The Many Faces of Kefir Fermented Dairy Products: Quality Characteristics, Flavour Chemistry, Nutritional Value, Health Benefits, and Safety
Abstract
1. Introduction
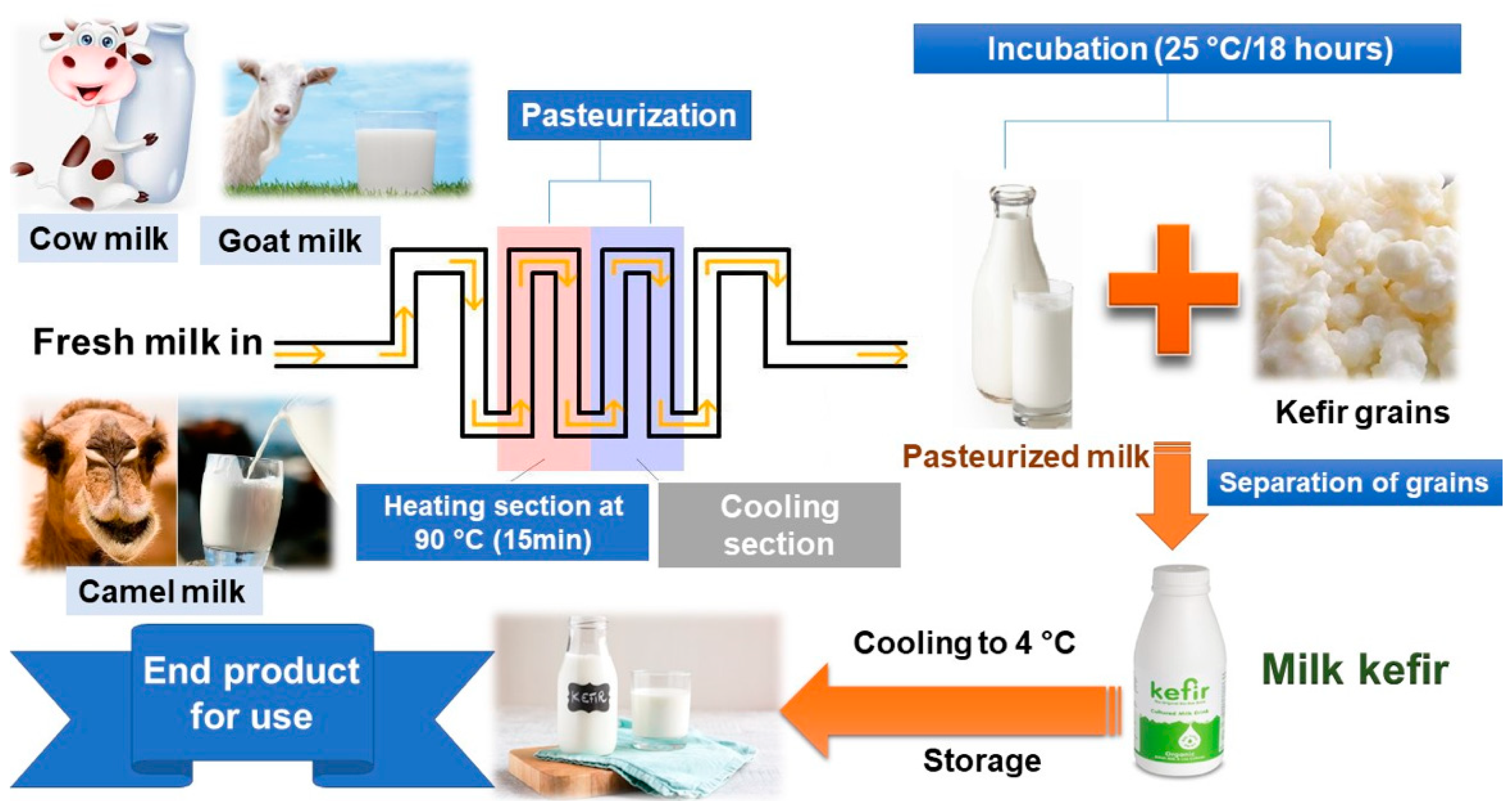
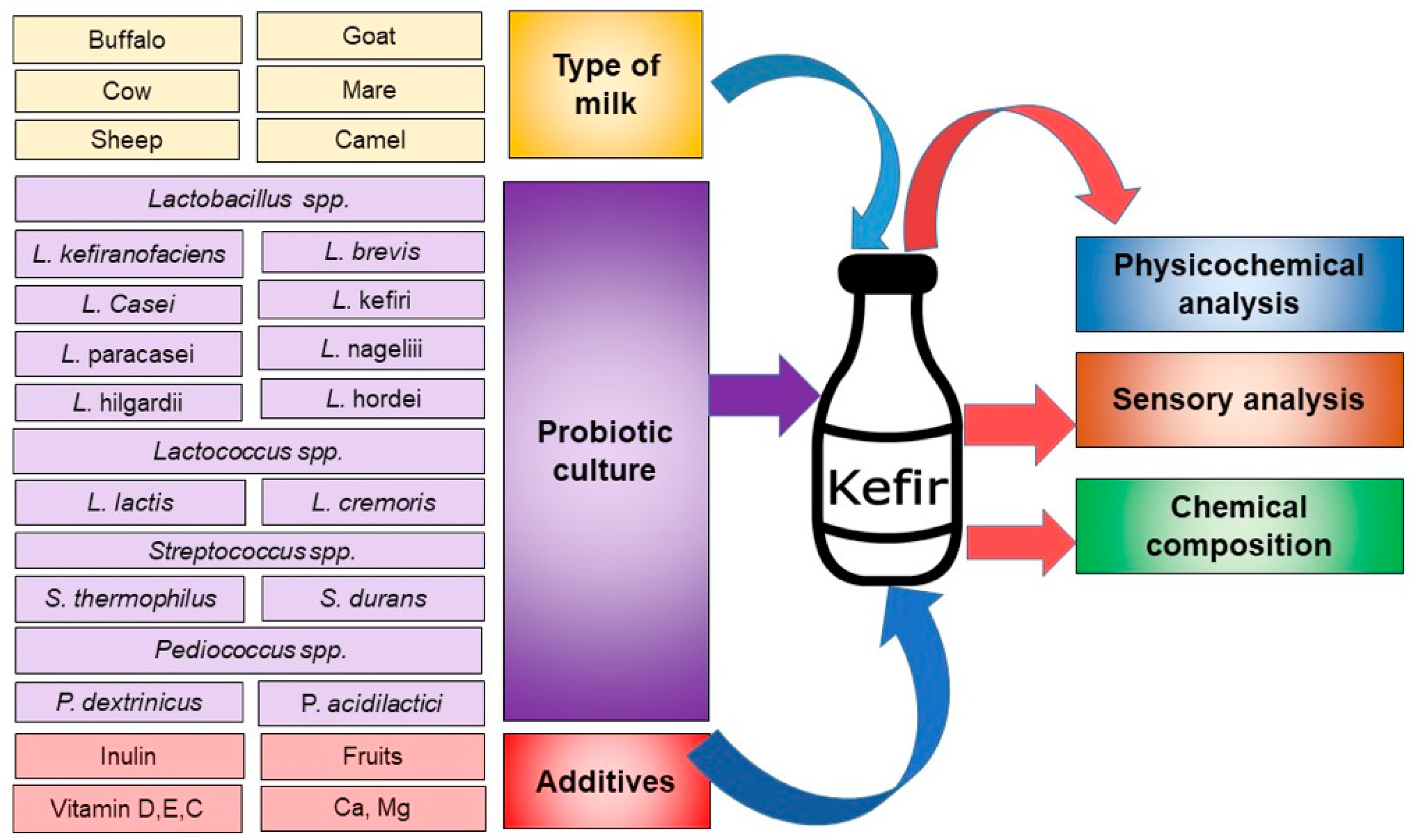
2. Prebiotic, Additives, and Production Methods Employed in Kefir Production
3. Physicochemical Parameters of Kefir in the Context of Its Different Production Methods
4. Sensory Analysis of the Different Kefir Types
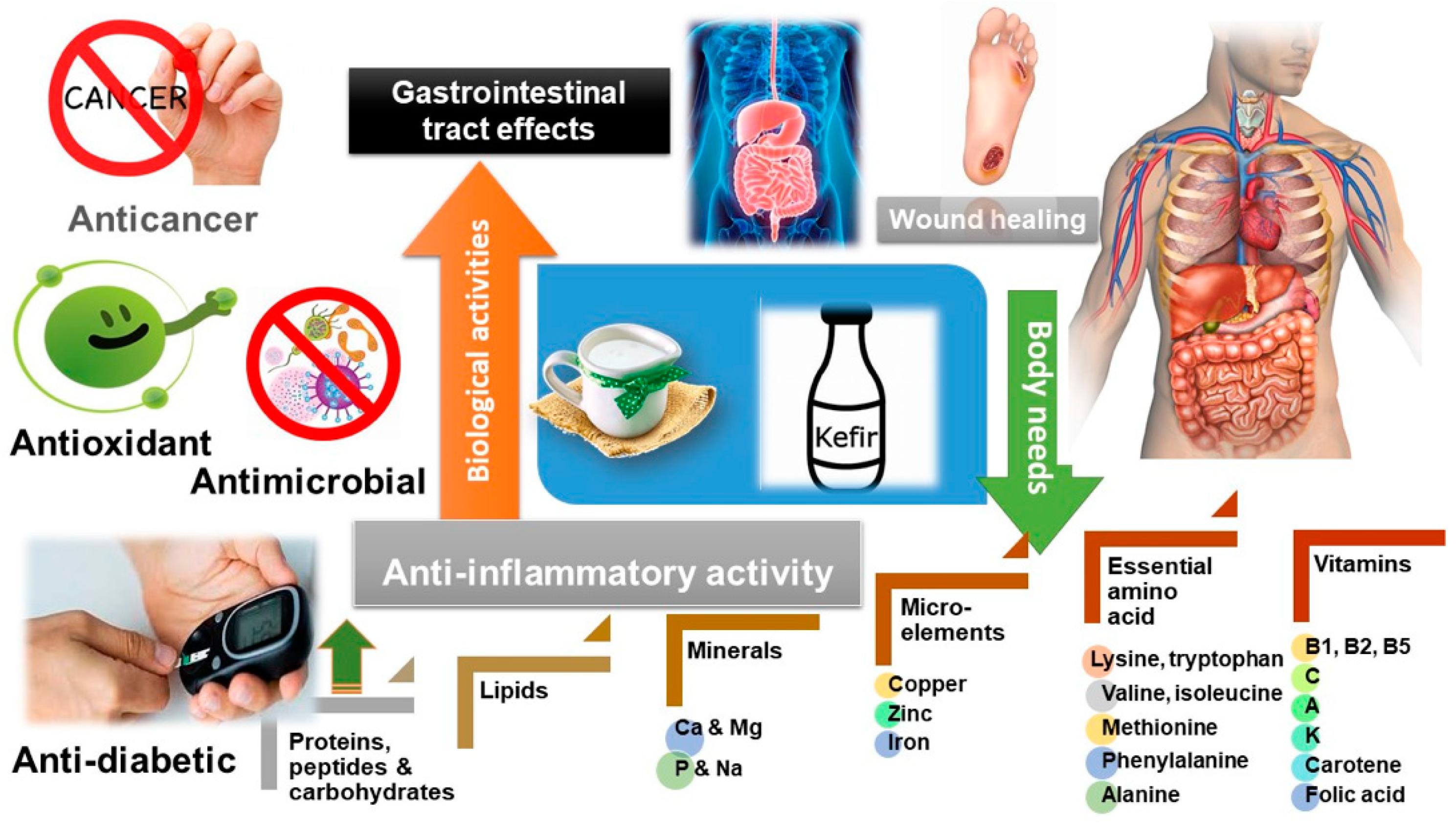
5. Kefir’s Nutritional Value & Health Benefits
6. Kefir’s Biological Properties in the Context of Its Different Manufacturing Practices
6.1. Anticancer Activity
6.2. Hypocholesterolemic Effect
6.3. Antimicrobial Activities
6.4. Wound Healing
6.5. Anti-Inflammatory and Antioxidant Activities
6.6. Anti-Diabetic Activity
6.7. Gastrointestinal Tract Effects and Gut Microbiota Modulation
7. Milk vs. Sugar Kefir Limitations and Safety
8. Concluding Remarks and Future Directions in Kefir
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Prado, M.R.; Blandón, L.M.; Vandenberghe, L.P.S. Milk kefir: Composition, microbial cultures, biological activities, and related products. Front. Microbiol. 2015, 6, 1177–1186. [Google Scholar] [CrossRef] [PubMed]
- Cais-Sokolińska, D.; Wójtowski, J.; Pikul, J.; Danków, R.; Majcher, M.; Teichert, J.; Bagnicka, E. Formation of volatile compounds in kefir made of goat and sheep milk with high polyunsaturated fatty acid content. J. Dairy Sci. 2015, 98, 6692–6705. [Google Scholar] [CrossRef] [PubMed]
- Kavas, G. Kefirs manufactured from camel (camelus dramedarius) milk and cow milk: Comparison of some chemical and microbial properties. Ital. J. Food Sci. 2015, 27, 357–365. [Google Scholar]
- Sun, Z.; Zhang, H.; Yu, Z.; Hou, Q.; Zheng, Y.; Zhong, Z.; Menghe, B.; Kwok, L. Bacterial microbiota compositions of naturally fermented milk are shaped by both geographic origin and sample type. J. Dairy Sci. 2016, 99, 7832–7841. [Google Scholar]
- Karaçalı, R.; Özdemİr, N.; Çon, A.H. Aromatic and functional aspects of kefir produced using soya milk and Bifidobacterium species. Int. J. Dairy Technol. 2018, 71, 921–933. [Google Scholar] [CrossRef]
- Kim, D.; Chon, J.; Kim, H.; Seo, K. Modulation of intestinal microbiota in mice by kefir administration. Food Sci. Biotechnol. 2015, 24, 1397–1403. [Google Scholar] [CrossRef]
- Liu, J.; Wang, S.; Chen, M.; Chen, H.; Yueh, P.; Lin, C. Hypocholesterolaemic effects of milk-kefir and soyamilk-kefir in cholesterol-fed hamsters. Br. J. Nutr. 2014, 95, 939–946. [Google Scholar] [CrossRef]
- Liu, J.; Wang, S.; Chen, M.; Yueh, P.; Lin, C. The anti-allergenic properties of milk kefir and soymilk kefir and their beneficial effects on the intestinal microflora. J. Sci. Food Agric. 2006, 2533, 2527–2533. [Google Scholar] [CrossRef]
- Miao, J.; Guo, H.; Chen, F.; Zhao, L.; He, L.; Ou, Y.; Huang, M.; Zhang, Y.; Guo, B.; Cao, Y.; et al. Antibacterial effects of a cell-penetrating peptide isolated from kefir. AgriculuraL Food Chem. 2016, 64, 3234–3242. [Google Scholar] [CrossRef]
- Sharifi, M.; Moridnia, A.; Mortazavi, D.; Salehi, M. Kefir: A powerful probiotics with anticancer properties. Med. Oncol. 2017, 34, 183–189. [Google Scholar] [CrossRef]
- Khoury, N.; El-Hayek, S.; Tarras, O.; El-Sabban, M.; El-Sibai, M.; Rizk, S. Kefir exhibits anti-proliferative and pro-apoptotic effects on colon adenocarcinoma cells with no significant effects on cell migration and invasion. Int. J. Oncol. 2014, 45, 2117–2127. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.; Huang, W.; Lin, J.; Chen, Y.; Huang, C.; Tung, Y. Kefir supplementation modifies gut microbiota composition, reduces physical fatigue, and improves exercise performance in mice. Nurients 2018, 10, 862. [Google Scholar] [CrossRef] [PubMed]
- Kıvanç, M.; Yapıcı, E. kefir as a probiotic dairy beverage: Determination lactic acid bacteria and yeast. ETP Int. J. Food Eng. 2016, 1, 55–60. [Google Scholar] [CrossRef]
- Cais-sokolińska, D.; Wójtowski, J.; Pikul, J. Rheological, texture and sensory properties of kefir from mare ’ s milk and its mixtures with goat and sheep milk. Mljekarstvo 2016, 66, 272–281. [Google Scholar] [CrossRef]
- Glibowski, P.; Kowalska, A. Rheological, texture and sensory properties of kefir with high performance and native inulin. J. Food Eng. J. 2012, 111, 299–304. [Google Scholar] [CrossRef]
- Fiorda, F.A.; de Melo Pereira, G.V.; Thomaz-Soccol, V.; Rakshit, S.K.; Pagnoncelli, M.G.B.; de Souza Vandenberghe, L.P.; Soccol, C.R. Microbiological, biochemical, and functional aspects of sugary kefir fermentation—A review. Food Microbiol. 2017, 66, 86–95. [Google Scholar] [CrossRef]
