Efficacy and Safety of Kudzu Flower–Mandarin Peel on Hot Flashes and Bone Markers in Women during the Menopausal Transition: A Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Test Materials
2.2. Subjects
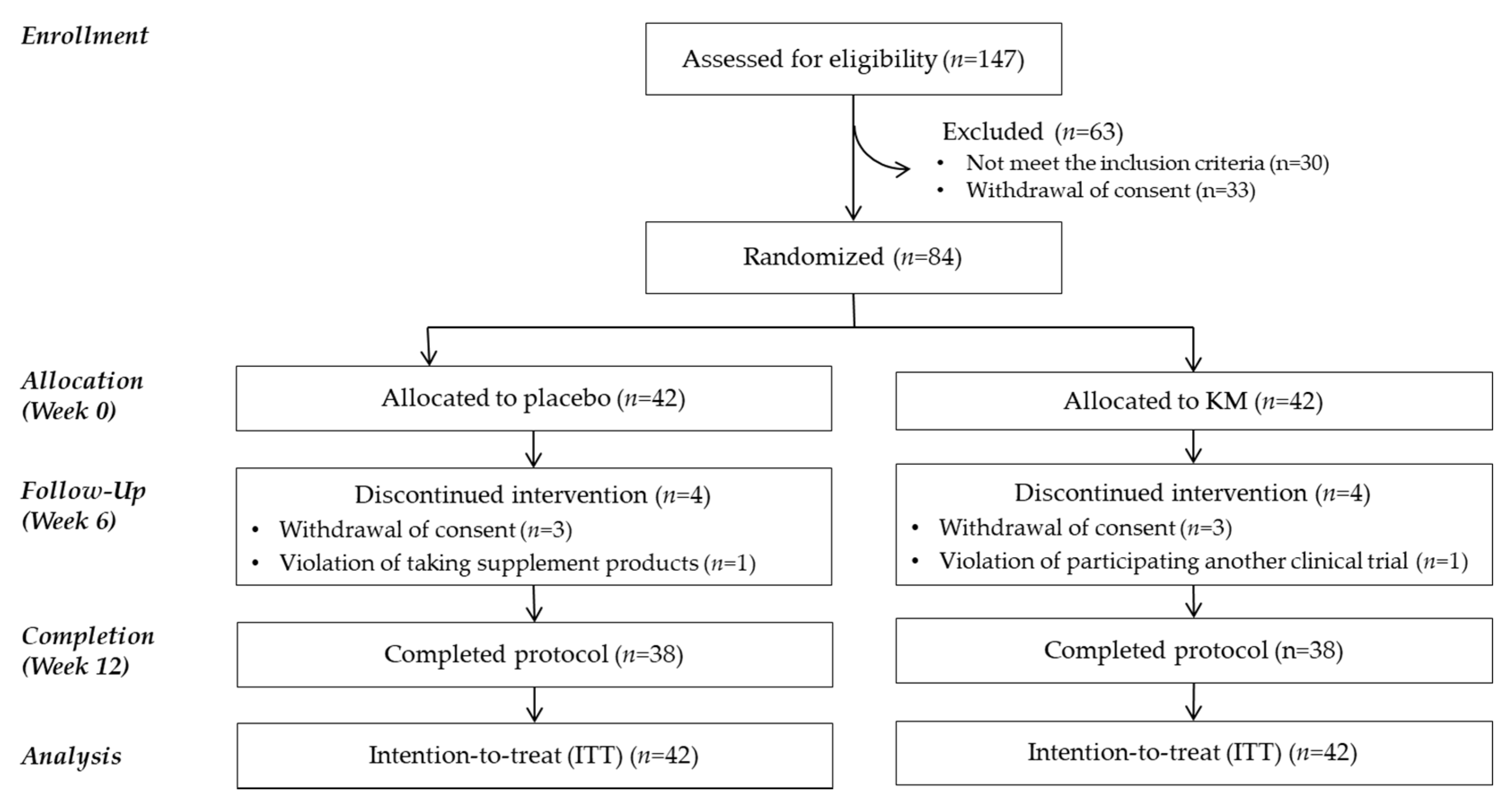
2.3. Study Design
2.4. Measurement of HFs
2.5. Measurement of Biochemical Markers in the Blood
2.6. Safety Measurements
2.7. Statistical Analysis
3. Results
3.1. Baseline Characteristics
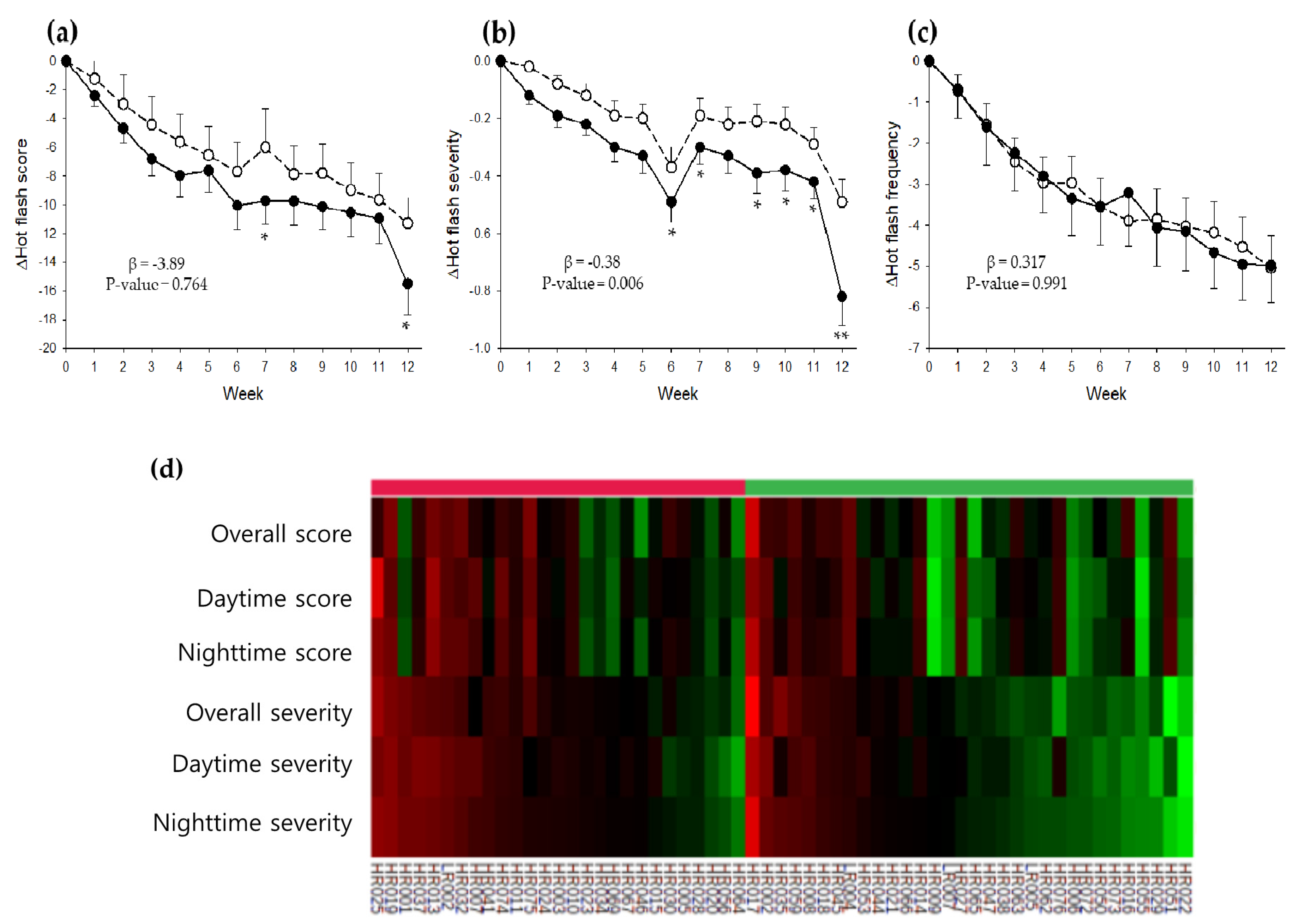
3.2. Effect of KM on HFs
3.3. Effect of KM on Bone Turnover Markers
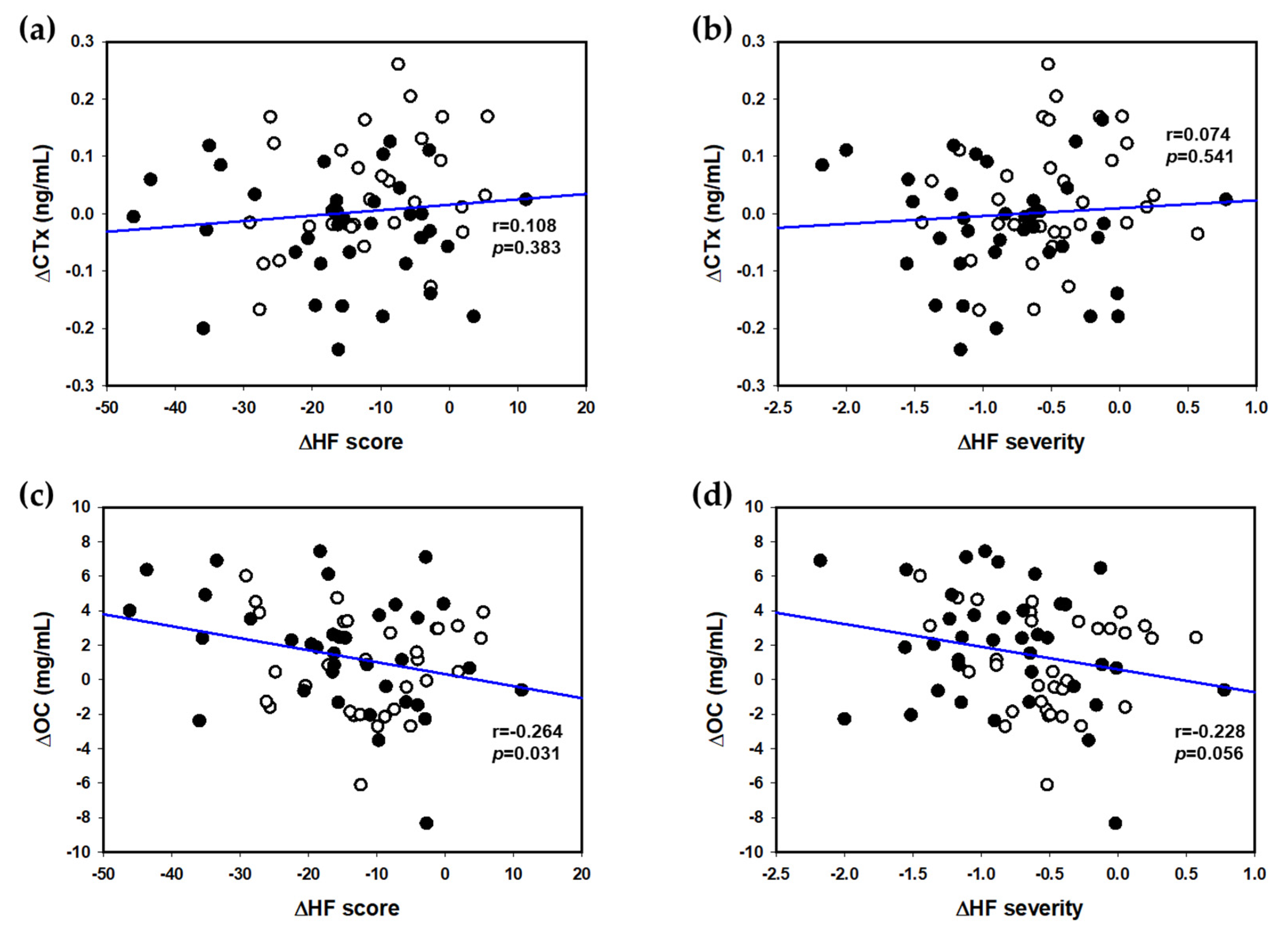
3.4. Associations between HF Scores and Bone Turnover Markers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dugan, S.A.; Powell, L.H.; Kravitz, H.M.; Everson Rose, S.A.; Karavolos, K.; Luborsky, J. Musculoskeletal pain and menopausal status. Clin. J. Pain 2006, 22, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Vousoura, E.; Spyropoulou, A.C.; Koundi, K.L.; Tzavara, C.; Verdeli, H.; Paparrigopoulos, T.; Augoulea, A.; Lambrinoudaki, I.; Zervas, I.M. Vasomotor and depression symptoms may be associated with different sleep disturbance patterns in postmenopausal women. Menopause 2015, 22, 1053–1057. [Google Scholar] [CrossRef] [PubMed]
- Utian, W.H. Psychosocial and socioeconomic burden of vasomotor symptoms in menopause: A comprehensive review. Health Qual. Life Outcomes 2005, 3, 47. [Google Scholar] [CrossRef] [PubMed]
- Nelson, H.D. Menopause. Lancet 2008, 371, 760–770. [Google Scholar] [CrossRef]
- Landgren, B.M.; Collins, A.; Csemiczky, G.; Burger, H.G.; Baksheev, L.; Robertson, D.M. Menopause transition: Annual changes in serum hormonal patterns over the menstrual cycle in women during a nine-year period prior to menopause. J. Clin. Endocr. Metab. 2004, 89, 2763–2769. [Google Scholar] [CrossRef]
- Deecher, D.C.; Dorries, K. Understanding the pathophysiology of vasomotor symptoms (hot flushes and night sweats) that occur in perimenopause, menopause, and postmenopause life stages. Arch. Women’s Ment. Health 2007, 10, 247–257. [Google Scholar] [CrossRef]
