ADORA2A C Allele Carriers Exhibit Ergogenic Responses to Caffeine Supplementation
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Participants
2.3. Exercise Protocol
2.4. Assessment of Blinding
2.5. Genetic Testing
2.6. Statistical Analysis
3. Results
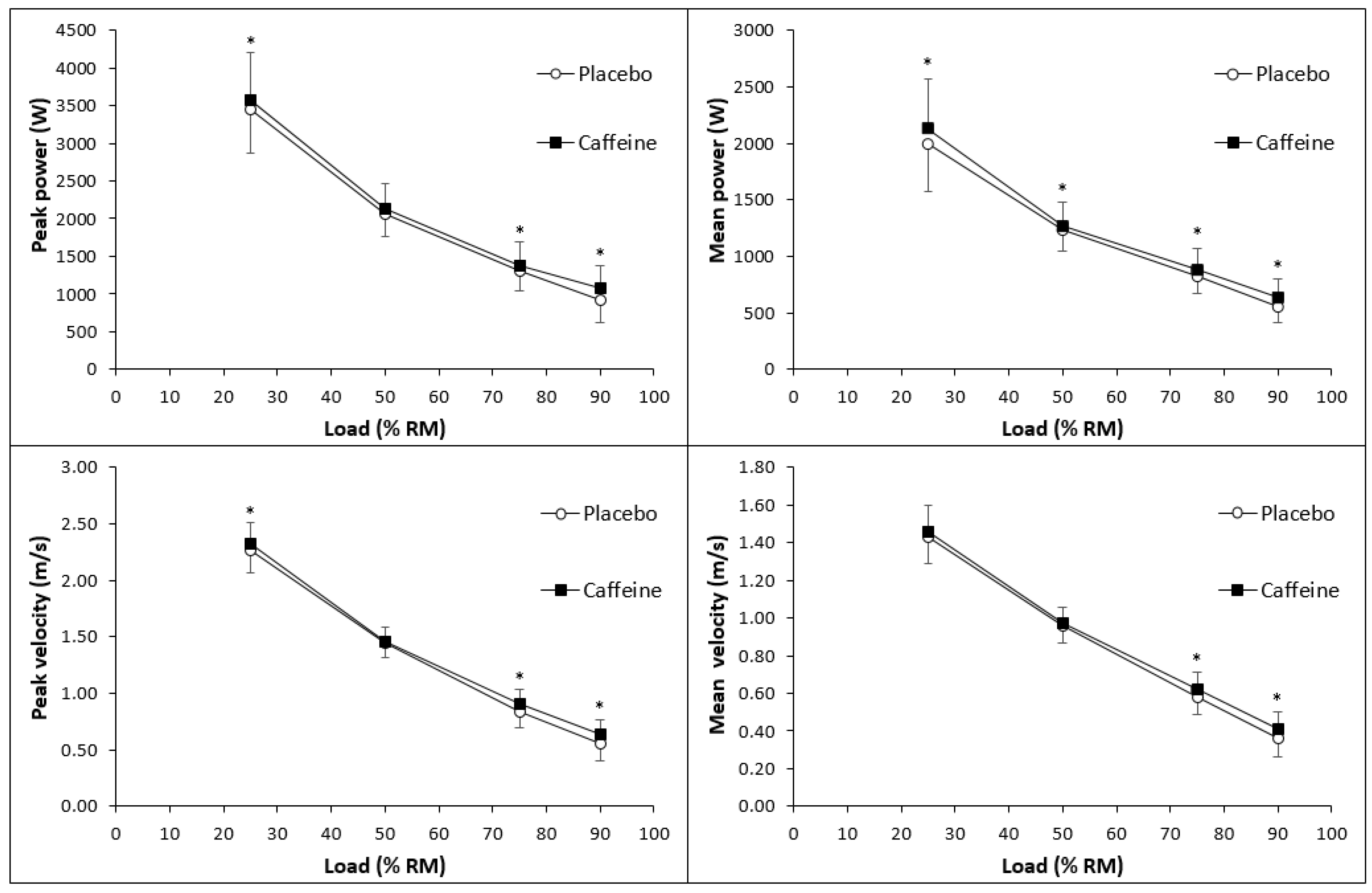
3.1. Exercise Performance
3.2. Assessment of Blinding
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Graham, T.E. Caffeine and exercise: Metabolism, endurance and performance. Sports. Med. 2001, 31, 785–807. [Google Scholar] [CrossRef] [PubMed]
- McLellan, T.M.; Caldwell, J.A.; Lieberman, H.R. A review of caffeine’s effects on cognitive, physical and occupational performance. Neurosci. Biobehav. Rev. 2016, 71, 294–312. [Google Scholar] [CrossRef] [PubMed]
- Grgic, J.; Grgic, I.; Pickering, C.; Schoenfeld, B.J.; Bishop, D.J.; Pedisic, Z. Wake up and smell the coffee: Caffeine supplementation and exercise performance-an umbrella review of 21 published meta-analyses. Br. J. Sports. Med. 2019. [Google Scholar] [CrossRef]
- Salinero, J.J.; Lara, B.; Del Coso, J. Effects of acute ingestion of caffeine on team sports performance: A systematic review and meta-analysis. Res. Sports Med. 2019, 27, 238–256. [Google Scholar] [CrossRef]
- Grgic, J.; Trexler, E.T.; Lazinica, B.; Pedisic, Z. Effects of caffeine intake on muscle strength and power: A systematic review and meta-analysis. J. Int. Soc. Sports Nutr. 2018, 15, 11. [Google Scholar] [CrossRef]
- Grgic, J.; Pickering, C. The effects of caffeine ingestion on isokinetic muscular strength: A meta-analysis. J. Sci. Med. Sport 2019, 22, 353–360. [Google Scholar] [CrossRef]
- Grgic, J. Caffeine ingestion enhances Wingate performance: A meta-analysis. Eur. J. Sport. Sci. 2018, 18, 219–225. [Google Scholar] [CrossRef]
- Grgic, J.; Mikulic, P.; Schoenfeld, B.J.; Bishop, D.J.; Pedisic, Z. The Influence of Caffeine Supplementation on Resistance Exercise: A Review. Sports Med. 2019, 49, 17–30. [Google Scholar] [CrossRef]
- Grgic, J. Are There Non-Responders to the Ergogenic Effects of Caffeine Ingestion on Exercise Performance? Nutrients 2018, 10, 1736. [Google Scholar] [CrossRef]
- Pickering, C.; Kiely, J. Are the Current Guidelines on Caffeine Use in Sport Optimal for Everyone? Inter-individual Variation in Caffeine Ergogenicity, and a Move Towards Personalised Sports Nutrition. Sports Med. 2018, 48, 7–16. [Google Scholar] [CrossRef]
