The Impact of Food Histamine Intake on Asthma Activity: A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
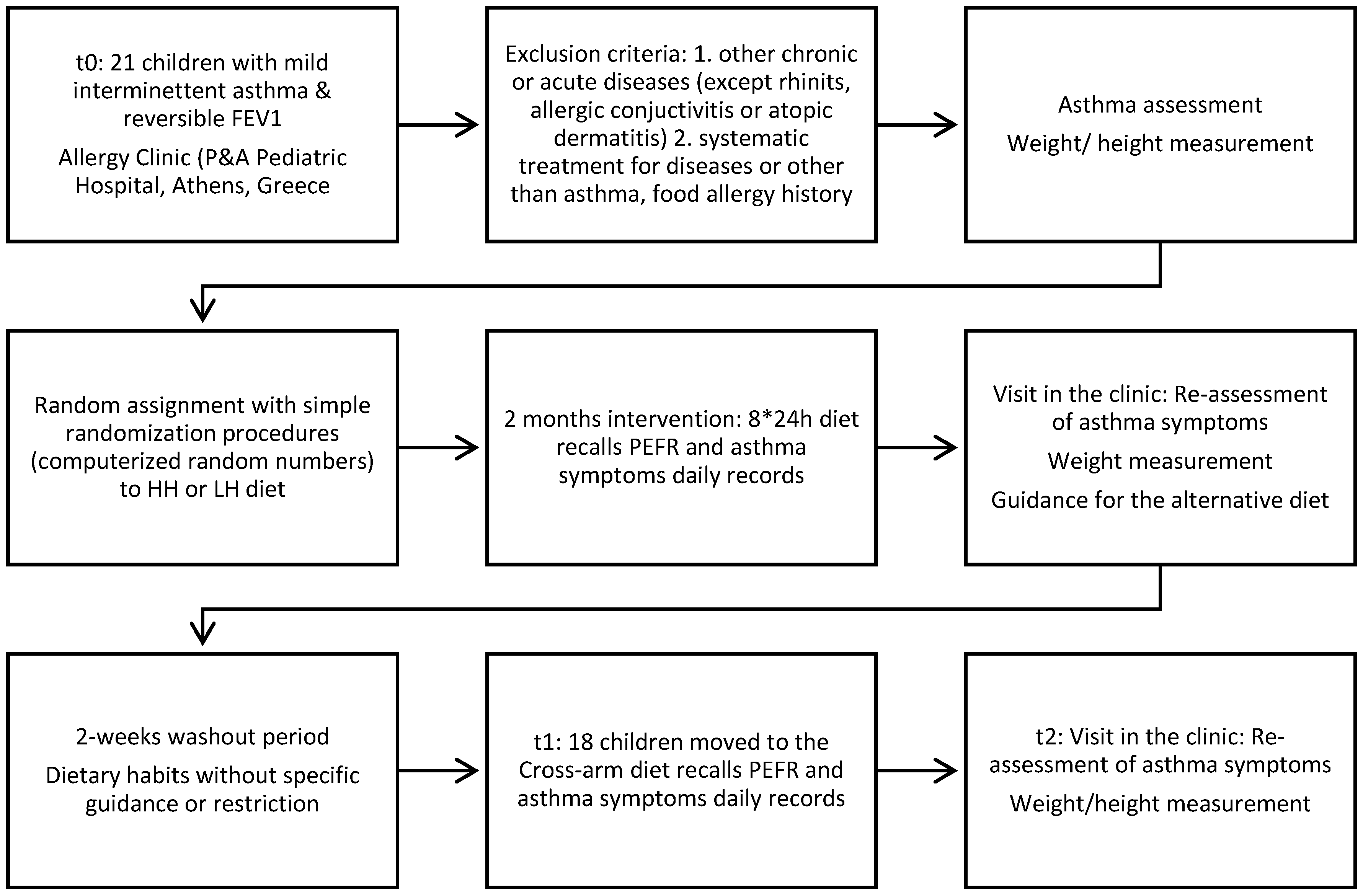
2.1. Patients
2.2. Study Design
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pavord, I.D.; Beasley, R.; Agusti, A.; Anderson, G.P.; Bel, E.; Brusselle, G.; Cullinan, P.; Custovic, A.; Ducharme, F.M.; Fahy, J.V.; et al. After asthma: Redefining airways diseases. Lancet 2018, 391, 350–400. [Google Scholar] [CrossRef]
- Papadopoulos, N.G.; Arakawa, H.; Lodrup-Carlsen, K.; Custovic, A.; Gern, J.; Lemanske, R.; Le Souef, P.; Mäkelä, M.; Roberts, G.; Wong, G.; et al. International consensus on (ICON) pediatric asthma. Allergy Eur. J. Allergy Clin. Immunol. 2012, 67, 976–997. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61 (Suppl. S6). [Google Scholar] [CrossRef] [PubMed]
- Calatayud-Sáez, F.M.; Calatayud Moscoso del Prado, B.; Gallego Fernández-Pacheco, J.G.; González-Martín, C.; Alguacil Merino, L.F. Mediterranean diet and childhood asthma. Allergol. Immunopathol. (Madr) 2016, 44, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Berentzen, N.E.; Van Stokkom, V.L.; Gehring, U.; Koppelman, G.H.; Schaap, L.A.; Smit, H.A.; Wijga, A.H. Associations of sugar-containing beverages with asthma prevalence in 11-year-old children: The PIAMA birth cohort. Eur. J. Clin. Nutr. 2015, 69, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.-Y.; Forno, E.; Alvarez, M.; Colón-Semidey, A.; Acosta-Perez, E.; Canino, G.; Celedón, J.C. Diet, Lung Function, and Asthma Exacerbations in Puerto Rican Children. Pediatr. Allergy Immunol. Pulmonol. 2017, 30, 202–209. [Google Scholar] [CrossRef]
- Guilleminault, L.; Williams, E.J.; Scott, H.A.; Berthon, B.S.; Jensen, M.; Wood, L.G. Diet and Asthma: Is It Time to Adapt Our Message? Nutrients 2017, 9, 1227. [Google Scholar] [CrossRef]
- Seaton, A.; Godden, D.J.; Brown, K. Increase in asthma: A more toxic environment or a more susceptible population? Thorax 1994, 49, 171–174. [Google Scholar] [CrossRef]
