Kava as a Clinical Nutrient: Promises and Challenges
Abstract
1. Introduction
2. Kava and Its Diversity
2.1. Different Forms of Kava
2.2. Kava Diversity
2.2.1. Cultivars
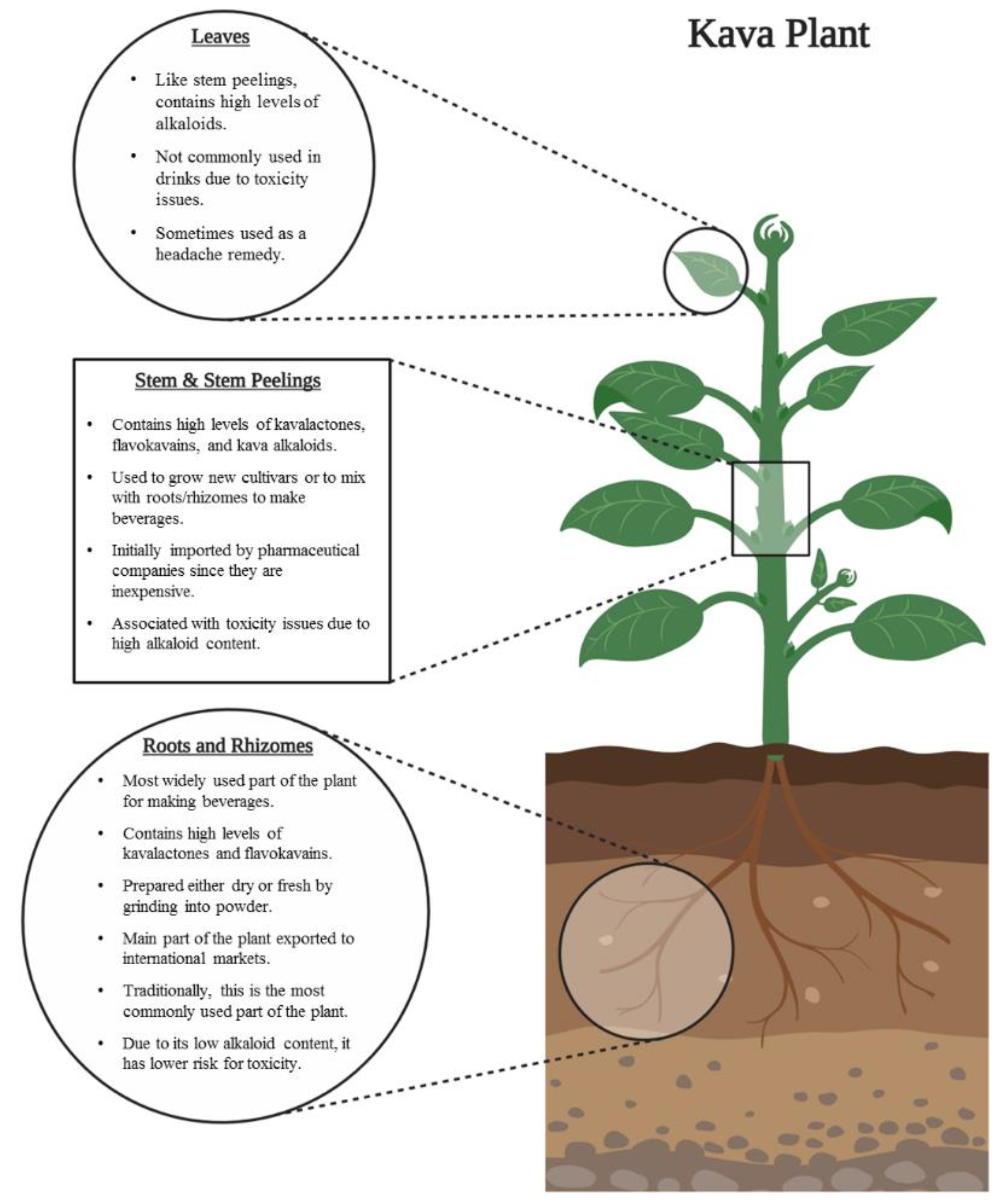
2.2.2. Plant Parts
2.2.3. Extraction Solvents
2.3. Kava Standardization
3. Regulation of the Inflammatory Responses by Kava and Its Components
3.1. Effects of Kava Components on Inflammation
| Component Used | Inflammation-Related Pharmacological Effects | References |
|---|---|---|
| Whole kava extract |
| [60,61] |
| Yangonin |
| [53,54,55] |
| Desmethoxyyangonin |
| [56] |
| Kavain |
| [51,53,62,63,64,65] |
| Dihydrokavain |
| [51] |
| Methysticin |
| [51,52,53,65] |
| Dihydromethysticin |
| [51] |
| Flavokavains |
| [57,58,59] |
3.2. Improving the Anti-Inflammatory Effects via Chemical Modifications
4. Neurological Functions of Kava and Their Mechanisms
4.1. Neuropharmacological Functions of Kava and Potential Mechanisms.
| Component Used | Neuropharmacological Effects and Mechanisms | References |
|---|---|---|
| Whole kava extract |
| [47,87,91] |
| Yangonin |
| [52,53,87,89,92] |
| Desmethoxyyangonin |
| [89,93] |
| Kavain |
| [47,52,53,82,86,87,89,90,94] |
| Dihydrokavain |
| [47,87] |
| Methysticin |
| [47,52,53,82] |
| Dihydromethysticin |
| [47,90] |
4.2. Clinical Evidence of Kava’s Neuropharmacological Effects
5. Kava and Cancer
5.1. Cancer Chemoprevention
5.1.1. Lung Cancer
5.1.2. Other Cancers
| Components | Cancer Type | Mechanism | Model | Reference |
|---|---|---|---|---|
| Kava | Lung cancer | Reduction in NNK induced DNA damage | A/J mice | [107] |
| Kavalactone-rich fraction | Lung cancer | Reduction in NNK induced DNA damage | A/J mice | [108] |
| Prostate cancer | Inhibition of angiogenesis and cell proliferation genes and upregulation of antitumor genes, immunity, muscle/neuro, and metabolism-related genes | Male C57BL/6J and female C57BL/6-Tg TRAMP 8247Ng/J mice | [116] | |
| Nonpolar extract | Colon cancer | Reduction in precancerous lesions | Rat | [115] |
| DHM | Lung cancer | Reduction in NNK induced DNA damage and NNAL detoxification | A/J mice | [33,109,111] |
| lung cancer | Reduction in NNK induced DNA damage | C57BL/6 Female Mice | [110] | |
| Flavokavain A | Urothelial cancer | Induction of apoptosis | UPII-SV40T mice | [113] |
| Prostate cancer | Inhibition of proliferation and induction of apoptosis | Female hemizygous C57BL/TGN TRAMP mice and male C57BL/6 mice | [114] |
5.2. Anticancer Activities of Kava and Its Components
5.2.1. Flavokavain A
5.2.2. Flavokavain B
5.2.3. Kavalactones
| Components | Cancer Type | Mechanism | Model | Reference |
|---|---|---|---|---|
| Water extract | Breast and colon cancer | Inhibition of proliferation | In vitro | [117] |
| kava root extract and flavokavain B | Prostate cancer | Downregulation of AR | Patient-derived prostate cancer xenografts in mice | [118] |
| Flavokavain A | Lung cancer | Anti-proliferation and induction of apoptosis, downregulation of P-gp | In vitro | [119] |
| Breast cancer | Induction of apoptosis, inhibition of metastasis and G2/M cell cycle arrest | In vitro | [15] | |
| Inducing G2/M cell cycle arrest and enhancing the activity of Herceptin | In vitro | [125] | ||
| enhancing antitumor immunity and inhibition of inflammation | Breast cancer-challenged mice | [124] | ||
| Bladder cancer | Inducing G2/M cell cycle arrest | In vitro | [122] | |
| Induction of apoptosis and inhibition of proliferation | In vitro and xenograft mouse model | [121] | ||
| Prostate cancer | Induction of G2/M cell cycle arrest and apoptosis and regulation of glutamine metabolism | In vitro | [123] | |
| Osteosarcoma | Inhibition of invasion through downregulation of SK | In vitro and osteosarcoma xenograft model | [120] | |
| Flavokavain B | Lung cancer | Induction of G2/M cell cycle arrest and apoptosis | In vitro | [132] |
| Induction of apoptosis and autophagy | In vitro | [139] | ||
| Induction of apoptosis and inhibition of migration and invasion | In vitro | [129] | ||
| Breast cancer | Induction of G2/M cell cycle arrest and inhibition of metastasis and angiogenesis | In vitro | [128] | |
| Induction of apoptosis and regulation of immune system | Xenograft model | [143] | ||
| SAR study | In vitro | [137] | ||
| Colon cancer | Induction of G2/M cell cycle arrest and apoptosis | In vitro | [134] | |
| Induction of G0/G1 cell cycle arrest | In vitro | [138] | ||
| Gastric cancer | Induction of autophagy | AGS-xenografted mice | [140] | |
| Thyroid cancer | Inhibition of cell proliferation, migration and invasion and induction of apoptosis and autophagy | In vitro | [142] | |
| Oral cancer | Induction of G2/M cell cycle arrest and apoptosis | In vitro | [136] | |
| Squamous carcinoma | Inhibition of proliferation and induction of apoptosis and G2/M cell cycle arrest | In vitro and xenograft mouse model | [135] | |
| Synovial sarcomas | Induction of apoptosis | In vitro | [133] | |
| Glioblastoma multiforme | Induction of autophagy | In vitro and intracranial xenograft model | [141] | |
| Cervical cancer | Induction of p21-mediated cell cycle arrest | In vitro | [144] | |
| Osteosarcoma | Inhibition of cell proliferation and induction of apoptosis and G2/M cell cycle arrest | In vitro | [131] | |
| Brain endothelial cell | Inhibition of angiogenesis | In vitro and zebrafish | [130] | |
| Prostate cancer | Enhancing the activity of bortezomib through promoting Skp2 degradation | In vitro | [145] | |
| Downregulation of AR | Patient-derived prostate cancer xenograft model | [118] | ||
| Acute myeloid leukemia | Induction of apoptosis and promoting the potency of daunorubicin via activation of NF-κB | In vitro | [146] | |
| dihydromethysticin | Osteosarcoma | Induction of apoptosis and cell cycle arrest | In vitro | [147] |
| Colorectal cancer | Inhibition of proliferation, migration, and invasion and induction of apoptosis and cell cycle arrest | In vitro and xenograft model | [148] | |
| Yangonin | Bladder cancer | Induction of autophagy | In vitro | [149] |
| Crude extract and kavalactones | Mouse leukemia | Inhibition of P-gp | In vitro | [150] |
