converting Enzyme (ACE) Inhibitory Peptides from Pea Proteins. Food Chem. 2013, 141, 3774–3780. [Google Scholar] [CrossRef]
- Watson, C.A.; Reckling, M.; Preissel, S.; Bachinger, J.; Bergkvist, G.; Kuhlman, T.; Lindström, K.; Nemecek, T.; Topp, C.F.; Vanhatalo, A. Grain legume production and use in European agricultural systems. In Advances in Agronomy; Elsevier: Amsterdam, The Netherlands, 2017; Volume 144, pp. 235–303. [Google Scholar]
- Magrini, M.; Cabanac, G.; Lascialfari, M.; Plumecocq, G.; Amiot, M.; Anton, M.; Arvisenet, G.; Baranger, A.; Bedoussac, L.; Chardigny, J. Peer-Reviewed Literature on Grain Legume Species in the WoS (1980–2018): A Comparative Analysis of Soybean and Pulses. Sustainability 2019, 11, 6833. [Google Scholar] [CrossRef]
- Sellami, M.H.; Pulvento, C.; Aria, M.; Stellacci, A.M.; Lavini, A. A Systematic Review of Field Trials to Synthesize Existing Knowledge and Agronomic Practices on Protein Crops in Europe. Agronomy 2019, 9, 292. [Google Scholar] [CrossRef]
- Manners, R.; Varela-Ortega, C.; van Etten, J. Protein-Rich Legume and Pseudo-Cereal Crop Suitability under Present and Future European Climates. Eur. J. Agron. 2020, 113, 125974. [Google Scholar] [CrossRef]
- Luo, Y.; Gu, Z.; Han, Y.; Chen, Z. The Impact of Processing on Phytic Acid, in Vitro Soluble Iron and Phy/Fe Molar Ratio of Faba Bean (Vicia Faba L.). J. Sci. Food Agric. 2009, 89, 861–866. [Google Scholar] [CrossRef]
- Gabriele, M.; Sparvoli, F.; Bollini, R.; Lubrano, V.; Longo, V.; Pucci, L. The Impact of Sourdough Fermentation on Non-Nutritive Compounds and Antioxidant Activities of Flours from Different Phaseolus Vulgaris L. Genotypes. J. Food Sci. 2019, 84, 1929–1936. [Google Scholar] [CrossRef] [PubMed]
- Lai, L.; Hsieh, S.; Huang, H.; Chou, C. Effect of Lactic Fermentation on the Total Phenolic, Saponin and Phytic Acid Contents as Well as Anti-Colon Cancer Cell Proliferation Activity of Soymilk. J. Biosci. Bioeng. 2013, 115, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Hirabayashi, M.; Matsui, T.; Yano, H. Fermentation of Soybean Flour with Aspergillus Usamii Improves Availabilities of Zinc and Iron in Rats. J. Nutr. Sci. Vitaminol. 1998, 44, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Rekha, C.R.; Vijayalakshmi, G. Bioconversion of Isoflavone Glycosides to Aglycones, Mineral Bioavailability and Vitamin B Complex in Fermented Soymilk by Probiotic Bacteria and Yeast. J. Appl. Microbiol. 2010, 109, 1198–1208. [Google Scholar] [CrossRef]
- Bahaciu, G.V.; Nicolae, C.G.; Șuler, A.D.; Segal, R. Germinated and Lactic Fermented Soybean Seeds, a Natural Alternative for Healthy Bones. A Scientific Approach. Bull. UASVM Food Sci. Technol. 2018, 75, 1. [Google Scholar] [CrossRef]
- Granito, M.; Frias, J.; Doblado, R.; Guerra, M.; Champ, M.; Vidal-Valverde, C. Nutritional Improvement of Beans (Phaseolus Vulgaris) by Natural Fermentation. Eur. Food Res. Technol. 2002, 214, 226–231. [Google Scholar] [CrossRef]
- Curiel, J.A.; Coda, R.; Centomani, I.; Summo, C.; Gobbetti, M.; Rizzello, C.G. Exploitation of the Nutritional and Functional Characteristics of Traditional Italian Legumes: The Potential of Sourdough Fermentation. Int. J. Food Microbiol. 2015, 196, 51–61. [Google Scholar] [CrossRef]
- Shimelis, E.A.; Rakshit, S.K. Influence of Natural and Controlled Fermentations on A-galactosides, Antinutrients and Protein Digestibility of Beans (Phaseolus Vulgaris L.). Int. J. Food Sci. Technol. 2008, 43, 658–665. [Google Scholar] [CrossRef]
