Erythrocyte Ascorbate Is a Potential Indicator of Steady-State Plasma Ascorbate Concentrations in Healthy Non-Fasting Individuals
Abstract
1. Introduction
2. Materials and Methods
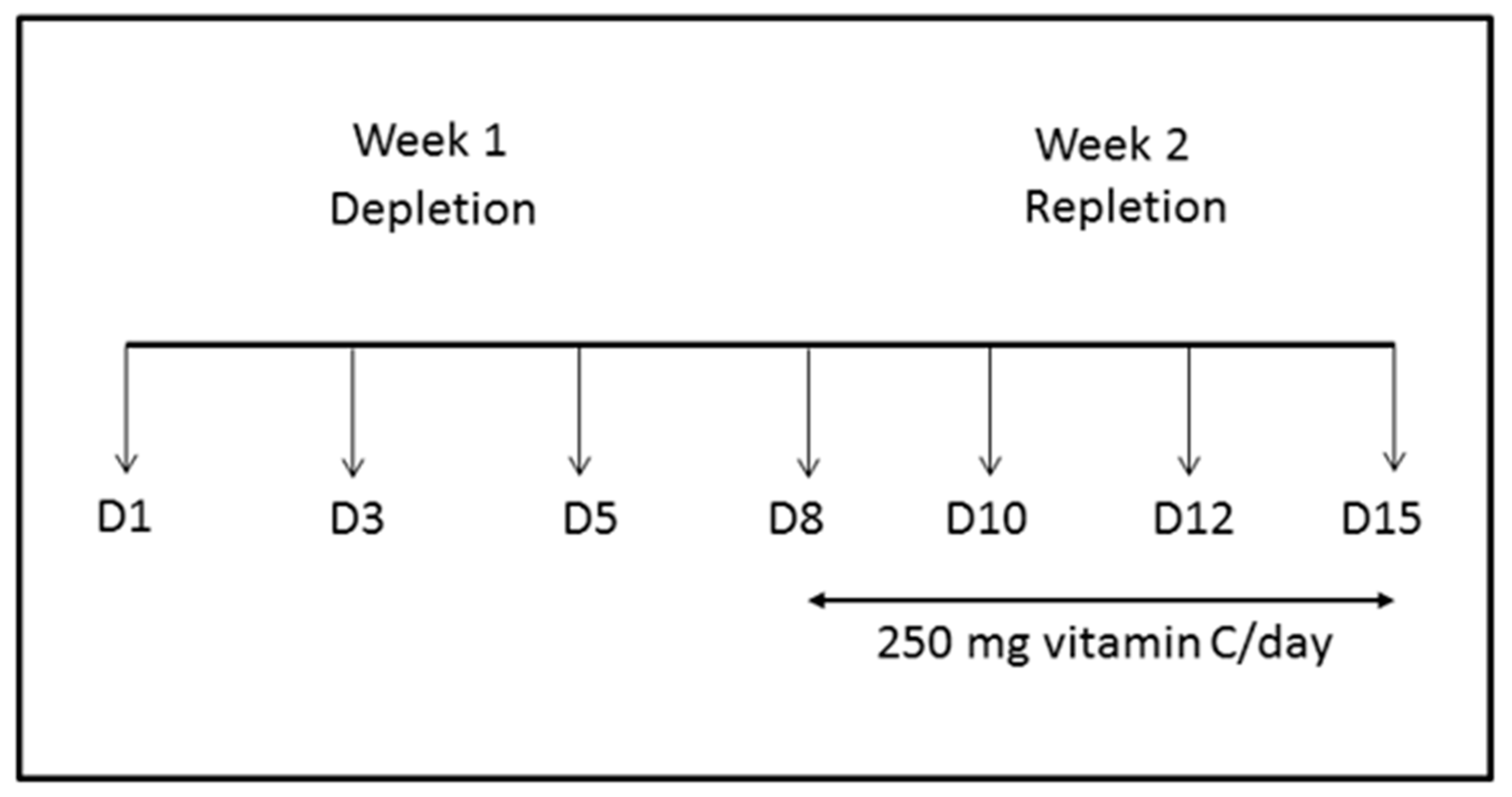
2.1. Steady-State Study
2.2. Pharmacokinetic Study
2.3. Plasma Ascorbate Sample Processing
2.4. Erythrocyte Ascorbate Sample Processing
2.5. Ascorbate HPLC Analysis
2.6. Statistical Analyses
3. Results
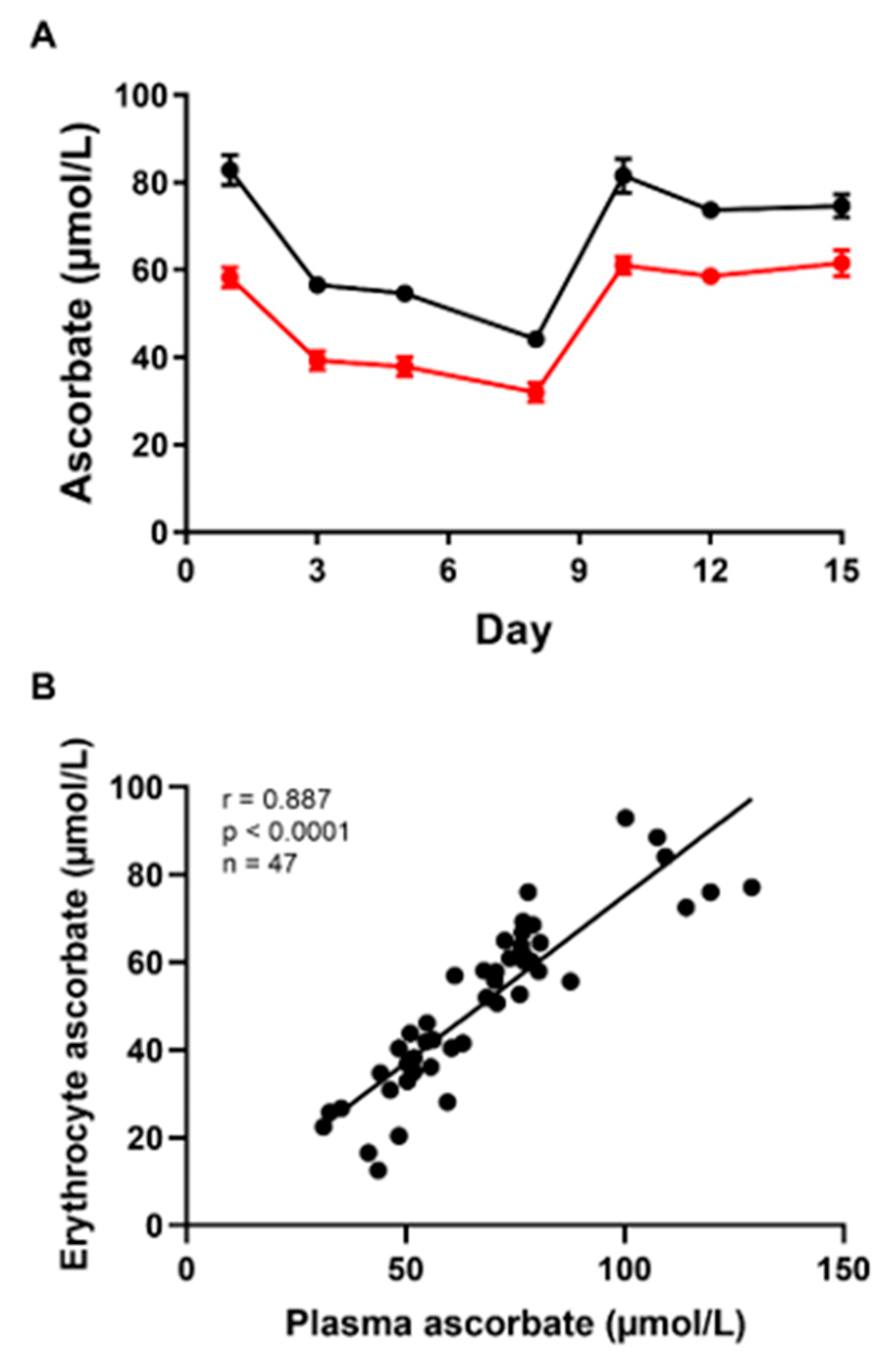
3.1. Steady-State Study
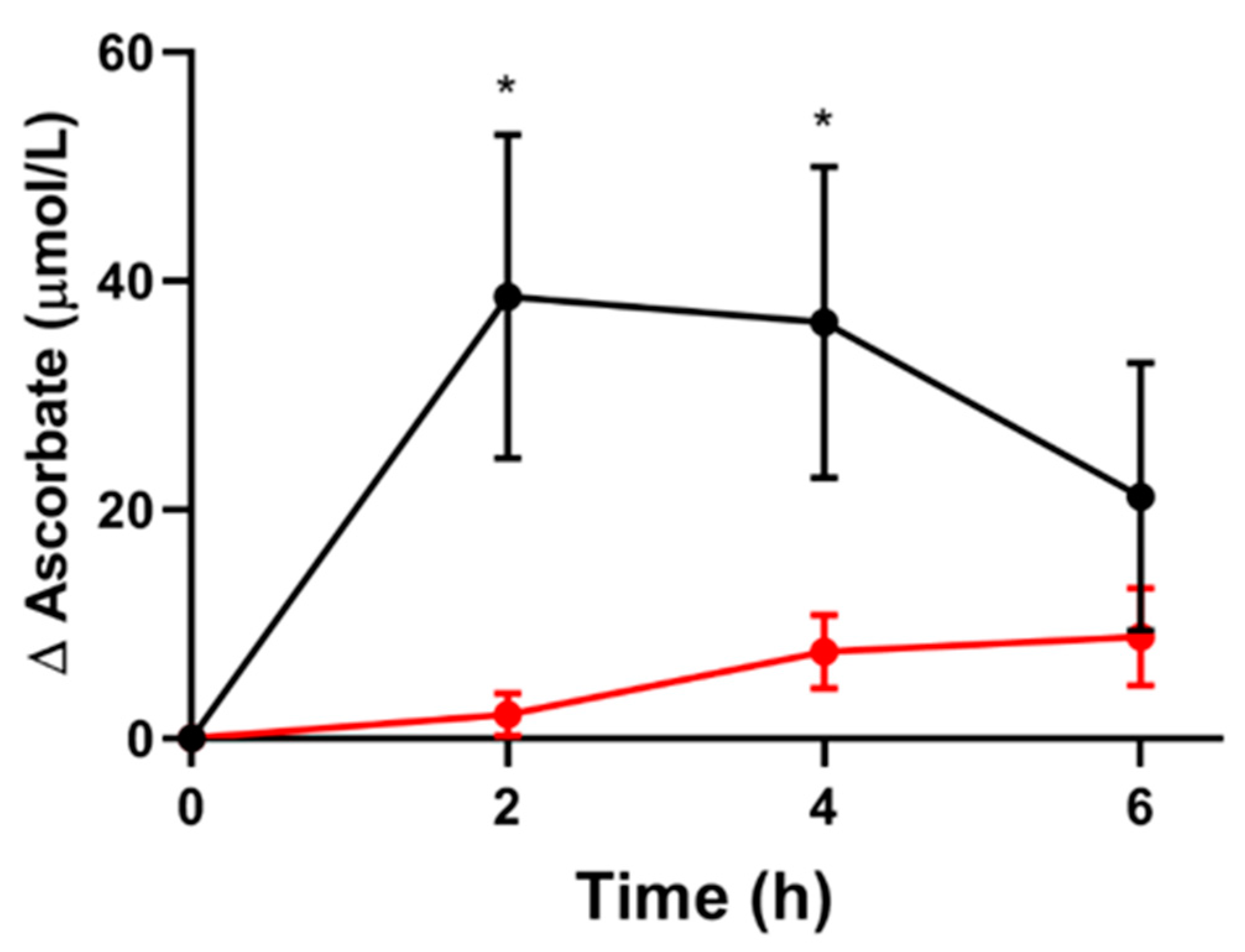
3.2. Short-Term Pharmacokinetic Study
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Smirnoff, N. Ascorbic acid metabolism and functions: A comparison of plants and mammals. Free. Radic. Boil. Med. 2018, 122, 116–129. [Google Scholar] [CrossRef] [PubMed]
- Khalife, R.; Grieco, A.; Khamisa, K.; Tinmouh, A.; McCudden, C.; Saidenberg, E. Scurvy, an old story in a new time: The hematologist’s experience. Blood Cells Mol. Dis. 2019, 76, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Padayatty, S.J.; Levine, M. Vitamin C: The known and the unknown and Goldilocks. Oral Dis. 2016, 22, 463–493. [Google Scholar] [CrossRef] [PubMed]
- Pearson, J.F.; Pullar, J.M.; Wilson, R.; Spittlehouse, J.K.; Vissers, M.C.M.; Skidmore, P.M.L.; Willis, J.; Cameron, V.A.; Carr, A.C. Vitamin C Status Correlates with Markers of Metabolic and Cognitive Health in 50-Year-Olds: Findings of the CHALICE Cohort Study. Nutrients 2017, 9, 831. [Google Scholar] [CrossRef] [PubMed]