- Rodstrom, K.; Bengtsson, C.; Lissner, L.; Milsom, I.; Sundh, V.; Bjorkelund, C. A longitudinal study of the treatment of hot flushes: The population study of women in Gothenburg during a quarter of a century. Menopause J. N. Am. Menopause Soc. 2002, 9, 156–161. [Google Scholar] [CrossRef]
- Abdia, F.; Alimoradi, Z.; Haqi, P.; Mandizad, F. Effects of phytoestrogens on bone mineral density during the menopause transition: A systematic review of randomized, controlled trials. Climacteric 2016, 19, 535–545. [Google Scholar] [CrossRef]
- Crandall, C.J.; Zheng, Y.; Crawford, S.L.; Thurston, R.C.; Gold, E.B.; Johnston, J.M.; Greendale, G.A. Presence of vasomotor symptoms is mineral density: A longitudinal associated with lower bone analysis. Menopause J. N. Am. Menopause Soc. 2009, 16, 239–246. [Google Scholar] [CrossRef]
- Tuomikoski, P.; Ylikorkala, O.; Mikkola, T.S. Postmenopausal hot flushes and bone mineral density: A longitudinal study. Acta Obstet. Gyn. Scan. 2015, 94, 198–203. [Google Scholar] [CrossRef]
- Rosendaal, F.R.; Helmerhorst, F.M.; Vandenbroucke, J.P. Female hormones and thrombosis. Arter. Thromb. Vasc. Biol. 2002, 22, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Isoflavones Market by Source (Soy, Red Clover), Application (Pharmaceuticals, Nutraceuticals, Cosmetics, and Food & Beverages), Form (Powder and Liquid), and Region (North America, Europe, Asia Pacific, RoW)–Global Forecast to 2025. Available online: https://www.marketsandmarkets.com/Market-Reports/isoflavones-market-161667005.html (accessed on 22 October 2020).
- Wang, S.; Gong, T.; Lu, J.; Kano, Y.; Yuan, D. Simultaneous determination of tectorigenin and its metabolites in rat plasma by ultra performance liquid chromatography/quadrupole time-of-flight mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2013, 933, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Branka Levaj, V.D.-U.; Bursackovacevic, D.; Krasnici, N. Determination of flavonoids in pulp and peel of mandarin fruits. Agric. Conspec. Sci. 2009, 74, 221–225. [Google Scholar]
- Lim, D.W.; Lee, Y.; Kim, Y.T. Preventive effects of Citrus unshiu peel extracts on bone and lipid metabolism in OVX rats. Molecules 2014, 19, 783–794. [Google Scholar] [CrossRef]