- Egounlety, M.; Aworh, O.C. Effect of Soaking, Dehulling, Cooking and Fermentation with Rhizopus Oligosporus on the Oligosaccharides, Trypsin Inhibitor, Phytic Acid and Tannins of Soybean (Glycine Max Merr.), Cowpea (Vigna Unguiculata L. Walp) and Groundbean (Macrotyloma Geocarpa Harms). J. Food Eng. 2003, 56, 249–254. [Google Scholar]
- Jakubczyk, A.; Karaś, M.; Złotek, U.; Szymanowska, U. Identification of Potential Inhibitory Peptides of Enzymes Involved in the Metabolic Syndrome obtained by Simulated Gastrointestinal Digestion of Fermented Bean (Phaseolus Vulgaris L.) Seeds. Food Res. Int. 2017, 100, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Bondia-Pons, I.; Nordlund, E.; Mattila, I.; Katina, K.; Aura, A.; Kolehmainen, M.; Orešič, M.; Mykkänen, H.; Poutanen, K. Postprandial Differences in the Plasma Metabolome of Healthy Finnish Subjects After Intake of a Sourdough Fermented Endosperm Rye Bread Versus White Wheat Bread. Nutr. J. 2011, 10, 116. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R. Improving the Protein Content and Composition of Cereal Grain. J. Cereal Sci. 2007, 46, 239–250. [Google Scholar] [CrossRef]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Whole Grain and Refined Grain Consumption and the Risk of Type 2 Diabetes: A Systematic Review and Dose–response Meta-Analysis of Cohort Studies. Eur. J. Epidemiol. 2013, 28, 845–858. [Google Scholar] [CrossRef]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Whole Grain Consumption and Risk of Cardiovascular Disease, Cancer, and all Cause and Cause Specific Mortality: Systematic Review and Dose-Response Meta-Analysis of Prospective Studies. BMJ 2016, 353, i2716. [Google Scholar] [CrossRef]
- De Mello, V.; Schwab, U.; Kolehmainen, M.; Koenig, W.; Siloaho, M.; Poutanen, K.; Mykkänen, H.; Uusitupa, M. A Diet High in Fatty Fish, Bilberries and Wholegrain Products Improves Markers of Endothelial Function and Inflammation in Individuals with Impaired Glucose Metabolism in a Randomised Controlled Trial: The Sysdimet Study. Diabetologia 2011, 54, 2755–2767. [Google Scholar] [CrossRef]
- Giacco, D.; Matanov, A.; Priebe, S. Providing Mental Healthcare to Immigrants: Current Challenges and New Strategies. Curr. Opin. Psychol. 2014, 27, 282–288. [Google Scholar] [CrossRef]
- Kallio, P.; Kolehmainen, M.; Laaksonen, D.E.; Pulkkinen, L.; Atalay, M.; Mykkänen, H.; Uusitupa, M.; Poutanen, K.; Niskanen, L. Inflammation Markers are Modulated by Responses to Diets Differing in Postprandial Insulin Responses in Individuals with the Metabolic Syndrome. Am. J. Clin. Nutr. 2008, 87, 1497–1503. [Google Scholar] [CrossRef]
- Ross, A.B.; Bruce, S.J.; Blondel-Lubrano, A.; Oguey-Araymon, S.; Beaumont, M.; Bourgeois, A.; Nielsen-Moennoz, C.; Vigo, M.; Fay, L.; Kochhar, S. A Whole-Grain Cereal-Rich Diet Increases Plasma Betaine, and Tends to Decrease Total and LDL-Cholesterol Compared with a Refined-Grain Diet in Healthy Subjects. Br. J. Nutr. 2011, 105, 1492–1502. [Google Scholar] [CrossRef] [PubMed]
- Koistinen, V.M.; Mattila, O.; Katina, K.; Poutanen, K.; Aura, A.; Hanhineva, K. Metabolic Profiling of Sourdough Fermented Wheat and Rye Bread. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Lappi, J.; Selinheimo, E.; Schwab, U.; Katina, K.; Lehtinen, P.; Mykkänen, H.; Kolehmainen, M.; Poutanen, K. Sourdough Fermentation of Wholemeal Wheat Bread Increases Solubility of Arabinoxylan and Protein and Decreases Postprandial Glucose and Insulin Responses. J. Cereal Sci. 2010, 51, 152–158. [Google Scholar] [CrossRef]