- Fredholm, B.B.; Yang, J.; Wang, Y. Low, but not high, dose caffeine is a readily available probe for adenosine actions. Mol. Asp. Med. 2017, 55, 20–25. [Google Scholar] [CrossRef]
- Davis, J.M.; Zhao, Z.; Stock, H.S.; Mehl, K.A.; Buggy, J.; Hand, G.A. Central nervous system effects of caffeine and adenosine on fatigue. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2003, 284, R399–R404. [Google Scholar] [CrossRef]
- Djordjevic, N.; Ghotbi, R.; Jankovic, S.; Aklillu, E. Induction of CYP1A2 by heavy coffee consumption is associated with the CYP1A2 -163C>A polymorphism. Eur. J. Clin. Pharmacol. 2010, 66, 697–703. [Google Scholar] [CrossRef]
- Algrain, H.A.; Thomas, R.M.; Carrillo, A.E.; Ryan, E.J.; Kim, C.H.; Lettan, R.B.; Ryan, E.J. The effects of a polymorphism in the cytochrome P450 CYP1A2 gene on performance enhancement with caffeine in recreational cyclists. J. Caffeine Res. 2016, 6, 34–39. [Google Scholar] [CrossRef]
- Giersch, G.E.; Boyett, J.C.; Hargens, T.A.; Luden, N.D.; Saunders, M.J.; Daley, H.; Hughey, C.A.; El-Sohemy, A.; Womack, C.J. The effect of the CYP1A2-163 C>A polymorphism on caffeine metabolism and subsequent cycling performance. J. Caffeine Adenosine Res. 2018, 8, 65–70. [Google Scholar] [CrossRef]
- Guest, N.; Corey, P.; Vescovi, J.; El-Sohemy, A. Caffeine, CYP1A2 genotype, and endurance performance in athletes. Med. Sci. Sports. Exerc. 2018, 50, 1570–1578. [Google Scholar] [CrossRef]
- Klein, C.S.; Clawson, A.; Martin, M.; Saunders, M.J.; Flohr, J.A.; Bechtel, M.K.; Dunham, W.; Hancock, M.; Womack, C.J. The effect of caffeine on performance in collegiate tennis players. J. Caffeine Res. 2012, 2, 111–116. [Google Scholar] [CrossRef]
- Pataky, M.W.; Womack, C.J.; Saunders, M.J.; Goffe, J.L.; D’lugos, A.C.; El-Sohemy, A.; Lunden, N.D. Caffeine and 3-km cycling performance: Effects of mouth rinsing, genotype, and time of day. Scand. J. Med. Sci. Sports 2016, 26, 613–619. [Google Scholar] [CrossRef]
- Puente, C.; Abián-Vicén, J.; Del Coso, J.; Lara, B.; Salinero, J.J. The CYP1A2-163C > A polymorphism does not alter the effects of caffeine on basketball performance. PLoS ONE 2018, 13, e0195943. [Google Scholar] [CrossRef]
- Salinero, J.J.; Lara, B.; Ruiz-Vicente, D.; Areces, F.; Puente-Torres, C.; Gallo-Salazar, C.; Pascual, T.; Del Coso, J. CYP1A2 genotype variations do not modify the benefits and drawbacks of caffeine during exercise: A pilot study. Nutrients 2017, 9, 269. [Google Scholar] [CrossRef]