- Kim, D.H.; Jeong, D.; Song, K.Y.; Seo, K.H. Comparison of traditional and backslopping methods for kefir fermentation based on physicochemical and microbiological characteristics. Food Sci. Technol. 2018, 97, 503–507. [Google Scholar] [CrossRef]
- Gulitz, A.; Stadie, J.; Wenning, M.; Ehrmann, M.A.; Vogel, R.F. The microbial diversity of water kefir. Int. J. Food Microbiol. 2011, 151, 284–288. [Google Scholar] [CrossRef]
- Shrivastava, N.; Ananthanarayan, L. Use of the backslopping method for accelerated and nutritionally enriched idli fermentation. J. Sci. Food Agric. 2015, 95, 2081–2087. [Google Scholar] [CrossRef]
- Yıldız-Akgül, F.; Yetişemiyen, A.; Şenel, E.; Yıldırım, Z. Microbiological, physicochemical, and sensory characteristics of kefir produced by secondary. Mljekarstvo 2018, 68, 201–213. [Google Scholar] [CrossRef]
- Lopitz-Otsoa, F.; Rementeria, A.; Elguezabala, N.; Garaizar, J. Kefir: A stmbiotic yeasts-bacteria community with alleged healthy capabilities. Rev. Iberoam Micol. 2006, 23, 67–74. [Google Scholar] [CrossRef]
- Machado, A.; Leite, D.O.; Antonio, M.; Miguel, L.; Peixoto, R.S.; Rosado, A.S.; Silva, J.T.; Margaret, V.; Paschoalin, F. Microbiological, technological and therapeutic properties of kefir: A natural probiotic beverage. Brazilian J. Microbiol. 2013, 44, 341–349. [Google Scholar]
- Teijeiro, M.; Pérez, P.F.; De Antoni, G.L.; Golowczyc, M.A. Suitability of kefir powder production using spray drying. Food Res. Int. 2018, 112, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Atalar, I.; Dervisoglu, M. Optimization of spray drying process parameters for ke fi r powder using response surface methodology. LWT Food Sci. Technol. 2015, 60, 751–757. [Google Scholar] [CrossRef]
- Bolla, P.A.; Serradell, M.D.L.A.; De Urraza, P.J.; De Antoni, G.L. Effect of freeze-drying on viability and in vitro probiotic properties of a mixture of lactic acid bacteria and yeasts isolated from kefir. J. Dairy Res. 2011, 78, 13–32. [Google Scholar] [CrossRef] [PubMed]
- Broeckx, G.; Vandenheuvel, D.; Claes, I.J.J.; Lebeer, S.; Kiekens, F. Drying techniques of probiotic bacteria as an important step towards the development of novel pharmabiotics. Int. J. Pharm. 2016, 505, 303–318. [Google Scholar] [CrossRef]
- Gul, O.; Mortas, M.; Atalar, I.; Dervisoglu, M.; Kahyaoglu, T. Manufacture and characterization of kefir made from cow and buffalo milk, using kefir grain and starter culture. J. Dairy Sci. 2015, 98, 1517–1525. [Google Scholar] [CrossRef]
- Setyawardani, T.; Rahardjo, A. Physiochemical and organoleptic features of goat milk kefir made of different kefir grain concentration on controlled fermentation. Anim. Prod. J. 2014, 16, 48–54. [Google Scholar]
- Zhou, J.; Liu, X.; Jiang, H.; Dong, M. Analysis of the microflora in Tibetan kefir grains using denaturing gradient gel electrophoresis. Food Microbiol. 2009, 26, 770–775. [Google Scholar] [CrossRef]
- Irigoyen, A.; Arana, I.; Castiella, M.; Torre, P.; Ib, F.C. Microbiological, physicochemical, and sensory characteristics of kefir during storage. Food Chem. 2005, 90, 613–620. [Google Scholar] [CrossRef]
- Ozcan, T.; Sahin, S.; Akpinar-Bayizit, A.; Yilmaz-Ersan, L. Assessment of antioxidant capacity by method comparison and amino acid characterisation in buffalo milk kefir. Int. J. Dairy Technol. 2019, 72, 65–73. [Google Scholar] [CrossRef]
- Ismaiel, A.A.; Ghaly, M.F.; El-Naggar, A.K. Some physicochemical analyses of kefir produced under different fermentation conditions. J. Sci. Ind. Res. (India). 2011, 70, 365–372. [Google Scholar]
- Arslan, S. A review: Chemical, microbiological and nutritional characteristics of kefir. CyTA J. Food 2015, 13, 340–345. [Google Scholar] [CrossRef]
- Hu, J.B.; Gunathilake, S.; Chen, Y.C.; Urban, P.L. On the dynamics of kefir volatome. RSC Adv. 2014, 4, 28865–28870. [Google Scholar] [CrossRef]
- Magalhães, K.T.; de Melo Pereira, G.V.; Campos, C.R.; Dragone, G.; Schwan, R.F. Brazilian kefir: Structure, microbial communities and chemical composition. Brazilian J. Microbiol. 2011, 42, 693–702. [Google Scholar] [CrossRef]
- Gao, W.; Zhang, L. Comparative analysis of the microbial community composition between Tibetan kefir grains and milks. Food Res. Int. 2019, 116, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.Z.; Liu, X.L.; Huang, K.H.; Dong, M.S.; Jiang, H.H. Application of the mixture design to design the formulation of pure cultures in Tibetan kefir. Agric. Sci. China 2008, 6, 1383–1389. [Google Scholar] [CrossRef]
- Aghlara, A.; Mustafa, S.; Manap, Y.A.; Mohamad, R. Characterization of headspace volatile flavor compounds formed during kefir production: Application of solid phase microextraction. Int. J. Food Prop. 2009, 12, 808–818. [Google Scholar] [CrossRef]