- Wantke, F.; Gotz, M.; Jarisch, R. Histamine-free diet: Treatment of choice for histamine-induced food intolerance and supporting treatment for chronical headaches. Clin. Exp. Allergy 1993, 23, 982–985. [Google Scholar] [CrossRef]
- Maintz, L.; Novak, N. Histamine and Histamine Intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef]
- Reese, I.; Ballmer-Weber, B.; Beyer, K.; Fuchs, T.; Kleine-Tebbe, J.; Klimek, L.; Lepp, U.; Niggemann, B.; Saloga, J.; Schäfer, C.; et al. German guideline for the management of adverse reactions to ingested histamine: Guideline of the German Society for Allergology and Clinical Immunology (DGAKI), the German Society for Pediatric Allergology and Environmental Medicine (GPA), the German Association of Allergologists (AeDA), and the Swiss Society for Allergology and Immunology (SGAI). Allergo J. Int. 2017, 26, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Dunford, P.J.; Holgate, S.T. The Role of Histamine in Asthma. In Histamine in Inflammation; Advances in Experimental Medicine and Biology; Thurmond, R.L., Ed.; Springer: Boston, MA, USA, 2010; Volume 709. [Google Scholar] [CrossRef]
- Yamauchi, K.; Ogasawara, M. The Role of Histamine in the Pathophysiology of Asthma and the Clinical Efficacy of Antihistamines in Asthma Therapy. Int. J. Mol. Sci. 2019, 20, 1733. [Google Scholar] [CrossRef] [PubMed]
- Barcik, W.; Pugin, B.; Westermann, P.; Perez, N.R.; Ferstl, R.; Wawrzyniak, M.; Smolinska, S.; Jutel, M.; Hessel, E.M.; Michalovich, D.; et al. Histamine-secreting microbes are increased in the gut of adult asthma patients. J. Allergy Clin. Immunol. 2016, 138, 1491–1494.e7. [Google Scholar] [CrossRef] [PubMed]
- Pocket Guide for Asthma Management and Prevention (for Adults and Children Older than 5 Years. Available online: https://ginasthma.org/wp-content/uploads/2019/04/GINA-2019-main-Pocket-Guide-wms.pdf (accessed on 2 July 2020).