- Marsh, A.J.; Hill, C.; Ross, R.P.; Cotter, P.D. Fermented Beverages with Health-Promoting Potential: Past and Future Perspectives. Trends Food Sci. Technol. 2014, 38, 113–124. [Google Scholar] [CrossRef]
- Repo-Carrasco-Valencia, R.A.; Encina, C.R.; Binaghi, M.J.; Greco, C.B.; Ronayne de Ferrer, P.A. Effects of Roasting and Boiling of Quinoa, Kiwicha and Kañiwa on Composition and Availability of Minerals in Vitro. J. Sci. Food Agric. 2010, 90, 2068–2073. [Google Scholar] [CrossRef] [PubMed]
- Dakhili, S.; Abdolalizadeh, L.; Hosseini, S.M.; Shojaee-Aliabadi, S.; Mirmoghtadaie, L. Quinoa Protein: Composition, Structure and Functional Properties. Food Chem. 2019, 299, 125161. [Google Scholar] [CrossRef]
- Committee for the Common Organisation of Agricultural Markets. EU Cereals Market. 2019/20 Marketing Year. Cereals; European Commission: Brussels, Belgium, 2019. [Google Scholar]
- Montemurro, M.; Coda, R.; Rizzello, C.G. Recent Advances in the use of Sourdough Biotechnology in Pasta Making. Foods 2019, 8, 129. [Google Scholar] [CrossRef]
- Belton, P.S.; Taylor, J.R. Sorghum and Millets: Protein Sources for Africa. Trends Food Sci. Technol. 2004, 15, 94–98. [Google Scholar] [CrossRef]
- Simnadis, T.G.; Tapsell, L.C.; Beck, E.J. Physiological Effects Associated with Quinoa Consumption and Implications for Research Involving Humans: A Review. Plant Foods Hum. Nutr. 2015, 70, 238–249. [Google Scholar] [CrossRef]
- Ludena Urquizo, F.E.; García Torres, S.M.; Tolonen, T.; Jaakkola, M.; Pena-Niebuhr, M.G.; von Wright, A.; Repo-Carrasco-Valencia, R.; Korhonen, H.; Plumed-Ferrer, C. Development of a Fermented Quinoa-based Beverage. Food Sci. Nutr. 2017, 5, 602–608. [Google Scholar] [CrossRef]
- Iglesias-Puig, E.; Monedero, V.; Haros, M. Bread with Whole Quinoa Flour and Bifidobacterial Phytases Increases Dietary Mineral Intake and Bioavailability. LWT 2015, 60, 71–77. [Google Scholar] [CrossRef]
- Multari, S.; Marsol-Vall, A.; Keskitalo, M.; Yang, B.; Suomela, J. Effects of Different Drying Temperatures on the Content of Phenolic Compounds and Carotenoids in Quinoa Seeds (Chenopodium Quinoa) from Finland. J. Food Compos. Anal. 2018, 72, 75–82. [Google Scholar] [CrossRef]
- Rasane, P.; Jha, A.; Sabikhi, L.; Kumar, A.; Unnikrishnan, V.S. Nutritional Advantages of Oats and Opportunities for its Processing as Value Added Foods-a Review. J. Food Sci. Technol. 2015, 52, 662–675. [Google Scholar] [CrossRef] [PubMed]
- Petkov, K.; Biel, W.; Kowieska, A.; Jaskowska, I. The Composition and Nutritive Value of Naked Oat Grain (Avena Sativa var. Nuda). J. Anim. Feed Sci. 2001, 10, 303–308. [Google Scholar] [CrossRef]
- Luana, N.; Rossana, C.; Curiel, J.A.; Kaisa, P.; Marco, G.; Rizzello, C.G. Manufacture and Characterization of a Yogurt-Like Beverage made with Oat Flakes Fermented by Selected Lactic Acid Bacteria. Int. J. Food Microbiol. 2014, 185, 17–26. [Google Scholar] [CrossRef]
- Espinosa-Páez, E.; Alanis-Guzmán, M.; Hernández-Luna, C.; Báez-González, J.; Amaya-Guerra, C.; Andrés-Grau, A. Increasing Antioxidant Activity and Protein Digestibility in Phaseolus Vulgaris and Avena Sativa by Fermentation with the Pleurotus Ostreatus Fungus. Molecules 2017, 22, 2275. [Google Scholar] [CrossRef]