- Wszolek, M.; Tamime, A.Y.; Muir, D.D.; Barclay, M.N.I. Properties of kefir made in scotland and poland using bovine, caprine and ovine milk with different starter cultures. LWT Food Sci. Technol. 2001, 34, 251–261. [Google Scholar] [CrossRef]
- Cais-Sokolińska, D.; Danków, R.; Pikul, J. Physicochemical and sensory characteristics of sheep kefir during storage. Acta Sci. Pol. Technol. Aliment. 2008, 7, 63–73. [Google Scholar]
- Wang, X.; Xiao, J.; Jia, Y.; Pan, Y.; Wang, Y. Lactobacillus kefiranofaciens, the sole dominant and stable bacterial species, exhibits colonization in Tibetan kefir distinct morphotypes upon grains. Heliyon 2018, 4, e00649. [Google Scholar] [CrossRef]
- Sabir, F.; Beyatli, Y.; Cokmus, C.; Onal-Darilmaz, D. Assessment of potential probiotic properties of Lactobacillus spp., Lactococcus spp., and Pediococcus spp. strains isolated from kefir. J. Food Sci. 2010, 75. [Google Scholar] [CrossRef]
- Dertli, E.; Hilmi, A. Microbial diversity of traditional kefir grains and their role on kefir aroma. LWT-Food Sci. Technol. 2017, 85, 151–157. [Google Scholar] [CrossRef]
- Ju, S.Y.; Kim, J.H.; Lee, P.C. Long-term adaptive evolution of Leuconostoc mesenteroides for enhancement of lactic acid tolerance and production. Biotechnol. Biofuels 2016, 9, 240–253. [Google Scholar] [CrossRef]
- Assadi, M.M.; Pourahmad, R.; Moazami, N. Use of isolated kefir starter cultures in kefir production. World J. Microbiol. Biotechnol. 2000, 16, 541–543. [Google Scholar] [CrossRef]
- Yüksekdag, Z.N.; Beyatli, Y.; Aslim, B. Determination of some characteristics coccoid forms of lactic acid bacteria isolated from Turkish kefirs with natural probiotic. LWT-Food Sci. Technol. 2004, 37, 663–667. [Google Scholar] [CrossRef]
- Gientka, I.; Kieliszek, M.; Jermacz, K.; Błazejak, S. Identification and characterization of oleaginous yeast isolated from kefir and its ability to accumulate intracellular fats in deproteinated potato wastewater with different carbon sources. Biomed Res. Int. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Leroi, F.; Pidoux, M. Characterization of interactions between Lactobacillus hilgardii and Saccharomyces florentinus isolated from sugary kefir grains. J. Appl. Bacteriol. 1993, 74, 54–60. [Google Scholar] [CrossRef]
- Zafar, S.; Owais, M. Ethanol production from crude whey by Kluyveromyces marxianus. Biochem. Eng. J. 2006, 27, 295–298. [Google Scholar] [CrossRef]
- Verce, M.; De Vuyst, L.; Weckx, S. Shotgun metagenomics of a water kefir fermentation ecosystem reveals a novel oenococcus species. Front. Microbiol. 2019, 10, 479–494. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Wang, S.; Chen, K.; Liu, J.; Chen, M. Microbiological and chemical properties of kefir manufactured by entrapped microorganisms isolated from kefir grains. J. Dairy Sci. 2009, 92, 3002–3013. [Google Scholar] [CrossRef]
- Carasi, P.; Racedo, S.M.; Jacquot, C.; Romanin, D.E.; Serradell, M.A.; Urdaci, M.C. Impact of kefir derived Lactobacillus kefiri on the mucosal immune response and gut microbiota. J. Immunol. Res. 2015, 2015, 1–12. [Google Scholar] [CrossRef]
- Beshkova, D.M.; Simova, E.D.; Simov, Z.I.; Frengova, G.I.; Spasov, Z.N. Pure cultures for making kefir. Food Microbiol. 2002, 19, 537–544. [Google Scholar] [CrossRef]
- Walsh, A.M.; Crispie, F.; Kilcawley, K.; O’Sullivan, O.; O’Sullivan, M.G.; Claesson, M.J.; Cotter, P.D. Microbial succession and flavor production in the fermented dairy beverage kefir. Msystems 2016, 1, e00052-16. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, X.; Tian, Z.; Yang, Y.; Yang, Z. Characterization of an exopolysaccharide produced by Lactobacillus plantarum YW11 isolated from Tibet Kefir. Carbohydr. Polym. 2015, 125, 16–25. [Google Scholar] [CrossRef]
- Chen, Z.; Shi, J.; Yang, X.; Nan, B.; Liu, Y.; Wang, Z. Chemical and physical characteristics and antioxidant activities of the exopolysaccharide produced by Tibetan kefir grains during milk fermentation. Int. Dairy J. 2015, 43, 15–21. [Google Scholar] [CrossRef]
- Jeong, D.; Kim, D.H.; Kang, I.B.; Kim, H.; Song, K.Y.; Kim, H.S.; Seo, K.H. Characterization and antibacterial activity of a novel exopolysaccharide produced by Lactobacillus kefiranofaciens DN1 isolated from kefir. Food Control 2017, 78, 436–442. [Google Scholar] [CrossRef]
- Öner, Z.; Karahan, A.G.; Çakmakçı, M.L. Effects of different milk types and starter cultures on kefir. Gida 2009, 7, 1–6. [Google Scholar]
- Paraskevopoulou, A.; Athanasiadis, I.; Blekas, G.; Koutinas, A.A.; Kanellaki, M. Influence of polysaccharide addition on stability of a cheese whey kefir-milk mixture. Food Hydrocoll. 2003, 17, 615–620. [Google Scholar] [CrossRef]