- Histamine Intolerance Awareness. The Food List. Available online: https://www.histamineintolerance.org.uk/about/the-food-diary/the-food-list/ (accessed on 9 September 2020).
- Chung, B.Y.; Park, S.Y.; Byun, Y.S.; Son, J.H.; Choi, Y.W.; Cho, Y.S.; Park, C.W.; Park, C.W. Effect of Different Cooking Methods on Histamine Levels in Selected Foods. Ann. Dermatol. 2017, 29, 706–714. [Google Scholar] [CrossRef]
- Sánchez-Pérez, S.; Comas-Basté, O.; Rabell-González, J.; Eveciana-Nogués, M.; Latorre-Moratalla, M.L.; Vidal-Carou, M. Biogenic Amines in Plant-Origin Foods: Are They Frequently Underestimated in Low-Histamine Diets? Foods 2018, 7, 205. [Google Scholar] [CrossRef]
- San Mauro Martin, I.; Brachero, S.; Garicano Vilar, E. Histamine intolerance and dietary management: A complete review. Allergol. Immunopathol. (Madr.) 2016, 44, 475–483. [Google Scholar] [CrossRef]
- Gibson, A.A.; Hsu, M.S.H.; Rangan, A.M.; Seimon, R.V.; Lee, C.M.Y.; Das, A.; Finch, C.H.; Sainsbury, A. Accuracy of hands v. household measures as portion size estimation aids. J. Nutr. Sci. 2016, 5, e29. [Google Scholar] [CrossRef]
- Adeniyi, A.; Erhabor, G.E.; The peak flow meter and its use in clinical practice. Afr. J. Respir. Med. 2011. Available online: https://www.africanjournalofrespiratorymedicine.com/articles/Peak%20flow%20meter.pdf (accessed on 15 October 2020).
- Konstantinou, G.N.; Xepapadaki, P.; Manousakis, E.; Makrinioti, H.; Kouloufakou-Gratsia, K.; Saxoni-Papageorgiou, P.; Papadopoulos, N.G. Assessment of airflow limitation, airway inflammation, and symptoms during virus-induced wheezing episodes in 4- to 6-year-old children. J. Allergy Clin. Immunol. 2013, 131. [Google Scholar] [CrossRef]
- Esha Research. Food Processor Nutrition Analysis Software. Available online: https://esha.com/products/food-processor/ (accessed on 2 July 2020).
- Mariscal-Arcas, M.; Rivas, A.; Velasco, J.; Ortega, M.; Caballero, A.M.; Olea-Serrano, F. Evaluation of the Mediterranean Diet Quality Index (KIDMED) in children and adolescents in Southern Spain. Public Health Nutr. 2009, 12, 1408–1412. [Google Scholar] [CrossRef]
- DRI Dietary Reference Intakes. Available online: https://www.ncbi.nlm.nih.gov/books/NBK222890/ (accessed on 2 July 2020).