- Schleicher, R.L.; Carroll, M.D.; Ford, E.S.; Lacher, D.A. Serum vitamin C and the prevalence of vitamin C deficiency in the United States: 2003–2004 National Health and Nutrition Examination Survey (NHANES). Am. J. Clin. Nutr. 2009, 90, 1252–1263. [Google Scholar] [CrossRef]
- Levine, M.; Conry-Cantilena, C.; Wang, Y.; Welch, R.W.; Washko, P.W.; Dhariwal, K.R.; Park, J.B.; Lazarev, A.; Graumlich, J.F.; King, J.; et al. Vitamin C pharmacokinetics in healthy volunteers: Evidence for a recommended dietary allowance. Proc. Natl. Acad. Sci. USA 1996, 93, 3704–3709. [Google Scholar] [CrossRef]
- Wilson, J.X. Regulation of Vitamin C Transport. Annu. Rev. Nutr. 2005, 25, 105–125. [Google Scholar] [CrossRef]
- Savini, I.; Rossi, A.; Pierro, C.; Avigliano, L.; Catani, M.V. SVCT1 and SVCT2: Key proteins for vitamin C uptake. Amino Acids 2008, 34, 347–355. [Google Scholar] [CrossRef]
- Du, J.; Cullen, J.J.; Buettner, G.R. Ascorbic acid: Chemistry, biology and the treatment of cancer. Biochim. Biophys. Acta Bioenerg. 2012, 1826, 443–457. [Google Scholar] [CrossRef]
- Sotiriou, S.; Gispert, S.; Cheng, J.; Wang, Y.; Chen, A.; Hoogstraten-Miller, S.; Miller, G.F.; Kwon, O.; Levine, M.; Guttentag, S.H.; et al. Ascorbic-acid transporter Slc23a1 is essential for vitamin C transport into the brain and for perinatal survival. Nat. Med. 2002, 8, 514–517. [Google Scholar] [CrossRef]