- Vasiliki, L.; Georgia, D.; Aikatarini, K. Effects of greek pomegranate extracts in the antioxidant properties and storage stability of kefir. Curr. Bioact. Compd. 2019, 15, 437–441. [Google Scholar]
- Gul, O.; Dervisoglu, M. Rheological, textural, colour and sensorial properties of kefir produced with buffalo milk using kefir grains and starter culture: A comparison with cows ’ milk kefir. Int. J. Dairy Technol. 2018, 71, 73–80. [Google Scholar] [CrossRef]
- Said, N.S.; Fahrodi, D.U.; Malaka, R.; Maruddin, F. The physicochemical, microbiology, and sensory characteristics of kefir goat milk with different levels of kefir grain. Trop. Anim. Sci. J. 2019, 42, 152–158. [Google Scholar]
- Bakhshandeh, T.; Pourahmad, R.; Sharifan, A.; Moghimi, A. Evaluation of flavor and aroma compounds present in kefir. J. Food Biosci. Technol. 2011, 1, 11–18. [Google Scholar]
- Dimitreli, G.; Petridis, D.; Kapageridis, N.; Mixiou, M. Effect of pomegranate juice and fir honey addition on the rheological and sensory properties of kefir-type products differing in their fat content. LWT-Food Sci. Technol. 2019, 111, 799–808. [Google Scholar] [CrossRef]
- Farag, M.A.; Wessjohann, L.A. Metabolome classification of commercial Hypericum perforatum (St. John’s Wort) preparations via UPLC-qTOF-MS and chemometrics. Planta Med. 2012, 78, 488–496. [Google Scholar] [CrossRef]
- Bensmira, M.; Jiang, B. Total phenolic compounds and antioxidant activity of a novel peanut based kefir. Food Sci. Biotechnol 2015, 24, 1055–1060. [Google Scholar] [CrossRef]
- Sarkar, S. Potential of kefir as a dietetic beverage—A review. Br. Food J. 2007, 109, 280–290. [Google Scholar] [CrossRef]
- Simova, E.; Simov, Z.; Beshkova, D.; Frengova, G.; Dimitrov, Z.; Spasov, Z. Amino acid profiles of lactic acid bacteria, isolated from kefir grains and kefir starter made from them. Int. J. Food Microbiol. 2006, 107, 112–123. [Google Scholar] [CrossRef]
- Grohmann, U.; Bronte, V. Control of immune response by amino acid metabolism. Immunol. Rev. 2010, 236, 243–264. [Google Scholar] [CrossRef]
- Bifari, F.; Nisoli, E. Branched-chain amino acids differently modulate catabolic and anabolic states in mammals: A pharmacological point of view. Br. J. Pharmacol. 2017, 174, 1366–1377. [Google Scholar] [CrossRef]
- Bakircioglu, D.; Topraksever, N.; Yurtsever, S.; Kizildere, M.; Kurtulus, Y.B. Investigation of macro, micro and toxic element concentrations of milk and fermented milks products by using an inductively coupled plasma optical emission spectrometer, to improve food safety in Turkey. Microchem. J. 2018, 136, 133–138. [Google Scholar] [CrossRef]
- Ebner, J.; Aşçi Arslan, A.; Fedorova, M.; Hoffmann, R.; Küçükçetin, A.; Pischetsrieder, M. Peptide profiling of bovine kefir reveals 236 unique peptides released from caseins during its production by starter culture or kefir grains. J. Proteomics 2015, 117, 41–57. [Google Scholar] [CrossRef] [PubMed]
- Miao, J.; Liu, G.; Ke, C.; Fan, W.; Li, C.; Chen, Y.; Dixon, W.; Song, M.; Cao, Y.; Xiao, H. Inhibitory effects of a novel antimicrobial peptide from kefir against Escherichia coli. Food Control 2016, 65, 63–72. [Google Scholar] [CrossRef]
- Amorim, F.G.; Coitinho, L.B.; Dias, A.T.; Gomes, A.; Friques, F.; Monteiro, B.L.; Dias, L.C.; Rezende, D.; De Melo, T.; Pereira, C.; et al. Identification of new bioactive peptides from kefir milk through proteopeptidomics: Bioprospection of antihypertensive molecules. Food Chem. 2019, 282, 109–119. [Google Scholar] [CrossRef]
- Rosa, D.D.; Grześkowiak, Ł.M.; Ferreira, C.L.; Fonseca, A.C.M.; Reis, S.A.; Dias, M.M.; Siqueira, N.P.; Silva, L.L.; Neves, C.A.; Oliveira, L.L.; et al. Kefir reduces insulin resistance and inflammatory cytokine expression in an animal model of metabolic syndrome. Food Funct. 2016, 7, 3390–3401. [Google Scholar] [CrossRef]
- Cho, H.-Y.; Choi, J.-W.; Kang, H.W.; Kim, M.-K.; Lim, W.-C.; Lee, I.-Y. Kefir prevented excess fat accumulation in diet-induced obese mice. Biosci. Biotechnol. Biochem. 2017, 81, 958–965. [Google Scholar] [CrossRef]
- Tsiouris, C.G.; Kelesi, M.; Vasilopoulos, G.; Kalemikerakis, I.; Papageorgiou, E.G. The efficacy of probiotics as pharmacological treatment of cutaneous wounds: Meta-analysis of animal studies. Eur. J. Pharm. Sci. 2017, 104, 230–239. [Google Scholar] [CrossRef]
- Chen, C.; Chan, H.M.; Kubow, S. Kefir extracts suppress in vitro proliferation of estrogen-dependent human breast cancer cells but not normal mammary epithelial cells. J. Med. Food 2007, 10, 416–422. [Google Scholar] [CrossRef]
- Dos Reis, S.A.; da Conceição, L.L.; Dias, M.M.; Siqueira, N.P.; Rosa, D.D.; de Oliveira, L.L.; da Matta, S.L.P.; Peluzio, M.D.C.G. Kefir reduces the incidence of pre-neoplastic lesions in an animal model for colorectal cancer. J. Funct. Foods 2019, 53, 1–6. [Google Scholar] [CrossRef]