- D’Vaz, N.; Meldrum, S.; Dunstan, J.A.; Martino, D.; McCarthy, S.; Metcalfe, J.; Tulic, M.K.; Mori, T.A.; Prescott, S.L. Postnatal fish oil supplementation in high-risk infants to prevent allergy: Randomized controlled trial. Pediatrics 2012, 130, 674–682. [Google Scholar] [CrossRef]
- Schindler, T.; Sinn, J.K.; Osborn, D.A. Polyunsaturated fatty acid supplementation in infancy for the prevention of allergy. Cochrane Database Syst. Rev. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Nagel, G.; Linseisen, J. Dietary intake of fatty acids, antioxidants and selected food groups and asthma in adults’. Eur. J. Clin. Nutr. 2005, 59, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xun, P.; Zamora, D.; Sood, A.; Liu, K.; Daviglus, M.; Iribarren, C.; Jacobs, D.; Shikany, J.M.; He, K. Intakes of long-chain omega-3 (n-3) PUFAs and fish in relation to incidence of asthma among American young adults: The CARDIA study. Am. J. Clin. Nutr. 2013, 97, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.-Y.; Forno, E.; Celedón, J.C. Vitamin D Insufficiency and Asthma in a US Nationwide Study. J. Allergy Clin. Immunol. Pract. 2017, 5, 790–796. [Google Scholar] [CrossRef]
- Yadav, M.; Mittal, K. Effect of vitamin D supplementation on moderate to severe bronchial asthma. Indian J. Pediatr. 2014, 81, 650–654. [Google Scholar] [CrossRef]
- Kerley, C.P.; Hutchinson, K.; Cormican, L.; Faul, J.; Greally, P.; Coghlan, D.; Elnazir, B. Vitamin D3 for uncontrolled childhood asthma: A pilot study. Pediatr. Allergy Immunol. 2016, 27, 404–412. [Google Scholar] [CrossRef]
- Wendell, S.G.; Baffi, C.; Holguin, F. Fatty acids, inflammation, and asthma. J. Allergy Clin. Immunol. 2014, 133, 1255–1264. [Google Scholar] [CrossRef]
- Allen, S.; Britton, J.R.; Leonardi-Bee, J.A. Association between antioxidant vitamins and asthma outcome measures: Systematic review and meta-analysis. Thorax 2009, 64, 610–619. [Google Scholar] [CrossRef]
- Ghaffari, J.; Khalilian, A.; Salehifar, E.; Khorasani, E.; Rezaii, M. Effect of zinc supplementation in children with asthma: A randomized, placebo-controlled trial in northern Islamic Republic of Iran. East. Mediterr. Health J. 2014, 20, 391–396. [Google Scholar] [CrossRef]
- Rago, D.; Rasmussen, M.A.; Lee-Sarwar, K.A.; Weiss, S.T.; Lasky-Su, J.; Stokholm, J.; Bønnelykke, K.; Chawes, B.L.; Bisgaard, H. Fish-oil supplementation in pregnancy, child metabolomics and asthma risk. EBioMedicine 2019, 46, 399–410. [Google Scholar] [CrossRef]
- Brigham, E.P.; Matsui, E.C.; Appel, L.J.; Bull, D.A.; Curtin-Brosnan, J.; Zhai, S.; White, K.; Charleston, J.B.; Hansel, N.N.; Diette, G.B.; et al. A pilot feeding study for adults with asthma: The healthy eating better breathing trial. PLoS ONE 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Strub, P.; Lavori, P.W.; Buist, A.S.; Camargo, C.A.; Nadeau, K.C.; Wilson, S.R.; Xiao, L. DASH for asthma: A pilot study of the DASH diet in not-well-controlled adult asthma. Contemp. Clin. Trials. 2013, 35, 55–67. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stoodley, I.; Williams, L.; Thompson, C.; Scott, H.; Wood, L. Evidence for lifestyle interventions in asthma. Breathe 2019, 15, e50–e61. [Google Scholar] [CrossRef] [PubMed]