- Chiba, H.; Uehara, M.; Wu, J.; Wang, X.; Masuyama, R.; Suzuki, K.; Kanazawa, K.; Ishimi, Y. Hesperidin, a citrus flavonoid, inhibits bone loss and decreases serum and hepatic lipids in ovariectomized mice. J. Nutr. 2003, 133, 1892–1897. [Google Scholar] [CrossRef] [PubMed]
- Han, N.R.; Nam, S.Y.; Hong, S.; Kim, H.Y.; Moon, P.D.; Kim, H.J.; Cho, H.; Lee, B.; Kim, H.M.; Jeong, H.J. Improvement effects of a mixed extract of flowers of Pueraria thomsonii Benth. and peels of Citrus unshiu Markovich on postmenopausal symptoms of ovariectomized mice. Biomed. Pharm. 2018, 103, 524–530. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.J.; Kim, M.H.; Kim, H.; Kim, H.Y.; Nam, S.Y.; Han, N.R.; Lee, B.; Cho, H.; Moon, P.D.; Kim, H.M. PCE17 and its active compounds exert an anti-osteoporotic effect through the regulation of receptor activator of nuclear factor-κB ligand in ovariectomized mice. J. Food Biochem. 2018, 42, e12561. [Google Scholar] [CrossRef]
- Fabricant, C.H.A.D. Pre-DSHEA List of Old Dietary Ingredients; Natural Products Association (NPA): Washington, DC, USA, 2017. [Google Scholar]
- Chang, A.; Kwak, B.Y.; Yi, K.; Kim, J.S. The effect of herbal extract (EstroG-100) on pre-, peri- and post-menopausal women: A randomized double-blind, placebo-controlled study. Phytother. Res. 2012, 26, 510–516. [Google Scholar] [CrossRef]
- Kim, S.Y.; Seo, S.K.; Choi, Y.M.; Jeon, Y.E.; Lim, K.J.; Cho, S.; Choi, Y.S.; Lee, B.S. Effects of red ginseng supplementation on menopausal symptoms and cardiovascular risk factors in postmenopausal women: A double-blind randomized controlled trial. Menopause 2012, 19, 461–466. [Google Scholar] [CrossRef]
- Zhou, X.; Han, D.; Xu, R.; Li, S.; Wu, H.; Qu, C.; Wang, F.; Wang, X.; Zhao, Y. A model of metabolic syndrome and related diseases with intestinal endotoxemia in rats fed a high fat and high sucrose diet. PLoS ONE 2014, 9, e115148. [Google Scholar] [CrossRef]
- Li, M.D.; Hung, A.; Lenon, G.B.; Yang, A.W.H. Chinese herbal formulae for the treatment of menopausal hot flushes: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0222383. [Google Scholar] [CrossRef]