- Corpe, C.P.; Tu, H.; Eck, P.; Wang, J.; Faulhaber-Walter, R.; Schnermann, J.; Margolis, S.; Padayatty, S.; Sun, H.; Wang, Y.; et al. Vitamin C transporter Slc23a1 links renal reabsorption, vitamin C tissue accumulation, and perinatal survival in mice. J. Clin. Investig. 2010, 120, 1069–1083. [Google Scholar] [CrossRef]
- Vera, J.C.; Rivas, C.I.; Fischbarg, J.; Golde, D.W. Mammalian facilitative hexose transporters mediate the transport of dehydroascorbic acid. Nature 1993, 364, 79–82. [Google Scholar] [CrossRef]
- Rumsey, S.C.; Kwon, O.; Xu, G.W.; Burant, C.; Simpson, I.; Levine, M. Glucose Transporter Isoforms GLUT1 and GLUT3 Transport Dehydroascorbic Acid. J. Boil. Chem. 1997, 272, 18982–18989. [Google Scholar] [CrossRef] [PubMed]
- Mendiratta, S.; Qu, Z.-C.; May, J.M. Erythrocyte Ascorbate Recycling: Antioxidant Effects in Blood. Free. Radic. Boil. Med. 1998, 24, 789–797. [Google Scholar] [CrossRef]
- May, J.M.; Qu, Z.C.; Qiao, H.; Koury, M.J. Maturational loss of the vitamin C transporter in erythrocytes. Biochem. Biophys. Res. Commun. 2007, 360, 295–298. [Google Scholar] [CrossRef] [PubMed]
- May, J.M. Ascorbate function and metabolism in the human erythrocyte. Front. Biosci. 1998, 3, d1–d10. [Google Scholar] [CrossRef]