- Vassilopoulou, E.; Efthymiou, D.; Elissavet, V.; Agorastos, A. Case Report Integrative Food, Nutrition and Metabolism. Integr. Food Nutr. Metab. 2019. [Google Scholar] [CrossRef]
| Food Group/Type of Diet | Low Histamine | High Histamine |
|---|---|---|
| Dairy products | Fresh pasteurised plain milk without additives (cow’s or goat’s), butter, plain yogurt, ricotta cheese, cottage cheese, cream cheese | All mature cheese, e.g., brie, camembert, cedar, Cheshire, Danish blue, edam, emmental, Gloucester, gouda, mozzarella, parmesan, Roquefort, yogurt with additives such as fruits, cereal, cacao milk, chocolate milk, plain whole fat long life milk |
| Fruit | Fresh not very mature apple, apricot, peach, pear, melon | Mature dates, kiwi, orange, papaya, tangerine, passion fruit, avocado, banana, fig, grape, lemon, pineapple, plum, berry, strawberry |
| Vegetables | Asparagus, cabbage, lettuce, green beans, onion, pepper, radish, turnip | Broccoli, cauliflower, eggplant, mushroom, tomato, sauerkraut, spinach, cucumber |
| Legumes | Peas, lentils, chickpeas | Beans, soybeans |
| Starchy products/grains | Bread with natural yeast without preservatives, oat cereals, pure corn flakes, potato, sweet potato, rice, spaghetti, any type of pasta excluded pasta stuffed with cheese, mince meat, cold-cuts, (e.g., tortellini) and colored pasta (with spinach, carrot, etc. | Bread with preservatives, long life bread, stuffed pasta with cheese, mince meat, cold cuts, colored pasta, cereals with chocolate, cereals with fruits, cereals with nuts, ready-made meals (frozen) |
| Nuts | Chestnut, sunflower seeds, pine nuts, pistachios, almonds, coconuts, currants, red currants, cashew nut, macadamia nut | Hazelnuts, brazil nuts, walnuts, pecan, peanuts, peanut butter |
| Spices | Herbs, spices except for anise, red pepper, curry (ready-made spicy products not allowed) | Malt vinegar, meat products, soy sauce, vinegar, Worcestershire sauce, yeast products |
| Meat/poultry/fish | Beef, chicken without skin, turkey without skin, fresh fish, lamb, rabbit, veal, cooked eggs, boiled unsalted turkey | Meat, fish and poultry older than two days, any frozen meat, any form of raw or smoked meat or fish, anchovies, beef liver, pies and pasta with meat, fish nuggets, salami, sausages, chicken livers, canned tuna, canned salmon |
| Fats/oils | All vegetable oils, homemade sauces prepared with permitted ingredients | Ready-made sauces, olives, oils with preservatives and colorants, olives |
| Beverages | Homemade lemonade | Tea, decaffeinated tea, ready-made juices, chocolate drinks, cacao-drink, soft drinks including cola-type, tomato juice, vegetable juices |