- Womack, C.J.; Saunders, M.J.; Bechtel, M.K.; Bolton, D.J.; Martin, M.; Luden, N.D.; Hancock, M. The influence of a CYP1A2 polymorphism on the ergogenic effects of caffeine. J. Int. Soc. Sports Nutr. 2012, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, R. The effect of CYP1A2 genotype on the ergogenic properties of caffeine during resistance exercise: A randomized, double-blind, placebo-controlled, crossover study. Ir. J. Med. Sci. 2019, 188, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Pickering, C.; Grgic, J. Caffeine and Exercise: What Next? Sports Med. 2019, 49, 1007–1030. [Google Scholar] [CrossRef] [PubMed]
- Cornelis, M.C.; El-Sohemy, A.; Campos, H. Genetic polymorphism of the adenosine A2A receptor is associated with habitual caffeine consumption. Am. J. Clin. Nutr. 2007, 86, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Loy, B.D.; O’Connor, P.J.; Lindheimer, J.B.; Covert, S.F. Caffeine is ergogenic for adenosine A2A receptor gene (ADORA2A) T allele homozygotes: A pilot study. J. Caffeine Res. 2015, 5, 73–81. [Google Scholar] [CrossRef]
- Grgic, J.; Mikulic, P. Caffeine ingestion acutely enhances muscular strength and power but not muscular endurance in resistance-trained men. Eur. J. Sport Sci. 2017, 17, 1029–1036. [Google Scholar] [CrossRef]
- Grgic, J.; Sabol, F.; Venier, S.; Mikulic, I.; Bratkovic, N.; Schoenfeld, B.J.; Pickering, C.; Bishop, D.J.; Pedisic, Z.; Mikulic, P. What Dose of Caffeine to Use: Acute Effects of 3 Doses of Caffeine on Muscle Endurance and Strength. Int. J. Sports Physiol. Perform. 2019. [Google Scholar] [CrossRef]
- Pallarés, J.G.; Fernández-Elías, V.E.; Ortega, J.F.; Muñoz, G.; Muñoz-Guerra, J.; Mora-Rodríguez, R. Neuromuscular responses to incremental caffeine doses: Performance and side effects. Med. Sci. Sports Exerc. 2013, 45, 2184–2192. [Google Scholar] [CrossRef]
- Venier, S.; Grgic, J.; Mikulic, P. Acute Enhancement of Jump Performance, Muscle Strength, and Power in Resistance-Trained Men After Consumption of Caffeinated Chewing Gum. Int. J. Sports. Physiol. Perform. 2020, 14, 1415–1421. [Google Scholar] [CrossRef]
- Venier, S.; Grgic, J.; Mikulic, P. Caffeinated Gel Ingestion Enhances Jump Performance, Muscle Strength, and Power in Trained Men. Nutrients 2019, 11, 937. [Google Scholar] [CrossRef]