- Committee, I.R. Guidelines for Data Processing and Analysis of the International Physical Activity Questionnaire (IPAQ)-Short and Long Forms. 2005. Available online: http://www.ipaq.ki.se/scoring.pdf (accessed on 22 October 2020).
- Buysse, D.J.; Reynolds, C.F., 3rd; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Kim, J.Y.; Yang, Y.J.; Yang, Y.K.; Oh, S.Y.; Hong, Y.C.; Lee, E.K.; Kwon, O. Diet quality scores and oxidative stress in Korean adults. Eur. J. Clin. Nutr. 2011, 65, 1271–1278. [Google Scholar] [CrossRef] [PubMed]
- Sloan, J.A.; Loprinzi, C.L.; Novotny, P.J.; Barton, D.L.; Lavasseur, B.I.; Windschitl, H. Methodologic lessons learned from hot flash studies. J. Clin. Oncol. 2001, 19, 4280–4290. [Google Scholar] [CrossRef]
- Fisher, W.I.; Thurston, R.C. Measuring hot flash phenomenonology using ambulatory prospective digital diaries. Menopause 2016, 23, 1222–1227. [Google Scholar] [CrossRef]
- Hilditch, J.R.; Lewis, J.; Peter, A.; van Maris, B.; Ross, A.; Franssen, E.; Guyatt, G.H.; Norton, P.G.; Dunn, E. A menopause-specific quality of life questionnaire: Development and psychometric properties. Maturitas 1996, 24, 161–175. [Google Scholar] [CrossRef]
- Zhu, D.; Li, X.; Macrae, V.E.; Simoncini, T.; Fu, X. Extragonadal Effects of Follicle-Stimulating Hormone on Osteoporosis and Cardiovascular Disease in Women during Menopausal Transition. Trends Endocrinol. Metab. 2018, 29, 571–580. [Google Scholar] [CrossRef]
- Lee, J.S.; Ettinger, B.; Stanczyk, F.Z.; Vittinghoff, E.; Hanes, V.; Cauley, J.A.; Chandler, W.; Settlage, J.; Beattie, M.S.; Folkerd, E.; et al. Comparison of methods to measure low serum estradiol levels in postmenopausal women. J. Clin. Endocrinol. Metab. 2006, 91, 3791–3797. [Google Scholar] [CrossRef]
- Herrmann, M.; Seibel, M.J.; Seibel, M. The amino- and carboxyterminal cross-linked telopeptides of collagen type I, NTX-I and CTX-I: A comparative review. Clin. Chim. Acta 2008, 393, 57–75. [Google Scholar] [CrossRef] [PubMed]
- Shetty, S.; Kapoor, N.; Bondu, J.D.; Thomas, N.; Paul, T.V. Bone turnover markers: Emerging tool in the management of osteoporosis. Indian J. Endocrinol. Metab. 2016, 20, 846–852. [Google Scholar] [CrossRef]