| Sweets/sweeteners | Sugar, homemade jams, homemade sweets with permitted ingredients | Cocoa, milk chocolate, black chocolate, white chocolate |
| Low Histamine * | |||||||
|---|---|---|---|---|---|---|---|
| Breakfast | 1 cup of fresh whole fat plain milk+ 2 slices of whole wheat bread made with fresh yeast+ 1tsp butter+ 1tsp honey | 1 cup of fresh whole fat milk+ 1slice of whole wheat bread made with fresh yeast+ 1tsp homemade peach jam | 1 cup of fresh whole fat plain milk+ 3 whole wheat bread rusks | 1 cup of fresh whole fat plain milk+ 1 whole wheat bread+ 1tsp honey | 200gr whole fat Greek yogurt+ 1 pear | 1 cup of fresh whole fat plain milk+ 2 slices of whole wheat bread made with fresh yeast+ 1tsp butter+ 1tsp honey | 1 cup of fresh whole fat plain milk+ 2 slices of whole wheat bread made with fresh yeast+ 1tsp butter+ 1tsp honey |
| Snack | 1 cup melon | 1 cup of fresh apple-pear juice | 1 apple | 1 pear | 1 cup of fresh juice from apple-pear | 1 apple | 1 cup of fresh apple juice |
| Lunch | 1 portion green fresh beans+ 1 slice of whole wheat bread+ potatoes | 1 portion stuffed zucchini or peppers with rice and beef or turkey mince meat+ 1slice of whole-wheat bread | 1 portion of boiled fresh fish+ 1 boiled potato+ boiled carrots, zucchini | 1 portion ratatouille vegetables (zucchini, carrots, peppers, onion, olive oil, potatoes)+ 1 slice whole wheat bread | 1 portion of pasta with turkey/chicken/beef mince meat+ boiled zucchini+ olive oil | 4–5 fried meatballs with chicken mince meat+ 15–20 slices of fried potatoes+ lettuce salad | 1 portion leek with rice, lemon and olive oil+ 1 slice whole wheat bread+ cottage or cream cheese |
| Snack | Oven-baked apple with egg and breadcrumbs, sugar and cinnamon | 1 milkshake with fresh whole fat plain milk, vanilla flavour, honey | Homemade popcorn with olive oil or butter | 1 slice of homemade vanilla cake | 1 homemade rice pudding | 1 full-fat yogurt+ 1tsp honey | 1 apple milk shake: full-fat milk, apple, cinnamon, honey |
| Dinner | Homemade Caesars Chicken salad with lettuce, boiled corn, cucumber, olive oil | 1 homemade chicken burger with whole wheat bread, lettuce | 1 homemade sandwich with lettuce, cottage cheese, boiled turkey ham, lettuce | 1 homemade souvlaki with chicken meat and whole wheat pita bread, fried potatoes | Egg salad: egg, tomato, lettuce, olive oil | Lettuce salad with baked lamb meat | Baked chicken meat with cucumber-lettuce salad and olive oil |
| High Histamine * | |||||||
| Breakfast | 1 cup of whole fat long life milk+ 2whole wheat homemade chocolate biscuits | 1 cup of whole fat long life milk+ 1slice of whole wheat bread+ 1tsp Nutella | 1 cup of whole fat long life chocolate milk+ 2 homemade vanilla biscuits | 1 cup of whole fat long life milk+ 1 whole wheat bread+ 1tsp honey | 1 fruit stirred yogurt with strawberries | 1 cup of whole fat long life milk+ ½ cup chocolate cereals | 1 cup of whole fat long life milk+ 2 slices of toasted bread+ 2tsp strawberry jam |