- Frikha, M.; Chaâri, N.; Mezghanni, N.; Souissi, N. Influence of warm-up duration and recovery interval prior to exercise on anaerobic performance. Biol. Sport 2016, 33, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Saunders, B.; de Oliveira, L.F.; da Silva, R.P.; de Salles Painelli, V.; Gonçalves, L.S.; Yamaguchi, G.; Mutti, T.; Maciel, E.; Roschel, H.; Artioli, G.G.; et al. Placebo in sports nutrition: A proof-of-principle study involving caffeine supplementation. Scand. J. Med. Sci. Sports 2017, 27, 1240–1247. [Google Scholar] [CrossRef] [PubMed]
- Pickering, C.; Kiely, J.; Suraci, B.; Collins, D. The magnitude of Yo-Yo test improvements following an aerobic training intervention are associated with total genotype score. PLoS ONE 2018, 13, e0207597. [Google Scholar] [CrossRef]
- Skinner, T.L.; Desbrow, B.; Arapova, J.; Schaumberg, M.A.; Osborne, J.; Grant, G.D.; Anoopkumar-Dukie, S.; Leveritt, M.D. Women Experience the Same Ergogenic Response to Caffeine as Men. Med. Sci. Sports Exerc. 2019, 51, 1195–1202. [Google Scholar] [CrossRef]
- Sabblah, S.; Dixon, D.; Bottoms, L. Sex differences on the acute effects of caffeine on maximal strength and muscular endurance. Comp. Exerc. Physiol. 2015, 11, 89–94. [Google Scholar] [CrossRef]
- Mielgo-Ayuso, J.; Marques-Jiménez, D.; Refoyo, I.; Del Coso, J.; León-Guereño, P.; Calleja-González, J. Effect of Caffeine Supplementation on Sports Performance Based on Differences Between Sexes: A Systematic Review. Nutrients 2019, 11, 2313. [Google Scholar] [CrossRef]
- Raya-González, J.; Rendo-Urteaga, T.; Domínguez, R.; Castillo, D.; Rodríguez-Fernández, A.; Grgic, J. Acute Effects of Caffeine Supplementation on Movement Velocity in Resistance Exercise: A Systematic Review and Meta-analysis. Sports Med. 2019. [Google Scholar] [CrossRef]
- Burke, L.M. Caffeine and sports performance. Appl. Physiol. Nutr. Metab. 2008, 33, 1319–1334. [Google Scholar] [CrossRef]
- Erblang, M.; Drogou, C.; Gomez-Merino, D.; Metlaine, A.; Boland, A.; Deleuze, J.F.; Thomas, C.; Sauvet, F.; Chennaoui, M. The Impact of Genetic Variations in ADORA2A in the Association between Caffeine Consumption and Sleep. Genes 2019, 10, 1021. [Google Scholar] [CrossRef]
- Roberts, S.P.; Stokes, K.A.; Trewartha, G.; Doyle, J.; Hogben, P.; Thompson, D. Effects of carbohydrate and caffeine ingestion on performance during a rugby union simulation protocol. J. Sports Sci. 2010, 28, 833–842. [Google Scholar] [CrossRef]
- Yeo, S.E.; Jentjens, R.L.; Wallis, G.A.; Jeukendrup, A.E. Caffeine increases exogenous carbohydrate oxidation during exercise. J. Appl. Physiol. 2005, 99, 844–850. [Google Scholar] [CrossRef]
