High-intensity Interval Training Promotes the Shift to a Health-Supporting Dietary Pattern in Young Adults
Abstract
1. Introduction
2. Materials and Methods
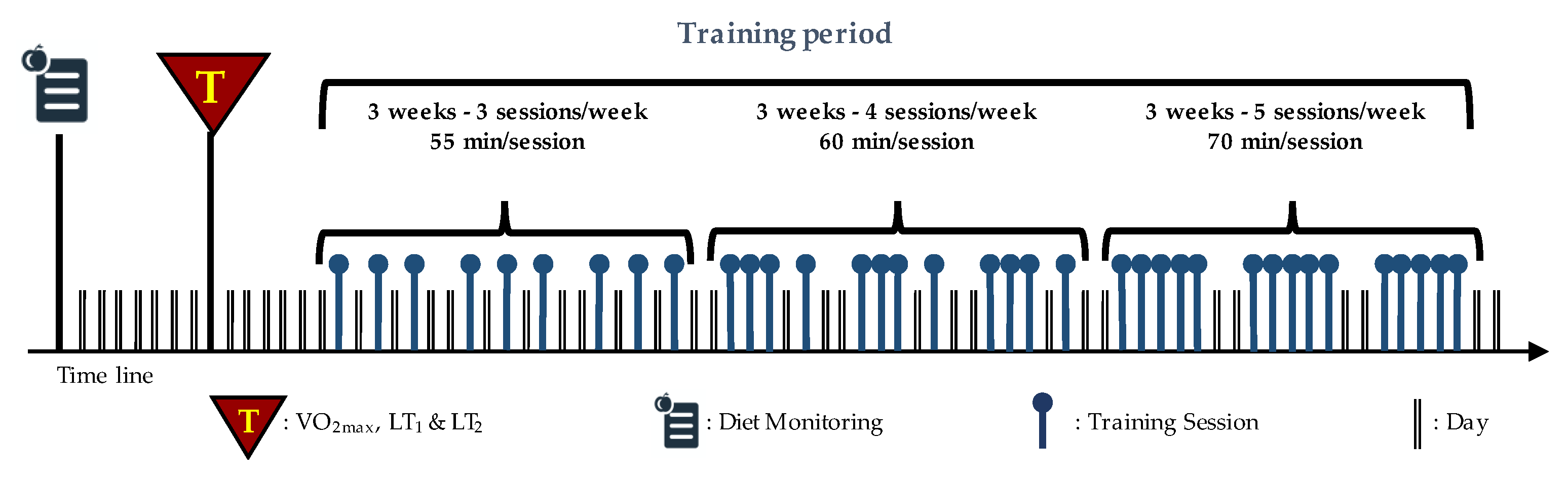
2.1. Study Design
2.2. Participants
2.3. Training Protocol
2.4. Body Composition Assessment
2.5. Diet Monitoring
- total energy intake and macronutrient quantity;
- starch, soluble and insoluble fibre, glycaemic index and glycaemic load;
- saturated, monounsaturated, polyunsaturated fat, omega-3, Omega-6, cholesterol;
- iron, vitamin C, A, E.
2.6. Statistical Analysis
3. Results
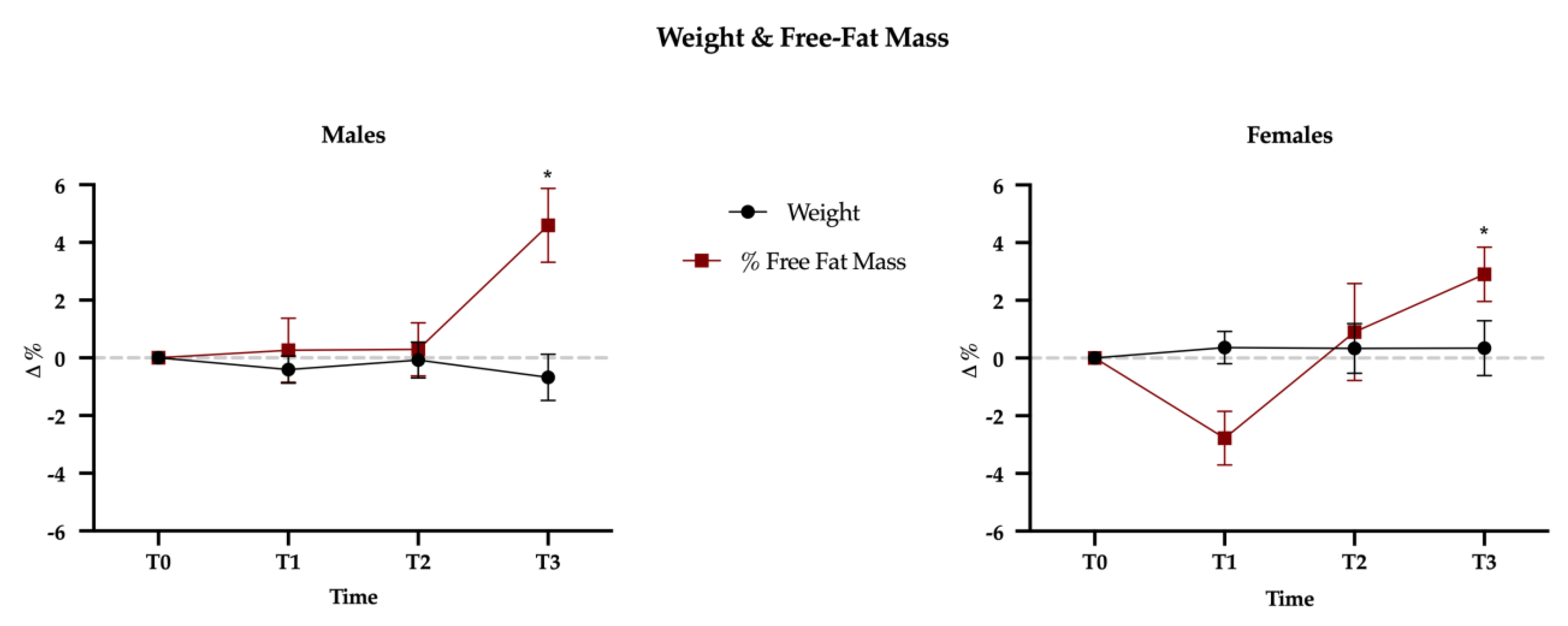
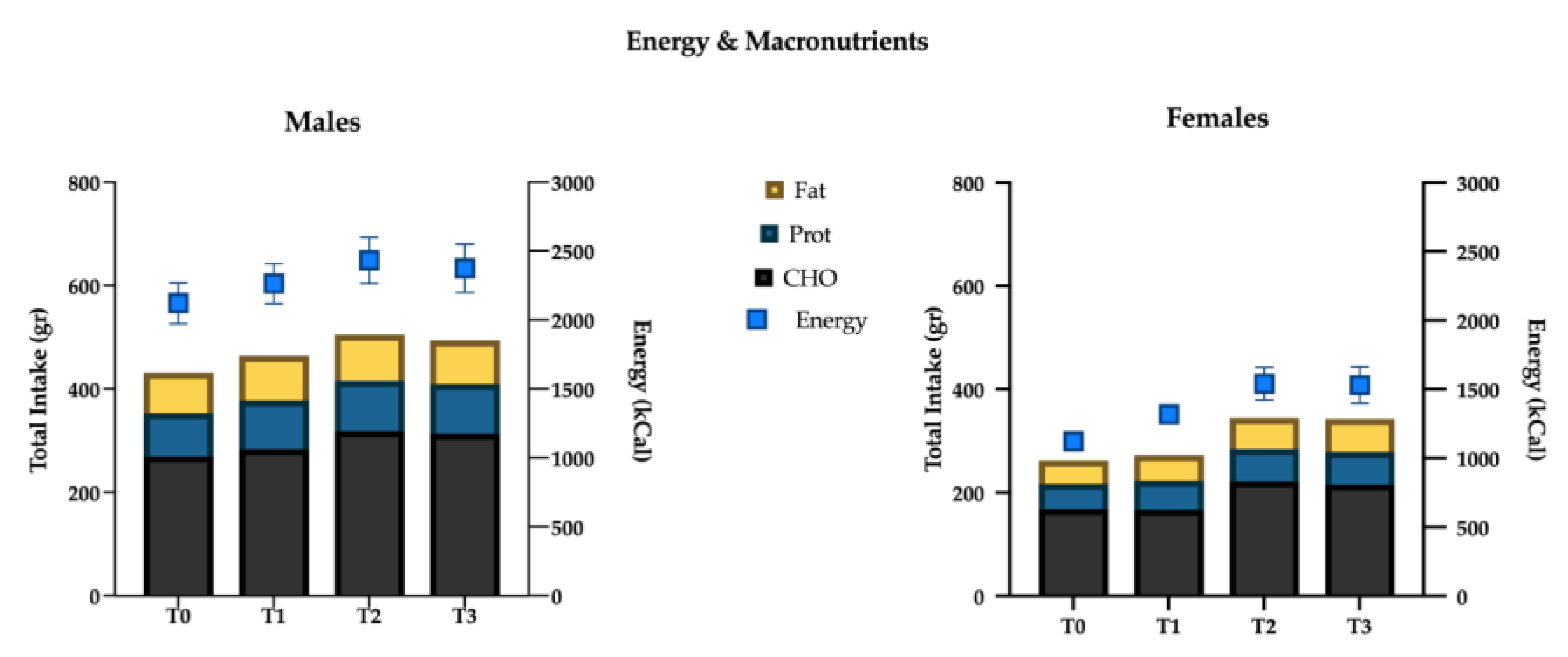
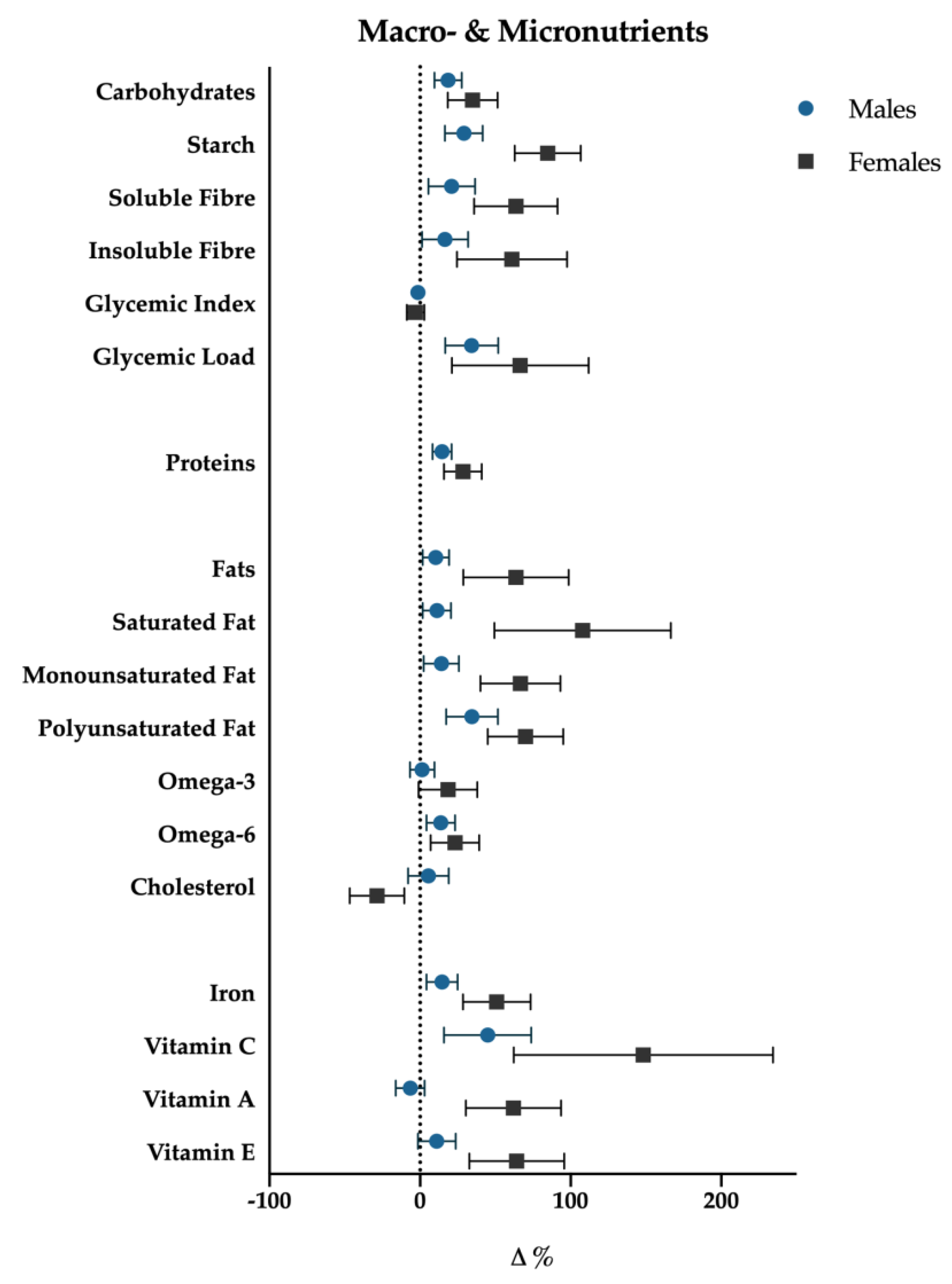
3.1. Weight, Body Composition, Macro- and Micronutrients
3.2. Factor Analysis
4. Discussion
4.1. Body Composition
4.2. Energy and Macronutrients Intake
4.3. Factor Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Koch, L.G.; Britton, S.L. Theoretical and Biological Evaluation of the Link between Low Exercise Capacity and Disease Risk. Cold Spring Harb. Perspect. Med. 2018, 8, a029868. [Google Scholar] [CrossRef]
- Bellisle, F. Food choice, appetite and physical activity. Public Health Nutr. 1999, 2, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, C.R.; Rakhmatullaeva, N.; Beckford, S.E.; Ammachathram, A.; Cristobal, A.; Koehler, K. Exercise and the Timing of Snack Choice: Healthy Snack Choice is Reduced in the Post-Exercise State. Nutrients 2018, 10, 1941. [Google Scholar] [CrossRef] [PubMed]
- Biddle, S.; Mutrie, N. Psychology of Physical Activity: Determinants, Well-being, and Interventions; Routledge: Abingdon, UK, 2008. [Google Scholar]
- Baker, C.W.; Brownell, K.D. Physical activity and maintenance of weight loss: Physiological and psychological mechanisms. In Physical Activity and Obesity; Bouchard, C., Ed.; Human Kinetics Europe: Leeds, UK, 2000. [Google Scholar]
- Andrade, A.M.; Coutinho, S.R.; Silva, M.N.; Mata, J.; Vieira, P.N.; Minderico, C.S.; Melanson, K.J.; Baptista, F.; Sardinha, L.B.; Teixeira, P.J. The effect of physical activity on weight loss is mediated by eating self-regulation. Patient Educ. Couns. 2010, 79, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Koehler, K.; Drenowatz, C. Integrated Role of Nutrition and Physical Activity for Lifelong Health. Nutrients 2019, 11, 1437. [Google Scholar] [CrossRef]