| Snack | 1 cup of fresh orange juice | 1 cup of packed mixed fruit juice | 1 banana | 1 cup of packed anana juice | 1 banana | 1 kiwi fruit | 1 cup of packed mixed fruit juice |
| Lunch | 1 portion of lentils+ mature yellow cheese (Cretan graviera)+ 1slice of wholewheat bread | 1 portion stuffed tomatoes with rice and frozen mince-meat+ 1slice of wholewheat bread+ feta cheese | 1 portion of frozen fish sticks+ 1boiled potato+ cauliflower or broccoli | 1 portion “tourlou” vegetables (aubergine, carrots, cucumber, tomato, olive oil, potatoes)+ 1 slice whole wheat bread+ feta cheese | 1 portion pasta with tomato sauce and mushrooms+ parmesan cheese+ Tomato salad | 4-5 fried meatballs from frozen minced-meat+ 15-20 slices of fried potatoes+ broccoli or cauliflower | 1 portion spinach with rice and tomato+ feta cheese+ 1 slice of whole wheat bread |
| Snack | 1 yogurt with chocolate slices | Homemade Banana milkshake (banana, whole milk, honey) | Fruit salad: grapes, orange, banana | 1 cup strawberries or grapes | chocolate ice cream | 50gr milk or back chocolate | Lemon or strawberry Sorbet |
| Dinner | 1 piece of ham and cheese pie | 1 burger with pork mince neat_ tomato salad | 1 hot dog: whole wheat bread, 1 sausage stuffed with cheese, 1tsp mustard, 1tsp ketchup | 1 souvlaki with pork meat and whole wheat pita bread, fried potatoes, tomato | Chef salad: Roquefort cheese, salami, cedar, boiled egg, tomato, lettuce, olive il | 1 slice of spinach pie | Baked pork meat with Greek salad |
| Type of Diet | ||||
|---|---|---|---|---|
| Low Histamine | High Histamine | p-Values ** | ||
| Symptoms duration in days | 5.5 (8) | 9.5 (4) | 0.753 | |
| Respiratory Symptoms | Upper * | 4.67 (11.5) | 12.5 (5.83) | 0.780 |
| Lower * | 4.25 (13.1) | 14 (12.45) | 0.727 | |
| Total * | 11.33 (53.5) | 30.17 (21.42) | 0.889 | |
| LH Period | HH Period | HH vs. LH | ||||||
|---|---|---|---|---|---|---|---|---|
| Remission | Episode | Remission | Episode | Remission | Episode | |||
| p-Value 1 | p-Value 1 | p-Value 2 | p-Value 3 | |||||
| Milk | 1.74 (±1.38) | 2.69 (±1.21) | 0.064 | 2.69 (±1.98) | 3.77 (±1.35) | 0.133 | 0.004 | 0.107 |
| Starchy food | 3.65 (2.94) | 6.04 (2.72) | 0.021 | 4.61 (3.28) | 6.27 (±1.85) | 0.046 | 0.972 | 0.141 |
| Meat | 4.01 (±2.91) | 5.19 (±2.32) | 0.382 | 3.25 (±2.05) | 4.4 (±1.78) | 0.133 | 0.196 | 0.207 |
| Olive oil | 3.24 (±3.12) | 0.26 (±0.44) | 0.014 | 0.28 (±0.44) | 4.23 (±3.16) | 0.002 | 0.002 | 0.007 |
| Butter | 0.318 (±0.83) | 0.05 (±0.14) | 0.299 | 2.56 (±2.62) | 0.46 (±0.5) | 0.043 | 0.014 | 0.007 |
| Fish | 0.51 (±0.89) | 0.75 (±0.59) | 0.184 | 3.7 (±2.93) | 0.29 (±0.47) | 0.003 | 0.059 | 0.003 |
| Chocolate | 0.268 (±0.7) | 0.28 (±0.45) | 0.657 | 0.9 (±0.74) | 3.33 (±3.06) | 0.007 | 0.002 | 0.015 |
| Egg | 0.05 (±0.15) | 0.04 (±0.08) | 0.809 | 0.89 (±1.47) | 0.18 (±0.34) | 0.053 | 0.027 | 0.006 |
| Sweats | 0.74 (±0.8) | 1.12 (±1.1) | 0.328 | 1.55 (±1.34) | 1.46 (±1.08) | 0.861 | 0.463 | 0.123 |