- Aird, T.P.; Davies, R.W.; Carson, B.P. Effects of fasted vs fed-state exercise on performance and post-exercise metabolism: A systematic review and meta-analysis. Scand. J. Med. Sci. Sports 2018, 28, 1476–1493. [Google Scholar] [CrossRef]
| Variable | Mean ± Standard Deviation |
|---|---|
| Age (years) | 29.3 ± 4.8 |
| Body mass (kg) | 80.3 ± 11.2 |
| Height (cm) | 183.1 ± 5.9 |
| 1RM in the bench press (normalized per body mass) | 1.1 ± 0.2 |
| Habitual caffeine intake (mg/day) | 143 ± 113 |
| Variable | Placebo | Caffeine | Hedges’ g and 95% CI | p-Value |
|---|---|---|---|---|
| Muscular endurance test | ||||
| Maximum repetitions at 85% 1RM | 6.9 ± 2.2 | 8.2 ± 2.1 | 0.58 (0.29, 0.91) | <0.001 |
| Mean power matched for repetitions (W) | 418 ± 116 | 492 ± 138 | 0.56 (0.32, 0.83) | <0.001 |
| Mean velocity matched for repetitions (m/s) | 0.27 ± 0.05 | 0.32 ± 0.05 | 0.96 (0.58, 1.41) | <0.001 |
| Peak power matched for repetitions (W) | 669 ± 250 | 740 ± 258 | 0.27 (0.14, 0.42) | <0.001 |
| Peak velocity matched for repetitions (m/s) | 0.41 ± 0.08 | 0.46 ± 0.07 | 0.64 (0.38, 0.94) | <0.001 |
| CMJ | ||||
| Vertical jump height (cm) | 35.0 ± 6.1 | 35.8 ± 5.9 | 0.13 (0.02, 0.25) | 0.034 |
| Wingate test | ||||
| Peak power in the Wingate test (W) | 859 ± 237 | 948 ± 229 | 0.37 (0.21, 0.55) | <0.001 |
| Mean power in the Wingate test (W) | 598 ± 101 | 634 ± 100 | 0.34 (0.17, 0.54) | <0.001 |
| Minimum power in the Wingate test (W) | 349 ± 103 | 392 ± 96 | 0.41 (0.07, 0.78) | 0.020 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grgic, J.; Pickering, C.; Bishop, D.J.; Del Coso, J.; Schoenfeld, B.J.; Tinsley, G.M.; Pedisic, Z. ADORA2A C Allele Carriers Exhibit Ergogenic Responses to Caffeine Supplementation. Nutrients 2020, 12, 741. https://doi.org/10.3390/nu12030741
Grgic J, Pickering C, Bishop DJ, Del Coso J, Schoenfeld BJ, Tinsley GM, Pedisic Z. ADORA2A C Allele Carriers Exhibit Ergogenic Responses to Caffeine Supplementation. Nutrients. 2020; 12(3):741. https://doi.org/10.3390/nu12030741
Chicago/Turabian StyleGrgic, Jozo, Craig Pickering, David J. Bishop, Juan Del Coso, Brad J. Schoenfeld, Grant M. Tinsley, and Zeljko Pedisic. 2020. "ADORA2A C Allele Carriers Exhibit Ergogenic Responses to Caffeine Supplementation" Nutrients 12, no. 3: 741. https://doi.org/10.3390/nu12030741
APA StyleGrgic, J., Pickering, C., Bishop, D. J., Del Coso, J., Schoenfeld, B. J., Tinsley, G. M., & Pedisic, Z. (2020). ADORA2A C Allele Carriers Exhibit Ergogenic Responses to Caffeine Supplementation. Nutrients, 12(3), 741. https://doi.org/10.3390/nu12030741