- Tu, H.; Wang, Y.; Li, H.; Brinster, L.R.; Levine, M. Chemical Transport Knockout for Oxidized Vitamin C, Dehydroascorbic Acid, Reveals Its Functions in vivo. EBioMedicine 2017, 23, 125–135. [Google Scholar] [CrossRef]
- Sage, J.M.; Carruthers, A. Human erythrocytes transport dehydroascorbic acid and sugars using the same transporter complex. Am. J. Physiol. Physiol. 2014, 306, C910–C917. [Google Scholar] [CrossRef]
- Williamson, D.; Winterbourn, C.C. Effect of oral administration of ascorbate on acetylphenylhydrazine-induced Heinz body formation. Br. J. Haematol. 1980, 46, 319–321. [Google Scholar] [CrossRef]
- Evans, R.M.; Currie, L.; Campbell, A. The distribution of ascorbic acid between various cellular components of blood, in normal individuals, and its relation to the plasma concentration. Br. J. Nutr. 1982, 47, 473–482. [Google Scholar] [CrossRef]
- Li, H.; Tu, H.; Wang, Y.; Levine, M. Vitamin C in mouse and human red blood cells: An HPLC assay. Anal. Biochem. 2012, 426, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Pullar, J.M.; Bayer, S.; Carr, A.C. Appropriate Handling, Processing and Analysis of Blood Samples Is Essential to Avoid Oxidation of Vitamin C to Dehydroascorbic Acid. Antioxidants 2018, 7, 29. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.; Wang, Y.; Padayatty, S.J.; Morrow, J. A new recommended dietary allowance of vitamin C for healthy young women. Proc. Natl. Acad. Sci. USA 2001, 98, 9842–9846. [Google Scholar] [CrossRef] [PubMed]