6. Safety of Kava and Its Hepatotoxic Risk
6.1. Kava Safety in Lab Animals
6.2. Safety Data of Traditional Kava
6.3. Safety of Kava as an Herbal Anxiolytic Drug
6.4. Safety of Kava in Recent Clinical Trials
6.5. Kava as a Dietary Supplement in the U.S.
6.6. Safety Issues of Kava with No or Limited Knowledge of Composition
6.7. Safety of Individual Kava Compounds
7. Conclusions and Future Directions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| WHO | World Health Organization |
| GC-MS | Gas Chromatography Mass Spectrometry |
| HPLC | High-performance liquid chromatography |
| TLC | Thin-Layer Chromatography |
| LC-MS | Liquid Chromatography–Mass Spectrometry |
| ROS | Reactive Oxygen Species |
| IL | Interleukin |
| TNF | Tumor Necrosis Factor |
| LPS | Lipopolysaccharide |
| NOS | Nitric Oxide Synthase |
| COX-2 | Cyclooxygenase 2 |
| NO | Nitric Oxide |
| PGE-2 | Prostaglandin E2 |
| NF-κB | Nuclear Factor kappa B |
| AP-1 | Activator Protein 1 |
| JNK | c-Jun N-terminal Kinase |
| MAPK | Mitogen-Activated Protein Kinase |
| FXR | Farnesoid X Receptor |
| Nrf2 | Nuclear Factor Erythroid 2-related Factor 2 |
| D-GaIN | D-galactosamine |
| AchE | Acetylcholinesterase |
| MAO | Monoamine oxidase |
| 5-HT | Serotonin |
| NMDA | N-Methyl-D-aspartate |
| GABA | γ-aminobutyric Acid |
| GAD | Generalized Anxiety Disorder |
| SSRI | Selective Serotonin Reuptake Inhibitor |
| NNK | 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone |
| B[a]P | Benzo[a]pyrene |
| DHM | Dihydromethysticin |
| DHK | Dihydrokavain |
| AhR | Aryl Hydrocarbon Receptor |
| NNAL | 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol |
| CYP2A5 | Cytochrome P450 2A5 |
| UCC | Urothelial cell carcinoma |
| TRAMP | Transgenic Adenocarcinoma of Mouse Prostate |
| Skp2 | S phase Kinase-associated Protein 2 |
| NECa | Neuroendocrine Carcinomas |
| AR | Androgen Receptor |
| NSCLC | Non-Small Cell Lung Cancer |
| UCK2 | Uridine-Cytidine Kinase 2 |
| XIAP | X-Linked Inhibitor of Apoptosis |
| FDA | Food and Drug Administration |
| ALT | Alanine Transaminase |
| AST | Aspartate transaminase |
| GGT | Gamma-glutamyltransferase |
| NTP | National Toxicology Program |
| BZD | Benzodiazepine |
| CIOMS | Council for International Organizations of Medical Sciences |
| ALP | Alkaline phosphatase |
| CAERS | CFSAN Adverse Event Reporting System |
| ADME | Absorption, distribution, metabolism, and excretion |
| CFSAN | Center for Food Safety and Applied Nutrition |
| QC | Quality control |
References
- Ulbricht, C.; Basch, E.; Boon, H.; Ernst, E.; Hammerness, P.; Sollars, D.; Tsourounis, C.; Woods, J.; Bent, S. Safety review of kava (Piper methysticum) by the Natural Standard Research Collaboration. J. Expert Opin. Drug Saf. 2005, 4, 779–794. [Google Scholar] [CrossRef] [PubMed]
- Lebot, V. Kava (Piper methysticum Forst. f.): The Polynesian dispersal of an Oceanian plant. In Islands, Plants, and Polynesians: An Introduction to Polynesian Ethnobotany; Dioscorides Press: Portland, OR, USA, 1991. [Google Scholar]
- Lebot, V.; Merlin, M.; Lindstrom, L. Kava: The Pacific Elixir: The Definitive Guide to Its Ethnobotany, History, and Chemistry; Inner Traditions/Bear & Co.: Rochester, VT, USA, 1997. [Google Scholar]
- Rivers, Z.; Xing, C.; Narayanapillai, S.J.M.C. Kava as a pharmacotherapy of anxiety disorders: Promises and concerns. J. Med. Chem. 2016, 6, 81–87. [Google Scholar] [CrossRef]
- Martin, A.C.; Johnston, C.X.; Hegeman, A.D. Measuring the chemical and cytotoxic variability of commercially available kava (Piper methysticum G. Forster). PLoS ONE 2014, 9, e111572. [Google Scholar] [CrossRef] [PubMed]
- December 2019 Highlights: International Merchandise Trade Statistics. Available online: https://vnso.gov.vu/index.php/economic-statistics/trade-news (accessed on 7 June 2020).
- Foreign Trade Report—The Central Bank of Samoa. Available online: https://cbs.gov.ws/index.php/statistics/foreign-trade-report/ (accessed on 7 June 2020).
- International Trade Statistics—Fiji Bureau of Statistics. Available online: https://www.statsfiji.gov.fj/index.php/statistics/economic-statistics/merchandise-trade-statistics (accessed on 7 June 2020).
- Foreign Trade—Tonga Statistics Department. Available online: https://tongastats.gov.to/statistics/economics/foreign-trade/#75-136-wpfd-2019 (accessed on 7 June 2020).
- Mohanty, M. Fiji Kava: Production, trade, role and challenges. J. Pac. Stud. 2017, 37, 5–30. [Google Scholar]
- Singh, Y.N.; Singh, N.N. Therapeutic potential of kava in the treatment of anxiety disorders. CNS Drugs 2002, 16, 731–743. [Google Scholar] [CrossRef]
- Singh, Y.N. Kava: An old drug in a new world. Cult. Crit. 2009, 71, 107–128. [Google Scholar] [CrossRef]
- Wang, J.; Qu, W.; Bittenbender, H.C.; Li, Q.X. Kavalactone content and chemotype of kava beverages prepared from roots and rhizomes of Isa and Mahakea varieties and extraction efficiency of kavalactones using different solvents. J. Food Sci. Technol. 2015, 52, 1164–1169. [Google Scholar] [CrossRef]
- Sarris, J.; LaPorte, E.; Schweitzer, I. Kava: A comprehensive review of efficacy, safety, and psychopharmacology. Aust. N. Z. J. Psychiatry 2011, 45, 27–35. [Google Scholar] [CrossRef]
- WHO. Kava: A Review of the Safety of Tradtional and Recreational Beverage Consumption; Food and Agriculture Organization of the United Nation: Rome, Italy, 2016; Volume 1, pp. 1–35. [Google Scholar]
- Aporosa, A.S.; Atkins, M.; Brunton, R. Kava drinking in traditional settings: Towards understanding effects on cognitive function. Hum. Psychopharmacol. 2020, 35, e2725. [Google Scholar] [CrossRef]
- Balick, M.J.; Lee, R. Traditional use of sakau (kava) in Pohnpei: Lessons for integrative medicine. Altern. Ther. Health Med. 2002, 8, 96–98. [Google Scholar]
- Showman, A.F.; Baker, J.D.; Linares, C.; Naeole, C.K.; Borris, R.; Johnston, E.; Konanui, J.; Turner, H. Contemporary Pacific and Western perspectives onawa (Piper methysticum) toxicology. Fitoterapia 2015, 100, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Pittler, M.H.; Ernst, E. Efficacy of kava extract for treating anxiety: Systematic review and meta-analysis. J. Clin. Psychopharmacol. 2000, 20, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Geier, F.; Konstantinowicz, T. Kava treatment in patients with anxiety. Phytother. Res. 2004, 18, 297–300. [Google Scholar] [CrossRef] [PubMed]
- Kuchta, K.; de Nicola, P.; Schmidt, M. Randomized, dose-controlled double-blind trial: Efficacy of an ethanolic kava (Piper methysticum rhizome) extract for the treatment of anxiety in elderly patients. Tradit. Kampo Med. 2018, 5, 3–10. [Google Scholar] [CrossRef]
- Sarris, J.; Kavanagh, D.J.; Byrne, G.; Bone, K.; Adams, J.; Deed, G. The Kava Anxiety Depression Spectrum Study (KADSS): A randomized, placebo-controlled crossover trial using an aqueous extract of Piper methysticum. Psychopharmacology 2009, 205, 399–407. [Google Scholar] [CrossRef]
- Sarris, J.; Stough, C.; Teschke, R.; Wahid, Z.T.; Bousman, C.A.; Murray, G.; Savage, K.M.; Mouatt, P.; Ng, C.; Schweitzer, I. Kava for the treatment of generalized anxiety disorder RCT: Analysis of adverse reactions, liver function, addiction, and sexual effects. Phytother. Res. 2013, 27, 1723–1728. [Google Scholar] [CrossRef]
- Pittler, M.H.; Ernst, E. Kava extract for treating anxiety. Cochrane Database Syst. Rev. 2003, 20, 84–89. [Google Scholar] [CrossRef]
- Witte, S.; Loew, D.; Gaus, W. Meta-analysis of the efficacy of the acetonic kava-kava extract WS® 1490 in patients with non-psychotic anxiety disorders. Phytother. Res. 2005, 19, 183–188. [Google Scholar] [CrossRef]
- Baker, J.D. Pills, potions, products: Kava’s transformations in new and nontraditional contexts. Contemp. Pac. 2012, 24, 233–265. [Google Scholar] [CrossRef][Green Version]
- Kuchta, K.; Schmidt, M.; Nahrstedt, A. German Kava Ban Lifted by Court: The Alleged Hepatotoxicity of Kava (Piper methysticum) as a Case of Ill-Defined Herbal Drug Identity, Lacking Quality Control, and Misguided Regulatory Politics. Planta Med. 2015, 81, 1647–1653. [Google Scholar] [CrossRef]
- Hu, Q.; Corral, P.; Narayanapillai, S.C.; Leitzman, P.; Upadhyaya, P.; O’Sullivan, M.G.; Hecht, S.S.; Lu, J.; Xing, C. Oral dosing of dihydromethysticin ahead of tobacco carcinogen NNK effectively prevents lung tumorigenesis in A/J mice. Chem. Res. Toxicol. 2020, 33, 1980–1988. [Google Scholar] [CrossRef] [PubMed]
- Parkman, C.A. Another FDA warning: Kava supplements. Case Manag. 2002, 13, 26–28. [Google Scholar] [CrossRef] [PubMed]
- Wolinski, C. This Ancient South Pacific Sipper Is Officially Trending. Available online: https://vinepair.com/articles/what-kava-kava-drink/ (accessed on 7 June 2020).
- Solomon, S. Counting on the Trendy to Revive Kava, a Traditional Drink. The New York Times, 24 February 2017. [Google Scholar]
- Olsen, L.R.; Grillo, M.P.; Skonberg, C. Constituents in Kava Extracts Potentially Involved in Hepatotoxicity: A Review. Chem. Res. Toxicol. 2011, 24, 992–1002. [Google Scholar] [CrossRef] [PubMed]
- Narayanapillai, S.C.; Balbo, S.; Leitzman, P.; Grill, A.E.; Upadhyaya, P.; Shaik, A.A.; Zhou, B.; O’Sullivan, M.G.; Peterson, L.A.; Lu, J.; et al. Dihydromethysticin from kava blocks tobacco carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone-induced lung tumorigenesis and differentially reduces DNA damage in A/J mice. Carcinogenesis 2014, 35, 2365–2372. [Google Scholar] [CrossRef] [PubMed]
- Pluskal, T.; Torrens-Spence, M.P.; Fallon, T.R.; De Abreu, A.; Shi, C.H.; Weng, J.-K. The biosynthetic origin of psychoactive kavalactones in kava. Nat. Plants 2019, 5, 867–878. [Google Scholar] [CrossRef]
- Coulter, D.; Tamayo, C.; Sotheeswaran, S.; Ulbricht, C.; World Health Organization. Assessment of the Risk of Hepatotoxicity with Kava Products; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Lebot, V.; Cabalion, P. Kavas of Vanuatu. Cultivars of Piper Methysticum Forst; South Pacific Commission: Noumea, France, 1988. [Google Scholar]
- Davis, R.I.; Brown, J.F. Kava (Piper methysticum) in the South Pacific: Its Importance, Methods of Cultivation, Cultivars, Diseases and Pests; Australian Centre for International Agricultural Research (ACIAR): Canberra, Australia, 1999.
- Côté, C.S.; Kor, C.; Cohen, J.; Auclair, K. Composition and biological activity of traditional and commercial kava extracts. Biochem. Biophys. Res. Commun. 2004, 322, 147–152. [Google Scholar] [CrossRef]
- Lebot, V.; Do, T.K.; Legendre, L. Detection of flavokavins (A, B, C) in cultivars of kava (Piper methysticum) using high performance thin layer chromatography (HPTLC). Food Chem. 2014, 151, 554–560. [Google Scholar] [CrossRef]
- Xuan, T.D.; Fukuta, M.; Wei, A.C.; Elzaawely, A.A.; Khanh, T.D.; Tawata, S. Efficacy of extracting solvents to chemical components of kava (Piper methysticum) roots. J. Nat. Med. 2007, 62, 188. [Google Scholar] [CrossRef]
- Lebot, V.; Michalet, S.; Legendre, L. Kavalactones and Flavokavins Profiles Contribute to Quality Assessment of Kava (Piper methysticum G. Forst.), the Traditional Beverage of the Pacific. Beverages 2019, 5, 34. [Google Scholar] [CrossRef]
- Teschke, R.; Lebot, V. Proposal for a kava quality standardization code. Food Chem. Toxicol. 2011, 49, 2503–2516. [Google Scholar] [CrossRef]
- Lebot, V.; Lévesque, J. The origin and distribution of kava (Piper methysticum Forst. f., Piperaceae): A phytochemical approach. Allertonia 1989, 5, 223–281. [Google Scholar]
- Lebot, V.; Levesque, J. Genetic control of kavalactone chemotypes in Piper methysticum cultivars. Phytochemistry 1996, 43, 397–403. [Google Scholar] [CrossRef]
- Kava Act 2002: Republic of Vanuatu Vanuatu Kava Act No. 7. Available online: https://biosecurity.gov.vu/images/Export/kava-act-2002.pdf (accessed on 7 June 2020).