| Nuts | 0.67 (±1.3) | 0.4 (±0.42) | 0.528 | 0.61 (±1.39) | 0.09 (±0.19) | 0.426 | 0.035 | 0.514 |
| Bacon or sausages or ham | 0.11 (±0.18) | 5.08 (±3.21) | 0.002 | 0.31 (±1.11) | 0.71 (±0.46) | 0.030 | 0.002 | 0.242 |
| Junk food | 1.01 (±1.27) | 0.89 (±1.15) | 0.889 | 0.12 (±0.3) | 2.03 (±1.41) | 0.002 | 0.055 | 0.017 |
| Fried oil | 0.79 (±1.25) | 0.14 (±0.13) | 0.726 | 1.23 (±1.59) | 1.26 (±0.94) | 0.917 | 0.003 | 0.105 |
| Juices | 0 | 0.8 (0.76) | 0.004 | 0.15 (0.56) | 0.12 (0.2) | 0.155 | 0.016 | 0.317 |
| Fruit | 0.29M (±0.37) | 0.82 (±0.61) | 0.546 | 1.27 (±2.35) | 0.59 (±0.49) | 0.726 | 0.221 | 0.302 |
| Vegetables | 0.66 (±1.02) | 0.1 (±0.23) | 0.052 | 0.4 (±0.51) | 0.95 (±0.79) | 0.028 | 0.002 | 0.806 |
| Type of Diet | High Histamine | Low Histamine | DRI |
|---|---|---|---|
| Sodium (mg) ** | 3200 | 1808 | 1500 |
| Potassium (mg) | 2922 | 2557 | 4500 |
| Calcium (mg) | 1365 | 807 | 1300 |
| Iron (mg) | 14.5 | 12 | 8 |
| Phosphorus (mg) | 1603 | 1208 | 1250 |
| Magnesium (mg) | 280 | 240 | 240 |
| Zinc (mg) ** | 22 | 8.9 | 8 |
| Copper (mg) | 1.3 | 1 | 0.7 |
| Manganese (mg) | 1.6 | 1.7 | 1.9 |
| Selenium (mg) | 0.13 | 0.1 | 0.04 |
| Fluoride (µg) | 223 | 300 | 200 |
| Chromium (mg) | 0.03 | 0.04 | 0.025 |
| Molybdenum (µg) | 43 | 49 | 34 |
| Histidine (mg) ** | 2400 | 1800 | NA |
| Vitamin A (RE) | 637 | 627 | 600 |
| Beta-carotene (ug) ** | 217 | 494 | NA |
| Vitamin C (mg) | 54 | 46 | 45 |
| Vitamin D (ug) | 4.5 | 4.46 | 15 |
| Vitamin E (IU) | 11.6 | 10.4 | 11 |
| Thiamin (mg) | 2 | 1.26 | 0.9 |
| Riboflavin (mg) | 2.4 | 1.67 | 0.9 |
| Niacin (mg) | 21.5 | 20.4 | 12 |
| Vit B6 (mg) | 1.65 | 1.5 | 1 |
| Vit B6 (mg) | 1.65 | 1.5 | 1 |
| Folate (µg) | 336 | 253 | 300 |
| Vit B12 (µg) | 4.98 | 3.8 | 1.8 |
| Biotin (µg) | 21.5 | 20.6 | 20 |
| Pantothenic Acid (mg) | 4.4 | 4 | 4 |
| Vitamin K (µg) | 30 | 40 | 60 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vassilopoulou, E.; Konstantinou, G.N.; Dimitriou, A.; Manios, Y.; Koumbi, L.; Papadopoulos, N.G. The Impact of Food Histamine Intake on Asthma Activity: A Pilot Study. Nutrients 2020, 12, 3402. https://doi.org/10.3390/nu12113402
Vassilopoulou E, Konstantinou GN, Dimitriou A, Manios Y, Koumbi L, Papadopoulos NG. The Impact of Food Histamine Intake on Asthma Activity: A Pilot Study. Nutrients. 2020; 12(11):3402. https://doi.org/10.3390/nu12113402
Chicago/Turabian StyleVassilopoulou, Emilia, George N. Konstantinou, Anastasia Dimitriou, Yannis Manios, Lemonica Koumbi, and Nikolaos G. Papadopoulos. 2020. "The Impact of Food Histamine Intake on Asthma Activity: A Pilot Study" Nutrients 12, no. 11: 3402. https://doi.org/10.3390/nu12113402
APA StyleVassilopoulou, E., Konstantinou, G. N., Dimitriou, A., Manios, Y., Koumbi, L., & Papadopoulos, N. G. (2020). The Impact of Food Histamine Intake on Asthma Activity: A Pilot Study. Nutrients, 12(11), 3402. https://doi.org/10.3390/nu12113402