- Lu, L.J.W.; Anderson, K.E.; Grady, J.J.; Kohen, F.; Nagamani, M. Decreased ovarian hormones during a soya diet: Implications for breast cancer prevention. Cancer Res. 2000, 60, 4112–4121. [Google Scholar] [PubMed]
- Kim, J.H.; Woo, J.H.; Kim, H.M.; Oh, M.S.; Jang, D.S.; Choi, J.H. Anti-Endometriotic Effects of Pueraria Flower Extract in Human Endometriotic Cells and Mice. Nutrients 2017, 9, 212. [Google Scholar] [CrossRef] [PubMed]
- Sternlicht, M.D.; Werb, Z. How matrix metalloproteinases regulate cell behavior. Annu. Rev. Cell Dev. Biol. 2001, 17, 463–516. [Google Scholar] [CrossRef] [PubMed]
- Perez, D.G.; Loprinzi, C.L.; Barton, D.L.; Pockaj, B.A.; Sloan, J.; Novotny, P.J.; Christensen, B.J. Pilot evaluation of mirtazapine for the treatment of hot flashes. J. Support Oncol. 2004, 2, 50–56. [Google Scholar]
- Carroll, D.G. Nonhormonal therapies for hot flashes in menopause. Am. Fam. Physician 2006, 73, 457–464. [Google Scholar]
- Kamiya, T.; Nagamine, R.; Sameshima-Kamiya, M.; Tsubata, M.; Ikeguchi, M.; Takagaki, K. The isoflavone-rich fraction of the crude extract of the Puerariae flower increases oxygen consumption and BAT UCP1 expression in high-fat diet-fed mice. Glob. J. Health Sci. 2012, 4, 147–155. [Google Scholar] [CrossRef]
- Simon, J.A.; Kaunitz, A.M.; Kroll, R.; Graham, S.; Bernick, B.; Mirkin, S. Oral 17β-estradiol/progesterone (TX-001HR) and quality of life in postmenopausal women with vasomotor symptoms. Menopause 2019, 26, 506–512. [Google Scholar] [CrossRef]
- Thurston, R.C.; El Khoudary, S.R.; Sutton-Tyrrell, K.; Crandall, C.J.; Gold, E.B.; Sternfeld, B.; Joffe, H.; Selzer, F.; Matthews, K.A. Vasomotor symptoms and lipid profiles in women transitioning through menopause. Obstet. Gynecol. 2012, 119, 753–761. [Google Scholar] [CrossRef]
- Massé, P.G.; Dosy, J.; Jougleux, J.L.; Caissie, M.; Howell, D.S. Bone mineral density and metabolism at an early stage of menopause when estrogen and calcium supplement are not used and without the interference of major confounding variables. J. Am. Coll. Nutr. 2005, 24, 354–360. [Google Scholar] [CrossRef]