- Tucker, M.; Reicks, M. Exercise as a gateway behavior for healthful eating among older adults: An exploratory study. J. Nutr. Educ. Behav. 2002, 34, S14–S19. [Google Scholar] [CrossRef]
- Joseph, R.J.; Alonso-Alonso, M.; Bond, D.S.; Pascual-Leone, A.; Blackburn, G.L. The neurocognitive connection between physical activity and eating behaviour. Obes. Rev. 2011, 12, 800–812. [Google Scholar] [CrossRef]
- Joo, J.; Williamson, S.A.; Vazquez, A.I.; Fernandez, J.R.; Bray, M.S. The influence of 15-week exercise training on dietary patterns among young adults. Int. J. Obes. 2019, 43, 1681–1690. [Google Scholar] [CrossRef]
- Karlsen, T.; Aamot, I.L.; Haykowsky, M.; Rognmo, O. High Intensity Interval Training for Maximizing Health Outcomes. Prog. Cardiovasc. Dis. 2017, 60, 67–77. [Google Scholar] [CrossRef]
- Buchheit, M.; Laursen, P.B. High-intensity interval training, solutions to the programming puzzle: Part I: Cardiopulmonary emphasis. Sports Med. 2013, 43, 313–338. [Google Scholar] [CrossRef]
- Alves, E.D.; Salermo, G.P.; Panissa, V.L.G.; Franchini, E.; Takito, M.Y. Effects of long or short duration stimulus during high-intensity interval training on physical performance, energy intake, and body composition. J. Exerc. Rehabil. 2017, 13, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Malek, M.H.; Housh, T.J.; Berger, D.E.; Coburn, J.W.; Beck, T.W. A new non-exercise-based Vo2max prediction equation for aerobically trained men. J. Strength Cond. Res. 2005, 19, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Y.; Haskell, W.L.; Farrell, S.W.; Lamonte, M.J.; Blair, S.N.; Curtin, L.R.; Hughes, J.P.; Burt, V.L. Cardiorespiratory fitness levels among US adults 20–49 years of age: Findings from the 1999–2004 National Health and Nutrition Examination Survey. Am. J. Epidemiol. 2010, 171, 426–435. [Google Scholar] [CrossRef] [PubMed]
- American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription, 10th ed.; Riebe, D., Ehrman, J.K., Liguori, G., Magal, M., Eds.; Wolters Kluwer Health: Philadelphia, PA, USA, 2018. [Google Scholar]