- Cai, S.; Gao, F.; Zhang, X.; Wang, O.; Wu, W.; Zhu, S.; Zhang, D.; Zhou, F.; Ji, B. Evaluation of Γ-Aminobutyric Acid, Phytate and Antioxidant Activity of Tempeh-Like Fermented Oats (Avena Sativa L.) Prepared with Different Filamentous Fungi. J. Food Sci. Technol. 2014, 51, 2544–2551. [Google Scholar] [CrossRef]
- Ozkaya, H.; Ozkaya, B.; Duman, B.; Turksoy, S. Effect of Dephytinization by Fermentation and Hydrothermal Autoclaving Treatments on the Antioxidant Activity, Dietary Fiber, and Phenolic Content of Oat Bran. J. Agric. Food Chem. 2017, 65, 5713–5719. [Google Scholar] [CrossRef]
- Eklund-Jonsson, C.; Sandberg, A.; Alminger, M.L. Reduction of Phytate Content while Preserving Minerals during Whole Grain Cereal Tempe Fermentation. J. Cereal Sci. 2006, 44, 154–160. [Google Scholar] [CrossRef]
- Zhang, H.; Önning, G.; Öste, R.; Gramatkovski, E.; Hulthén, L. Improved Iron Bioavailability in an Oat-Based Beverage: The Combined Effect of Citric Acid Addition, Dephytinization and Iron Supplementation. Eur. J. Nutr. 2007, 46, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; White, P.J. In Vitro Fermentation of Oat Flours from Typical and High Β-Glucan Oat Lines. J. Agric. Food Chem. 2009, 57, 7529–7536. [Google Scholar] [CrossRef] [PubMed]
- Davila-Gay, A.; Blachier, F.; Gotteland, M.; Andriamihaja, M.; Benetti, P.; Sanz, Y.; Tomé, D. Intestinal Luminal Nitrogen Metabolism: Role of the Gut Microbiota and Consequences for the Host. Pharmacol. Res. 2013, 68, 95–107. [Google Scholar] [CrossRef] [PubMed]
- Kedia, G.; Vázquez, J.A.; Pandiella, S.S. Enzymatic Digestion and in Vitro Fermentation of Oat Fractions by Human Lactobacillus Strains. Enzym. Microb. Technol. 2008, 43, 355–361. [Google Scholar] [CrossRef]
- Kamal-Eldin, A.; Lærke, H.N.; Knudsen, K.B.; Lampi, A.; Piironen, V.; Adlercreutz, H.; Katina, K.; Poutanen, K.; Åman, P. Physical, Microscopic and Chemical Characterisation of Industrial Rye and Wheat Brans from the Nordic Countries. Food Nutr. Res. 2009, 53, 1912. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.M.; Damstrup, M.L.; Dal Thomsen, A.; Rasmussen, S.K.; Hansen, Å. Phytase Activity and Degradation of Phytic Acid during Rye Bread Making. Eur. Food Res. Technol. 2007, 225, 173–181. [Google Scholar] [CrossRef]
- Morifuji, M.; Ishizaka, M.; Baba, S.; Fukuda, K.; Matsumoto, H.; Koga, J.; Kanegae, M.; Higuchi, M. Comparison of Different Sources and Degrees of Hydrolysis of Dietary Protein: Effect on Plasma Amino Acids, Dipeptides, and Insulin Responses in Human Subjects. J. Agric. Food Chem. 2010, 58, 8788–8797. [Google Scholar] [CrossRef]
- Waters, D.M.; Mauch, A.; Coffey, A.; Arendt, E.K.; Zannini, E. Lactic Acid Bacteria as a Cell Factory for the Delivery of Functional Biomolecules and Ingredients in Cereal-Based Beverages: A Review. Crit. Rev. Food Sci. Nutr. 2015, 55, 503–520. [Google Scholar] [CrossRef]
- Bos, C.; Juillet, B.; Fouillet, H.; Turlan, L.; Daré, S.; Luengo, C.; N’tounda, R.; Benamouzig, R.; Gausserès, N.; Tomé, D. Postprandial Metabolic Utilization of Wheat Protein in Humans. Am. J. Clin. Nutr. 2005, 81, 87–94. [Google Scholar] [CrossRef]
- Balk, J.; Connorton, J.M.; Wan, Y.; Lovegrove, A.; Moore, K.L.; Uauy, C.; Sharp, P.A.; Shewry, P.R. Improving Wheat as a Source of Iron and Zinc for Global Nutrition. Nutr. Bull. 2019, 44, 53–59. [Google Scholar] [CrossRef]