- Bezerra, M.C.; Carvalho, J.F.; Prokopowitsch, A.S.; Pereira, R.M. RANK, RANKL and osteoprotegerin in arthritic bone loss. Braz. J. Med. Biol. Res. 2005, 38, 161–170. [Google Scholar] [CrossRef]
- Ma, C.Y.; Xu, K.; Meng, J.H.; Ran, J.S.; Moqbel, S.A.A.; Liu, A.; Yan, S.G.; Wu, L.D. Tectorigenin inhibits RANKL-induced osteoclastogenesis via suppression of NF-kappa B signalling and decreases bone loss in ovariectomized C57BL/6. J. Cell. Mol. Med. 2018, 22, 5121–5131. [Google Scholar] [CrossRef]
- Wang, J.; Tang, Y.; Lv, X.; Zhang, J.; Ma, B.; Wen, X.; Bao, Y.; Wang, G. Tectoridin inhibits osteoclastogenesis and bone loss in a murine model of ovariectomy-induced osteoporosis. Exp. Gerontol. 2020, 111057. [Google Scholar] [CrossRef] [PubMed]
- Horcajada, M.N.; Habauzit, V.; Trzeciakiewicz, A.; Morand, C.; Gil-Izquierdo, A.; Mardon, J.; Lebecque, P.; Davicco, M.J.; Chee, W.S.S.; Coxam, V.; et al. Hesperidin inhibits ovariectomized-induced osteopenia and shows differential effects on bone mass and strength in young and adult intact rats. J. Appl. Physiol. 2008, 104, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Salamone, L.M.; Gregg, E.; Wolf, R.L.; Epstein, R.S.; Black, D.; Palermo, L.; Kuller, L.H.; Cauley, J.A. Are menopausal symptoms associated with bone mineral density and changes in bone mineral density in premenopausal women? Maturitas 1998, 29, 179–187. [Google Scholar] [CrossRef]
- Lee, S.J.; Kanis, J.A. An association between osteoporosis and premenstrual symptoms and postmenopausal symptoms. Bone Miner. 1994, 24, 127–134. [Google Scholar] [CrossRef]
- Scoutellas, V.; O’Neill, T.W.; Lunt, M.; Reeve, J.; Silman, A.J. Does the presence of postmenopausal symptoms influence susceptibility to vertebral deformity? European Vertebral Osteoporosis Study (EVOS) Group. Maturitas 1999, 32, 179–187. [Google Scholar] [CrossRef]
- Pal, L.; Bevilacqua, K.; Zeitlian, G.; Shu, J.; Santoro, N. Implications of diminished ovarian reserve (DOR) extend well beyond reproductive concerns. Menopause 2008, 15, 1086–1094. [Google Scholar] [CrossRef]