- Gervasi, M.; Sisti, D.; Amatori, S.; Donati Zeppa, S.; Annibalini, G.; Piccoli, G.; Vallorani, L.; Benelli, P.; Rocchi, M.B.L.; Barbieri, E.; et al. Effects of a commercially available branched-chain amino acid-alanine-carbohydrate-based sports supplement on perceived exertion and performance in high intensity endurance cycling tests. J. Int. Soc. Sports Nutr. 2020, 17, 6. [Google Scholar] [CrossRef] [PubMed]
- Seiler, K.S.; Kjerland, G.O. Quantifying training intensity distribution in elite endurance athletes: Is there evidence for an “optimal” distribution? Scand. J. Med. Sci. Sports 2006, 16, 49–56. [Google Scholar] [CrossRef]
- Girden, E.R. ANOVA: Repeated Measures; Sage Publications, Inc.: Thousand Oaks, CA, USA, 1992; Volume 84. [Google Scholar]
- West, S.L.; Caterini, J.; Banks, L.; Wells, G.D. The Epidemic of Obesity and Poor Physical Activity Participation: Will We Ever See a Change? J. Funct. Morphol. Kinesiol. 2018, 3, 34. [Google Scholar] [CrossRef]
- Terranova, C.O.; Brakenridge, C.L.; Lawler, S.P.; Eakin, E.G.; Reeves, M.M. Effectiveness of lifestyle-based weight loss interventions for adults with type 2 diabetes: A systematic review and meta-analysis. Diabetes Obes. Metab. 2015, 17, 371–378. [Google Scholar] [CrossRef]
- Curioni, C.C.; Lourenco, P.M. Long-term weight loss after diet and exercise: A systematic review. Int. J. Obes. 2005, 29, 1168–1174. [Google Scholar] [CrossRef]
- Jayawardene, W.P.; Torabi, M.R.; Lohrmann, D.K. Exercise in Young Adulthood with Simultaneous and Future Changes in Fruit and Vegetable Intake. J. Am. Coll. Nutr. 2016, 35, 59–67. [Google Scholar] [CrossRef]
- Fisher, G.; Brown, A.W.; Bohan Brown, M.M.; Alcorn, A.; Noles, C.; Winwood, L.; Resuehr, H.; George, B.; Jeansonne, M.M.; Allison, D.B. High Intensity Interval— vs. Moderate Intensity—Training for Improving Cardiometabolic Health in Overweight or Obese Males: A Randomized Controlled Trial. PLoS ONE 2015, 10, e0138853. [Google Scholar] [CrossRef]
- Miguet, M.; Fearnbach, S.N.; Metz, L.; Khammassi, M.; Julian, V.; Cardenoux, C.; Pereira, B.; Boirie, Y.; Duclos, M.; Thivel, D. Effect of HIIT versus MICT on body composition and energy intake in dietary restrained and unrestrained adolescents with obesity. Appl. Physiol. Nutr. Metab. 2019. [Google Scholar] [CrossRef] [PubMed]