- Hunter, P. The inflammation theory of disease. The growing realization that chronic inflammation is crucial in many diseases opens new avenues for treatment. EMBO Rep. 2012, 13, 968–970. [Google Scholar] [CrossRef] [PubMed]
- Jamieson, D.D.; Duffield, P.H. The antinociceptive actions of kava components in mice. Clin. Exp. Pharmacol. Physiol. 1990, 17, 495–507. [Google Scholar] [CrossRef]
- Nuki, G. Pain control and the use of non-steroidal analgesic anti-inflammatory drugs. Br. Med. Bull. 1990, 46, 262–278. [Google Scholar] [CrossRef]
- Folmer, F.; Blasius, R.; Morceau, F.; Tabudravu, J.; Dicato, M.; Jaspars, M.; Diederich, M. Inhibition of TNFα-induced activation of nuclear factor κB by kava (Piper methysticum) derivatives. Biochem. Pharmacol. 2006, 71, 1206–1218. [Google Scholar] [CrossRef]
- Meyer, H.J. [ANTAGONISTIC EFFECTS OF GEUNINE KAWA PYRONES IN EXPERIMENTAL INFLAMMATIONS AND FEVER]. Klin. Wochenschr. 1965, 43, 469–470. [Google Scholar] [CrossRef]
- Pollastri, M.P.; Whitty, A.; Merrill, J.C.; Tang, X.; Ashton, T.D.; Amar, S. Identification and characterization of kava-derived compounds mediating TNF-alpha suppression. Chem. Biol. Drug Des. 2009, 74, 121–128. [Google Scholar] [CrossRef]
- Fragoulis, A.; Siegl, S.; Fendt, M.; Jansen, S.; Soppa, U.; Brandenburg, L.O.; Pufe, T.; Weis, J.; Wruck, C.J. Oral administration of methysticin improves cognitive deficits in a mouse model of Alzheimer’s disease. Redox Biol. 2017, 12, 843–853. [Google Scholar] [CrossRef]
- Wruck, C.J.; Götz, M.E.; Herdegen, T.; Varoga, D.; Brandenburg, L.O.; Pufe, T. Kavalactones protect neural cells against amyloid beta peptide-induced neurotoxicity via extracellular signal-regulated kinase 1/2-dependent nuclear factor erythroid 2-related factor 2 activation. Mol. Pharmacol. 2008, 73, 1785–1795. [Google Scholar] [CrossRef]
- Dong, R.; Wang, J.; Gao, X.; Wang, C.; Liu, K.; Wu, J.; Liu, Z.; Sun, H.; Ma, X.; Meng, Q. Yangonin protects against estrogen-induced cholestasis in a farnesoid X receptor-dependent manner. Eur. J. Pharmacol. 2019, 857, 172461. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.; Gao, X.; Wang, C.; Ning, C.; Liu, K.; Liu, Z.; Sun, H.; Ma, X.; Sun, P.; Meng, Q. Protective effects of yangonin from an edible botanical Kava against lithocholic acid-induced cholestasis and hepatotoxicity. Eur. J. Pharmacol. 2018, 824, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.W.; Feng, J.H.; Huang, C.C.; Cheng, Y.W.; Chien, S.C.; Wang, S.Y.; Shyur, L.F. A plant kavalactone desmethoxyyangonin prevents inflammation and fulminant hepatitis in mice. PLoS ONE 2013, 8, e77626. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wei, Z.; Teichmann, A.T.; Wieland, F.H.; Wang, A.; Lei, X.; Zhu, Y.; Yin, J.; Fan, T.; Zhou, L.; et al. Development of a novel nitric oxide (NO) production inhibitor with potential therapeutic effect on chronic inflammation. Eur. J. Med. Chem. 2020, 193, 112216. [Google Scholar] [CrossRef] [PubMed]
- Kwon, D.J.; Ju, S.M.; Youn, G.S.; Choi, S.Y.; Park, J. Suppression of iNOS and COX-2 expression by flavokawain A via blockade of NF-κB and AP-1 activation in RAW 264.7 macrophages. Food Chem. Toxicol. 2013, 58, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Abu, N.; Mohameda, N.E.; Tangarajoo, N.; Yeap, S.K.; Akhtar, M.N.; Abdullah, M.P.; Omar, A.R.; Alitheen, N.B. In vitro Toxicity and in vivo Immunomodulatory Effects of Flavokawain A and Flavokawain B in Balb/C Mice. Nat. Prod. Commun. 2015, 10, 1199–1202. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Yang, L.; Wang, J.; Hu, G.; Liu, Z.; Yan, D.; Serikuly, N.; Alpyshov, E.T.; Demin, K.A.; Galstyan, D.S.; et al. Behavioral and physiological effects of acute and chronic kava exposure in adult zebrafish. Neurotoxicol. Teratol. 2020, 79, 106881. [Google Scholar] [CrossRef]
- Shimoda, L.M.; Park, C.; Stokes, A.J.; Gomes, H.H.; Turner, H. Pacific island ’Awa (Kava) extracts, but not isolated kavalactones, promote proinflammatory responses in model mast cells. Phytother. Res. 2012, 26, 1934–1941. [Google Scholar] [CrossRef]
- Huck, O.; You, J.; Han, X.; Cai, B.; Panek, J.; Amar, S. Reduction of Articular and Systemic Inflammation by Kava-241 in a Porphyromonas gingivalis-Induced Arthritis Murine Model. Infect. Immun. 2018, 86, e00356-18. [Google Scholar] [CrossRef]
- Huck, O.; Han, X.; Mulhall, H.; Gumenchuk, I.; Cai, B.; Panek, J.; Iyer, R.; Amar, S. Identification of a Kavain Analog with Efficient Anti-inflammatory Effects. Sci. Rep. 2019, 9, 12940. [Google Scholar] [CrossRef]
- Terazawa, R.; Akimoto, N.; Kato, T.; Itoh, T.; Fujita, Y.; Hamada, N.; Deguchi, T.; Iinuma, M.; Noda, M.; Nozawa, Y.; et al. A kavalactone derivative inhibits lipopolysaccharide-stimulated iNOS induction and NO production through activation of Nrf2 signaling in BV2 microglial cells. Pharmacol. Res. 2013, 71, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Rowe, A.; Braet, F.; Ramzan, I. Macrophage depletion ameliorates kavalactone damage in the isolated perfused rat liver. J. Toxicol. Sci. 2012, 37, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Cai, B.; Panek, J.S.; Amar, S. Kava analogues as agents for treatment of periodontal diseases: Synthesis and initial biological evaluation. Bioorg. Med. Chem. Lett. 2018, 28, 2667–2669. [Google Scholar] [CrossRef] [PubMed]
- Botello, J.F.; Corral, P.; Bian, T.; Xing, C. Kava and its Kavalactones Inhibit Norepinephrine-induced Intracellular Calcium Influx in Lung Cancer Cells. Planta Med. 2020, 86, 26–31. [Google Scholar] [CrossRef]
- Volgin, A.; Yang, L.; Amstislavskaya, T.; Demin, K.; Wang, D.; Yan, D.; Wang, J.; Wang, M.; Alpyshov, E.; Hu, G.; et al. DARK Classics in Chemical Neuroscience: Kava. ACS Chem. Neurosci. 2020. [Google Scholar] [CrossRef]
- Yadhu, N.; Singh, M.B. Kava: An Overview. HerbalGram 1997, 39, 33. [Google Scholar]
- Singh, Y.N. Effects of kava on neuromuscular transmission and muscle contractility. J. Ethnopharmacol. 1983, 7, 267–276. [Google Scholar] [CrossRef]
- Gleitz, J.; Friese, J.; Beile, A.; Ameri, A.; Peters, T. Anticonvulsive action of (+/-)-kavain estimated from its properties on stimulated synaptosomes and Na+ channel receptor sites. Eur. J. Pharmacol. 1996, 315, 89–97. [Google Scholar] [CrossRef]
- Seitz, U.; Ameri, A.; Pelzer, H.; Gleitz, J.; Peters, T. Relaxation of evoked contractile activity of isolated guinea-pig ileum by (+/-)-kavain. Planta Med. 1997, 63, 303–306. [Google Scholar] [CrossRef]
- Martin, H.B.; Stofer, W.D.; Eichinger, M.R. Kavain inhibits murine airway smooth muscle contraction. Planta Med. 2000, 66, 601–606. [Google Scholar] [CrossRef]
- Cairney, S.; Maruff, P.; Clough, A.R. The neurobehavioural effects of kava. Aust. N. Z. J. Psychiatry 2002, 36, 657–662. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.W. Substance, Symbol And Practice. Canberra Anthropol. 1995, 18, 97–118. [Google Scholar] [CrossRef]
- Warnecke, G. [Psychosomatic dysfunctions in the female climacteric. Clinical effectiveness and tolerance of Kava Extract WS 1490]. Fortschr. Der Med. 1991, 109, 119–122. [Google Scholar]