- Rizk, S.; Maalouf, K.; Baydoun, E. The antiproliferative effect of kefir cell-free fraction on hut-102 malignant t lymphocytes. Clin. Lymphoma Myeloma 2009, 9, S198–S203. [Google Scholar] [CrossRef]
- Hatmal, M.M.; Nuirat, A.; Zihlif, M.A.; Taha, M.O. Exploring the influence of culture conditions on kefir’s anticancer properties. J. Dairy Sci. 2018, 101, 3771–3777. [Google Scholar] [CrossRef] [PubMed]
- De Paula Melo, A.F.; Mendonça, M.C.P.; de Mendonça Rosa-Castro, R. The protective effects of fermented kefir milk on azoxymethane-induced aberrant crypt formation in mice colon. Tissue Cell 2018, 52, 51–56. [Google Scholar] [CrossRef]
- Boeneke, C.; O’Brien, K.; Lisano, J.K.; Hayward, R.; Smoak, P.; Mathias, A.; Stewart, L.K.; Christensen, M.; Hydock, D.S. Milk and kefir maintain aspects of health during doxorubicin treatment in rats. J. Dairy Sci. 2019, 102, 1910–1917. [Google Scholar]
- Yamane, T.; Sakamoto, T.; Nakagaki, T.; Nakano, Y. Lactic acid bacteria from kefir increase cytotoxicity of natural killer cells to tumor cells. Foods 2018, 7, 48. [Google Scholar] [CrossRef]
- Esener, O.B.B.; Balkan, B.M.; Armutak, E.I.; Uvez, A.; Yildiz, G.; Hafizoglu, M.; Yilmazer, N.; Gurel-Gurevin, E. Donkey milk kefir induces apoptosis and suppresses proliferation of Ehrlich ascites carcinoma by decreasing iNOS in mice. Biotech. Histochem. 2018, 93, 424–431. [Google Scholar] [CrossRef]
- Gao, J.; Gu, F.; Ruan, H.; Chen, Q.; He, J.; He, G. Induction of apoptosis of gastric cancer cells SGC7901 in vitro by a cell-free fraction of Tibetan kefir. Int. Dairy J. 2013, 30, 14–18. [Google Scholar] [CrossRef]
- Osada, K.; Nagira, K.; Teruya, K.; Tachibana, H.; Shirahata, S.; Murakami, H. Enhancement of interferon-β production with sphingomyelin from fermented milk. Biotherapy 1993, 7, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Güzel-Seydim, Z.B.; Ece Cagdas, M.D.; Seydim, A.C. Effect of kefir on fusobacterium nucleatum potentially causing intestinal cancer. Funct. Foods Heal. Dis. 2016, 6, 469–477. [Google Scholar] [CrossRef]
- Wong, B.; Kruse, G.; Kutikova, L.; Ray, K.K.; Mata, P.; Bruckert, E. Cardiovascular disease risk associated with familial hypercholesterolemia: A systematic review of the literature. Clin. Ther. 2016, 38, 1696–1709. [Google Scholar] [CrossRef]
- Chen, H.L.; Tung, Y.T.; Tsai, C.L.; Lai, C.W.; Lai, Z.L.; Tsai, H.C.; Lin, Y.L.; Wang, C.H.; Chen, C.M. Kefir improves fatty liver syndrome by inhibiting the lipogenesis pathway in leptin-deficient ob/ob knockout mice. Int. J. Obes. 2014, 38, 1172–1179. [Google Scholar] [CrossRef]
- Kim, D.H.; Kim, H.; Jeong, D.; Kang, I.B.; Chon, J.W.; Kim, H.S.; Song, K.Y.; Seo, K.H. Kefir alleviates obesity and hepatic steatosis in high-fat diet-fed mice by modulation of gut microbiota and mycobiota: Targeted and untargeted community analysis with correlation of biomarkers. J. Nutr. Biochem. 2017, 44, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Andrade, T.U.; Porto, M.L.; Filete, P.F.; Abdalla, D.S.P.; Lima, E.M.; Pereira, T.M.C.; Meyrelles, S.S.; Lenz, D.; Vasquez, E.C.; Santanna, A.F.; et al. Chronic administration of the soluble, nonbacterial fraction of kefir attenuates lipid deposition in LDLr −/− mice. Nutrition 2016, 35, 100–105. [Google Scholar]
- Tung, Y.T.; Chen, H.L.; Wu, H.S.; Ho, M.H.; Chong, K.Y.; Chen, C.M. Kefir peptides prevent hyperlipidemia and obesity in high-fat-diet-induced obese rats via lipid metabolism modulation. Mol. Nutr. Food Res. 2018, 62, 1700505–1700513. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.; Choi, J.; Lim, W.; Kim, M. Kefir inhibits 3T3-L1 adipocyte differentiation through down-regulation of adipogenic. J. Sci. Food Agric. 2013, 93, 485–490. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, X.; Wang, J.; Wu, F.; Sui, Y.; Yang, L.; Wang, Z. Lactobacillus plantarum strains as potential probiotic cultures with cholesterol-lowering activity. J. Dairy Sci. 2013, 96, 2746–2753. [Google Scholar] [CrossRef]
- Huang, Y.; Wu, F.; Wang, X.; Sui, Y.; Yang, L.; Wang, J. Characterization of Lactobacillus plantarum Lp27 isolated from Tibetan kefir grains: A potential probiotic bacterium with cholesterol-lowering effects. J. Dairy Sci. 2013, 96, 2816–2825. [Google Scholar] [CrossRef]
- Zheng, Y.; Lu, Y.; Wang, J.; Yang, L.; Pan, C.; Huang, Y. Probiotic properties of lactobacillus strains isolated from tibetan kefir grains. PLoS One 2013, 8, e69868–e69877. [Google Scholar] [CrossRef]