- Laursen, P.B.; Jenkins, D.G. The scientific basis for high-intensity interval training: Optimising training programmes and maximising performance in highly trained endurance athletes. Sports Med. 2002, 32, 53–73. [Google Scholar] [CrossRef]
- Jung, W.S.; Hwang, H.; Kim, J.; Park, H.Y.; Lim, K. Effect of interval exercise versus continuous exercise on excess post-exercise oxygen consumption during energy-homogenized exercise on a cycle ergometer. J. Exerc. Nutr. Biochem. 2019, 23, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Scheurink, A.J.; Ammar, A.A.; Benthem, B.; van Dijk, G.; Sodersten, P.A. Exercise and the regulation of energy intake. Int. J. Obes. Relat. Metab. Disord. 1999, 23, S1–S6. [Google Scholar] [CrossRef] [PubMed]
- Birkenhead, K.L.; Slater, G. A Review of Factors Influencing Athletes’ Food Choices. Sports Med. 2015, 45, 1511–1522. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Tanaka, K.; Yabushita, N.; Sakai, T.; Shigematsu, R. Effects of exercise frequency on functional fitness in older adult women. Arch. Gerontol. Geriatr. 2007, 44, 163–173. [Google Scholar] [CrossRef]
- Westcott, W.L.; Winett, R.A.; Annesi, J.J.; Wojcik, J.R.; Anderson, E.S.; Madden, P.J. Prescribing physical activity: Applying the ACSM protocols for exercise type, intensity, and duration across 3 training frequencies. Phys. Sportsmed. 2009, 37, 51–58. [Google Scholar] [CrossRef]
- Hazell, T.J.; Hamilton, C.D.; Olver, T.D.; Lemon, P.W. Running sprint interval training induces fat loss in women. Appl. Physiol. Nutr. Metab. 2014, 39, 944–950. [Google Scholar] [CrossRef]
- Trapp, E.G.; Chisholm, D.J.; Freund, J.; Boutcher, S.H. The effects of high-intensity intermittent exercise training on fat loss and fasting insulin levels of young women. Int. J. Obes. 2008, 32, 684–691. [Google Scholar] [CrossRef]
- Astorino, T.A.; Heath, B.; Bandong, J.; Ordille, G.M.; Contreras, R.; Montell, M.; Schubert, M.M. Effect of periodized high intensity interval training (HIIT) on body composition and attitudes towards hunger in active men and women. J. Sports Med. Phys. Fit. 2018, 58, 1052–1062. [Google Scholar] [CrossRef]