- Warnecke, G.; Pfaender, H.; Gerster, G.; Gracza, E. Wirksamkeit von kawa-kawa extrakt beim klimakterischen syndrom. Z. Phytother. 1990, 11, 81–86. [Google Scholar]
- Cawte, J. Parameters of kava used as a challenge to alcohol. Aust. N. Z. J. Psychiatry 1986, 20, 70–76. [Google Scholar] [CrossRef]
- Boonen, G.; Haberlein, H. Influence of genuine kavapyrone enantiomers on the GABA-A binding site. Planta Med. 1998, 64, 504–506. [Google Scholar] [CrossRef]
- Dinh, L.D.; Simmen, U.; Bueter, K.B.; Bueter, B.; Lundstrom, K.; Schaffner, W. Interaction of various Piper methysticum cultivars with CNS receptors in vitro. Planta Med. 2001, 67, 306–311. [Google Scholar] [CrossRef]
- Uebelhack, R.; Franke, L.; Schewe, H.L. Inhibition of platelet MAO-B by kava pyrone-enriched extract from Piper methysticum Forster (kava-kava). Pharmacopsychiatry 1998, 31, 187–192. [Google Scholar] [CrossRef]
- Magura, E.I.; Kopanitsa, M.V.; Gleitz, J.; Peters, T.; Krishtal, O.A. Kava extract ingredients, (+)-methysticin and (+/-)-kavain inhibit voltage-operated Na+-channels in rat CA1 hippocampal neurons. Neuroscience 1997, 81, 345–351. [Google Scholar] [CrossRef]
- Garrett, K.M.; Basmadjian, G.; Khan, I.A.; Schaneberg, B.T.; Seale, T.W. Extracts of kava (Piper methysticum) induce acute anxiolytic-like behavioral changes in mice. Psychopharmacology 2003, 170, 33–41. [Google Scholar] [CrossRef]
- Davies, L.P.; Drew, C.A.; Duffield, P.; Johnston, G.A.R.; Jamieson, D.D. Kava Pyrones and Resin—Studies on Gaba-a, Gaba-B and Benzodiazepine Binding-Sites in Rodent Brain. Pharmacol. Toxicol. 1992, 71, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Noor, N. Anxiolytic action and safety of Kava: Effect on rat brain acetylcholinesterase activity and some serum biochemical parameters. Afr. J. Pharm. Pharmacol. 2010, 4, 823–828. [Google Scholar]
- Gleitz, J.; Beile, A.; Peters, T. (±)-Kavain inhibits the veratridine- and KCl-induced increase in intracellular Ca2+ and glutamate-release of rat cerebrocortical synaptosomes. Neuropharmacology 1996, 35, 179–186. [Google Scholar] [CrossRef]
- Hegazy, N.H.; Breitinger, H.G.; Breitinger, U. Kavalactones from Kava (Piper methysticum) root extract as modulators of recombinant human glycine receptors. Biol. Chem. 2019, 400, 1205–1215. [Google Scholar] [CrossRef]
- Prinsloo, D.; van Dyk, S.; Petzer, A.; Petzer, J.P. Monoamine Oxidase Inhibition by Kavalactones from Kava (Piper Methysticum). Planta Med. 2019, 85, 1136–1142. [Google Scholar] [CrossRef]
- Baum, S.S.; Hill, R.; Rommelspacher, H. Effect of kava extract and individual kavapyrones on neurotransmitter levels in the nucleus accumbens of rats. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 1998, 22, 1105–1120. [Google Scholar] [CrossRef]
- Walden, J.; von Wegerer, J.; Winter, U.; Berger, M.; Grunze, H. Effects of kawain and dihydromethysticin on field potential changes in the hippocampus. Prog Neuro-Psychopharmacol. Biol. Psychiatry 1997, 21, 697–706. [Google Scholar] [CrossRef]
- Jussofie, A.; Schmiz, A.; Hiemke, C. Kavapyrone enriched extract from Piper methysticum as modulator of the GABA binding site in different regions of rat brain. Psychopharmacology 1994, 116, 469–474. [Google Scholar] [CrossRef]
- Ligresti, A.; Villano, R.; Allarà, M.; Ujváry, I.; Di Marzo, V. Kavalactones and the endocannabinoid system: The plant-derived yangonin is a novel CB₁ receptor ligand. Pharmacol. Res. 2012, 66, 163–169. [Google Scholar] [CrossRef]
- Tzeng, Y.M.; Lee, M.J. Neuroprotective properties of kavalactones. Neural Regen. Res. 2015, 10, 875–877. [Google Scholar] [CrossRef]
- Chua, H.C.; Christensen, E.T.; Hoestgaard-Jensen, K.; Hartiadi, L.Y.; Ramzan, I.; Jensen, A.A.; Absalom, N.L.; Chebib, M. Kavain, the Major Constituent of the Anxiolytic Kava Extract, Potentiates GABAA Receptors: Functional Characteristics and Molecular Mechanism. PLoS ONE 2016, 11, e0157700. [Google Scholar] [CrossRef] [PubMed]
- Ooi, S.L.; Henderson, P.; Pak, S.C. Kava for Generalized Anxiety Disorder: A Review of Current Evidence. J. Altern. Complement. Med. 2018, 24, 770–780. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.; Leiras, C. The effectiveness and safety of Kava Kava for treating anxiety symptoms: A systematic review and analysis of randomized clinical trials. Complement. Ther. Clin. Pract. 2018, 33, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Connor, K.M.; Payne, V.; Davidson, J.R. Kava in generalized anxiety disorder: Three placebo-controlled trials. Int. Clin. Psychopharmacol. 2006, 21, 249–253. [Google Scholar] [CrossRef]
- Sarris, J.; Byrne, G.J.; Bousman, C.A.; Cribb, L.; Savage, K.M.; Holmes, O.; Murphy, J.; Macdonald, P.; Short, A.; Nazareth, S.; et al. Kava for generalised anxiety disorder: A 16-week double-blind, randomised, placebo-controlled study. Aust. N. Z. J. Psychiatry 2020, 54, 288–297. [Google Scholar] [CrossRef]
- Sarris, J.; Stough, C.; Bousman, C.A.; Wahid, Z.T.; Murray, G.; Teschke, R.; Savage, K.M.; Dowell, A.; Ng, C.; Schweitzer, I. Kava in the Treatment of Generalized Anxiety Disorder: A Double-Blind, Randomized, Placebo-Controlled Study. J. Clin. Psychopharmacol. 2013, 33, 643–648. [Google Scholar] [CrossRef]
- Steiner, G.G. Kava as an anticraving agent: Preliminary data. Pac. Health Dialog. 2001, 8, 335–339. [Google Scholar]
- Aporosa, S.A. De-mythologizing and re-branding of kava as the new ‘world drug’ of choice. Drug Sci. Policy Law 2019, 5, 2050324519876131. [Google Scholar] [CrossRef]
- Bilia, A.R.; Gallon, S.; Vincieri, F.F. Kava-kava and anxiety: Growing knowledge about the efficacy and safety. Life Sci. 2002, 70, 2581–2597. [Google Scholar] [CrossRef]
- Münte, T.F.; Heinze, H.J.; Matzke, M.; Steitz, J. Effects of Oxazepam and an Extract of Kava Roots (Piper methysticum) on Event-Related Potentials in a Word Recognition Task. Neuropsychobiology 2004, 27, 46–53. [Google Scholar]
- Emser, W.; Bartylla, K. Improvement in quality of sleep: Effect of kava extract WS 1490 on the sleep patterns in healthy people. TW Neurol. Psychiatr. 1991, 5, 636–642. [Google Scholar]
- Lehrl, S. Clinical efficacy of kava extract WS 1490 in sleep disturbances associated with anxiety disorders. Results of a multicenter, randomized, placebo-controlled, double-blind clinical trial. J. Affect. Disord. 2004, 78, 101–110. [Google Scholar] [CrossRef]
- Steiner, G.G. The correlation between cancer incidence and kava consumption. Hawaii Med. J. 2000, 59, 420–422. [Google Scholar] [PubMed]
- Johnson, T.E.; Kassie, F.; Sullivan, M.G.; Negia, M.; Hanson, T.E.; Upadhyaya, P.; Ruvolo, P.P.; Hecht, S.S.; Xing, C. Chemopreventive Effect of Kava on 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanone plus Benzo[a]pyrene–Induced Lung Tumorigenesis in A/J Mice. Cancer Prev. Res. 2008, 1, 430. [Google Scholar] [CrossRef]