- Woods, N.F.; Mitchell, E.S. Symptoms during the perimenopause: Prevalence, severity, trajectory, and significance in women’s lives. Am. J. Med. 2005, 118 (Suppl. 2), 14–24. [Google Scholar] [CrossRef]
- Shams, T.; Firwana, B.; Habib, F.; Alshahrani, A.; Alnouh, B.; Murad, M.H.; Ferwana, M. SSRIs for hot flashes: A systematic review and meta-analysis of randomized trials. J. Gen. Intern. Med. 2014, 29, 204–213. [Google Scholar] [CrossRef]
| Variables | Placebo (n = 42) | KM (n = 42) | p-Value |
|---|---|---|---|
| Age (years) | 52.0 ± 0.6 | 51.5 ± 0.5 | 0.526 |
| Amenorrhea period (months) | 29.8 ± 4.5 | 20.7 ± 2.9 | 0.096 |
| Hot flash score | 34.8 ± 6.7 | 27.8 ±3.3 | 0.354 |
| Kupperman Index | 25.2 ± 1.3 | 24.1 ± 1.3 | 0.532 |
| Alcohol drinker (Y/N) | 21/21 | 19/23 | 0.662 |
| Alcohol amount (g/week) | 8.0 ± 2.2 | 6.5 ± 1.8 | 0.598 |
| Smoker (Y/N) | 0/42 | 1/41 | 1.000 |
| Smoking amount (cigarettes/day) | 0.0 ± 0.0 | 0.1 ± 0.1 | 0.183 |
| Body weight (kg) | 59.8 ± 1.5 | 57.7 ± 1.2 | 0.268 |
| BMI (kg/m2) | 23.7 ± 0.5 | 23.1 ± 0.5 | 0.352 |
| Waist circumference (cm) | 83.0 ± 1.6 | 79.1 ± 1.3 | 0.059 |
| Physical activity (MET-min/week) | 1499.5 ± 148.7 | 1905.2 ± 251.7 | 0.170 |
| Vigorous activity (h/week) | 0.4 ± 0.2 | 0.2 ± 0.1 | 0.435 |
| RFS | 23.0 ± 1.2 | 27.0 ± 1.3 | 0.028 |
| SBP (mmHg) | 118.2 ± 1.6 | 118.2 ± 2.1 | 0.979 |
| DBP (mmHg) | 73.2 ± 1.4 | 72.7 ± 1.6 | 0.807 |
| Variables | Placebo (n = 42) | KM (n = 42) | Estimate | p-Value | |
|---|---|---|---|---|---|
| Retrospective HF scores | |||||
| KI | Week 0 | 6.86 ± 0.44 | 5.24 ± 0.37 | 0.83 | 0.191 |
| Week 12 | 4.63 ± 0.47 | 4.21 ± 0.4 | |||
| p-value | <0.001 | 0.013 | |||
| MenQOL score | Week 0 | 15.17 ± 0.7 | 13.64 ± 0.64 | 0.555 | 0.163 |
| Week 12 | 9.97 ± 0.79 | 10.26 ± 0.75 | |||
| p-value | <0.001 | <0.001 | |||
| Safety parameters | |||||
| Endometrial thickness (mm) | Week 0 | 5.4 ± 0.4 | 5.4 ± 0.6 | 0.52 | 0.380 |
| Week 12 | 5.0 ± 0.4 | 5.6 ± 0.5 | |||
| p-value | 0.14 | 0.793 | |||
| FSH (mIU/mL) | Week 0 | 63.4 ± 5.8 | 71.0 ± 6.5 | −10.81 | 0.037 |
| Week 12 | 68.4 ± 5.5 | 66.2 ± 7.5 | |||
| p-value | 0.062 | 0.271 | |||
| Estradiol (pg/mL) | Week 0 | 42.1 ± 12.8 | 37.5 ± 11.6 | 46.23 | 0.043 |