- Thivel, D.; Isacco, L.; Montaurier, C.; Boirie, Y.; Duche, P.; Morio, B. The 24-h energy intake of obese adolescents is spontaneously reduced after intensive exercise: A randomized controlled trial in calorimetric chambers. PLoS ONE 2012, 7, e29840. [Google Scholar] [CrossRef] [PubMed]
- Thivel, D.; Finlayson, G.; Blundell, J.E. Homeostatic and neurocognitive control of energy intake in response to exercise in pediatric obesity: A psychobiological framework. Obes. Rev. 2019, 20, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Poon, E.T.; Sun, F.H.; Chung, A.P.; Wong, S.H. Post-Exercise Appetite and Ad Libitum Energy Intake in Response to High-Intensity Interval Training versus Moderate— or Vigorous—Intensity Continuous Training among Physically Inactive Middle-Aged Adults. Nutrients 2018, 10, 1408. [Google Scholar] [CrossRef]
- Dorling, J.; Broom, D.R.; Burns, S.F.; Clayton, D.J.; Deighton, K.; James, L.J.; King, J.A.; Miyashita, M.; Thackray, A.E.; Batterham, R.L.; et al. Acute and Chronic Effects of Exercise on Appetite, Energy Intake, and Appetite-Related Hormones: The Modulating Effect of Adiposity, Sex, and Habitual Physical Activity. Nutrients 2018, 10, 1140. [Google Scholar] [CrossRef]
- Johnson, R.K. Dietary intake—How do we measure what people are really eating? Obes. Res. 2002, 10, 63S–68S. [Google Scholar] [CrossRef] [PubMed]
- Moshfegh, A.J.; Rhodes, D.G.; Baer, D.J.; Murayi, T.; Clemens, J.C.; Rumpler, W.V.; Paul, D.R.; Sebastian, R.S.; Kuczynski, K.J.; Ingwersen, L.A.; et al. The US Department of Agriculture Automated Multiple-Pass Method reduces bias in the collection of energy intakes. Am. J. Clin. Nutr. 2008, 88, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Olendzki, B.C.; Pagoto, S.L.; Hurley, T.G.; Magner, R.P.; Ockene, I.S.; Schneider, K.L.; Merriam, P.A.; Hebert, J.R. Number of 24-hour diet recalls needed to estimate energy intake. Ann. Epidemiol. 2009, 19, 553–559. [Google Scholar] [CrossRef]
- Subar, A.F.; Kipnis, V.; Troiano, R.P.; Midthune, D.; Schoeller, D.A.; Bingham, S.; Sharbaugh, C.O.; Trabulsi, J.; Runswick, S.; Ballard-Barbash, R.; et al. Using intake biomarkers to evaluate the extent of dietary misreporting in a large sample of adults: The OPEN study. Am. J. Epidemiol. 2003, 158, 1–13. [Google Scholar] [CrossRef]