- Leitzman, P.; Narayanapillai, S.C.; Balbo, S.; Zhou, B.; Upadhyaya, P.; Shaik, A.A.; Sullivan, M.G.; Hecht, S.S.; Lu, J.; Xing, C. Kava Blocks 4-(Methylnitrosamino)-1-(3-pyridyl)-1-Butanone–Induced Lung Tumorigenesis in Association with Reducing O6-methylguanine DNA Adduct in A/J Mice. Cancer Prev. Res. 2014, 7, 86. [Google Scholar] [CrossRef]
- Puppala, M.; Narayanapillai, S.C.; Leitzman, P.; Sun, H.; Upadhyaya, P.; O’Sullivan, M.G.; Hecht, S.S.; Xing, C. Pilot in Vivo Structure-Activity Relationship of Dihydromethysticin in Blocking 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanone-Induced O(6)-Methylguanine and Lung Tumor in A/J Mice. J. Med. Chem. 2017, 60, 7935–7940. [Google Scholar] [CrossRef] [PubMed]
- Narayanapillai, S.C.; Lin, S.-H.; Leitzman, P.; Upadhyaya, P.; Baglole, C.J.; Xing, C. Dihydromethysticin (DHM) Blocks Tobacco Carcinogen 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK)-Induced O6-Methylguanine in a Manner Independent of the Aryl Hydrocarbon Receptor (AhR) Pathway in C57BL/6 Female Mice. Chem. Res. Toxicol. 2016, 29, 1828–1834. [Google Scholar] [CrossRef] [PubMed]
- Narayanapillai, S.C.; von Weymarn, L.B.; Carmella, S.G.; Leitzman, P.; Paladino, J.; Upadhyaya, P.; Hecht, S.S.; Murphy, S.E.; Xing, C. Dietary Dihydromethysticin Increases Glucuronidation of 4-(Methylnitrosamino)-1-(3-Pyridyl)-1-Butanol in A/J Mice, Potentially Enhancing Its Detoxification. Drug Metab. Dispos. 2016, 44, 422. [Google Scholar] [CrossRef]
- Wang, Y.; Narayanapillai, S.C.; Tessier, K.M.; Strayer, L.G.; Upadhyaya, P.; Hu, Q.; Kingston, R.; Salloum, R.G.; Lu, J.; Hecht, S.S.; et al. The Impact of One-week Dietary Supplementation with Kava on Biomarkers of Tobacco Use and Nitrosamine-based Carcinogenesis Risk among Active Smokers. Cancer Prev. Res. 2020, 13, 483. [Google Scholar] [CrossRef]
- Liu, Z.; Xu, X.; Li, X.; Liu, S.; Simoneau, A.R.; He, F.; Wu, X.-R.; Zi, X. Kava chalcone, flavokawain A, inhibits urothelial tumorigenesis in the UPII-SV40T transgenic mouse model. Cancer Prev. Res. 2013, 6, 1365–1375. [Google Scholar] [CrossRef]
- Li, X.; Yokoyama, N.N.; Zhang, S.; Ding, L.; Liu, H.-m.; Lilly, M.B.; Mercola, D.; Zi, X. Flavokawain A induces deNEDDylation and Skp2 degradation leading to inhibition of tumorigenesis and cancer progression in the TRAMP transgenic mouse model. Oncotarget 2015, 6, 41809–41824. [Google Scholar] [CrossRef] [PubMed]
- Triolet, J.; Shaik, A.A.; Gallaher, D.D.; O’Sullivan, M.G.; Xing, C. Reduction in colon cancer risk by consumption of kava or kava fractions in carcinogen-treated rats. Nutr. Cancer 2012, 64, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.-N.; Zhang, J.; Jiang, P.; Datta, P.; Leitzman, P.; O’Sullivan, M.G.; Jiang, C.; Xing, C.; Lü, J. Gene expression signatures associated with suppression of TRAMP prostate carcinogenesis by a kavalactone-rich Kava fraction. Mol. Carcinog. 2016, 55, 2291–2303. [Google Scholar] [CrossRef] [PubMed]
- Einbond, L.S.; Negrin, A.; Kulakowski, D.M.; Wu, H.A.; Antonetti, V.; Jalees, F.; Law, W.; Roller, M.; Redenti, S.; Kennelly, E.J.; et al. Traditional preparations of kava (Piper methysticum) inhibit the growth of human colon cancer cells in vitro. Phytomedicine 2017, 24, 1–13. [Google Scholar] [CrossRef]
- Li, X.; Liu, Z.; Xu, X.; Blair, C.A.; Sun, Z.; Xie, J.; Lilly, M.B.; Zi, X. Kava components down-regulate expression of AR and AR splice variants and reduce growth in patient-derived prostate cancer xenografts in mice. PLoS ONE 2012, 7, e31213. [Google Scholar] [CrossRef]
- Li, J.; Zheng, L.; Yan, M.; Wu, J.; Liu, Y.; Tian, X.; Jiang, W.; Zhang, L.; Wang, R. Activity and mechanism of flavokawain A in inhibiting P-glycoprotein expression in paclitaxel resistance of lung cancer. Oncol. Lett. 2020, 19, 379–387. [Google Scholar] [CrossRef]
- Zhang, Y.; Zvi, Y.S.; Batko, B.; Zaphiros, N.; O’Donnell, E.F.; Wang, J.; Sato, K.; Yang, R.; Geller, D.S.; Koirala, P.; et al. Down-regulation of Skp2 expression inhibits invasion and lung metastasis in osteosarcoma. Sci. Rep. 2018, 8, 14294. [Google Scholar] [CrossRef]
- Zi, X.; Simoneau, A.R. Flavokawain A, a Novel Chalcone from Kava Extract, Induces Apoptosis in Bladder Cancer Cells by Involvement of Bax Protein-Dependent and Mitochondria-Dependent Apoptotic Pathway and Suppresses Tumor Growth in Mice. Cancer Res. 2005, 65, 3479. [Google Scholar] [CrossRef]
- Tang, Y.; Simoneau, A.R.; Xie, J.; Shahandeh, B.; Zi, X. Effects of the Kava Chalcone Flavokawain A Differ in Bladder Cancer Cells with Wild-type versus Mutant p53. Cancer Prev. Res. 2008, 1, 439. [Google Scholar] [CrossRef]
- Wang, K.; Zhang, W.; Wang, Z.; Gao, M.; Wang, X.; Han, W.; Zhang, N.; Xu, X. Flavokawain A inhibits prostate cancer cells by inducing cell cycle arrest and cell apoptosis and regulating the glutamine metabolism pathway. J. Pharm. Biomed. Anal. 2020, 186, 113288. [Google Scholar] [CrossRef]
- Nadiah, A.; Nurul Elyani, M.; Swee Keong, Y.; Kian Lam, L.; Akhtar, M.N.; Aimi Jamil, Z.; Beh Boon, K.; Mohd Puad, A.; Abdul Rahman, O.; Noorjahan Banu, A. In Vivo Anti-Tumor Effects of Flavokawain A in 4T1 Breast Cancer Cell-Challenged Mice. Anti-Cancer Agents Med. Chem. 2015, 15, 905–915. [Google Scholar] [CrossRef]
- Jandial, D.D.; Krill, L.S.; Chen, L.; Wu, C.; Ke, Y.; Xie, J.; Hoang, B.H.; Zi, X. Induction of G2M Arrest by Flavokawain A, a Kava Chalcone, Increases the Responsiveness of HER2-Overexpressing Breast Cancer Cells to Herceptin. Molecules 2017, 22, 462. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Yu, X.; Zhuang, C.; Villalta, P.; Lin, Y.; Lu, J.; Xing, C. Unambiguous Identification of beta-Tubulin as the Direct Cellular Target Responsible for the Cytotoxicity of Chalcone by Photoaffinity Labeling. ChemMedChem 2016, 11, 1436–1445. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, C.; Zhang, W.; Sheng, C.; Zhang, W.; Xing, C.; Miao, Z. Chalcone: A Privileged Structure in Medicinal Chemistry. Chem. Rev. 2017, 117, 7762–7810. [Google Scholar] [CrossRef]
- Abu, N.; Akhtar, M.N.; Yeap, S.K.; Lim, K.L.; Ho, W.Y.; Abdullah, M.P.; Ho, C.L.; Omar, A.R.; Ismail, J.; Alitheen, N.B. Flavokawain B induced cytotoxicity in two breast cancer cell lines, MCF-7 and MDA-MB231 and inhibited the metastatic potential of MDA-MB231 via the regulation of several tyrosine kinases In vitro. BMC Complement. Altern. Med. 2016, 16, 86. [Google Scholar] [CrossRef]
- Hua, R.; Pei, Y.; Gu, H.; Sun, Y.; He, Y. Antitumor effects of flavokawain-B flavonoid in gemcitabine-resistant lung cancer cells are mediated via mitochondrial-mediated apoptosis, ROS production, cell migration and cell invasion inhibition and blocking of PI3K/AKT Signaling pathway. J. BUON 2020, 25, 262–267. [Google Scholar]
- Rossette, M.C.; Moraes, D.C.; Sacramento, E.K.; Romano-Silva, M.A.; Carvalho, J.L.; Gomes, D.A.; Caldas, H.; Friedman, E.; Bastos-Rodrigues, L.; De Marco, L. The In Vitro and In Vivo Antiangiogenic Effects of Flavokawain, B. Phytother. Res. 2017, 31, 1607–1613. [Google Scholar] [CrossRef]
- Ji, T.; Lin, C.; Krill, L.S.; Eskander, R.; Guo, Y.; Zi, X.; Hoang, B.H. Flavokawain B, a kava chalcone, inhibits growth of human osteosarcoma cells through G2/M cell cycle arrest and apoptosis. Mol. Cancer 2013, 12, 55. [Google Scholar] [CrossRef]