- Tu, H.; Li, H.; Wang, Y.; Niyyati, M.; Wang, Y.; Leshin, J.; Levine, M. Low Red Blood Cell Vitamin C Concentrations Induce Red Blood Cell Fragility: A Link to Diabetes Via Glucose, Glucose Transporters, and Dehydroascorbic Acid. EBioMedicine 2015, 2, 1735–1750. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pullar, J.M.; Dunham, S.; Dachs, G.U.; Vissers, M.C.M.; Carr, A.C. Erythrocyte Ascorbate Is a Potential Indicator of Steady-State Plasma Ascorbate Concentrations in Healthy Non-Fasting Individuals. Nutrients 2020, 12, 418. https://doi.org/10.3390/nu12020418
Pullar JM, Dunham S, Dachs GU, Vissers MCM, Carr AC. Erythrocyte Ascorbate Is a Potential Indicator of Steady-State Plasma Ascorbate Concentrations in Healthy Non-Fasting Individuals. Nutrients. 2020; 12(2):418. https://doi.org/10.3390/nu12020418
Chicago/Turabian StylePullar, Juliet M., Susannah Dunham, Gabi U. Dachs, Margreet C. M. Vissers, and Anitra C. Carr. 2020. "Erythrocyte Ascorbate Is a Potential Indicator of Steady-State Plasma Ascorbate Concentrations in Healthy Non-Fasting Individuals" Nutrients 12, no. 2: 418. https://doi.org/10.3390/nu12020418
APA StylePullar, J. M., Dunham, S., Dachs, G. U., Vissers, M. C. M., & Carr, A. C. (2020). Erythrocyte Ascorbate Is a Potential Indicator of Steady-State Plasma Ascorbate Concentrations in Healthy Non-Fasting Individuals. Nutrients, 12(2), 418. https://doi.org/10.3390/nu12020418