| Week 12 | 23.6 ± 8.4 | 67.8 ± 19.1 | |||
| p-value | 0.192 | 0.115 | |||
| LH (mIU/mL) | Week 0 | 34.7 ± 2.8 | 35.9 ± 3.1 | −0.09 | 0.986 |
| Week 12 | 38.3 ± 2.7 | 40.1 ± 4.5 | |||
| p-value | 0.27 | 0.267 | |||
| Variables | Placebo (n = 42) | KM (n = 42) | Estimate | p-Value | |
|---|---|---|---|---|---|
| Markers of bone resorption | |||||
| CTx (ng/mL) | Week 0 | 0.43 ± 0.03 | 0.43 ± 0.03 | −0.05 | 0.027 |
| Week 12 | 0.47 ± 0.03 | 0.39 ± 0.03 | |||
| p-value | 0.098 | 0.134 | |||
| NTx (ng/mL) | Week 0 | 3.50 ± 0.32 | 3.94 ± 0.45 | −0.80 | 0.25 |
| Week 12 | 3.53 ± 0.36 | 3.06 ± 0.23 | |||
| p-value | 0.922 | 0.124 | |||
| Markers of bone formation | |||||
| OC (mg/mL) | Week 0 | 18.35 ± 0.92 | 17.64 ± 0.98 | 1.03 | 0.17 |
| Week 12 | 19.49 ± 0.87 | 19.48 ± 1.12 | |||
| p-value | 0.124 | 0.001 | |||
| BALP (mg/mL) | Week 0 | 15.21 ± 0.73 | 13.48 ± 0.73 | 0.67 | 0.276 |
| Week 12 | 14.73 ± 0.70 | 13.53 ± 0.84 | |||
| p-value | 0.443 | 0.435 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.E.; Jeong, H.; Hur, S.; Lee, J.; Kwon, O. Efficacy and Safety of Kudzu Flower–Mandarin Peel on Hot Flashes and Bone Markers in Women during the Menopausal Transition: A Randomized Controlled Trial. Nutrients 2020, 12, 3237. https://doi.org/10.3390/nu12113237
Kim JE, Jeong H, Hur S, Lee J, Kwon O. Efficacy and Safety of Kudzu Flower–Mandarin Peel on Hot Flashes and Bone Markers in Women during the Menopausal Transition: A Randomized Controlled Trial. Nutrients. 2020; 12(11):3237. https://doi.org/10.3390/nu12113237
Chicago/Turabian StyleKim, Ji Eon, Hyeyun Jeong, Soohee Hur, Junho Lee, and Oran Kwon. 2020. "Efficacy and Safety of Kudzu Flower–Mandarin Peel on Hot Flashes and Bone Markers in Women during the Menopausal Transition: A Randomized Controlled Trial" Nutrients 12, no. 11: 3237. https://doi.org/10.3390/nu12113237
APA StyleKim, J. E., Jeong, H., Hur, S., Lee, J., & Kwon, O. (2020). Efficacy and Safety of Kudzu Flower–Mandarin Peel on Hot Flashes and Bone Markers in Women during the Menopausal Transition: A Randomized Controlled Trial. Nutrients, 12(11), 3237. https://doi.org/10.3390/nu12113237