- An, J.; Gao, Y.; Wang, J.; Zhu, Q.; Ma, Y.; Wu, J.; Sun, J.; Tang, Y. Flavokawain B induces apoptosis of non-small cell lung cancer H460 cells via Bax-initiated mitochondrial and JNK pathway. Biotechnol. Lett. 2012, 34, 1781–1788. [Google Scholar] [CrossRef][Green Version]
- Sakai, T.; Eskander, R.N.; Guo, Y.; Kim, K.J.; Mefford, J.; Hopkins, J.; Bhatia, N.N.; Zi, X.; Hoang, B.H. Flavokawain B, a kava chalcone, induces apoptosis in synovial sarcoma cell lines. J. Orthop. Res. 2012, 30, 1045–1050. [Google Scholar] [CrossRef]
- Kuo, Y.-F.; Su, Y.-Z.; Tseng, Y.-H.; Wang, S.-Y.; Wang, H.-M.; Chueh, P.J. Flavokawain B, a novel chalcone from Alpinia pricei Hayata with potent apoptotic activity: Involvement of ROS and GADD153 upstream of mitochondria-dependent apoptosis in HCT116 cells. Free Radic. Biol. Med. 2010, 49, 214–226. [Google Scholar] [CrossRef] [PubMed]
- Lin, E.; Lin, W.-H.; Wang, S.-Y.; Chen, C.-S.; Liao, J.-W.; Chang, H.-W.; Chen, S.-C.; Lin, K.-Y.; Wang, L.; Yang, H.-L.; et al. Flavokawain B inhibits growth of human squamous carcinoma cells: Involvement of apoptosis and cell cycle dysregulation in vitro and in vivo. J. Nutr. Biochem. 2012, 23, 368–378. [Google Scholar] [CrossRef] [PubMed]
- Hseu, Y.-C.; Lee, M.-S.; Wu, C.-R.; Cho, H.-J.; Lin, K.-Y.; Lai, G.-H.; Wang, S.-Y.; Kuo, Y.-H.; Senthil Kumar, K.J.; Yang, H.-L. The Chalcone Flavokawain B Induces G2/M Cell-Cycle Arrest and Apoptosis in Human Oral Carcinoma HSC-3 Cells through the Intracellular ROS Generation and Downregulation of the Akt/p38 MAPK Signaling Pathway. J. Agric. Food Chem. 2012, 60, 2385–2397. [Google Scholar] [CrossRef] [PubMed]
- Abu Bakar, A.; Akhtar, M.N.; Mohd Ali, N.; Yeap, S.K.; Quah, C.K.; Loh, W.-S.; Alitheen, N.B.; Zareen, S.; Ul-Haq, Z.; Shah, S.A.A. Design, Synthesis and Docking Studies of Flavokawain B Type Chalcones and Their Cytotoxic Effects on MCF-7 and MDA-MB-231 Cell Lines. Molecules 2018, 23, 616. [Google Scholar] [CrossRef]
- Malami, I.; Abdul, A.B.; Abdullah, R.; Kassim, N.K.B.; Rosli, R.; Yeap, S.K.; Waziri, P.; Etti, I.C.; Bello, M.B. Crude Extracts, Flavokawain B and Alpinetin Compounds from the Rhizome of Alpinia mutica Induce Cell Death via UCK2 Enzyme Inhibition and in Turn Reduce 18S rRNA Biosynthesis in HT-29 Cells. PLoS ONE 2017, 12, e0170233. [Google Scholar] [CrossRef]
- Hseu, Y.-C.; Huang, Y.-C.; Thiyagarajan, V.; Mathew, D.C.; Lin, K.-Y.; Chen, S.-C.; Liu, J.-Y.; Hsu, L.-S.; Li, M.-L.; Yang, H.-L. Anticancer activities of chalcone flavokawain B from Alpinia pricei Hayata in human lung adenocarcinoma (A549) cells via induction of reactive oxygen species-mediated apoptotic and autophagic cell death. J. Cell Physiol. 2019, 234, 17514–17526. [Google Scholar] [CrossRef]
- Chang, C.-T.; Hseu, Y.-C.; Thiyagarajan, V.; Lin, K.-Y.; Way, T.-D.; Korivi, M.; Liao, J.-W.; Yang, H.-L. Chalcone flavokawain B induces autophagic-cell death via reactive oxygen species-mediated signaling pathways in human gastric carcinoma and suppresses tumor growth in nude mice. Arch. Toxicol. 2017, 91, 3341–3364. [Google Scholar] [CrossRef]
- Wang, J.; Qi, Q.; Zhou, W.; Feng, Z.; Huang, B.; Chen, A.; Zhang, D.; Li, W.; Zhang, Q.; Jiang, Z.; et al. Inhibition of glioma growth by flavokawain B is mediated through endoplasmic reticulum stress induced autophagy. Autophagy 2018, 14, 2007–2022. [Google Scholar] [CrossRef]
- He, Q.; Liu, W.; Sha, S.; Fan, S.; Yu, Y.; Chen, L.; Dong, M. Adenosine 5’-monophosphate-activated protein kinase-dependent mTOR pathway is involved in flavokawain B-induced autophagy in thyroid cancer cells. Cancer Sci. 2018, 109, 2576–2589. [Google Scholar] [CrossRef]
- Abu, N.; Mohamed, N.E.; Yeap, S.K.; Lim, K.L.; Akhtar, M.N.; Zulfadli, A.J.; Kee, B.B.; Abdullah, M.P.; Omar, A.R.; Alitheen, N.B. In vivo antitumor and antimetastatic effects of flavokawain B in 4T1 breast cancer cell-challenged mice. Drug Des. Dev. Ther. 2015, 9, 1401–1417. [Google Scholar] [CrossRef]
- Yeap, S.K.; Abu, N.; Akthar, N.; Ho, W.Y.; Ky, H.; Tan, S.W.; Alitheen, N.B.; Kamarul, T. Gene Expression Analysis Reveals the Concurrent Activation of Proapoptotic and Antioxidant-Defensive Mechanisms in Flavokawain B-Treated Cervical Cancer HeLa Cells. Integr. Cancer Ther. 2017, 16, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Pham, V.; Tippin, M.; Fu, D.; Rendon, R.; Song, L.; Uchio, E.; Hoang, B.H.; Zi, X. Flavokawain B targets protein neddylation for enhancing the anti-prostate cancer effect of Bortezomib via Skp2 degradation. Cell Commun. Signal. 2019, 17, 25. [Google Scholar] [CrossRef]
- Lee, J.J.; Koh, K.-N.; Park, C.-J.; Jang, S.; Im, H.J.; Kim, N. The Combination of Flavokawain B and Daunorubicin Induces Apoptosis in Human Myeloid Leukemic Cells by Modifying NF-κB. Anticancer Res. 2018, 38, 2771–2778. [Google Scholar] [PubMed]
- Dai, J.-Q.; Huang, Y.-G.; He, A.-N. Dihydromethysticin kavalactone induces apoptosis in osteosarcoma cells through modulation of PI3K/Akt pathway, disruption of mitochondrial membrane potential and inducing cell cycle arrest. Int. J. Clin. Exp. Pathol. 2015, 8, 4356–4366. [Google Scholar] [PubMed]
- Pan, H.; Liu, F.; Wang, J.; Zhao, M.; Wang, D.; Jia, C.; Wang, T.; Chen, Z.; Fan, Y.; Liang, D.; et al. Dihydromethysticin, a natural molecule from Kava, suppresses the growth of colorectal cancer via the NLRC3/PI3K pathway. Mol. Carcinog. 2020, 59, 575–589. [Google Scholar] [CrossRef]
- Liu, Z.; Ha, U.S.; Yu, K.; Wu, C.; Yokoyama, N.; Zi, X. Kavalactone yangonin induces autophagy and sensitizes bladder cancer cells to flavokawain A and docetaxel via inhibition of the mTOR pathway. J. Biomed. Res. 2017, 31, 408–418. [Google Scholar] [CrossRef]
- Weiss, J.; Sauer, A.; Frank, A.; Unger, M. Extracts and Kavalactones of Piper Methysticum G. Forst (Kava-kava) inhibit P-Glycoprotein in vitro. Drug Metab. Dispos. 2005, 33, 1580. [Google Scholar] [CrossRef]
- Tarbah, F.; Mahler, H.; Kardel, B.; Weinmann, W.; Hafner, D.; Daldrup, T. Kinetics of kavain and its metabolites after oral application. J. Chromatogr. B 2003, 789, 115–130. [Google Scholar] [CrossRef]
- Shaik, A.A.; Hermanson, D.L.; Xing, C. Identification of methysticin as a potent and non-toxic NF-kappaB inhibitor from kava, potentially responsible for kava’s chemopreventive activity. Bioorg. Med. Chem. Lett. 2009, 19, 5732–5736. [Google Scholar] [CrossRef]
- Zhou, P.; Gross, S.; Liu, J.H.; Yu, B.Y.; Feng, L.L.; Nolta, J.; Sharma, V.; Piwnica-Worms, D.; Qiu, S.X. Flavokawain B, the hepatotoxic constituent from kava root, induces GSH-sensitive oxidative stress through modulation of IKK/NF-kappaB and MAPK signaling pathways. FASEB J. 2010, 24, 4722–4732. [Google Scholar] [CrossRef]
- Narayanapillai, S.C.; Leitzman, P.; O’Sullivan, M.G.; Xing, C. Flavokawains a and B in kava, not dihydromethysticin, potentiate acetaminophen-induced hepatotoxicity in C57BL/6 mice. Chem. Res. Toxicol. 2014, 27, 1871–1876. [Google Scholar] [CrossRef]