- Hiza, H.A.; Casavale, K.O.; Guenther, P.M.; Davis, C.A. Diet quality of Americans differs by age, sex, race/ethnicity, income, and education level. J. Acad. Nutr. Diet. 2013, 113, 297–306. [Google Scholar] [CrossRef]
- Johnson, R.; Robertson, W.; Towey, M.; Stewart-Brown, S.; Clarke, A. Changes over time in mental well-being, fruit and vegetable consumption and physical activity in a community-based lifestyle intervention: A before and after study. Public Health 2017, 146, 118–125. [Google Scholar] [CrossRef]
- Braakhuis, A.J. Effect of vitamin C supplements on physical performance. Curr. Sports Med. Rep. 2012, 11, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Pedlar, C.R.; Brugnara, C.; Bruinvels, G.; Burden, R. Iron balance and iron supplementation for the female athlete: A practical approach. Eur. J. Sport Sci. 2018, 18, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Hankinson, A.L.; Daviglus, M.L.; Bouchard, C.; Carnethon, M.; Lewis, C.E.; Schreiner, P.J.; Liu, K.; Sidney, S. Maintaining a high physical activity level over 20 years and weight gain. JAMA 2010, 304, 2603–2610. [Google Scholar] [CrossRef] [PubMed]
- Leow, S.; Jackson, B.; Alderson, J.A.; Guelfi, K.J.; Dimmock, J.A. A Role for Exercise in Attenuating Unhealthy Food Consumption in Response to Stress. Nutrients 2018, 10, 176. [Google Scholar] [CrossRef] [PubMed]
- Cena, H.; Calder, P.C. Defining a Healthy Diet: Evidence for The Role of Contemporary Dietary Patterns in Health and Disease. Nutrients 2020, 12, 334. [Google Scholar] [CrossRef]
- Alamgir, A.N.M. Therapeutic Use of Medicinal Plants and Their Extracts: Volume 2; Springer International Publishing: Berlin/Heidelberg, Germany, 2018; Progress in Drug Research 74. [Google Scholar] [CrossRef]
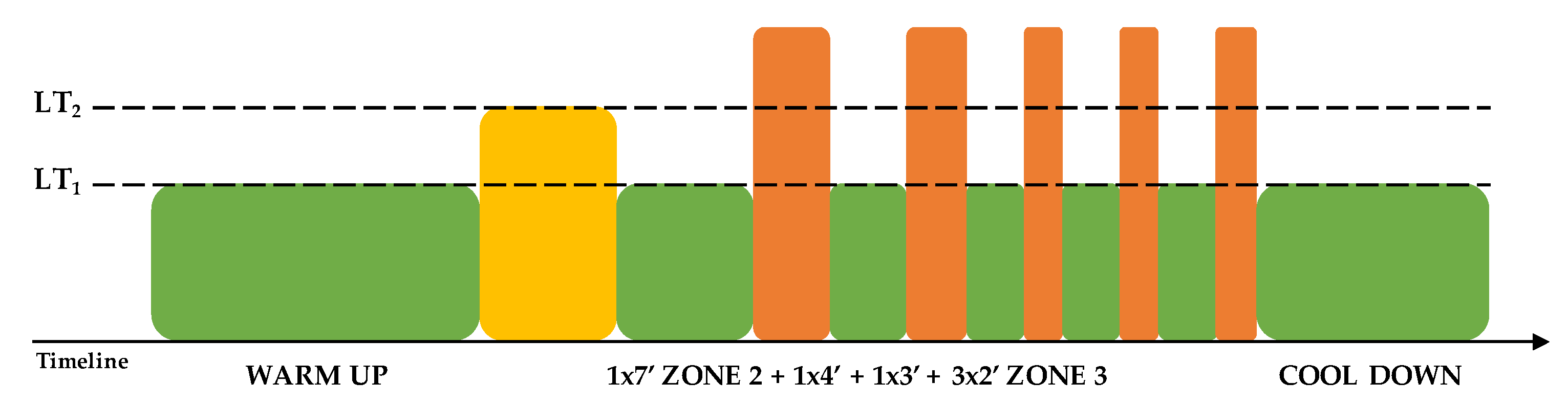
| T0 | T1 | T2 | T3 | |||||
|---|---|---|---|---|---|---|---|---|
| Males | Females | Males | Females | Males | Females | Males | Females | |
| Weight (kg) | 68.2 ± 10.0 | 52.9 ± 5.4 | 67.8 ± 9.3 | 53.0 ± 5.3 | 67.9 ± 8.6 | 53.0 ± 5.6 | 67.4 ± 7.9 | 53.0 ± 5.5 |
| Free-Fat Mass (%) | 83.7 ± 5.9 | 75.0 ± 2.9 | 83.7 ± 5.9 | 72.9 ± 3.4 | 83.3 ± 4.6 | 75.7 ± 5.0 | 87.3 ± 4.3 | 77.2 ± 3.8 |
| Energy (kcal) | 2122 ± 663 | 1121 ± 189 | 2266 ± 646 | 1317 ± 238 | 2432 ± 745 | 1541 ± 412 | 2375 ± 777 | 1531 ± 460 |