- Chen, H.L.; Tsai, T.C.; Tsai, Y.C.; Liao, J.W.; Yen, C.C.; Chen, C.M. Kefir peptides prevent high-fructose corn syrup-induced non-alcoholic fatty liver disease in a murine model by modulation of inflammation and the JAK2 signaling pathway. Nutr. Diabetes 2016, 6, e237–e245. [Google Scholar] [CrossRef]
- Abdel-mogheith, S.; El-gendy, A.O. Exploring the antimicrobial and hepatoprotective effects of kefir; a probiotic fermented milk. J. Pure Appl. Microbiol. 2017, 11, 759–772. [Google Scholar] [CrossRef]
- Ismaiel, A.A.; Ghaly, M.F.; El-Naggar, A.K. Milk kefir: Ultrastructure, antimicrobial activity and efficacy on aflatoxin b1 production by Aspergillus flavus. Curr. Microbiol. 2011, 62, 1602–1609. [Google Scholar] [CrossRef]
- Macuamule, C.L.S.; Wiid, I.J.; van Helden, P.D.; Tanner, M.; Witthuhn, R.C. Effect of milk fermentation by kefir grains and selected single strains of lactic acid bacteria on the survival of Mycobacterium bovis BCG. Int. J. Food Microbiol. 2016, 217, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Nicoli, J.R.; Neumann, E.; Arantes, R.M.E.; Abatemarco Júnior, M.; Nunes, Á.C.; Sandes, S.H.C.; Ricci, M.F. Protective effect of Lactobacillus diolivorans 1Z, isolated from Brazilian kefir, against Salmonella enterica serovar Typhimurium in experimental murine models. Front. Microbiol. 2018, 9, 2856–2867. [Google Scholar]
- Miao, J.; Guo, H.; Ou, Y.; Liu, G.; Fang, X.; Liao, Z.; Ke, C.; Chen, Y.; Zhao, L.; Cao, Y. Purification and characterization of bacteriocin F1, a novel bacteriocin produced by Lactobacillus paracasei subsp. tolerans FX-6 from Tibetan kefir, a traditional fermented milk from Tibet, China. Food Control 2014, 42, 48–53. [Google Scholar] [CrossRef]
- De Montijo-Prieto, S.; Moreno, E.; Bergillos-Meca, T.; Lasserrot, A.; Ruiz-López, M.D.; Ruiz-Bravo, A.; Jiménez-Valera, M. A Lactobacillus plantarum strain isolated from kefir protects against intestinal infection with Yersinia enterocolitica O9 and modulates immunity in mice. Res. Microbiol. 2015, 166, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Jeong, D.; Kim, D.H.; Song, K.Y.; Seo, K.H. Antimicrobial and anti-biofilm activities of Lactobacillus kefiranofaciens DD2 against oral pathogens. J. Oral Microbiol. 2018, 10, 1472985–1472993. [Google Scholar] [CrossRef]
- Kakisu, E.; Bolla, P.; Abraham, A.G.; de Urraza, P.; De Antoni, G.L. Lactobacillus plantarum isolated from kefir: Protection of cultured Hep-2 cells against Shigella invasion. Int. Dairy J. 2013, 33, 22–26. [Google Scholar] [CrossRef]
- Wang, R.; Lechtenberg, M.; Sendker, J.; Petereit, F.; Deters, A.; Hensel, A. Wound-healing plants from TCM: In vitro investigations on selected TCM plants and their in fl uence on human dermal fi broblasts and keratinocytes. Fitoterapia 2013, 84, 308–317. [Google Scholar] [CrossRef]
- Rodrigues, K.L.; Gaudino Caputo, L.R.; Tavares Carvalho, J.C.; Evangelista, J.; Schneedorf, J.M. Antimicrobial and healing activity of kefir and kefiran extract. Int. J. Antimicrob. Agents 2005, 25, 404–408. [Google Scholar] [CrossRef]
- Barzegari, A.A.; Hashemzaei, M.; Majdani, R.; Alihemmati, A.-R. Effects of topical treatment of second-degree burn wounds with Lactobacillus acidophilus on the wound healing process in male rats. Pharm. Biomed. Res. 2018, 3, 23–30. [Google Scholar] [CrossRef]
- Mohseni, S.; Bayani, M.; Bahmani, F.; Tajabadi-Ebrahimi, M.; Bayani, M.A.; Jafari, P.; Asemi, Z. The beneficial effects of probiotic administration on wound healing and metabolic status in patients with diabetic foot ulcer: A randomized, double-blind, placebo-controlled trial. Diabetes. Metab. Res. Rev. 2018, 34, 1–8. [Google Scholar] [CrossRef]
- Oryan, A.; Alemzadeh, E.; Eskandari, M.H. Kefir accelerates burn wound healing through inducing fibroblast cell migration in vitro and modulating the expression of IL-1β, TGF-β1, and bFGF genes in vivo. Probiotics Antimicrob. Proteins 2018, 11, 788–790. [Google Scholar]
- Huseini, H.F.; Rahimzadeh, G.; Fazeli, M.R.; Mehrazma, M.; Salehi, M. Evaluation of wound healing activities of kefir products. Burns 2012, 38, 719–723. [Google Scholar] [CrossRef] [PubMed]
- Seo, M.K.; Park, E.J.; Ko, S.Y.; Choi, E.W.; Kim, S. Therapeutic effects of kefir grain Lactobacillus -derived extracellular vesicles in mice with 2, 4, 6-trinitrobenzene sulfonic acid-induced inflammatory bowel disease. J. Dairy Sci. 2018, 101, 8662–8671. [Google Scholar] [CrossRef] [PubMed]
- Radhouani, H.; Gonçalves, C.; Maia, F.R.; Oliveira, J.M.; Reis, R.L. Biological performance of a promising Kefiran-biopolymer with potential in regenerative medicine applications: A comparative study with hyaluronic acid. J. Mater. Sci. Mater. Med. 2018, 29, 124–133. [Google Scholar] [CrossRef]
- Yilmaz-Ersan, L.; Ozcan, T.; Akpinar-Bayizit, A.; Sahin, S. Comparison of antioxidant capacity of cow and ewe milk kefirs. J. Dairy Sci. 2018, 101, 3788–3798. [Google Scholar] [CrossRef]