- Asseng, S.; Martre, P.; Maiorano, A.; Rötter, R.P.; O’Leary, G.J.; Fitzgerald, G.J.; Girousse, C.; Motzo, R.; Giunta, F.; Babar, M.A. Climate Change Impact and Adaptation for Wheat Protein. Glob. Chang. Biol. 2019, 25, 155–173. [Google Scholar] [CrossRef]
- Lena, G.D.; Patroni, E.; Quaglia, G.B. Improving the Nutritional Value of Wheat Bran by a White-rot Fungus. Int. J. Food Sci. Technol. 1997, 32, 513–519. [Google Scholar] [CrossRef]
- Dallagnol, A.M.; Pescuma, M.; De Valdez, G.F.; Rollán, G. Fermentation of Quinoa and Wheat Slurries by Lactobacillus Plantarum CRL 778: Proteolytic Activity. Appl. Microbiol. Biotechnol. 2013, 97, 3129–3140. [Google Scholar] [CrossRef] [PubMed]
- Kostekli, M.; Karakaya, S. Protease Inhibitors in various Flours and Breads: Effect of Fermentation, Baking and in Vitro Digestion on Trypsin and Chymotrypsin Inhibitory Activities. Food Chem. 2017, 224, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Makokha, A.O.; Oniang’o, R.K.; Njoroge, S.M.; Kamar, O.K. Effect of Traditional Fermentation and Malting on Phytic Acid and Mineral Availability from Sorghum (Sorghum Bicolor) and Finger Millet (Eleusine Coracana) Grain Varieties Grown in Kenya. Food Nutr. Bull. 2002, 23, 241–245. [Google Scholar] [CrossRef]
- Mohapatra, D.; Patel, A.S.; Kar, A.; Deshpande, S.S.; Tripathi, M.K. Effect of Different Processing Conditions on Proximate Composition, Anti-Oxidants, Anti-Nutrients and Amino Acid Profile of Grain Sorghum. Food Chem. 2019, 271, 129–135. [Google Scholar] [CrossRef]
- Mugula, J.K.; Sørhaug, T.; Stepaniak, L. Proteolytic Activities in Togwa, a Tanzanian Fermented Food. Int. J. Food Microbiol. 2003, 84, 1–12. [Google Scholar] [CrossRef]
- Ochanda, S.O.; Akoth, O.C.; Mwasaru, A.M.; Kagwiria, O.J.; Mathooko, F.M. Effects of Malting and Fermentation Treatments on Group B-Vitamins of Red Sorghum, White Sorghum and Pearl Millets in Kenya. J. Appl. Biosci. 2010, 34, 2128–2134. [Google Scholar]
- Väkeväinen, K.; Ludena-Urquizo, F.; Korkala, E.; Lapveteläinen, A.; Peräniemi, S.; von Wright, A.; Plumed-Ferrer, C. Potential of Quinoa in the Development of Fermented Spoonable Vegan Products. LWT 2020, 120, 108912. [Google Scholar] [CrossRef]
- Carrizo, S.L.; de LeBlanc, A.D.M.; LeBlanc, J.G.; Rollán, G.C. Quinoa Pasta Fermented with Lactic Acid Bacteria Prevents Nutritional Deficiencies in Mice. Food Res. Int. 2020, 127, 108735. [Google Scholar] [CrossRef]
- Castro-Alba, V.; Lazarte, C.E.; Perez-Rea, D.; Carlsson, N.; Almgren, A.; Bergenståhl, B.; Granfeldt, Y. Fermentation of Pseudocereals Quinoa, Canihua, and Amaranth to Improve Mineral Accessibility through Degradation of Phytate. J. Sci. Food Agric. 2019, 99, 5239–5248. [Google Scholar] [CrossRef]
- Castro-Alba, V.; Lazarte, C.E.; Perez-Rea, D.; Sandberg, A.; Carlsson, N.; Almgren, A.; Bergenståhl, B.; Granfeldt, Y. Effect of Fermentation and Dry Roasting on the Nutritional Quality and Sensory Attributes of Quinoa. Food Sci. Nutr. 2019, 7, 3902–3911. [Google Scholar] [CrossRef] [PubMed]