| Carbohydrate (g) | 270.2 ± 58.3 | 168.3 ± 33.6 | 284.2 ± 75.4 | 167.3 ± 23.9 | 318.0 ± 106.1 | 222.0 ± 97.5 | 313.6 ± 99.8 | 216.2 ± 85.7 |
| Protein (g) | 83.4 ± 16.8 | 49.2 ± 6.3 | 94.2 ± 26.6 | 55.5 ± 10.7 | 98.3 ± 29.5 | 62.3 ± 21.7 | 96.3 ± 29.5 | 62.5 ± 21.7 |
| Fat (g) | 77.9 ± 23.8 | 44.5 ± 14.9 | 86.0 ± 30.6 | 49.7 ± 14.3 | 88.7 ± 31.5 | 59.8 ± 21.6 | 84.4 ± 35.1 | 63.4 ± 31.4 |
| Starch (g) | 129.9 ± 44.2 | 48.4 ± 15.5 | 132.8 ± 49.0 | 57.3 ± 17.1 | 148.0 ± 58.1 | 77.6 ± 27.6 | 158.0 ± 61.1 | 81.7 ± 26.2 |
| Soluble Fibre (g) | 2.6 ± 1.2 | 1.0 ± 0.4 | 2.6 ± 1.2 | 1.1 ± 0.4 | 2.8 ± 1.4 | 1.1 ± 0.4 | 3.0 ± 1.6 | 1.5 ± 0.6 |
| Insoluble Fibre (g) | 7.5 ± 3.4 | 3.2 ± 1.4 | 6.8 ± 3.3 | 3.5 ± 1.3 | 7.6 ± 4.3 | 3.6 ± 1.5 | 8.5 ± 6.6 | 4.8 ± 2.9 |
| Glycaemic Index | 55.3 ± 5.1 | 58.9 ± 7.3 | 54.5 ± 3.8 | 55.5 ± 4.3 | 55.9 ± 3.7 | 56.7 ± 5.7 | 54.4 ± 3.4 | 56.5 ± 2.6 |
| Glycaemic Load | 89.9 ± 27.4 | 47.7 ± 24.8 | 94.1 ± 24.1 | 48.8 ± 12.5 | 110.1 ± 33.0 | 54.2 ± 14.8 | 113.1 ± 34.8 | 59.9 ± 16.7 |
| Saturated Fats (g) | 23.3 ± 10.9 | 11.7 ± 4.1 | 25.1 ± 9.6 | 14.9 ± 4.9 | 25.9 ± 9.4 | 18.4 ± 7.2 | 24.3 ± 9.7 | 20.8 ± 13.4 |
| Monounsaturated Fats (g) | 27.5 ± 12.1 | 10.6 ± 6.4 | 28.5 ± 12.3 | 14.1 ± 7.4 | 29.3 ± 11.2 | 16.5 ± 8.7 | 28.8 ± 13.3 | 16.7 ± 11.9 |
| Polyunsaturated Fats (g) | 8.4 ± 3.4 | 3.8 ± 1.1 | 9.0 ± 3.1 | 4.6 ± 1.6 | 9.6 ± 2.8 | 5.8 ± 2.8 | 10.0 ± 4.3 | 6.2 ± 3.2 |
| Omega-3 (% kcal/kcal tot) | 0.4 ± 0.2 | 0.5 ± 0.2 | 0.4 ± 0.1 | 0.5 ± 0.2 | 0.4 ± 0.1 | 0.5 ± 0.2 | 0.4 ± 0.1 | 0.5 ± 0.4 |
| Omega-6 (% kcal/kcal tot) | 3.0 ± 1.1 | 2.5 ± 1.0 | 3.0 ± 0.6 | 2.7 ± 0.8 | 3.1 ± 0.4 | 2.7 ± 1.2 | 3.1 ± 0.6 | 2.8 ± 1.0 |
| Cholesterol (mg) | 238.9 ± 73.8 | 116.4 ± 58.5 | 217.1 ± 73.5 | 95.0 ± 22.9 | 265.1 ± 65.1 | 88.6 ± 60.3 | 236.5 ± 66.9 | 86.6 ± 60.5 |
| Iron (mg) | 9.0 ± 2.9 | 4.2 ± 1.0 | 8.8 ± 2.6 | 4.9 ± 1.7 | 9.4 ± 2.9 | 5.8 ± 2.8 | 9.8 ± 3.9 | 6.3 ± 3.8 |
| Vitamin C (mg) | 68.1 ± 30.4 | 39.0 ± 21.8 | 60.3 ± 22.1 | 37.5 ± 18.4 | 66.2 ± 26.6 | 45.5 ± 22.0 | 80.7 ± 49.8 | 59.3 ± 35.4 |
| Vitamin A (mcg) | 1081.0 ± 537.9 | 512.7 ± 604.1 | 982.2 ± 433.4 | 590.7 ± 469.6 | 980.4 ± 458.1 | 666.5 ± 623.9 | 910.3 ± 507.6 | 540.6 ± 328.9 |