- Teschke, R.; Sarris, J.; Glass, X.; Schulze, J. Kava, the anxiolytic herb: Back to basics to prevent liver injury? Br. J. Clin. Pharmacol. 2011, 71, 445–448. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M. Are kavalactones the hepatotoxic principle of kava extracts? The pitfalls of the glutathione theory. J. Altern. Complement. Med. 2003, 9, 183–187; author reply 187–188. [Google Scholar] [CrossRef] [PubMed]
- Peterson, L.A.; Balbo, S.; Fujioka, N.; Hatsukami, D.K.; Hecht, S.S.; Murphy, S.E.; Stepanov, I.; Tretyakova, N.; Turesky, R.J.; Villalta, P.W. Applying tobacco, environmental and dietary-related biomarkers to understand cancer etiology and evaluate prevention strategies. Cancer Epidemiol. Biomark. Prev. 2020, 29, 1904–1919. [Google Scholar] [CrossRef] [PubMed]
- Moulds, R.F.; Malani, J. Kava: Herbal panacea or liver poison? Med. J. Aust. 2003, 178, 451–453. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Sarris, J.; Lebot, V. Contaminant hepatotoxins as culprits for kava hepatotoxicity—Fact or fiction? Phytother. Res. 2013, 27, 472–474. [Google Scholar] [CrossRef]
- Teschke, R.; Sarris, J.; Lebot, V. Kava hepatotoxicity solution: A six-point plan for new kava standardization. Phytomedicine 2011, 18, 96–103. [Google Scholar] [CrossRef]
- Teschke, R.; Qiu, S.X.; Xuan, T.D.; Lebot, V. Kava and kava hepatotoxicity: Requirements for novel experimental, ethnobotanical and clinical studies based on a review of the evidence. Phytother. Res. 2011, 25, 1263–1274. [Google Scholar] [CrossRef]
- Teschke, R.; Qiu, S.X.; Lebot, V. Herbal hepatotoxicity by kava: Update on pipermethystine, flavokavain B, and mould hepatotoxins as primarily assumed culprits. Dig. Liver Dis. 2011, 43, 676–681. [Google Scholar] [CrossRef]
- Teschke, R.; Genthner, A.; Wolff, A. Kava hepatotoxicity: Comparison of aqueous, ethanolic, acetonic kava extracts and kava-herbs mixtures. J. Ethnopharmacol. 2009, 123, 378–384. [Google Scholar] [CrossRef]
- Teschke, R.; Schwarzenboeck, A.; Hennermann, K.H. Kava hepatotoxicity: A clinical survey and critical analysis of 26 suspected cases. Eur. J. Gastroenterol. Hepatol. 2008, 20, 1182–1193. [Google Scholar] [CrossRef] [PubMed]
- Anke, J.; Ramzan, I. Kava Hepatotoxicity: Are we any closer to the truth? Planta Med. 2004, 70, 193–196. [Google Scholar] [PubMed]
- Johnson, T.E.; Hermanson, D.; Wang, L.; Kassie, F.; Upadhyaya, P.; O’Sullivan, M.G.; Hecht, S.S.; Lu, J.; Xing, C. Lung tumorigenesis suppressing effects of a commercial kava extract and its selected compounds in A/J mice. Am. J. Chin. Med. 2011, 39, 727–742. [Google Scholar] [CrossRef] [PubMed]
- Stickel, F.; Baumuller, H.M.; Seitz, K.; Vasilakis, D.; Seitz, G.; Seitz, H.K.; Schuppan, D. Hepatitis induced by Kava (Piper methysticum rhizoma). J. Hepatol. 2003, 39, 62–67. [Google Scholar] [CrossRef]
- Program, N.T. NTP Technical Report on the Toxicology and Carcinogenesis Studies of Kava Kava Extract in F344/N Rats and B6C3F1 Mice; NTP TR 571, No. 12-5913; NIH: Bethesda, MA, USA, 2012; pp. 1–192.
- McElroy, J.P.; Carmella, S.G.; Heskin, A.K.; Tang, M.K.; Murphy, S.E.; Reisinger, S.A.; Jensen, J.A.; Hatsukami, D.K.; Hecht, S.S.; Shields, P.G. Effects of cessation of cigarette smoking on eicosanoid biomarkers of inflammation and oxidative damage. PLoS ONE 2019, 14, e0218386. [Google Scholar] [CrossRef]
- Toll, B.A.; O’Malley, S.S.; McKee, S.A.; Salovey, P.; Krishnan-Sarin, S. Confirmatory factor analysis of the Minnesota Nicotine Withdrawal Scale. Psychol. Addict. Behav. 2007, 21, 216–225. [Google Scholar] [CrossRef]
- Carmella, S.G.; Heskin, A.K.; Tang, M.K.; Jensen, J.; Luo, X.; Le, C.T.; Murphy, S.E.; Benowitz, N.L.; McClernon, F.J.; Vandrey, R.; et al. Longitudinal stability in cigarette smokers of urinary eicosanoid biomarkers of oxidative damage and inflammation. PLoS ONE 2019, 14, e0215853. [Google Scholar] [CrossRef]
- Teschke, R. Kava hepatotoxicity—A clinical review. Ann. Hepatol. 2010, 9, 251–265. [Google Scholar] [CrossRef]
- Clouatre, D.L. Kava kava: Examining new reports of toxicity. Toxicol. Lett. 2004, 150, 85–96. [Google Scholar] [CrossRef]
- Kessler, R.C.; Chiu, W.T.; Demler, O.; Merikangas, K.R.; Walters, E.E. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch. Gen. Psychiatry 2005, 62, 617–627. [Google Scholar] [CrossRef]
- Bachhuber, M.A.; Hennessy, S.; Cunningham, C.O.; Starrels, J.L. Increasing Benzodiazepine Prescriptions and Overdose Mortality in the United States, 1996–2013. Am. J. Public Health 2016, 106, 686–688. [Google Scholar] [CrossRef] [PubMed]
- Fluyau, D.; Revadigar, N.; Manobianco, B.E. Challenges of the pharmacological management of benzodiazepine withdrawal, dependence, and discontinuation. Ther. Adv. Psychopharmacol. 2018, 8, 147–168. [Google Scholar] [CrossRef] [PubMed]
- Weinberger, D.R. Anxiety at the frontier of molecular medicine. N. Engl. J. Med. 2001, 344, 1247–1249. [Google Scholar] [CrossRef] [PubMed]
- Sarris, J.; Scholey, A.; Schweitzer, I.; Bousman, C.; Laporte, E.; Ng, C.; Murray, G.; Stough, C. The acute effects of kava and oxazepam on anxiety, mood, neurocognition; and genetic correlates: A randomized, placebo-controlled, double-blind study. Hum. Psychopharmacol. 2012, 27, 262–269. [Google Scholar] [CrossRef]
- Wang, Y.; Eans, S.; Stacy, H.; Narayanapillai, S.; Sharma, A.; Fujioka, N.; Haddad, L.; McLaughlin, J.; Avery, B.A.; Xing, C. A stable isotope dilution tandem mass spectrometry method of major kavalactones and its applications. PLoS ONE 2018, 13, e0197940. [Google Scholar] [CrossRef]
- Berry, J.; Gilbert, A.; Grodnitzky, J. Cases of Kava Impairment in Iowa Drivers. J. Forensic. Sci. 2019, 64, 1943–1949. [Google Scholar] [CrossRef]
- Herberg, K.W. Effect of Kava-Special Extract WS 1490 combined with ethyl alcohol on safety-relevant performance parameters. Blutalkohol 1993, 30, 96–105. [Google Scholar]
- Foo, H.; Lemon, J. Acute effects of kava, alone or in combination with alcohol, on subjective measures of impairment and intoxication and on cognitive performance. Drug Alcohol Rev. 1997, 16, 147–155. [Google Scholar] [CrossRef]
- Sarris, J.; Laporte, E.; Scholey, A.; King, R.; Pipingas, A.; Schweitzer, I.; Stough, C. Does a medicinal dose of kava impair driving? A randomized, placebo-controlled, double-blind study. Traffic Inj. Prev. 2013, 14, 13–17. [Google Scholar] [CrossRef]
- Christl, S.U.; Seifert, A.; Seeler, D. Toxic hepatitis after consumption of traditional kava preparation. J. Travel Med. 2009, 16, 55–56. [Google Scholar] [CrossRef]
- Cox, L.S.; Tiffany, S.T.; Christen, A.G. Evaluation of the brief questionnaire of smoking urges (QSU-brief) in laboratory and clinical settings. Nicotine Tob. Res. 2001, 3, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Ketola, R.A.; Viinamaki, J.; Rasanen, I.; Pelander, A.; Goebeler, S. Fatal kavalactone intoxication by suicidal intravenous injection. Forensic. Sci. Int. 2015, 249, e7–e11. [Google Scholar] [CrossRef] [PubMed]