- Valencia, S.; Svanberg, U.; Sandberg, A.-S.; Ruales, J. Processing of Quinoa (Chenopodium Quinoa, Willd): Effects on in Vitro Iron Availability and Phytate Hydrolysis. Int. J. Food Sci. Nutr. 1999, 50, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Rizzello, C.G.; Lorusso, A.; Montemurro, M.; Gobbetti, M. Use of Sourdough Made with Quinoa (Chenopodium Quinoa) Flour and Autochthonous Selected Lactic Acid Bacteria For Enhancing the Nutritional, Textural and Sensory Features of White Bread. Food Microbiol. 2016, 56, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Adeyeye, S.A.O.; Adebayo-Oyetoro, A.O.; Fayemi, O.E.; Tiamiyu, H.K.; Oke, E.K.; Soretire, A.A. Effect of Co-Fermentation on Nutritional Composition, Anti-Nutritional Factors and Acceptability of Cookies from Fermented Sorghum (Sorghum Bicolor) and Soybeans (Glycine Max) Flour Blends. J. Culin. Sci. Technol. 2019, 17, 59–74. [Google Scholar] [CrossRef]
- Lorusso, A.; Verni, M.; Montemurro, M.; Coda, R.; Gobbetti, M.; Rizzello, C.G. Use of Fermented Quinoa Flour for Pasta Making and Evaluation of the Technological and Nutritional Features. LWT 2017, 78, 215–221. [Google Scholar] [CrossRef]
- Poulsen, H.D.; Blaabjerg, K. Fermentation of Rapeseed Meal, Sunflower Meal and Faba Beans in Combination with Wheat Bran Increases Solubility of Protein and Phosphorus. J. Sci. Food Agric. 2017, 97, 244–251. [Google Scholar] [CrossRef]
- Uusitupa, M.; Schwab, U.; Mäkimattila, S.; Karhapää, P.; Sarkkinen, E.; Maliranta, H.; Agren, J.; Penttilä, I. Effects of Two High-Fat Diets with Different Fatty Acid Compositions on Glucose and Lipid Metabolism in Healthy Young Women. Am. J. Clin. Nutr. 1994, 59, 1310–1316. [Google Scholar] [CrossRef]
- Lankinen, M.; Schwab, U.; Gopalacharyulu, P.V.; Seppänen-Laakso, T.; Yetukuri, L.; Sysi-Aho, M.; Kallio, P.; Suortti, T.; Laaksonen, D.E.; Gylling, H. Dietary Carbohydrate Modification Alters Serum Metabolic Profiles in Individuals with the Metabolic Syndrome. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 249–257. [Google Scholar] [CrossRef]
- Lankinen, M.; Schwab, U.; Kolehmainen, M.; Paananen, J.; Poutanen, K.; Mykkänen, H.; Seppänen-Laakso, T.; Gylling, H.; Uusitupa, M.; Orešič, M. Whole Grain Products, Fish and Bilberries Alter Glucose and Lipid Metabolism in a Randomized, Controlled Trial: The Sysdimet Study. PLoS ONE 2011, 6, e22646. [Google Scholar] [CrossRef]
- Burd, N.A.; Cermak, N.M.; Kouw, I.W.; Gorissen, S.H.; Gijsen, A.P.; Van Loon, L.J. The use of Doubly Labeled Milk Protein to Measure Postprandial Muscle Protein Synthesis Rates in Vivo in Humans. J. Appl. Physiol. 2014, 117, 1363–1370. [Google Scholar] [CrossRef]
- Van Loon, L.; Boirie, Y.; Gijsen, A.P.; Fauquant, J.; De Roos, A.L.; Kies, A.K.; Lemosquet, S.; Saris, W.; Koopman, R. The Production of Intrinsically Labeled Milk Protein Provides a Functional Tool for Human Nutrition Research. J. Dairy Sci. 2009, 92, 4812–4822. [Google Scholar] [CrossRef] [PubMed]
- Pennings, B.; Pellikaan, W.F.; Senden, J.M.; van Vuuren, A.M.; Sikkema, J.; van Loon, L.J. The Production of Intrinsically Labeled Milk and Meat Protein is Feasible and Provides Functional Tools for Human Nutrition Research. J. Dairy Sci. 2011, 94, 4366–4373. [Google Scholar] [CrossRef] [PubMed]
- van Vliet, S.; Beals, J.W.; Parel, J.T.; Hanna, C.D.; Utterback, P.L.; Dilger, A.C.; Ulanov, A.V.; Li, Z.; Paluska, S.A.; Moore, D.R. Development of Intrinsically Labeled Eggs and Poultry Meat for use in Human Metabolic Research. J. Nutr. 2016, 146, 1428–1433. [Google Scholar] [CrossRef] [PubMed]
- Evenepoel, P.; Claus, D.; Geypens, B.; Hiele, M.; Geboes, K.; Rutgeerts, P.; Ghoos, Y. Amount and Fate of Egg Protein Escaping Assimilation in the Small Intestine of Humans. Am. J. Physiol.-Gastrointest. Liver Physiol. 1999, 277, G935–G943. [Google Scholar] [CrossRef]