- Sabokbar, N.; Khodaiyan, F.; Moosavi-Nasab, M. Optimization of processing conditions to improve antioxidant activities of apple juice and whey based novel beverage fermented by kefir grains. J. Food Sci. Technol. 2015, 52, 3422–3432. [Google Scholar] [CrossRef]
- Nurliyani; Sadewa, A.H.; Sunarti. Kefir properties prepared with goat milk and black rice (Oryza sativa L.) extract and its influence on the improvement of pancreatic β-cells in diabetic rats. Emirates J. Food Agric. 2015, 27, 727–735. [Google Scholar] [CrossRef]
- Bourrie, B.C.T.; Cotter, P.D.; Willing, B.P. Traditional kefir reduces weight gain and improves plasma and liver lipid profiles more successfully than a commercial equivalent in a mouse model of obesity. J. Funct. Foods 2018, 46, 29–37. [Google Scholar] [CrossRef]
- Harmayani, E. Antidiabetic potential of kefir combination from goat milk and soy milk in rats induced with streptozotocin-nicotinamide. Korean J. Food Sci. Anim. Resour. 2016, 35, 847–858. [Google Scholar]
- Hamet, M.F.; Medrano, M.; Pérez, P.F.; Abraham, A.G. Oral administration of kefiran exerts a bifidogenic effect on BALB/c mice intestinal microbiota. Benef. Microbes 2016, 7, 237–246. [Google Scholar] [CrossRef]
- Bolla, P.A.; Abraham, A.G.; Pérez, P.F.; De Los Angeles Serradell, M. Kefir-isolated bacteria and yeasts inhibit Shigella flexneri invasion and modulate pro-inflammatory response on intestinal epithelial cells. Benef. Microbes 2016, 7, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Franco, M.C.; Golowczyc, M.A.; De Antoni, G.L.; Pe, P.F. Administration of kefir-fermented milk protects mice against Giardia intestinalis infection Printed in Great Britain. J. Med. Microbiol. 2013, 62, 1815–1822. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Teruya, K.; Myojin-maekawa, Y.; Shimamoto, F.; Watanabe, H.; Nakamichi, N.; Tokumaru, K.; Tokumaru, S.; Shirahata, S. Protective effects of the fermented milk kefir on x-ray irradiation- induced intestinal damage in b6c3f1 mice. Biol. Pharm. Bull. 2013, 36, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Bolla, P.A.; Carasi, P.; Angeles, M.; De Antoni, G.L. Anaerobe protective effect of a mixture of kefir isolated lactic acid bacteria and yeasts in a hamster model of clostridium dif fi cile infection. Anaerobe 2013, 21, 28–33. [Google Scholar] [CrossRef]
- Corona, O.; Randazzo, W.; Miceli, A.; Guarcello, R.; Francesca, N.; Erten, H.; Moschetti, G.; Settanni, L. Characterization of kefir-like beverages produced from vegetable juices. LWT-Food Sci. Technol. 2016, 66, 572–581. [Google Scholar] [CrossRef]
- Bernardeau, M.; Vernoux, J.P.; Henri-Dubernet, S.; Guéguen, M. Safety assessment of dairy microorganisms: The Lactobacillus genus. Int. J. Food Microbiol. 2008, 126, 278–285. [Google Scholar] [CrossRef]
- Leite, A.M.O.; Miguel, M.A.L.; Peixoto, R.S.; Paschoalin, V.M.F.; Mayo, B. Probiotic potential of selected lactic acid bacteria strains isolated from Brazilian kefir grains. J. Dairy Sci. 2015, 98, 3622–3632. [Google Scholar] [CrossRef]
- Yin, W.; Uthumporn, K.; Rosma, U.; Irfan, A. Evaluation of probiotic potential and anti-hyperglycemic properties of a novel Lactobacillus strain isolated from water kefir grains. Food Sci. Biotechnol. 2018, 27, 1369–1376. [Google Scholar]
- Ansari, F.; Khodaiyan, F.; Rezaei, K.; Rahmani, A. Modelling of aflatoxin G1 reduction by kefir grain using response surface methodology. J. Environ. Heal. Sci. Eng. 2015, 13, 1–7. [Google Scholar] [CrossRef][Green Version]
- Rosa, D.D.; Peluzio, M.d.C.G.; Bueno, T.P.; Cañizares, E.V.; Miranda, L.S.; Dorbignyi, B.M.; Dubí, D.C.; Castaño, I.E.; Grzeskowiak, Ł.M.; Ferreira, C.L.L.F. Evaluation of the subchronic toxicity of kefir by oral administration in Wistar rats. Nutr. Hosp. 2014, 29, 1352–1359. [Google Scholar]
- Carasi, P.; Jacquot, C.; Romanin, D.E.; Elie, A.M.; De Antoni, G.L.; Urdaci, M.C.; de los Angeles Serradell, M. Safety and potential beneficial properties of Enterococcus strains isolated from kefir. Int. Dairy J. 2014, 39, 193–200. [Google Scholar]
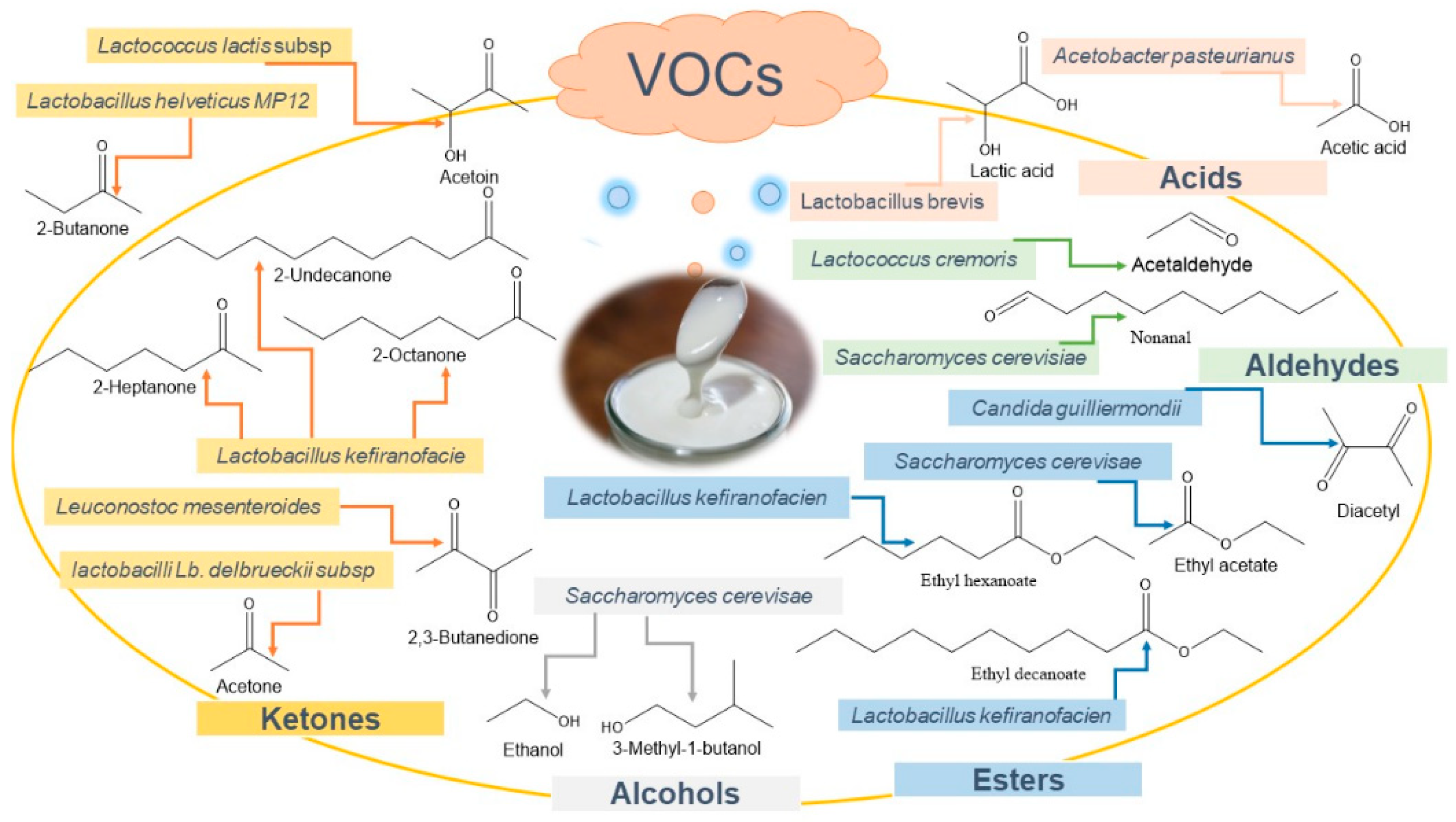
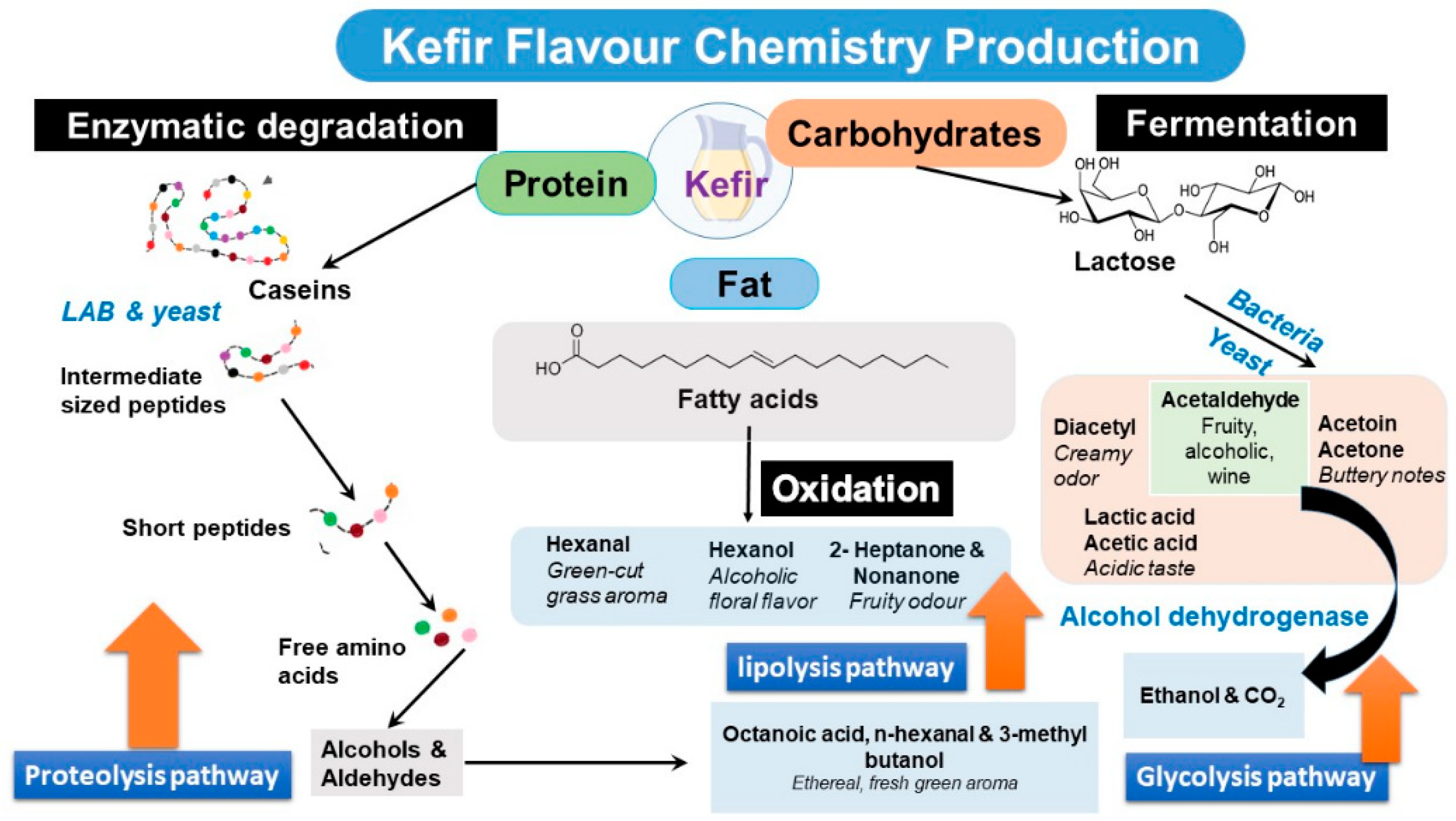
| Microorganisms (Kefir Type) | Produced Metabolites | Odor and FlavorDescription | Reference |
|---|---|---|---|
| Lactobacillus kefiranofaciens (Tibetan kefir) Lactobacillus acidophilus Z1L (Turkish homemade kefir) Lactobacillus brevis Lactobacillus helveticus Z5L Lactobacillus casei Z7L1 (Turkish homemade kefir) Lactobacillus harbinensis (Water kefir) Leuconostoc mesenteroides Lactococcus cremoris Lactococcus lactis Streptococcus durans Pediococcus dextrinicus ZN1P Pediococcus acidilactici ZN10P Pediococcus pentosaceus ZN13P (Candida kefir) Candida guilliermondii Saccharomyces Jorentinus (Sugar kefir) | Lactic acid | Sour flavor | [41,42,43,44,45,46,47,48] |