- Dong, R.; Yang, X.; Wang, C.; Liu, K.; Liu, Z.; Ma, X.; Sun, H.; Huo, X.; Fu, T.; Meng, Q. Yangonin protects against non-alcoholic fatty liver disease through farnesoid X receptor. Phytomedicine 2019, 53, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Jhoo, J.W.; Freeman, J.P.; Heinze, T.M.; Moody, J.D.; Schnackenberg, L.K.; Beger, R.D.; Dragull, K.; Tang, C.S.; Ang, C.Y. In vitro cytotoxicity of nonpolar constituents from different parts of kava plant (Piper methysticum). J. Agric. Food Chem. 2006, 54, 3157–3162. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xu, X.; Ji, T.; Liu, Z.; Gu, M.; Hoang, B.H.; Zi, X. Dietary feeding of Flavokawain A, a Kava chalcone, exhibits a satisfactory safety profile and its association with enhancement of phase II enzymes in mice. Toxicol. Rep. 2014, 1, 2–11. [Google Scholar] [CrossRef] [PubMed]

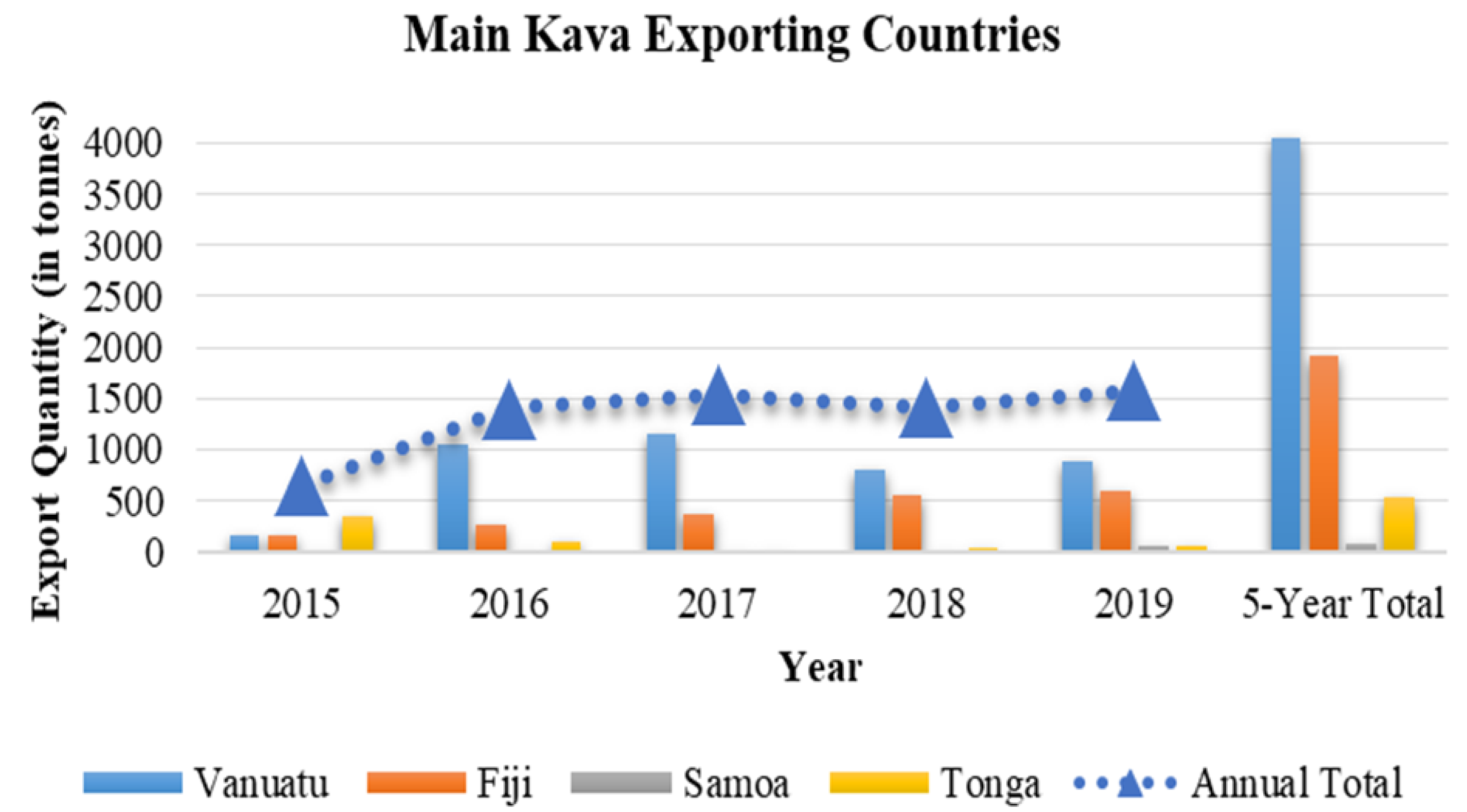
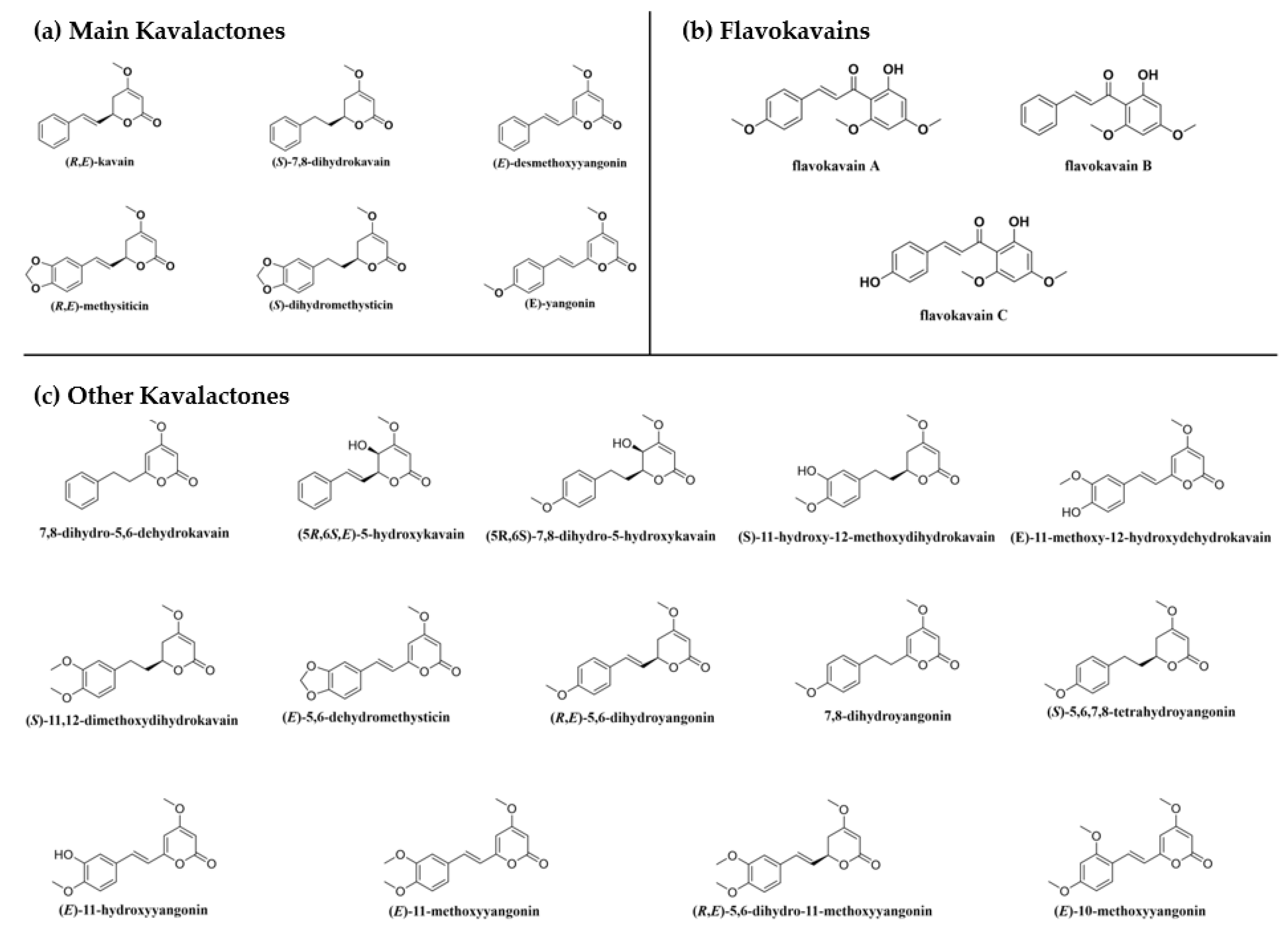
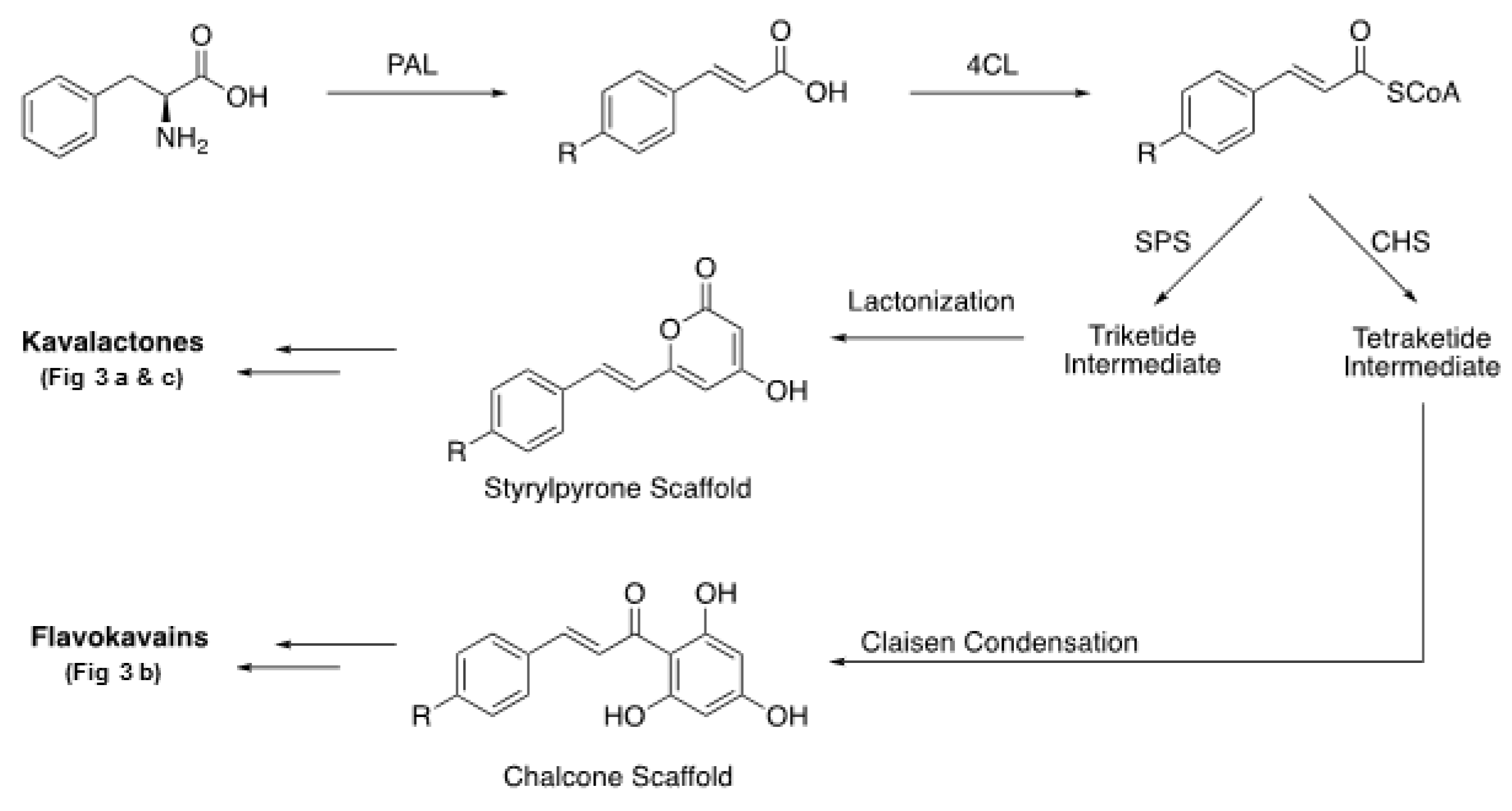
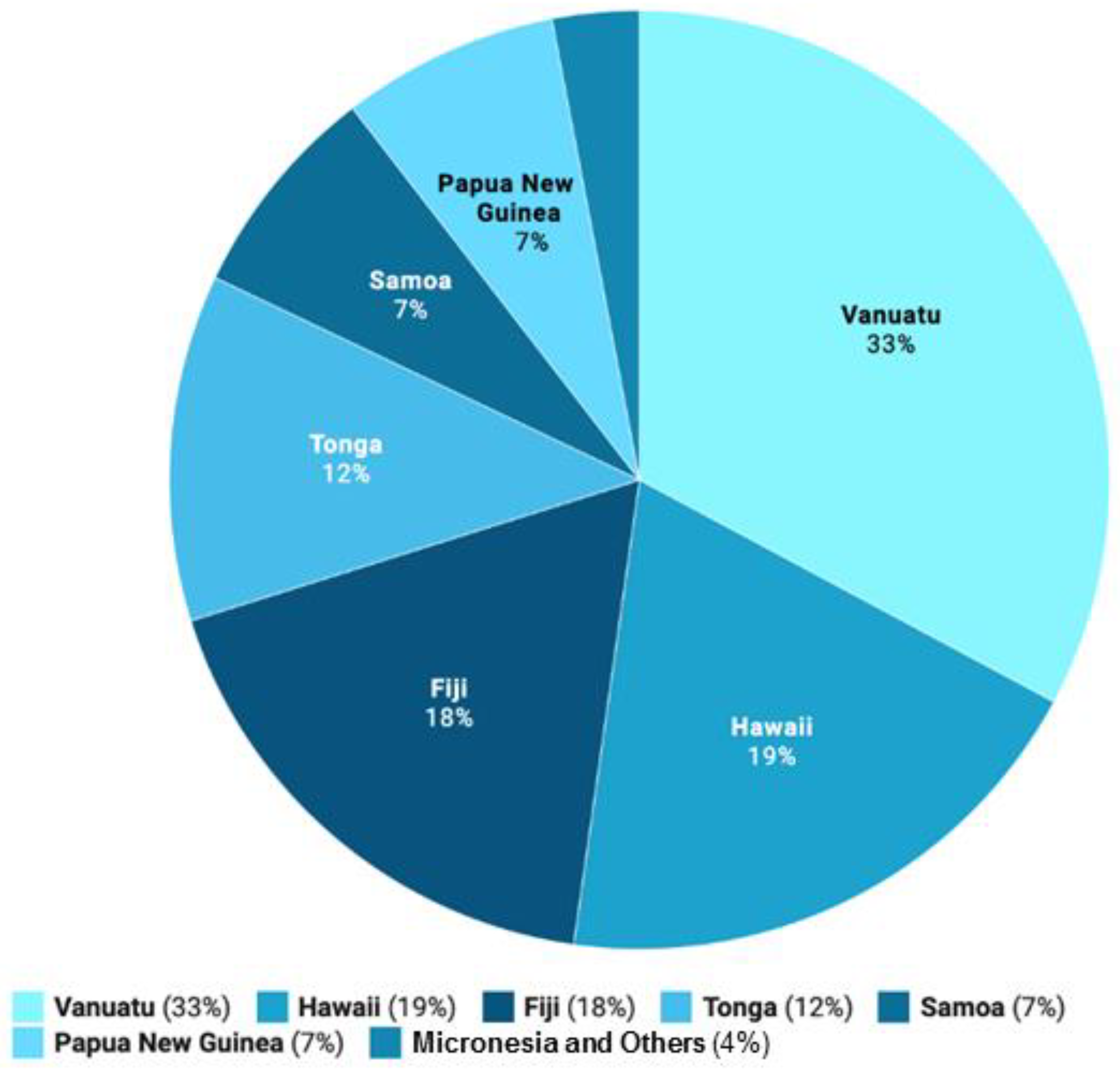
| Major Regions | Traditional Kava Dose | References |
|---|---|---|
| Vanuatu | Female: 750 mg/day; male: 1000 mg/day | [15] |
| Fiji | >8000 mg/drink | [16] |
| Tonga | 2400 mg/day | [17] |
| Hawaii | 1000–1500 mg/drink | [18] |
| Noble | ||||||||
|---|---|---|---|---|---|---|---|---|
| Puariki | Kelai | Borogu | Borogu | Borogu | Borogu | Borogu | Borogu | |
| DMY 1 (%) | 0.71 | 0.91 | 0.74 | 0.84 | 0.7 | 0.51 | 0.67 | 0.61 |
| DHK 2 (%) | 1.49 | 2.21 | 2.33 | 2.16 | 2.29 | 2.05 | 1.94 | 1.44 |
| Y 3 (%) | 0.99 | 1.57 | 2.06 | 1.94 | 1.88 | 1.74 | 1.69 | 1.34 |
| K 4 (%) | 2.7 | 4.05 | 3.26 | 3.53 | 3.21 | 2.74 | 2.88 | 2.42 |
| DHM 5 (%) | 0.63 | 0.71 | 0.95 | 0.96 | 0.96 | 0.95 | 0.82 | 0.74 |
| M 6 (%) | 0.43 | 0.64 | 0.71 | 0.86 | 0.79 | 0.69 | 0.71 | 0.69 |
| Total (%) | 6.95 | 10.09 | 10.05 | 10.29 | 9.83 | 8.68 | 8.71 | 7.24 |
| Chemotype | 423156 | 423156 | 423516 | 423561 | 423561 | 423561 | 423561 | 423561 |
| Borogu | Borogu | Borogu | Borogu | Borogu | Borogu | Silese | Ahouia | |
| DMY 1 (%) | 0.78 | 0.38 | 0.47 | 0.46 | 0.43 | 0.45 | 0.64 | 0.78 |
| DHK 2 (%) | 2.17 | 1.12 | 1.48 | 1.17 | 1.24 | 1.47 | 1.5 | 1.83 |
| Y 3 (%) | 1.91 | 0.74 | 0.88 | 0.84 | 0.8 | 0.7 | 1.43 | 1.84 |
| K 4 (%) | 3.73 | 1.92 | 2.35 | 2.38 | 2.01 | 2.23 | 2.69 | 3.27 |
| DHM 5 (%) | 1.19 | 0.66 | 0.72 | 0.48 | 0.69 | 0.57 | 0.75 | 0.86 |
| M 6 (%) | 1.13 | 0.63 | 0.67 | 0.67 | 0.66 | 0.6 | 0.75 | 0.73 |
| Total (%) | 10.91 | 5.45 | 6.57 | 6 | 5.83 | 6.02 | 7.76 | 9.31 |
| Chemotype | 423561 | 423561 | 423561 | 423651 | 423561 | 423651 | 423651 | 432516 |
| Tuidei | ||||||||
| Tarivoravora | Abogae | Gelav | Marino | Rongrongwul | PD Palisi | PA Palisi | PC Palisi | |
| DMY 1 (%) | 0.71 | 0.61 | 0.8 | 0.63 | 0.84 | 0.59 | 0.57 | 0.41 |
| DHK 2 (%) | 2.7 | 2.63 | 3.03 | 2.46 | 4.04 | 2.23 | 1.95 | 2.24 |
| Y 3 (%) | 1.66 | 1.35 | 1.98 | 1.49 | 1.62 | 1.41 | 1.43 | 1.29 |
| K 4 (%) | 2.46 | 1.89 | 2.7 | 1.93 | 2.65 | 1.83 | 1.74 | 1.69 |
| DHM 5 (%) | 1.78 | 1.77 | 2.07 | 1.71 | 2.44 | 1.77 | 1.71 | 1.52 |
| M 6 (%) | 1.42 | 1.15 | 1.46 | 0.96 | 1.32 | 1.39 | 1.39 | 1.3 |
| Total (%) | 10.73 | 9.4 | 12.04 | 9.18 | 12.91 | 9.22 | 8.79 | 8.45 |
| Chemotype | 245361 | 245361 | 245361 | 245361 | 245361 | 245361 | 245361 | 245631 |
| Narango Palisi | Sara Palisi | Tarivarus | Malogro | Ring | ||||
| DMY 1 (%) | 0.48 | 0.57 | 0.91 | 0.36 | 0.81 | |||
| DHK 2 (%) | 2.12 | 2.06 | 5.21 | 2.17 | 0.1 | |||
| Y 3 (%) | 1.31 | 1.1 | 1.86 | 1.24 | 0.45 | |||
| K 4 (%) | 1.62 | 1.91 | 2.6 | 1.6 | 0.13 | |||
| DHM 5 (%) | 1.74 | 1.93 | 3.09 | 1.66 | 1.98 | |||
| M 6 (%) | 1.45 | 1.46 | 1.31 | 1.38 | 0.37 | |||
| Total (%) | 8.72 | 9.03 | 14.98 | 8.41 | 3.84 | |||
| Chemotype | 254631 | 254631 | 254361 | 254631 | 521364 | |||
| Patient Identification | Specific Information |
|---|---|
| BfArm 93015209 38 years Female | Acetonic kava extract (210 mg/day, 3.5 m). Oral contraceptive, Diazepam, and L-Thyroxine ALT 2305 U/L, AST 1048 U/L and ALP 307 U/L |
| BfArm 01006939 36 years Male | Acetonic kava extract (70 mg/d, 1.5 m) ALT 2341 U/L, AST 2425 U/L, and ALP 530 U/L |
| BfArM 01010536 45 years Female | Ethanolic kava extract (45 mg/d, 4 m) ALT 1000 U/L, AST 700 U/L, and ALP 360 U/L |
| BfArM 02001414 46 years Female | Ethanolic kava extract (360 mg/d, 1 m) ALT 1442 U/L, AST 683 U/L, and ALP 325 U/L |
| BrArM 02002090 26 years Female | Ethanolic kava extract (50 mg/d, 0.25 m) Sulfasalazine, Diclofenac, Progesterone, Omeprazole, Butylscopolaminium bromide ALT 572 U/L, AST 220 U/L, and ALP 163 U/L |
| BfArM 39 years Female | Ethanolic kava extract (60 mg/d, 6 m) Oral contraceptive, Paroxetine, and St. John’s wort ALT 600 U/L, AST 400 U/L, and ALP 183 U/L |
| BfArM 60 years Female | Ethanolic kava extract (1200 mg/d, 12 m) Etilefrine and Pretanide ALT > 1000 U/L, AST > 1000 U/L, and ALP > 500 U/L |
| IKS 2000–3502 50 years Male | Acetonic kava extract (280 mg/d, 2 m) ALT max 3627 U/L, AST max 3360 U/L, and ALP max 430 U/L |
| IKS 1999–2596 46 years Female | Acetonic kava extract (140 mg/d, 3m) Hydrochlorothiazine, Valsartan, and Propranolol ALT max 1900 U/L, AST max 2005 U/L, and ALP not recorded |
| 59 years Female | Aqueous kava extract (unknown daily dose, 1 m) Lisinopril, Phenobarbital and Fenofibrate ALT 568 U/L, AST 672 U/L, and ALP not recorded |
| 55 years Female | Aqueous kava extract (2.571 mg/d, 1.25 m) ALT 1666 U/L, AST 1569 U/L and ALP not recorded |
| 56 years Female | Kava mixture (180 mg/d, 3 m) Passiflora incarnate, Scutellaria lateriflora, vitamins, and mineral supplements ALT 4539 U/L, ALP 190 U/L, and AST not recorded |
| 14 years Female | Kava mixture (200 mg/d, 4 m) St. John’s wort, catnip root, Siberian ginseng root, and other 20 ingredients ALT > 4400 U/L, AST > 3500 U/L and AST not recorded |
| 34 years Female | Aqueous powdered kava extract, ethanolic extract before (120 mg/d, 3 m) L-Thyroxine and potassium iodine ALT 884 U/L, AST 547 U/L, and ALP 319 U/L |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bian, T.; Corral, P.; Wang, Y.; Botello, J.; Kingston, R.; Daniels, T.; Salloum, R.G.; Johnston, E.; Huo, Z.; Lu, J.; et al. Kava as a Clinical Nutrient: Promises and Challenges. Nutrients 2020, 12, 3044. https://doi.org/10.3390/nu12103044
Bian T, Corral P, Wang Y, Botello J, Kingston R, Daniels T, Salloum RG, Johnston E, Huo Z, Lu J, et al. Kava as a Clinical Nutrient: Promises and Challenges. Nutrients. 2020; 12(10):3044. https://doi.org/10.3390/nu12103044
Chicago/Turabian StyleBian, Tengfei, Pedro Corral, Yuzhi Wang, Jordy Botello, Rick Kingston, Tyler Daniels, Ramzi G. Salloum, Edward Johnston, Zhiguang Huo, Junxuan Lu, and et al. 2020. "Kava as a Clinical Nutrient: Promises and Challenges" Nutrients 12, no. 10: 3044. https://doi.org/10.3390/nu12103044
APA StyleBian, T., Corral, P., Wang, Y., Botello, J., Kingston, R., Daniels, T., Salloum, R. G., Johnston, E., Huo, Z., Lu, J., Liu, A. C., & Xing, C. (2020). Kava as a Clinical Nutrient: Promises and Challenges. Nutrients, 12(10), 3044. https://doi.org/10.3390/nu12103044