| Vitamin E (mg) | 8.4 ± 3.8 | 3.6 ± 2.5 | 8.3 ± 3.8 | 4.5 ± 2.7 | 8.8 ± 4.4 | 5.2 ± 2.8 | 8.7 ± 4.7 | 5.2 ± 3.9 |
| Component | Factor 1 | Factor 2 | Factor 3 |
|---|---|---|---|
| Δ Fat | 0.865 | 0.278 | 0.043 |
| Δ Protein | 0.821 | 0.088 | 0.395 |
| Δ Carbohydrate | 0.731 | −0.030 | 0.466 |
| Δ Energy | 0.713 | 0.037 | 0.517 |
| Δ Monounsaturated fat | 0.694 | 0.326 | 0.206 |
| Δ Saturated fat | 0.647 | 0.003 | 0.209 |
| Δ Vitamin E | 0.629 | 0.439 | 0.392 |
| Δ Omega 6 | 0.140 | 0.848 | 0.206 |
| Δ Omega 3 | 0.016 | 0.714 | 0.061 |
| Δ Polyunsaturated fat | 0.472 | 0.697 | 0.444 |
| Δ Insoluble fibre | 0.282 | 0.281 | 0.857 |
| Δ Soluble fibre | 0.135 | 0.286 | 0.836 |
| Δ Vitamin C | 0.275 | 0.043 | 0.783 |
| Δ Vitamin A | 0.268 | 0.161 | 0.644 |
| Δ Starch | 0.551 | 0.118 | 0.583 |
| Δ Iron | 0.515 | 0.168 | 0.573 |
| Eigenvalues | 8.62 | 1.55 | 1.29 |
| % of total variance | 30.10 | 44.68 | 71.61 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donati Zeppa, S.; Sisti, D.; Amatori, S.; Gervasi, M.; Agostini, D.; Piccoli, G.; Bertuccioli, A.; Rocchi, M.B.L.; Stocchi, V.; Sestili, P. High-intensity Interval Training Promotes the Shift to a Health-Supporting Dietary Pattern in Young Adults. Nutrients 2020, 12, 843. https://doi.org/10.3390/nu12030843
Donati Zeppa S, Sisti D, Amatori S, Gervasi M, Agostini D, Piccoli G, Bertuccioli A, Rocchi MBL, Stocchi V, Sestili P. High-intensity Interval Training Promotes the Shift to a Health-Supporting Dietary Pattern in Young Adults. Nutrients. 2020; 12(3):843. https://doi.org/10.3390/nu12030843
Chicago/Turabian StyleDonati Zeppa, Sabrina, Davide Sisti, Stefano Amatori, Marco Gervasi, Deborah Agostini, Giovanni Piccoli, Alexander Bertuccioli, Marco B.L. Rocchi, Vilberto Stocchi, and Piero Sestili. 2020. "High-intensity Interval Training Promotes the Shift to a Health-Supporting Dietary Pattern in Young Adults" Nutrients 12, no. 3: 843. https://doi.org/10.3390/nu12030843
APA StyleDonati Zeppa, S., Sisti, D., Amatori, S., Gervasi, M., Agostini, D., Piccoli, G., Bertuccioli, A., Rocchi, M. B. L., Stocchi, V., & Sestili, P. (2020). High-intensity Interval Training Promotes the Shift to a Health-Supporting Dietary Pattern in Young Adults. Nutrients, 12(3), 843. https://doi.org/10.3390/nu12030843