| Lactobacillus harbinensis (Water kefir) Leuconostoc mesenteroides Acetobacter pasteurianus Lactococcus cremoris | Acetic acid | Vinegar, green, fruity, sour | [43] |
| Lactobacillus kefiri Lactobacillus harbinensis Lactobacillus hilgardii (Water kefir) Leuconostoc mesenteroides (Candida kefir) Saccharomyces turicensis Saccharomyces florentinus Kluyveromyces marxianus | Ethanol | Alcoholic flavor | [35,43,44,45,48,49,50,51,52] |
| LactobacillusBrevis Lactobacillus kefiri Leuconostocmesenteroides (Candida kefir) Saccharomycesturicensis Saccharomycescerevisae Pichia Kurdriavzevii | CO2 | Sharp odor and a sour taste | [45,51,52,53] |
| Lactobacillus kefiranofaciens | Octanoic acid | Cheesy, rancid, pungent, sweet, soapy, goaty | [54] |
| Lactobacillus kefiranofaciens (Tibetan kefir) Lactobacillus kefiri (Brazilian kefir) | Kefiran | - | [41] |
| Lactobacillus kefiranofaciens | n-Decanoic acid | Soapy, waxy, stale, buttery, fruity, grassy, cheesy, milky | [54] |
| Lactobacillus kefiranofaciens | Ethyl decanoate | Fruity, grape, cognac | [54] |
| Lactobacillus kefiranofaciens | Ethyl hexanoate | Animal, cardboard | [54] |
| Lactobacillus nagelii, Lactobacillus hilgardii & Lactobacillus hordei (Water kefir) Lactobacillus plantarum YW11 (Tibet Kefir) Lactobacillus acidophilus Z1L (Turkish homemade kefir) Lactobacillus helveticus Z5L Lactobacillus casei Z7Ll Lactococcus cremoris Z11S & Lactococcus lactis Z3S (Turkish homemade kefirs) Pediococcus dextrinicus ZN1P Pediococcus acidilactici ZN10P Pediococcus pentosaceus ZN13P Kazachstania unispora | Exopolysaccharides (EPS) | [18,42,55,56] | |
| Lactobacillus kefiranofaciens DN1 | New exopolysaccharide (EPS), EPS_DN1 | - | [57] |
| Lactobacillus acidophilus Z1L & Lactococcuscremoris Z11S (Turkish homemade kefir) Pediococcus dextrinicus ZN1P Pediococcus acidilactici ZN10P Pediococcus pentosaceus ZN13P Candida guilliermondii Streptococcus thermophiles | H2O2 | - | [42,46,47] |
| Lactobacillus hilgardii Saccharomyces florentinus (Sugar kefir) | Pyruvate | - | [48,50] |
| Lactobacillus hilgardii Saccharomyces florentinus (Sugar kefir) | Propionate | - | [48,50] |
| Lactobacillus hilgardii Saccharomyces florentinus (Sugar kefir) | Acetate | - | [48,50] |
| Lactobacillus hilgardii Saccharomyces florentinus (Sugar kefir) | Succinate | - | [48,50] |
| Lactobacillus hilgardii Saccharomyces florentinus (Sugar kefir) | Fumarate | - | [48,50] |
| Lactobacillus hilgardii (Sugar kefir) | Mannitol | - | [48,50] |
| Leuconostocmesenteroides | 2,3-Butanedione | Buttery flavors | [43,46,47] |
| Lactococcus cremoris Streptococcus thermophilus Streptococcus durans Candidaguilliermondii | Acetaldehyde | Fruity, alcoholic, wine | [47] |
| Candidaguilliermondii | Diacetyl | Creamy odor | [18,35,41,50] |
| Saccharomycescerevisae (Brazilian kefir, water kefir, and Tibetan kefir grains) | Glycerol | - | [18,35,41,50] |
| Saccharomycescerevisae | Nonanal | Green, citrus, fatty, floral | [54] |
| Saccharomycescerevisae | Phenylethyl alcohol | Unclean, rose, violet-like, honey, floral, spicy | [54] |
| Saccharomycescerevisae | Octanal | Green, fatty, soapy, fruity, orange peel | [54] |
| Saccharomycescerevisae | Ethyl acetate | Solvent, pineapple, fruity, apples | [54] |
| Saccharomycescerevisae | 3-Methyl-butanol | Penetrating, alcohol, wine-like, plastic | [54] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farag, M.A.; Jomaa, S.A.; Abd El-Wahed, A.; R. El-Seedi, H. The Many Faces of Kefir Fermented Dairy Products: Quality Characteristics, Flavour Chemistry, Nutritional Value, Health Benefits, and Safety. Nutrients 2020, 12, 346. https://doi.org/10.3390/nu12020346
Farag MA, Jomaa SA, Abd El-Wahed A, R. El-Seedi H. The Many Faces of Kefir Fermented Dairy Products: Quality Characteristics, Flavour Chemistry, Nutritional Value, Health Benefits, and Safety. Nutrients. 2020; 12(2):346. https://doi.org/10.3390/nu12020346
Chicago/Turabian StyleFarag, Mohamed A., Suzan A. Jomaa, Aida Abd El-Wahed, and Hesham R. El-Seedi. 2020. "The Many Faces of Kefir Fermented Dairy Products: Quality Characteristics, Flavour Chemistry, Nutritional Value, Health Benefits, and Safety" Nutrients 12, no. 2: 346. https://doi.org/10.3390/nu12020346
APA StyleFarag, M. A., Jomaa, S. A., Abd El-Wahed, A., & R. El-Seedi, H. (2020). The Many Faces of Kefir Fermented Dairy Products: Quality Characteristics, Flavour Chemistry, Nutritional Value, Health Benefits, and Safety. Nutrients, 12(2), 346. https://doi.org/10.3390/nu12020346