- Bos, C.; Airinei, G.; Mariotti, F.; Benamouzig, R.; Bérot, S.; Evrard, J.; Fénart, E.; Tomé, D.; Gaudichon, C. The Poor Digestibility of Rapeseed Protein is Balanced by its very High Metabolic Utilization in Humans. J. Nutr. 2007, 137, 594–600. [Google Scholar] [CrossRef]
- Devi, S.; Varkey, A.; Sheshshayee, M.S.; Preston, T.; Kurpad, A.V. Measurement of Protein Digestibility in Humans by a Dual-Tracer Method. Am. J. Clin. Nutr. 2018, 107, 984–991. [Google Scholar] [CrossRef]
- Rafii, M.; Elango, R.; Ball, R.O.; Pencharz, P.B.; Courtney-Martin, G. Metabolic Availability of the Limiting Amino Acids Lysine and Tryptophan in Cooked White African Cornmeal Assessed in Healthy Young Men using the Indicator Amino Acid Oxidation Technique. J. Nutr. 2018, 148, 917–924. [Google Scholar] [CrossRef]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F. INFOGEST Static in Vitro Simulation of Gastrointestinal Food Digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef]
- Vinderola, G.; Gueimonde, M.; Gomez-Gallego, C.; Delfederico, L.; Salminen, S. Correlation between in Vitro and in Vivo Assays in Selection of Probiotics from Traditional Species of Bacteria. Trends Food Sci. Technol. 2017, 68, 83–90. [Google Scholar] [CrossRef]
- Bering, S.; Suchdev, S.; Sjøltov, L.; Berggren, A.; Tetens, I.; Bukhave, K. A Lactic Acid-Fermented Oat Gruel Increases Non-Haem Iron Absorption from a Phytate-Rich Meal in Healthy Women of Childbearing Age. Br. J. Nutr. 2006, 96, 80–85. [Google Scholar] [CrossRef]
- Lopez, H.W.; Duclos, V.; Coudray, C.; Krespine, V.; Feillet-Coudray, C.; Messager, A.; Demigné, C.; Rémésy, C. Making Bread with Sourdough Improves Mineral Bioavailability from Reconstituted Whole Wheat Flour in Rats. Nutrition 2003, 19, 524–530. [Google Scholar] [CrossRef]
- Hirabayashi, M.; Matsui, T.; Yano, H. Fermentation of Soybean Meal withAspergillus Usamii Improves Zinc Availability in Rats. Biol. Trace Elem. Res. 1998, 61, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Marles, R.J.; Roe, A.L.; Oketch-Rabah, H.A. US Pharmacopeial Convention Safety Evaluation of Menaquinone-7, a Form of Vitamin K. Nutr. Rev. 2017, 75, 553–578. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Kakuda, H.; Gao, Y.H.; Tsukamoto, Y. Prolonged Intake of Fermented Soybean (Natto) Diets Containing Vitamin K 2 (Menaquinone-7) Prevents Bone Loss in Ovariectomized Rats. J. Bone Miner. Metab. 2000, 18, 71–76. [Google Scholar] [CrossRef]
- Lucas, M.M.; Stoddard, F.L.; Annicchiarico, P.; Frias, J.; Martinez-Villaluenga, C.; Sussmann, D.; Duranti, M.; Seger, A.; Zander, P.M.; Pueyo, J.J. The Future of Lupin as a Protein Crop in Europe. Front. Plant Sci. 2015, 6, 705. [Google Scholar] [CrossRef]
- Zaworska, A.; Frankiewicz, A.; Kasprowicz-Potocka, M. The Influence of Narrow-Leafed Lupin Seed Fermentation on their Chemical Composition and Ileal Digestibility and Microbiota in Growing Pigs. Arch. Anim. Nutr. 2017, 71, 285–296. [Google Scholar] [CrossRef]
- Alireza Sadeghi, M.; Bhagya, S. Quality Characterization of Pasta Enriched with Mustard Protein Isolate. J. Food Sci. 2008, 73, S229–S237. [Google Scholar] [CrossRef]
- Schutyser, M.; Pelgrom, P.; Van der Goot, A.J.; Boom, R.M. Dry Fractionation for Sustainable Production of Functional Legume Protein Concentrates. Trends Food Sci. Technol. 2015, 45, 327–335. [Google Scholar] [CrossRef]
- van der Goot, A.J.; Pelgrom, P.J.; Berghout, J.A.; Geerts, M.E.; Jankowiak, L.; Hardt, N.A.; Keijer, J.; Schutyser, M.A.; Nikiforidis, C.V.; Boom, R.M. Concepts for further Sustainable Production of Foods. J. Food Eng. 2016, 168, 42–51. [Google Scholar] [CrossRef]
- Barisone, G.A.; O’Donnell, R.T.; Ma, Y.; Abuhay, M.W.; Lundeberg, K.; Gowda, S.; Tuscano, J.M. A Purified, Fermented, Extract of Triticum Aestivum has Lymphomacidal Activity Mediated Via Natural Killer Cell Activation. PLoS ONE 2018, 13, e0190860. [Google Scholar] [CrossRef]
- Chandra-Hioe, M.V.; Wong, C.H.; Arcot, J. The Potential use of Fermented Chickpea and Faba Bean Flour as Food Ingredients. Plant Foods Hum. Nutr. 2016, 71, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Wang, J.; Yang, S.; Feng, J.; Jia, F.; Zhang, C. Protein Degradation in Wheat Sourdough Fermentation with Lactobacillus Plantarum M616. Interdiscip. Sci. 2015, 7, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Angelov, A.; Yaneva-Marinova, T.; Gotcheva, V. Oats as a Matrix of Choice for Developing Fermented Functional Beverages. J. Food Sci. Technol. 2018, 55, 2351–2360. [Google Scholar] [CrossRef] [PubMed]
- Dlusskaya, E.; Jänsch, A.; Schwab, C.; Gänzle, M.G. Microbial and Chemical Analysis of a Kvass Fermentation. Eur. Food Res. Technol. 2008, 227, 261–266. [Google Scholar] [CrossRef]
- Osimani, A.; Garofalo, C.; Aquilanti, L.; Milanović, V.; Clementi, F. Unpasteurised Commercial Boza as a Source of Microbial Diversity. Int. J. Food Microbiol. 2015, 194, 62–70. [Google Scholar] [CrossRef]
- Axel, C.; Zannini, E.; Arendt, E.K. Mold Spoilage of Bread and its Biopreservation: A Review of Current Strategies for Bread Shelf Life Extension. Crit. Rev. Food Sci. Nutr. 2017, 57, 3528–3542. [Google Scholar] [CrossRef]
- Gaggia, F.; Di Gioia, D.; Baffoni, L.; Biavati, B. The Role of Protective and Probiotic Cultures in Food and Feed and their Impact in Food Safety. Trends Food Sci. Technol. 2011, 22, S58–S66. [Google Scholar] [CrossRef]
- Dalié, D.; Deschamps, A.M.; Richard-Forget, F. Lactic Acid bacteria–Potential for Control of Mould Growth and Mycotoxins: A Review. Food Control 2010, 21, 370–380. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kårlund, A.; Gómez-Gallego, C.; Korhonen, J.; Palo-oja, O.-M.; El-Nezami, H.; Kolehmainen, M. Harnessing Microbes for Sustainable Development: Food Fermentation as a Tool for Improving the Nutritional Quality of Alternative Protein Sources. Nutrients 2020, 12, 1020. https://doi.org/10.3390/nu12041020
Kårlund A, Gómez-Gallego C, Korhonen J, Palo-oja O-M, El-Nezami H, Kolehmainen M. Harnessing Microbes for Sustainable Development: Food Fermentation as a Tool for Improving the Nutritional Quality of Alternative Protein Sources. Nutrients. 2020; 12(4):1020. https://doi.org/10.3390/nu12041020
Chicago/Turabian StyleKårlund, Anna, Carlos Gómez-Gallego, Jenni Korhonen, Outi-Maaria Palo-oja, Hani El-Nezami, and Marjukka Kolehmainen. 2020. "Harnessing Microbes for Sustainable Development: Food Fermentation as a Tool for Improving the Nutritional Quality of Alternative Protein Sources" Nutrients 12, no. 4: 1020. https://doi.org/10.3390/nu12041020
APA StyleKårlund, A., Gómez-Gallego, C., Korhonen, J., Palo-oja, O.-M., El-Nezami, H., & Kolehmainen, M. (2020). Harnessing Microbes for Sustainable Development: Food Fermentation as a Tool for Improving the Nutritional Quality of Alternative Protein Sources. Nutrients, 12(4), 1020. https://doi.org/10.3390/nu12041020






